Chapter 11: Lipid and Amino Acid Metabolism

Chapter 11: Lipid and Amino Acid Metabolism
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- Which of the following hormones does NOT directly regulate the activity of hormone-sensitive lipase in adipose tissue?
- Insulin
- Glucagon
- Epinephrine
- Cortisol
- What is the fate of long-chain fatty acids that are contained within micelles?
- Transport into chylomicrons released into the lymphatic system
- Transport into chylomicrons released into the circulatory system
- Direct diffusion across the intestine into the lymphatic system
- Direct diffusion across the intestine into the circulatory system
- During fatty acid mobilization, which of the following occur(s)?
- HSL is activated.
- Free fatty acids are released.
- Gluconeogenesis proceeds in adipocytes.
- I only
- III only
- I and II only
- II and III only
- How do chylomicrons and VLDL differ?
- Chylomicrons contain apoproteins, VLDL do not.
- Chylomicrons are synthesized in the intestine, VLDL are synthesized in the liver.
- Chylomicrons transport triacylglycerol, VLDL transport cholesterol.
- VLDL are another term for chylomicron remnants; they differ in age.
- Which of the following could result from an absence of apolipoproteins?
- An inability to secrete lipid transport lipoproteins
- An inability to endocytose lipoproteins
- A decreased ability to remove excess cholesterol from blood vessels
- I only
- III only
- I and II only
- I, II, and III
- Statin drugs inhibit HMG-CoA reductase. As such, they are likely prescribed for:
- hypercholesterolemia (high cholesterol).
- hypertriglyceridemia (high triacylglycerol).
- hypocholesterolemia (low cholesterol).
- visceral adiposity (obesity).
- What is the function of LCAT?
- LCAT catalyzes the production of cholesteryl esters.
- LCAT catalyzes the production of cholesterol.
- LCAT catalyzes the transfer of cholesteryl esters.
- LCAT catalyzes the transfer of cholesterol.
- Which fatty acid can be synthesized by humans?
- 12:0
- 16:0
- 16:1
- 18:3
- Which of the following best characterizes the process of fatty acid synthesis?
- Two reductions followed by a dehydration and bond formation
- Reduction followed by activation, bond formation, dehydration, and reduction
- Activation followed by bond formation, reduction, dehydration, and reduction
- Activation followed by bond formation, oxidation, dehydration, and reduction
- Which of the following best describes how the body creates or processes ketone bodies?
- Ketone bodies are produced in the peroxisomes of liver cells during periods of fasting.
- Acetyl-CoA is converted into acetoacetate, 3-hydroxybutyrate, or acetone, all of which are utilized for energy.
- Ketone bodies are produced in the mitochondria of liver cells after prolonged fasting.
- The brain is the first organ to use ketone bodies.
- The majority of triacylglycerols stored in adipocytes originate from:
- synthesis in the adipocyte.
- dietary intake.
- ketone bodies.
- synthesis in the liver.
- 2,4-Dienoyl-CoA reductase is used in the oxidation of:
- saturated fatty acids.
- monounsaturated fatty acids.
- polyunsaturated fatty acids.
- cholesterol.
- Which of the following is true regarding ketolysis?
- Ketolysis occurs only in the brain.
- Ketolysis occurs in the liver.
- Ketolysis generates acetyl-CoA.
- Ketolysis increases glucose metabolism.
- Which of the following amino acids will provide the most energy when degraded?
- Glycine
- Alanine
- Valine
- Isoleucine
- Which of the following is LEAST likely to result from protein degradation and processing by the liver?
- Fatty acids
- Glucose
- Acetoacetate
- 3-Hydroxybutyrate
Answer Key
- B
- A
- C
- B
- D
- A
- A
- B
- C
- C
- D
- C
- C
- D
- A
Chapter 11: Lipid and Amino Acid Metabolism
CHAPTER 11
LIPID AND AMINO ACID METABOLISM
In This Chapter
11.1 Lipid Digestion and Absorption
Digestion
Micelle Formation
Absorption
11.2 Lipid Mobilization 11.3 Lipid Transport
Chylomicrons
VLDL (Very-Low-Density Lipoprotein)
IDL (Intermediate-Density Lipoprotein)
LDL (Low-Density Lipoprotein)
HDL (High-Density Lipoprotein)
Apolipoproteins
11.4 Cholesterol Metabolism
Sources
Specific Enzymes
11.5 Fatty Acids and Triacylglycerols
Nomenclature
Synthesis
Oxidation
11.6 Ketone Bodies
Ketogenesis
Ketolysis
11.7 Protein Catabolism Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 2% of all questions about Biochemistry on the MCAT.
This chapter covers material from the following AAMC content category:
1D: Principles of bioenergetics and fuel molecule metabolism
Introduction
For weeks before the winter season begins, bears and certain mammals increase their food intake to prepare for hibernation. During this time, they increase their weight by storing energy. Different organisms store fuel and supplies in different ways. Hamsters store extra food in pouches in their cheeks. Cacti absorb and conserve water in preparation for dry seasons. But hibernating animals store extra calories as fat. Over the course of the winter, fat stores are mobilized and metabolized for basic bodily functions, which are minimal during hibernation. Come spring and summer, these reserves will be replenished in preparation for the next winter season. Humans also store extra energy as fat. While we may not hibernate through the winter, fat stores allow us to store energy to use during prolonged periods without food.
As discussed in Chapter 8 of MCAT Biochemistry Review, lipids play a major role in maintaining the structure and function of cells; however, they also have important roles as storage molecules for energy and in biological signaling. In this chapter, we’ll examine the metabolism of lipids, starting with ingestion of food particles and continuing through absorption, transport, and energy catabolism. We will also cover energy storage via lipid synthesis, as well as the metabolism of cholesterol and ketone bodies. In addition, we will learn about how protein degradation feeds into lipid and carbohydrate pathways and the urea cycle.
MCAT EXPERTISE
Chapter 11 contains some of the most complex and difficult material in this book. However, the content within this chapter is not particularly high-yield on Test Day, as can be seen in the chapter profile above. This chapter is still worth reviewing, but its content will be most relevant to students seeking full mastery of all testable content for an extremely competitive score.
11.1 Lipid Digestion and Absorption
LEARNING OBJECTIVES
After Chapter 11.1, you will be able to:
- Differentiate between lipid digestion in the stomach and lipid digestion in the intestines
- Identify the methods by which lipids can enter circulation
- Describe the structure of a micelle
In addition to being a major source of energy in the body, lipids serve a variety of other functions in the body. For instance, some fat-soluble vitamins play roles as coenzymes; prostaglandins and steroid hormones are necessary in the control and maintenance of homeostasis. Aberrant lipid metabolism may also be associated with clinical manifestations such as atherosclerosis and obesity.
Digestion
Dietary fat consists mainly of triacylglycerols, with the remainder comprised of cholesterol, cholesteryl esters, phospholipids, and free fatty acids. Lipid digestion is minimal in the mouth and stomach; lipids are transported to the small intestine essentially intact. Upon entry into the duodenum, emulsification occurs, which is the mixing of two normally immiscible liquids (in this case, fat and water). Formation of an emulsion increases the surface area of the lipid, which permits greater enzymatic interaction and processing. Emulsification is aided by bile, which contains bile salts, pigments, and cholesterol; bile is secreted by the liver and stored in the gallbladder. Finally, the pancreas secretes pancreatic lipase, colipase, and cholesterol esterase into the small intestine; together, these enzymes hydrolyze the lipid components to 2-monoacylglycerol, free fatty acids, and cholesterol. Figure 11.1 summarizes the digestion and absorption of dietary lipid components.
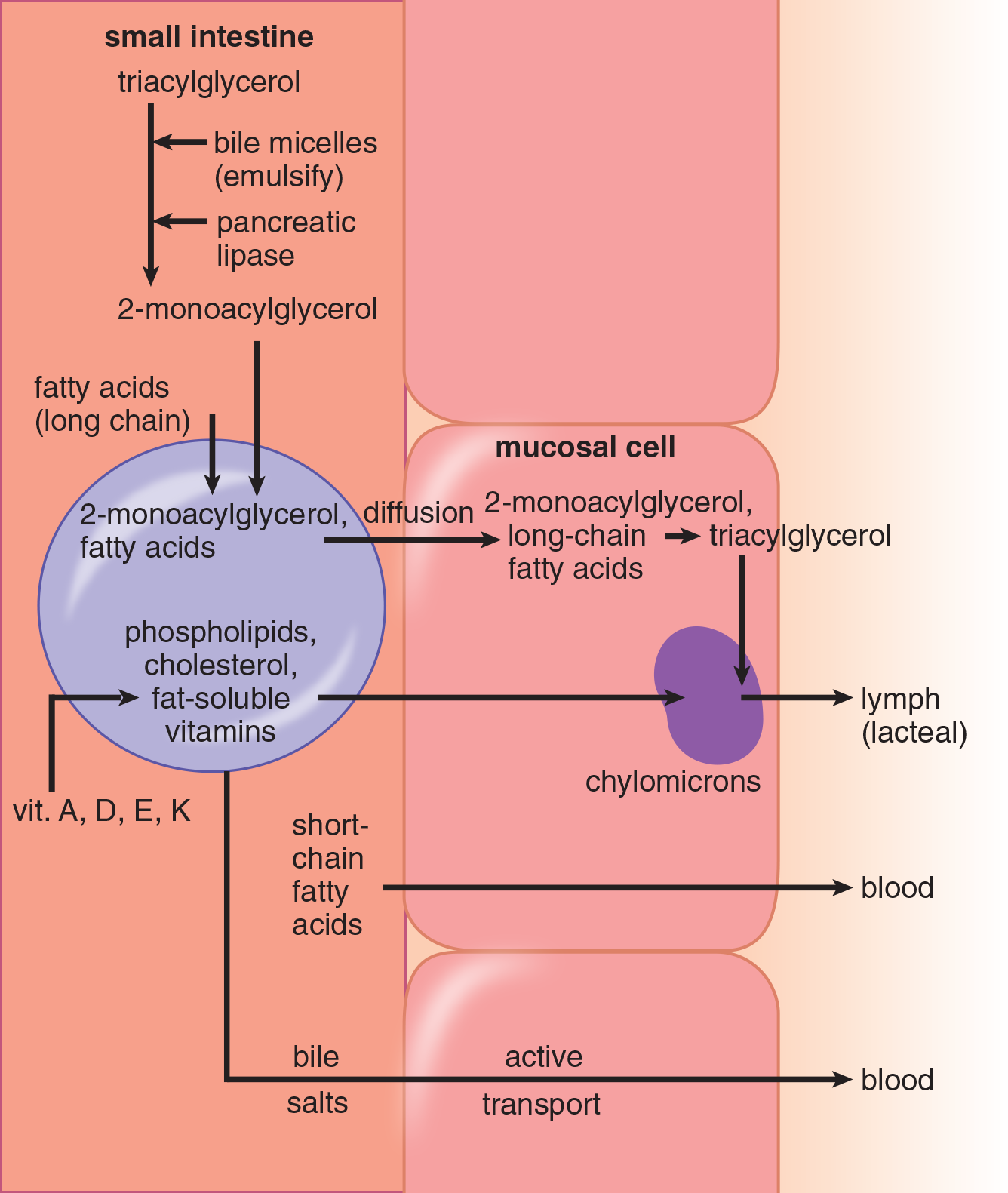
Figure 11.1 Absorption of Lipids
BRIDGE
Digestion is covered in Chapter 9 of MCAT Biology Review
Micelle Formation
Emulsification is followed by absorption of fats by intestinal cells. Free fatty acids, cholesterol, 2-monoacylglycerol, and bile salts contribute to the formation of micelles, which are clusters of amphipathic lipids that are soluble in the aqueous environment of the intestinal lumen. Essentially, micelles are water-soluble spheres with a lipid-soluble interior. Micelles are vital in digestion, transport, and absorption of lipid-soluble substances starting from the duodenum all the way to the end of the ileum. At the end of the ileum, bile salts are actively reabsorbed and recycled; any fat that remains in the intestine will pass into the colon, and ultimately ends up in the stool.
Absorption
Micelles diffuse to the brush border of the intestinal mucosal cells where they are absorbed. The digested lipids pass through the brush border, where they are absorbed into the mucosa and re-esterified to form triacylglycerols and cholesteryl esters and packaged, along with certain apoproteins, fat-soluble vitamins, and other lipids, into chylomicrons. Chylomicrons leave the intestine via lacteals, the vessels of the lymphatic system, and re-enter the bloodstream via the thoracic duct, a long lymphatic vessel that empties into the left subclavian vein at the base of the neck. The more water-soluble short-chain fatty acids can be absorbed by simple diffusion directly into the bloodstream.
BRIDGE
Absorption in the small intestine and colon follows a characteristic pattern. This is a good time to review digestion, discussed in Chapter 9 of MCAT Biology Review, to create a complete schema for the absorption and metabolism of all the macronutrients.
MCAT CONCEPT CHECK 11.1:
Before you move on, assess your understanding of the material with these questions.
- When lipids leave the stomach, what stages of digestion have been accomplished? What enzymes are added to accomplish the next phase?
___________________________________
- True or False: All lipids enter the circulation through the lymphatic system.
- Describe the structure of a micelle.
____________________________________
11.2 Lipid Mobilization
LEARNING OBJECTIVES
After Chapter 11.2, you will be able to:
- Identify the conditions and hormones that promote lipid mobilization
- Predict the ratio of free fatty acids per glycerol molecule broken down via lipid mobilization
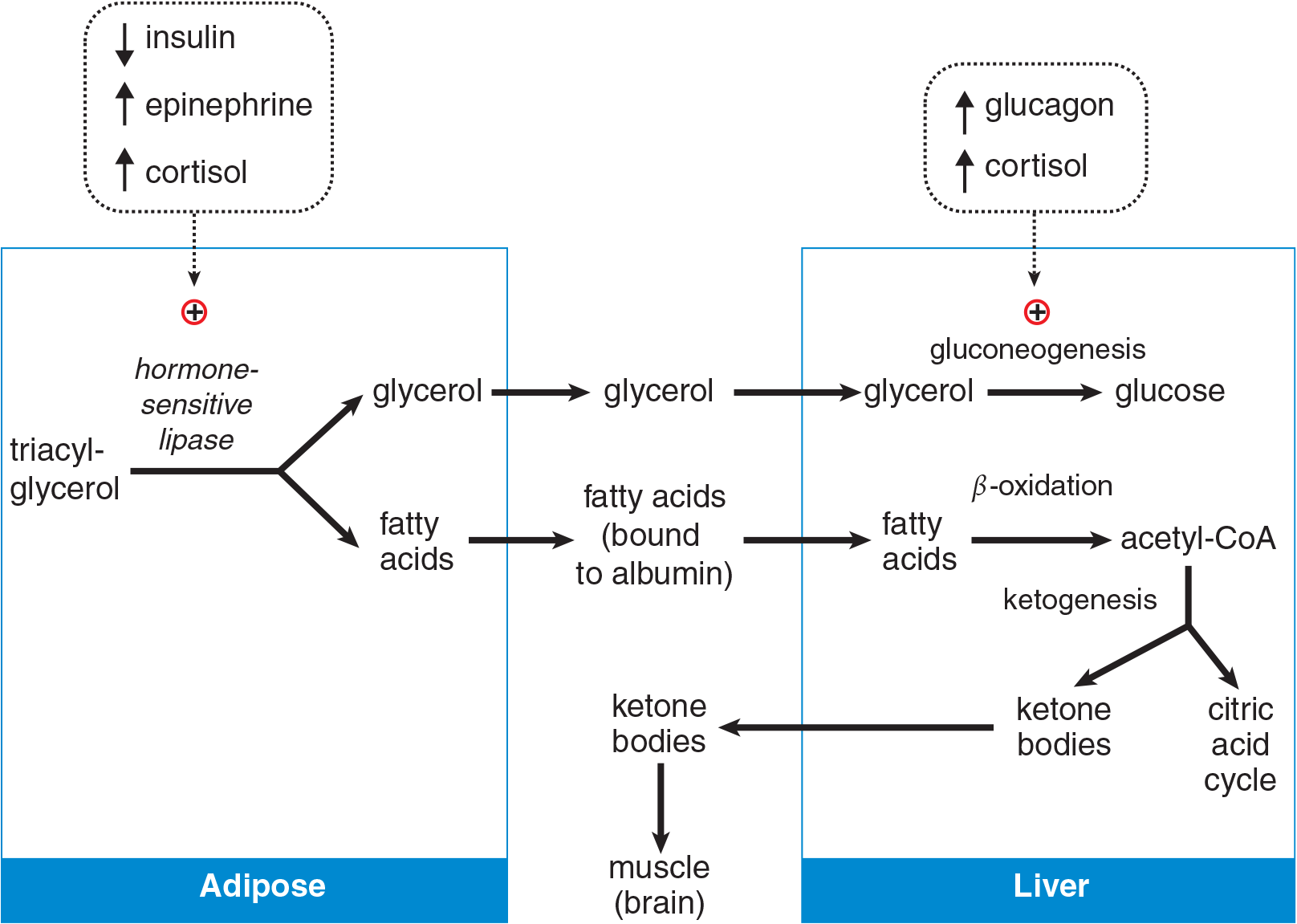
Figure 11.2 Mobilization of Triacylglycerols and Metabolism by the Liver
At night, the body is in the postabsorptive state, utilizing energy stores instead of food for fuel. In the postabsorptive state, fatty acids are released from adipose tissue and used for energy. Although human adipose tissue does not respond directly to glucagon, a fall in insulin levels activates a hormone-sensitive lipase (HSL) that hydrolyzes triacylglycerols, yielding fatty acids and glycerol. Epinephrine and cortisol can also activate HSL, as shown in Figure 11.2; we will discuss the effects of these hormones on metabolism in more detail in the next chapter. Released glycerol from fat may be transported to the liver for glycolysis or gluconeogenesis. HSL is effective within adipose cells, but lipoprotein lipase (LPL) is necessary for the metabolism of chylomicrons and very-low-density lipoproteins (VLDL). LPL is an enzyme that can release free fatty acids from triacylglycerols in these lipoproteins.
MCAT CONCEPT CHECK 11.2:
Before you move on, assess your understanding of the material with these questions.
- A patient who has diabetes begins insulin injections for management of blood glucose levels. What is the expected impact on the patient's weight?
_______________________________________
- What is the ratio of free fatty acids to glycerol produced through lipid mobilization?
_______________________________________
11.3 Lipid Transport
LEARNING OBJECTIVES
After Chapter 11.3, you will be able to:
- Describe the means of transport for free fatty acids in the blood
- Order lipoproteins according to percentage of protein and lipid transported
- Explain the relationship between the liver and the intestinal epithelium in relation to lipoproteins
- Recall the major structural components of a lipoprotein
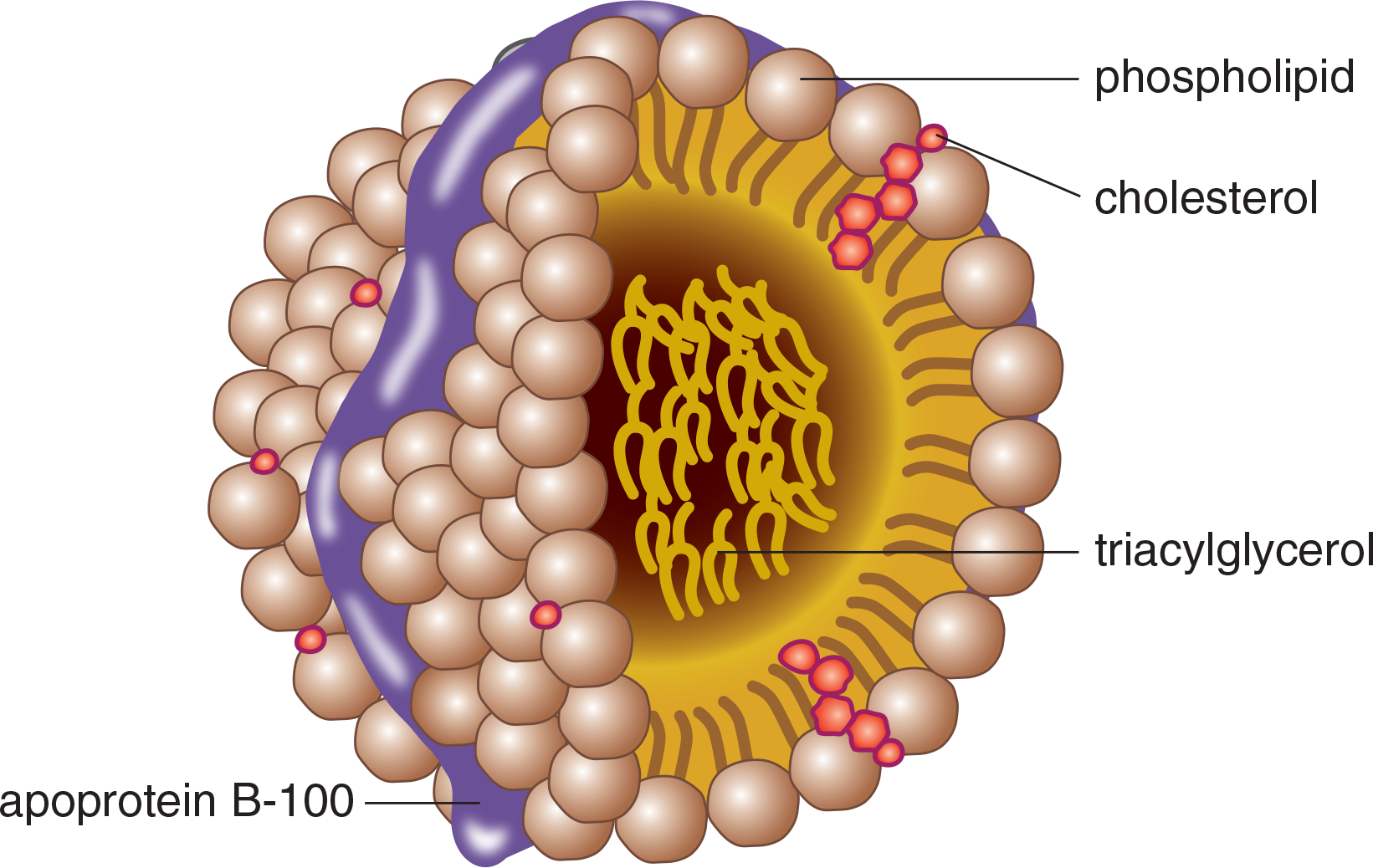
Figure 11.3 Lipoprotein Structure
While free fatty acids are transported through the blood in association with albumin, a carrier protein, triacylglycerol and cholesterol are transported in the blood as lipoproteins: aggregates of apolipoproteins and lipids, as shown in Figure 11.3. Lipoproteins are named according to their density, which increases in direct proportion to the percentage of protein in the particle. Chylomicrons are the least dense, with the highest fat-to-protein ratio. VLDL (very-low-density lipoprotein) is slightly more dense, followed by IDL (intermediate-density), LDL (low-density), and HDL (high-density). The main functions of each lipoprotein are shown in Table 11.1. Note that chylomicrons and VLDL primarily carry triacylglycerols, but also contain small quantities of cholesteryl esters. LDL and HDL are primarily cholesterol transport molecules.
Table 11.1. Classes of Lipoproteins
LIPOPROTEIN FUNCTIONS
Chylomicrons Transport dietary triacylglycerols, cholesterol, and cholesteryl esters from intestine to tissues
VLDL Transports triacylglycerols and fatty acids from liver to tissues
IDL (VLDL remnants) Picks up cholesteryl esters from HDL to become LDL Picked up by the liver
LDL Delivers cholesterol into cells
HDL Picks up cholesterol accumulating in blood vessels Delivers cholesterol to liver and steroidogenic tissues Transfers apolipoproteins to other lipoproteins
Chylomicrons
Chylomicrons are highly soluble in both lymphatic fluid and blood and function in the transport of dietary triacylglycerols, cholesterol, and cholesteryl esters to other tissues. Assembly of chylomicrons occurs in the intestinal lining and results in a nascent chylomicron that contains lipids and apolipoproteins.
VLDL (Very-Low-Density Lipoprotein)
VLDL metabolism is similar to that of chylomicrons; however, VLDL is produced and assembled in liver cells. Like chylomicrons, the main function of VLDL is the transport of triacylglycerols to other tissues. VLDLs also contain fatty acids that are synthesized from excess glucose or retrieved from chylomicron remnants.
KEY CONCEPT
Chylomicrons and VLDL primarily carry triacylglycerols. LDL and HDL primarily carry cholesterol. IDL is intermediate; it is a transition state between VLDL and LDL, occurring as the primary lipid within the lipoprotein changes from triacylglycerol to cholesterol.
IDL (Intermediate-Density Lipoprotein)
Once triacylglycerol is removed from VLDL, the resulting particle is referred to as either a VLDL remnant or IDL. Some IDL is reabsorbed by the liver by apolipoproteins on its exterior, and some is further processed in the bloodstream. For example, some IDL picks up cholesteryl esters from HDL to become LDL. IDL thus exists as a transition particle between triacylglycerol transport (associated with chylomicrons and VLDL) and cholesterol transport (associated with LDL and HDL). This process is shown in Figure 11.4.
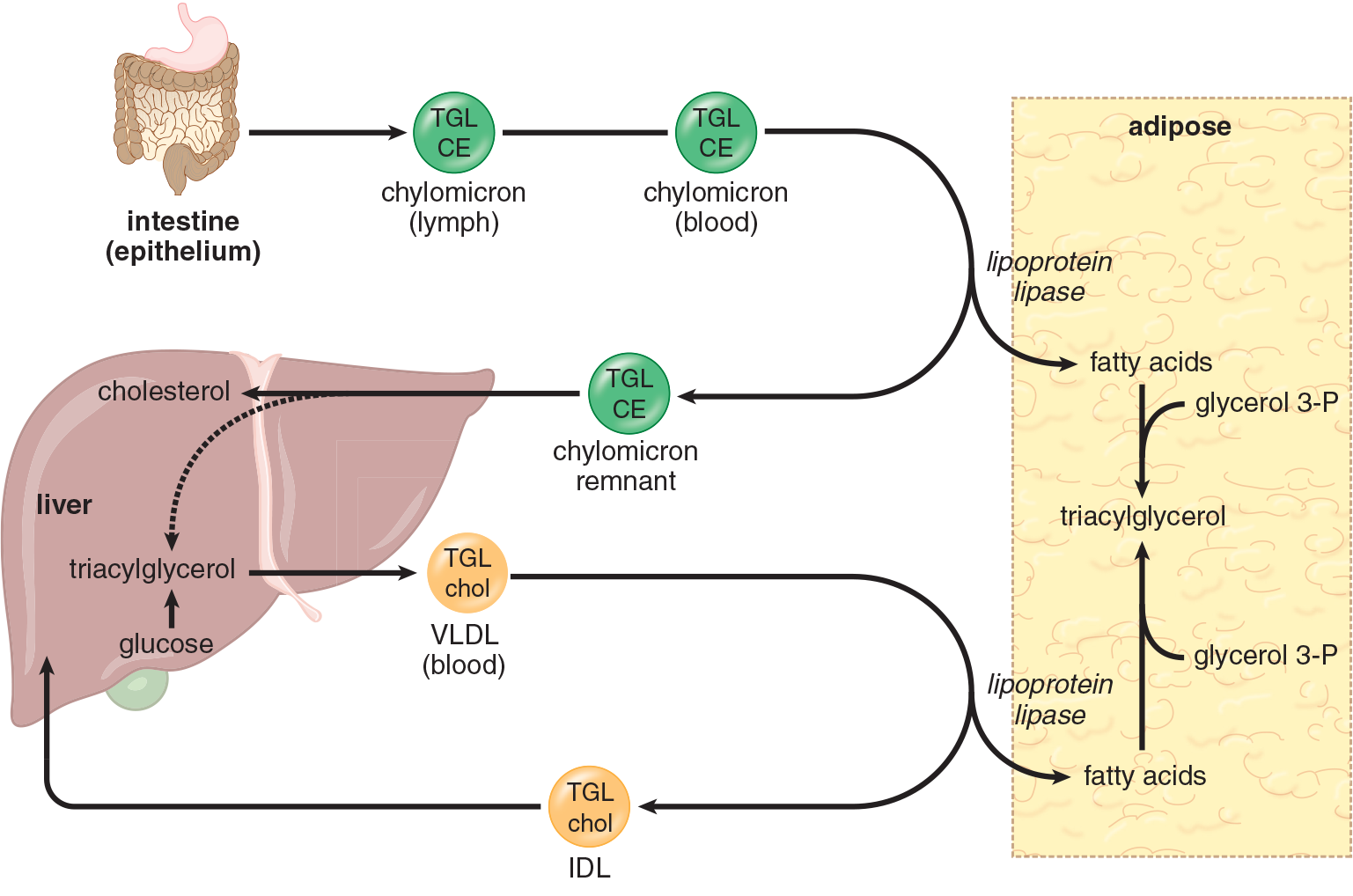
Figure 11.4 Lipid Transport in Lipoproteins TGL = triacylglycerol; CE = cholesteryl esters; chol = cholesterol
LDL (Low-Density Lipoprotein)
Although both LDL and HDL are primarily cholesterol particles, the majority of the cholesterol measured in blood is associated with LDL. The normal role of LDL is to deliver cholesterol to tissues for biosynthesis. However, cholesterol also plays an important role in cell membranes. In addition, bile acids and salts are made from cholesterol in the liver, and many other tissues require cholesterol for steroid hormone synthesis (steroidogenesis).
HDL (High-Density Lipoprotein)
HDL is synthesized in the liver and intestines and released as dense, protein-rich particles into the blood. HDL contains apolipoproteins used for cholesterol recovery—that is, the cleaning up of excess cholesterol from blood vessels for excretion. HDL also delivers some cholesterol to steroidogenic tissues and transfers necessary apolipoproteins to some of the other lipoproteins.
REAL WORLD
When a physician orders a blood test for cholesterol, they are actually measuring levels of LDL and HDL in the blood. HDL is often considered “good” cholesterol because it picks up excess cholesterol from blood vessels for excretion.
Apolipoproteins
Apolipoproteins, also referred to as apoproteins, form the protein component of the lipoproteins described above. Apolipoproteins are receptor molecules and are involved in signaling. While it is highly unlikely that specific functions of each apolipoprotein will be tested on the MCAT, they are briefly summarized below to illustrate their diverse purposes:
- apoA-I: activates LCAT, an enzyme that catalyzes cholesterol esterification
- apoB-48: mediates chylomicron secretion
- apoB-100: permits uptake of LDL by the liver
- apoC-II: activates lipoprotein lipase
- apoE: permits uptake of chylomicron remnants and VLDL by the liver
MCAT CONCEPT CHECK 11.3:
Before you move on, assess your understanding of the material with these questions.
- What is the primary method of transporting free fatty acids in the blood?
_________________________________________
- Order the lipoproteins from greatest percentage of protein to least percentage of protein. Circle the molecules that are primarily involved in triacylglycerol transport.
________________________________________
- Lipoproteins are synthesized primarily by which two organs?
_________________________________________
-
When physicians order a lipid panel to evaluate a patient, which value do they prefer to see over a minimum threshold rather than below a maximum?
________________________________________
11.4 Cholesterol Metabolism
LEARNING OBJECTIVES
After Chapter 11.4, you will be able to:
- Predict optimal conditions for HMG-CoA reductase activity
- Recall the functions of the citrate shuttle, HMG-CoA reductase, LCAT, and CETP
Cholesterol is a ubiquitous component of all cells in the human body and plays a major role in the synthesis of cell membranes, steroid hormones, bile acids, and vitamin D.
Sources
Most cells derive their cholesterol from LDL or HDL, but some cholesterol may be synthesized de novo. De novo synthesis of cholesterol occurs in the liver and is driven by acetyl-CoA and ATP. The citrate shuttle carries mitochondrial acetyl-CoA into the cytoplasm, where synthesis occurs. NADPH (from the pentose phosphate pathway) supplies reducing equivalents. Synthesis of mevalonic acid in the smooth endoplasmic reticulum (SER) is the rate-limiting step in cholesterol biosynthesis and is catalyzed by 3-hydroxy-3-methylglutaryl (HMG) CoA reductase. Cholesterol synthesis is regulated in several ways. First, increased levels of cholesterol can inhibit further synthesis by a feedback inhibition mechanism. Next, insulin promotes cholesterol synthesis. Control over de novo cholesterol synthesis is also dependent on regulation of HMG-CoA reductase gene expression in the cell.
Specific Enzymes
Specialized enzymes involved in the transport of cholesterol include LCAT and CETP. Lecithin–cholesterol acyltransferase (LCAT) is an enzyme found in the bloodstream that is activated by HDL apoproteins. LCAT adds a fatty acid to cholesterol, which produces soluble cholesteryl esters such as those in HDL. HDL cholesteryl esters can be distributed to other lipoproteins like IDL, which becomes LDL by acquiring these cholesteryl esters. The cholesteryl ester transfer protein (CETP) facilitates this transfer process.
MCAT CONCEPT CHECK 11.4:
Before you move on, assess your understanding of the material with these questions.
- Under what conditions is HMG-CoA reductase most active? In what cellular region does it exist?
______________________________________
______________________________________
______________________________________
- What proteins are specific to the formation and transmission of cholesteryl esters, and what are their functions?
______________________________________
______________________________________
11.5 Fatty Acids and Triacylglycerols
LEARNING OBJECTIVES
After Chapter 11.5, you will be able to:
- Recall and sequence the five steps in the addition of acetyl-CoA to a growing fatty acid chain
- Differentiate β-oxidation of unsaturated fatty acids from that of saturated fatty acids
- Identify the cellular locations involved in fatty acid synthesis and modification
- Name fatty acids and predict their structure based on their nomenclature:
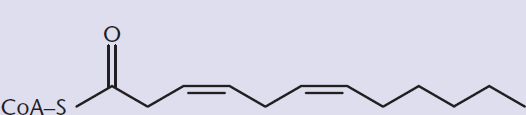
Fatty acids are long-chain carboxylic acids. The carboxyl carbon is carbon 1, and carbon 2 is referred to as the α-carbon. Fatty acids found within the body occur as salts that are capable of forming micelles or are esterified to other compounds, such as the membrane lipids discussed in Chapter 8 of MCAT Biochemistry Review.
Nomenclature
When describing a fatty acid, the total number of carbons is given along with the number of double bonds, written as carbons:double bonds. Further description can be given by indicating the position and isomerism of the double bonds in an unsaturated fatty acid. Saturated fatty acids have no double bonds while unsaturated fatty acids have one or more double bonds. Humans can synthesize only a few of the unsaturated fatty acids; the rest come from essential fatty acids found in the diet that are transported in chylomicrons as triacylglycerols from the intestine. Two important essential fatty acids are α-linolenic acid and linoleic acid. These polyunsaturated fatty acids, as well as other acids formed from them, are important in maintaining cell membrane fluidity, which is critical for proper functioning of the cell. The omega (ω) numbering system is also used for unsaturated fatty acids. The ω designation describes the position of the last double bond relative to the end of the chain and identifies the major precursor fatty acid. For example, linoleic acid (18:2 cis,cis-9,12) is the precursor of the ω-6 family, which includes arachidonic acid. α-Linolenic acid (18:3 all-cis-9,12,15) is the primary precursor of the ω-3 family. Double bonds in natural fatty acids are generally in the cis configuration.
REAL WORLD
Trans double bonds are uncommon in natural fatty acids; they predominate in fatty acids found in margarine and other foods that use partial hydrogenation of vegetable oils in their preparation. Compared with liquid oils, these partially hydrogenated fatty acids are solids at room temperature. These fatty acids contribute to arterial diseases and decreased membrane fluidity.
Synthesis
Fatty acids used by the body for fuel are supplied primarily by the diet. In addition, excess carbohydrate and protein acquired from the diet can be converted to fatty acids and stored as energy reserves in the form of triacylglycerols. Lipid and carbohydrate synthesis are often called nontemplate synthesis processes because they do not rely directly on the coding of a nucleic acid, unlike protein and nucleic acid synthesis.
Fatty Acid Biosynthesis
Fatty acid biosynthesis, shown in Figure 11.5, occurs in the liver and its products are subsequently transported to adipose tissue for storage. Adipose tissue can also synthesize smaller quantities of fatty acids. Both of the major enzymes of fatty acid synthesis, acetyl-CoA carboxylase and fatty acid synthase, are also stimulated by insulin. Palmitic acid (palmitate) is the primary end product of fatty acid synthesis.
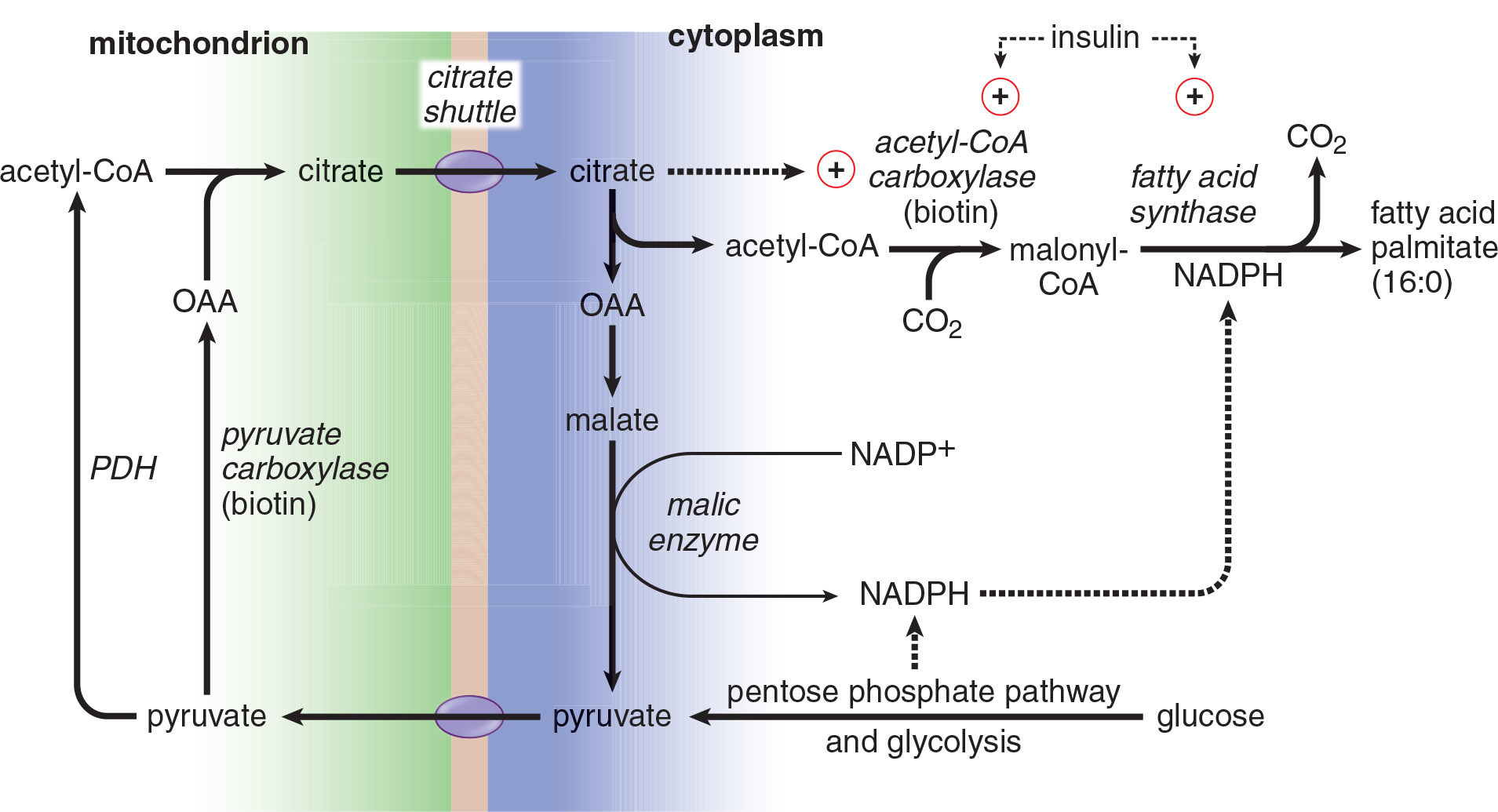
Figure 11.5 Fatty Acid Synthesis from Glucose
Acetyl-CoA Shuttling
Following a large meal, acetyl-CoA accumulates in the mitochondrial matrix and needs to be moved to the cytosol for fatty acid biosynthesis. Acetyl-CoA is the product of the pyruvate dehydrogenase complex, and it couples with oxaloacetate to form citrate at the beginning of the citric acid cycle. Remember that isocitrate dehydrogenase is the rate-limiting enzyme of citric acid cycle; as the cell becomes energetically satisfied, it slows the citric acid cycle, which causes citrate accumulation. Citrate can then diffuse across the mitochondrial membrane. In the cytosol, citrate lyase splits citrate back into acetyl-CoA and oxaloacetate. The oxaloacetate can then return to the mitochondrion to continue moving acetyl-CoA.
Acetyl-CoA Carboxylase
Acetyl-CoA is activated in the cytoplasm for incorporation into fatty acids by acetyl-CoA carboxylase, the rate-limiting enzyme of fatty acid biosynthesis. Acetyl-CoA carboxylase requires biotin and ATP to function, and adds CO2 to acetyl-CoA to form malonyl-CoA. The enzyme is activated by insulin and citrate. The CO2 added to form malonyl-CoA is never actually incorporated into the fatty acid because it is removed by fatty acid synthase during addition of the activated acetyl group to the fatty acid.
Fatty Acid Synthase
Fatty acid synthase is more appropriately called palmitate synthase because palmitate is the only fatty acid that humans can synthesize de novo. Fatty acid synthase is a large multienzyme complex found in the cytosol that is rapidly induced in the liver following a meal high in carbohydrates because of elevated insulin levels. The enzyme complex contains an acyl carrier protein (ACP) that requires pantothenic acid (vitamin B5). NADPH is also required to reduce the acetyl groups added to the fatty acid. Eight acetyl-CoA groups are required to produce palmitate (16:0). Fatty acyl-CoA may be elongated and desaturated, to a limited extent, using enzymes associated with the smooth endoplasmic reticulum (SER). The steps involved in fatty acid biosynthesis are shown in Figure 11.6 and include attachment to an acyl carrier protein, bond formation between activated malonyl-CoA (malonyl-ACP) and the growing chain, reduction of a carbonyl group, dehydration, and reduction of a double bond. These reactions occur over and over again until the sixteen-carbon palmitate molecule is created. Many of these reactions are reversed in β-oxidation.
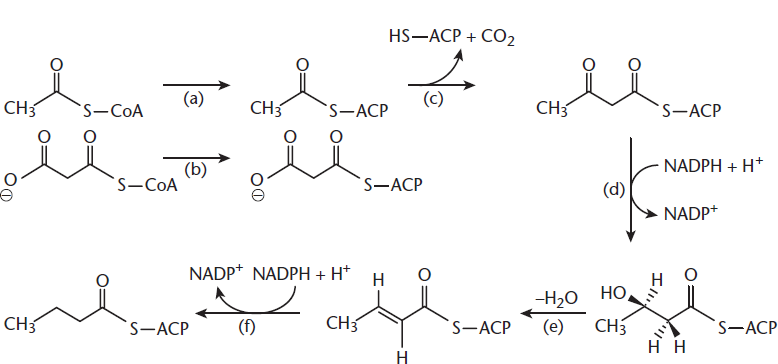
Figure 11.6 Action of Fatty Acid Synthase Reactions include activation of the growing chain (a) and malonyl-CoA (b) with ACP, bond formation between these activated molecules (c), reduction of a carbonyl to a hydroxyl group (d), dehydration (e), and reduction to a saturated fatty acid (f).
Triacylglycerol (Triglyceride) Synthesis
Triacylglycerols, the storage form of fatty acids, are formed by attaching three fatty acids (as fatty acyl-CoA) to glycerol. Triacylglycerol formation from fatty acids and glycerol 3-phosphate occurs primarily in the liver and somewhat in adipose tissue, with a small contribution directly from the diet, as well. In the liver, triacylglycerols are packaged and sent to adipose tissue as very-low-density lipoproteins (VLDL), leaving only a small amount of stored triacylglycerols.
KEY CONCEPT
Fatty acid synthesis and β-oxidation are reverse processes. Both involve transport across the mitochondrial membrane, followed by a series of redox reactions, but always in the opposite direction of one another. Understanding one process will enable you to answer questions about both pathways.
Oxidation
Most fatty acid catabolism proceeds via ****β*-***oxidation that occurs in the mitochondria; however, peroxisomal β-oxidation also occurs. Branched-chain fatty acids may also undergo α-oxidation, depending on the branch points, while ω-oxidation in the endoplasmic reticulum produces dicarboxylic acids. You should be aware that these processes exist; however, the mechanisms are beyond the scope of the MCAT. We will take an in-depth look at β-oxidation, which will be much more heavily tested. Insulin indirectly inhibits β-oxidation while glucagon stimulates this process.
Activation
When fatty acids are metabolized, they first become activated by attachment to CoA, which is catalyzed by fatty-acyl-CoA synthetase. The product is generically referred to as a fatty acyl-CoA or acyl-CoA. Specific examples would be acetyl-CoA containing a 2-carbon acyl group, or palmitoyl-CoA with a 16-carbon acyl group.
Fatty Acid Entry into Mitochondria
Short-chain fatty acids (2 to 4 carbons) and medium-chain fatty acids (6 to 12 carbons) diffuse freely into mitochondria, where they are oxidized. In contrast, while long-chain fatty acids (14 to 20 carbons) are also oxidized in the mitochondria, they require transport via a carnitine shuttle, as shown in Figure 11.7. Carnitine acyltransferase I is the rate-limiting enzyme of fatty acid oxidation. Very long chain fatty acids (over 20 carbons) are oxidized elsewhere in the cell.
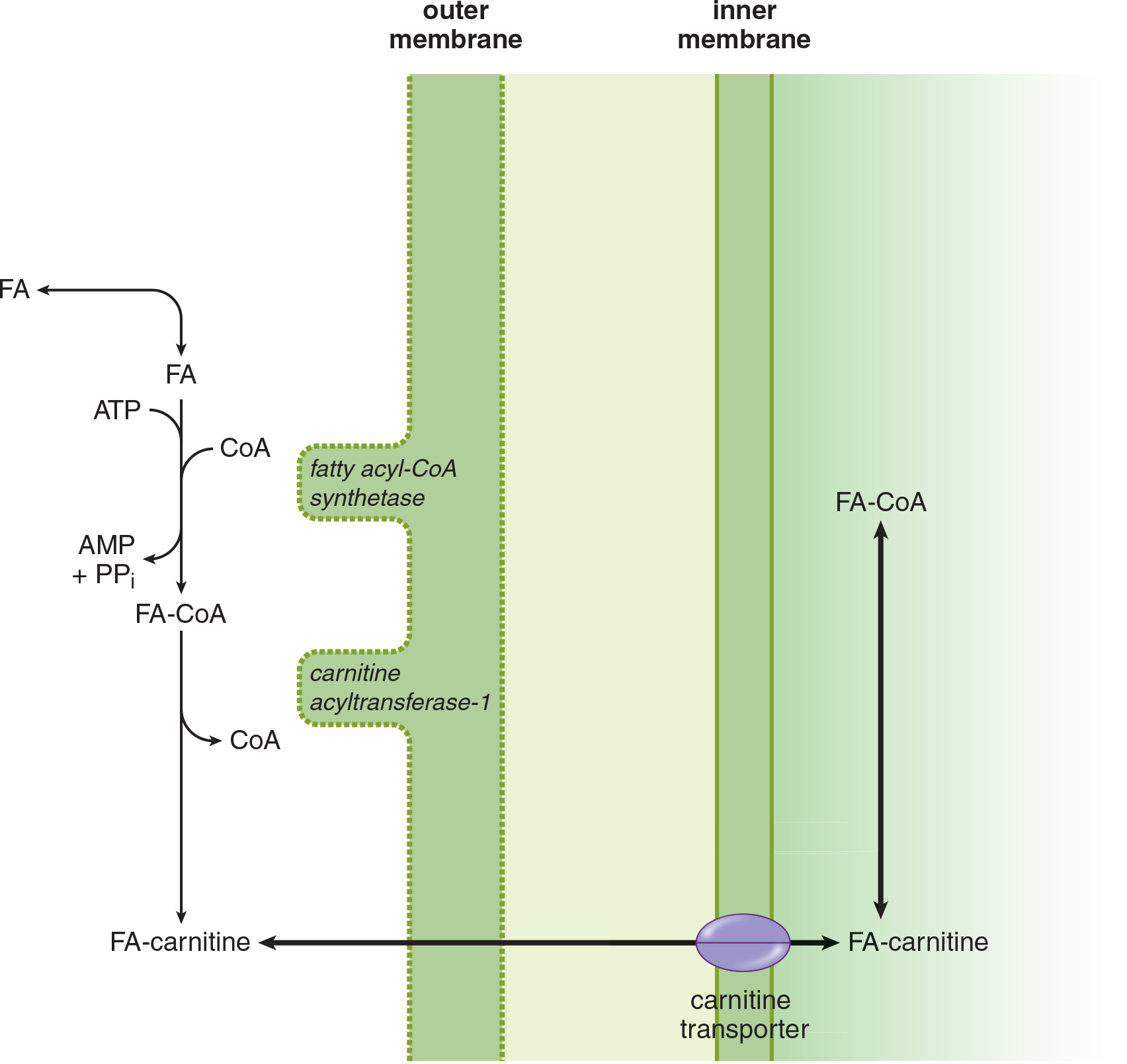
Figure 11.7 Fatty Acid Activation and Transport
*β*-Oxidation in Mitochondria
β-Oxidation reverses the process of fatty acid synthesis by oxidizing and releasing (rather than reducing and linking) molecules of acetyl-CoA. The pathway is a repetition of four steps; each four-step cycle releases one acetyl-CoA and reduces NAD+ and FAD (producing NADH and FADH2). The FADH2 and NADH are oxidized in the electron transport chain, producing ATP. In muscle and adipose tissue, acetyl-CoA enters the citric acid cycle. In the liver, acetyl-CoA, which cannot be converted to glucose, stimulates gluconeogenesis by activating pyruvate carboxylase. In a fasting state, the liver produces more acetyl-CoA from β-oxidation than is used in the citric acid cycle. Much of the acetyl-CoA is used to synthesize ketone bodies (essentially two acetyl-CoA molecules linked together) that are released into the bloodstream and transported to other tissues.
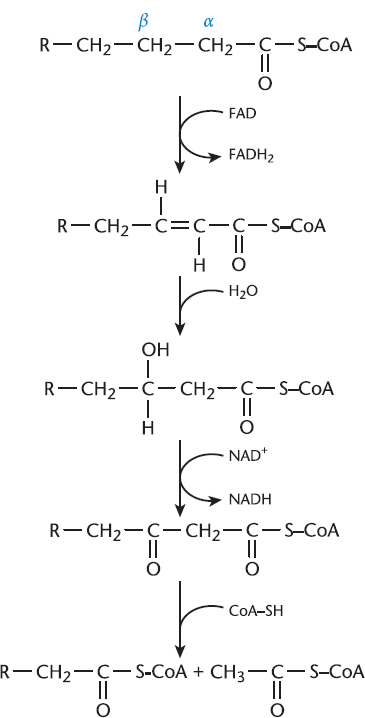
Figure 11.8 β-Oxidation
The four steps of β-oxidation, illustrated in Figure 11.8, are:
- Oxidation of the fatty acid to form a double bond
- Hydration of the double bond to form a hydroxyl group
- Oxidation of the hydroxyl group to form a carbonyl (β-ketoacid)
- Splitting of the β-ketoacid into a shorter acyl-CoA and acetyl-CoA
This process then continues until the chain has been shortened to two carbons, creating a final acetyl-CoA.
Fatty acids with an odd number of carbon atoms undergo β-oxidation in the same manner as even-numbered carbon fatty acids for the most part. The only difference is observed during the final cycle, where even-numbered fatty acids for the most part yield two acetyl-CoA molecules (from the four-carbon remaining fragment) and odd-numbered fatty acids yield one acetyl-CoA and one propionyl-CoA (from the five-carbon remaining fragment), as shown in Figure 11.9. Propionyl-CoA is converted to methylmalonyl-CoA by propionyl-CoA carboxylase, which requires biotin (vitamin B7). Methylmalonyl-CoA is then converted into succinyl-CoA by methylmalonyl-CoA mutase, which requires cobalamin (vitamin B12). Succinyl-CoA is a citric acid cycle intermediate and can also be converted to malate to enter the gluconeogenic pathway in the cytosol. Odd-carbon fatty acids thus represent an exception to the rule that fatty acids cannot be converted to glucose in humans.
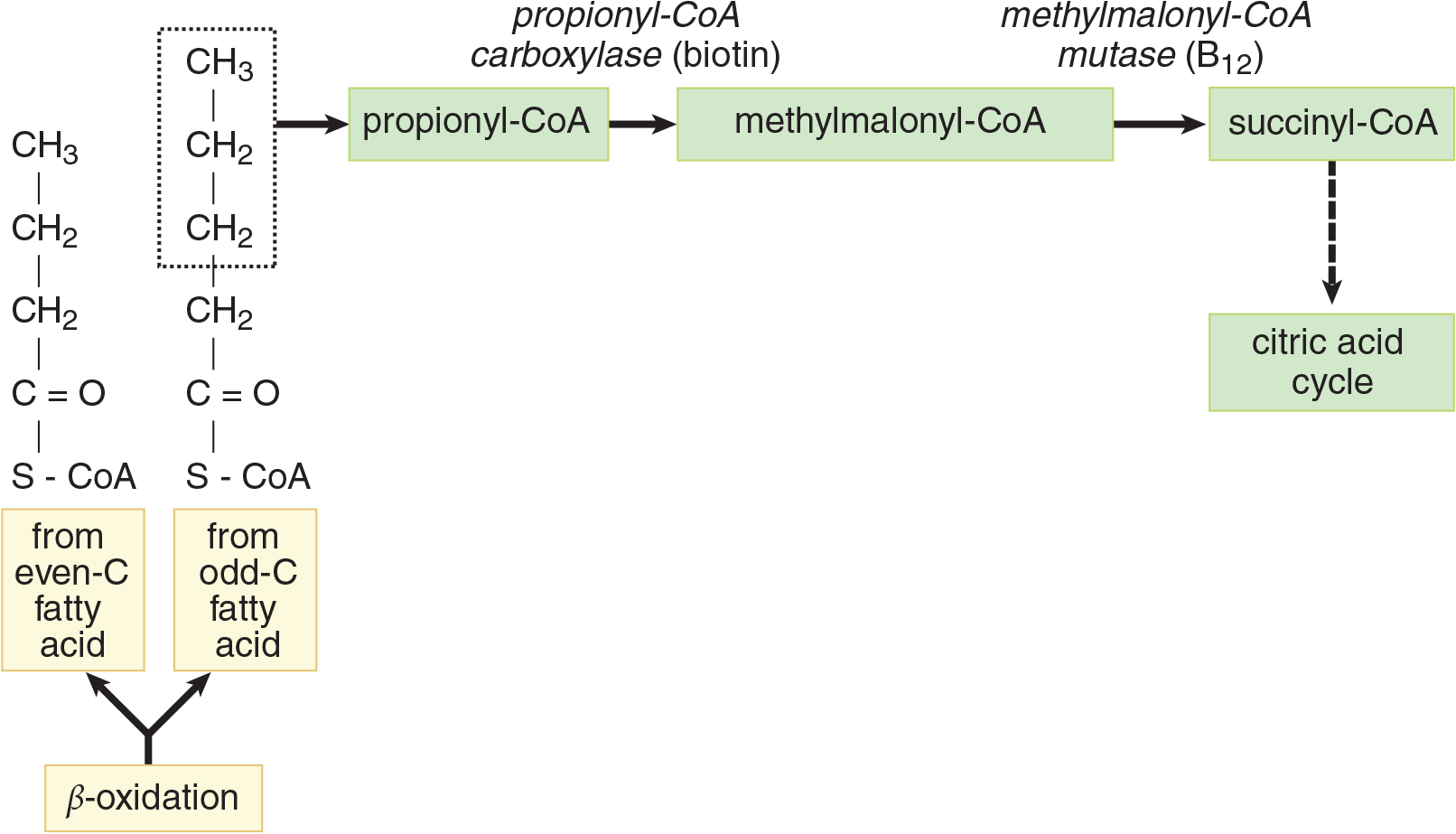
Figure 11.9 The Propionic Acid Pathway
Until now we’ve been discussing the oxidation of saturated fatty acids. In unsaturated fatty acids, two additional enzymes are necessary because double bonds can disturb the stereochemistry needed for oxidative enzymes to act on the fatty acid. To function, these enzymes can have at most one double bond in their active site; this bond must be located between carbons 2 and 3. Enoyl-CoA isomerase, shown in Figure 11.10, rearranges cis double bonds at the 3,4 position to trans double bonds at the 2,3 position once enough acetyl-CoA has been liberated to isolate the double bond within the first three carbons. In monounsaturated fatty acids this single step permits β-oxidation to proceed.
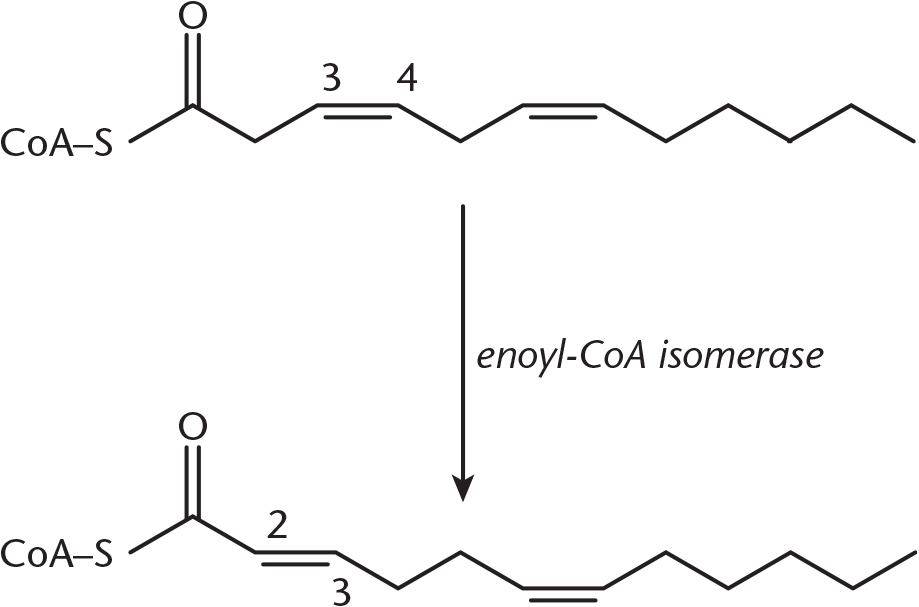
Figure 11.10 Reaction of Enoyl-CoA Isomerase
In polyunsaturated fatty acids, a further reduction is required using 2,4-dienoyl-CoA reductase to convert two conjugated double bonds to just one double bond at the 3,4 position, where it will then undergo the same rearrangement as monounsaturated fatty acids (as shown in Figure 11.11) to form a trans2,3 double bond.
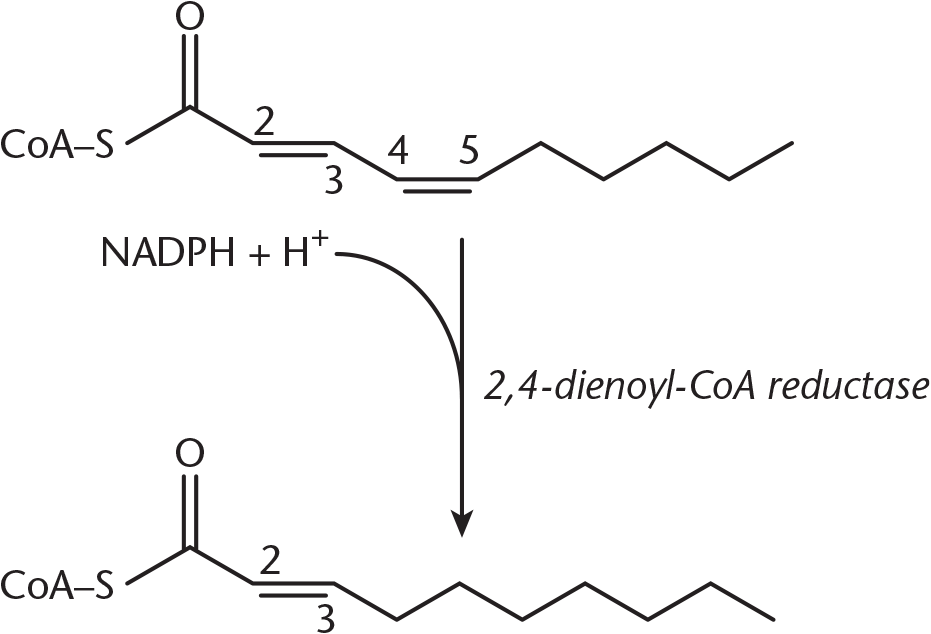
Figure 11.11 Reaction of 2,4-Dienoyl-CoA Reductase
BIOCHEMISTRY GUIDED EXAMPLE WITH EXPERT THINKING
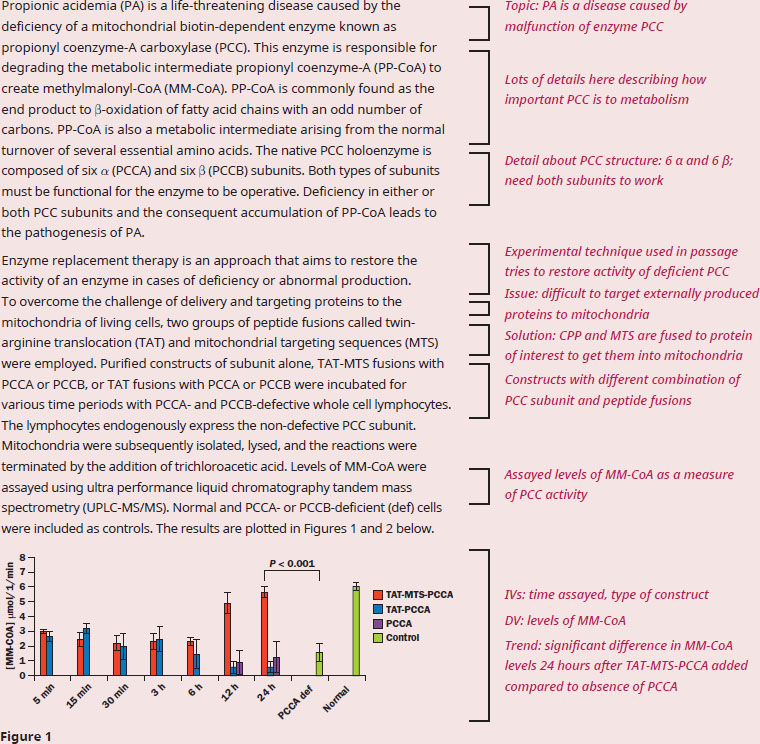
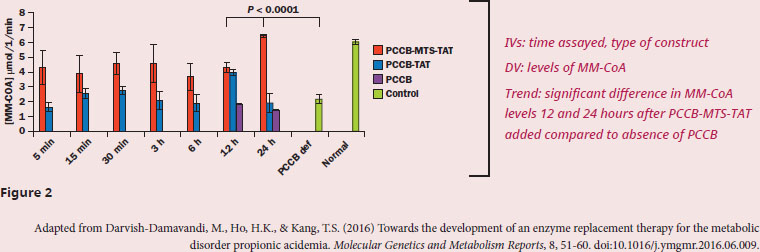
According to the data, are MTS-TAT-PCC constructs able to successfully rescue PCC activity in deficient mitochondria?
This question begins with "according to the data", meaning it will require us to understand and analyze the data presented in the context of the experiment described in the passage. Given we have multiple, complex figures, it’s important to get some context on the experiments and what the data represent first. The researchers are exploring treatment options for a disease caused by deficient PCC, which is an important metabolic enzyme in the mitochondria. We’re told that PCC requires both types of subunits in order to function properly. Common methods of restoring protein activity in cells are at the transcriptional/translational level within the cell, but the method employed in this study produces the required protein outside of the cell, then adds the protein back into the cell. We should know from our content background that proteins are generally polar, so the challenge is getting the protein past the lipid bilayers of both the cellular and the mitochondrial membranes. The researcher’s solution to this problem is to fuse PCC subunits to peptides that not only get PCC through the membranes, but also target the mitochondria. Also, note that the cell lines still express the non-deficient subunit: the cell lines in Figure 1 still express functional PCCB, so PCCA must be added and delivered to the mitochondria for there to be PCC activity; the opposite is true for the cell lines in Figure 2.
Diving into the data, the experimental layout and the results are similar for both PCCA and PCCB fusions. When the PCC-subunit is fused to TAT alone, or in the absence of fusion, the levels of MM-CoA are similar. However, when the PCC-subunit is fused to both MTS and TAT, we see significantly higher levels of MM-CoA. The first paragraph tells us that PCC will catalyze PP-CoA to produce MM-CoA, the product being measured in the experiment, so high levels of MM-CoA correlate to PCC activity.
Therefore, these results indicate that PCC activity can be rescued by exogenously expressing PCC-subunits and fusing them to both mitochondrial-targeting and cell-permeating proteins.
MCAT CONCEPT CHECK 11.5:
Before you move on, assess your understanding of the material with these questions.
- Draw the following fatty acids: palmitic acid, 18:3 (all-cis-9,12,15), an ω-6.
- What are the five steps in the addition of acetyl-CoA to a growing fatty acid chain?
- ___________________________
- ___________________________
- ___________________________
- ___________________________
- ___________________________
- How does β-oxidation of unsaturated fatty acids differ from that of saturated fatty acids?
______________________________________
- True or False: Fatty acids are synthesized in the cytoplasm and modified by enzymes in the smooth endoplasmic reticulum.
11.6 Ketone Bodies
LEARNING OBJECTIVES
After Chapter 11.6, you will be able to:
- Predict when fatty acids would be used to create ketone bodies rather than glucose
- Recall the conditions that favor ketogenesis and ketolysis
In the fasting state, the liver converts excess acetyl-CoA from β-oxidation of fatty acids into the **ketone bodies acetoacetate,3-hydroxybutyrate(β***-hydroxybutyrate), andacetone*, which can be used for energy in various tissues. Cardiac and skeletal muscle and the renal cortex can metabolize acetoacetate and 3-hydroxybutyrate to acetyl-CoA. During fasting periods, muscle will metabolize ketones as rapidly as the liver releases them, preventing accumulation in the bloodstream. After a week of fasting, ketones reach a concentration in the blood that is high enough for the brain to begin metabolizing them. The processes of ketogenesis and ketolysis are shown in Figure 11.12.
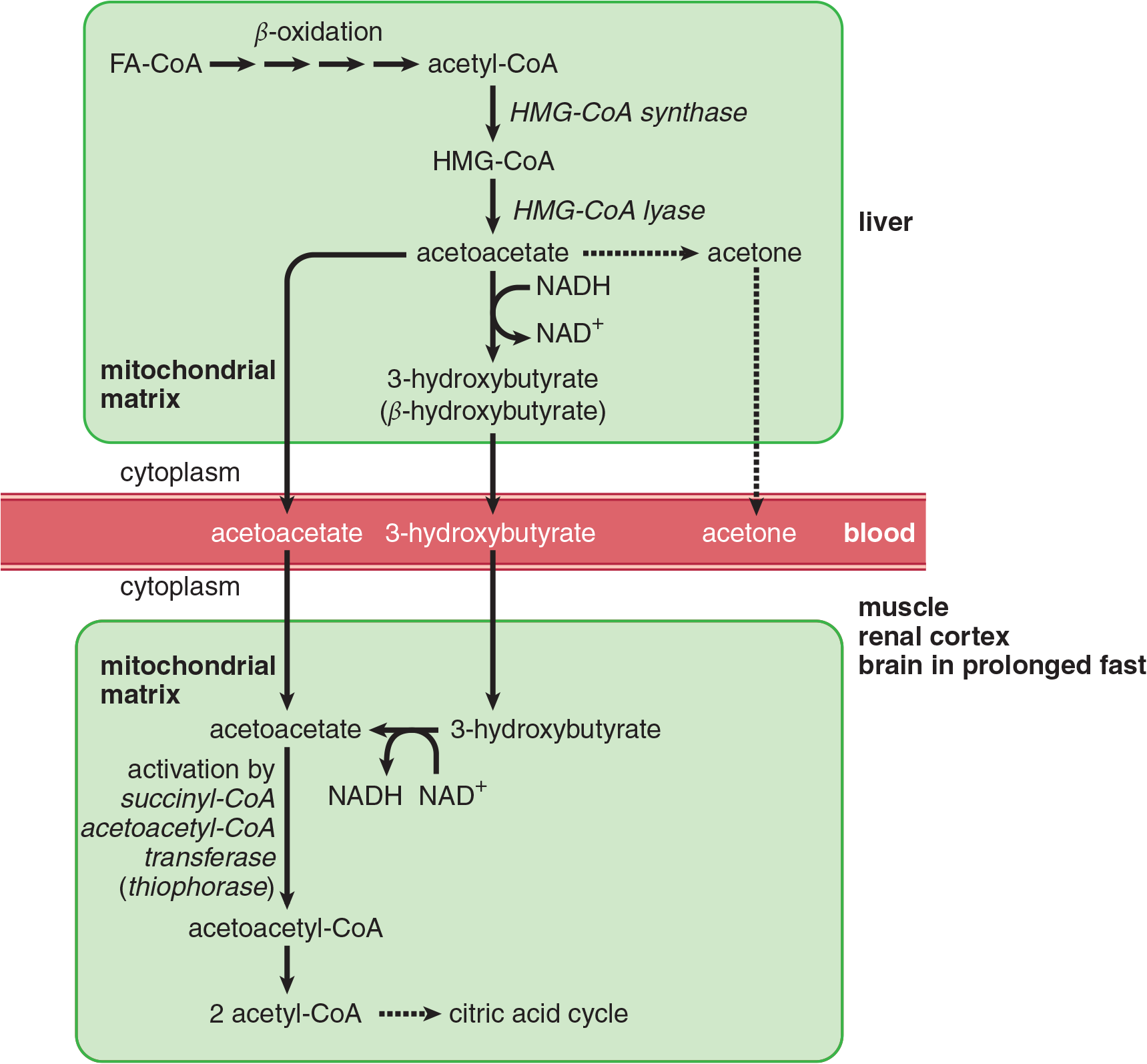
Figure 11.12 Ketogenesis and Ketolysis
KEY CONCEPT
Ketone bodies are essentially transportable forms of acetyl-CoA. They are produced by the liver and used by other tissues during prolonged starvation.
Ketogenesis
Ketogenesis occurs in the mitochondria of liver cells when excess acetyl-CoA accumulates in the fasting state. HMG-CoA synthase forms HMG-CoA, and HMG-CoA lyase breaks down HMG-CoA into acetoacetate, which can subsequently be reduced to 3-hydroxybutyrate. Acetone is a minor side product that cannot be directly turned into acetyl-CoA, but can be converted into lactate and pyruvate. Figure 11.13 shows the creation of acetoacetate, β-hydroxybutyrate (3-hydroxybutyrate), and acetone from acetyl-CoA.
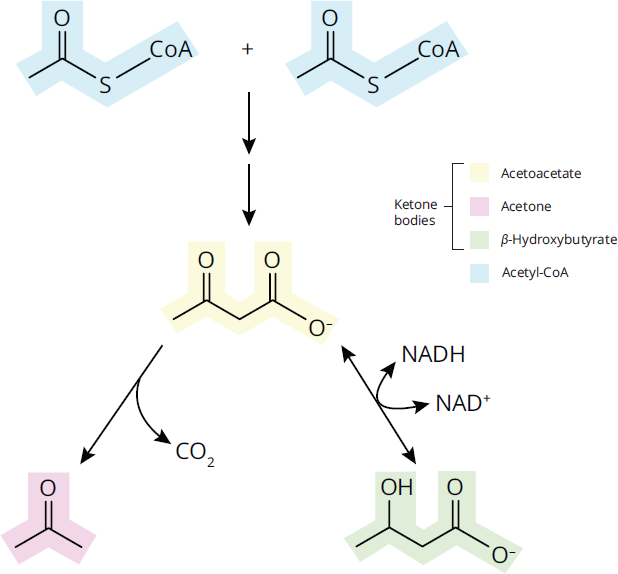
Figure 11.13 Ketone Bodies
Ketolysis
Acetoacetate picked up from the blood is activated in the mitochondria by succinyl-CoA acetoacetyl-CoA transferase (commonly called thiophorase), an enzyme present only in tissues outside the liver. During this reaction, acetoacetate is oxidized to acetoacetyl-CoA. The liver lacks this enzyme, so it cannot catabolize the ketone bodies that it produces.
REAL WORLD
A significant increase in ketone levels in the blood can lead to ketoacidosis, a potentially dangerous medical condition. This occurs most often with fatty acid breakdown in type 1 (insulin-dependent) diabetes mellitus.
Ketolysis in the Brain
During a prolonged fast (longer than one week), the brain begins to derive up to two-thirds of its energy from ketone bodies. In the brain, when ketones are metabolized to acetyl-CoA, pyruvate dehydrogenase is inhibited. Glycolysis and glucose uptake in the brain decreases. This important switch spares essential protein in the body, which otherwise would be catabolized to form glucose by gluconeogenesis in the liver, and allows the brain to indirectly metabolize fatty acids as ketone bodies.
MCAT CONCEPT CHECK 11.6:
Before you move on, assess your understanding of the material with these questions.
- Why are fatty acids used to create ketone bodies instead of creating glucose?
______________________________________
- What conditions and tissues favor ketogenesis? Ketolysis?
__________________________________
- Ketogenesis:
___________________________________
- Ketolysis:
11.7 Protein Catabolism
LEARNING OBJECTIVES
After Chapter 11.7, you will be able to:
- Identify common sources for acetyl-CoA used in lipid synthesis
- Recall the location(s) associated with large amounts of protein digestion
- Predict what will happen to the carbon skeleton, amino groups, and side chains of proteins that have been broken down
Protein is very rarely used as an energy source because it is so important for other functions; routinely breaking down protein would result in serious illness. However, under conditions of extreme energy deprivation, proteins can be used for energy. In order to provide a reservoir of amino acids for protein building by the cell, proteins must be digested and absorbed.
KEY CONCEPT
Metabolism is directed toward conserving tissues to the greatest extent possible, especially the brain and heart. Digestion of protein compromises muscle—potentially that of the heart—so it is unlikely to occur under normal conditions.
Proteolysis (the breakdown of proteins) begins in the stomach with pepsin and continues with the pancreatic proteases trypsin, chymotrypsin, and carboxypeptidases A and B, all of which are secreted as zymogens. Protein digestion is completed by the small intestinal brush-border enzymes dipeptidase and aminopeptidase. The main end products of protein digestion are amino acids, dipeptides, and tripeptides. Absorption of amino acids and small peptides through the luminal membrane is accomplished by secondary active transport linked to sodium. At the basal membrane, simple and facilitated diffusion transports amino acids into the bloodstream. Figure 11.14 illustrates the major transport mechanisms involved in moving amino acids across the luminal and basal membranes of intestinal cells.
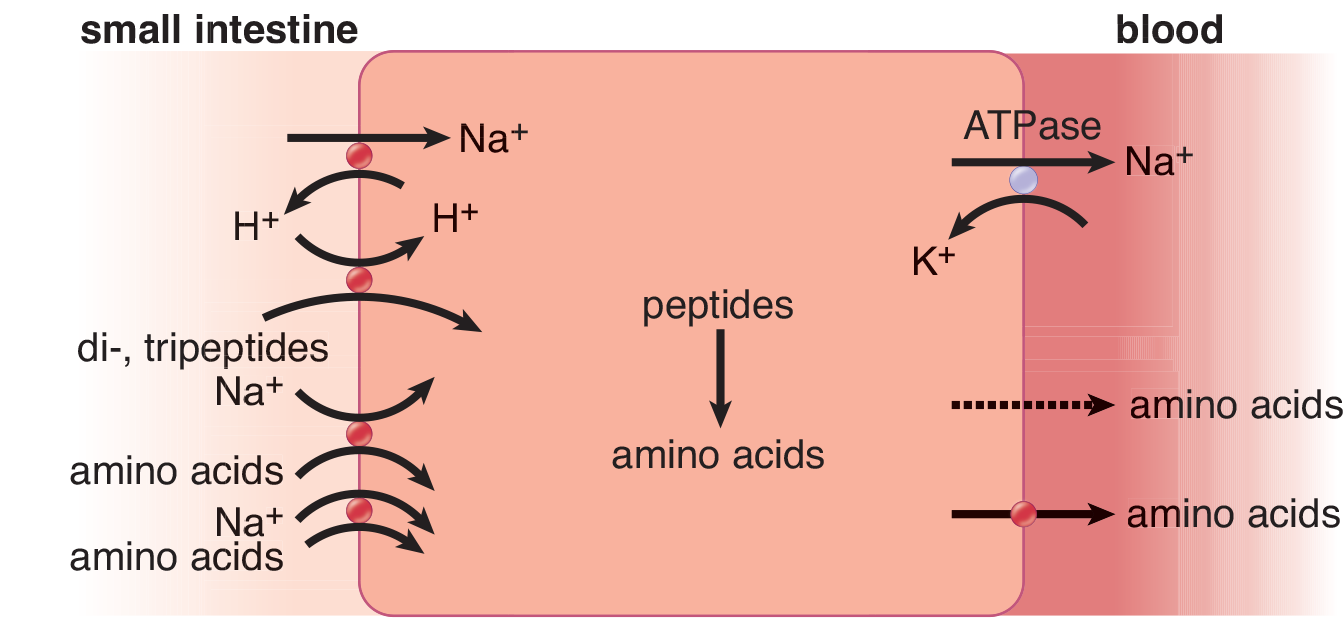
Figure 11.14 Absorption of Amino Acids and Peptides in the Intestine
Protein obtained from the diet or from the body (during prolonged fasting or starvation) may be used as an energy source. Body protein is catabolized primarily in muscle and liver. Amino acids released from proteins usually lose their amino group through transamination or deamination. The remaining carbon skeleton can be used for energy. Amino acids are classified by their ability to turn into specific metabolic intermediates: glucogenic amino acids (all but leucine and lysine) can be converted into glucose through gluconeogenesis; ketogenic amino acids (leucine and lysine, as well as isoleucine, phenylalanine, threonine, tryptophan, and tyrosine, which are also glucogenic as well) can be converted into acetyl-CoA and ketone bodies.
The amino groups removed by transamination or deamination constitute a potential toxin to the body in the form of ammonia, and must be excreted safely. The urea cycle, shown in Figure 11.15, occurs in the liver and is the body’s primary way of removing excess nitrogen from the body. The MCAT is highly unlikely to test on the steps and intermediates of the urea cycle directly, but it is provided here as a point of reference.
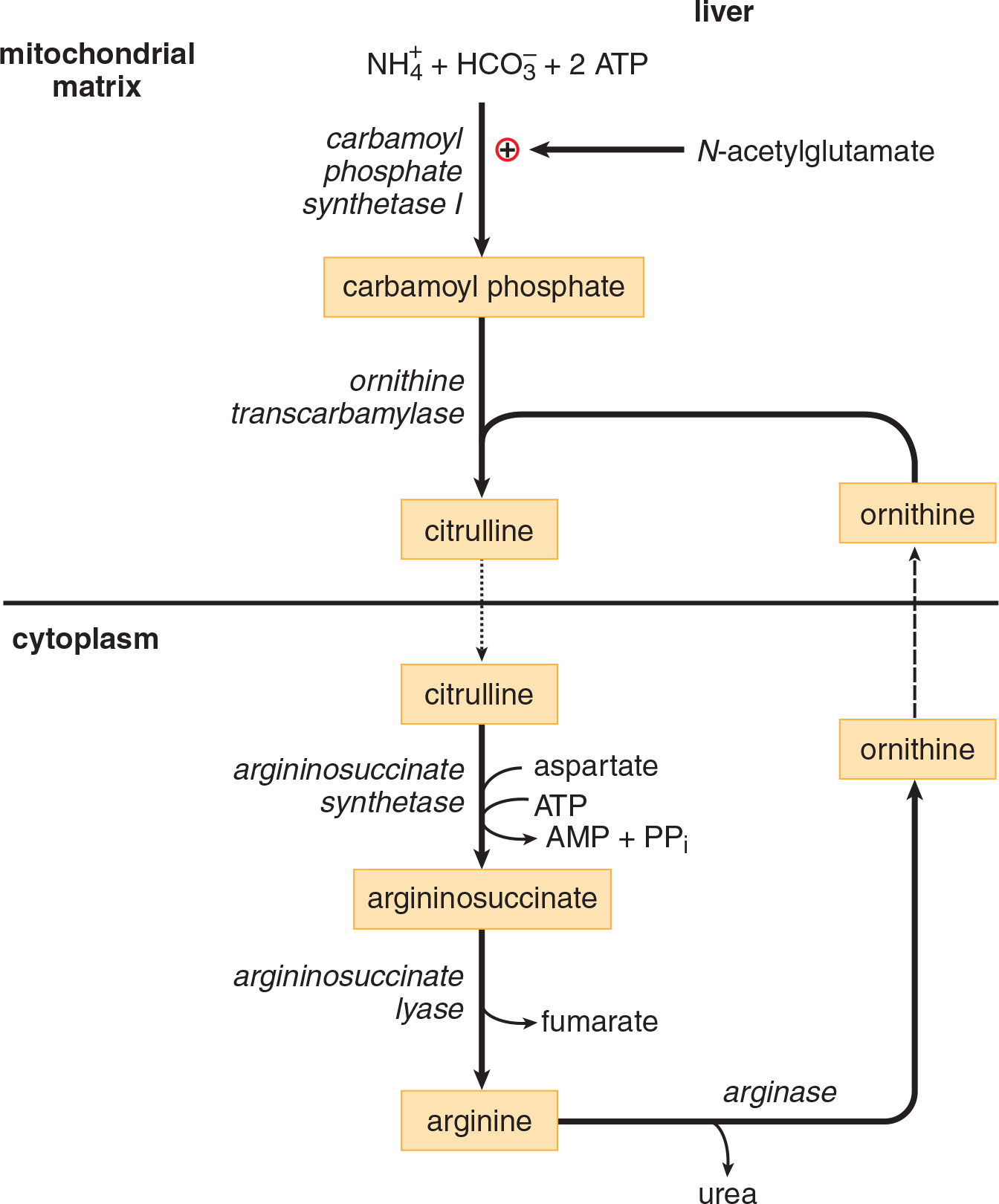
Figure 11.15 The Urea Cycle
The fate of the side chain from each amino acid depends on its chemistry. Basic amino acid side chains feed into the urea cycle, while the other side chains act like the carbon skeleton and produce energy through gluconeogenesis or ketone production.
MCAT CONCEPT CHECK 11.7:
Before you move on, assess your understanding of the material with these questions.
- True or False: Bodily proteins will commonly be broken down to provide acetyl-CoA for lipid synthesis.
- Where does the bulk of protein digestion occur?
____________________________________
- During protein processing, what is the eventual fate of each of the following components: carbon skeleton, amino group, and side chains?
_________________________________
- Carbon skeleton:
_________________________________
- Amino group:
__________________________________
- Side chains:
Conclusion
At this point, we have examined all of the vital metabolic processes of the cell. In this chapter, we reviewed dietary lipids and different ways that lipids are metabolized in the cell. We also covered lipid transport in blood and lymphatic fluid and the mobilization of lipids from adipocytes. In addition, we went over the structure, synthesis, and breakdown of fatty acids required to address the energy needs of the cell. The importance of ketone bodies and how they are utilized by the cell during periods of starvation were also reviewed. Finally, we went over digestion and metabolism of proteins and amino acids.
Metabolism of the different macromolecules does not occur in isolation, as you’ve already seen: the acetyl-CoA produced in fatty acid oxidation regulates the pyruvate dehydrogenase complex and pyruvate carboxylase to create a shift in carbohydrate metabolism from glycolysis and the citric acid cycle to gluconeogenesis. In the next chapter, we’ll dive into how the different pathways fit together and will integrate the metabolic knowledge that you’ve compiled in Chapters 9, 10, and 11 of MCAT Biochemistry Review.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Lipid Digestion and Absorption
- Mechanical digestion of lipids occurs primarily in the mouth and stomach.
- Chemical digestion of lipids occurs in the small intestine and is facilitated by bile, pancreatic lipase, colipase, and cholesterol esterase.
- Digested lipids may form micelles for absorption or be absorbed directly.
- Short-chain fatty acids are absorbed across the intestine into the blood.
- Long-chain fatty acids are absorbed as micelles and assembled into chylomicrons for release into the lymphatic system.
Lipid Mobilization
- Lipids are mobilized from adipocytes by hormone-sensitive lipase.
- Lipids are mobilized from lipoproteins by lipoprotein lipase.
Lipid Transport
- Chylomicrons are the transport mechanism for dietary triacylglycerol molecules and are transported via the lymphatic system.
- VLDL transports newly synthesized triacylglycerol molecules from the liver to peripheral tissues in the bloodstream.
- IDL is a VLDL remnant in transition between triacylglycerol and cholesterol transport; it picks up cholesteryl esters from HDL.
- LDL primarily transports cholesterol for use by tissues.
- HDL is involved in the reverse transport of cholesterol.
- Apoproteins control interactions between lipoproteins.
Cholesterol Metabolism
- Cholesterol may be obtained through dietary sources or through de novo synthesis in the liver.
- The key enzyme in cholesterol biosynthesis is HMG-CoA reductase.
- LCAT catalyzes the formation of cholesteryl esters for transport with HDL.
- CETP catalyzes the transition of IDL to LDL by transferring cholesteryl esters from HDL.
Fatty Acids and Triacylglycerols
- Fatty acids are carboxylic acids, typically with a single long chain, although they can be branched.
- Saturated fatty acids have no double bonds between carbons. Unsaturated fatty acids have one or more double bonds.
- Fatty acids are synthesized in the cytoplasm from acetyl-CoA transported out of the mitochondria.
- Synthesis includes five steps: activation, bond formation, reduction, dehydration, and a second reduction.
- These steps are repeated eight times to form palmitic acid, the only fatty acid that humans can synthesize.
- Fatty acid oxidation occurs in the mitochondria following transport by the carnitine shuttle.
- β-Oxidation uses cycles of oxidation, hydration, oxidation, and cleavage.
- Branched and unsaturated fatty acids require special enzymes.
- Unsaturated fatty acids use an isomerase and an additional reductase during cleavage.
Ketone Bodies
- Ketone bodies form (ketogenesis) during a prolonged starvation state due to excess acetyl-CoA in the liver.
- Ketolysis regenerates acetyl-CoA for use as an energy source in peripheral tissues.
- The brain can derive up to two-thirds of its energy from ketone bodies during prolonged starvation.
Protein Catabolism
- Protein digestion occurs primarily in the small intestine.
- Catabolism of cellular proteins occurs only under conditions of starvation.
- Carbon skeletons of amino acids are used for energy, either through gluconeogenesis or ketone body formation. Amino groups are fed into the urea cycle for excretion. The fate of a side chain depends on its chemistry.
ANSWERS TO CONCEPT CHECKS
**11.1**
- Physical digestion is accomplished in the mouth and the stomach, reducing the particle size. Beginning in the small intestine, pancreatic lipase, colipase, cholesterol esterase, and bile assist in the chemical digestion of lipids. In the more distal portion of the small intestine, absorption occurs.
- False. Small free fatty acids enter the circulation directly.
- Micelles are collections of lipids with their hydrophobic ends oriented toward the center and their charged ends oriented toward the aqueous environment. Micelles collect lipids within their hydrophobic centers.
**11.2**
- An increase in insulin levels will increase lipid storage and decrease lipid mobilization from adipocytes, leading to weight gain in patients who have diabetes and begin insulin injections.
- The ratio of free fatty acids to glycerol is 3:1. A triacylglycerol molecule is composed of glycerol and three fatty acids.
**11.3**
- Free fatty acids remain in the blood, bonded to albumin and other carrier proteins. A much smaller amount will remain unbonded.
- With respect to protein content, HDL > LDL > IDL > VLDL > chylomicrons. VLDL and chylomicrons are the primary triacylglycerol transporters. HDL and LDL are mostly involved in cholesterol transport.
- Lipoproteins are synthesized primarily by the intestine and liver.
- As mentioned in the chapter, HDL is often considered “good” cholesterol because it picks up excess cholesterol from blood vessels for excretion. Because of this crucial role, HDL values are checked for being over a minimum value.
**11.4**
- HMG-CoA reductase is most active in the absence of cholesterol and when stimulated by insulin. Cholesterol reduces the activity of HMG-CoA reductase, which is located in the smooth endoplasmic reticulum.
- LCAT catalyzes the esterification of cholesterol to form cholesteryl esters. CETP promotes the transfer of cholesteryl esters from HDL to IDL, forming LDL.
**11.5**
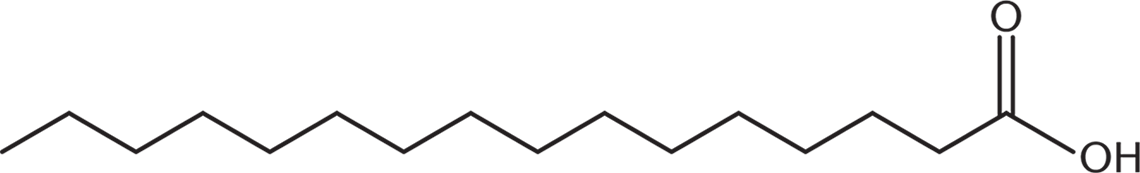
- Palmitic acid (16:0):
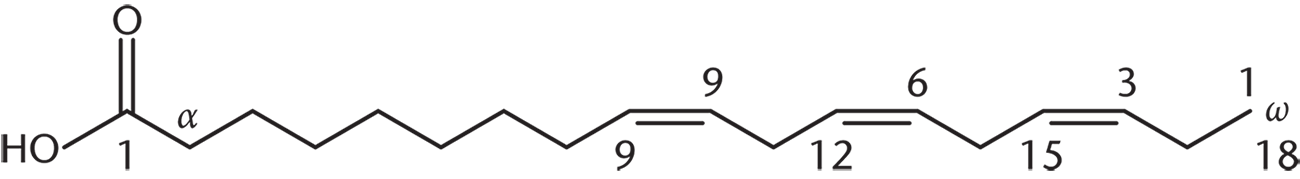
α-Linolenic acid (18:3 all-cis-9,12,15), an ω-3 fatty acid:
Linoleic acid (18:2 cis,cis-9,12), an ω-6 fatty acid:
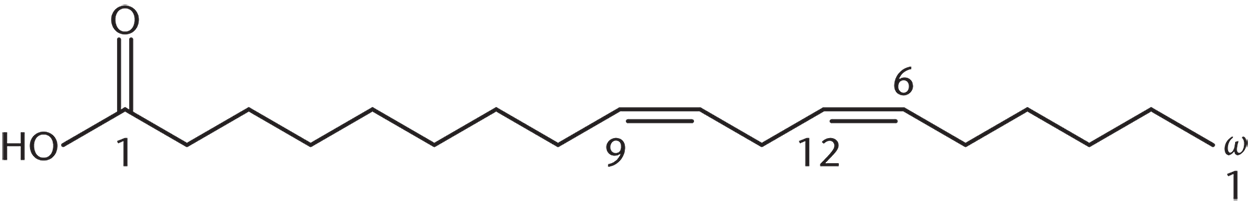
Note: As long as the last double bond is in the same position relative to the end of the chain, many answers are possible for the ω-6 fatty acid.
- The steps in the attachment of acetyl-CoA to a fatty acid chain are attachment to acyl carrier protein, bond formation between molecules, reduction of a carbonyl group, dehydration, and reduction of a double bond. These steps are shown in Figure 11.6.
- There is an additional isomerase and an additional reductase for the β-oxidation of unsaturated fatty acids, which provide the stereochemistry necessary for further oxidation.
- True.
**11.6**
- Fatty acid degradation results in large amounts of acetyl-CoA, which cannot enter the gluconeogenic pathway to produce glucose. Only odd-numbered fatty acids can act as a source of carbon for gluconeogenesis; even then, only the final malonyl-CoA molecule can be used. Energy is packaged into ketone bodies for consumption by the brain and muscles.
- Ketogenesis is favored by a prolonged fast and occurs in the liver. It is stimulated by increasing concentrations of acetyl-CoA. Ketolysis is also favored during a prolonged fast, but is stimulated by a low-energy state in muscle and brain tissues and does not occur in the liver.
**11.7**
- False. Proteins are more valuable to the cell than lipids, thus they will not commonly be broken down for lipid synthesis.
- The bulk of protein digestion occurs in the small intestine.
- The carbon skeleton is transported to the liver for processing into glucose or ketone bodies. The amino group will feed into the urea cycle for excretion. Side chains are processed depending on their composition. Basic side chains will be processed like amino groups, while other functional groups will be treated like the carbon skeleton.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. B
This question asks for the hormone that does not directly regulate the activity of hormone-sensitive lipase (HSL). Hormone sensitive lipase is an enzyme that cleaves fatty acids from their glycerol backbones, releasing these free fatty acids for use in energy production. HSL is directly activated by epinephrine and cortisol, so (C) and (D) can be eliminated. Regarding insulin vs. glucagon, observe that free fatty acid mobilization, and therefore HSL activity, is generally associated with low blood sugar, which is also associated with elevated glucagon levels. However, glucagon does not directly regulate the activity of HSL. Rather, HSL is activated by a drop in insulin levels. For this reason, insulin does directly regulate HSL activity, and therefore (A) can be eliminated. Only glucagon, (B), does not directly influence HSL activity. Therefore (B) is the correct answer.
2. A
Short-chain fatty acids are soluble in the intestinal lumen, and thus do not interact with micelles as longer fatty acid chains do. The long-chain fatty acids are taken up by the intestinal cells and packaged into triacylglycerols for transport as chylomicrons. Chylomicrons exit the intestine through lacteals that feed into the lymphatic system, which joins with the bloodstream in the base of the neck through the thoracic duct.
3. C
During fatty acid mobilization, there is a breakdown of triacylglycerols in adipocytes by hormone-sensitive lipase (HSL). This breakdown results in the release of three fatty acids and a glycerol molecule. The glycerol may be used by the liver for gluconeogenesis, but adipocytes do not have the ability to carry out gluconeogenesis.
4. B
Chylomicrons and VLDL are very similar. Both contain apolipoproteins and primarily transport triacylglycerols, eliminating (A) and (C). The only major difference between them is the tissue of origin. Chylomicrons transport dietary triacylglycerol and originate in the small intestine, while VLDL transport newly synthesized triacylglycerols and originate in the liver.
5. D
While the transport and lipid binding functions of most lipoproteins are independent of the apolipoprotein component, the interaction of these lipoproteins with the environment is controlled almost exclusively by apolipoproteins. Lipoproteins cannot exit or enter cells without apolipoproteins, and are unable to transfer lipids without specialized apolipoproteins or cholesterol-specific enzymes.
6. A
Statins are drugs that are prescribed to treat high cholesterol and act as competitive inhibitors of HMG-CoA reductase. HMG-CoA reductase is the rate-limiting enzyme of de novo cholesterol synthesis; inhibition of this enzyme lowers production of cholesterol, thus lowering overall levels of cholesterol.
7. A
LCAT adds a fatty acid to cholesterol, producing cholesteryl esters, which dissolve in the core of HDL, allowing HDL to transport cholesterol from the periphery to the liver.
8. B
Humans can only synthesize one fatty acid, palmitic acid. Palmitic acid is fully saturated and therefore does not contain any double bonds. Palmitic acid has 16 carbons, and is synthesized from eight molecules of acetyl-CoA. In shorthand notation, palmitic acid is written as 16:0 (16 carbons, no double bonds).
9. C
The steps in fatty acid synthesis are activation (attachment to acyl carrier protein), bond formation (between malonyl-CoA and the growing fatty acid chain), reduction (of a carboxyl group), dehydration, and reduction (of a double bond).
10. C
Ketone bodies are produced in the mitochondria of liver cells as acetyl-CoA accumulates during fasting, consistent with (C). On the other hand, to eliminate (B), observe that while acetyl-CoA is indeed converted to acetoacetate, 3-hydroxybutyrate, or acetone, the acetone that is produced is only a byproduct and is not used for energy, which conflicts with the assertion in (B) that "all of which are used for energy." Muscle cells will begin using ketone bodies for energy before the brain. The brain will only start using ketone bodies once they have accumulated to higher concentrations in the blood, eliminating choice (D).
11. D
The liver is the major metabolic organ in the body and is responsible for much of the synthesis and interconversion of fuel sources. Most of the triacylglycerols that are synthesized in the liver are transported as VLDL to adipose tissue for storage. Both the adipocytes, (A), and dietary intake, (B), constitute a minor source of triacylglycerols.
12. C
In order for the enzymes of fatty acid oxidation to operate, there can be, at most, one double bond in the area of enzyme activity, and it must be oriented between carbons 2 and 3. In order to accomplish this in monounsaturated fatty acids, an isomerase is employed. When there are multiple double bonds that fall within the enzymatic binding site, both an isomerase and 2,4-dienoyl-CoA reductase are required for the oxidative enzymes to act on the fatty acid. For this question, simply recognizing that dienoyl refers to having multiple double bonds is sufficient to arrive at the answer.
13. C
Ketolysis is the breakdown of ketone bodies to acetyl-CoA for energy. This process occurs in the brain and muscle tissues, but cannot occur in the liver, which lacks an enzyme necessary for ketone body breakdown. Ketolysis is not associated with an increase in glucose metabolism because it most often occurs under conditions of starvation.
14. D
The energy contribution of an amino acid depends on its ability to be turned into glucose through gluconeogenesis (glucogenic amino acids), ketone bodies (ketogenic amino acids), or both. All of the amino acids listed in the answer choices are glucogenic; isoleucine is also ketogenic. The energy acquired from an amino acid will also depend on the number of carbons it can donate to these energy-creating processes, which depends on the size of its side chain. Isoleucine has the largest side chain of the answer choices, and will thus contribute the most energy per molecule.
15. A
The degradation of protein and processing by the liver implies a prolonged starvation state; protein will not be used for energy unless absolutely necessary. Thus, gluconeogenesis is the most likely process. When gluconeogenesis is not possible, easily metabolized molecules, such as ketone bodies, are synthesized. Fatty acid production occurs when energy is being stored; proteins would not be broken down to store energy in fatty acids.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 1
- Amino Acids, Peptides, and Proteins
- Biochemistry Chapter 5
- Lipid Structure and Function
- Biochemistry Chapter 8
- Biological Membranes
- Biochemistry Chapter 12
- Bioenergetics and Regulation of Metabolism
- Biology Chapter 9
- The Digestive System
- Biology Chapter 10
- Homeostasis