12.1 Reading the Passage
12.1 Reading the Passage
Passages related to organic chemistry may come in the form of a passage that is exclusively related to organic chemistry or a passage that covers multiple subject areas, especially biochemistry. There is a clear relationship between these two sciences; an understanding of organic chemistry structures and reactions is essential to understanding the behavior of biochemical systems. Therefore, success in organic chemistry on the MCAT depends on your ability to apply your knowledge of organic chemistry to biochemical systems.
Passage Types
Organic chemistry passages are either information passages or experiment passages. (See Table 12.1.)
Information Passages
- Often short and accompanied by diagrams illustrating reactions or mechanisms.
- May be integrated with other subject areas, such as biochemistry or biology.
- May present a new reaction or a series of related reactions, or may describe a compound or an experimental technique.
Experiment Passages
- Presentation of one or more experiments.
- Experimental data may be in the form of percentage yield from a synthesis, a written summary of the reaction, or a description of the appearance or the spectroscopic properties of a product.
- The product of a synthesis may then be used in a biochemistry experiment, meaning that the synthesis reaction and the biochemistry experiment go hand in hand.
Table 12.1.Organic Chemistry Passage Types
Information Passages Experiment Passages
Content Read like a textbook; may integrate concepts of organic chemistry and biochemistry. Read like a lab report or a journal article that summarizes an experimental procedure. The product of an organic chemistry reaction may be the substrate for a biochemistry experiment.
Questions Many questions do not require information from the passage. Those that do are likely to be more theoretical. Often focus on the hypothesis, procedure, and outcome. If multiple experiments are described, questions are likely to focus on the relationships among the experiments.
Reading and Distilling the Passage
Organic chemistry passages often feature synthesis pathways or molecular structures. In addition, the passage may be a mixture of organic chemistry and biochemistry rather than a pure organic chemistry passage. When approaching a passage, be sure to think about how the organic chemistry science applies to a living system, and look for the connections between the sciences presented in the passage.
- Preview for difficulty
- Note the structure of the passage, the location of the paragraphs, and any figures such as charts, graphs, tables, or diagrams.
- Determine whether the passage is an experiment or an information passage.
- Determine the topic and the degree of difficulty.
- Identify whether this passage requires a large time investment.
- Decide whether this passage is one to do now or later.
- Choose your approach
- Using information from the Preview step, Choose an appropriate Distill approach for the passage (Interrogate, Outline, or Highlight).
- Interrogation should be chosen for experiment passages.
- Outlining should be chosen for information passages that are dense or detail heavy.
- Highlighting should be chosen for information passages that are light on details.
- Read and Distill key themes
- While reading the passage, your aim is to distill the major takeaway of each paragraph and identify testable information using one of the following approaches.
- Interrogate: Thoroughly examine the experimental passage by identifying the key components of experimental design and interrogating why specific procedures were done and how they connect to the overall purpose of the experiment.
- Outline: Create a brief label for each paragraph that summarizes the contents of the paragraph, allowing you to return quickly to the passage when demanded by a question.
- Highlight: Highlight one to three terms per paragraph that can pull your attention back to testable information when demanded by a question.
12.2 Answering the Questions
12.2 Answering the Questions
Questions that are exclusively related to organic chemistry are often very straightforward. However, questions that require integration of organic chemistry with another topic may be more difficult. Once again, the same question types seen in the other sciences apply.
- Discrete questions
- Questions not associated with a descriptive passage.
- Are preceded with a warning such as, “Questions 12–15 do not refer to a passage and are independent of each other.”
- Likely to test basic principles of organic chemistry, such as structures and reactions.
- Questions that stand alone from the passage
- One of the most common organic chemistry questions types.
- Often requires analysis of structures.
- When evaluating structures, many of the wrong answer choices contain structures that are simply not possible given the basic concepts of organic chemistry.
- Questions that require data from the passage
- Often require analysis of data or an experimental design.
- Often require evaluation of a synthesis process with a different substrate, thus requiring the application of information from the passage to a new situation.
- Questions that require the goal of the passage
- The nature of these questions depends on what else is in the passage.
- If the passage is exclusively organic chemistry, the goal of the passage is likely to be the outcome of an experiment or a synthetic pathway.
- If the passage contains organic chemistry integrated with biology or biochemistry, the goal of the passage depends on the content of the passage as a whole and the context in which organic chemistry is discussed.
Attacking the Questions
MCAT organic chemistry questions may be related only to organic chemistry or may be organic chemistry in the context of another subject area. In addition, language is often used to disguise simple organic chemistry questions as more difficult ones. Pay special attention to the task of the question to ensure that questions like these are discovered and answered correctly.
- Type the question
- Read the question; peek at the answer choices for patterns, but don't analyze closely.
- Assess the topic and the degree of difficulty.
- Identify the level of time involvement: is this question likely to take a tremendous amount of time to identify the answer? If so, skip it and come back after you do the other questions in the passage set.
- Good questions to do now in organic chemistry are those that stand alone from the passage because these are generally quick and easy points.
- Rephrase the question stem
- Rephrase the question, focusing on the task(s) to be accomplished.
- Simplify the phrasing of the original question stem.
- Translate the question into a specific set of tasks to be accomplished using the passage and your background knowledge.
- Investigate potential solutions
- Complete the task(s) identified in your Rephrase step.
- Analyze the data; evaluate the experimental design; locate the information required; and connect the information, data, and experimental design with the information you already know.
- Predict what you can about the answer.
- Be flexible if your initial approach fails.
- Match your prediction to an answer choice
- Search the answer choices for a response that is synonymous with your prediction, or eliminate answers that are not correct.
- Select an answer and move on.
- If you cannot find a match to your prediction, eliminate wrong answers, select a response from the remaining choices, and move on.
MCAT questions testing organic chemistry usually focus on large concepts, not minor details. As such, you will find that many of the organic chemistry problems you will practice with actually test the same concept in new situations. Incorporate this into your Rephrase step! Often complex question stems can be Rephrased to generic questions, such as, Which carbon is chiral? Which atom is the electrophile?
THINgs to watch out for
Wrong answer choices in organic chemistry often contain molecular structures that are simply not possible or do not make sense. Look for incorrect charge distribution, wrong numbers of bonds for atoms within the molecule, double bonds in unusual locations, and so on.
12.6 Organic Chemistry on Your Own
12.6 Organic Chemistry on Your Own
Organic Chemistry Passage III (Questions 1–6)
Humans can synthesize only 11 of the proteogenic amino acids. Nine others are known as essential amino acids and must be supplied through one’s diet—although some essential amino acids may be interconverted (the sulfur-containing and aromatic amino acids are interchangeable in the body).
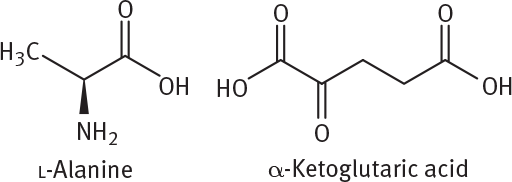
De novo synthesis of amino acids usually starts with the nonessential amino acid glutamate (the conjugate base of glutamic acid). Glutamate is formed from the molecule α-ketoglutarate, a product of the Krebs cycle. In amino acid synthesis, α-ketoglutarate is aminated by ammonium to form glutamate. Glutamate can then be used to transaminate a number of different precursors into their respective amino acids. The transamination converts glutamate to α-ketoglutarate. For example, pyruvate, shown in Figure 1, can be aminated by glutamate to form alanine.

Figure 1.Pyruvate
Amino acid synthesis in the lab follows a variety of other pathways, using molecules not usually found in the human body. The Strecker synthesis starts with a carefully chosen aldehyde. The aldehyde is reacted with ammonium ions, leading to an iminium intermediate. The iminium intermediate is then attacked by a cyanide ion that forms an aminonitrile. Subsequently, this aminonitrile is converted to a carboxylic acid by the addition of water and acid, proceeding through a 1,2-diamino diol intermediate.
Amino acids have unique isoelectric points (pI), a pH where the amino acid will have a net neutral charge. The pI is determined by the appropriate pKa of each functional group. The pI can be found for individual amino acids or for a polypeptide chain. In a polypeptide chain, most of the carboxylic acid and amino groups are bound and thus have no charge. Therefore the charge and, subsequently, the pI are influenced most significantly by the side chains in the polypeptide.
-
Which of the following amino acids will be negatively charged at physiological pH?
-
Glutamic acid
-
Arginine
-
Valine
-
Phenylalanine
-
The following structure shows an ionized form of tyrosine and its pKa values. Based on this information, what is tyrosine’s pI?

-
5.64
-
6.17
-
9.04
-
9.57
-
Which of the following is a significant disadvantage of using Strecker amino acid synthesis to create amino acids for the body?
-
The ammonium ion causes the reaction to proceed too quickly to control.
-
The nucleophile used can also attack side chains with carbonyls.
-
The nucleophilic attack on the carbonyl causes racemization.
-
All amino acids formed from this synthesis are useless biologically.
-
Given the structure of l-alanine and α-ketoglutaric acid, what is the structure of l-glutamic acid?
-

-

-

-

-
Suppose a portion of a peptide chain contains a large amount of phenylalanine, alanine, and valine residues. If the peptide is part of an enzyme that is dissolved in the cytoplasm, where on the enzyme is this region likely to be located?
-
In the active site of the enzyme
-
In the allosteric site of the enzyme
-
In the interior of the enzyme
-
On the exterior of the enzyme
-
Which of the following setups would be most appropriate for isoelectric focusing of protein molecules?
-
A pH gradient (0–14) from left to right, with the anode on the left and the cathode on the right
-
A pH gradient (0–14) from right to left, with the anode on the left and the cathode on the right
-
A pH gradient (0–14) from bottom to top, with the anode on the right and the cathode on the left
-
A pH gradient (0–14) from top to bottom, with the anode on the right and the cathode on the left
-
Organic Chemistry Practice Passage Explanations
Organic Chemistry Practice Passage Explanations
- (A)
At physiological pH, the carboxylic acid and the amino group of an amino acid have a negative charge and positive charge, respectively. This implies that if a molecule is to be negatively charged at physiological pH, the side chain must carry a negative charge. Based on this prediction, look for an amino acid with an acidic side chain. A match is found with an acid in (A).
- (A)
When calculating the pI for an amino acid, the side chain must be considered. In this case, the side chain is a relatively unreactive phenol group—which will remain uncharged until it donates a proton, requiring the pH to be near or above its pKa. At a low pH, the amine group will be protonated and the carboxylic acid group will be neutral (as shown). At a pH equal to the pKa of the carboxylic acid, approximately half of the carboxylic acids will be deprotonated and carry a negative charge. As the pH nears the pKa of the protonated amino group, approximately half the amino groups will lose their proton and become neutral while the other half remains positively charged. The pH between these two pKa values is where the number of ionized carboxyl groups and ionized amine groups is the same. This is the pI, which is calculated as the average of the two pKa values:
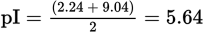
or (A).
- (C)
In the Strecker synthesis, before the ammonium attacks the aldehyde, the carbonyl carbon is sp2 hybridized. This means the electrophile is planar, so the nucleophile can attack from either the top or the bottom. This implies that there will be a racemic mixture of amino acids (for all amino acids except glycine). d-amino acids are not useful biologically because essentially all amino acids in the body are of the l- form. This means that approximately half of the amino acids produced will not be useful, thus potentiating a disadvantage to Strecker synthesis, or (C).
- (B)
The correct form of glutamic acid is similar in chirality to the alanine shown in the question. This means that the configuration at the α-carbon should be S. The passage provides a few clues that can elucidate the molecular formula for glutamic acid. The second paragraph states that pyruvate can be aminated by glutamate to form alanine; in the process, glutamate is deaminated to α-ketoglutarate (pyruvate + glutamate alanine + α-ketoglutarate). Therefore, there must be five carbons in glutamate and its conjugate acid, glutamic acid. This rules out (C). (D) can be eliminated because it lacks an amine group and is therefore not an amino acid. Between the remaining answers, (B) correctly matches the chirality shown in alanine.
- (C)
The location of a certain section of a polypeptide chain depends on the types of amino acids contained in that chain. The chain in this question contains nonpolar amino acids, which are also known as hydrophobic amino acids. Interactions between these residues and water are energetically unfavorable, and hydrophobic amino acids will group together in a nonpolar environment. This means that they are unlikely to be located on any part of the enzyme that is exposed to water, so (C) is the correct answer.
- (A)
This question asks how isoelectric focusing works in agar. The idea behind isoelectric focusing is that at a certain pH, the molecules are neutral; at other pH levels, the molecules have charges. Charged particles abide by Coulomb’s law: they experience a force from other charges from the anode or cathode and accelerate. In an electrolytic cell, the anode is the source of positive charge and the cathode is the source of negative charge. Positively charged molecules migrate toward the cathode, and negatively charged molecules migrate toward the anode. Molecules become more positive as the conditions become more acidic. This means that the cathode should be on the opposite side of the gel from the acidic side so that when the protein is in acidic conditions and has a net positive charge, it will travel toward the basic side (the cathode). As the protein moves toward the cathode, the pH increases and the molecule begins to lose its positive charge. Once it has lost its charge, the protein will no longer experience a force from other charges and will stop moving. This matches (A).