Chapter 12: Bioenergetics and Regulation of Metabolism

Chapter 12: Bioenergetics and Regulation of Metabolism
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- Adding heat to a closed biological system will do all of the following EXCEPT:
- increase the internal energy of the system.
- increase the average of the vibrational, rotational, and translational energies.
- cause the system to do work to maintain a fixed internal energy.
- increase the enthalpy of the system.
- At 25 °C the ΔG° for a certain reaction A ⇌ B + 2 C is 0. If the concentration of A, B, and C in the cell at 25 °C are all 10 mM, how does the ΔGcompare to the measurement taken with 1 M concentrations?
- ΔGis greater than ΔG°, thus the reaction is spontaneous.
- ΔGis less than ΔG°, thus the reaction is spontaneous.
- ΔGis greater than ΔG°, thus the reaction is nonspontaneous.
- ΔGis less than ΔG°, thus the reaction is nonspontaneous.
- Which of the following statements is true about the hydrolysis of ATP?
- The free energy of ATP hydrolysis is independent of pH.
- One mole of creatine phosphate can phosphorylate two moles of ADP.
- The free energy of hydrolysis of ATP is nearly the same as for ADP.
- ATP yields cyclic AMP after two hydrolysis reactions.
- The reduction half-reaction in the last step of the electron transport chain is:
- O2+ 4e–+ 4 H+→2 H2O
- NADPH→NADP++e–+ H+
- NADP++e–+ H+→NADPH
- Ubiquinone (Q)→Ubiquinol (QH2)
- The ability to exist in both an oxidized and a reduced state is characteristic of:
- adenosine triphosphate (ATP).
- electron carriers.
- regulatory enzymes.
- peptide hormones.
- What energy state was described in the introduction to this chapter?
- Absorptive
- Postabsorptive
- Starvation
- Vegetative
- Which of the following correctly pairs a tissue with its preferred energy source in the well fed state?
- Liver - ketone bodies
- Adipose tissue - fatty acids
- Brain - amino acids
- Cardiac muscle - fatty acids
- How do hormonal controls of glycogen metabolism differ from allosteric controls?
- Hormonal control is systemic and covalent.
- Hormonal control is local and covalent.
- Hormonal control is systemic and noncovalent.
- Hormonal control is local and noncovalent.
- Which of the following tissues is most dependent on insulin?
- Active skeletal muscle
- Resting skeletal muscle
- Cardiac muscle
- Smooth muscle
- Glucocorticoids have been implicated in stress-related weight gain because:
- they increase appetite and decrease satiety signals.
- they increase the activity of catabolic hormones.
- they increase glucose levels, which causes insulin secretion.
- they interfere with activity of the leptin receptor.
- In the absence of oxygen, which tissue will experience damage most rapidly?
- Skin
- Brain
- Red blood cells
- Liver
- A respiratory quotient approaching 0.7 indicates metabolism primarily of which macromolecule?
- Carbohydrates
- Lipids
- Nucleic acids
- Amino acids
- Which of the following side effects would be anticipated in someone taking leptin to promote weight loss?
- Drowsiness
- Increased appetite
- Irritability
- Fever
- Which of the following statements is FALSE?
- Growth hormone participates in glucose counterregulation.
- T4 acts more slowly than T3.
- ATP stores are turned over more than 10,000 times daily.
- Catecholamines stimulate the sympathetic nervous system.
- Which process is expected to begin earliest in a prolonged fast?
- Ketone bodies are used by the brain.
- Glycogen storage is halted.
- Proteins are broken down.
- Enzyme phosphorylation and dephosphorylation.
Answer Key
- C
- B
- C
- A
- B
- B
- D
- A
- B
- C
- B
- B
- A
- C
- D
Chapter 12: Bioenergetics and Regulation of Metabolism
CHAPTER 12
BIOENERGETICS AND REGULATION OF METABOLISM
In This Chapter
12.1 Thermodynamics and Bioenergetics
Biological Systems
Enthalpy, Entropy, and Free Energy
Physiological Conditions
12.2 The Role of ATP
ATP as an Energy Carrier
Hydrolysis and Coupling
Phosphoryl Group Transfers
12.3 Biological Oxidation and Reduction
Half-Reactions
Electron Carriers
12.4 Metabolic States
Postprandial (Absorptive) State
Postabsorptive (Fasting) State
Prolonged Fasting (Starvation)
12.5 Hormonal Regulation of Metabolism
Insulin and Glucagon
Glucocorticoids
Catecholamines
Thyroid Hormones
12.6 Tissue-Specific Metabolism
Liver
Adipose Tissue
Skeletal Muscle
Cardiac Muscle
Brain
12.7 Integrative Metabolism
Analysis of Metabolism
Regulation of Body Mass
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 9% of all questions about biochemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1D: Principles of bioenergetics and fuel molecule metabolism
5E: Principles of chemical thermodynamics and kinetics
Introduction
You got up this morning with a really ambitious plan: study for the MCAT! The day started with a big breakfast, and then you dove into MCAT Biochemistry Review. A few chapters in you noticed your stomach growling, but you were having so much fun that you ignored it. A little while later, your body realized it wasn’t getting any more food for a while, but it still needed energy. Where does it come from?
The human body is an incredible system. When we skip lunch on a study day, we produce hormones that help raise the level of certain energy molecules in the bloodstream, mainly glucose. This is a good thing because the brain relies solely on glucose for most of its metabolism, and we always want to be thinking at our peak. Glucose in the blood comes from either our diet, such as when we eat a big breakfast, or from our fuel stores, through the processes of gluconeogenesis and glycogenolysis. These processes, just like the formation and consumption of ATP, are highly regulated.
In this chapter, we’ll highlight the basic principles of bioenergetics, including thermodynamics: the sources of energy and the reactions that play a key role in moving that energy around. Then we’ll examine the different energy states of the body before taking a look at the intimate relationship of hormones with metabolism. We’ll spend some time examining the regulation of metabolism, regulatory enzymes for some common pathways, and how specific tissues preferentially metabolize particular macronutrients. By the end of this chapter, you’ll be able to tell where and how your food is being used, and you probably won’t choose to skip lunch again—no matter how much fun you’re having!
12.1 Thermodynamics and Bioenergetics
LEARNING OBJECTIVES
After Chapter 12.1, you will be able to:
- Describe the relationship between ΔG° and ΔG°′
- Explain why heat can be used as a measure of internal energy for biological systems
- Predict the spontaneity of a reaction given the signs of ΔHand ΔS
If we take a look back at what we learned about thermodynamics in Chapter 3 of MCAT Physics and Math Review and Chapter 7 of MCAT General Chemistry Review, it becomes evident that we already know quite a bit. However, most of the data that we’ve seen so far has been obtained under standard-state conditions (25 °C, 1 atm pressure, and 1 M concentrations). These assumptions work in a chemistry lab, but must be adjusted for application in the human body.
Biological Systems
Biological systems are often considered open systems because they can exchange both energy and matter with the environment. Energy is exchanged in the form of mechanical work when something is moved over a distance, or as heat energy. Matter is exchanged through food consumption and elimination, as well as respiration. Most biochemical studies are performed on the cellular or subcellular level rather than in an entire organism. These systems can be considered closed because there is no exchange of matter with the environment. In such a system, we can make useful simplifications about the internal energy, U. Internal energy is the sum of all of the different interactions between and within atoms in a system; vibration, rotation, linear motion, and stored chemical energies all contribute.
MCAT EXPERTISE
The energy of chemical reactions is described as part of general chemistry, while work is generally associated with physics. Be aware that on Test Day, you will see crossover that allows you to draw on knowledge of the other subjects and to use that background information to your advantage.
Because the system is closed, the change in internal energy can come only in the form of work or heat. This can be represented mathematically through the First Law of Thermodynamics, ΔU = Q – W. Work in thermodynamics refers to changes in pressure and volume. These are constant in most living systems, so the only quantity of interest in determining internal energy is heat.
Enthalpy, Entropy, and Free Energy
Bioenergetics is the term used to describe energy states in biological systems. Changes in free energy (ΔG) provide information about chemical reactions and can predict whether a chemical reaction is favorable and will occur. In biological systems, ATP plays a crucial role in transferring energy from energy-releasing catabolic processes to energy-requiring anabolic processes.
Whether a chemical reaction proceeds is determined by the degree to which enthalpy and entropy change during a chemical reaction. Enthalpy measures the overall change in heat of a system during a reaction. At constant pressure and volume, enthalpy (ΔH) and thermodynamic heat exchange (Q) are equal. Changes in entropy (ΔS) measure the degree of disorder or energy dispersion in a system. While the MCAT will not test on the level of statistical thermodynamics, this conceptual understanding of entropy (ΔS) will be helpful. Entropy carries the units
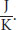
When combined together mathematically, along with temperature (T), these quantities can be related through the Gibbs free energy equation:
ΔG = ΔH − TΔS
Equation 12.1
which predicts the direction in which a chemical reaction proceeds spontaneously. Spontaneous reactions proceed in the forward direction, exhibit a net loss of free energy, and therefore have a negative ΔG. In contrast, nonspontaneous reactions, which would be spontaneous in the reverse direction, exhibit a net gain of energy and have a positive ΔG. Free energy approaches zero as the reaction proceeds to equilibrium and there is no net change in concentration of reactants or products.
BRIDGE
Enthalpy, entropy, and free energy are discussed more thoroughly in Chapter 7 of MCAT General Chemistry Review.
Physiological Conditions
The change in free energy (ΔG) that we have been discussing up to this point predicts changes occurring at any concentration of products and reactants and at any temperature. In contrast, standard free energy (ΔG°) is the energy change that occurs at standard concentrations of 1 M, pressure of 1 atm, and temperature of 25 °C. These can be related by the equation:
ΔG = ΔG° + RT ln(Q)
Equation 12.2
where R is the universal gas constant, T is the temperature, and Q is the reaction quotient. Biochemical analysis works well under all standard conditions except one: pH. A 1 M concentration of protons would correspond to a pH of 0, which is far too acidic for most biochemical reactions. Therefore, in the modified standard state, [H+] = 10−7 M and the pH is 7. With this additional condition, ΔG° is given the special symbol ΔG°′, indicating that it is standardized to the neutral buffers used in biochemistry. Note that if the concentrations of other reactants and products differ from 1 M, these must still be adjusted for in the equation above.
The shift in ΔG as a result of changing concentration is not universally toward or away from spontaneity. There is a general trend that reactions with more products than reactants have a more negative ΔG, while reactions with more reactants than products have a more positive ΔG. While this trend is useful for making quick assessments, always double check with numbers on Test Day.
MCAT CONCEPT CHECK 12.1:
Before you move on, assess your understanding of the material with these questions.
- What conditions does ΔG°′ adjust for that are not considered with ΔG°?
_____________________________________
- Why can heat be used as a measure of internal energy in living systems?
______________________________________
______________________________________
- Complete the following table relating the change in entropy and enthalpy of a reaction with whether the reaction is spontaneous.
**+ΔH −ΔH +ΔS −Δ*S***
12.2 The Role of ATP
LEARNING OBJECTIVES
After Chapter 12.2, you will be able to:
- Predict the impact of ATP coupling on the energetics of a reaction
- Calculate free energy change for an overall reaction given energetic data for component reactions
- Relate the structure of ATP to its role as an energy carrier
The human body can make use of different energy sources with roughly the same efficiency, but all nutrient molecules are not created equally. For example, fats are much more energy-rich than carbohydrates, proteins, or ketones. Complete combustion of fat results in 9
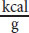
of energy, compared with only 4
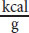
derived from carbohydrates, proteins, or ketones. Because fats are so much more energy-dense than other biomolecules, they are preferred for long-term energy storage. Think of the difference between fats and carbohydrates like the difference between a 16-GB and an 8-GB storage drive. The storage drive with a greater capacity occupies the same amount of physical space, but holds twice as much data. While different energy sources provide greater or lesser caloric values, the end goal is to have energy in a readily available form. For the cell, this is adenosine triphosphate (ATP), shown in Figure 12.1.
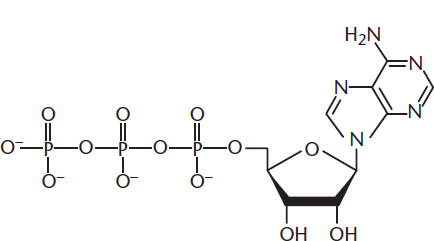
Figure 12.1 Adenosine Triphosphate (ATP)
ATP as an Energy Carrier
ATP is the major energy currency in the body. It is a mid-level energy carrier, as seen in Table 12.1, and is formed from substrate-level phosphorylation, as well as oxidative phosphorylation. Why do we want ATP to be a mid-level carrier and not a higher-level one? Think about your wallet. If you never had the ability to get change back after a purchase, what type of bill would you want in abundance? One dollar bills! Similarly, ATP cannot get back the “leftover” free energy after a reaction, so it’s best to use a carrier with a smaller free energy. ATP provides about 30
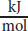
of energy under physiological conditions. If a reaction only requires 10
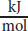
to overcome a positive ΔG value, then 20
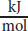
have been wasted. The waste would be even higher with a higher-energy compound like creatine phosphate.
Table 12.1. Free Energy of Hydrolysis for Key Metabolic Phosphate Compounds
COMPOUND
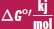
FUNCTION
cAMP −50.4 Second messenger
Creatine phosphate −43.3 Direct phosphorylation in muscle
ATP −30.5 Energy turnover in all cell types
Glucose 6-phosphate −13.9 Intermediate of glycolysis and gluconeogenesis
AMP −9.2 ATP synthesis
Remember that most of the ATP in a cell is produced by mitochondrial ATP synthase, as described in Chapter 10 of MCAT Biochemistry Review, but some ATP is produced during glycolysis and (indirectly from GTP) in the citric acid cycle. ATP consists of an adenosine molecule attached to three phosphate groups, and is generated from ADP and Pi with energy input from an exergonic reaction or electrochemical gradient. ATP is consumed either through hydrolysis or the transfer of a phosphate group to another molecule. If one phosphate group is removed, adenosine diphosphate (ADP) is produced; if two phosphate groups are removed, adenosine monophosphate (AMP) is the result. In a single day, an average-sized person uses about 90 percent of body weight in ATP but only has about 50 grams of ATP available at any given time. Continuous recycling of ATP, ADP, and Pi more than 1000 times per day accounts for this discrepancy.
What makes ATP such a good energy carrier is its high-energy phosphate bonds. The negative charges on the phosphate groups experience repulsive forces with one another, and the ADP and Pi molecules that form after hydrolysis are stabilized by resonance. While ATP doesn’t rapidly break down on its own in the cell, it is much more stable after hydrolysis. This accounts for the very negative value of ΔG. Under standard conditions ΔG° is about −55
At pH 7 and with excess magnesium, the standard free energy change is still −30.5
ADP, which also displays charge repulsion and resonance stabilization after hydrolysis, has similar ΔG values, but AMP has a much smaller ΔG° near −9.2
Hydrolysis and Coupling
ATP hydrolysis is most likely to be encountered in the context of coupled reactions. Many coupled reactions use ATP as an energy source. For example, the movement of sodium and potassium against their electrochemical gradients requires energy, which is harnessed from the hydrolysis of ATP.
ATP cleavage is the transfer of a high-energy phosphate group from ATP to another molecule. Generally, this activates or inactivates the target molecule. With these phosphoryl group transfers, the overall free energy of the reaction will be determined by taking the sum of the free energies of the individual reactions.
KEY CONCEPT
ATP is used to fuel energetically unfavorable reactions or to activate or inactivate other molecules.
Phosphoryl Group Transfers
ATP can provide a phosphate group as a reactant. For example, in the phosphorylation of glucose in the early stages of glycolysis, ATP donates a phosphate group to glucose to form glucose 6-phosphate. The information in Table 12.1 indicates the free energy of hydrolysis, which can be conceptualized as the transfer of the phosphate group to water. To determine the free energy of phosphoryl group transfer to another biological molecule, one could use Hess’s law and calculate the difference in free energy between the reactants and products:
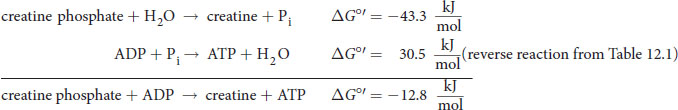
BRIDGE
Hess’s law, discussed in Chapter 7 of MCAT General Chemistry Review, applies for all of the state functions, including pressure, density, temperature, volume, enthalpy, internal energy, free energy, and entropy.
MCAT CONCEPT CHECK 12.2:
Before you move on, assess your understanding of the material with these questions.
- How does coupling with ATP hydrolysis alter the energetics of a reaction?
_________________________________________
- Explain why ATP is an inefficient molecule for long-term energy storage.
_________________________________________
- Using Table 12.1, calculate the free energy change for the synthesis of ATP from cAMP and inorganic phosphate. Note: cAMP is hydrolyzed to AMP, and the free energy of hydrolysis for ATP and ADP is approximately equal.
_________________________________________
12.3 Biological Oxidation and Reduction
LEARNING OBJECTIVES
After Chapter 12.3, you will be able to:
- Explain the benefits of analyzing half reactions for biological oxidation–reduction reactions
- Recall soluble electron carriers, such as NADH and CoQ, and the pathways they are paired with
Many key enzymes in ATP synthesis and other biochemical pathways have oxidoreductase activity.
Half-Reactions
Just as you practiced with general chemistry, an important skill in biochemistry is to be able to divide oxidation–reduction reactions into their half-reaction components to determine the number of electrons being transferred. For example, in lactic acid fermentation, pyruvate and NADH are converted to lactate and NAD+ by lactate dehydrogenase. This reaction can be broken down into half-reactions as follows:
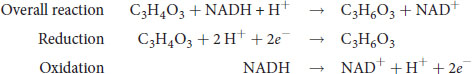
Remember that spontaneous oxidation–reduction reactions have a negative value of ΔG and a positive value of E (electromotive force).
BRIDGE
Oxidation–reduction reactions, discussed in Chapter 11 of MCAT General Chemistry Review and Chapter 4 of MCAT Organic Chemistry Review, are a staple of general chemistry and are characteristic of oxidoreductase enzymes. Take a moment to identify the oxidizing and reducing agents in the reaction catalyzed by lactate dehydrogenase.
Electron Carriers
In the cytoplasm, there are several molecules that act as high-energy electron carriers. These are all soluble and include NADH, NADPH, FADH2, ubiquinone, cytochromes, and glutathione. Some of these electron carriers are used by the mitochondrial electron transport chain, which leads to the oxidative phosphorylation of ADP to ATP. As electrons are passed down the electron transport chain, they give up their free energy to form the proton-motive force across the inner mitochondrial membrane. The oxidized and reduced forms of NADH and FADH2 are shown in Figure 12.2. In addition to soluble electron carriers, there are membrane-bound electron carriers embedded within the inner mitochondrial membrane. One such carrier is flavin mononucleotide (FMN), which is bonded to complex I of the electron transport chain and can also act as a soluble electron carrier. In general, proteins with prosthetic groups containing iron–sulfur clusters are particularly well suited for the transport of electrons.
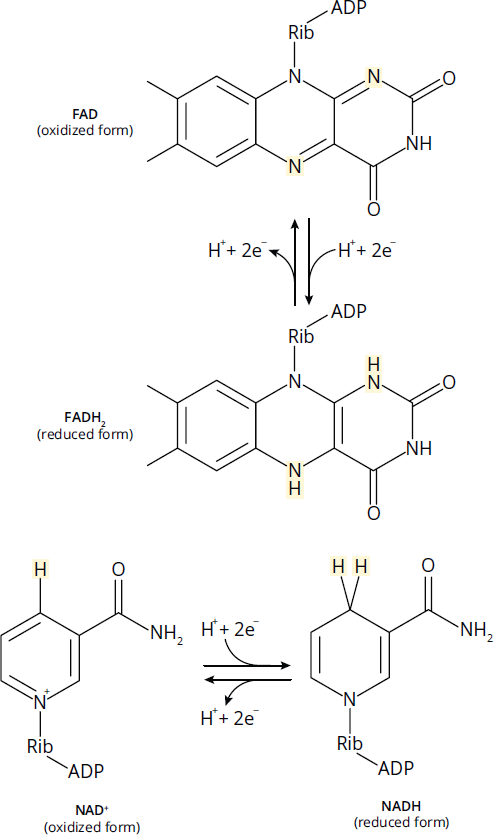
Figure 12.2 Oxidized and Reduced Forms of NADH and FADH2
Flavoproteins
Flavoproteins contain a modified vitamin B2, or riboflavin. They are nucleic acid derivatives, generally either flavin adenine dinucleotide (FAD) or flavin mononucleotide (FMN). Flavoproteins are most notable for their presence in the mitochondria and chloroplasts as electron carriers. Flavoproteins are also involved in the modification of other B vitamins to active forms. Finally, flavoproteins function as coenzymes for enzymes in the oxidation of fatty acids, the decarboxylation of pyruvate, and the reduction of glutathione. The structure of FAD is shown in Figure 12.3 in comparison to the structure of NAD+.
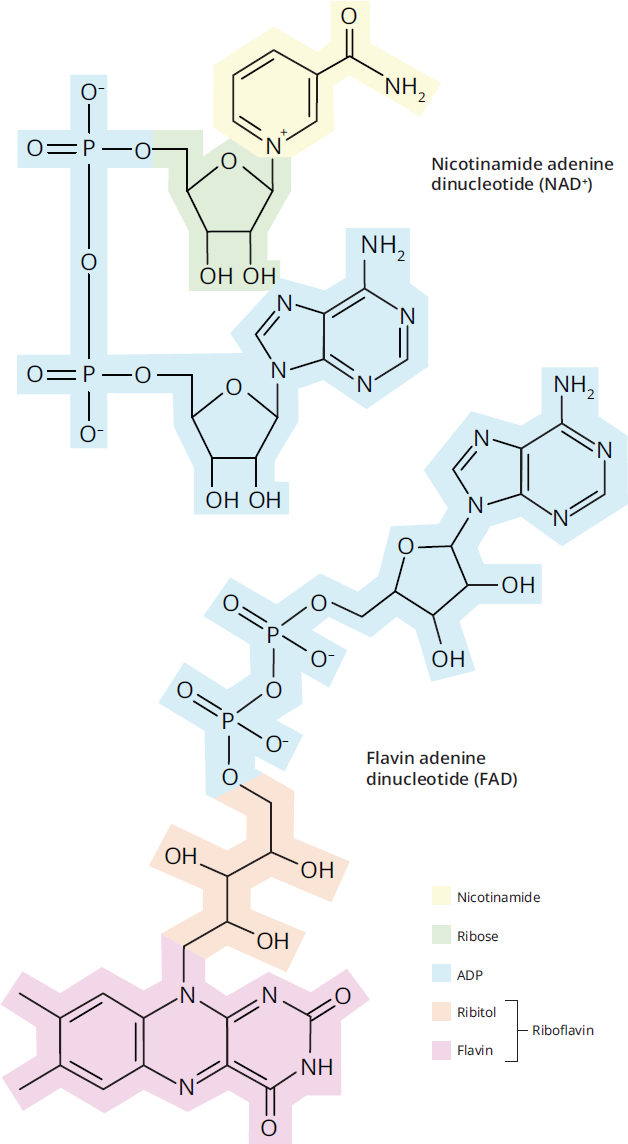
Figure 12.3 Structure of NAD+ and FAD
REAL WORLD
Deficiency of riboflavin, a key component of flavoproteins, leads to a lack of growth, failure to thrive, and eventual death in experimental models. In humans, riboflavin deficiency is very rare, but may occur in severely malnourished individuals.
MCAT CONCEPT CHECK 12.3:
Before you move on, assess your understanding of the material with these questions.
- What is an advantage of analyzing the half-reactions in biological oxidation and reduction reactions?
_________________________________
- Name three soluble electron carriers and their relevant metabolic pathways in the cell.
Electron Carrier Metabolic Pathway(s)
12.4 Metabolic States
LEARNING OBJECTIVES
After Chapter 12.4, you will be able to:
- Identify disequilibriums that are maintained at energetic cost within the cell
- Compare the behavior of different tissues during postprandial, postabsorptive, and prolonged fasting states
- Associate the three metabolic states with related hormone activity
One of the key differences between general chemistry and biochemistry is whether or not equilibrium is seen as a desirable state. Biochemists emphatically believe that it is not! Equilibrium is a fixed state, which prevents us from storing any energy for later use or creating an excitable environment. Instead, biochemists seek a state of homeostasis. Homeostasis is a physiological tendency toward a relatively stable state that is maintained and adjusted, often with the expenditure of energy. Most compounds in the body are actually maintained at a homeostatic level that is different from equilibrium, which allows us to store potential energy; for example, keeping sodium concentrations much higher outside a neuron than inside it creates a gradient that stores energy. In this state, reactions can proceed such that equilibrium is put off for a long time (someone born today can delay equilibrium for about 80 years).
The pathways that are operational in fuel metabolism depend on the nutritional status of the organism. Shifts between storage and mobilization of a particular fuel, as well as shifts among the types of fuel being used, are very pronounced when going from the well-fed state to an overnight fast, and finally to a prolonged state of starvation. We’ll take a look at how fuel metabolism is regulated in each state. Remember that in addition to the “big-picture view” discussed here, the specific regulatory steps of each pathway are discussed in the previous chapters of MCAT Biochemistry Review: Chapter 9 (glycolysis, glycogenesis, glycogenolysis, gluconeogenesis, and the pentose phosphate pathway), Chapter 10 (the citric acid cycle, electron transport chain, and oxidative phosphorylation), and Chapter 11 (fatty acid and cholesterol synthesis, β-oxidation, ketogenesis and ketolysis, and amino acid metabolism).
Postprandial (Absorptive) State
The postprandial state, also called the absorptive or well-fed state, occurs shortly after eating. This state is marked by greater anabolism (synthesis of biomolecules) and fuel storage than catabolism (breakdown of biomolecules for energy). Nutrients flood in from the gut and make their way via the hepatic portal vein to the liver, where they can be stored or distributed to other tissues of the body. The postprandial state generally lasts three to five hours after eating a meal.
Just after eating, blood glucose levels rise and stimulate the release of insulin. The three major target tissues for insulin are the liver, muscle, and adipose tissue, as shown in Figure 12.4. Insulin promotes glycogen synthesis in liver and muscle. After the glycogen stores are filled, the liver converts excess glucose to fatty acids and triacylglycerols. Insulin promotes triacylglycerol synthesis in adipose tissue and protein synthesis in muscle, as well as glucose entry into both tissues. After a meal, most of the energy needs of the liver are met by the oxidation of excess amino acids.
Two types of cells—nervous tissue and red blood cells—are notably insensitive to insulin. Nervous tissue derives energy from oxidizing glucose to CO2 and water in both the well-fed and normal fasting states. Only in prolonged fasting does this situation change. Red blood cells can only use glucose anaerobically for all their energy needs, regardless of the individual’s metabolic state.
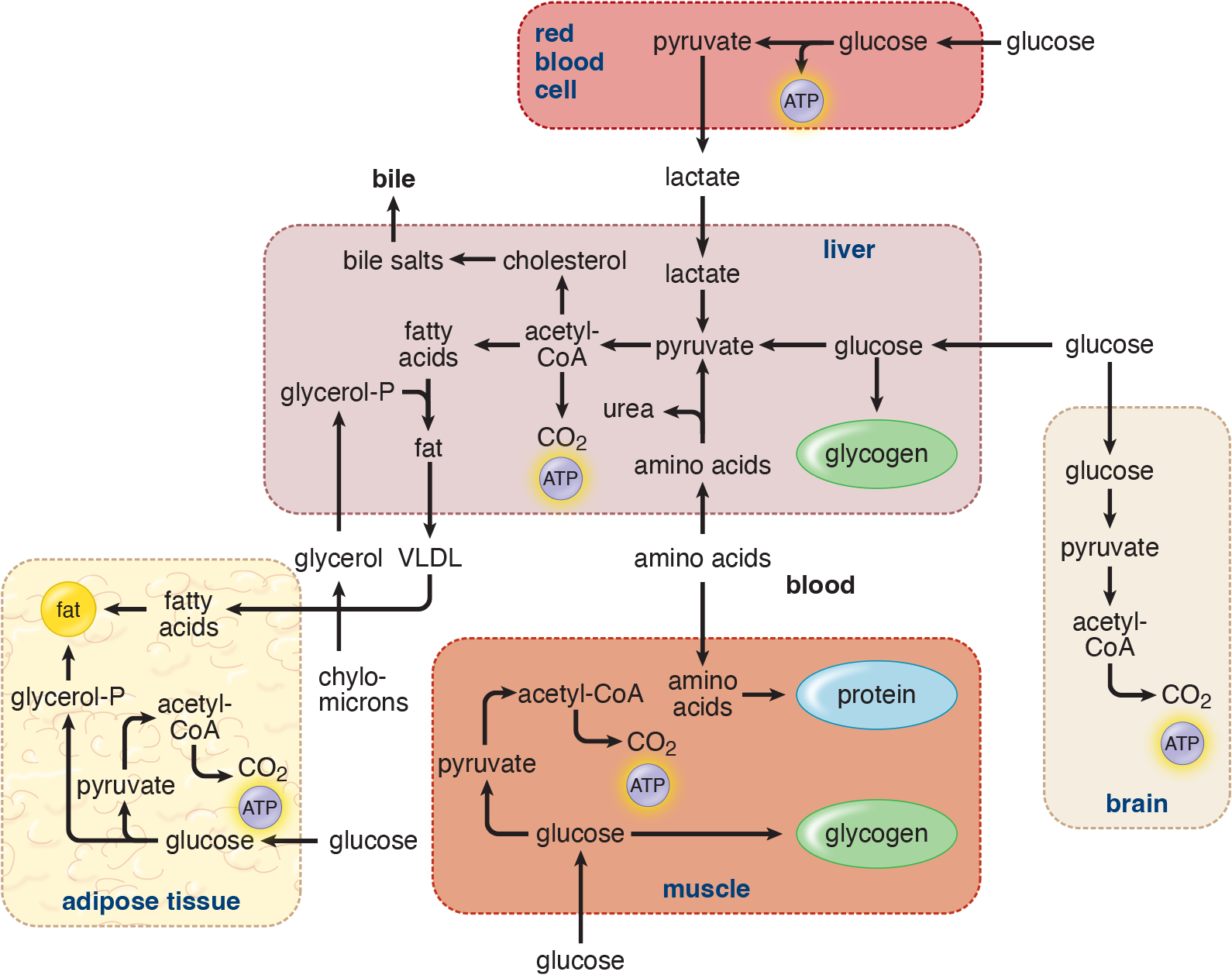
Figure 12.4 Metabolic Profile of the Postprandial (Absorptive) State
Postabsorptive (Fasting) State
Glucagon, cortisol, epinephrine, norepinephrine, and growth hormone oppose the actions of insulin. These hormones are sometimes termed counterregulatory hormones because of their effects on skeletal muscle, adipose tissue, and the liver, which are opposite to the actions of insulin. In the liver, glycogen degradation and the release of glucose into the blood are stimulated, as shown in Figure 12.5. Hepatic gluconeogenesis is also stimulated by glucagon, but the response is slower than that of glycogenolysis. Whereas glycogenolysis begins almost immediately at the beginning of the postabsorptive state, gluconeogenesis takes about 12 hours to hit maximum velocity.
The release of amino acids from skeletal muscle and fatty acids from adipose tissue are both stimulated by the decrease in insulin and by an increase in levels of epinephrine. Once carried into the liver, amino acids and fatty acids can provide the necessary carbon skeletons and energy required for gluconeogenesis.
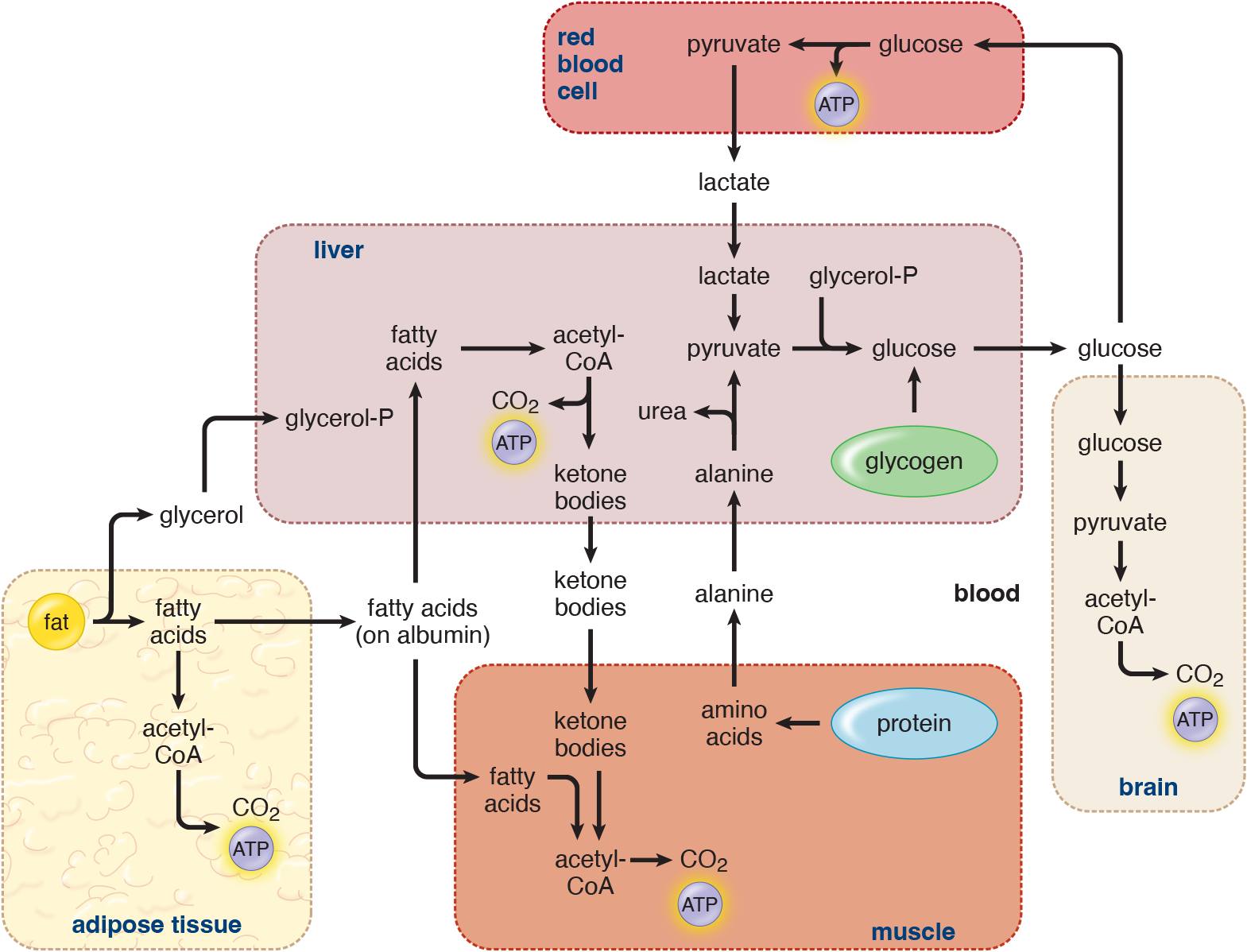
Figure 12.5 Metabolic Profile of the Postabsorptive (Fasting) State
Prolonged Fasting (Starvation)
Levels of glucagon and epinephrine are markedly elevated during starvation. Increased levels of glucagon relative to insulin result in rapid degradation of glycogen stores in the liver. As liver glycogen stores are depleted, gluconeogenic activity continues and plays an important role in maintaining blood glucose levels during prolonged fasting; after about 24 hours, gluconeogenesis is the predominant source of glucose for the body. Lipolysis is rapid, resulting in excess acetyl-CoA that is used in the synthesis of ketone bodies. Once levels of fatty acids and ketones are high enough in the blood, muscle tissue will utilize fatty acids as its major fuel source and the brain will adapt to using ketones for energy. After several weeks of fasting, the brain derives approximately two-thirds of its energy from ketones and one-third from glucose. The shift from glucose to ketones as the major fuel reduces the quantity of amino acids that must be degraded to support gluconeogenesis, which spares proteins that are vital for other functions. Cells that have few, if any, mitochondria, like red blood cells, continue to be dependent on glucose for their energy.
MCAT CONCEPT CHECK 12.4:
Before you move on, assess your understanding of the material with these questions.
- Provide an example of disequilibrium that is maintained at the expense of cellular energy.
_____________________________________
- What tissue is least able to change its fuel source in periods of prolonged starvation?
_____________________________________
- During what stage is there the greatest decrease in the circulating concentration of insulin?
_____________________________________
12.5 Hormonal Regulation of Metabolism
LEARNING OBJECTIVES
After Chapter 12.5, you will be able to:
- Describe the impact of key metabolic hormones, such as insulin, glucagon, and thyroid hormones, on metabolic function
- Apply knowledge of the metabolic hormones to a given disease state
- Recall the general structures and traits of metabolic hormones:
If each cell were acting independently of one another, metabolism would be a random process that could not be coordinated with outside events like meals or exertion. In order to make the most efficient use of the resources available, metabolism must be regulated across the entire organism. This regulation is accomplished best through hormonal means. Water-soluble peptide hormones, like insulin, are able to rapidly adjust the metabolic processes of cells via second messenger cascades, while certain fat-soluble amino acid–derivative hormones, like thyroid hormones, and steroid hormones, like cortisol, enact longer-range effects by exerting regulatory actions at the transcriptional level. Hormone levels are regulated by feedback loops with other endocrine structures, such as the hypothalamic–pituitary axis, or by the biomolecule upon which they act; for example, insulin causes a decrease in blood glucose, which removes the trigger for continued insulin release. Next, we’ll examine the specific actions of several hormones involved in the regulation of metabolism and in maintaining homeostasis, including insulin and glucagon, epinephrine, glucocorticoids, and thyroid hormones.
Insulin and Glucagon
Insulin
Insulin is a peptide hormone secreted by the β-cells of the pancreatic islets of Langerhans, as shown in Figure 12.6. It is a key player in the uptake and storage of glucose. Glucose is absorbed by peripheral tissues via facilitated transport mechanisms that utilize glucose transporters located in the cell membrane. The tissues that require insulin for effective uptake of glucose are adipose tissue and resting skeletal muscle. Tissues in which glucose uptake is not affected by insulin include:
- Nervous tissue
- Kidney tubules
- Intestinal mucosa
- Red blood cells (erythrocytes)
- β-cells of the pancreas

Figure 12.6 Insulin (Light Brown) in Pancreatic β-Cells
Take note of the differences between these types of tissues. Some tissues that require insulin actively store glucose when it is present in high concentrations, while other tissues that do not require insulin must still be able to absorb glucose even when the glucose concentration is low.
Insulin impacts the metabolism of the different nutrient classes in different ways. For carbohydrates, insulin increases the uptake of glucose and increases carbohydrate metabolism in muscle and fat. Increased glucose in muscle can be used as additional fuel to burn during exercise, or can be stored as glycogen. Insulin also increases glycogen synthesis in the liver by increasing the activity of glucokinase and glycogen synthase, while decreasing the activity of enzymes that promote glycogen breakdown (glycogen phosphorylase and *glucose**-6-phosphatase*).
While the primary effects of insulin are on carbohydrate metabolism, it also changes the way that the body processes other macromolecules. For instance, insulin increases amino acid uptake by muscle cells, thereby increasing levels of protein synthesis and decreasing breakdown of essential proteins. Insulin also exhibits a significant impact on the metabolism of fats, especially in the liver and adipocytes. The effects of insulin on the metabolism of fats are described below.
Insulin increases:
- Glucose and triacylglycerol uptake by fat cells
- Lipoprotein lipase activity, which clears VLDL and chylomicrons from the blood
- Triacylglycerol synthesis (lipogenesis) in adipose tissue and the liver from acetyl-CoA
Insulin decreases:
- Triacylglycerol breakdown (lipolysis) in adipose tissue
- Formation of ketone bodies by the liver
The most important controller of insulin secretion is plasma glucose. Above a threshold of 100
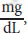
or about 5.6 mM glucose, insulin secretion is directly proportional to plasma glucose. For glucose to promote insulin secretion, it must not only enter the β-cell but also be metabolized, increasing intracellular ATP concentration. Increased ATP leads to calcium release in the cell, which promotes exocytosis of preformed insulin from intracellular vesicles. Insulin secretion is also affected by signaling initiated by other hormones, such as glucagon and somatostatin.
Glucagon
Glucagon is a peptide hormone secreted by the α-cells of the pancreatic islets of Langerhans, as shown in Figure 12.7. The primary target for glucagon action is the hepatocyte. Glucagon acts through second messengers to cause the following effects:
- Increased liver glycogenolysis. Glucagon activates glycogen phosphorylase and inactivates glycogen synthase.
- Increased liver gluconeogenesis. Glucagon promotes the conversion of pyruvate to phosphoenolpyruvate by pyruvate carboxylase and phosphoenolpyruvate carboxykinase (PEPCK). Glucagon increases the conversion of fructose 1,6-bisphosphate to fructose 6-phosphate by fructose-1,6-bisphosphatase.
- Increased liver ketogenesis and decreased lipogenesis.
- Increased lipolysis in the liver. Glucagon activates *hormone-sensitive lipas**e* in the liver. Because the action is on the liver and not the adipocyte, glucagon is not considered a major fat-mobilizing hormone.

Figure 12.7 Glucagon (Dark Brown) in Pancreatic α-Cells
Low plasma glucose (hypoglycemia) is the most important physiological promoter of glucagon secretion, and elevated plasma glucose (hyperglycemia) is the most important inhibitor. Amino acids, especially basic amino acids (arginine, lysine, histidine), also promote the secretion of glucagon. Thus, glucagon is secreted in response to the ingestion of a meal rich in proteins.
REAL WORLD
Patients with type 1 diabetes mellitus are incapable of synthesizing insulin, but still synthesize glucagon. This combination increases blood sugar much more than if an individual were to lose all pancreatic function or to develop insulin insensitivity.
Functional Relationship of Glucagon and Insulin
Insulin, associated with a well-fed, absorptive metabolic state, and glucagon, associated with a postabsorptive metabolic state, usually oppose each other with respect to pathways of energy metabolism. Enzymes that are phosphorylated by glucagon are generally dephosphorylated by insulin; enzymes that are phosphorylated by insulin are generally dephosphorylated by glucagon. Figure 12.8 displays a feedback diagram of the interaction of insulin and glucagon on plasma glucose concentration, as well as fat and protein metabolism.
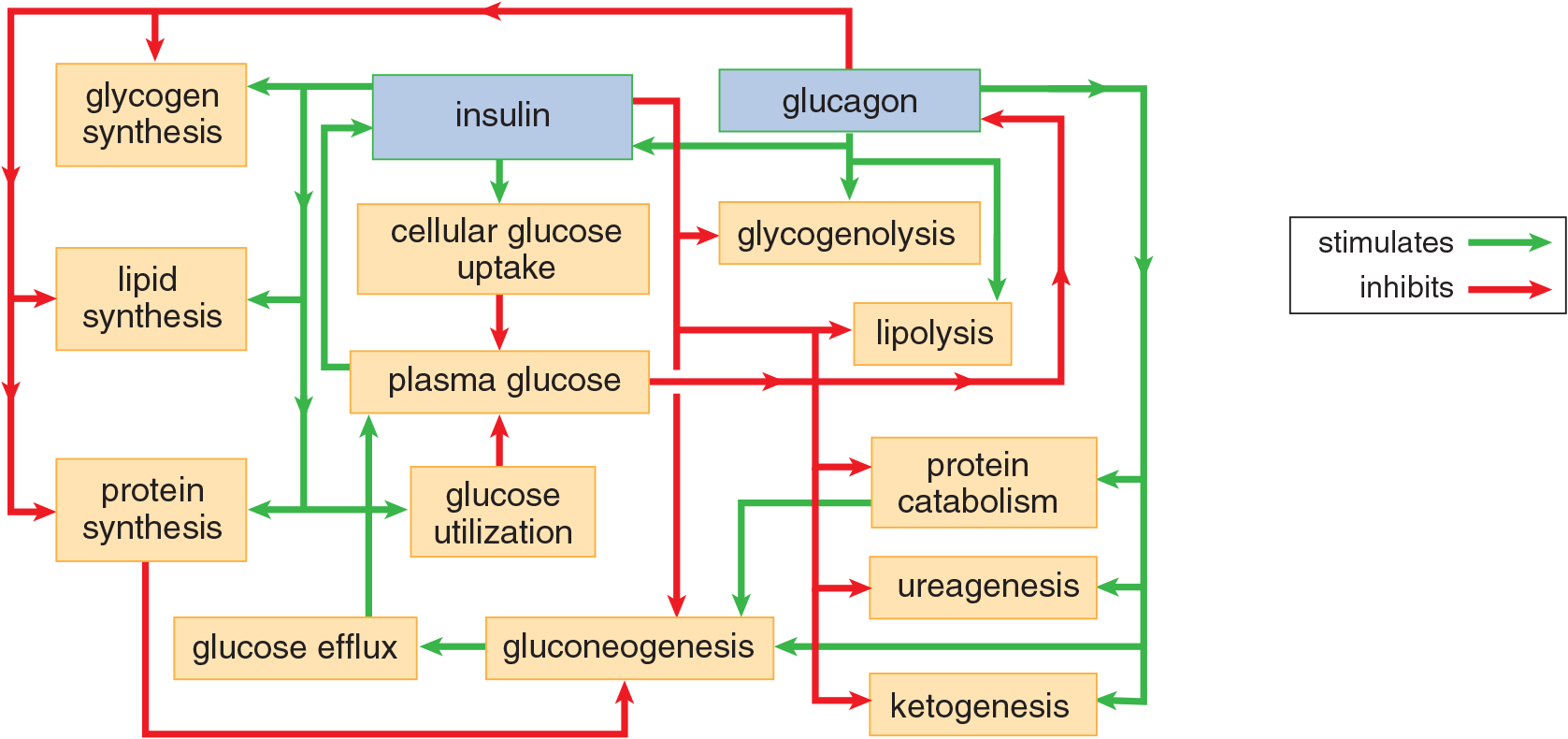
Figure 12.8 Relationship of Glucagon and Insulin in Metabolism
Glucocorticoids
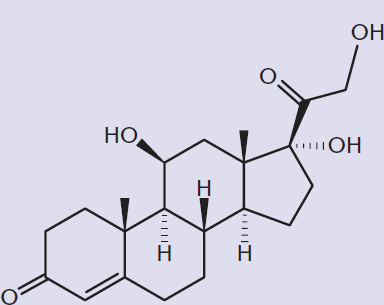
Glucocorticoids from the adrenal cortex are responsible for part of the stress response. In order to make a getaway in the “fight-or-flight” response, glucose must be rapidly mobilized from the liver in order to fuel actively contracting muscle cells while fatty acids are released from adipocytes. Glucocorticoids, especially cortisol, are secreted with many forms of stress, including exercise, cold, and emotional stress. Cortisol, shown in Figure 12.9, is a steroid hormone that promotes the mobilization of energy stores through the degradation and increased delivery of amino acids and increased lipolysis. Cortisol also elevates blood glucose levels, increasing glucose availability for nervous tissue through two mechanisms. First, cortisol inhibits glucose uptake in most tissues (muscle, lymphoid, and fat) and increases hepatic output of glucose via gluconeogenesis, particularly from amino acids. Second, cortisol has a permissive function that enhances the activity of glucagon, epinephrine, and other catecholamines. Long-term exposure to glucocorticoids may be required clinically, but causes persistent hyperglycemia, which stimulates insulin. This actually promotes fat storage in the adipose tissue, rather than lipolysis.
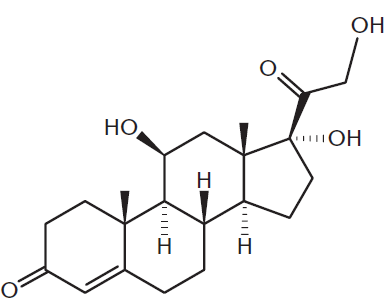
Figure 12.9 Structure of Cortisol
BRIDGE
The endocrine system, discussed in Chapter 5 of MCAT Biology Review, is a major regulator of homeostasis. Like the glucocorticoids and catecholamines, mineralocorticoids and sex hormones are also synthesized by the adrenal glands and play a more minor role in metabolism.
An enlarged adrenal gland (with a tumor of the adrenal cortex) is shown in Figure 12.10. While the adrenal cortex produces steroid hormones (glucocorticoids, mineralocorticoids, and sex hormones), the adrenal medulla produces catecholamines.

Figure 12.10 Adrenal Gland (Enlarged) The adrenal cortex (yellow) and adrenal medulla (brown interior) are visible on both slices.
Catecholamines
Catecholamines are secreted by the adrenal medulla and include epinephrine and norepinephrine, also known as adrenaline and noradrenaline. The structures of these hormones are shown in Figure 12.11. Catecholamines increase the activity of liver and muscle glycogen phosphorylase, thus promoting glycogenolysis. This increases glucose output by the liver. Glycogenolysis also increases in skeletal muscle, but because muscle lacks glucose-6-phosphatase, glucose cannot be released by skeletal muscle into the bloodstream; instead, it is metabolized by the muscle tissue itself. Catecholamines act on adipose tissue to increase lipolysis by increasing the activity of hormone-sensitive lipase. Glycerol from triacylglycerol breakdown is a minor substrate for gluconeogenesis. Epinephrine also acts directly on target organs like the heart to increase the basal metabolic rate through the sympathetic nervous system. This increase in metabolic function is often associated with an adrenaline rush.
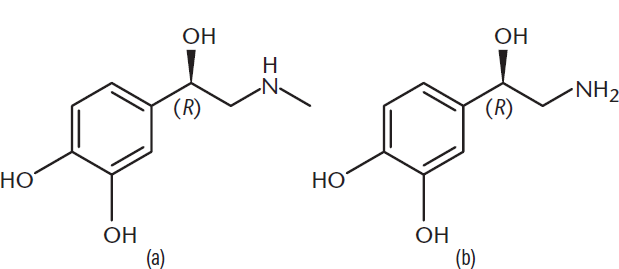
Figure 12.11 Structures of Adrenal Catecholamines (a) Epinephrine; (b) Norepinephrine
Thyroid Hormones
Thyroid hormone activity is largely permissive. In other words, thyroid hormone levels are kept more or less constant, rather than undulating with changes in metabolic state. Thyroid hormones increase the basal metabolic rate, as evidenced by increased O2 consumption and heat production when they are secreted. The increase in metabolic rate produced by a dose of thyroxine (T4) occurs after a latency of several hours but may last for several days, while triiodothyronine (T3) produces a more rapid increase in metabolic rate and has a shorter duration of activity. The subscript numbers refer to the number of iodine atoms in the hormone; iodine atoms are represented by purple spheres in the structures shown in Figure 12.12. T4 can be thought of as the precursor to T3; deiodonases (enzymes that remove iodine from a molecule) are located in target tissues and convert T4 to T3. Thyroid hormones have their primary effects in lipid and carbohydrate metabolism. They accelerate cholesterol clearance from the plasma and increase the rate of glucose absorption from the small intestine. Epinephrine requires thyroid hormones to have a significant metabolic effect.
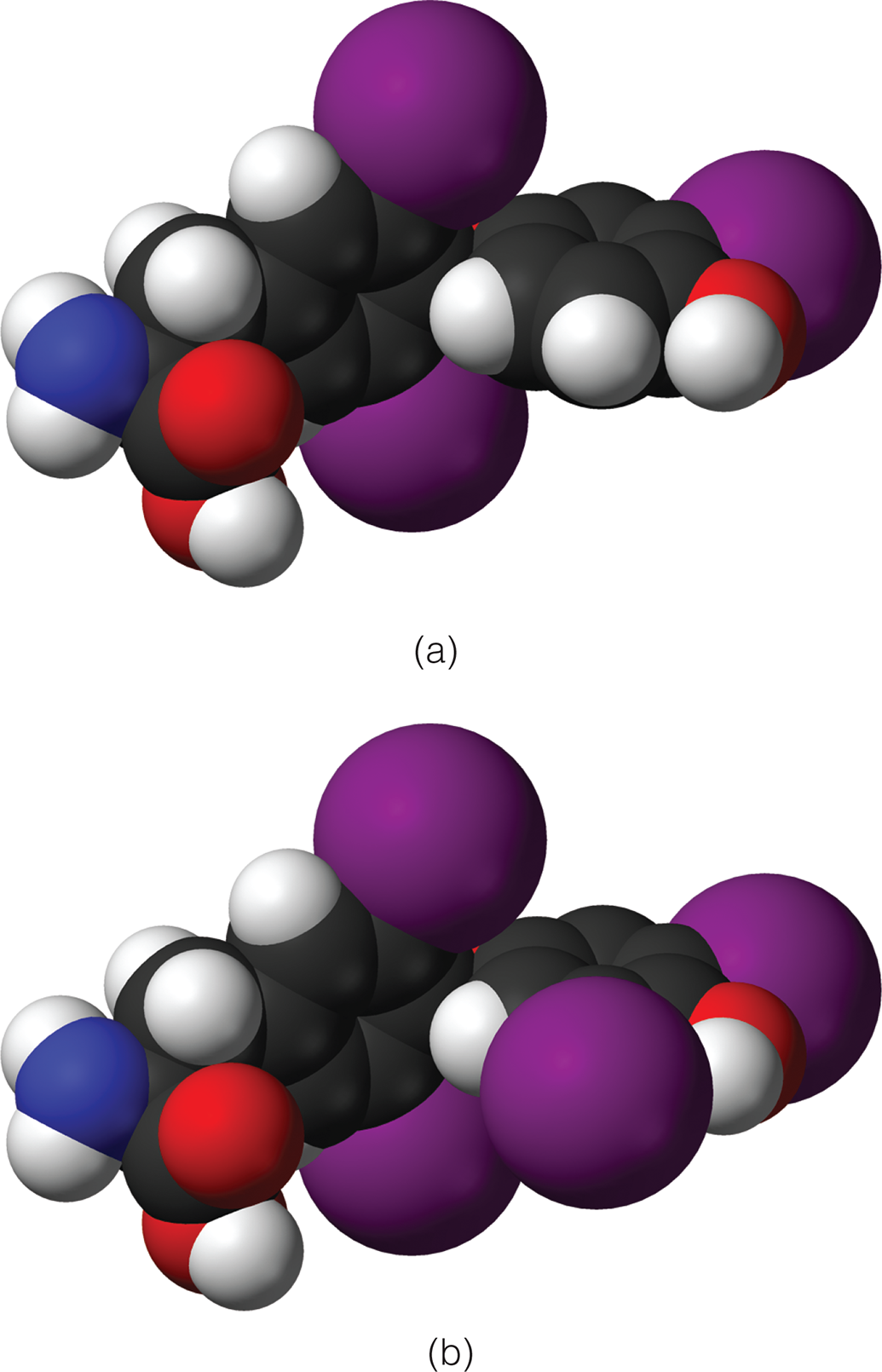
Figure 12.12 Structures of Thyroid Hormones (a) Triiodothyronine (T3); (b) Thyroxine (T4)
REAL WORLD
While thyroid hormones are not responsible for day-to-day adjustments in metabolism, insufficient thyroid hormone levels (hypothyroidism) can cause symptoms including cold intolerance, fatigue, weight gain, and depression as metabolism suffers. Excessive thyroid hormone levels (hyperthyroidism) can cause rapid weight loss, anxiety, jitteriness, and fever.
MCAT CONCEPT CHECK 12.5:
Before you move on, assess your understanding of the material with these questions.
- Describe the primary metabolic function of each of the following hormones:
______________________________
- Insulin:
______________________________
- Glucagon:
______________________________
- Cortisol:
______________________________
- Catecholamines:
________________________
- Thyroid hormones (T3 / T4):
- Thyroid storm is a potentially lethal state of extreme hyperthyroidism in which T3 and T4 levels are significantly above normal limits. What vital sign abnormalities might be expected in a patient with thyroid storm?
__________________________________
12.6 Tissue-Specific Metabolism
LEARNING OBJECTIVES
After Chapter 12.6, you will be able to:
- Identify the preferred fuel sources and fuel quantities for different tissue types, including skeletal muscle and the brain
- Recall the metabolic functions of the liver
Tissues have evolved so that their metabolic needs are met in a way corresponding to their form and function. The major sites of metabolic activity in the body are the liver, skeletal and cardiac muscles, brain, and adipocytes. Connective tissue and epithelial cells do not make major contributions to the consumption of energy. Remember though, that epithelial cells are the primary secretory cells, so they are involved in the regulation of metabolism. We have already discussed how the body operates under different nutritional conditions. The organ-specific patterns of fuel utilization in the well-fed and fasting states are summarized in Table 12.2.
Table 12.2. Preferred Fuels in the Well-Fed and Fasting States
ORGAN WELL-FED FASTING
Liver Glucose and amino acids Fatty acids
Resting skeletal muscle Glucose Fatty acids, ketones
Cardiac muscle Fatty acids Fatty acids, ketones
Adipose tissue Glucose Fatty acids
Brain Glucose Glucose (ketones in prolonged fast)
Red blood cells Glucose Glucose
Liver
Two major roles of the liver in fuel metabolism are to maintain a constant level of blood glucose under a wide range of conditions and to synthesize ketones when excess fatty acids are being oxidized. After a meal, glucose concentration in the portal blood is elevated. The liver extracts excess glucose and uses it to replenish its glycogen stores. Any glucose remaining in the liver is then converted to acetyl-CoA and used for fatty acid synthesis. The increase in insulin after a meal stimulates both glycogen synthesis and fatty acid synthesis in the liver. The fatty acids are converted to triacylglycerols and released into the blood as very-low-density lipoproteins (VLDL). In the well-fed state, the liver derives most of its energy from the oxidation of excess amino acids. Between meals and during prolonged fasts, the liver releases glucose into the blood. The increase in glucagon during fasting promotes both glycogen degradation and gluconeogenesis. Lactate from anaerobic metabolism, glycerol from triacylglycerols, and amino acids provide carbon skeletons for glucose synthesis.
Adipose Tissue
After a meal, elevated insulin levels stimulate glucose uptake by adipose tissue. Insulin also triggers fatty acid release from VLDL and chylomicrons (which carry triacylglycerols absorbed from the gut). Lipoprotein lipase, an enzyme found in the capillary bed of adipose tissue, is also induced by insulin. The fatty acids that are released from lipoproteins are taken up by adipose tissue and re-esterified to triacylglycerols for storage. The glycerol phosphate required for triacylglycerol synthesis comes from glucose that is metabolized in adipocytes as an alternative product of glycolysis. Insulin can also effectively suppress the release of fatty acids from adipose tissue. During the fasting state, decreased levels of insulin and increased epinephrine activate hormone-sensitive lipase in fat cells, allowing fatty acids to be released into circulation.
Skeletal Muscle
Resting Muscle
The major fuels of skeletal muscle are glucose and fatty acids. Because of its enormous bulk, skeletal muscle is the body’s major consumer of fuel. After a meal, insulin promotes glucose uptake in skeletal muscle, which replenishes glycogen stores and amino acids used for protein synthesis. Both excess glucose and amino acids can also be oxidized for energy. In the fasting state, resting muscle uses fatty acids derived from free fatty acids circulating in the bloodstream. Ketone bodies may also be used if the fasting state is prolonged.
Active Muscle
The primary fuel used to support muscle contraction depends on the magnitude and duration of exercise as well as the major fibers involved. A very short-lived source of energy (2–7 seconds) comes from creatine phosphate, which transfers a phosphate group to ADP to form ATP. Skeletal muscle has stores of both glycogen and some triacylglycerols. Blood glucose and free fatty acids may also be used. Short bursts of high-intensity exercise are also supported by anaerobic glycolysis drawing on stored muscle glycogen. During moderately high-intensity, continuous exercise, oxidation of glucose and fatty acids are both important, but after 1 to 3 hours of continuous exercise at this level, muscle glycogen stores become depleted, and the intensity of exercise declines to a rate that can be supported by oxidation of fatty acids.
BRIDGE
Fast-twitch muscle fibers have a high capacity for anaerobic glycolysis but are quick to fatigue. They are involved primarily in short-term, high-intensity exercise. Slow-twitch muscle fibers in arm and leg muscles are well vascularized and primarily oxidative. They are used during prolonged, low-to-moderate intensity exercise and resist fatigue. Slow-twitch fibers and the number of their mitochondria increase dramatically in trained endurance athletes. The musculoskeletal system is discussed in Chapter 11 of MCAT Biology Review.
Cardiac Muscle
Unlike other tissues of the body, cardiac myocytes prefer fatty acids as their major fuel, even in the well-fed state. When ketones are present during prolonged fasting, they can also be used. Thus, not surprisingly, cardiac myocytes most closely parallel skeletal muscle during extended periods of exercise. In patients with cardiac hypertrophy (thickening of the heart muscle), this situation reverses to some extent. In a failing heart, glucose oxidation increases and β-oxidation falls.
Brain
Although the brain represents only 2 percent of total body weight, it obtains 15 percent of the cardiac output, uses 20 percent of the total O2, and consumes 25 percent of the total glucose, the brain’s primary fuel. Blood glucose levels are tightly regulated to maintain a sufficient glucose supply for the brain (and sufficient concentration while studying). Normal function depends on a continuous glucose supply from the bloodstream. In hypoglycemic conditions
hypothalamic centers in the brain sense a fall in blood glucose level, and the release of glucagon and epinephrine is triggered. Fatty acids cannot cross the blood–brain barrier and are therefore not used at all as an energy source. Between meals, the brain relies on blood glucose supplied by either hepatic glycogenolysis or gluconeogenesis. Only during prolonged fasting does the brain gain the capacity to use ketone bodies for energy, and even then, the ketone bodies only supply approximately two-thirds of the fuel; the remainder is glucose.
BIOCHEMISTRY GUIDED EXAMPLE WITH EXPERT THINKING
Given the information in the passage, if the researchers were to assay mice that were fed a ketogenic diet versus normal chow, how would the NAD+/NADH ratio in ketogenic mice compare to that of normal mice? Why would the researchers likely be more interested in measuring NAD+/NADH ratios specifically in the brain?
The first question asks us to compare the NAD+/NADH ratio in KD mice to that of mice that were fed a normal diet, which means we should be combining our content background with the information in the passage to generate an answer. We know normal diets tend to be high in carbohydrates, whereas a ketogenic diet will be high in ketone bodies. Diagram 1 demonstrates that the metabolism of glucose and the metabolism of β-hydroxybutyrate each produce two acetyl-CoA molecules. However, the metabolism of glucose converts four NAD+ molecules to NADH, while the metabolism of β-hydroxybutyrate only converts one NAD+ molecule to NADH. This difference generates a higher NAD+/NADH ratio in those mice that are subsisting on a KD diet, as NADH levels are relatively constant (due to its production from other sources in the body) but NAD+ availability has increased.
The answer to the second question is superficially answered in the first paragraph—many of the diseases mentioned are focused on the brain (epilepsy, neurodegenerative diseases, brain cancer). However, if we dig a little deeper into our content background, we know that the brain can use only glucose or ketone bodies as its source of energy and there are no energy storage molecules, like glycogen or adipose cells, present in the brain. So, even in the early days of a ketogenic diet, while other tissues are still using alternative energy sources such as fatty acids, the brain is already shifting to utilizing a higher proportion of ketone bodies. Thus, an increased NAD+/NADH ratio, as well as its potential associated benefits, will occur in the brain sooner and to a greater extent than anywhere else in the body. The passage states that NAD+ activates pathways involving sirtuin enzymes, which reduce inflammation and fight the aging process. Such physiological changes also indirectly explain why a ketogenic diet could potentially help in so many brain-based diseases.
In short, mice on ketogenic diets would likely have a higher NAD+/NADH ratio compared to mice on a normal diet, and NAD+ levels in the brain would likely be more susceptible to diet changes because the brain only relies on plasma glucose and ketone bodies for energy.
MCAT CONCEPT CHECK 12.6:
Before you move on, assess your understanding of the material with these questions.
- What is the preferred fuel for most cells in the well-fed state? What is the exception and its preferred fuel?
- Preferred fuel: ________
- Exception: ___Preferred fuel:_____
- What organ consumes the greatest amount of glucose relative to its percentage of body mass?
____________________________________
- Describe the major metabolic functions of the liver.
_____________________________________
_____________________________________
_____________________________________
12.7 Integrative Metabolism
LEARNING OBJECTIVES
After Chapter 12.7, you will be able to:
- Predict changes to respiratory quotient with changes in activity
- Explain the role of leptin, ghrelin, and orexin in regulating body mass
- Apply knowledge of measurement methods for metabolism to analytical approaches in a lab setting
Analysis of Metabolism
There are several methods of analyzing metabolic control of an organism. In humans, levels of glucose, thyroid hormones and thyroid-stimulating hormone, insulin, glucagon, oxygen, and carbon dioxide can all be measured in the blood. Because these hormones and substrates have a predictable effect on metabolism, they can be used as indicators of metabolic function. They can also be used as indicators of disorders, as in the case of blood glucose or thyroid-stimulating hormone.
MCAT EXPERTISE
The MCAT does not expect you to know what levels are healthy for any of these indicators, but can easily pose data interpretation questions related to them.
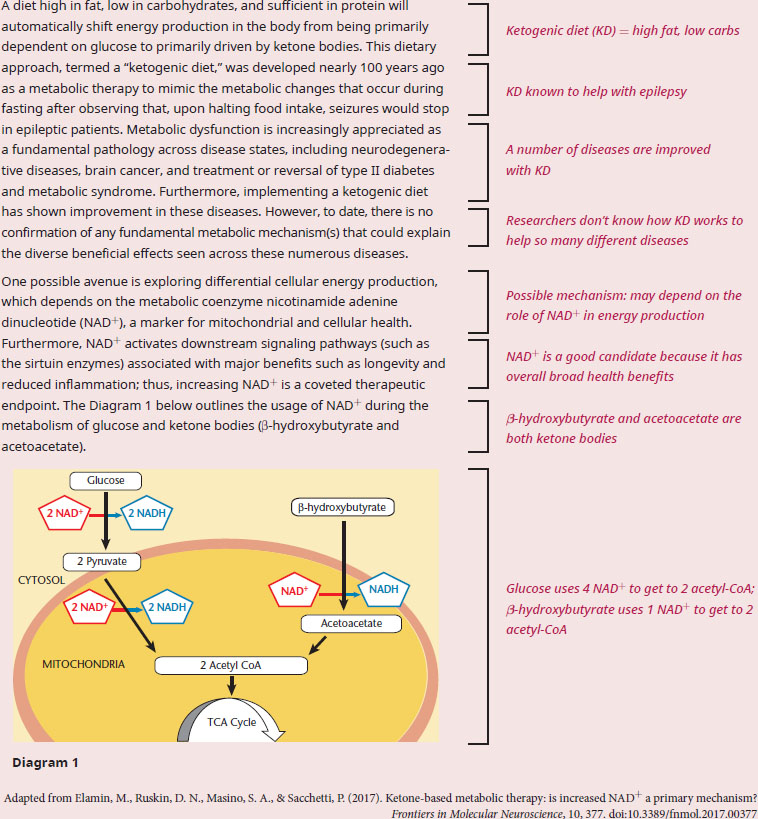
Respirometry allows accurate measurement of the respiratory quotient, which differs depending on the fuels being used by the organism. The respiratory quotient (RQ) can be measured experimentally, and can be calculated as:
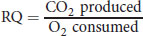
Equation 12.3
for the complete combustion of a given fuel source. The respiratory quotient for carbohydrates is around 1.0, while the respiratory quotient for lipids is around 0.7. In resting individuals, the respiratory quotient is generally around 0.8, indicating that both fat and glucose are consumed. The respiratory quotient changes under conditions of high stress, starvation, and exercise as predicted by the actions of different hormones.
Calorimeters can measure basal metabolic rate (BMR) based on heat exchange with the environment. Human calorimetry makes use of large insulated chambers with specialized heat sinks to determine energy expenditure. Because of the isolationist nature of testing and the expense of creating a calorimetry chamber, other measures of BMR are preferred. Because of previous experimentation, BMR can be estimated based on age, weight, height, and phenotypical sex.
Regulation of Body Mass
Until now, we’ve been discussing metabolism on a very small scale, but metabolic controls are also involved in maintaining body mass (weight loss or gain). Body mass is determined by several factors, including water, carbohydrates, proteins, and lipids, while nucleic acids do not contribute significantly to its maintenance. The overall mass of carbohydrates and proteins tends to be stable over time, although it can be modified slightly by periods of prolonged starvation or by significant muscle-building activities. Water is very quickly adjusted by the endocrine system and the kidneys; therefore, it does not factor into our discussion of obesity and weight regulation. Water is the primary source of frequent minor weight fluctuations because it is subject to rapid adjustment. Therefore, lipids, stored in adipocytes, are the primary factor in the gradual change of body mass over time.
An individual who is maintaining body weight consumes the same amount of energy that is spent on average each day. If energy consumed is greater than energy expenditure over a significant period of time, then fat stores begin to accumulate. The opposite is also seen. If an energy deficit exists where calories consumed are less than calories burned, then a decrease in weight is observed. As individuals increase in mass, basal metabolic rate (the amount of energy required for one sedentary day) also increases. Thus, a caloric excess will cause an increase in body mass until equilibrium is reached between the new basal metabolic rate and the existing intake. In weight loss the reverse trend is seen.
This effect does have a threshold that differs between individuals. Small adjustments in intake, even over a prolonged period of time, are partially or fully compensated by changes in energy expenditure. Similarly, a small increase or decrease in activity level will be compensated by changes in hunger. Deliberate alterations of body mass require alterations above this threshold level, which is larger in negative energy balance than in positive energy balance—in other words, larger changes must be made to lose weight than to gain it.
Diet (energy intake) and exercise (energy expenditure), genetics, socioeconomic status, and geography all play key roles in weight control. As described earlier, hormonal control by thyroid hormones, cortisol, epinephrine, glucagon, and insulin is critical to the integration of metabolism. In addition, there are hormones that control hunger and satiety, including ghrelin, orexin, and leptin. Have you ever wondered why, even if you don’t feel hungry, when you walk into your favorite restaurant you’re suddenly ravenous? This is the job of ghrelin and orexin. Ghrelin is secreted by the stomach in response to signals of an impending meal. Sight, sound, taste, and especially smell all act as signals for its release. Ghrelin increases appetite and also stimulates secretion of orexin. Orexin further increases appetite, and is also involved in alertness and the sleep–wake cycle. Hypoglycemia is also a trigger for orexin release. Leptin is a hormone secreted by fat cells that decreases appetite by suppressing orexin production. Genetic variations in the leptin molecule and its receptors have been implicated in obesity; a knockout mouse unable to produce leptin is shown on the left in Figure 12.13. These messengers and receptors are the target of current research; for now, questions regarding body mass modifications on the MCAT mostly come down to diet and exercise.
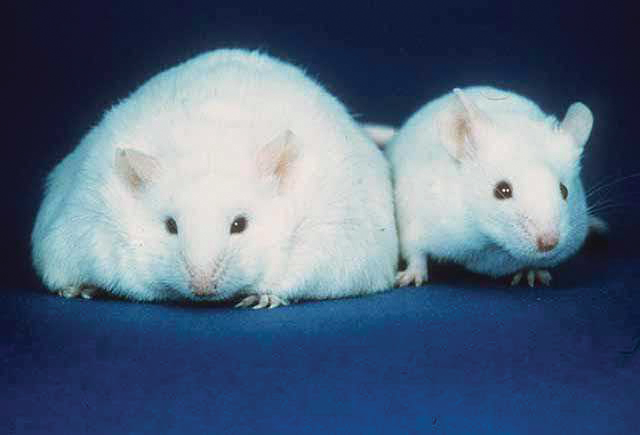
Figure 12.13 Leptin Knockout Mouse (left) Compared to Normal Mouse (right)
BRIDGE
Motivation, a psychological concept discussed in Chapter 5 of MCAT Behavioral Sciences Review, is often linked with physiological drives and signaling pathways. The hypothalamus, which produces orexin and responds to leptin and ghrelin, is responsible for regulating hunger, thirst, and libido.
Body mass can be measured and tracked using the body mass index (BMI), which is given by:
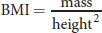
Equation 12.4
where mass is measured in kilograms and height is measured in meters. A normal BMI is considered to be between 18.5 and 25; values lower than this are considered underweight. A BMI between 25 and 30 is considered overweight, whereas a BMI over 30 is considered obese.
MCAT CONCEPT CHECK 12.7:
Before you move on, assess your understanding of the material with these questions.
- How is the respiratory quotient expected to change when a person transitions from resting to brief exercise?
_______________________________________
- True or False: Body mass can be predicted by the leptin receptor phenotype and caloric intake alone.
- True or False: It is easier to gain weight than to lose weight.
- If you were designing a study to assess metabolism, which measurement method would you choose? Defend your answer.
________________________________________
Conclusion
In this chapter, we reviewed the principles of thermodynamics and thermochemistry that were introduced in general chemistry and physics and their applications to biological systems. We looked at the specific energy molecules of human metabolism, sources of energy, and key reaction types in ATP synthesis and hydrolysis. We compared different energy states and their impact on overall metabolism and tissue-specific metabolism.
At this point you should have a decent idea about how to determine what energy sources are being used from experimental data, and be able to make predictions about the changes in metabolism under varying conditions. Congratulations, because this is the last chapter of MCAT Biochemistry Review and you’re just about ready to tackle any of the challenges that you will face on Test Day. Continue practicing, and try not to skip lunch!
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Thermodynamics and Bioenergetics
- Biological systems are considered:
- Open, wherein matter and energy can be exchanged with the environment, or
- Closed, wherein only energy can be exchanged with the environment.
- This determination is made based on the examination of the entire organism or an isolated process.
- Changes in enthalpy in a closed biological system are equal to changes in internal energy, which is equal to heat exchange within the environment.
- No work is performed in a closed biological system because pressure and volume remain constant.
- Entropy is a measure of energy dispersion in a system.
- Physiological concentrations are usually much less than standard concentrations.
- Free energy calculations must be adjusted for pH (ΔG°′), temperature (37 °C = 98.6°F = 310 K), and concentrations.
The Role of ATP
- ATP is a mid-level energy molecule.
- ATP contains high-energy phosphate bonds that are stabilized upon hydrolysis by resonance, ionization, and loss of charge repulsion.
- ATP provides energy through hydrolysis and coupling to energetically unfavorable reactions.
- ATP can also participate in phosphoryl group transfers as a phosphate donor.
Biological Oxidation and Reduction
- Biological oxidation and reduction reactions can be broken down into component half-reactions.
- Half-reactions provide useful information about stoichiometry and thermodynamics.
- Many oxidation–reduction reactions involve an electron carrier to transport high-energy electrons.
- Electron carriers may be soluble or membrane-bound.
- Flavoproteins are one subclass of electron carriers that are derived from riboflavin (vitamin B2).
Metabolic States
- Equilibrium is an undesirable state for most biochemical reactions because organisms need to harness free energy to survive.
- In the postprandial/well-fed (absorptive) state, insulin secretion is high and anabolic metabolism prevails.
- In the postabsorptive (fasting) state, insulin secretion decreases while glucagon and catecholamine secretion increases.
- This state is observed in short-term fasting (overnight).
- There is a transition to catabolic metabolism.
- Prolonged fasting (starvation) dramatically increases glucagon and catecholamine secretion.
- Most tissues rely on fatty acids.
- At maximum,
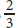
of the brain’s energy can be derived from ketone bodies.
Hormonal Regulation of Metabolism
- Insulin and glucagon have opposing activities during most aspects of metabolism.
- Insulin causes a decrease in blood glucose levels by increasing cellular uptake.
- Insulin increases the rate of anabolic metabolism.
- Insulin secretion by pancreatic β-cells is regulated by blood glucose levels.
- Glucagon increases blood glucose levels by promoting gluconeogenesis and glycogenolysis in the liver.
- Glucagon secretion by pancreatic α-cells is stimulated by both low glucose and high amino acid levels.
- Glucocorticoids increase blood glucose in response to stress by mobilizing fat stores and inhibiting glucose uptake.
- Glucocorticoids increase the impact of glucagon and catecholamines.
- Catecholamines promote glycogenolysis and increase basal metabolic rate through their sympathetic nervous system activity.
- Thyroid hormones modulate the impact of other metabolic hormones and have a direct impact on basal metabolic rate.
- T3 is more potent than T4, but has a shorter half-life and is available in lower concentrations in the blood.
- T4 is converted to T3 at the tissues.
Tissue-Specific Metabolism
- The liver is the most metabolically diverse tissue.
- Hepatocytes are responsible for the maintenance of blood glucose levels by glycogenolysis and gluconeogenesis in response to pancreatic hormone stimulation.
- The liver also participates in the processing of lipids and cholesterol, bile, urea, and toxins.
- Adipose tissue stores lipids under the influence of insulin and releases them under the influence of epinephrine.
- Skeletal muscle metabolism differs based on the current activity level and fiber type.
- Resting muscle conserves carbohydrates in glycogen stores and uses free fatty acids from the bloodstream.
- Active muscle may use anaerobic metabolism, oxidative phosphorylation of glucose, direct phosphorylation from creatine phosphate, or fatty acid oxidation, depending on fiber type and exercise duration.
- Cardiac muscle uses fatty acid oxidation in both the well-fed and fasting states.
- The brain and other nervous tissues consume glucose in all metabolic states, except for prolonged fasts, where up to
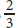
of the brain’s fuel may come from ketone bodies.
Integrative Metabolism
- Metabolic rates can be measured using calorimetry, respirometry, consumption tracking, or measurement of blood concentrations of substrates and hormones.
- Composition of fuel that is actively consumed by the body is estimated by the respiratory quotient (RQ).
- Body mass regulation is multifactorial with consumption and activity as modifiable factors.
- The hormones leptin, ghrelin, and orexin, as well as their receptors, play a role in body mass.
- Long-term changes in body mass result from changes in lipid storage.
- Changes in consumption or activity must surpass a threshold to cause weight change. The threshold is lower for weight gain than for weight loss.
- Body mass can be measured and tracked using the body mass index (BMI).
ANSWERS TO CONCEPT CHECKS
**12.1**
- ΔG°′ adjusts only for the pH of the environment by fixing it at 7. Temperature and concentrations of all other reagents are still fixed at their values from standard conditions and must be adjusted for if they are not 1 M.
- The cellular environment has a relatively fixed volume and pressure, which eliminates work from our calculations of internal energy; if ΔU = Q – W and W = 0, ΔU = Q.
-
**+ΔH −ΔH +Δ*S*** Spontaneous at high temperatures Spontaneous
**−Δ*S*** Nonspontaneous Spontaneous at low temperatures
**12.2**
- ATP hydrolysis yields about 30
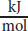
of energy, which can be harnessed to drive other reactions forward. This may either allow a nonspontaneous reaction to occur or increase the rate of a spontaneous reaction.
- ATP is an intermediate-energy storage molecule and is not energetically dense. The high-energy bonds in ATP and the presence of a significant charge make it an inefficient molecule to pack into a small space. Long-term storage molecules are characterized by energy density and stable, nonrepulsive bonds, primarily seen in lipids.
-

**12.3**
- Analyzing half-reactions can help to determine the number of electrons being transferred. This type of analysis also facilitates balancing equations and the determination of electrochemical potential if reduction potentials are provided.
-
Electron Carrier Metabolic Pathway(s) NADH Glycolysis, fermentation, citric acid cycle, electron transport chain
NADPH Pentose phosphate pathway, lipid biosynthesis, bleach formation, oxidative stress, photosynthesis
Ubiquinone (CoQ) Electron transport chain
Cytochromes Electron transport chain
Glutathione Oxidative stress
**12.4**
- Any excitable cell is maintained in a state of disequilibrium. Classic examples include muscle tissue and neurons. In addition, cell volume and membrane transport are regulated by the action of the sodium–potassium pump, which can maintain a stable disequilibrium state in most tissues.
- Cells that rely solely on anaerobic respiration are the least adaptable to different energy sources. Therefore, red blood cells are the least flexible during periods of prolonged starvation and stay reliant on glucose.
- During the postabsorptive state, there is the greatest decrease in insulin levels. The concentrations of the counterregulatory hormones (glucagon, cortisol, epinephrine, norepinephrine, and growth hormone) begin to rise.
**12.5**
- Insulin promotes glucose uptake by adipose tissue and muscle, glucose utilization in muscle cells, and macromolecule storage (glycogenesis, lipogenesis). Glucagon increases blood glucose levels by promoting glycogenolysis, gluconeogenesis, lipolysis, and ketogenesis. Cortisol increases lipolysis and amino acid mobilization, while decreasing glucose uptake in certain tissues and enhancing the activity of other counterregulatory hormones. Catecholamines increase glycogenolysis in muscle and liver and lipolysis in adipose tissue. Thyroid hormones increase basic metabolic rate and potentiate the activity of other hormones.
- Thyroid storm presents with hyperthermia (high temperature), tachycardia (fast heart rate), hypertension (high blood pressure), and tachypnea (high respiratory rate).
**12.6**
- The preferred fuel for most cells in the well-fed state is glucose; the exception is cardiac muscle, which prefers fatty acids.
- The brain consumes the greatest amount of glucose relative to its percentage of body mass.
- The liver is responsible for maintaining a steady-state concentration of glucose in the blood through glucose uptake and storage, glycogenolysis, and gluconeogenesis. The liver also participates in cholesterol and fat metabolism, the urea cycle, bile synthesis, and the detoxification of foreign substances.
**12.7**
- As a person begins to exercise, the proportion of energy derived from glucose increases. This transition to almost exclusively carbohydrate metabolism will cause the respiratory quotient to approach 1.
- False; energy expenditure, genetics, socioeconomic status, geography, and other hormones also play a role in body mass regulation.
- True; the threshold is lower for uncompensated weight gain than it is for uncompensated weight loss. Therefore, it is easier to surpass this threshold and gain weight than to lose weight.
- The methods described in the text include chemical analysis, which is objective and can quantify specific metabolic substrates, products, and enzymes; calorimetry, which is most accurate for basal metabolic rate but also most expensive; respirometry, which provides basic information about fuel sources; and caloric analysis at constant weight (food and exercise logs), which is the least invasive. Any of these answers could be defended.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. C
In a closed biological system, enthalpy, heat, and internal energy are all directly related because there is no change in pressure or volume. Because pressure and volume are fixed, work cannot be done, thus (C) is correct.
2. B
To solve this question, we can use the equation ΔG= ΔG° + RT ln Q. Q, the reaction quotient, is
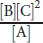
for this reaction. Plugging in the variables, we get:
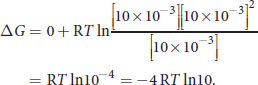
Because both R and Tare positive, and natural log values being greater than 0, we know that ΔGmust be negative and therefore lower than the original value. A negative ΔGcorresponds to a spontaneous reaction.
3. C
The hydrolysis of ATP is energetically favorable because there are repulsive negative charges that are relieved when hydrolyzed, and the new compounds are stabilized by resonance. This is true of both ATP and ADP. Some of the other answer choices are tempting, though. In (A), ATP hydrolysis relies on pH because a protonated ATP molecule contains less negative charge and therefore experiences less repulsive force. For (B), the energy released by one mole of creatine phosphate upon hydrolysis is not sufficient to phosphorylate two moles of ADP according to Table 12.1; creatine phosphate donates one phosphate group to a molecule of ADP, so one mole of creatine phosphate will phosphorylate one mole of ADP. For (D), the removal of two phosphate groups from ATP yields AMP, not cyclic AMP.
4. A
Reduction is a gain of electrons, which eliminates (B) because it is an oxidation reaction. NADPH, (C), is a product of the pentose phosphate pathway. Ubiquinone, (D), transfers electrons during the course of the electron transport chain, but is not the final electron acceptor. This title belongs to oxygen.
5. B
In order to transport electrons, electron carriers like flavoproteins must be able to exist in a stable oxidized state and a stable reduced form. ATP can be dephosphorylated but is generally not oxidized or reduced. Regulatory enzymes may also be phosphorylated or dephosphorylated but are not generally oxidized or reduced.
6. B
Skipping a single meal is not a prolonged fast. However, the increase in hormones that promote gluconeogenesis and glycogenolysis indicates that the absorptive phase has ended.
7. D
In the well-fed state, the liver runs off of glucose and amino acids, the brain and adipose tissue run off of glucose, and cardiac muscle runs off of fatty acids. Only (D) correctly pairs the tissue with its energy source.
8. A
Hormonal controls are coordinated to regulate the metabolic activity of the entire organism, while allosteric controls can be local or systemic. The modification of the enzymes of glycogen metabolism by insulin and glucagon is either through phosphorylation or dephosphorylation, both of which modify covalent bonds.
9. B
Adipose tissue and resting skeletal muscle require insulin for glucose uptake. Active skeletal muscle, (A), uses creatine phosphate and glycogen (regulated by epinephrine and AMP) to maintain its energy requirements.
10. C
Short-term glucocorticoid exposure causes a release of glucose and the hydrolysis of fats from adipocytes. However, if this glucose is not used for metabolism, it causes an increase in glucose level which promotes fat storage. The net result is the release of glucose from the liver to be converted into lipids in the adipose tissue under insulin stimulation.
11. B
The brain uses aerobic metabolism of glucose exclusively and therefore is very sensitive to oxygen levels. The extremely high oxygen requirement of the brain (20% of the body’s oxygen content) relative to its size (2% of total body weight) implies that brain is the most sensitive organ to oxygen deprivation.
12. B
The respiratory quotient (RQ) gives an indication of the primary fuel being utilized. An RQ around 0.7 indicates lipid metabolism, 0.8–0.9 indicates amino acid metabolism, (D), and 1.0 indicates carbohydrate metabolism, (A). Nucleic acids do not contribute significantly to the respiratory quotient.
13. A
Leptin acts to decrease appetite by inhibiting the production of orexin. Orexin is also associated with alertness, so decreasing the level of orexin in the body is expected to cause drowsiness. Even without this information, the answer should be apparent because the body tends to maintain an energy balance. If consumption decreases, energy expenditures are expected to decrease as well.
14. C
ATP stores are turned over about 1,000 times per day, not 10,000.
15. D
A prolonged fast is characterized by an increase in glucagon, which accomplishes its cellular activity by phosphorylating and dephosphorylating metabolic enzymes. Glycogen storage, (B), is then halted, but this requires enzyme regulation by glucagon to occur. Later in the postabsorptive state, protein breakdown, (C), begins. Eventually, in starvation, ketone bodies, (A), are used by the brain for its main energy source.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(12.1)Gibbs free energy: ΔG=ΔH−TΔS
(12.2)Modified standard state: ΔG=ΔG° + RTln(Q)
(12.3)Respiratory quotient:
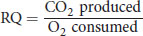
(12.4)Body mass index:
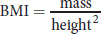
SHARED CONCEPTS
- Biochemistry Chapter 9
- Carbohydrate Metabolism I
- Biochemistry Chapter 10
- Carbohydrate Metabolism II
- Biochemistry Chapter 11
- Lipid and Amino Acid Metabolism
- Biology Chapter 5
- The Endocrine System
- General Chemistry Chapter 11
- Oxidation–Reduction Reactions
- Physics and Math Chapter 3
- Thermodynamics