Chapter 8: The Immune System

Chapter 8: The Immune System
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- In DiGeorge syndrome, the thymus can be completely absent. The absence of the thymus would leave an individual unable to mount specific defenses against which of the following types of pathogens?
- Viruses
- Bacteria
- Parasites
- Fungi
- Which of the following are NOT involved in cell-mediated immunity?
- Memory cells
- Plasma cells
- Cytotoxic cells
- Suppressor cells
- The lymphatic system:
- transports hormones throughout the body.
- transports chylomicrons to the circulatory system.
- causes extravasation of fluid into tissues.
- is the site of mast cell activation.
- Which of the following are involved in antibody production?
- Plasma cells
- Memory cells
- Helper T-cells
- Cytotoxic cells
- Which of the following is NOT true of the innate immune system?
- Includes macrophages to mediate inflammation
- Is always active against infection
- Recognizes unique features of a pathogen
- Contains cells derived from a hematopoietic stem cell
- Which of the following is an example of adaptive immunity?
- PRRs recognize that a pathogen is an invasive parasite and eosinophils are recruited to the area.
- Complement is activated, causing osmotic instability in a bacterium.
- Memory B-cells generated through vaccination are activated when their antigen is encountered.
- Dendritic cells sample bacteria within a laceration and travel to the lymph nodes to present the antigen.
- Which of the following is true regarding passive and active immunity?
- Active immunity requires weeks to build, whereas passive immunity is acquired immediately.
- Active immunity is short lived, whereas passive immunity is long lived.
- Active immunity may be acquired during pregnancy through the placenta.
- Passive immunity may be acquired through vaccination.
- Where are most self-reactive T-cells eliminated?
- Spleen
- Lymph nodes
- Bone marrow
- Thymus
- What is the response of the immune system to downregulation of MHC molecules on somatic cells?
- B-cells are activated and antibodies are released.
- T-cells are activated, resulting in a cytotoxic response.
- Natural killer cells induce apoptosis of affected cells.
- Macrophages engulf the pathogen and display its antigens.
- Which of the following correctly indicates the response of CD8+ T-cells when activated?
- Secretion of cytotoxic chemicals
- Causing isotype switching
- Presentation of antigens
- Activation of B-cells
- Lymphoma is cancer of the cells of lymphoid lineage. These cells often reside within lymph nodes. What type of cell is NOT likely to cause a lymphoma?
- CD8+ T-cells
- B-cells
- Macrophages
- Th1 cells
- Upon encountering an antigen, only T-cells with a specific T-cell receptor are activated. This is an example of:
- innate immunity.
- a cytotoxic T-cell response.
- humoral immunity.
- clonal selection.
- Which cell type is a phagocyte that attacks bacterial pathogens in the bloodstream?
- Neutrophils
- Eosinophils
- Basophils
- Dendritic cells
- What type of immunity is likely to be affected by removal of the spleen?
- Cytotoxic immunity
- Humoral immunity
- Innate immunity
- Passive immunity
- Which of the following is NOT an example of a nonspecific defense mechanism?
- Skin provides a physical barrier against invasion.
- Macrophages engulf and destroy foreign particles.
- An inflammatory response is initiated in response to physical damage.
- Cytotoxic T-cells destroy cells displaying foreign antigens.
Answer Key
- A
- B
- B
- A
- C
- C
- A
- D
- C
- A
- C
- D
- A
- B
- D
Chapter 8: The Immune System
CHAPTER 8
THE IMMUNE SYSTEM
In This Chapter
8.1 Structure of the Immune System
Innate and Adaptive Immunity
Anatomy
8.2 The Innate Immune System
Noncellular Nonspecific Defenses
Cells of the Innate Immune System
8.3 The Adaptive Immune System
Cells of the Adaptive Immune System
Activation of the Adaptive Immune System
Recognition of Self and Nonself
Immunization
8.4 The Lymphatic System
Structure
Function
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 6% of all questions about biology on the MCAT.
This chapter covers material from the following AAMC content category:
3B: Structure and integrative functions of the main organ systems
Introduction
Over the past few decades, the public’s imagination has been captured by alarming reports of “flesh-eating” bacteria and diseases. While these bacteria do not actually eat flesh, that doesn’t make them any less dangerous. “Flesh-eating” bacteria cause a condition called necrotizing fasciitis, a serious disease that requires aggressive medical and surgical treatment including intravenous antibiotics, surgical debridement (removal) of the necrotic tissue, and sometimes even amputation. Many different types of bacteria, including group A Streptococcus, Clostridium perfringens, and methicillin-resistant Staphylococcus aureus (MRSA), can cause necrotizing fasciitis. The massive destruction of skin, muscle, and connective tissue by the release of bacterial toxins called superantigens is life-threatening. These superantigens cause the immune system to become nonspecifically overactivated. Necrotizing fasciitis is dangerous not only because of the bacteria and subsequent inflammatory response, but also because the destruction of skin—a nonspecific immune defense—leaves the body susceptible to superinfection (infection with another pathogen).
In this chapter, we will consider this type of nonspecific defense, as well as specific immune defenses. The human body relies on the interaction between the innate (nonspecific) and adaptive (specific) immune systems in order to protect itself from disease. However, the immune system is not always perfect in its responses. Sometimes the immune system may become nonspecifically overactivated, as is the case with necrotizing fasciitis, or it may become activated against the same human it is supposed to protect. We will discuss the individual parts of the immune system and how these parts work together to protect from disease. In addition, we will discuss the concept of autoimmunity, or disease that results from immune attack against oneself. We’ll briefly touch on vaccines, which take advantage of our understanding of the immune system to protect us from life-threatening infections. The immune system is largely integrated with the lymphatic system, which will be discussed in this chapter as well.
8.1 Structure of the Immune System
LEARNING OBJECTIVES
After Chapter 8.1, you will be able to:
- Distinguish between innate and adaptive immunity
- Draw comparisons between B- and T-cells, including their development, maturation, functions, specificity, and control mechanisms
- Identify immune cells as granulocytes or agranulocytes
Each day, the human body is exposed to numerous bacteria, viruses, fungi, and even parasites. Yet our bodies are able to protect us from infection most of the time. Even when we do get sick, the immune system is usually able to contain and eliminate the infection.
Innate and Adaptive Immunity
In order to fight infection, the human body has two different divisions of the immune system: innate and adaptive immunity, compared in Figure 8.1. Innate immunity is composed of defenses that are always active against infection, but lack the ability to target specific invaders; for this reason, it is also called nonspecific immunity. Adaptive or specific immunity refers to the defenses that target a specific pathogen. This system is slower to act, but can maintain immunological memory of an infection to mount a faster attack in subsequent infections.
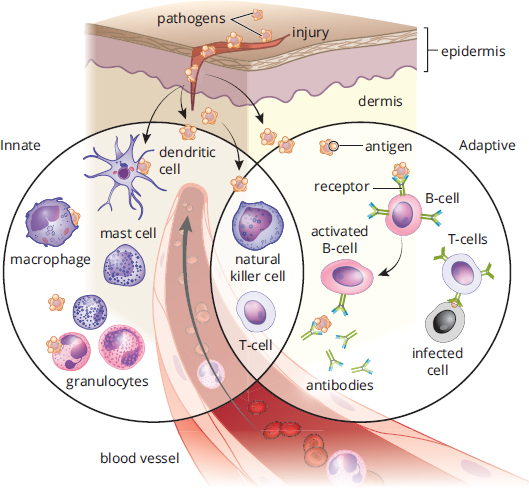
Figure 8.1. Divisions of the Immune System
MCAT EXPERTISE
The immune system is a topic where cell biology, biochemistry, anatomy, and biology interact. Thus, it is a topic that allows the MCAT to ask questions that integrate these four topics. As we discuss these complex ideas, focus on the big picture first, creating a mental image to help you put these concepts together. Then go back and plug in the details. This approach will foster your ability to associate structure with function and to think critically about the topic on Test Day.
Anatomy
The immune system is not housed in a single organ, as shown in Figure 8.2. The structure and components that serve as nonspecific defenses often serve functions in other organ systems. The bone marrow produces all of the leukocytes (white blood cells) that participate in the immune system through the process of hematopoiesis, discussed in Chapter 7 of MCAT Biology Review. The spleen is a location of blood storage and activation of B-cells, which turn into plasma cells to produce antibodies as part of adaptive immunity. Note that when B-cells leave the bone marrow, they are considered mature but naïve (because they have not yet been exposed to an antigen). Because these antibodies dissolve and act in the blood (rather than within cells), this division of adaptive immunity is called humoral immunity. T-cells, another class of adaptive immune cells, mature in the thymus, a small gland just in front of the pericardium, the sac that protects the heart. T-cells are the agents of cell-mediated immunity because they coordinate the immune system and directly kill virally infected cells. Finally, lymph nodes, a major component of the lymphatic system, provide a place for immune cells to communicate and mount an attack; B-cells can be activated here as well. Other immune tissue is found in close proximity to the digestive system, which is a site of potential invasion by pathogens. These tissues are commonly called gut-associated lymphoid tissue (GALT) and include the tonsils and adenoids in the head, Peyer’s patches in the small intestine, and lymphoid aggregates in the appendix.
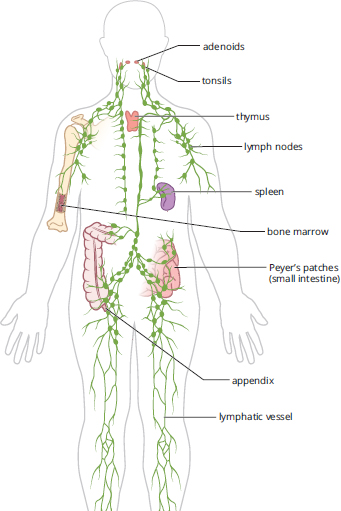
Figure 8.2. Anatomy of the Immune and Lymphatic Systems
REAL WORLD
Lay people not involved in medicine often say that their “glands” are swollen when they feel a sensation of fullness around the throat. From a biological standpoint, lymph nodes are not glands—they do not secrete products into the bloodstream (endocrine) or ducts (exocrine). What they are actually experiencing is known medically as lymphadenopathy, or the swelling of the lymph nodes that occurs with activation of the immune system.
KEY CONCEPT
Organs of the immune system:
- Lymph nodes filter lymph and are a site where immune responses can be mounted.
- Bone marrow is the site of immune cell production.
- The thymus is the site of T-cell maturation.
- The spleen acts as a storage area for white blood cells and platelets, a recycling center for red blood cells, and a filter of blood and lymph for the immune system.
Leukocytes are produced in the bone marrow through hematopoiesis, shown in Figure 8.3. (Note: This is the same image shown in Figure 7.7, but is copied here for convenience.) Leukocytes are divided into two groups of cells: granulocytes and agranulocytes. These names refer to the presence or absence of granules in the cytoplasm. These granules contain toxic enzymes and chemicals, which can be released by exocytosis, and are particularly effective against bacterial, fungal, and parasitic pathogens. Both granulocytes and agranulocytes come from a common precursor: hematopoietic stem cells. Remember from Chapter 7 of MCAT Biology Review that hematopoietic stem cells are also the cell type that gives rise to red blood cells and platelets. Granulocytes include cells such as neutrophils, eosinophils, and basophils. The names of these cells actually refer to the way that the cells appear after staining with certain chemicals. Agranulocytes include the lymphocytes, which are responsible for antibody production, immune system modulation, and targeted killing of infected cells. Monocytes, which are phagocytic cells in the bloodstream, are also considered agranulocytes. They become macrophages in tissues; many tissues have resident populations of macrophages with specific names (such as microglia in the central nervous system, Langerhans cells in the skin, and osteoclasts in bone).
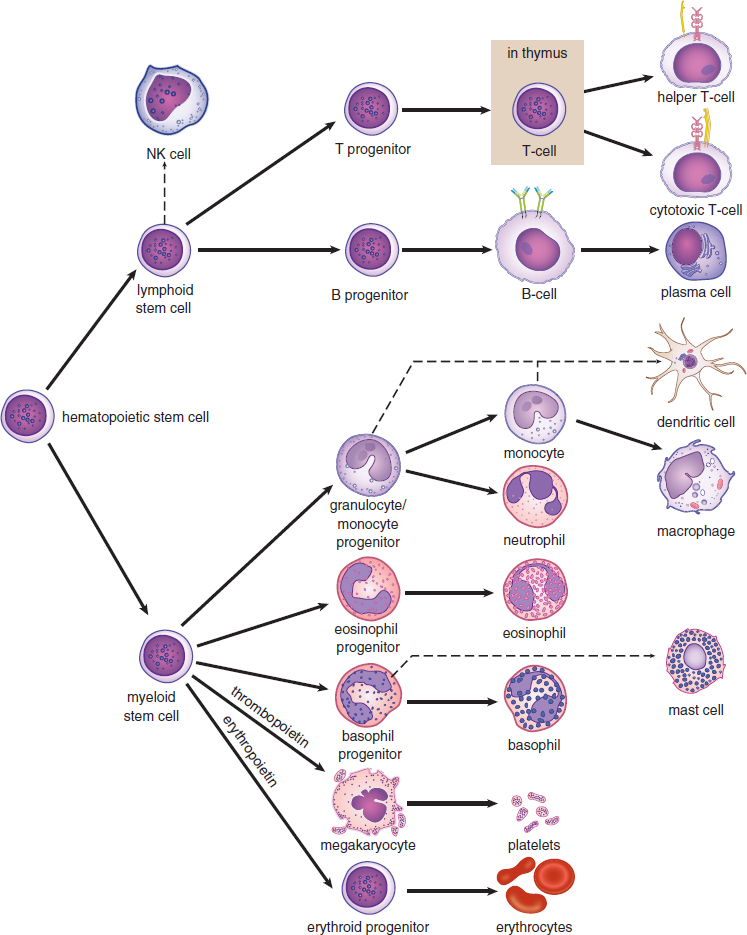
Figure 8.3. Hematopoiesis
REAL WORLD
One of the tests often ordered by physicians is known as a complete blood count (CBC). This test counts the number of red blood cells, platelets, and white blood cells. If the CBC is ordered with a differential, it means that each type of white blood cell is counted. Any cell populations that are abnormal may indicate a particular type of infection given the right clinical conditions. For example, if 90 percent of the white blood cells are neutrophils, it is possible that a bacterial infection is present. If 20 percent of the white blood cells are eosinophils, it is possible that an invasive parasitic infection is present.
Innate immunity refers to the responses cells can carry out without learning; for this reason, it is also known as the nonspecific immune response. Conversely, adaptive immunity is developed as immune cells learn to recognize and respond to particular antigens, and is often aptly referred to as the specific immune response. We can also divide the specific immune system into humoral immunity (driven by B-cells and antibodies) and cell-mediated immunity (driven by T-cells).
MCAT CONCEPT CHECK 8.1
Before you move on, assess your understanding of the material with these questions.
- What are the differences between innate and adaptive immunity?
- Innate immunity:
- Adaptive immunity:
- Compare and contrast B- and T-cells:
Cell Type Site of Development Site of Maturation Major Functions Specific or Nonspecific? Humoral or Cell-Mediated?
B-cell T-cell
- Which cells are considered granulocytes? Which are considered agranulocytes?
- Granulocytes:
- Agranulocytes:
8.2 The Innate Immune System
LEARNING OBJECTIVES
After Chapter 8.2, you will be able to:
- Describe the immunologic functions of each of the noncellular nonspecific immune defenses, including defensins and mucus
- Identify the immune cell type used for antigen presentation
- Differentiate between MHC-I and MHC-II
- Recall the stimuli required for activating natural killer cells, neutrophils, eosinophils, basophils, and mast cells
The innate immune system consists of cells and structures that offer nonspecific protection.
Noncellular Nonspecific Defenses
Our first line of defense is the skin (integument). In Chapter 10 of MCAT Biology Review, we will discuss the specific homeostatic functions of the skin; for now, we’ll focus on how skin protects the body. The skin provides a physical barrier between the outside world and our internal organs, preventing most bacteria, viruses, fungi, and parasites from entering the body. Additionally, antibacterial enzymes called defensins can be found on the skin. Sweat also has antimicrobial properties. The skin is an important first line of defense: a cut or abrasion on the skin provides an entry point for pathogens into the body. Deeper wounds allow pathogens to penetrate deeper into the body.
As discussed in Chapter 6 of MCAT Biology Review, the respiratory system also has mechanisms to prevent pathogens from entering the body. The respiratory passages are mucous membranes, lined with cilia to trap particulate matter and push it up toward the oropharynx, where it can be swallowed or expelled. While mucus helps to trap particulates like smoke and dirt, it also helps to prevent bacteria and viruses from gaining access to the lung tissue below. Several other mucous membranes, including those around the eye and in the oral cavity, produce a nonspecific bacterial enzyme called lysozyme, which is secreted in tears and saliva, respectively.
The Gastrointestinal Tract
The gastrointestinal tract also plays a role in nonspecific immunity. First, the stomach secretes acid, resulting in the elimination of most pathogens. In addition, the gut is colonized by bacteria. Most of these bacteria lack the necessary characteristics to cause infection. Because there is already such a large bacterial population in the gut, many potential invaders are not able to compete and are thus kept at bay. Many antibiotics reduce the population of gut flora, providing an opportunity for the growth of pathogens resistant to that antibiotic.
REAL WORLD
The GI tract of a newborn baby is particularly susceptible to infection because the newborn’s immune system is underdeveloped and the GI tract is not yet colonized. Breast milk contains a family of antibodies that are particularly effective on mucosal surfaces and help to defend newborn babies against gastrointestinal infections.
Complement
The complement system consists of a number of proteins in the blood that act as a nonspecific defense against bacteria. Complement can be activated through a classical pathway (which requires the binding of an antibody to a pathogen) or an alternative pathway (which does not require antibodies). The complement proteins punch holes in the cell walls of bacteria, making them osmotically unstable. Despite the association with antibodies, complement is considered a nonspecific defense because it cannot be modified to target a specific organism over others.
Interferons
To protect against viruses, cells that have been infected with viruses produce interferons, proteins that prevent viral replication and dispersion. Interferons cause nearby cells to decrease production of both viral and cellular proteins. They also decrease the permeability of these cells, making it harder for a virus to infect them. In addition, interferons upregulate MHC class I and class II molecules, resulting in increased antigen presentation and better detection of the infected cells by the immune system, as described in the next section. Interferons are responsible for many “flu-like” symptoms that occur during viral infection, including malaise, tiredness, muscle soreness, and fever.
Cells of the Innate Immune System
So, what happens when bacteria, viruses, fungi, or parasites breach noncellular defenses? The cells of the innate immune system are always poised and ready to attack.
Macrophages
Macrophages, a type of agranulocyte, reside within the tissues. These cells derive from blood-borne monocytes and can become a resident population within a tissue (becoming a permanent, rather than transient, cell group in the tissue). Many of these resident macrophages are highlighted throughout MCAT Biology Review, including microglia in the central nervous system, Langerhans cells in the skin, and osteoclasts in bone. When a bacterial invader enters a tissue, the macrophages become activated. The activated macrophage does three things. First, it phagocytizes the invader through endocytosis. Then, it digests the invader using enzymes. Finally, it presents little pieces of the invader (mostly peptides) to other cells using a protein called major histocompatibility complex (MHC). MHC binds to a pathogenic peptide (also called an antigen) and carries it to the cell surface, where it can be recognized by cells of the adaptive immune system. In addition, macrophages release cytokines, chemical substances that stimulate inflammation and recruit additional immune cells to the area.
MHC molecules come in two main classes: class I and class II. All nucleated cells in the body display MHC class I molecules. Any protein produced within a cell can be loaded onto MHC-I and presented on the surface of the cell, as shown in Figure 8.4. This allows the immune system to monitor the health of these cells and to detect if the cells have been infected with a virus or another intracellular pathogen; only those cells that are infected would be expected to present an unfamiliar (nonself) protein on their surfaces. Therefore, the MHC-I pathway is often called the endogenous pathway because it binds antigens that come from inside the cell. Cells that have been invaded by intracellular pathogens can then be killed by a certain group of T-cells (cytotoxic T-lymphocytes) to prevent infection of other cells.
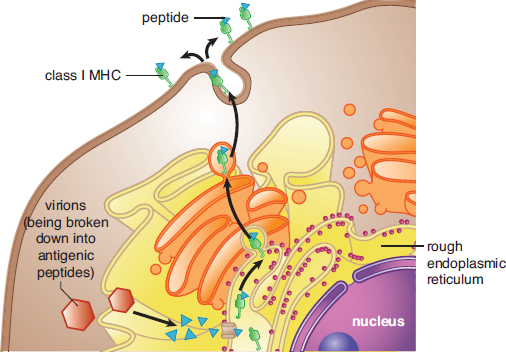
Figure 8.4. Endogenous Pathway for Antigen Presentation (MHC Class I) MHC-I exists in all nucleated cells.
REAL WORLD
MHC molecules are highly variable among humans, but can usually be divided into particular subtypes. People with certain MHC subtypes have different susceptibility to disease, and individuals with certain subtypes are considerably more susceptible to autoimmune diseases. It is also important to match the MHC types of the donor and recipient as closely as possible during transplants to avoid rejection.
MHC class II molecules are mainly displayed by professional antigen-presenting cells like macrophages, as shown in Figure 8.5. Remember that these phagocytic cells pick up pathogens from the environment, process them, and then present them on MHC-II. An antigen is a substance (usually a pathogenic protein) that can be targeted by an antibody. While antibody production is the domain of the adaptive immune system, it is important to understand that cells of the innate immune system also present antigens. Because these antigens originated outside the cell, the MHC-II pathway is often called the exogenous pathway. The presentation of an antigen by an immune cell may result in the activation of both the innate and adaptive immune systems. Professional antigen-presenting cells include macrophages, dendritic cells in the skin, some B-cells, and certain activated epithelial cells.
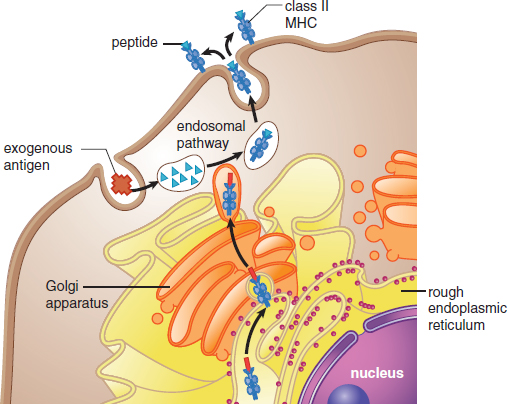
Figure 8.5. Exogenous Pathway for Antigen Presentation (MHC Class II) MHC-II exists only in professional antigen-presenting cells, like macrophages, dendritic cells, some B-cells, and some activated epithelial cells.
KEY CONCEPT
Major histocompatibility complex molecules are joined with antigens. The MHC–antigen complex then goes to the cell surface to display the antigen. This allows the immune system to monitor the health of cells. MHC-I is in all nucleated cells and presents endogenous antigens. MHC-II is in antigen-presenting cells and presents exogenous antigens.
Macrophages and dendritic cells also have special receptors known as pattern recognition receptors (PRR), the best-described of which are toll-like receptors (TLR). PRRs are able to recognize the category of the invader (bacterium, virus, fungus, or parasite). This allows for the production of appropriate cytokines to recruit the right type of immune cells; each immune cell has different weapons that can target particular groups of pathogens.
Natural Killer Cells
In the arms race between the human immune system and pathogens, some pathogens have found ways to avoid certain defenses. For example, some viruses cause downregulation of MHC molecules, making it harder for T-cells to recognize the presence of an infection. Natural killer (NK) cells, a type of nonspecific lymphocyte, are able to detect the downregulation of MHC and induce apoptosis in these virally infected cells. Cancer cells may also downregulate MHC production, so NK cells also offer protection from the growth of cancer as well.
KEY CONCEPT
The innate immune cells are nonspecific and form the first line of defense against pathogens.
Innate Immunity
Macrophages envelop pathogens and break them down.
Granulocytes contain granules and also contribute to inflammation. The three types of granulocytes are neutrophils, eosinophils, and basophils.
Dendritic cells induce adaptive immune cells to attack pathogens by presenting them with antigens from those pathogens.
Natural killer cells destroy cancer cells and body cells infected with pathogens.
Mast cells release histamine and other inflammatory agents.
Granulocytes
In addition to macrophages, the granulocytes, which include neutrophils, eosinophils, and basophils (and closely related mast cells), are also involved in nonspecific defense. Neutrophils are the most populous leukocyte in blood and are very short lived (a bit more than five days). These cells are phagocytic, like macrophages, and target bacteria. Neutrophils can follow bacteria using chemotaxis—the movement of an organism according to chemical stimuli; in this case, the neutrophil senses products given off by bacteria, moving up the concentration gradient to the source. Neutrophils can also detect bacteria once they have been opsonized (marked with an antibody from a B-cell). Other cells, like natural killer cells, macrophages, monocytes, and eosinophils, also contain receptors for antibodies and can attack opsonized bacteria. Dead neutrophil collections are responsible for the formation of pus during an infection.
Eosinophils contain bright red-orange granules and are primarily involved in allergic reactions and invasive parasitic infections. Upon activation, eosinophils release large amounts of histamine, an inflammatory mediator. This results in vasodilation and increased leakiness of the blood vessels, allowing additional immune cells (especially macrophages and neutrophils) to move out of the bloodstream and into the tissue. Inflammation is particularly useful against extracellular pathogens, including bacteria, fungi, and parasites.
KEY CONCEPT
Histamine causes inflammation by inducing vasodilation and the movement of fluid and cells from the bloodstream into tissues.
Finally, basophils contain large purple granules and are involved in allergic responses. They are the least populous leukocyte in the bloodstream under normal conditions. Mast cells are closely related to basophils, but have smaller granules and exist in the tissues, mucosa, and epithelium. Both basophils and mast cells release large amounts of histamine in response to allergens, leading to inflammatory responses.
MCAT CONCEPT CHECK 8.2
Before you move on, assess your understanding of the material with these questions.
- For each of the noncellular nonspecific immune defenses listed below, provide a brief description of its immunologic function:
- Skin:
- Defensins:
- Lysozyme:
- Mucus:
- Stomach acid:
- Normal gastrointestinal flora:
- Complement:
- Which cells are professional antigen-presenting cells?
- What are the differences between MHC-I and MHC-II?
- MHC-I:
- MHC-II:
- What stimulus activates each of the following types of cells?
- Natural killer cells:
- Neutrophils:
- Eosinophils:
- Basophils and mast cells:
8.3 The Adaptive Immune System
LEARNING OBJECTIVES
After Chapter 8.3, you will be able to:
- Recall the major classes of lymphocytes and their functions
- Describe the three main effects that circulating antibodies can have on a pathogen
- Describe the effects of positive and negative selection on T-cell maturation
- Explain why the secondary response to a pathogen is more efficient than the primary response
- Differentiate between passive and active immunity
- Explain how antibodies become specific for a given antigen
The adaptive immune system can identify specific invaders and mount an attack against that pathogen. The response is variable and depends on the identity of the pathogen. The adaptive immune system can be divided into two divisions: humoral immunity and cell-mediated (cytotoxic) immunity. Each involves the identification of the specific pathogen and organization of an appropriate immune response.
Cells of the Adaptive Immune System
The adaptive immune system consists mainly of two types of lymphocytes, B-cells and T-cells. B-cells govern the humoral response, while T-cells mount the cell-mediated response. All cells of the immune system are created in the bone marrow, but B- and T-cells mature in different locations. B-cells mature in the bone marrow and spleen (although the B in their name originally stood for the bursa of Fabricius, an organ found in birds), and T-cells mature in the thymus. When we are exposed to a pathogen, it may take a few days for the physical symptoms to be relieved. This occurs because the adaptive immune response takes time to form specific defenses against the pathogen.
KEY CONCEPT
B-cells mature in the bone marrow. T-cells mature in the thymus.
Humoral Immunity
Humoral immunity, which involves the production of antibodies, may take as long as a week to become fully effective after initial infection. These antibodies are specific to the antigens of the invading microbe. Antibodies are produced by B-cells, which are lymphocytes that originate and mature in the bone marrow and are activated in the spleen and lymph nodes.
KEY CONCEPT
The adaptive immune cells target invaders with greater specificity, but respond slower since they are replicated in response to an antigen.
Adaptive Immunity
B-cells respond to antigens by dividing and making antibodies that can neutralize pathogens directly or indirectly with the help of other leukocytes.
T-cells have a variety of functions in adaptive immune response. Cytotoxic T-cells detect specific antigens and destroy the infected cells that present them. Suppressor and helper T-cells play coordinating roles in immune response. Memory T-cells prepare for potential reinfections.
Antibodies (also called immunoglobulins [Ig]) can carry out many different jobs in the body. Just as antigens can be displayed on the surface of cells or can float freely in blood, chyle (lymphatic fluid), or air, so too can antibodies be present on the surface of a cell or secreted into body fluids. When an antibody binds to an antigen, the response will depend on the location. For antibodies secreted into body fluids, there are three main possibilities: first, once bound to a specific antigen, antibodies may attract other leukocytes to phagocytize those antigens immediately. This is called opsonization, as described earlier. Second, antibodies may cause pathogens to clump together or agglutinate, forming large insoluble complexes that can be phagocytized. Third, antibodies can block the ability of a pathogen to invade tissues, essentially neutralizing it. For cell-surface antibodies, the binding of antigen to a B-cell causes activation of that cell, resulting in its proliferation and formation of plasma and memory cells, as described later in this chapter. In contrast, when antigen binds to antibodies on the surface of a mast cell, it causes degranulation (exocytosis of granule contents), releasing histamine and causing an inflammatory allergic reaction.
Antibodies are Y-shaped molecules that are made up of two identical heavy chains and two identical light chains, as shown in Figure 8.6. Disulfide linkages and noncovalent interactions hold the heavy and light chains together. Each antibody has an antigen-binding region at the end of which is called the variable region (domain), at the tips of the Y. The branches of the Y are known as the fragment antigen binding region (Fab region). Within this region, there are specific polypeptide sequences that will bind one, and only one, specific antigenic sequence. Part of the reason it takes so long to initiate the antibody response is that each B-cell undergoes hypermutation of its antigen-binding region, trying to find the best match for the antigen. Only those B-cells that can bind the antigen with high affinity survive, providing a mechanism for generating specificity called clonal selection. The remaining part of the antibody molecule is known as the constant region (domain), also known as the fragment crystallizable region (Fc region). It is this region that cells such as natural killer cells, macrophages, monocytes, and eosinophils have receptors for, and that can initiate the complement cascade. Each B-cell makes only one type of antibody, but we have many B-cells, so our immune system can recognize many antigens. Further, antibodies come in five different isotypes (IgM, IgD, IgG, IgE, and IgA). While the specific purposes of each antibody isotype is outside the scope of the MCAT, you should know that the different types can be used at different times during the adaptive immune response, for different types of pathogens, or in different locations in the body. Cells can change which isotype of antibody they produce when stimulated by specific cytokines in a process called isotype switching.
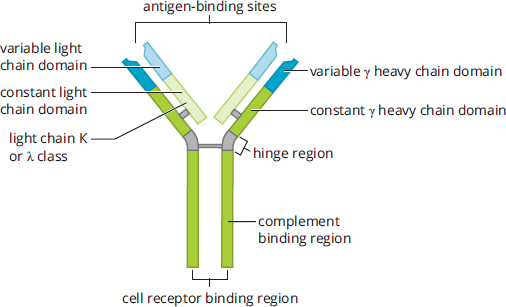
Figure 8.6. Structure of an Antibody Molecule
Not all B-cells that are generated actively or constantly produce antibodies. Antibody production is an energetically expensive process, and there is no reason to expend energy to produce antibodies that are not needed. Instead, naïve B-cells (those that have not yet been exposed to an antigen) wait in the lymph nodes for their particular antigen to come along. Upon exposure to the correct antigen, a B-cell will proliferate and produce two types of daughter cells. Plasma cells produce large amounts of antibodies, whereas memory B-cells stay in the lymph node, awaiting reexposure to the same antigen. This initial activation takes approximately seven to ten days and is known as the primary response. The plasma cells will eventually die, but the memory cells may last the lifetime of the organism. If the same microbe is ever encountered again, the memory cells rapidly proliferate and differentiate into plasma cells to produce antibodies specific to that pathogen. This immune response, called the secondary response, will be more rapid and robust. The development of these lasting memory cells is the basis of the efficacy of vaccination.
Cytotoxic Immunity
Whereas humoral immunity is based on the activity of B-cells, cell-mediated immunity involves the T-cells. T-cells mature in the thymus, where they undergo both positive and negative selection. Positive selection refers to allowing only the maturation of cells that can respond to the presentation of antigen on MHC (cells that cannot respond to MHC undergo apoptosis because they will not be able to respond in the periphery). Negative selection refers to causing apoptosis in cells that are self-reactive (activated by proteins produced by the organism itself). The maturation of T-cells is facilitated by thymosin, a peptide hormone secreted by thymic cells. Once the T-cell has left the thymus, it is mature but naïve. Upon exposure to antigen, T-cells will also undergo clonal selection so that only those with the highest affinity for a given antigen proliferate.
There are three major types of T-cells: helper T-cells, suppressor T-cells, and killer (cytotoxic) T-cells. Helper T-cells (Th), also called CD4+ T-cells, coordinate the immune response by secreting chemicals known as lymphokines. These molecules are capable of recruiting other immune cells (such as plasma cells, cytotoxic T-cells, and macrophages) and increasing their activity. The loss of these cells, as occurs in human immunodeficiency virus (HIV) infection, prevents the immune system from mounting an adequate response to infection; in advanced HIV infection, also called acquired immunodeficiency syndrome (AIDS), even weak pathogens can cause devastating consequences as opportunistic infections. CD4+ T-cells respond to antigens presented on MHC-II molecules. Because MHC-II presents exogenous antigens, CD4+ T-cells are most effective against bacterial, fungal, and parasitic infections.
REAL WORLD
“CD” in immunology stands for cluster of differentiation and includes cell-surface markers that can be detected by the lab technique called flow cytometry; these markers give an indication of the types of leukocytes under investigation, how many are present, and their state of maturity.
Cytotoxic T-cells (Tc or CTL, for cytotoxic T-lymphocytes), also called CD8+ T-cells, are capable of directly killing virally infected cells by injecting toxic chemicals that promote apoptosis into the infected cell. CD8+ T-cells respond to antigens presented on MHC-I molecules. Because MHC-I presents endogenous antigens, CD8+ T-cells are most effective against viral (and intracellular bacterial or fungal) infections.
KEY CONCEPT
CD4+ T-cells are better at fighting extracellular infections, while CD8+ T-cells are better at targeting intracellular infections.
Suppressor or regulatory T-cells (Treg) also express CD4, but can be differentiated from helper T-cells because they also express a protein called Foxp3. These cells help to tone down the immune response once infection has been adequately contained. These cells also turn off self-reactive lymphocytes to prevent autoimmune diseases: this is termed self-tolerance.
MNEMONIC
CD × MHC = 8
CD4+ cells respond to MHC-II (4 × 2 = 8)
CD8+ cells respond to MHC-I (8 × 1 = 8)
Finally, memory T-cells can be generated. Similar to memory B-cells, these cells lie in wait until the next exposure to the same antigen. When activated, they carry out a more robust and rapid response.
REAL WORLD
Many suppressor T-cells were formerly self-reactive T-cells that have been turned off. When a suppressor T-cell inactivates another lymphocyte, it can either target it for destruction or promote its conversion into another suppressor T-cell.
A summary of the different types of lymphocytes in adaptive (specific) immunity is shown in Figure 8.7.
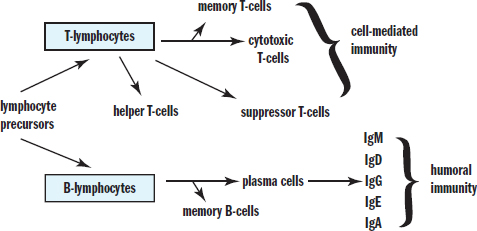
Figure 8.7. Lymphocytes of Specific Immunity This diagram shows the differentiation of lymphocyte precursors and the cell types involved in specific immunity.
Activation of the Adaptive Immune System
It is important to note that the innate and adaptive immune systems are not really disparate entities that function separately. The proper functioning of the entire immune system depends on the interactions between these two systems. There are five types of infectious pathogens: bacteria, viruses, fungi, parasites (including protozoa, worms, and insects), and prions (for which there are no immune defenses). While the immune system’s response depends on the specific identity of the pathogen, we present two classic examples: a bacterial (extracellular pathogen) infection and a viral (intracellular pathogen) infection. Keep in mind that this categorization is imperfect; for example, some bacteria, like Mycobacterium tuberculosis and Listeria monocytogenes, are actually intracellular pathogens.
Bacterial (Extracellular Pathogen) Infections
Macrophages are like the sentinels of the human body, always on the lookout for potential invaders. Let’s say a person suffers a laceration and bacteria are introduced into the body via this laceration. First, macrophages (and other antigen-presenting cells) engulf the bacteria and subsequently release inflammatory mediators. These cells also digest the bacteria and present antigens from the pathogen on their surfaces in conjunction with MHC-II. The cytokines attract inflammatory cells, including neutrophils and additional macrophages. Mast cells are activated by the inflammation and degranulate, resulting in histamine release and increased leakiness of the capillaries. This augments the ability of the immune cells to leave the bloodstream to travel to the affected tissue. The dendritic cell then leaves the affected tissue and travels to the nearest lymph node, where it presents the antigen to B-cells. B-cells that produce the correct antibody proliferate through clonal selection to create plasma cells and memory cells. Antibodies then travel through the bloodstream to the affected tissue, where they tag the bacteria for destruction.
At the same time, dendritic cells are also presenting the antigen to T-cells, activating a T-cell response. In particular, CD4+ T-cells are activated. These cells come in two types, called Th1 and Th2. Th1 cells release interferon gamma (IFN-γ), which activates macrophages and increases their ability to kill bacteria. Th2 cells help activate B-cells and are more common in parasitic infections.
After the pathogen has been eliminated, plasma cells die, but memory B- and T-cells remain. These memory cells allow for a much faster secondary response upon exposure to the pathogen at a later time.
Viral (Intracellular Pathogen) Infections
In a viral infection, the virally infected cell will begin to produce interferons, which reduce the permeability of nearby cells (decreasing the ability of the virus to infect these cells), reduce the rate of transcription and translation in these cells (decreasing the ability of the virus to multiply), and cause systemic symptoms (malaise, muscle aching, fever, and so on). These infected cells also present intracellular proteins on their surface in conjunction with MHC-I; in a virally infected cell, at least some of these intracellular proteins will be viral proteins.
CD8+ T-cells will recognize the MHC-I and antigen complex as foreign and will inject toxins into the cell to promote apoptosis. In this way, the infection can be shut down before it is able to spread to nearby cells. In the event that the virus downregulates the production and presentation of MHC-I molecules, natural killer cells will recognize the absence of MHC-I and will accordingly cause apoptosis of these cells.
Again, once the pathogen has been cleared, memory T-cells will be generated that can allow a much faster response to be mounted upon a second exposure.
Recognition of Self and Nonself
Self-antigens are the proteins and carbohydrates present on the surface of every cell of the body. Under normal circumstances, these self-antigens signal to immune cells that the cell is not foreign and should not be attacked. However, when the immune system fails to make the distinction between self and foreign, it may attack cells expressing particular self-antigens, a condition known as autoimmunity. Note that autoimmunity is only one potential problem with immune functioning: another problem arises when the immune system misidentifies a foreign antigen as dangerous when, in fact, it is not. Pet dander, pollen, and peanuts are not inherently threatening to human life, yet some people’s immune systems are hypersensitive to these antigens and become overactivated when these antigens are encountered. This is called an allergic reaction. Allergies and autoimmunity are part of a family of immune reactions classified as hypersensitivity reactions.
The human body strives to prevent autoimmune reactions very early in T-cell and B-cell maturation processes. T-cells are educated in the thymus. Part of this education involves the elimination of T-cells that respond to self-antigens, called negative selection. Immature B-cells that respond to self-antigens are eliminated before they leave the bone marrow. However, this process is not perfect, and occasionally a cell that responds to self-antigens is allowed to survive. Most autoimmune diseases can be treated with a number of therapies; one common example is administration of glucocorticoids (modified versions of cortisol), which have potent immunosuppressive qualities.
REAL WORLD
Autoimmune diseases can result in destruction of tissues, which may cause various deficiencies. Type I diabetes mellitus results from autoimmune destruction of the β-cells of the pancreas. This results in an inability to produce insulin, characterized by high blood sugars and excessive utilization of fats and proteins for energy. Other examples of autoimmune diseases include multiple sclerosis, myasthenia gravis, psoriasis, systemic lupus erythematosus, rheumatoid arthritis, Graves’ disease, and Guillain–Barré syndrome.
Immunization
Often, diseases can have significant, long-term consequences. Infection with the poliovirus, for example, can cause permanent paralysis. Polio used to be a widespread illness; however, today we hardly hear about outbreaks of polio because of a highly effective vaccination program that led to the elimination of polio from many parts of the world.
Immunization can be achieved in an active or passive fashion. In active immunity, the immune system is stimulated to produce antibodies against a specific pathogen. The means by which we are exposed to this pathogen may either be natural or artificial. Through natural exposure, antibodies are generated by B-cells once an individual becomes infected. Artificial exposure (through vaccines) also results in the production of antibodies; however, the individual never experiences true infection. Instead, the individual receives an injection or intranasal spray containing an antigen that will activate B-cells to produce antibodies to fight the specific infection. The antigen may be a weakened or killed form of the microbe, or it may be a part of the microbe’s protein structure.
Immunization may also be achieved passively. Passive immunity results from the transfer of antibodies to an individual. The immunity is transient because only the antibodies, and not the plasma cells that produce them, are given to the individual. Natural examples are the transfer of antibodies across the placenta during pregnancy to protect the fetus and the transfer of antibodies from a mother to her nursing infant through breast milk. In some cases of exposure, such as to the rabies virus or tetanus, intravenous immunoglobulin may be given to prevent the pathogen from spreading.
REAL WORLD
In 1998, a paper published in The Lancet claimed to have found a link between vaccines and autism. This paper was withdrawn from The Lancet after it was demonstrated that the primary author had an undisclosed conflict of interest and that the results were scientifically inaccurate. In fact, no well-designed scientific study has yet shown this link to exist. Nevertheless, reporting of this unsubstantiated connection in the lay media has influenced some parents and guardians to avoid immunizing their children. Since 1998, outbreaks of measles and mumps in the United States and other industrialized nations have raised concerns about the resurgence of illnesses that were previously thought to have been eliminated from these countries. Vaccines do carry risks, including rare cases of encephalitis (brain inflammation) and Guillain–Barré syndrome (an autoimmune disease in which the myelin of peripheral nerves is attacked), but so too do the pathogens these vaccines protect against.
BIOLOGY GUIDED EXAMPLE WITH EXPERT THINKING
The development of immune-mediated liver failure during viral hepatitis has recently been linked to the action of CD8+ T-cells. Several effector mechanisms have been identified in preclinical models, including the cytokines interferon (IFN) and tumor necrosis factor (TNF), as well as apoptosis-inducing molecules such as Fas Ligand (FasL) and perforin-1 (Prf1). To better study these factors, a mouse model of acute hepatitis was developed by immunizing mice with OVA epitope (a short peptide sequence derived from the protein ovalbumin) and then infected with recombinant adenovirus expressing OVA peptide 30 days later. Acute liver damage was observed, marked by an increase in serum alanine aminotransferase (sALT), a marker of liver damage, and a decrease in body weight. Topic CD8+ T-cells and hepatitis These are all components of the immune system that may lead to immune-mediated liver failure New model to study immune-mediated liver damage during acute hepatitis Damage to liver = elevated sALT, decrease in body weight
To study the contribution of FasL and TNF to liver pathology, mice were injected with Anti-FasL, anti-TNF, or IgG control antibodies before adenovirus challenge. Researchers measured sALT levels and body weight in the days post challenge (Figure 1). To better elucidate the role of perforin-1, OVA-specific T-cells (OT-1 cells) with either WT perforin or perforin-1 KO (OT-1 x Prf1–/– cells), were injected into naive mice prior to adenovirus challenge. Serum ALT and weight measurements were taken following infection (Figure 2). Exp 1: test FasL and TNF contribution, antibodies added that will prevent factors from exerting their normal biological function Exp 2: test role of perforin-1, add T-cells with and without perforin-1, measure sALT and body mass
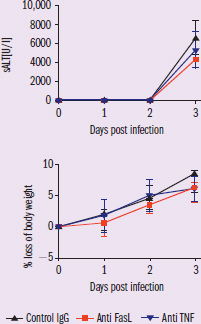
Figure 1 Figure 1 = antibody study IV: antibody treatment DVs: sALT levels and % loss of body weight Trend: in both graphs, the three groups have overlapping error bars
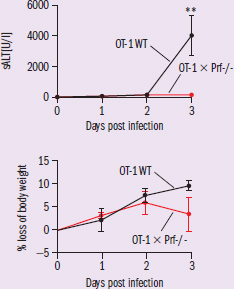
Figure 2 Figure 2 = cell transfer study IV: presence or absence of perforin-1 DVs: same as previous Trends: mice given perforin-1 knockout cells have no increase in sALT and lose less weight compared to controls
Adapted from Welz, M., Eickhoff, S., Abdullah, Z., Trebicka, J., Gartlan, K. H., Spicer, J. A., & Kastenmüller, W. (2018). Perforin inhibition protects from lethal endothelial damage during fulminant viral hepatitis. Nature Communications, 9(1), 4805. doi:10.1038/s41467-018-07213-x.
Based on the results, which of the three parameters tested, FAS Ligand, Tumor Necrosis Factor, or Perforin, is most likely involved in the destruction of liver tissue during acute infection?
This question is really testing our understanding of the experimental design and comprehension of the results. Let’s start with the experimental design. Paragraph 1 tells us that there are four main factors that are believed to be involved in immune-mediated liver damage: FasL, TNF, perforin-1, and INF. To test the involvement of these factors, the researchers designed a new experimental model. They first injected mice with OVA peptide to induce a primary immune response and the development of memory cells. Thirty days later, the researchers infected the mice with adenovirus expressing OVA. This will result in activation of memory cells and a secondary immune response, which the researchers showed was able to induce immune-controlled liver damage.
In the first experiment, the mice were injected with antibodies against FasL and TNF prior to adenovirus infection. These antibodies will bind to FasL and TNF, rendering them unable to function, similar to knocking out the gene (but significantly cheaper than developing transgenic mice!). Figure 1 shows the results. We notice that there is no significant difference in sALT levels nor body weight loss between mice treated with the control antibody and mice treated with either FasL or TNF antibody. We see the same increase in sALT and the same decrease in weight loss, indicating that removing TNF and FasL function did not prevent liver damage. This suggests that these two factors are not required for the immune-mediated liver damage.
In the second experiment, we test the involvement of perforin-1. The experimental setup is slightly different. Instead of injecting OVA peptide, OVA specific T-cells were injected into mice. Two different types of cells were used, normal and perforin-1 knockout. The mice were then infected with adenovirus as before. This enables us to examine the contribution of perforin-1 in liver damage. Figure 2 shows the results of the experiment. We can note that there is a significant difference in sALT levels between control and perforin-1 knockout (KO) cells. There is no elevation in sALT in the knockout cells, whereas there is an increase in control cells. This suggests that perforin-1 contributes to liver damage. Furthermore, mice given perforin-1 KO cells lose less weight compared to mice given control cells. Because a knockout of the perforin-1 gene led to a decrease in immune-mediated liver damage in mice, perforin-1 plays an important role in immune-mediated liver damage.
Because the results show no significant difference between the control and TNF and FasL antibody treated mice, TNF and FasL do not play a role in immune-mediated liver damage. However, a statistically significant difference between control and perforin-1 knockout cells was observed, indicating a role for perforin-1 in immune-mediated liver damage.
MCAT CONCEPT CHECK 8.3
Before you move on, assess your understanding of the material with these questions.
- For each of the lymphocytes listed below, what are its main functions?
- Plasma cell:
- Memory B-cell:
- Helper T-cell:
- Cytotoxic T-cell:
- Suppressor (regulatory) T-cell:
- Memory T-cell:
- What are the three main effects circulating antibodies can have on a pathogen?
-
-
-
- How do antibodies become specific for a given antigen?
- A T-cell appropriately passes through positive selection, but then inappropriately passes through negative selection. What will this T-cell be reactive toward?
- Which cells account for the fact that the secondary response to a pathogen is much more rapid and robust than the primary response?
- What is the difference between active and passive immunity?
- Active immunity:
- Passive immunity:
8.4 The Lymphatic System
LEARNING OBJECTIVES
After Chapter 8.4, you will be able to:
- Predict how a blockage of flow from a lymph node would impact the lymphatic system and the body as a whole
- Describe the factors and structures linking lymphatic and cardiovascular circulation
The immune system and the lymphatic system are intimately related. B-cells proliferate and develop within the lymphatic system, especially the lymph nodes. This system also serves other functions for the body.
Structure
The lymphatic system, along with the cardiovascular system, is a type of circulatory system. It is made up of one-way vessels that become larger as they move toward the center of the body. These vessels carry lymphatic fluid (lymph) and most join to form a large thoracic duct in the posterior chest, which then delivers the fluid into the left subclavian vein (near the heart).
Lymph nodes are small, bean-shaped structures along the lymphatic vessels. Lymph nodes contain a lymphatic channel, as well as an artery and a vein. The lymph nodes provide a space for the cells of the immune system to be exposed to possible pathogens.
REAL WORLD
Certain cancers, especially breast cancer, are prone to spread via lymphatic channels. Mastectomy, a surgery that removes the breast, is one of the treatments for breast cancer. In order to ensure that all of the cancer has been removed, local lymph nodes are often removed at the same time.
Function
The lymphatic system serves many different purposes for the body by providing a secondary system for circulation.
Equalization of Fluid Distribution
At the capillaries, fluid leaves the bloodstream and goes into the tissues. The quantity of fluid that leaves the tissues at the arterial end of the capillary bed depends on both hydrostatic and oncotic pressures (Starling forces). Remember that the oncotic pressure of the blood draws water back into the vessel at the venule end, once hydrostatic pressure has decreased. Because the net pressure drawing fluid in at the venule end is slightly less than the net pressure pushing fluid out at the arterial end, a small amount of fluid remains in the tissues. Lymphatic vessels drain these tissues and subsequently return the fluid to the bloodstream.
The lymphatics offer some protection against pathology. For example, if the blood has a low concentration of albumin (a key plasma protein), the oncotic pressure of the blood is decreased, and less water is driven back into the bloodstream at the venule end. Thus, this fluid will collect in the tissues. Provided that the lymphatic channels are not blocked, much of this fluid may eventually return to the bloodstream via the lymphatics. Only when the lymphatics are overwhelmed does edema occur—swelling due to fluid collecting in tissue.
Transportation of Biomolecules
The lymphatic system also transports fats from the digestive system into the bloodstream. Lacteals, small lymphatic vessels, are located at the center of each villus in the small intestine. Fats, packaged into chylomicrons by intestinal mucosal cells, enter the lacteal for transport. Lymphatic fluid carrying many chylomicrons takes on a milky white appearance and is called chyle.
Immunity
As stated previously in this chapter, lymph nodes are a place for antigen-presenting cells and lymphocytes to interact. B-cells proliferate and mature in the lymph nodes in collections called germinal centers.
MCAT CONCEPT CHECK 8.4
Before you move on, assess your understanding of the material with these questions.
- Filariasis is the name for an infection with a member of a certain group of parasites, most notably Wuchereria bancrofti. This parasite resides in lymph nodes and causes blockage of flow. If an individual had a W. bancrofti infection in the lymph nodes of the thigh, what would likely happen?
- What structure is primarily responsible for returning materials from lymphatic circulation to the cardiovascular system?
Conclusion
The ability to fend off microbial invasion is critical to our survival. The immune system is throughout the body and involves multiple different organs and cell types. Nonspecific mechanisms, such as intact skin, mucous membranes, defensins, lysozyme, complement, interferons, natural killer cells, neutrophils, eosinophils, basophils, and monocytes/macrophages, constitute a complex first line of defense; these mechanisms comprise the innate immune system, which is capable of an immediate response but cannot target a specific pathogen or maintain immunologic memory. The adaptive immune system, comprised of B- and T-cells (lymphocytes), allows our immune system to target specific pathogens and learn from past exposure. Thus, once we are infected with a certain strain of virus, activation of specific immunity confers long-term protection against that particular virus. We take advantage of this secondary response through immunization, and we can see the problems specificity can have when a self-antigen is labeled as foreign, leading to autoimmune disease.
If the immune system is focused on destroying pathogens, including bacteria, then it’s an interesting transition we make in the next chapter. We’re going from a “sterilization” system to one in which bacterial colonization is the norm. From oral flora to the normal gut bacteria, our ability to digest and absorb nutrients is intimately linked to symbiotic bacteria throughout the digestive tract. In the next chapter, we will explore the anatomy and physiology of the digestive system, which provides us with the raw materials to generate energy, build proteins, and carry out activities of daily living.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Structure of the Immune System
- The immune system can be divided into innate and adaptive immunity.
- Innate immunity is composed of defenses that are always active, but that cannot target a specific invader and cannot maintain immunologic memory; also called nonspecific immunity.
- Adaptive immunity is composed of defenses that take time to activate, but that target a specific invader and can maintain immunologic memory; also called specific immunity.
- The immune system is dispersed in the body.
- Immune cells come from the bone marrow.
- The spleen and lymph nodes are sites where immune responses can be mounted, and in which B-cells are activated.
- The thymus is the site of T-cell maturation.
- Gut-associated lymphoid tissue (GALT) includes the tonsils and adenoids.
- Leukocytes, or white blood cells, are involved in immune defenses.
The Innate Immune System
- Many of the nonspecific defenses are noncellular.
- The skin acts as a physical barrier and secretes antimicrobial compounds, like defensins.
- Mucus on mucous membranes traps pathogens; in the respiratory system, the mucus is propelled upward by cilia and can be swallowed or expelled.
- Tears and saliva contain lysozyme, an antibacterial compound.
- The stomach produces acid, killing most pathogens. Colonization of the gut helps prevent overgrowth by pathogenic bacteria through competition.
- The complement system can punch holes in the cell walls of bacteria, making them osmotically unstable.
- Interferons are given off by virally infected cells and help prevent viral replication and dispersion to nearby cells.
- Many of the nonspecific defenses are also cellular.
- Macrophages ingest pathogens and present them on major histocompatibility complex (MHC) molecules. They also secrete cytokines.
- MHC class I (MHC-I) is present in all nucleated cells and displays endogenous antigen (proteins from within the cell) to cytotoxic T-cells (CD8+ cells).
- MHC class II (MHC-II) is present in professional antigen-presenting cells (macrophages, dendritic cells, some B-cells, and certain activated epithelial cells) and displays exogenous antigen (proteins from outside the cell) to helper T-cells (CD4+ cells).
- Dendritic cells are antigen-presenting cells in the skin.
- Natural killer cells attack cells not presenting MHC molecules, including virally infected cells and cancer cells.
- Granulocytes include neutrophils, eosinophils, and basophils.
- Neutrophils ingest bacteria, particularly opsonized bacteria (those marked with antibodies). They can follow bacteria using chemotaxis.
- Eosinophils are used in allergic reactions and invasive parasitic infections. They release histamine, causing an inflammatory response.
- Basophils are used in allergic reactions. Mast cells are related cells found in the skin.
The Adaptive Immune System
- Humoral immunity is centered on antibody production by plasma cells, which are activated B-cells.
- Antibodies target a particular antigen. They contain two heavy chains and two light chains. They have a constant region and a variable region; the tip of the variable region is the antigen-binding region.
- When activated, the antigen-binding region undergoes hypermutation to improve the specificity of the antibody produced. Cells may be given signals to switch isotypes of antibody (IgM, IgD, IgG, IgE, IgA).
- Circulating antibodies can opsonize pathogens (mark them for destruction), cause agglutination (clumping) into insoluble complexes that are ingested by phagocytes, or neutralize pathogens.
- Cell-surface antibodies can activate immune cells or mediate allergic reactions.
- Memory B-cells lie in wait for a second exposure to a pathogen and can then mount a more rapid and vigorous immune response (secondary response).
- Cell-mediated (cytotoxic)immunity is centered on the functions of T-cells.
- T-cells undergo maturation in the thymus through positive selection (only selecting for T-cells that can react to antigen presented on MHC) and negative selection (causing apoptosis in self-reactive T-cells). The peptide hormone thymosin promotes T-cell development.
- Helper T-cells (Th or CD4+) respond to antigen on MHC-II and coordinate the rest of the immune system, secreting lymphokines to activate various arms of immune defense. Th1 cells secrete interferon gamma, which activates macrophages. Th2 cells activate B-cells, primarily in parasitic infections.
- Cytotoxic T-cells (Tc, CTL, or CD8+) respond to antigen on MHC-I and kill virally infected cells.
- Suppressor (regulatory)T-cells (Treg) tone down the immune response after an infection and promote self-tolerance.
- Memory T-cells serve a similar function to memory B-cells.
- In autoimmune conditions, a self-antigen is identified as foreign, and the immune system attacks the body’s own cells.
- In allergic reactions, nonthreatening exposures incite an inflammatory response.
- Immunization is a method of inducing active immunity (activation of B-cells that produce antibodies to an antigen) prior to exposure to a particular pathogen.
- Passive immunity is the transfer of antibodies to an individual.
The Lymphatic System
- The lymphatic system is a circulatory system that consists of one-way vessels with intermittent lymph nodes.
- The lymphatic system connects to the cardiovascular system via the thoracic duct in the posterior chest.
- The lymphatic system equalizes fluid distribution, transports fats and fat-soluble compounds in chylomicrons, and provides sites for mounting immune responses.
ANSWERS TO CONCEPT CHECKS
**8.1**
- Innate immunity consists of defenses that are always active against pathogens, but that are not capable of targeting specific invaders. It takes longer to mount a response with adaptive immunity, but the response targets a specific pathogen and maintains immunologic memory of the infection to mount a faster response during subsequent infections.
-
Cell Type Site of Development Site of Maturation Major Functions Specific or Nonspecific? Humoral or Cell-Mediated?
B-cell Bone marrow Bone marrow (but are activated in spleen or lymph nodes) Produce antibodies Specific Humoral
T-cell Bone marrow Thymus Coordinate immune system and directly kill infected cells Specific Cell-mediated
- Granulocytes include neutrophils, eosinophils, and basophils. Agranulocytes include B- and T-cells (lymphocytes) and monocytes (macrophages).
**8.2**
- Skin provides a physical barrier and secretes antimicrobial enzymes. Defensins are examples of antibacterial enzymes on the skin. Lysozyme is antimicrobial and is present in tears and saliva. Mucus is present on mucous membranes and traps incoming pathogens; in the respiratory system, cilia propel the mucus upward so it can be swallowed or expelled. Stomach acid is an antimicrobial substance in the digestive system. The normal gastrointestinal flora provides competition, making it hard for pathogenic bacteria to grow in the gut. Complement is a set of proteins in the blood that can create holes in bacteria.
- Professional antigen-presenting cells include macrophages, dendritic cells in the skin, some B-cells, and certain activated epithelial cells.
- MHC-I is found in all nucleated cells and presents pieces of proteins (peptides) created within the cell (endogenous antigens); this can allow for detection of cells infected with intracellular pathogens (especially viruses). MHC-II is only found in antigen-presenting cells and presents proteins that result from the digestion of extracellular pathogens that have been brought in by endocytosis (exogenous antigens).
- Natural killer cells are activated by cells that do not present MHC (such as virally infected cells and cancer cells). Neutrophils are activated by bacteria, especially those that have been opsonized (tagged with an antibody on their surface). Eosinophils are activated by invasive parasites and allergens. Basophils and mast cells are activated by allergens.
**8.3**
- Plasma cells form from B-cells exposed to antigen and produce antibodies. Memory B-cells also form from B-cells exposed to antigen and lie in wait for a second exposure to a given antigen to mount a rapid, robust response. Helper T-cells coordinate the immune system through lymphokines and respond to antigen bound to MHC-II. Cytotoxic T-cells directly kill virally infected cells and respond to antigen bound to MHC-I. Suppressor (regulatory) T-cells quell the immune response after a pathogen has been cleared and promote self-tolerance. Memory T-cells, like memory B-cells, lie in wait until a second exposure to a pathogen to mount a rapid, robust response.
- Circulating antibodies can mark a pathogen for destruction by phagocytic cells (opsonization), cause agglutination of the pathogen into insoluble complexes that can be taken up by phagocytic cells, or neutralize the pathogen by preventing it from invading tissues.
- B-cells originally mature in the bone marrow and have some specificity at that point; however, antibodies that can respond to a given antigen undergo hypermutation, or rapid mutation of their antigen-binding sites. Only those B-cells that have the highest affinity for the antigen survive and proliferate, increasing the specificity for the antigen over time.
- Positive selection occurs when T-cells in the thymus that are able to respond to antigen presented on MHC are allowed to survive (those that do not respond undergo apoptosis). Negative selection occurs when T-cells that respond to self-antigens undergo apoptosis before leaving the thymus. A T-cell that appropriately passes through positive selection, but then inappropriately passes through negative selection will be reactive to self-antigens.
- Memory cells allow the immune system to carry out a much more rapid and robust secondary response.
- Active immunity refers to the stimulation of the immune system to produce antibodies against a pathogen. Passive immunity refers to the transfer of antibodies to prevent infection, without stimulation of the plasma cells that produce these antibodies.
**8.4**
- Fluid would be unable to return from the lower leg, and edema would result. This infection leads to elephantiasis, severe swelling of the limb with thickening of the skin.
- The thoracic duct carries lymphatic fluid into the left subclavian vein.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. A
T-lymphocytes, which mature in the thymus, are the only specific defense against intracellular pathogens. While some bacteria, fungi, and parasites can live intracellularly, viruses—by definition—must replicate within cells. The absence of T-cells would leave an individual unable to fight viral infections with specific defenses.
2. B
The lymphocytes involved in cell-mediated immunity are the T-lymphocytes, or T-cells. There are four types of T-cells, each playing a different role in cell-mediated immunity: cytotoxic T-cells, helper T-cells, memory T-cells, and suppressor T-cells. Thus, from the answer choices, the only cells not involved in cell-mediated immunity are the plasma cells, which are differentiated immunoglobulin-secreting B-lymphocytes involved in humoral immunity. (B)is therefore the correct answer.
3. B
The main function of the lymphatic system is to collect excess interstitial fluid and return it to the circulatory system, maintaining the balance of body fluids. However, this is not one of the answer choices. In addition, the lymphatic system accepts chylomicrons from the small intestine and delivers them to the cardiovascular circulation. Transport of hormones is a function of the cardiovascular system, eliminating (A). The lymphatic system absorbs fluid that has been pushed into tissues, but does not cause the extravasation of the fluid, eliminating (C). Mast cells reside in (and are activated in) the skin and mucous membranes, eliminating (D).
4. A
Antibodies are produced by plasma cells derived from B-lymphocytes. The other cells are all types of T-lymphocytes, although memory B-cells can also exist. Upon secondary antigen exposure, memory B-cells rapidly proliferate and differentiate into plasma cells to produce antibodies. Memory B-cells do not directly produce antibodies.
5. C
In this question, true statements should be eliminated. The innate immune system does include phagocytes like macrophages, which can activate an inflammatory response to recruit additional immune cells, eliminating (A). The innate immune system acts near entry points into the body and is always active, eliminating (B). Furthermore, all blood cells, including all cells of the immune system, are derived from hematopoietic stem cells in the bone marrow, eliminating (D). By contrast, the innate immune system does not recognize specific antigens on a pathogen, making (C) correct.
6. C
Adaptive immunity involves the activation of B-cells and T-cells specific to the encountered antigen. Any choice that conforms to this paradigm will be correct. (C)indicates that B-cells are activated. Pattern recognition receptors, or PRRs, in (A)recognize patterns common to certain pathogens, but do not identify the specific pathogen. Complement is an example of a blood-borne nonspecific defense against bacteria, eliminating (B). Dendritic cells traveling to the lymph nodes (D) are a part of the interaction between the innate and adaptive immune systems, but the dendritic cells themselves are nonspecific.
7. A
Active immunity refers to the production of antibodies during an immune response. Active immunity may be conferred on an individual by vaccination, such as when an individual is injected with a weakened, inactive, or modified form of a particular antigen that stimulates the immune system to produce antibodies. Active immunity may require weeks to build. Passive immunity, on the other hand, involves the transfer of antibodies through, for example, breast milk. Another example of passive immunity would be during pregnancy, when some maternal antibodies cross the placenta and enter fetal circulation, conferring passive immunity to the fetus. Although passive immunity is acquired immediately, it is very short lived, lasting only as long as the antibodies circulate in blood.
8. D
T-cells mature in the thymus, where they are “educated.” This education involves the elimination of T-cells with improper binding to MHC–antigen complexes (positive selection) and self-reactive T-cells (negative selection). Thus, self-reactive T-cells are eliminated in the thymus.
9. C
Healthy cells exhibit MHC class I molecules. Natural killer cells monitor the expression of MHC molecules on the surface of cells. Viral infection and cancer often cause a reduction in the expression of MHC class I molecules on the cell surface. Natural killer cells detect this lack of MHC and induce apoptosis in the affected cells.
10. A
CD8+ T-cells are largely responsible for the cytotoxic immune response. By releasing toxic chemicals into virally infected cells, CD8+ T-cells are able to kill these cells in an effort to contain viral infections. Isotype switching refers to changes in the isotype of antibody produced, which is not caused by CD8+ cells, eliminating (B). Antigens are presented by macrophages, dendritic cells, certain epithelial cells, and some B-cells, eliminating (C). B-cells are not activated by cytotoxic T-lymphocytes, eliminating (D).
11. C
Lymphocytes arise from the lymphoid lineage, which includes B-cells and T-cells. Thus, all types of B- and T-cells are capable of causing lymphoma. Macrophages, however, are not lymphocytes and are not likely to cause lymphoma.
12. D
When the adaptive immune system encounters an antigen, only the cells with antibodies or T-cell receptors specific to that antigen are activated. This is known as clonal selection. While a T-cell response may be a cytotoxic response, it could also be the activation of helper T-cells; plus, this does not explain the specificity of the response, eliminating (B).
13. A
The only phagocytes that attack bacteria on this list are neutrophils and dendritic cells. Dendritic cells are able to sample and present any type of material, and reside in the skin. Neutrophils, on the other hand, are present in the bloodstream and can attack bacteria present there or in tissues. Eosinophils and basophils are involved in the development of allergies; eosinophils also defend against parasites.
14. B
The spleen is a location where B-cells mature and proliferate. Therefore, removal of the spleen is likely to result in a reduction of humoral immunity. In fact, many people receive vaccinations prior to removal of the spleen in order to bolster their immunity.
15. D
The body employs a number of nonspecific defense mechanisms against foreign invasion. The skin and mucous membranes provide a physical barrier against bacterial invasion. In addition, sweat contains enzymes that attack bacterial cell walls. Certain passages, such as the respiratory tract, are lined with ciliate mucus-coated epithelia, which filter and trap foreign particles. Macrophages engulf and destroy foreign particles. The inflammatory response is initiated in response to physical damage. The only choice that is not a nonspecific defense mechanism is (D), the correct answer. Cytotoxic T-cells are involved in (specific) cell-mediated immunity.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
Biochemistry Chapter 3
Nonenzymatic Protein Function and Protein Analysis
Biology Chapter 1
The Cell
Biology Chapter 6
The Respiratory System
Biology Chapter 7
The Cardiovascular System
Biology Chapter 9
The Digestive System
Biology Chapter 10
Homeostasis