Chapter 1: Atomic Structure

Chapter 1: Atomic Structure
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of the following is the correct electron configuration for Zn2+?
- 1s22s22p63s23p64s03d10
- 1s22s22p63s23p64s23d8
- 1s22s22p63s23p64s23d10
- 1s22s22p63s23p64s03d8
-
Which of the following quantum number sets is possible?
- n = 2; l = 2; ml = 1; m s = + 1 2
- n = 2; l = 1; ml = −1; m s = + 1 2
- n = 2; l = 0; ml = −1; m s = − 1 2
- n = 2; l = 0; ml = 1; m s = − 1 2
-
What is the maximum number of electrons allowed in a single atomic energy level in terms of the principal quantum number n ?
- 2n
- 2n + 2
- 2n2
- 2n2 + 2
-
Which of the following equations describes the maximum number of electrons that can fill a subshell?
- 2l + 2
- 4l + 2
- 2l2
- 2l2 + 2
-
Which of the following atoms only has paired electrons in its ground state?
- Sodium
- Iron
- Cobalt
- Helium
-
An electron returns from an excited state to its ground state, emitting a photon at λ = 500 nm. What would be the magnitude of the energy change if one mole of these photons were emitted? (Note: h = 6.626 × 10−34 J ·s, NA = 6.02 × 1023 mol−1)
- 3.98 × 10−21 J
- 3.98 × 10−19 J
- 2.39 × 103 J
- 2.39 × 105 J
-
Suppose an electron falls from n = 4 to its ground state, n = 1. Which of the following effects is most likely?
- A photon is absorbed.
- A photon is emitted.
- The electron moves into a p-orbital.
- The electron moves into a d-orbital.
-
Which of the following isotopes of carbon is LEAST likely to be found in nature?
- 6C
- 12C
- 13C
- 14C
-
Which of the following best explains the inability to measure position and momentum exactly and simultaneously according to the Heisenberg uncertainty principle?
- Imprecision in the definition of the meter and kilogram
- Limits on accuracy of existing scientific instruments
- Error in one variable is increased by attempts to measure the other
- Discrepancies between the masses of nuclei and of their component particles
-
Which of the following electronic transitions would result in the greatest gain in energy for a single hydrogen electron?
- An electron moves from n = 6 to n = 2.
- An electron moves from n = 2 to n = 6.
- An electron moves from n = 3 to n = 4.
- An electron moves from n = 4 to n = 3.
-
Suppose that an atom fills its orbitals as shown:

Such an electron configuration most clearly illustrates which of the following laws of atomic physics?
- Hund’s rule
- Heisenberg uncertainty principle
- Bohr model
- Rutherford model
-
How many total electrons are in a 133Cs cation?
- 54
- 55
- 78
- 132
-
The atomic weight of hydrogen is 1.008 amu. What is the percent composition of hydrogen by isotope, assuming that hydrogen’s only isotopes are 1H and 2D?
- 92% H, 8% D
- 99.2% H, 0.8% D
- 99.92% H, 0.08% D
- 99.992% H, 0.008% D
-
Consider the two sets of quantum numbers shown in the table, which describe two different electrons in the same atom.
n l ml ms
2 1 1 + 1 2
3 1 −1 + 1 2
Which of the following terms best describes these two electrons?
- Parallel
- Opposite
- Antiparallel
- Paired
-
Which of the following species is represented by the electron configuration 1s22s22p63s23p64s13d5?
- Cr
- Mn+
- Fe2+
- I only
- I and II only
- II and III only
- I, II, and III
Answer Key
- A
- B
- C
- B
- D
- D
- B
- A
- C
- B
- A
- A
- B
- A
- B
Chapter 1: Atomic Structure
CHAPTER 1
ATOMIC STRUCTURE
In This Chapter
1.1 Subatomic Particles
Protons
Neutrons
Electrons
1.2 Atomic Mass vs. Atomic Weight
Atomic Mass
Atomic Weight
1.3 Rutherford, Planck, and Bohr
Bohr Model
Applications of the Bohr Model
1.4 Quantum Mechanical Model of Atoms
Quantum Numbers
Electron Configurations
Hund’s Rule
Valence Electrons
Concept Summary
CHAPTER PROFILE
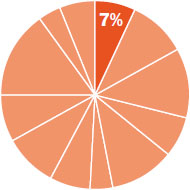
The content in this chapter should be relevant to about 7% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content category:
4E: Atoms, nuclear decay, electronic structure, and atomic chemical behavior
Introduction
Chemistry is the investigation of the atoms and molecules that make up our bodies, our possessions, the food we eat, and the world around us. There are different branches of chemistry, three of which are tested directly on the MCAT: general (inorganic) chemistry, organic chemistry, and biochemistry. Ultimately, all investigations in chemistry are seeking to answer the questions that confront us in the form—the shape, structure, mode, and essence—of the physical world that surrounds us.
Many students feel similarly about general chemistry and physics: But I’m premed, they say. Why do I need to know any of this? What good will this be when I’m a doctor? Do I only need to know this for the MCAT? Recognize that to be an effective doctor, one must understand the physical building blocks that make up the human body. Pharmacologic treatment is based on chemistry; many diagnostic tests used every day detect changes in the chemistry of the body.
So, let’s get down to the business of learning and remembering the principles of the physical world that help us understand what all this “stuff ” is, how it works, and why it behaves the way it does—at both the molecular and macroscopic levels. In the process of reading through these chapters and applying your knowledge to practice questions, you’ll prepare yourself for success not only on the Chemical and Physical Foundations of Biological Systems section of the MCAT but also in your future career as a physician.
This first chapter starts our review of general chemistry with a consideration of the fundamental unit of matter—the atom. First, we focus on the subatomic particles that make it up: protons, neutrons, and electrons. We will also review the Bohr and quantum mechanical models of the atom, with a particular focus on the similarities and differences between them.
MCAT EXPERTISE
The building blocks of the atom are also the building blocks of knowledge for the general chemistry concepts tested on the MCAT. By understanding these particles, we will be able to use that knowledge as the “nucleus” of understanding for all of general chemistry.
1.1 Subatomic Particles
LEARNING OBJECTIVES
After Chapter 1.1, you will be able to:
- Identify the subatomic particles most important for determining various traits of an atom, including charge, atomic number, and isotope
- Determine the number of protons, neutrons, and electrons within an isotope, such as 14C
Although you may have encountered in your university-level chemistry classes such subatomic particles as quarks, leptons, and gluons, the MCAT’s approach to atomic structure is much simpler. There are three subatomic particles that you must understand: protons, neutrons, and electrons.
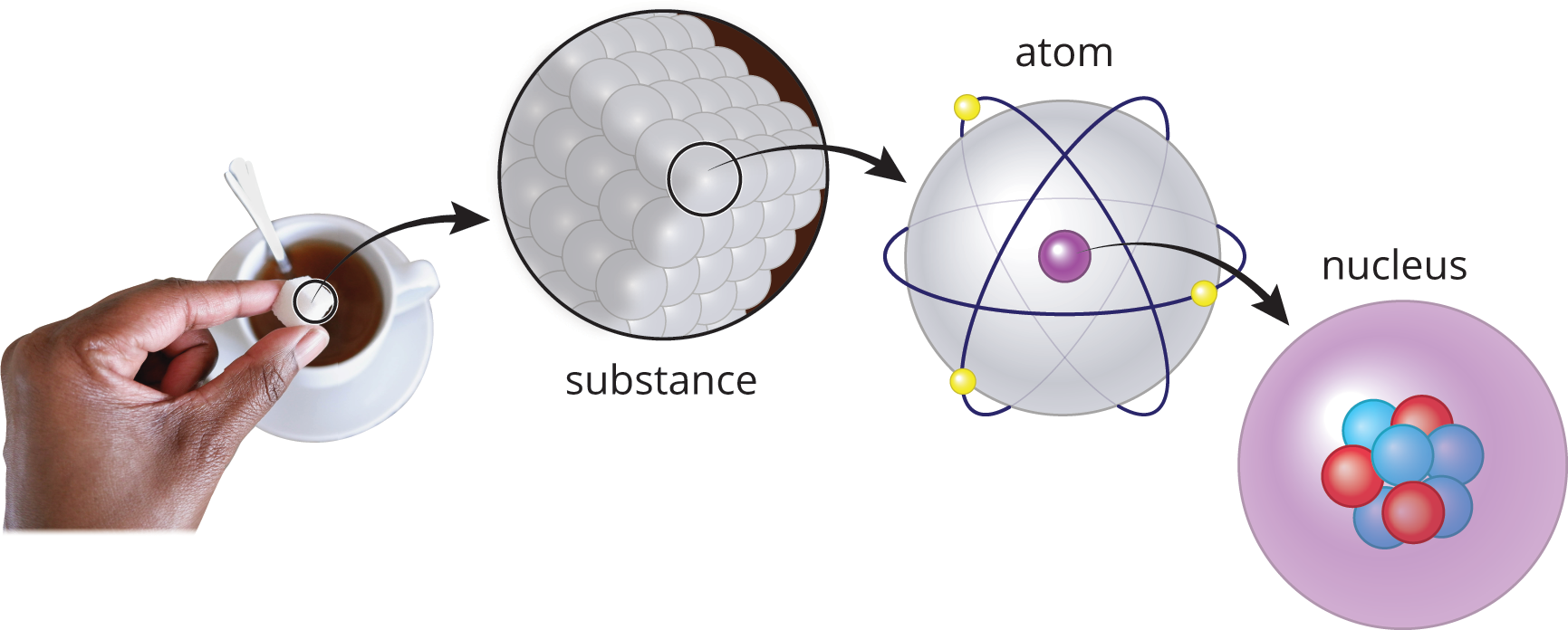
Figure 1.1. Matter: From Macroscopic to Microscopic
Protons
Protons are found in the nucleus of an atom, as shown in Figure 1.1. Each proton has an amount of charge equal to the fundamental unit of charge (e = 1.6 × 10−19 C), and we denote this fundamental unit of charge as “+1 e” or simply “+1” for the proton. Protons have a mass of approximately one atomic mass unit (amu). The atomic number (Z) of an element, as shown in Figure 1.2, is equal to the number of protons found in an atom of that element. As such, it acts as a unique identifier for each element because elements are defined by the number of protons they contain. For example, all atoms of oxygen contain eight protons; all atoms of gadolinium contain 64 protons. While all atoms of a given element have the same atomic number, they do not necessarily have the same mass—as we will see in our discussion of isotopes.

Figure 1.2. Potassium, from the Periodic Table Potassium has the symbol K (Latin:kalium), atomic number 19, and atomic weight of approximately 39.1.
Neutrons
Neutrons, as the name implies, are neutral—they have no charge. A neutron’s mass is only slightly larger than that of the proton, and together, the protons and the neutrons of the nucleus make up almost the entire mass of an atom. Every atom has a characteristic mass number (A), which is the sum of the protons and neutrons in the atom’s nucleus. A given element can have a variable number of neutrons; thus, while atoms of the same element always have the same atomic number, they do not necessarily have the same mass number. Atoms that share an atomic number but have different mass numbers are known as isotopes of the element, as shown in Figure 1.3. For example, carbon (Z = 6) has three naturally occurring isotopes: 6 12 C , with six protons and six neutrons; 6 13 C , with six protons and seven neutrons; and 6 14 C , with six protons and eight neutrons. The convention Z A X is used to show both the atomic number (Z) and the mass number (A) of atom X.
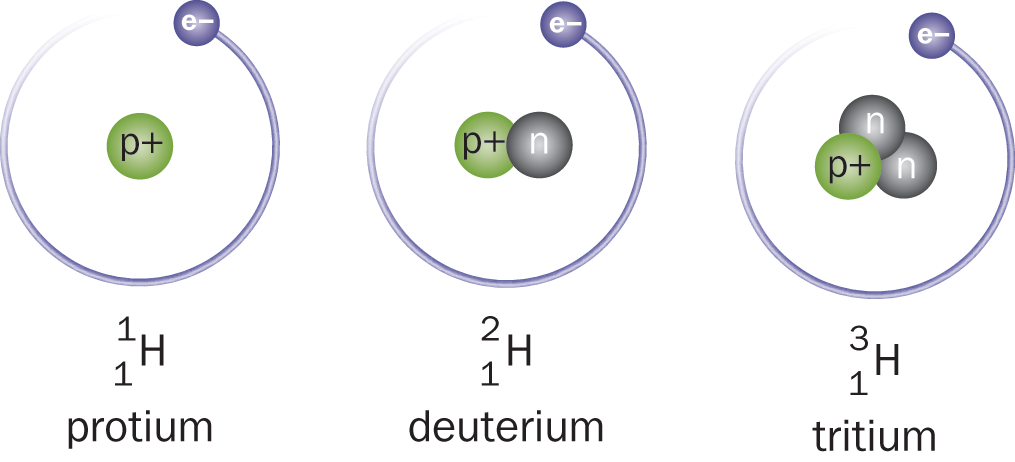
Figure 1.3. Various Isotopes of Hydrogen Atoms of the same element have the same atomic number (Z= 1) but may have varying mass numbers (A= 1, 2, or 3).
Electrons
Electrons move through the space surrounding the nucleus and are associated with varying levels of energy. Each electron has a charge equal in magnitude to that of a proton, but with the opposite (negative) sign, denoted by “−1 e” or simply “–1.” The mass of an electron is approximately 1 2000 that of a proton. Because subatomic particles’ masses are so small, the electrostatic force of attraction between the unlike charges of the proton and electron is far greater than the gravitational force of attraction based on their respective masses.
Electrons move around the nucleus at varying distances, which correspond to varying levels of electrical potential energy. The electrons closer to the nucleus are at lower energy levels, while those that are further out (in higher electron shells) have higher energy. The electrons that are farthest from the nucleus have the strongest interactions with the surrounding environment and the weakest interactions with the nucleus. These electrons are called valence electrons; they are much more likely to become involved in bonds with other atoms because they experience the least electrostatic pull from their own nucleus. Generally speaking, the valence electrons determine the reactivity of an atom. As we will discuss in Chapter 3 ofMCAT General Chemistry Review, the sharing or transferring of these valence electrons in bonds allows elements to fill their highest energy level to increase stability. In the neutral state, there are equal numbers of protons and electrons; losing electrons results in the atom gaining a positive charge, while gaining electrons results in the atom gaining a negative charge. A positively charged atom is called a cation, and a negatively charged atom is called an anion.
BRIDGE
Valence electrons will be very important to us in both general and organic chemistry. Knowing how tightly held those electrons are will allow us to understand many of an atom’s properties and how it interacts with other atoms, especially in bonding. Bonding is so important that it is discussed in Chapter 3 of both MCAT General Chemistry Review and MCAT Organic Chemistry Review.
Some basic features of the three subatomic particles are shown in Table 1.1.
Table 1.1. Subatomic Particles
SUBATOMIC PARTICLE SYMBOL RELATIVE MASS CHARGE LOCATION
Proton p, p+, or 11H 1 +1 Nucleus
Neutron n0 or 01n 1 0 Nucleus
Electron e− or −10e 0 −1 Orbitals
Example: Determine the number of protons, neutrons, and electrons in a nickel-58 atom and in a nickel-60 +2 cation.
Solution: 58Ni has an atomic number of 28 and a mass number of 58. Therefore, 58Ni will have 28 protons, 28 electrons, and 58 – 28, or 30, neutrons.
60Ni2+ has the same number of protons as the neutral 58Ni atom. However, 60Ni2+ has a positive charge because it has lost two electrons; thus, Ni2+ will have 26 electrons. Also, the mass number is two units higher than for the 58Ni atom, and this difference in mass must be due to two extra neutrons; thus, it has a total of 32 neutrons.
BRIDGE
Solutions to concept checks for a given chapter in MCAT General Chemistry Review can be found near the end of the chapter in which the concept check is located, following the Concept Summary for that chapter.
MCAT CONCEPT CHECK 1.1:
Before you move on, assess your understanding of the material with these questions.
-
Which subatomic particle is the most important for determining each of the following properties of an atom?
- Charge: ________________________________
- Atomic number: ________________________________
- Isotope: ____________________________
-
In nuclear medicine, isotopes are created and used for various purposes; for instance, 18O is created from 18F. Determine the number of protons, neutrons, and electrons in each of these species.
Particle Protons Neutrons Electrons 18O 18F
1.2 Atomic Mass *vs.* Atomic Weight
LEARNING OBJECTIVES
After Chapter 1.2, you will be able to:
- Describe atomic mass and atomic weight
- Recall the units of molar mass
- Predict the number of protons, neutrons, and electrons in a given isotope
There are a few different terms used by chemists to describe the heaviness of an element: atomic mass and mass number, which are essentially synonymous, and atomic weight. While the atomic weight is a constant for a given element and is reported in the periodic table, the atomic mass or mass number varies from one isotope to another. In this section, carefully compare and contrast the different definitions of these terms—because they are similar, they can be easy to mix up on the MCAT.
KEY CONCEPT
- Atomic number (Z) = number of protons
- Mass number (A) = number of protons + number of neutrons
- Number of protons = number of electrons (in a neutral atom)
- Electrons are not included in mass calculations because they are much smaller.
Atomic Mass
As we’ve seen, the mass of one proton is approximately one amu. The size of the atomic mass unit is defined as exactly 1 12 the mass of the carbon-12 atom, approximately 1.66 × 10−24 g. Because the carbon-12 nucleus has six protons and six neutrons, an amu is approximately equal to the mass of a proton or a neutron. The difference in mass between protons and neutrons is extremely small; in fact, it is approximately equal to the mass of an electron.
The atomic mass of an atom (in amu) is nearly equal to its mass number, the sum of protons and neutrons (in reality, some mass is lost as binding energy, as discussed in Chapter 9 of MCAT Physics and Math Review). Atoms of the same element with varying mass numbers are called isotopes (from the Greek for “same place”). Isotopes differ in their number of neutrons and are referred to by the name of the element followed by the mass number; for example, carbon-12 or iodine-131. Only the three isotopes of hydrogen, shown in Figure 1.3, are given unique names: protium (Greek: “first”) has one proton and an atomic mass of 1 amu; deuterium (“second”) has one proton and one neutron and an atomic mass of 2 amu; tritium (“third”) has one proton and two neutrons and an atomic mass of 3 amu. Because isotopes have the same number of protons and electrons, they generally exhibit similar chemical properties.
Atomic Weight
In nature, almost all elements exist as two or more isotopes, and these isotopes are usually present in the same proportions in any sample of a naturally occurring element. The weighted average of these different isotopes is referred to as the atomic weight and is the number reported on the periodic table. For example, chlorine has two main naturally occurring isotopes: chlorine-35 and chlorine-37. Chlorine-35 is about three times more abundant than chlorine-37; therefore, the atomic weight of chlorine is closer to 35 than 37. On the periodic table, it is listed as 35.5. Figure 1.4 illustrates the half-lives of the different isotopes of the elements; because half-life corresponds with stability, it also helps determine the relative proportions of these different isotopes.
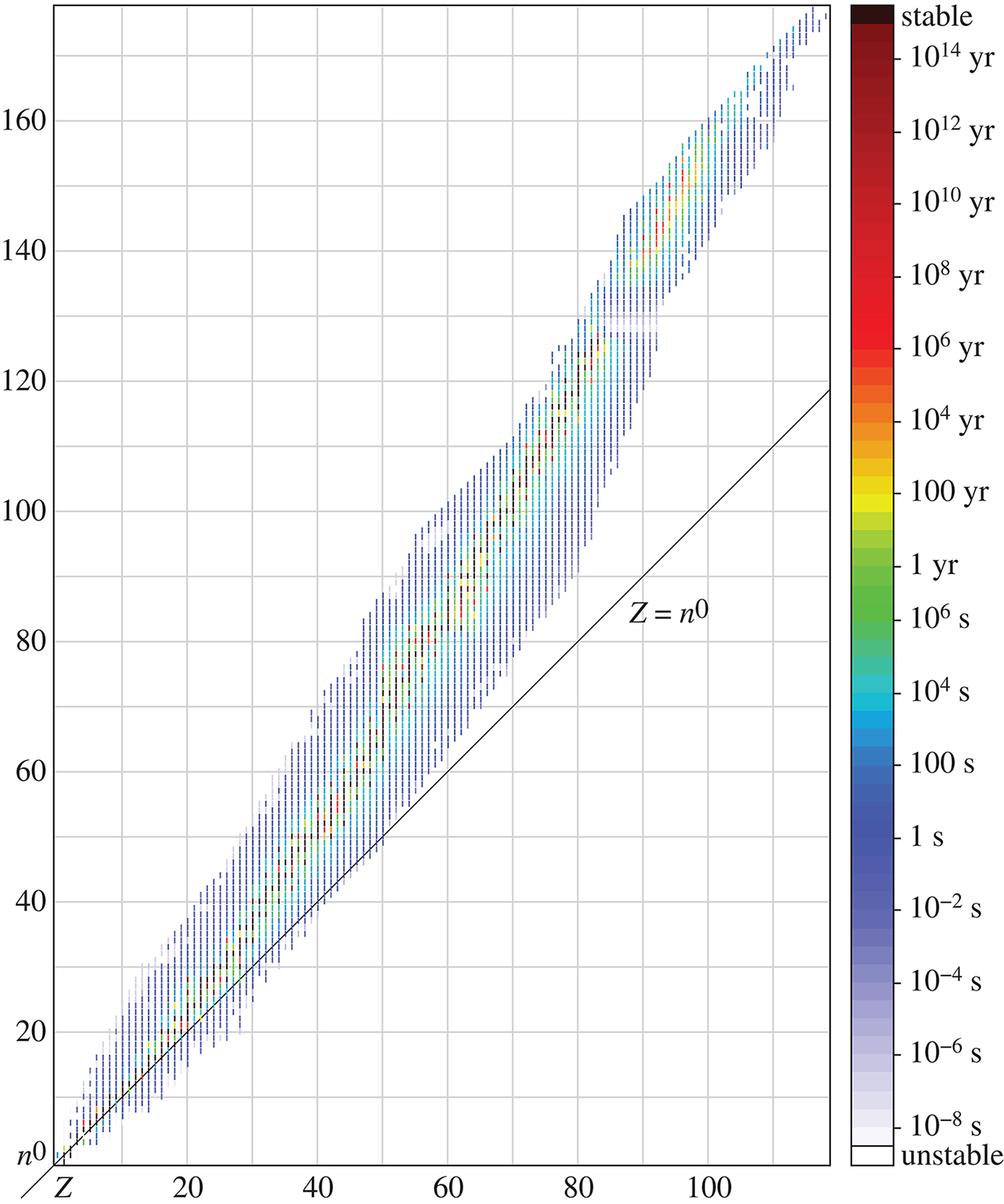
Figure 1.4. Half-Lives of the Different Isotopes of Elements Half-life is a marker of stability; generally, longer-lasting isotopes are more abundant.
KEY CONCEPT
When an element has two or more isotopes, no one isotope will have a mass exactly equal to the element’s atomic weight. Bromine, for example, is listed in the periodic table as having a mass of 79.9 amu. This is an average of the two naturally occurring isotopes, bromine-79 and bromine-81, which occur in almost equal proportions. There are no bromine atoms with an actual mass of 79.9 amu.
The utility of the atomic weight is that it represents both the mass of the “average” atom of that element, in amu, and the mass of one mole of the element, in grams. A mole is a number of “things” (atoms, ions, molecules) equal to Avogadro’s number, NA = 6.02 × 1023. For example, the atomic weight of carbon is 12.0 amu, which means that the average carbon atom has a mass of 12.0 amu (carbon-12 is far more abundant than carbon-13 or carbon-14), and 6.02 × 1023 carbon atoms have a combined mass of 12.0 grams.
MNEMONIC
Atomic mass is nearly synonymous with mass number. Atomic weight is a weighted average of naturally occurring isotopes of that element.
Example: Element Q consists of three different isotopes: A, B, and C. Isotope A has an atomic mass of 40 amu and accounts for 60 percent of naturally occurring Q. Isotope B has an atomic mass of 44 amu and accounts for 25 percent of Q. Finally, isotope C has an atomic mass of 41 amu and accounts for 15 percent of Q. What is the atomic weight of element Q?
Solution: The atomic weight is the weighted average of the naturally occurring isotopes of that element:
0.60 (40 amu) + 0.25 (44 amu) + 0.15 (41 amu) = 24.00 amu + 11.00 amu + 6.15 amu = 41.15 amu
MCAT CONCEPT CHECK 1.2:
Before you move on, assess your understanding of the material with these questions.
-
What are the definitions of atomic mass and atomic weight?
- Atomic mass: ___________________________________
- Atomic weight: ___________________________________
-
While molar mass is typically written in grams per mole ( g mol ) , is the ratio moles per gram ( mol g ) also acceptable?
_______________________________________
-
Calculate and compare the subatomic particles that make up the following atoms.
Isotope Protons Neutrons Electrons 19O 16O 17O 19F 16F 238U 240U
1.3 Rutherford, Planck, and Bohr
LEARNING OBJECTIVES
After Chapter 1.3, you will be able to:
- Calculate the energy of transition for a valence electron that jumps energy levels
- Calculate the wavelength of an emitted photon given the energy emitted by an electron
- Calculate the energy of a photon given its wavelength
MCAT EXPERTISE
The “High-Yield” badge on this section indicates that the content is frequently tested on the MCAT.
In 1910, Ernest Rutherford provided experimental evidence that an atom has a dense, positively charged nucleus that accounts for only a small portion of the atom’s volume. Eleven years earlier, Max Planck developed the first quantum theory, proposing that energy emitted as electromagnetic radiation from matter comes in discrete bundles called quanta. The energy of a quantum, he determined, is given by the Planck relation:
E = hf
Equation 1.1
where h is a proportionality constant known as Planck’s constant, equal to 6.626 × 10−34 J ·s, and f (sometimes designated by the Greek letter nu, ν) is the frequency of the radiation.
BRIDGE
Recall from Chapter 8 of MCAT Physics Review that the speed of light (or any wave) can be calculated using v = fλ. The speed of light, c, is 3 × 10 8 m s . This equation can be incorporated into the equation for quantum energy to provide different derivations.
Bohr Model
In 1913, Danish physicist Niels Bohr used the work of Rutherford and Planck to develop his model of the electronic structure of the hydrogen atom. Starting from Rutherford’s findings, Bohr assumed that the hydrogen atom consisted of a central proton around which an electron traveled in a circular orbit. He postulated that the centripetal force acting on the electron as it revolved around the nucleus was created by the electrostatic force between the positively charged proton and the negatively charged electron.
Bohr used Planck’s quantum theory to correct certain assumptions that classical physics made about the pathways of electrons. Classical mechanics postulates that an object revolving in a circle, such as an electron, may assume an infinite number of values for its radius and velocity. The angular momentum (L = mvr) and kinetic energy ( K = 1 2 m v 2 ) of the object could therefore take on any value. However, by incorporating Planck’s quantum theory into his model, Bohr placed restrictions on the possible values of the angular momentum. Bohr predicted that the possible values for the angular momentum of an electron orbiting a hydrogen nucleus could be given by:
L = n h 2 π
Equation 1.2
where n is the principal quantum number, which can be any positive integer, and h is Planck’s constant. Because the only variable is the principal quantum number, the angular momentum of an electron changes only in discrete amounts with respect to the principal quantum number. Note the similarities between quantized angular momentum and Planck’s concept of quantized energy.
MCAT EXPERTISE
When you see a formula in your review or on Test Day, focus on ratios and relationships. This simplifies our calculations to a conceptual understanding, which is usually enough to lead us to the right answer. Further, the MCAT tends to ask how changes in one variable may affect another variable, rather than a plug-and-chug application of complex equations.
Bohr then related the permitted angular momentum values to the energy of the electron to obtain:
E = − R H n 2
Equation 1.3
where RH is the experimentally determined Rydberg unit of energy, equal to 2.18 × 10 − 18 J electron . Therefore, like angular momentum, the energy of the electron changes in discrete amounts with respect to the quantum number. A value of zero energy was assigned to the state in which the proton and electron are separated completely, meaning that there is no attractive force between them. Therefore, the electron in any of its quantized states in the atom will have an attractive force toward the proton; this is represented by the negative sign in Equation 1.3. Ultimately, the only thing the energy equation is saying is that the energy of an electron increases—becomes less negative—the farther out from the nucleus that it is located (increasing n). This is an important point: while the magnitude of the fraction is getting smaller, the actual value it represents is getting larger (becoming less negative).
KEY CONCEPT
At first glance, it may not be clear that the energy (E) is directly proportional to the principal quantum number (n) in Equation 1.3. Take notice of the negative sign, which causes the values to approach zero from a more negative value as n increases (thereby increasing the energy). Negative signs are as important as a variable’s location in a fraction when it comes to determining proportionality.
Think of the concept of quantized energy as being similar to the change in gravitational potential energy that you experience when you ascend or descend a flight of stairs. Unlike a ramp, on which you could take an infinite number of steps associated with a continuum of potential energy changes, a staircase only allows you certain changes in height and, as a result, allows only certain discrete (quantized) changes of potential energy.
Bohr came to describe the structure of the hydrogen atom as a nucleus with one proton forming a dense core, around which a single electron revolved in a defined pathway (orbit) at a discrete energy value. If one could transfer an amount of energy exactly equal to the difference between one orbit and another, this could result in the electron “jumping” from one orbit to a higher-energy one. These orbits had increasing radii, and the orbit with the smallest, lowest-energy radius was defined as the ground state (n = 1). More generally, the ground state of an atom is the state of lowest energy, in which all electrons are in the lowest possible orbitals. In Bohr’s model, if the electron was promoted to an orbit with a larger radius (higher energy), the atom was said to be in the excited state. In general, an atom is in an excited state when at least one electron has moved to a subshell of higher than normal energy. Bohr likened his model of the hydrogen atom to the planets orbiting the sun, in which each planet traveled along a roughly circular pathway at set distances—and energy values—from the sun. Bohr’s Nobel Prize-winning model was reconsidered over the next two decades but remains an important conceptualization of atomic behavior. In particular, remember that we now know that electrons are not restricted to specific pathways, but tend to be localized in certain regions of space.
MCAT EXPERTISE
Note that all systems tend toward minimal energy; thus on the MCAT, atoms of any element will generally exist in the ground state unless subjected to extremely high temperatures or irradiation.
Applications of the Bohr Model
The Bohr model of the hydrogen atom (and other one-electron systems, such as He+ and Li2+) is useful for explaining the atomic emission and absorption spectra of atoms.
MNEMONIC
As electrons go from a lower energy level to a higher energy level, they get AHED:
- Absorb light
- Higher potential
- Excited
- Distant (from the nucleus)
Atomic Emission Spectra
At room temperature, the majority of atoms in a sample are in the ground state. However, electrons can be excited to higher energy levels by heat or other energy forms to yield excited states. Because the lifetime of an excited state is brief, the electrons will return rapidly to the ground state, resulting in the emission of discrete amounts of energy in the form of photons, as shown in Figure 1.5.
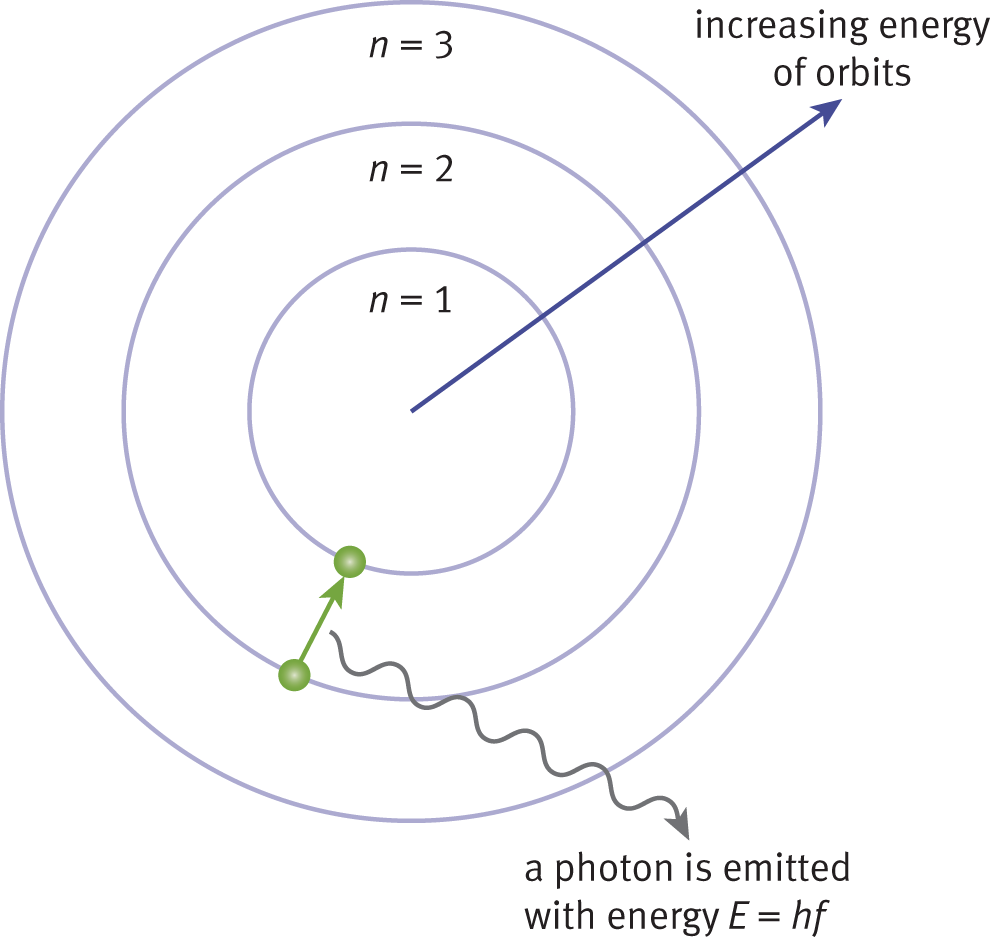
Figure 1.5. Atomic Emission of a Photon as a Result of a Ground State Transition
The electromagnetic energy of these photons can be determined using the following equation:
E = h c λ
Equation 1.4
where h is Planck’s constant, c is the speed of light in a vacuum ( 3.00 × 10 8 m s ) , and λ is the wavelength of the radiation. Note that Equation 1.4 is just a combination of two other equations: E = hf and c = f λ.
The electrons in an atom can be excited to different energy levels. When these electrons return to their ground states, each will emit a photon with a wavelength characteristic of the specific energy transition it undergoes. As described above, these energy transitions do not form a continuum, but rather are quantized to certain values. Thus, the spectrum is composed of light at specified frequencies. It is sometimes called a line spectrum, where each line on the emission spectrum corresponds to a specific electron transition. Because each element can have its electrons excited to a different set of distinct energy levels, each possesses a unique atomic emission spectrum, which can be used as a fingerprint for the element. One particular application of atomic emission spectroscopy is in the analysis of stars and planets: while a physical sample may be impossible to procure, the light from a star can be resolved into its component wavelengths, which are then matched to the known line spectra of the elements as shown in Figure 1.6.
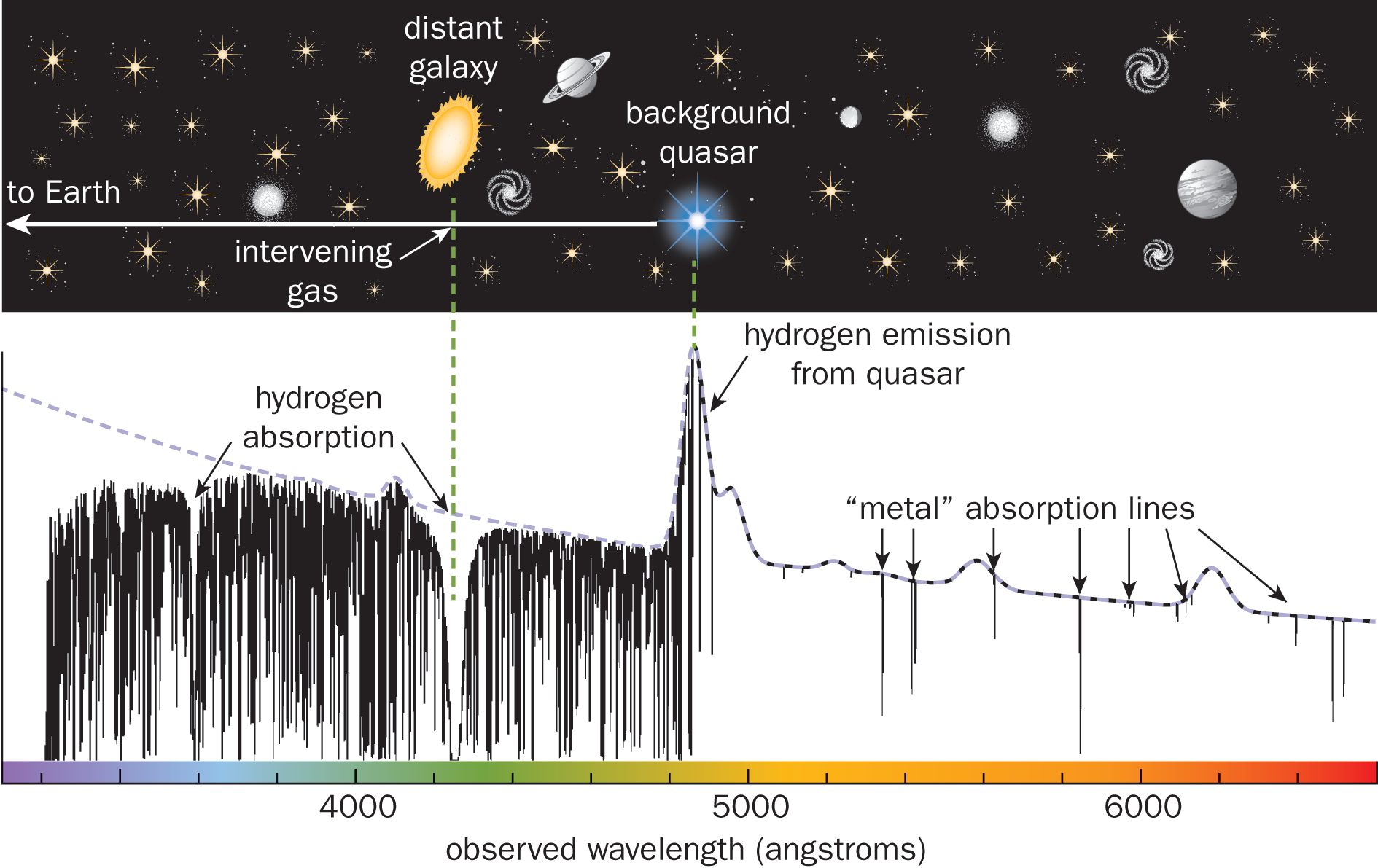
Figure 1.6. Line Spectrum with Transition Wavelengths for Various Celestial Bodies
REAL WORLD
Emissions from electrons dropping from an excited state to a ground state give rise to fluorescence. What we see is the color of the emitted light.
The Bohr model of the hydrogen atom explained the atomic emission spectrum of hydrogen, which is the simplest emission spectrum among all the elements. The group of hydrogen emission lines corresponding to transitions from energy levels n ≥ 2 to n = 1 is known as the Lyman series. The group corresponding to transitions from energy levels n ≥ 3 to n = 2 is known as the Balmer series and includes four wavelengths in the visible region. The Lyman series includes larger energy transitions than the Balmer series; it therefore has shorter photon wavelengths in the UV region of the electromagnetic spectrum. The Paschen series corresponds to transitions from n ≥ 4 to n = 3. These energy transition series can be seen in Figure 1.7.
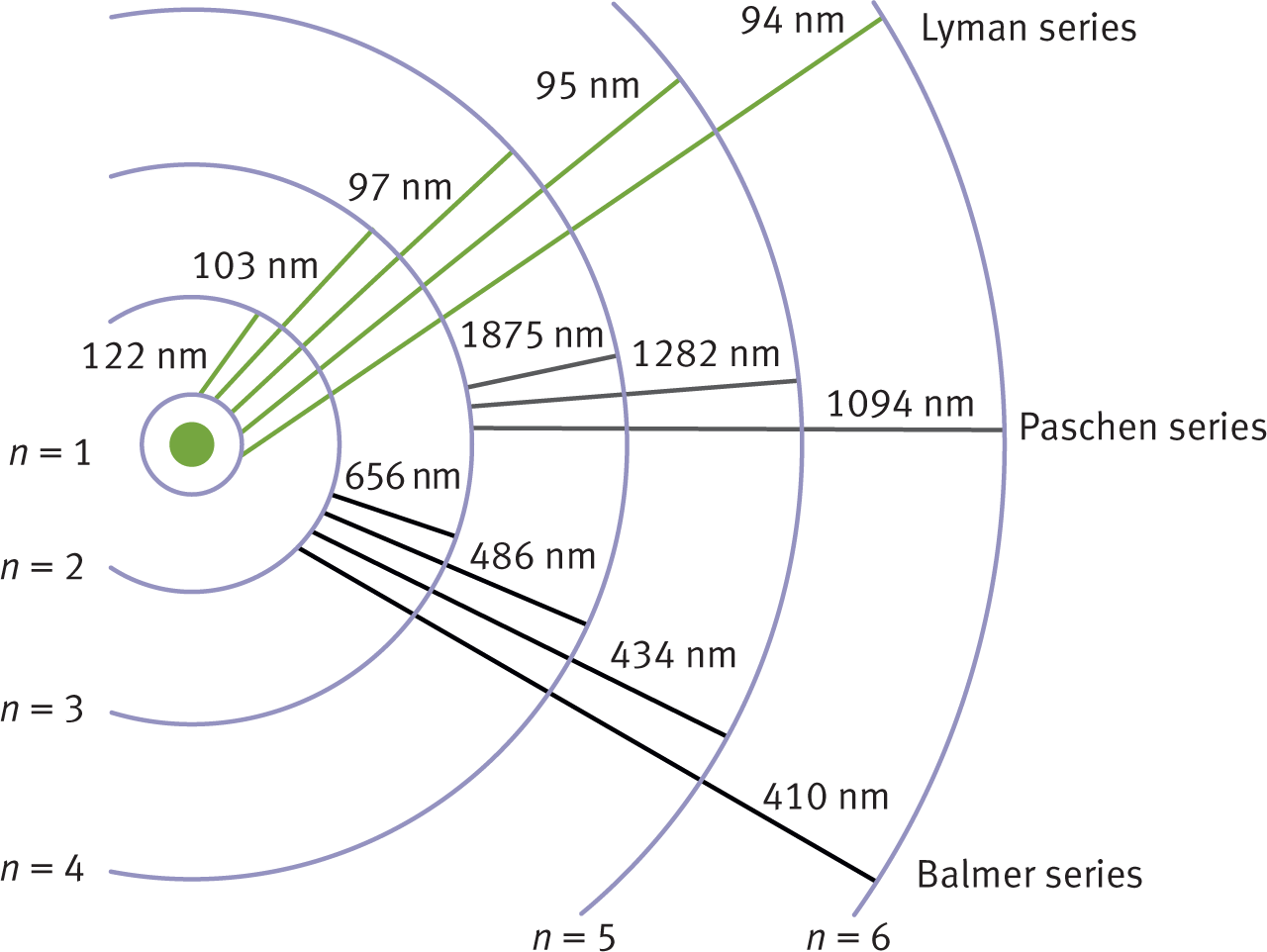
Figure 1.7. Wavelengths of Electron Orbital Transitions Energy is inversely proportional to wavelength: E = h f = h c λ .
The energy associated with a change in the principal quantum number from a higher initial value ni to a lower final value nf is equal to the energy of the photon predicted by Planck’s quantum theory. Combining Bohr’s and Planck’s calculations, we can derive:
E = h c λ = R H [ 1 n i 2 − 1 n f 2 ]
Equation 1.5
This complex-appearing equation essentially says: The energy of the emitted photon corresponds to the difference in energy between the higher-energy initial state and the lower-energy final state.
KEY CONCEPT
It may seem strange to see an equation that has initial minus final, where most equations usually have final minus initial. But ultimately, this equation is designed to work just like you'd expect: If an atom emits a photon, the equation gives a negative value for energy, indicating a decrease. You can easily check this for yourself by using ni= 2 and nf= 1.
Atomic Absorption Spectra
When an electron is excited to a higher energy level, it must absorb exactly the right amount of energy to make that transition. This means that exciting the electrons of a particular element results in energy absorption at specific wavelengths. Thus, in addition to a unique emission spectrum, every element possesses a characteristic absorption spectrum. Not surprisingly, the wavelengths of absorption correspond exactly to the wavelengths of emission because the difference in energy between levels remains unchanged. Identification of elements in the gas phase requires absorption spectra.
BRIDGE
ΔE is the same for absorption or emission between any two energy levels according to the conservation of energy, as discussed in Chapter 2 of MCAT Physics and Math Review. This is also the same as the energy of the photon of light absorbed or emitted.
Atomic emission and absorption spectra are complex topics, but the takeaway is that each element has a characteristic set of energy levels. For electrons to move from a lower energy level to a higher energy level, they must absorb the right amount of energy to do so. They absorb this energy in the form of light. Similarly, when electrons move from a higher energy level to a lower energy level, they emit the same amount of energy in the form of light.
REAL WORLD
Absorption is the basis for the color of compounds. We see the color of the light that is not absorbed by the compound.
MCAT CONCEPT CHECK 1.3:
Before you move on, assess your understanding of the material with these questions.
Note: For these questions, try to estimate the calculations without a calculator to mimic Test Day conditions. Double-check your answers with a calculator and refer to the answers for confirmation of your results.
-
The valence electron in a lithium atom jumps from energy level n = 2 to n = 4. What is the energy of this transition in joules? In eV? (Note: R H = 2.18 × 10 − 18 J electron = 13.6 eV electron )
_______________________________________
-
If an electron emits 3 eV of energy, what is the corresponding wavelength of the emitted photon? (Note: 1 eV = 1.60 × 10−19 J, h = 6.626 × 10−34 J· s)
_______________________________________
-
Calculate the energy of a photon of wavelength 662 nm. (Note: h = 6.626 × 10−34 J· s)
_______________________________________
1.4 Quantum Mechanical Model of Atoms
LEARNING OBJECTIVES
After Chapter 1.4, you will be able to:
- Identify the four quantum numbers, the potential range of values for each, and their relationship to the electron they represent
- Compare the orbital diagram for a neutral atom, such as sulfur (S), to an ion such as S2–
- Differentiate between paramagnetic and diamagnetic compounds
- Determine the number of valence electrons in a given atom
While Bohr’s model marked a significant advancement in the understanding of the structure of atoms, his model ultimately proved inadequate to explain the structure and behavior of atoms containing more than one electron. The model’s failure was a result of not taking into account the repulsion between multiple electrons surrounding the nucleus. Modern quantum mechanics has led to a more rigorous and generalizable study of the electronic structure of atoms. The most important difference between Bohr’s model and the modern quantum mechanical model is that Bohr postulated that electrons follow a clearly defined circular pathway or orbit at a fixed distance from the nucleus, whereas modern quantum mechanics has shown that this is not the case. Rather, we now understand that electrons move rapidly and are localized within regions of space around the nucleus called orbitals. The confidence by which those in Bohr’s time believed they could identify the location (or pathway) of the electron was now replaced by a more modest suggestion that the best we can do is describe the probability of finding an electron within a given region of space surrounding the nucleus. In the current quantum mechanical model, it is impossible to pinpoint exactly where an electron is at any given moment in time. This is expressed best by the Heisenberg uncertainty principle: It is impossible to simultaneously determine, with perfect accuracy, the momentum and the position of an electron. If we want to assess the position of an electron, the electron has to stop (thereby removing its momentum); if we want to assess its momentum, the electron has to be moving (thereby changing its position). This can be seen visually in Figure 1.8.
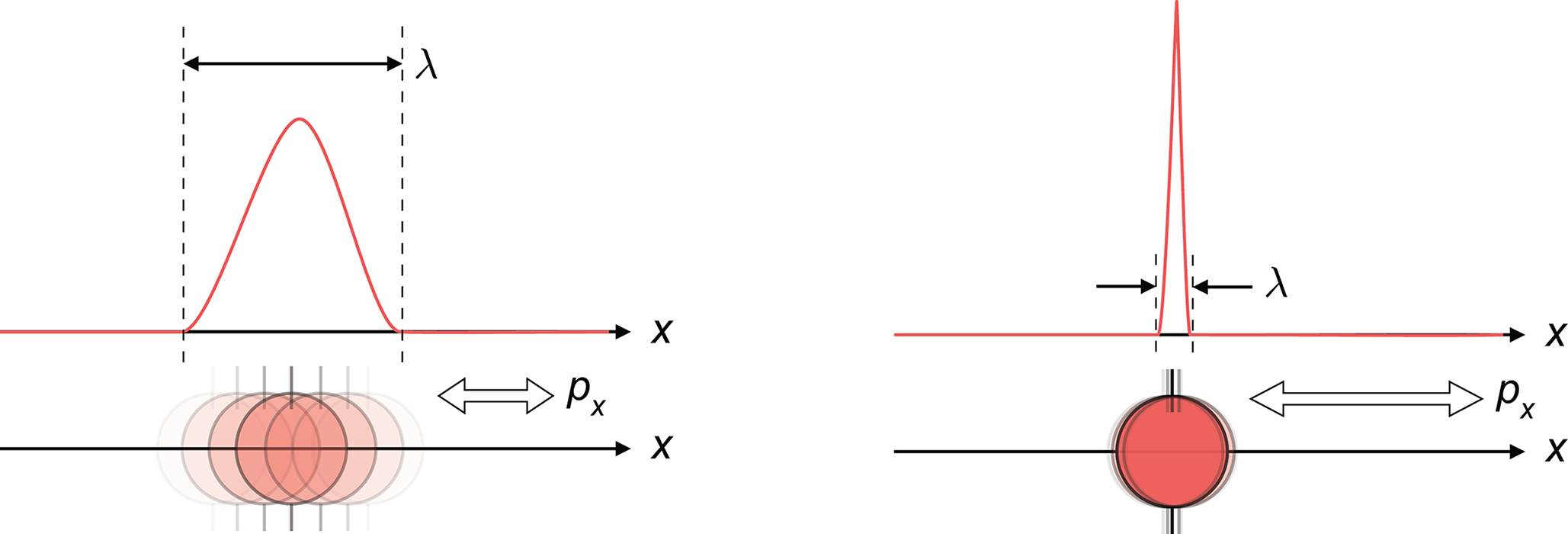
Figure 1.8. Heisenberg Uncertainty Principle Known momentum and uncertain position (left); known position but uncertain momentum (right).λ= confidence interval of position;px= confidence interval of momentum.
Quantum Numbers
Modern atomic theory postulates that any electron in an atom can be completely described by four quantum numbers: n, l, ml, and ms. Furthermore, according to the Pauli exclusion principle, no two electrons in a given atom can possess the same set of four quantum numbers. The position and energy of an electron described by its quantum numbers are known as its energy state. The value of n limits the values of l, which in turn limit the values of ml. In other words, for a given value of n, only particular values of l are permissible; given a value of l, only particular values of ml are permissible. The values of the quantum numbers qualitatively give information about the size, shape, and orientation of the orbitals. As we examine the four quantum numbers more closely, pay attention especially to l and ml because these two tend to give students the greatest difficulty.
MCAT EXPERTISE
Think of the quantum numbers as becoming more specific as one goes from n to l to ml to *m**s. This is like an address: one lives in a particular state (n), in a particular city (l), on a particular street (ml), at a particular house number (m**s*).
Principal Quantum Number (*n*)
The first quantum number is commonly known as the principal quantum number and is denoted by the letter n. This is the quantum number used in Bohr’s model that can theoretically take on any positive integer value. The larger the integer value of n, the higher the energy level and radius of the electron’s shell. Within each shell, there is a capacity to hold a certain number of electrons, given by:
Maximum number of electrons within a shell = 2n2
Equation 1.6
where n is the principal quantum number. The difference in energy between two shells decreases as the distance from the nucleus increases because the energy difference is a function of [ 1 n i 2 − 1 n f 2 ] . For example, the energy difference between the n = 3 and the n = 4 shells ( 1 9 − 1 16 ) is less than the energy difference between the n = 1 and the n = 2 shells ( 1 1 − 1 4 ) . This can be seen in Figure 1.7. Remember that electrons do not travel in precisely defined orbits; it just simplifies the visual representation of the electrons’ motion.
BRIDGE
Remember, a larger integer value for the principal quantum number indicates a larger radius and higher energy. This is similar to gravitational potential energy, as discussed in Chapter 2 of MCAT Physics Review, where the higher or farther the object is above the Earth, the higher its potential energy will be.
Azimuthal Quantum Number (*l*)
The second quantum number is called theazimuthal (angular momentum) quantum number and is designated by the letter l. The second quantum number refers to the shape and number of subshells within a given principal energy level (shell). The azimuthal quantum number is very important because it has important implications for chemical bonding and bond angles. The value of n limits the value of l in the following way: for any given value of n, the range of possible values for l is 0 to (n – 1). For example, within the first principal energy level, n = 1, the only possible value for l is 0; within the second principal energy level, n = 2, the possible values for l are 0 and 1. A simpler way to remember this relationship is that the n-value also tells you the number of possible subshells. Therefore, there’s only one subshell (l = 0) in the first principal energy level; there are two subshells (l = 0 and 1) within the second principal energy level; there are three subshells (l = 0, 1, and 2) within the third principal energy level, and so on.
KEY CONCEPT
For any principal quantum number n, there will be n possible values for l, ranging from 0 to (n – 1).
Spectroscopic notation refers to the shorthand representation of the principal and azimuthal quantum numbers. The principal quantum number remains a number, but the azimuthal quantum number is designated by a letter: the l = 0 subshell is called s; the l = 1 subshell is called p; the l = 2 subshell is called d; and the l = 3 subshell is called f. Thus, an electron in the shell n = 4 and subshell l = 2 is said to be in the 4d subshell. The spectroscopic notation for each subshell is demonstrated in Figure 1.9.
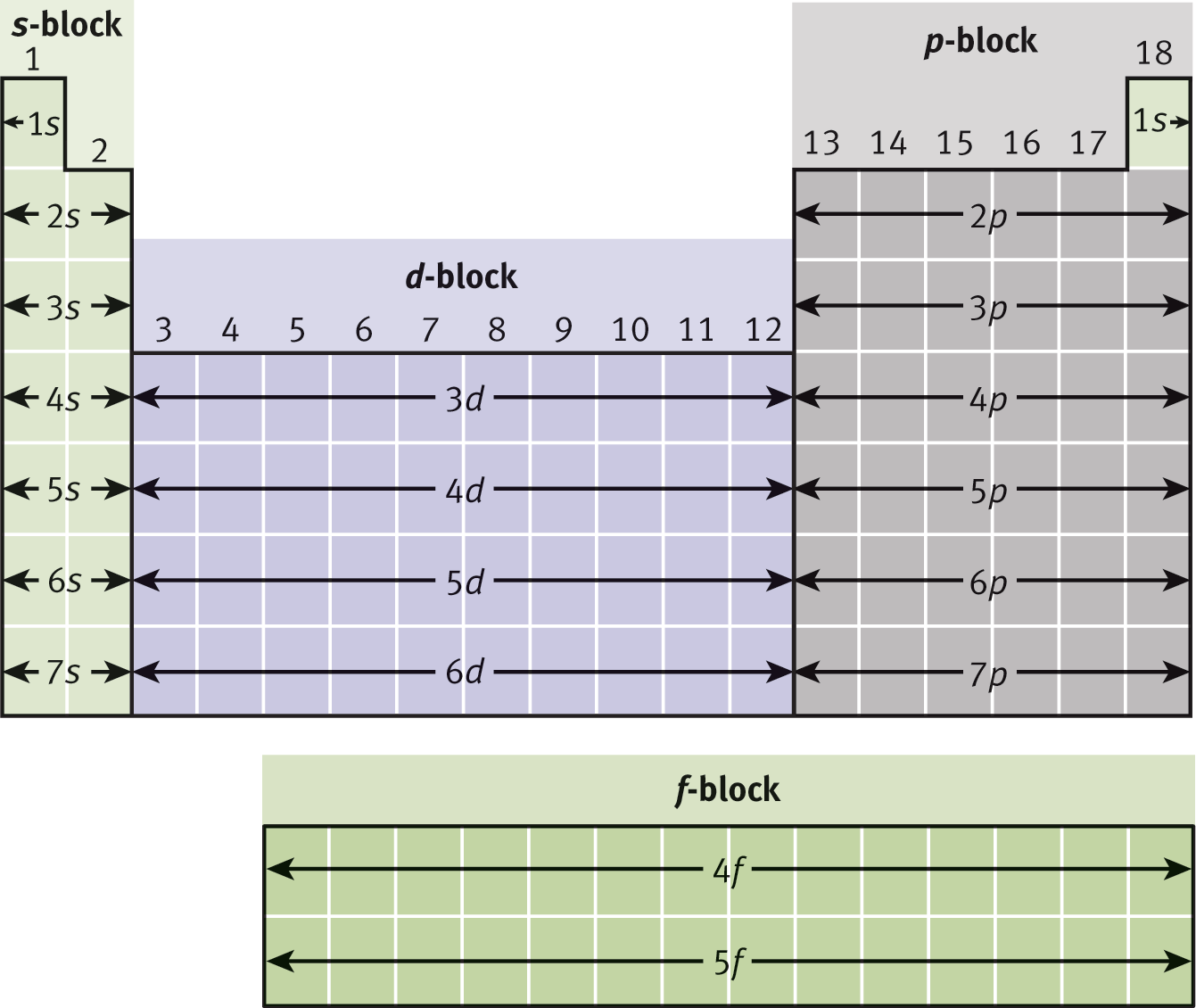
Figure 1.9. Spectroscopic Notation for Every Subshell on the Periodic Table
Within each subshell, there is a capacity to hold a certain number of electrons, given by:
Maximum number of electrons within a subshell = 4l + 2
Equation 1.7
where l is the azimuthal quantum number. The energies of the subshells increase with increasing l value; however, the energies of subshells from different principal energy levels may overlap. For example, the 4s subshell will have a lower energy than the 3d subshell.
Figure 1.10 provides an example of computer-generated probability maps of the first few electron clouds in a hydrogen atom. This provides a rough visual representation of the shapes of different subshells.
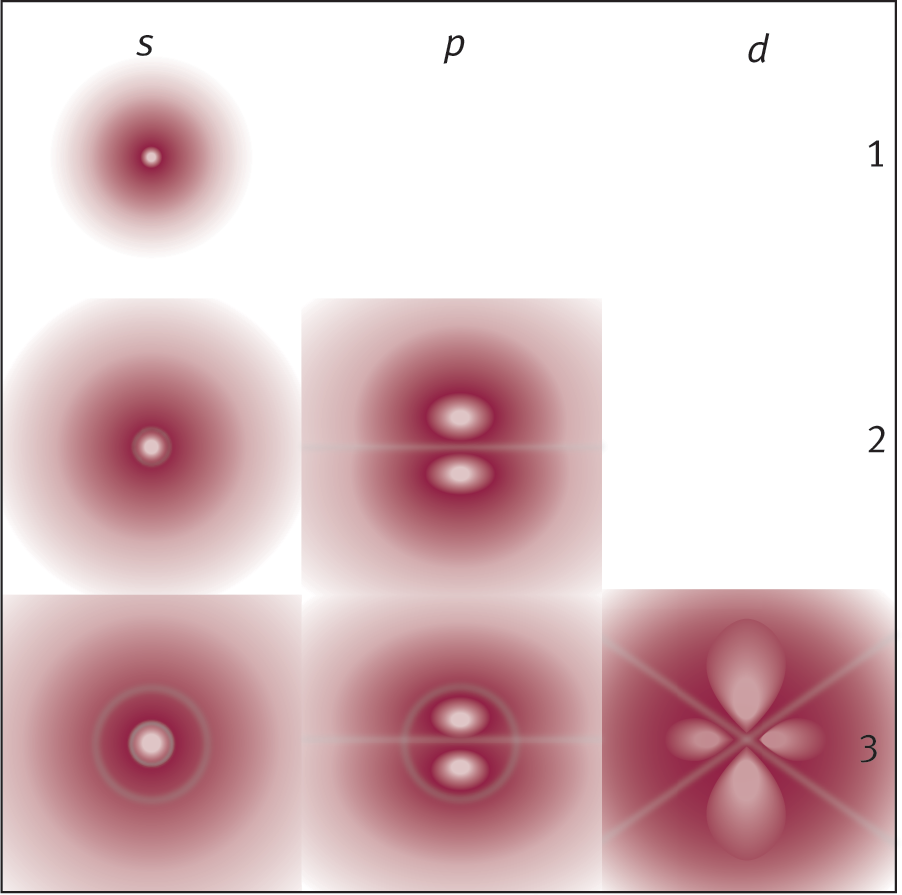
Figure 1.10. Electron Clouds of Various Subshells
Magnetic Quantum Number (*ml*)
The third quantum number is the magnetic quantum number and is designated *m**l. The magnetic quantum number specifies the particular orbital within a subshell where an electron is most likely to be found at a given moment in time. Each orbital can hold a maximum of two electrons. The possible values ofmlare the integers between –land +l, including 0. For example, thessubshell, withl= 0, limits the possiblemlvalues to 0, and because there is a single value ofml, there is only one orbital in thessubshell. Thepsubshell, withl= 1, limits the possiblemlvalues to −1, 0, and +1, and because there are three values forml, there are three orbitals in thepsubshell. Thedsubshell has five orbitals (−2 to +2), and thefsubshell has seven orbitals (−3 to +3). The shape of the orbitals, like the number of orbitals, is dependent on the subshell in which they are found. The orbitals in thessubshell are spherical, while the three orbitals in thepsubshell are each dumbbell-shaped and align along thex-, y-,andz-axes. In fact, thep-orbitals are often referred to asp**x,p**y, andp**z. The first five orbitals—1s, 2s, 2p**x, 2p**y, and 2p**z*—are demonstrated in Figure 1.11. Note the similarity to the images in Figure 1.10.
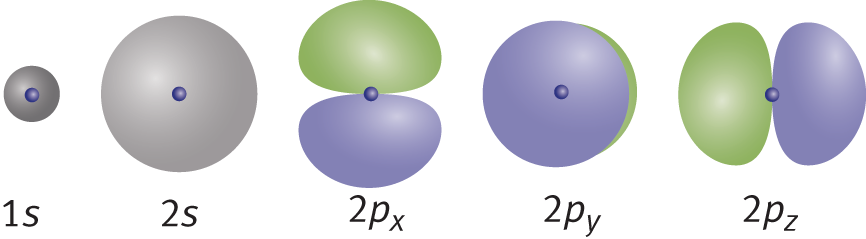
Figure 1.11. The First Five Atomic Orbitals
KEY CONCEPT
For any value of l, there will be 2l + 1 possible values for ml. For any n, this produces n2 orbitals. For any value of n, there will be a maximum of 2n2 electrons (two per orbital).
The shapes of the orbitals in the d and f subshells are much more complex, and the MCAT will not expect you to answer questions about their appearance. The shapes of orbitals are defined in terms of a concept called probability density, the likelihood that an electron will be found in a particular region of space.
Take a look at the 2p block in the periodic table. As mentioned above, 2p contains three orbitals. If each orbital can contain two electrons, then six electrons can be added during the course of filling the 2p-orbitals. As atomic number increases, so too does the number of electrons (assuming the species is neutral). Therefore, it should be no surprise that the p block contains six groups of elements. The s block contains two elements in each row of the periodic table, the d block contains ten elements, and the f block contains fourteen elements.
Spin Quantum Number (*ms*)
The fourth quantum number is called the spin quantum number and is denoted by *m**s. In classical mechanics, an object spinning about its axis has an infinite number of possible values for its angular momentum. However, this does not apply to the electron, which has two spin orientations designated + 1 2 and − 1 2 . Whenever two electrons are in the same orbital, they must have opposite spins. In this case, they are often referred to as being paired. Electrons in different orbitals with the samem**s* values are said to have parallel spins.
The quantum numbers for the orbitals in the second principal energy level, with their maximum number of electrons noted in parentheses, are shown in Table 1.2.
Table 1.2. Quantum Numbers for the Second Principal Energy Level
**n**
2 (8)
l 0 (2)
1 (6)
ml 0 (2) +1 (2) 0 (2) −1(2)
ms + 1 2 , − 1 2 + 1 2 , − 1 2 + 1 2 , − 1 2 + 1 2 , − 1 2
Electron Configurations
For a given atom or ion, the pattern by which subshells are filled, as well as the number of electrons within each principal energy level and subshell, are designated by its electron configuration. Electron configurations use spectroscopic notation, wherein the first number denotes the principal energy level, the letter designates the subshell, and the superscript gives the number of electrons in that subshell. For example, 2p4 indicates that there are four electrons in the second (p) subshell of the second principal energy level. This also implies that the energy levels below 2p (that is, 1s and 2s) have already been filled, as shown in Figure 1.12.
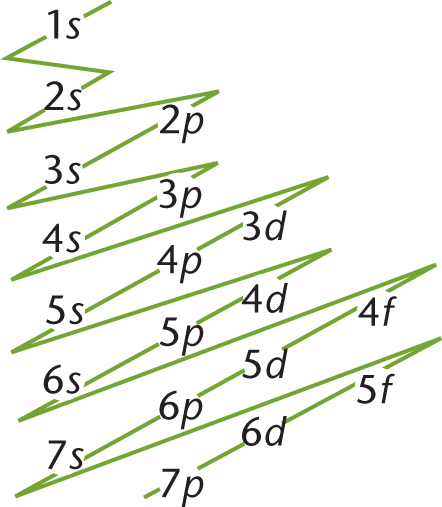
Figure 1.12. Electron Subshell Flow Diagram
MCAT EXPERTISE
Remember that the shorthand used to describe the electron configuration is derived directly from the quantum numbers.
To write out an atom’s electron configuration, one needs to know the order in which subshells are filled. Electrons fill from lower- to higher-energy subshells, according to the Aufbau principle (also called the building-up principle), and each subshell will fill completely before electrons begin to enter the next one. The order need not be memorized because there are two very helpful ways of recalling this. The ***n+l* rule** can be used to rank subshells by increasing energy. This rule states that the lower the sum of the values of the first and second quantum numbers, n + l, the lower the energy of the subshell. This is a helpful rule to remember for Test Day. If two subshells possess the same n + l value, the subshell with the lower n value has a lower energy and will fill with electrons first.
Example: Which will fill first, the 5d subshell or the 6s subshell?
Solution: For 5d, n = 5 and l = 2, so n + l = 7. For 6s, n = 6 and l = 0, so n + l = 6. Therefore, the 6s subshell has lower energy and will fill first.
An alternative way to approach electron configurations is through simply reading the periodic table. One must remember that the lowest s subshell is 1s, the lowest p subshell is 2p, the lowest d subshell is 3d, and the lowest f subshell is 4f. This can be seen in Figure 1.9. Then, we can simply read across the periodic table to get to the element of interest, filling subshells along the way. To do this, we must know the correct position of the lanthanide and actinide series (the f block), as shown in Figure 1.13. In most representations of the periodic table, the f block is pulled out and placed below the rest of the table. This is purely an effect of graphic design—placing the f block in its correct location results in a lot of excess white space on a page.
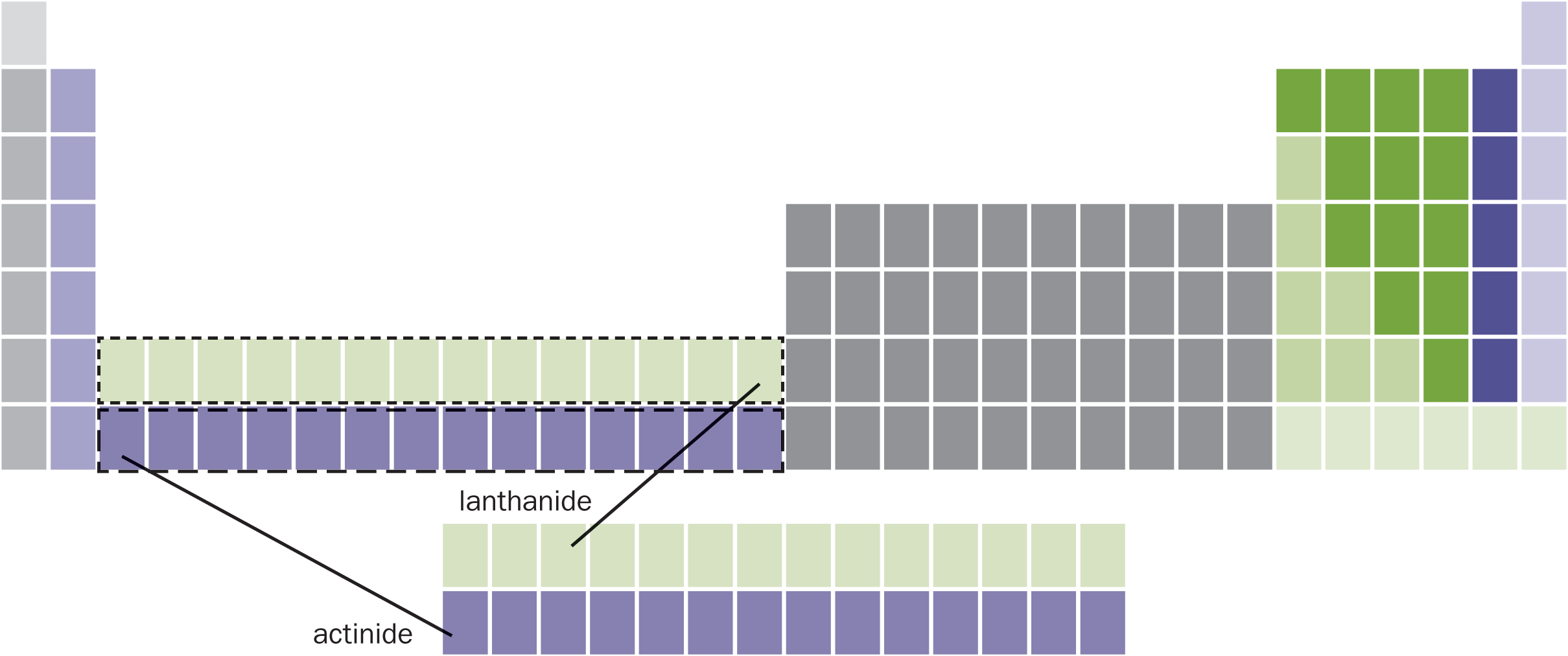
Figure 1.13. Periodic Table with Lanthanide and Actinide Series Inserted Thefblock fits between thesblock andd block in the periodic table.
MCAT EXPERTISE
Many general chemistry courses teach the flow diagram in Figure 1.12 as a method to determine the order of subshell filling in electron configurations. However, on Test Day, it can be both time-consuming and error-prone, resulting in incorrect electron configurations. Learning to read the periodic table, as described here, is the best method.
Electron configurations can be abbreviated by placing the noble gas that precedes the element of interest in brackets. For example, the electron configuration of any element in period four (starting with potassium) can be abbreviated by starting with [Ar].
Example: What is the electron configuration of osmium (Z = 76)?
Solution: The noble gas that comes just before osmium is xenon (Z = 54). Therefore, the electron configuration can begin with [Xe]. Continuing across the periodic table, we pass through the 6s subshell (cesium and barium), the 4f subshell (the lanthanide series; remember its position on the periodic table!), and into the 5d subshell. Osmium is the sixth element in the 5d subshell, so the configuration is [Xe] 6s24f145d6.
This method works for neutral atoms, but how does one write the electron configuration of an ion? Negatively charged ions (anions) have additional electrons that fill according to the same rules as above; for example, if fluorine’s electron configuration is [He] 2s22p5, then F– is [He] 2s22p6. Positively charged ions (cations) are a bit more complicated: start with the neutral atom, and remove electrons from the subshells with the highest value for n first. If multiple subshells are tied for the highest n value, then electrons are removed from the subshell with the highest l value among these.
Example: What is the electron configuration of Fe3+?
Solution: The electron configuration of iron is [Ar] 4s23d6. Electrons are removed from the 4s subshell before the 3d subshell because it has a higher principal quantum number. Therefore, Fe3+ has a configuration of [Ar] 3d5, not [Ar] 4s23d3.
Hund’s Rule
In subshells that contain more than one orbital, such as the 2p subshell with its three orbitals, the orbitals will fill according to Hund’s rule, which states that, within a given subshell, orbitals are filled such that there are a maximum number of half-filled orbitals with parallel spins. Like finding a seat on a crowded bus, electrons would prefer to have their own seat (orbital) before being forced to double up with another electron. Of course, the basis for this preference is electron repulsion: electrons in the same orbital tend to be closer to each other and thus repel each other more than electrons placed in different orbitals.
Example: According to Hund’s rule, what are the orbital diagrams for nitrogen and iron?
Solution: Nitrogen has an atomic number of 7. Thus, its electron configuration is 1s22s22p3. According to Hund’s rule, the two s-orbitals will fill completely, while the three p-orbitals will each contain one electron, all with parallel spins.
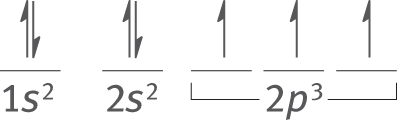
Iron has an atomic number of 26. As determined earlier, its electron configuration is [Ar] 4s23d6. The electrons will fill all of the subshells except for the 3d, which will contain four orbitals with parallel (upward) spin and one orbital with electrons of both spin directions.
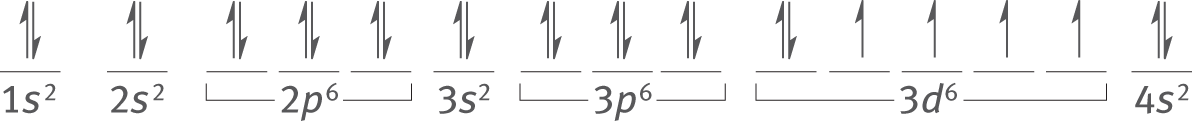
Subshells may be listed either in the order in which they fill (4s before 3d) or with subshells of the same principal quantum number grouped together, as shown here. Both methods are correct.
An important corollary from Hund’s rule is that half-filled and fully filled orbitals have lower energies (higher stability) than other states. This creates two notable exceptions to electron configuration that are often tested on the MCAT: chromium (and other elements in its group) and copper (and other elements in its group). Chromium (Z = 24) should have the electron configuration [Ar] 4s23d4 according to the rules established earlier. However, moving one electron from the 4s subshell to the 3d subshell allows the 3d subshell to be half-filled: [Ar] 4s13d5 (remember that s subshells can hold two electrons and d subshells can hold ten). While moving the 4s electron up to the 3d-orbital is energetically unfavorable, the extra stability from making the 3d subshell half-filled outweighs that cost. Similarly, copper (Z = 29) has the electron configuration [Ar] 4s13d10, rather than [Ar] 4s23d9; a full d subshell outweighs the cost of moving an electron out of the 4s subshell. Other elements in the same group have similar behavior, moving one electron from the highest s subshell to the highest d subshell. Similar shifts can be seen with f subshells, but they are never observed for the p subshell; the extra stability doesn’t outweigh the cost.
The presence of paired or unpaired electrons affects the chemical and magnetic properties of an atom or molecule. Materials composed of atoms with unpaired electrons will orient their spins in alignment with a magnetic field, and the material will thus be weakly attracted to the magnetic field. These materials are considered paramagnetic. An example is shown in Figure 1.14 where a set of iron orbs is influenced by a magnet. The metallic spheres that are close enough to be induced by the magnet are attracted to the magnet and move toward it.

Figure 1.14. Attraction of Paramagnetic Iron Spheres to a Magnet
MNEMONIC
Remember that paramagnetic means that a magnetic field will cause parallel spins in unpaired electrons and therefore cause an attraction.
Materials consisting of atoms that have only paired electrons will be slightly repelled by a magnetic field and are said to bediamagnetic. In Figure 1.15, a piece of pyrolytic graphite is suspended in the air over strong neodymium magnets. All the electrons in this allotrope (configuration) of carbon are paired because of covalent bonding between layers of the material, and are thus opposed to being reoriented. Given sufficiently strong magnetic fields beneath an object, any diamagnetic substance can be made to levitate.

Figure 1.15. Diamagnetic Pyrolytic Graphite
REAL WORLD
The concept behind “maglev” or magnetic levitation is no longer science fiction. Using powerful magnetic fields and strongly diamagnetic materials, some transportation systems have developed frictionless, high speed rail networks such as Japan’s SCMaglev.
Valence Electrons
The valence electrons of an atom are those electrons that are in its outermost energy shell, are most easily removed, and are available for bonding. In other words, the valence electrons are the “active” electrons of an atom and to a large extent dominate the chemical behavior of the atom. For elements in Groups IA and IIA (Groups 1 and 2), only the highest s subshell electrons are valence electrons. For elements in Groups IIIA through VIIIA (Groups 13 through 18), the highest s and p subshell electrons are valence electrons. For transition elements, the valence electrons are those in the highest s and d subshells, even though they have different principal quantum numbers. For the lanthanide and actinide series, the valence electrons are those in the highest s and f subshells, even though they have different principal quantum numbers. All elements in period three (starting with sodium) and below may accept electrons into their d subshell, which allows them to hold more than eight electrons in their valence shell. This allows them to violate the octet rule, as discussed in Chapter 3 of MCAT General Chemistry Review.
MCAT EXPERTISE
The valence electron configuration of an atom helps us understand its properties and is ascertainable from the periodic table (the only “cheat sheet” available on the MCAT!). On Test Day, you will be able to access a periodic table by clicking on the button labeled “Periodic Table” on the bottom left of the screen. Use it as needed!
Example: Which electrons are the valence electrons of elemental vanadium, elemental selenium, and the sulfur atom in a sulfate ion?
Solution: Vanadium has five valence electrons: two in its 4s subshell and three in its 3d subshell.
Selenium has six valence electrons: two in its 4s subshell and four in its 4p subshell. Selenium’s 3d electrons are not part of its valence shell.
Sulfur in a sulfate ion has 12 valence electrons: its original six plus six more from the oxygens to which it is bonded. Sulfur’s 3s and 3p subshells can contain only eight of these 12 electrons; the other four electrons have entered the sulfur atom’s 3d subshell, which is normally empty in elemental sulfur.
MCAT CONCEPT CHECK 1.4:
Before you move on, assess your understanding of the material with these questions.
-
If given the following quantum numbers, which element(s) do they likely refer to? (Note: Assume that these quantum numbers describe the valence electrons in the element.)
***n l* Possible Elements 2 1 3 0 5 3 4 2**
-
Write out and compare an orbital diagram for a neutral oxygen (O) atom and an O2– ion.
_______________________________________
-
Magnetic resonance angiography (MRA) is a technique that can resolve defects like stenotic (narrowed) arteries. A contrast agent like gadolinium or manganese injected into the blood stream interacts with the strong magnetic fields of the MRI device to produce such images. Based on their orbital configurations, are these contrast agents paramagnetic or diamagnetic?
_______________________________________
-
Determine how many valence electrons come from each subshell in the following atoms:
**Atom s-electrons p-electrons d-electrons f-electrons Total Valence Electrons P in PO43− O in PO43− Ir Cf**
Conclusion
Congratulations! You’ve made it through the first chapter! Now that we have covered topics related to the most fundamental unit of matter—the atom—you’re set to advance your understanding of the physical world in more complex ways. This chapter described the characteristics and behavior of the three subatomic particles: the proton, neutron, and electron. In addition, it compared and contrasted two models of the atom. The Bohr model is adequate for describing the structure of one-electron systems, such as the hydrogen atom or the helium ion, but fails to adequately describe the structure of more complex atoms. The quantum mechanical model theorizes that electrons are found not in discrete orbits, but in “clouds of probability,” or orbitals, by which we can predict the likelihood of finding electrons within given regions of space surrounding the nucleus. Both theories tell us that the energy levels available to electrons are not infinite but discrete and that the energy difference between levels is a precise amount called a quantum. The four quantum numbers completely describe the location and energy of any electron within a given atom. Finally, we learned two simple recall methods for the order in which electrons fill the shells and subshells of an atom and that the valence electrons are the reactive electrons in an atom. In the next chapter, we’ll take a look at how the elements are organized on the periodic table and will then turn our attention to their bonding behavior—based on valence electrons—in Chapter 3 of MCAT General Chemistry Review.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Subatomic Particles
- Aproton has a positive charge and mass around 1 amu; a neutron has no charge and mass around 1 amu; an electron has a negative charge and negligible mass.
- The nucleus contains the protons and neutrons, while the electrons move around the nucleus.
- The atomic number is the number of protons in a given element.
- The mass number is the sum of an element’s protons and neutrons.
Atomic Mass *vs.* Atomic Weight
- Atomic mass is essentially equal to the mass number, the sum of an element’s protons and neutrons.
- Isotopes are atoms of a given element (same atomic number) that have different mass numbers. They differ in the number of neutrons.
- Most isotopes are identified by the element followed by the mass number (such as carbon-12, carbon-13, and carbon-14).
- The three isotopes of hydrogen go by different names: protium, deuterium, and tritium.
- Atomic weight is the weighted average of the naturally occurring isotopes of an element. The periodic table lists atomic weights, not atomic masses.
Rutherford, Planck, and Bohr
- Rutherford first postulated that the atom had a dense, positively charged nucleus that made up only a small fraction of the volume of the atom.
- In the Bohr model of the atom, a dense, positively charged nucleus is surrounded by electrons revolving around the nucleus in orbits with distinct energy levels.
- The energy difference between energy levels is called a quantum, first described by Planck.
- Quantization means that there is not an infinite range of energy levels available to an electron; electrons can exist only at certain energy levels. The energy of an electron increases the farther it is from the nucleus.
- The atomic absorption spectrum of an element is unique; for an electron to jump from a lower energy level to a higher one, it must absorb an amount of energy precisely equal to the energy difference between the two levels.
- When electrons return from the excited state to the ground state, they emit an amount of energy that is exactly equal to the energy difference between the two levels; every element has a characteristic atomic emission spectrum, and sometimes the electromagnetic energy emitted corresponds to a frequency in the visible light range.
Quantum Mechanical Model of Atoms
- The quantum mechanical model posits that electrons do not travel in defined orbits but rather are localized in orbitals; an orbital is a region of space around the nucleus defined by the probability of finding an electron in that region of space.
- The Heisenberg uncertainty principle states that it is impossible to know both an electron’s position and its momentum exactly at the same time.
- There are four quantum numbers; these numbers completely describe any electron in an atom.
- The principal quantum number, n, describes the average energy of a shell.
- The azimuthal quantum number, l, describes the subshells within a given principal energy level (s, p, d, and f).
- The magnetic quantum number, ml, specifies the particular orbital within a subshell where an electron is likely to be found at a given moment in time.
- The spin quantum number, ms, indicates the spin orientation ( ± 1 2 ) of an electron in an orbital.
- The electron configuration uses spectroscopic notation (combining the n and l values as a number and letter, respectively) to designate the location of electrons.
- For example, 1s22s22p63s2 is the electron configuration for magnesium: a neutral magnesium atom has 12 electrons—two in the s subshell of the first energy level, two in the s subshell of the second energy level, six in the p subshell of the second energy level, and two in the s subshell of the third energy level; the two electrons in the 3s subshell are the valence electrons for the magnesium atom.
- Electrons fill the principal energy levels and subshells according to increasing energy, which can be determined by the *n*** + *l***rule.
- Electrons fill orbitals according to Hund’s rule, which states that subshells with multiple orbitals (p, d, and f) fill electrons so that every orbital in a subshell gets one electron before any of them gets a second.
- Paramagnetic materials have unpaired electrons that align with magnetic fields, attracting the material to a magnet.
- Diamagnetic materials have all paired electrons, which cannot easily be realigned; they are repelled by magnets.
- Valence electrons are those electrons in the outermost shell available for interaction (bonding) with other atoms.
- For the representative elements (those in Groups 1, 2, and 13−18), the valence electrons are found in s- and/or p-orbitals.
- For the transition elements, the valence electrons are found in s- and either d- or f-orbitals.
- Many atoms interact with other atoms to form bonds that complete an octet in the valence shell.
ANSWERS TO CONCEPT CHECKS
**1.1**
- Charge is determined by the number of electrons present. Atomic number is determined by the number of protons. Isotope is determined by the number of neutrons (while protons make up part of the mass number, it is the number of neutrons that explains the variability between isotopes).
- 18O: 8 p+, 10 n0, 8 e–. 18F: 9 p+, 9 n0, 9 e–.
**1.2**
- Atomic mass is (just slightly less than) the sum of the masses of protons and neutrons in a given atom of an element. Atoms of the same element with different mass numbers are isotopes of each other. The atomic weight is the weighted average of the naturally occurring isotopes of an element.
- This ratio is an equivalent concept. It is therefore acceptable, as long as units can be cancelled in dimensional analysis.
-
Isotope Protons Neutrons Electrons 19O 8 11 8
16O 8 8 8
17O 8 9 8
19F 9 10 9
16F 9 7 9
238U 92 146 92
240U 92 148 92
**1.3**
-
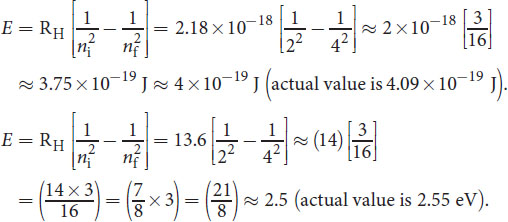
-
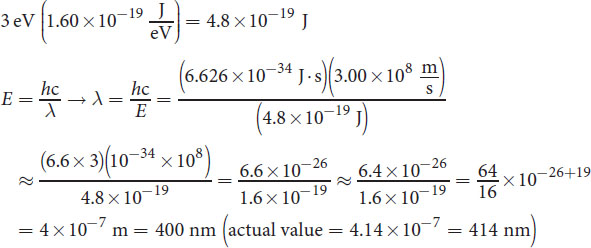
-
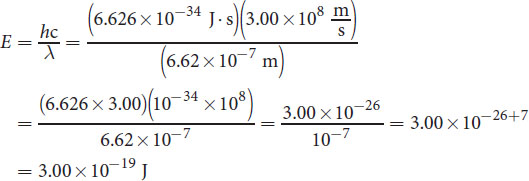
**1.4**
-
***n l* Possible Elements**
2 1 2p: B, C, N, O, F, Ne
3 0 3s: Na, Mg
5 3 5f: Actinide series
4 2 4d: Y, Zr, Nb, Mo, Tc, Ru, Rh, Pd, Ag, Cd
- Both O and O2– have fully filled 1s- and 2s-orbitals. O has four electrons in the 2p subshell; two are paired, and the other two each have their own orbital. O2– has six electrons in the 2p subshell, all of which are paired in the three p-orbitals.
- Both these molecules have unfilled valence electron shells with relatively few paired electrons; therefore, they are paramagnetic.
-
**Atom s-electrons p-electrons d-electrons f-electrons Total Valence Electrons P in PO43−** 2 6 2 0 10
O in PO43− 2 6 0 0 8
Ir 2 0 7 0 9
Cf 2 0 0 10 12
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. A
Remember that when electrons are removed from an element, forming a cation, they will be removed from the subshell with the highest n value first. Zn0 has 30 electrons, so it would have an electron configuration of 1s22s22p63s23p64s23d10. The 4s subshell has the highest principal quantum number, so it is emptied first, forming 1s22s22p63s23p64s03d10. (B) implies that electrons are pulled out of the * d** *subshell, (C) presents the configuration of the uncharged zinc atom, and (D) shows the configuration that would exist if four electrons were removed.
2. B
The azimuthal quantum number l cannot be higher than n – 1, ruling out (A). The ml number, which describes the chemical’s magnetic properties, can only be an integer value between –l and l. It cannot be equal to ±1 if l = 0; this would imply that the s subshell has three orbitals (−1, 0, and 1) when we know it can only have one. This rules out (C) and (D).
3. C
For any value of n, there will be a maximum of 2n2 electrons; that is, two per orbital. This can also be determined from the periodic table. There are only two elements (H and He) that have valence electrons in the n = 1 shell. Eight elements (Li to Ne) have valence electrons in the n = 2 shell. This is the only equation that matches this pattern.
4. B
This formula describes the number of electrons in terms of the azimuthal quantum number l, which ranges from 0 to n – 1, with n being the principal quantum number. A table of the maximum number of electrons per subshell is provided here:
**Subshell Azimuthal Quantum Number (l) Number of Electrons**
s 0 2
p 1 6
d 2 10
f 3 14
5. D
The only answer choice without unpaired electrons in its ground state is helium. Recall from the chapter that a diamagnetic substance is identified by the lack of unpaired electrons in its shell. A substance without unpaired electrons, like helium, cannot be magnetized by an external magnetic field and is actually slightly repelled. Elements that come at the end of a block (Group IIA, the group containing Zn, and the noble gases, most notably) have only paired electrons.
6. D
The problem requires the MCAT favorite equation
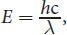
where h = 6.626 × 10−34 J ·s (Planck’s constant),
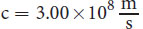
is the speed of light, and λ is the wavelength of the light. This question asks for the energy of one mole of photons, so we must multiply by Avogadro’s number, NA = 6.02 × 1023 mol−1.
The setup is:
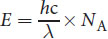
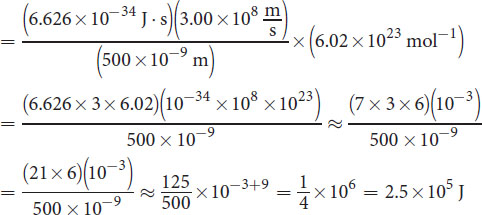
As we rounded during calculation, we must find the answer choice that most closely matches this value. (D) is the closest match.
7. B
Because the electron is moving into the n = 1 shell, the only subshell available is the 1s subshell, which eliminates (C) and (D). There will be some energy change, however, as the electron must lose energy to return to the minimum-energy ground state. That will require emitting radiation in the form of a photon.
8. A
Recall that the superscript refers to the mass number of an atom, which is equal to the number of protons plus the number of neutrons present in an element. Sometimes a text will list the atomic number, Z, as a subscript under the mass number, A. According to the periodic table, carbon contains six protons; therefore, its atomic number is 6. Isotopes all have the same number of protons, but differ in the number of neutrons. Almost all atoms with Z > 1 have at least one neutron. Carbon is most likely to have a mass number of 12, for six protons and six neutrons, as in (B).(C) and (D) are possible isotopes that would have more neutrons than 12C. The 6C isotope is unlikely. It would mean that there are 6 protons and 0 neutrons. As shown in Figure 1.4, this would be a highly unstable isotope.
9. C
The limitations placed by the Heisenberg uncertainty principle are caused by limitations inherent in the measuring process: if a particle is moving, it has momentum, but trying to measure that momentum necessarily creates uncertainty in the position. Even if we had an exact definition of the meter, as in (A), or perfect measuring devices, as in (B), we still wouldn’t be able to measure position and momentum simultaneously and exactly.
10. B
For the electron to gain energy, it must absorb energy from photons to jump up to a higher energy level. There is a bigger jump between n = 2 and n = 6 than there is between n = 3 and n = 4.
11. A
The MCAT covers the topics in this chapter qualitatively more often than quantitatively. It is critical to be able to distinguish the fundamental principles that determine electron organization, which are usually known by the names of the scientists who discovered or postulated them. The Heisenberg uncertainty principle, (B), refers to the inability to know the momentum and position of a single electron simultaneously. The Bohr model, (C), was an early attempt to describe the behavior of the single electron in a hydrogen atom. The Rutherford model, (D), described a dense, positively charged nucleus. The element shown here, phosphorus, is often used to demonstrate Hund’s rule because it contains a half-filled p subshell. Hund’s rule explains that electrons fill empty orbitals first before doubling up electrons in the same orbital.
12. A
The quickest way to solve this problem is to use the periodic table and find out how many protons are in Cs atoms; there are 55. Neutral Cs atoms would also have 55 electrons. A stable Cs cation will have a single positive charge because it has one unpaired s-electron. This translates to one fewer electron than the number of protons or 54 electrons.
13. B
The easiest way to approach this problem is to set up a system of two algebraic equations, where H and D are the percentages of H (mass = 1 amu) and D (mass = 2 amu), respectively. Your setup should look like the following system:
H + D = 1 (percent H + percent D = 100%) 1 H + 2 D = 1.008 (atomic weight calculation)
Rearranging the first equation and substituting into the second yields (1 – D) + 2D = 1.008, or D = 0.008. 0.008 is 0.8%, so there is 0.8% D.
14. A
The terms in the answer choices refer to the magnetic spin of the two electrons. The quantum number *m**srepresents this property as a measure of an electron’s intrinsic spin. These electrons’ spins are parallel, in that their spins are aligned in the same direction (m**s* = +
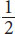
for both species).
15. B
When dealing with ions, you cannot directly approach electronic configurations based on the number of electrons they currently hold. First examine the neutral atom’s configuration, and then determine which electrons are removed.
Neutral Atom’s Configuration Ion’s Configuration
Cr0: [Ar] 4s13d5 —
Mn0: [Ar] 4s23d5 Mn+: [Ar] 4s13d5
Fe0: [Ar] 4s23d6 Fe2+: [Ar] 4s03d6
Due to the stability of half-filled d-orbitals, neutral chromium assumes the electron configuration of [Ar] 4s13d5. Mn must lose one electron from its initial configuration to become the Mn+ cation. That electron would come from the 4s subshell according to the rule that the first electron removed comes from the highest-energy shell. Fe must lose two electrons to become Fe2+. They’ll both be lost from the same orbital; the only way Fe2+ could hold the configuration in the question stem would be if one d-electron and one s-electron were lost together.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(1.1) Planck relation(frequency): E = hf
(1.2) Angular momentum of an electron(Bohr model):
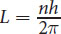
(1.3) Energy of an electron(Bohr model):
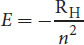
(1.4) Planck relation(wavelength):
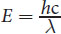
(1.5) Energy of electron transition(Bohr model):
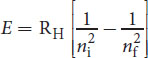
(1.6) Maximum number of electrons within a shell: 2n2
(1.7) Maximum number of electrons within a subshell: 4l + 2
SHARED CONCEPTS
General Chemistry Chapter 2
The Periodic Table
General Chemistry Chapter 3
Bonding and Chemical Interactions
Organic Chemistry Chapter 3
Bonding
Physics and Math Chapter 2
Work and Energy
Physics and Math Chapter 8
Light and Optics
Physics and Math Chapter 9
Atomic and Nuclear Phenomena