5.2 Fundamental Concepts of Skill 3
5.2 Fundamental Concepts of Skill 3
Skill 3 questions ask you to apply scientific concepts to your understanding of research in both the life sciences and the behavioral sciences. However, this information is rarely, if ever, covered in your undergraduate science classes. The scientific method is usually mentioned, but it is unlikely that it is covered in the level of detail that you will need on Test Day. This section discusses the basic concepts that you need to be successful on Skill 3 questions.
The Scientific Method
The scientific method is the basic paradigm of all scientific inquiry. It is the established protocol for transitioning from a question to a new body of knowledge. The steps in the scientific method are as follows.
Generate a Testable Question
- Occurs after observing something anomalous in another scientific inquiry or daily life.
Gather Data and Resources
- Phase of journal and database searches and information compilation.
- Look at all information, not just those consistent with the opinion of the investigator.
Form a Hypothesis
- Often in the form of an if-then statement, which will be tested in subsequent steps.
Collect New Data
- Collect data by experimentation (manipulation and control of variables of interest) or by observation (usually involves no changes in the subject’s environment).
Analyze the Data
- Look for trends.
- Perform mathematical manipulations to solidify the relationship(s) among the variables.
Interpret the Data and Existing Hypothesis
- Consider whether the data analysis is consistent with the original hypothesis.
- If data are inconsistent, consider alternate hypotheses.
Publish
- Provide an opportunity for peer review.
- Summarize what was done during the previous steps in the publication.
Verify Results
- Repeat the experiment to verify results under new conditions.
Basic Concepts in Scientific Research
Basic science research—the kind conducted in a laboratory, not on people—is generally the easiest to design because the experimenter has the most control. Often, a causal relationship is being examined because the hypothesis generally states a condition and an outcome. To make generalizations about our experiments, the outcome of interest must not be obscured. In addition, there must also be a method by which causality may be demonstrated, which is relatively simple in basic science research but less so in other research areas. This requires the use of a control, or standard, and an identified set of variables.
Controls
In basic science research, conditions are applied to multiple trials of the same experiment that are as near to identical as possible.
- A control or standard is included as a method of verifying results.
- Controls can also be used to separate experimental conditions.
- Positive and negative controls are used as points of comparison, or a group of controls can be used to create a curve of known values.
- Positive controls are those that ensure a change in the dependent variable when it is expected. For example, if a new assay is developed for the detection of a human immunodeficiency virus (HIV) infection, a number of blood samples known to contain HIV virus can act as a positive control.
- Negative controls ensure that the dependent variable does not change when no change is expected. For example, the same new HIV assay would be used to test samples known to be without the virus. In pharmaceutical trials, a negative control could be used to assess the placebo effect. An observed or a reported change when an individual is given an inactive substance, such as a sugar pill, is an example of the placebo effect.
Causality
By manipulating all of the relevant experimental conditions, basic science researchers can often establish causality. Causality is an if-then relationship and is often the hypothesis being tested.
- Independent variable: the variable that is manipulated or changed.
- Dependent variable: the variable that is measured or observed.
- If a change in the independent variable always causes a change in the dependent variable and if the change in the dependent variable does not occur without a change in the independent variable, a relationship is said to be causal.
Error Sources
In basic science research, experimental bias is usually minimal. The most likely way for an experimenter’s personal opinions to be incorporated is through the generation of a faulty hypothesis from incomplete early data and resource collection. Other sources of error include the manipulation of results by eliminating trials without appropriate background or by failing to publish works that contradict the experimenter’s own hypothesis.
The low levels of bias introduced by the experimenter do not eliminate all error from basic science research. Measurements are especially important in the laboratory sciences, and the instruments may give faulty readings. Instrument error may affect accuracy, precision, or both. Accuracy, also called validity, is the ability of an instrument to measure a true value. Precision, also called reliability, is the ability of an instrument to read consistently or within a narrow range. Because bias is a systematic error in data, only an inaccurate tool will introduce bias. However, an imprecise tool will still introduce error.
Human Subjects Research
Research using human subjects is considerably more complex, and the level of experimental control is invariably lower than in basic science research. In human subjects research, there are both experimental and observational studies.
Experimental Approach
Experimental research, similar to basic science research, attempts to establish causality. An independent variable is manipulated, and changes in a dependent variable are identified and quantified (if possible). Because subjects are in less-controlled conditions, the data analysis phase is more complicated than in laboratory studies. Two of the most fundamental concepts of the experimental approach are randomization and blinding.
Randomization
- Method used to control for differences between subject groups in biomedical research.
- Uses an algorithm to place each subject into either a control group that receives no treatment (or a sham treatment) or one or more treatment groups.
- Results are measured in all groups.
- Ideally, each group is perfectly matched on conditions such as age and gender.
Blinding
- Many measures in biomedical research are subjective. The perception of the subject and the investigator may be biased by knowing the group to which the subject has been assigned.
- When a study is blinded, the subject and/or the investigator are not aware of the group in which the subject has been placed.
- In single-blind experiments, only the patient or the assessor is blinded.
- In double-blind experiments, neither the subject nor the assessor (or even the investigator) is aware of the group into which a subject has been placed.
- The lack of blinding results in a diminished placebo effect in the control group, but the presence of the placebo affects the treatment group.
In biomedical research, data analysis must account for variables outside the independent and dependent variables. Most often, these include gender and age, lifestyle variables such as smoking, body mass index, and other factors that may affect the measured outcomes. Confounding variables, or variables that are not controlled or measured, also may affect the outcome.
Observational Approach
The observational approach is often adopted to study certain causal relationships for which an experiment is either impractical or unethical. Observational studies in medicine fall into three categories: cohort studies, cross-sectional studies, and case-control studies.
Cohort Studies
- Subjects are sorted into two groups based on differences in risk factors (exposures) and then assessed at various intervals to determine how many subjects in each group have a certain outcome.
Cross-Sectional Studies
- Patients are categorized into different groups at a single point in time based on the presence or absence of a characteristic, such as a disease.
Case-Control Studies
- Subjects are separated into two groups based on the presence or absence of some outcome.
- The study looks backward to assess how many subjects in each group had exposure to a particular risk factor.
Identifying causality isn’t necessarily simple. Hill’s criteria describe the components of an observed relationship that increases the likelihood of causality in that relationship, as shown in Table 5.1. Although only the first criterion, temporality, is necessary for the relationship to be causal, it is not sufficient. An increased likelihood of causality is signified by an increased number of met criteria. Hill’s criteria do not provide an absolute guideline on causality of a relationship. Thus, for any observational study, the relationship should be described as a correlation.
Table 5.1.Hill’s Criteria
Criterion Description
Temporality Exposure (independent variable) must occur before the outcome (dependent variable).
Strength Greater changes in the independent variable will cause a similar change in the dependent variable if the relationship is causal.
Dose-response relationship As the independent variable increases, there is a proportional increase in the response (dependent variable).
Consistency The relationship is found in multiple settings.
Plausibility The presence of a reasonable mechanism for the relationship between the variables is supported by existing literature.
Consideration of alternate explanations If all other plausible explanations have been eliminated, the remaining explanation is more likely.
Experiment An experiment can confirm causality.
Specificity Change in the outcome (dependent) variable is produced only by an associated change in the independent variable.
Coherence New data and hypotheses are consistent with the current state of scientific knowledge.
Error Sources
In addition to the measurement error found in basic science research, we must be aware of bias and error introduced by using human subjects as part of an experimental or observational model. As mentioned earlier, bias is a systematic error. As such, it generally does not impact the precision of the data but, rather, skews the data in one direction or the other. Bias is a result of flaws in the data collection phase of an experimental or observational study. Confounding is an error during analysis (see Figure 5.1).
Selection Bias
- Most prevalent type of bias.
- Occurs when subjects used for the study are not representative of the target population.
- May apply in cases where one gender is more prevalent than another or when there are differences in the age profile of the experiment group and the population.
- Measurement and the assessment of selection bias occur before any intervention.
Detection Bias
- Results from educated professionals applying knowledge in an inconsistent manner.
- Often occurs when prior studies have indicated that there is a correlation between two variables; when the researcher finds one of the variables, then he or she is more likely to search for the second, possibly related variable. That makes the second variable more likely to be found because the investigator is looking for it.
Observation Bias
- Also known as the Hawthorne effect.
- Occurs when the behavior of study participants is altered when the participants are aware that they are being studied.
- Systematic and occurs prior to data analysis.
Confounding
- Data analysis error.
- Data may or may not be flawed, but an incorrect relationship is characterized.
- Variables that are not controlled or measured but are present.
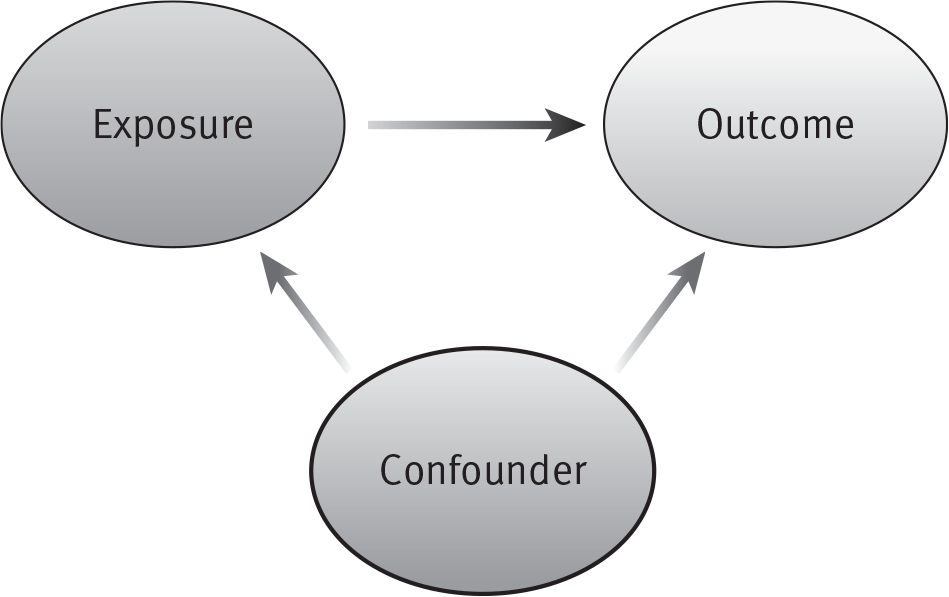
Figure 5.1. Relationship Between Confounder, Exposure, and Outcome
Ethical Issues in Research
In medicine, there are four core ethical tenets: beneficence, nonmaleficence, patient autonomy, and justice.
- Beneficence: an obligation to act in the patient’s best interest
- Nonmaleficence: an obligation to avoid treatments or interventions in which the potential for harm outweighs the potential for benefit
- Patient autonomy: the responsibility to respect patients’ decisions and choices about their own health care
- Justice: the responsibility to treat similar patients with similar care and to distribute health care resources fairly
In research, these principles are replaced by a slightly modified set as defined by the Belmont Report, a landmark document published by the National Commission for the Protection of Human Subjects in Biomedical and Behavioral Research. According to the Belmont Report, the three necessary pillars of research include respect for persons, justice, and a slightly more inclusive version of beneficence.
Respect for Persons
- Includes the need for honesty between the subjects and the researcher and generally—but not always—prohibits deception.
- Also includes the process of informed consent, in which a patient must be adequately counseled about the procedures, risks, benefits, and goals of a study to make a knowledgeable decision about whether or not to participate. Consent may be withdrawn at any time.
- Prohibits a coercive influence over the subjects.
- Institutional review boards are in place to provide systematic protections against unethical studies.
- Vulnerable persons, including children, pregnant women, and prisoners, require special protections above and beyond those taken with the general population.
- Confidentiality is generally considered to be part of respect for persons during research.
Justice
- Applies to both the selection of a research topic and the execution of the research.
- Morally relevant differences are defined as those differences among individuals that are considered an appropriate reason to treat the individuals differently. These differences include age and population size. Race, ethnicity, sexual orientation, and financial status are not considered morally relevant differences. However, religion is a special case in that certain interventions are prohibited by a given religion. Thus, avoidance of that treatment in individuals of that religion is consistent with patient autonomy.
- Risks inherent in the study must be distributed fairly so as to not impose undue harm on a particular group. However, when there is a population that is likely to receive a greater benefit from a study, this group may necessarily bear a greater proportion of the risk. Thus, the likelihood of benefit may be a morally relevant difference among individuals in certain situations.
Beneficence
- The intent of a study must be to cause a net positive change for both the study population and the general population. The study must be conducted in such a way as to minimize any potential harm.
- Research should be conducted in the least invasive, least painful, or least traumatic way possible. When choosing between two methods of measurement, the less painful and less invasive method should be employed.