Chapter 2: The Periodic Table

Chapter 2: The Periodic Table
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Lithium and sodium have similar chemical properties. For example, both can form ionic bonds with chloride. Which of the following best explains this similarity?
- Both lithium and sodium ions are positively charged.
- Lithium and sodium are in the same group of the periodic table.
- Lithium and sodium are in the same period of the periodic table.
- Both lithium and sodium have low atomic weights.
-
Carbon and silicon are the basis of biological life and synthetic computing, respectively. While these elements share many chemical properties, which of the following best describes a difference between the two elements?
- Carbon has a smaller atomic radius than silicon.
- Silicon has a smaller atomic radius than carbon.
- Carbon has fewer valence electrons than silicon.
- Silicon has fewer valence electrons than carbon.
-
Which of the following elements has the highest electronegativity?
- Mg
- Cl
- Zn
- I
-
Ionization energy contributes to an atom’s chemical reactivity. Which of the following shows an accurate ranking of ionization energies from lowest to highest?
- First ionization energy of Be < second ionization energy of Be < first ionization energy of Li
- First ionization energy of Be < first ionization energy of Li < second ionization energy of Be
- First ionization energy of Li < first ionization energy of Be < second ionization energy of Be
- First ionization energy of Li < second ionization energy of Be < first ionization energy of Be
-
Antimony is used in some antiparasitic medications—specifically those targeting Leishmania donovani. What type of element is antimony?
- Metal
- Metalloid
- Halogen
- Nonmetal
-
The properties of atoms can be predicted, to some extent, by their location within the periodic table. Which property or properties increase in the direction of the arrows shown?
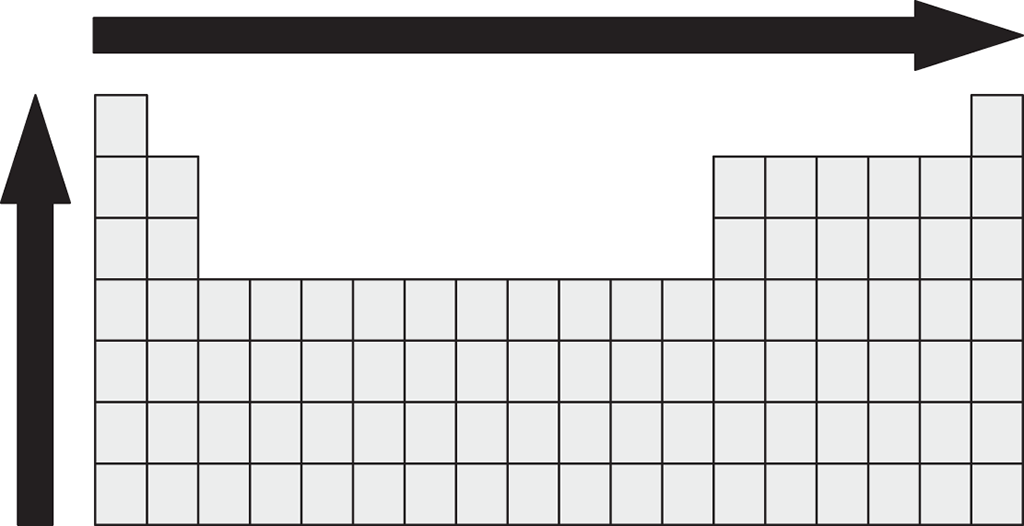
- Electronegativity
- Atomic radius
- First ionization energy
- I only
- I and II only
- I and III only
- II and III only
-
Metals are often used for making wires that conduct electricity. Which of the following properties of metals explains why?
- Metals are malleable.
- Metals have low electronegativities.
- Metals have valence electrons that can move freely.
- Metals have high melting points.
-
Which of the following is an important property of the group of elements shaded in the periodic table below?
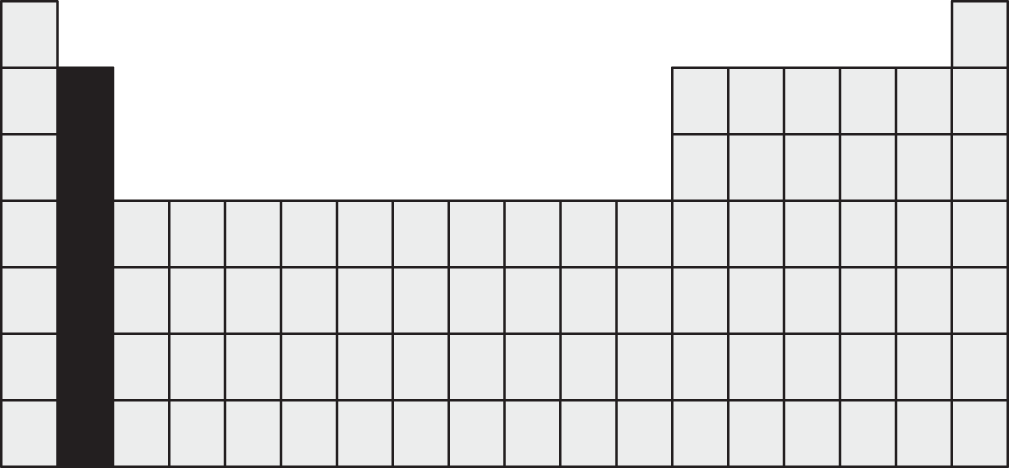
- These elements are the best electrical conductors in the periodic table.
- These elements form divalent cations.
- The second ionization energy for these elements is lower than the first ionization energy.
- The atomic radii of these elements decrease as one moves down the column.
-
When dissolved in water, which of the following ions is most likely to form a complex ion with H2O?
- Na+
- Fe2+
- Cl–
- S2–
-
How many valence electrons are present in elements in the third period?
- 2
- 3
- The number decreases as the atomic number increases.
- The number increases as the atomic number increases.
-
Arsenic mediates some of its toxic effects via the inhibition of acetyl-CoA formation and the inhibition of the enzyme succinic dehydrogenase to decrease ATP production. In addition to its metabolic effects, arsenic can also replace phosphorus in many reactions. Which of the following best explains the similarity between these two elements?
- Similar atomic numbers
- Similar number of electrons
- Similar valence configuration
- Similar atomic radius
-
Of the four atoms depicted here, which has the highest electron affinity?
-
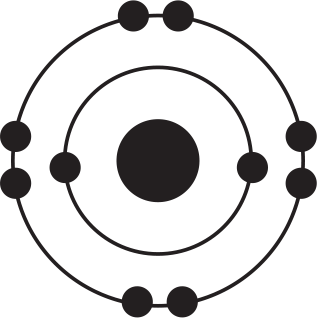
-
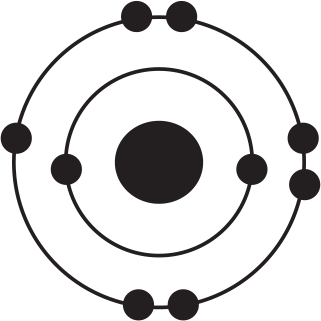
-
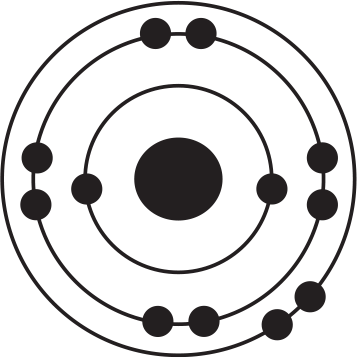
-
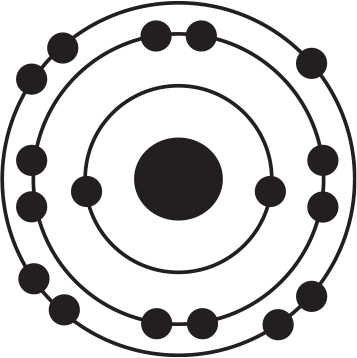
-
Which of the following atoms or ions has the largest effective nuclear charge?
- Cl
- Cl–
- K
- K+
-
Why do halogens often form ionic bonds with alkaline earth metals?
- The alkaline earth metals have much higher electron affinities than the halogens.
- By sharing electrons equally, the alkaline earth metals and halogens both form full octets.
- Within the same row, the halogens have smaller atomic radii than the alkaline earth metals.
- The halogens have much higher electron affinities than the alkaline earth metals.
-
What is the highest-energy orbital of elements with valence electrons in the n = 3 shell?
- s-orbital
- p-orbital
- d-orbital
- f-orbital
Answer Key
- B
- A
- B
- C
- B
- C
- C
- B
- B
- D
- C
- D
- D
- D
- C
Chapter 2: The Periodic Table
CHAPTER 2
THE PERIODIC TABLE
In This Chapter
2.1 The Periodic Table 2.2 Types of Elements
Metals
Nonmetals
Metalloids
2.3 Periodic Properties of the Elements
Atomic and Ionic Radii
Ionization Energy
Electron Affinity
Electronegativity
2.4 The Chemistry of Groups
Alkali Metals (IA)
Alkaline Earth Metals (IIA)
Chalcogens (VIA)
Halogens (VIIA)
Noble Gases (VIIIA)
Transition Metals (B)
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 10% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content category:
4E: Atoms, nuclear decay, electronic structure, and atomic chemical behavior
Introduction
The pharmacological history of lithium is an interesting window into the scientific and medical communities’ attempts to take advantage of the chemical and physical properties of an element for human benefit. By the mid-1800s, the medical community was showing great interest in theories that linked uric acid to a myriad of maladies. When it was discovered that solutions of lithium carbonate dissolved uric acid, therapeutic preparations containing lithium carbonate salt became popular. Even nonmedical companies tried to profit from lithium’s reputation as a cure-all by adding it to their soft drinks.
Eventually, fascination with theories of uric acid wore off, and lithium’s time in the spotlight seemed to be coming to an end. Then, in the 1940s, doctors began to recommend salt-restricted diets for cardiac patients. Lithium chloride was made commercially available as a sodium chloride (table salt) substitute. Unfortunately, lithium is quite toxic at fairly low concentrations, and when medical literature in the late 1940s reported several incidents of severe poisonings and multiple deaths—some associated with only minor lithium overdosing—U.S. companies voluntarily withdrew all lithium salts from the market. Right around this time, John Cade, a psychiatrist from Australia, proposed the use of lithium salts for the treatment of mania. Cade’s clinical trials were quite successful. In fact, his use of lithium salts to control mania was the first instance of successful medical treatment of a mental illness, and lithium carbonate became commonly prescribed in Europe for manic behavior. Not until 1970 did the U.S. Food and Drug Administration finally approve the use of lithium carbonate for manic symptoms.
Lithium (Li) is the element with the atomic number 3. It is a very soft alkali metal, and under standard conditions, it is the least dense solid element (specific gravity = 0.53). Lithium is so reactive that it does not naturally occur on earth in its elemental form and is found only in various salt compounds.
Why would medical scientists pay attention to this particular element? What would make doctors believe that lithium chloride would be a good substitute for sodium chloride for patients on salt-restricted diets? The answers lie in the periodic table.
2.1 The Periodic Table
LEARNING OBJECTIVES
After Chapter 2.1, you will be able to:
- Explain how the modern periodic table is organized
- Differentiate between representative and nonrepresentative elements
In 1869, the Russian chemist Dmitri Mendeleev published the first version of his Periodic Table of the Elements, which showed that ordering the known elements according to atomic weight revealed a pattern of periodically recurring physical and chemical properties. Since then, the periodic table has been revised, using the work of English physicist Henry Moseley, to organize the elements based on increasing atomic number (the number of protons in an element) rather than atomic weight. Using this revised table, many properties of elements that had not yet been discovered could be predicted. The periodic table creates a visual representation of the periodic law, which states: the chemical and physical properties of the elements are dependent, in a periodic way, upon their atomic numbers.
The modern periodic table arranges the elements into periods (rows) and groups or families (columns), based on atomic number. There are seven periods representing the principal quantum numbers n = 1 through n = 7 for the s- and p-block elements. Each period is filled sequentially, and each element in a given period has one more proton and one more electron than the element to its left (in their neutral states). Groups contain elements that have the same electronic configuration in their valence shell and share similar chemical properties.
BRIDGE
Recall from Chapter 1 of MCAT General Chemistry Review that periods (rows) graphically represent the principal quantum number, and groups (columns) help to determine the valence electron configuration.
The electrons in the valence shell, known as the valence electrons, are the farthest from the nucleus and have the greatest amount of potential energy. Their higher potential energy and the fact that they are held less tightly by the nucleus allows them to become involved in chemical bonds with the valence electrons of other atoms; thus, the valence shell electrons largely determine the chemical reactivity and properties of the element.
MCAT EXPERTISE
Relating valence electrons to reactivity is important. Elements with similar valence electron configurations generally behave in similar ways, as long as they are the same type (metal, nonmetal, or metalloid).
The Roman numeral above each group represents the number of valence electrons elements in that group have in their neutral state. The Roman numeral is combined with the letter A or B to separate the elements into two larger classes. The A elements are known as the representative elements and include groups IA through VIIIA. The elements in these groups have their valence electrons in the orbitals of either s or p subshells. The B elements are known as the nonrepresentative elements and include both the transition elements, which have valence electrons in the s and d subshells, and the lanthanide and actinide series, which have valence electrons in the s and f subshells. For the representative elements, the Roman numeral and the letter designation determine the electron configuration. For example, an element in Group VA has five valence electrons with the configuration s2p3. As described in Chapter 1 of MCAT General Chemistry Review, the nonrepresentative elements may have unexpected electron configurations, such as chromium (4s13d5) and copper (4s13d10). In the modern IUPAC identification system, the groups are numbered 1 to 18 and are not subdivided into Group A and Group B elements.
MCAT CONCEPT CHECK 2.1:
Before you move on, assess your understanding of the material with these questions.
-
Mendeleev’s table was arranged by atomic weight, but the modern periodic table is arranged by:
_______________________________________
-
Which of the following are representative elements (A), and which are nonrepresentative (B)?
Element A or B Element A or B Element A or B Element A or B Ag Al K P Pb Li Pu U Cu Cf Zn B N Np O He
2.2 Types of Elements
LEARNING OBJECTIVES
After Chapter 2.2, you will be able to:
- Classify elements as metal, nonmetal, or metalloid
- Predict the traits of an element given its location on a periodic table:
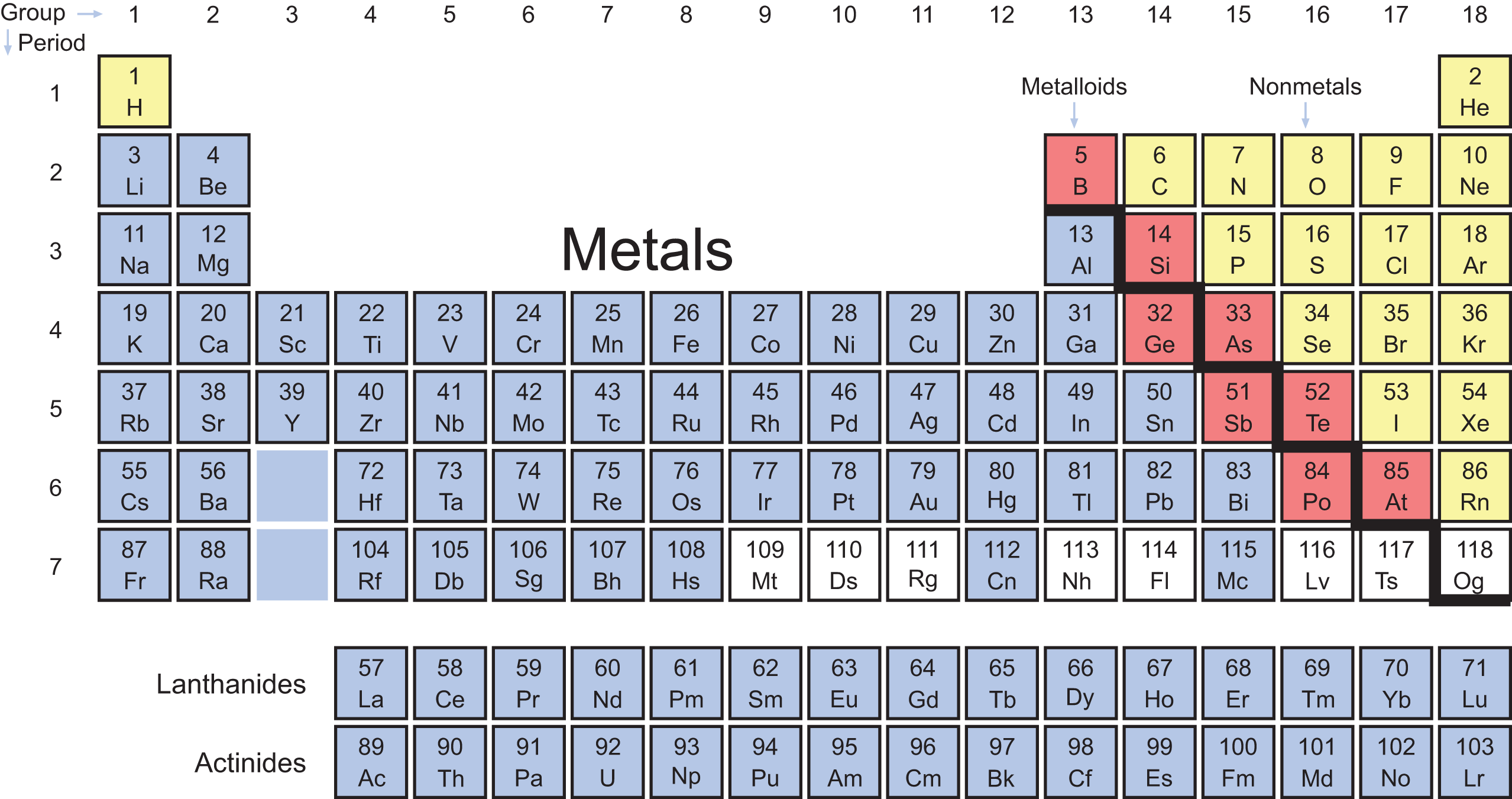
When we consider the trends of chemical reactivity and physical properties together, we can begin to identify groups of elements with similar characteristics. These larger collections are divided into three categories: metals, nonmetals, and metalloids (also called semimetals).
Metals
Metals are found on the left side and in the middle of the periodic table. They include the active metals, the transition metals, and the lanthanide and actinide series of elements. Metals are lustrous (shiny) solids, except for mercury, which is a liquid under standard conditions. They generally have high melting points and densities, but there are exceptions, such as lithium, which has a density about half that of water. Metals have the ability to be deformed without breaking; the ability of metal to be hammered into shapes is called malleability, and its ability to be pulled or drawn into wires is called ductility. At the atomic level, a metal is defined by a low effective nuclear charge, low electronegativity (high electropositivity), large atomic radius, small ionic radius, low ionization energy, and low electron affinity. All of these characteristics are manifestations of the ability of metals to easily give up electrons.
Many of the transition metals (Group B elements) have two or more oxidation states (charges when forming bonds with other atoms). Because the valence electrons of all metals are only loosely held to their atoms, they are free to move, which makes metals good conductors of heat and electricity. The valence electrons of the active metals are found in the s subshell; those of the transition metals are found in the s and d subshells; and those of the lanthanide and actinide series elements are in the s and f subshells. Some transition metals—copper, nickel, silver, gold, palladium, and platinum—are relatively nonreactive, a property that makes them ideal for the production of coins and jewelry.
KEY CONCEPT
Alkali and alkaline earth metals are both metallic in nature because they easily lose electrons from the s subshell of their valence shells.
An example of a metal is shown in Figure 2.1 with a copper wire. The wire exhibits luster, malleability, and ductility. It is used as a wire because it also exhibits good heat and electrical conductivity.

Figure 2.1. Copper (Cu) Metal Wire
Nonmetals
Nonmetals are found predominantly on the upper right side of the periodic table. Nonmetals are generally brittle in the solid state and show little or no metallic luster. They have high ionization energies, electron affinities, and electronegativities, as well as small atomic radii and large ionic radii. They are usually poor conductors of heat and electricity. All of these characteristics are manifestations of the inability of nonmetals to easily give up electrons. Nonmetals are less unified in their chemical and physical properties than the metals.
Carbon, shown in Figure 2.2, is a stereotypical nonmetal that retains a solid structure but is brittle, nonlustrous, and generally a poor conductor of heat and electricity.

Figure 2.2. Charcoal, Composed of the Nonmetal Carbon (C)
Metalloids
Separating the metals and nonmetals are a stair-step group of elements called the metalloids. The metalloids are also called semimetals because they share some characteristics with both metals and nonmetals. The electronegativities and ionization energies of the metalloids lie between those of metals and nonmetals. Their physical properties—densities, melting points, and boiling points—vary widely and can be combinations of metallic and nonmetallic characteristics. For example, silicon (Si) has a metallic luster but is brittle and a poor conductor. The reactivities of the metalloids are dependent on the elements with which they are reacting. Boron (B), for example, behaves like a nonmetal when reacting with sodium (Na) and like a metal when reacting with fluorine (F). The elements classified as metalloids form a “staircase” on the periodic table and include boron, silicon, germanium (Ge), arsenic (As), antimony (Sb), tellurium (Te), polonium (Po), and astatine (At). While there is debate over polonium and astatine’s status as metalloids, most sources label them as such. Figure 2.3 color-codes the major classifications of elements on the periodic table.
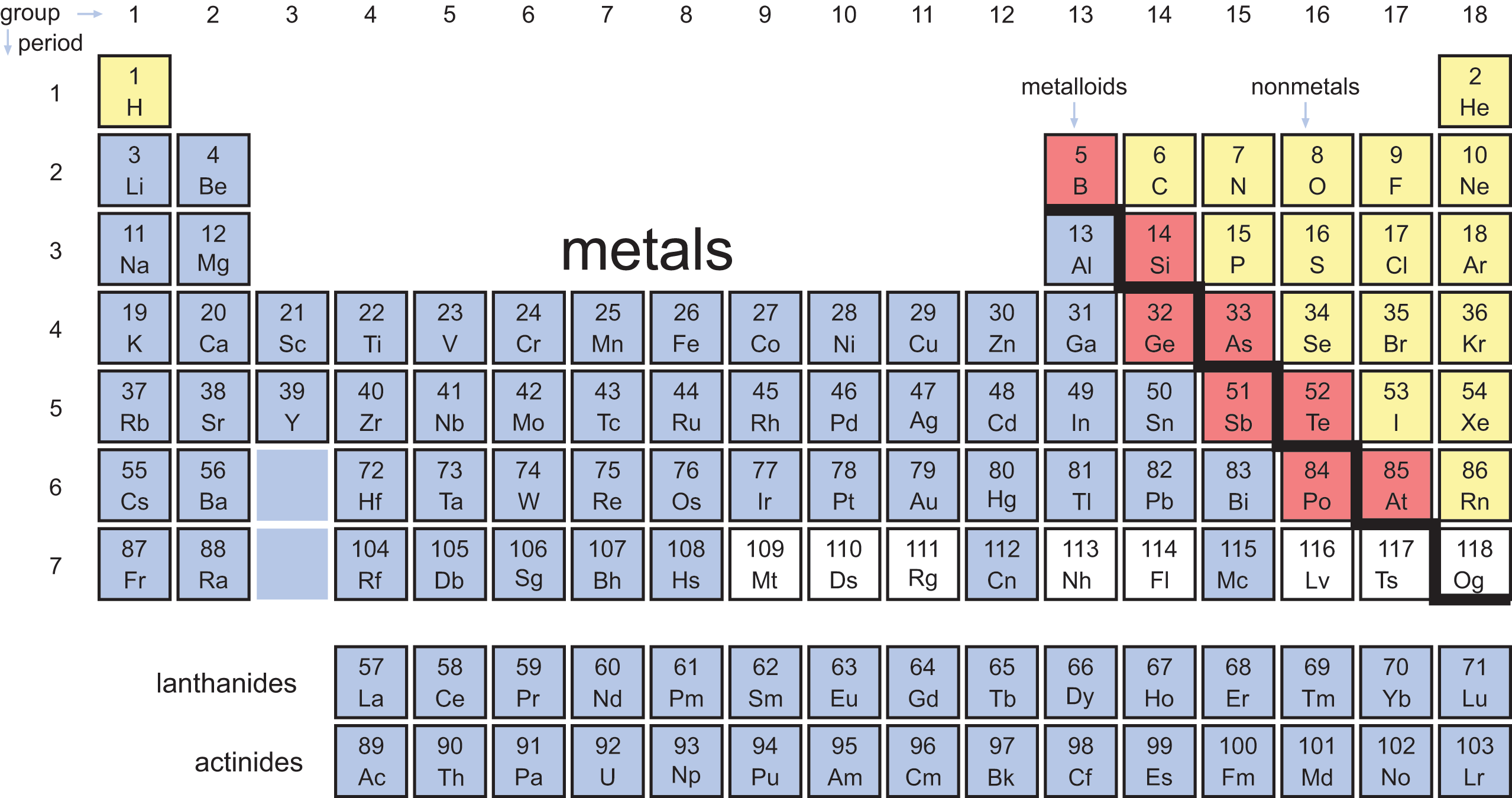
Figure 2.3. Periodic Table, Coded by Element Type
REAL WORLD
Metalloids share some properties with metals and others with nonmetals. For instance, metalloids make good semiconductors due to their partial conductivity of electricity.
MCAT CONCEPT CHECK 2.2:
Before you move on, assess your understanding of the material with these questions.
-
Based on their location in the periodic table, identify a few elements that likely possess the following properties:
- Luster: ________________________________
- Poor conductivity of heat and electricity: ________________________________
- Good conductivity but brittle: ____________________________
-
Classify the following elements as metals (M), nonmetals (NM), or metalloids (MO):
Element Class Element Class Element Class Element Class Ag Al K P Pb Li Pu U Cu As Zn B Si Np O He
2.3 Periodic Properties of the Elements
LEARNING OBJECTIVES
After Chapter 2.3, you will be able to:
- Compare the atomic radius of neutral atoms to their ions
- Rank elements by ionization energy, electron affinity, electronegativity, or atomic radius:

The MCAT does not expect you to have memorized the entire periodic table. Fortunately, the periodic table is a guide unto itself, a self-referencing localization system for all of the elements. Remember, the modern table is organized in such a way to represent visually the periodicity of chemical and physical properties of the elements. The periodic table, then, can provide you with a tremendous amount of information that otherwise would have to be memorized. Note, though, that while you do not need to memorize the periodic table for the MCAT, you do need to understand the trends within the periodic table that help predict the chemical and physical behaviors of the elements.
MCAT EXPERTISE
Don’t try to memorize the periodic table. You will have access to it on Test Day through the test interface. Do understand its configuration and trends so that you can use it efficiently to get a higher score!
Before exploring the periodic trends, let’s take stock of three key rules that control how valence electrons work in an atom. First, as we’ve already mentioned, as one moves from left to right across a period, electrons and protons are added one at a time. As the positivity of the nucleus increases, the electrons surrounding the nucleus, including those in the valence shell, experience a stronger electrostatic pull toward the center of the atom. This causes the electron cloud, which is the outer boundary defined by the valence shell electrons, to move closer and bind more tightly to the nucleus. This electrostatic attraction between the valence shell electrons and the nucleus is known as the effective nuclear charge (Zeff), a measure of the net positive charge experienced by the outermost electrons. This pull is somewhat mitigated by nonvalence electrons that reside closer to the nucleus. For elements in the same period, Zeff increases from left to right. The parts of an atom responsible for Zeff are illustrated in Figure 2.4.
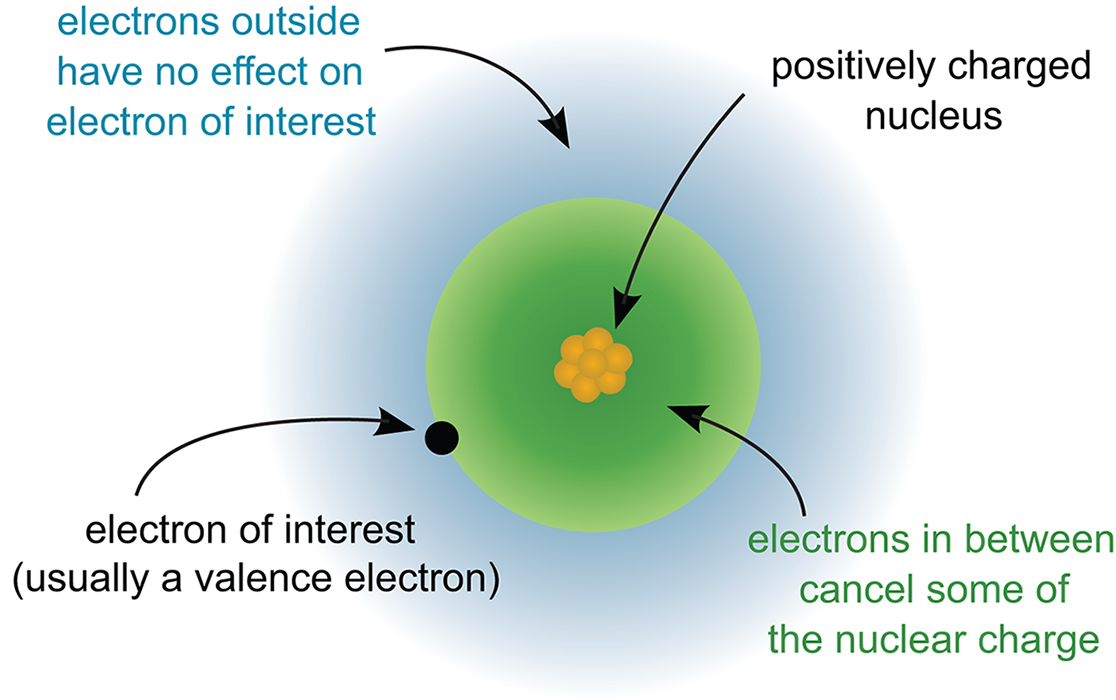
Figure 2.4. Factors that Determine Effective Nuclear Charge (Zeff)
BRIDGE
Zeff relies on the principles of electrostatic forces defined in Chapter 5 of MCAT Physics and Math Review. The values q1 and q2 can represent the net charge of the nucleus and valence electron shell, respectively. The larger each charge gets (going to the right in the periodic table), the higher the value of Zeff.
Second, as one moves down the elements of a given group, the principal quantum number increases by one each time. This means that the valence electrons are increasingly separated from the nucleus by a greater number of filled principal energy levels, which can also be called inner shells. The result of this increased separation is a reduction in the electrostatic attraction between the valence electrons and the positively charged nucleus. These outermost electrons are held less tightly as the principal quantum number increases. As one goes down in a group, the increased shielding created by the inner shell electrons cancels the increased positivity of the nucleus. Thus, the Zeff is more or less constant among the elements within a given group. Despite this fact, the valence electrons are held less tightly to the nucleus as one moves down a group due to the increased separation between valence electrons and the nucleus.
Third, elements can also gain or lose electrons in order to achieve a stable octet formation representative of the noble (inert) gases (Group VIIIA or Group 18). In Chapter 3 of MCAT General Chemistry Review, we will discuss how the octet rule is hardly a rule at all because there are many exceptions. For now, keep in mind that elements, especially the ones that have biological roles, tend to be most stable with eight electrons in their valence shell.
These three facts are guiding principles as we work toward an understanding of the trends demonstrated in the periodic table. In fact, the trend for effective nuclear charge across a period and the impact of increasing the number of inner shells down a group will help derive all the trends we discuss below.
Atomic and Ionic Radii
Think of an atom as a cloud of electrons surrounding a dense core of protons and neutrons. The atomic radius of an element is thus equal to one-half of the distance between the centers of two atoms of an element that are briefly in contact with each other. The distance between two centers of circles in contact is akin to a diameter, making this radius calculation simple. The atomic radius cannot be measured by examining a single atom because the electrons are constantly moving around, making it impossible to mark the outer boundary of the electron cloud.
KEY CONCEPT
Atomic radius refers to the size of a neutral element, while an ionic radius is dependent on how the element ionizes based on its element type and group number.
As we move across a period from left to right, protons and electrons are added one at a time to the atoms. Because the electrons are being added only to the outermost shell and the number of inner-shell electrons remains constant, the increasing positive charge of the nucleus pulls the outer electrons more closely inward and holds them more tightly. The Zeff increases left to right across a period, and as a result, atomic radius decreases from left to right across a period.
MCAT EXPERTISE
Atomic radius is essentially opposite that of all other periodic trends. While others increase going up and to the right, atomic radius increases going down and to the left.
As we move down a group, the increasing principal quantum number implies that the valence electrons will be found farther away from the nucleus because the number of inner shells is increasing, separating the valence shell from the nucleus. Although the Zeff remains essentially constant, the atomic radius increases down a group. Within each group, the largest atom will be at the bottom, and within each period, the largest atom will be in Group IA (Group 1). For reference, the largest atomic radius in the periodic table belongs to cesium (Cs, 260 pm), and the smallest belongs to helium (He, 25 pm). Francium is typically not considered because it is exceptionally rare in nature. Figure 2.5 displays a graph of atomic radius vs. atomic number, with Group IA elements possessing the largest atomic radius in each row.
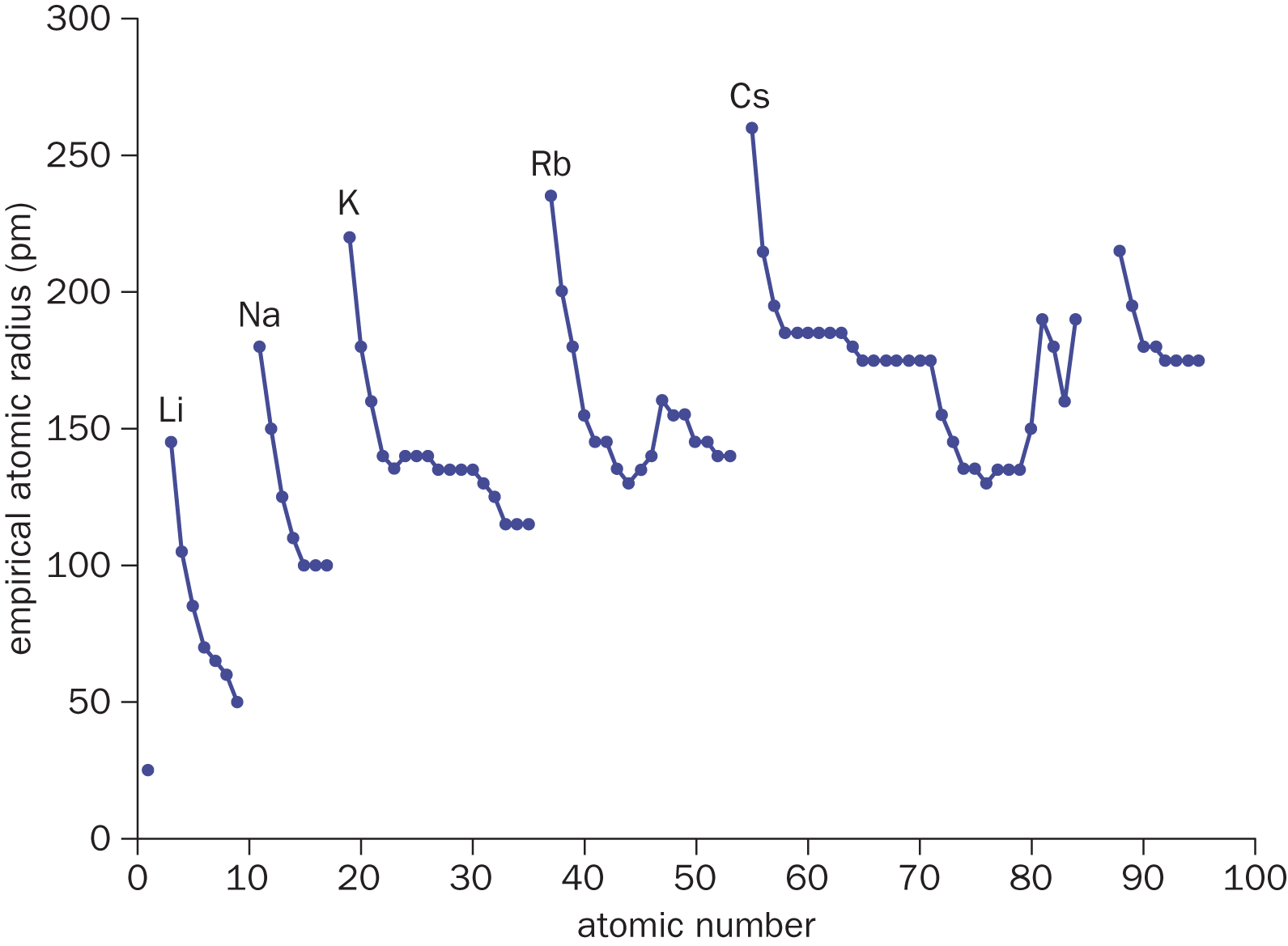
Figure 2.5. Atomic Radius (in pm) vs. Atomic Number
Unlike atomic radii, ionic radii will require some critical thinking and periodic table geography to determine. In order to understand ionic radii, we must make two generalizations. One is that metals lose electrons and become positive, while nonmetals gain electrons and become negative. The other is that metalloids can go in either direction, but tend to follow the trend based on which side of the metalloid line they fall on. Thus, silicon (Si) behaves more like a nonmetal, while germanium (Ge) tends to act more like a metal. On the MCAT, these generalizations can also be inferred from information found in passages and questions, such as oxidation states in compounds.
For nonmetals close to the metalloid line, their group number dictates that they require more electrons than other nonmetals to achieve the electronic configuration seen in Group VIIIA (Group 18). These nonmetals gain electrons while their nuclei maintain the same charge. Therefore, these nonmetals close to the metalloid line possess a larger ionic radius than their counterparts closer to Group VIIIA.
For metals, the trend is similar but opposite. Metals closer to the metalloid line have more electrons to lose to achieve the electronic configuration seen in Group VIIIA. Because of this, the ionic radius of metals near the metalloid line is dramatically smaller than that of other metals. Metals closer to Group IA have fewer electrons to lose and therefore experience a less drastic reduction in radius during ionization. These changes are illustrated in Figure 2.6. Note that tellurium (Te) behaves as a nonmetal and boron (B) behaves as a metal; under varying conditions, these metalloids can have opposite behavior.
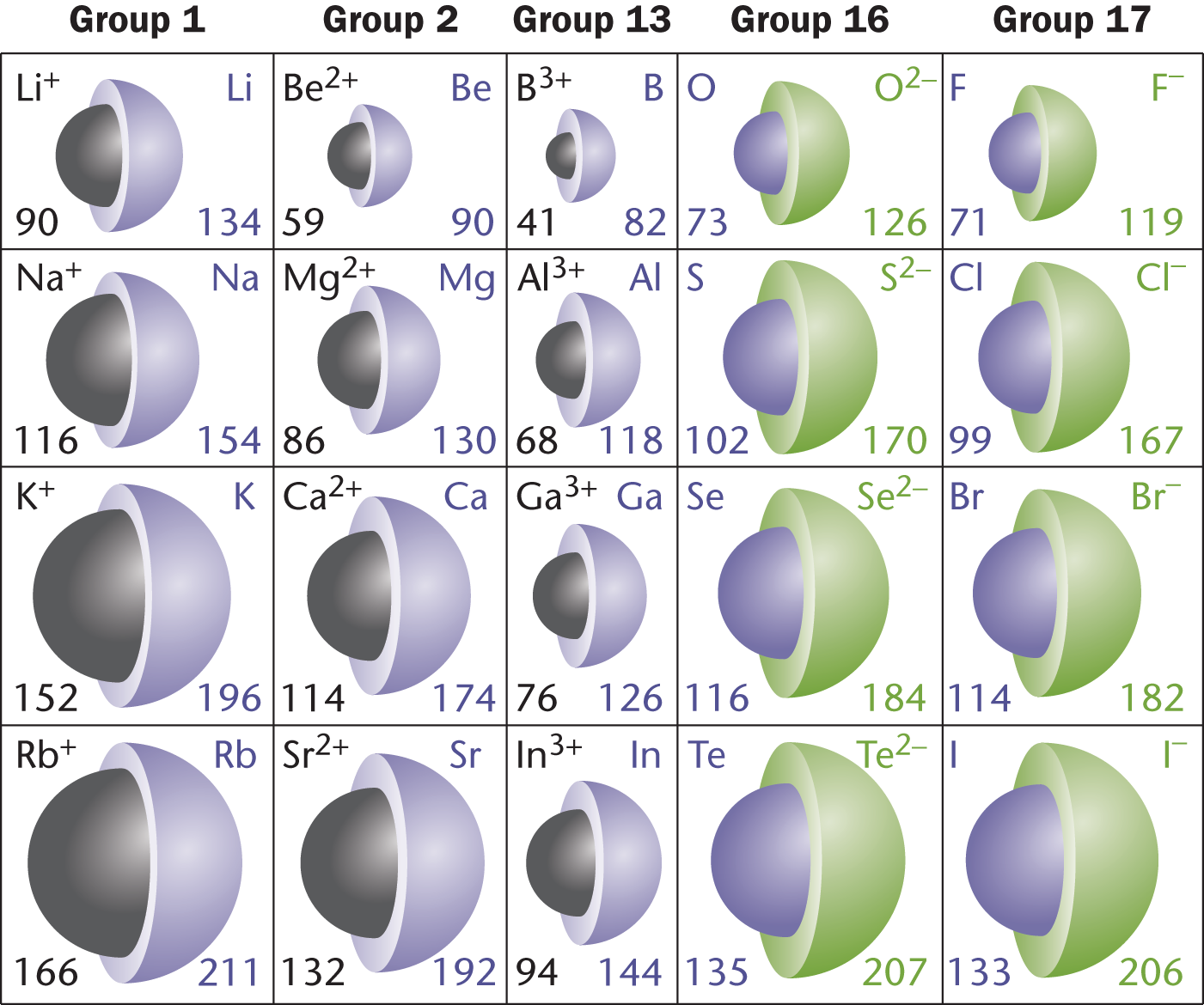
Figure 2.6. Ionic Radii (in pm) for Various Metals and Nonmetals Neutral atoms are shown in purple; cations in black; anions in green.
Ionization Energy
Ionization energy (IE), also known as ionization potential, is the energy required to remove an electron from a gaseous species. Removing an electron from an atom always requires an input of heat, which makes it an endothermic process. The greater the atom’s Zeff or the closer the valence electrons are to the nucleus, the more tightly bound they are. This makes it more difficult to remove one or more electrons, increasing the ionization energy. Thus, ionization energy increases from left to right across a period and from bottom to top in a group. The subsequent removal of a second or third electron requires increasing amounts of energy because the removal of more than one electron means that the electrons are being removed from an increasingly cationic (positive) species. The energy necessary to remove the first electron is called the first ionization energy; the energy necessary to remove the second electron from the univalent cation (X+) to form the divalent cation (X2+) is called the second ionization energy, and so on. For example:
Mg ( g ) → Mg + ( g ) + e − first ionization energy = 738 kJ mol Mg + ( g ) → Mg 2 + ( g ) + e − second ionization energy = 1450 kJ mol
Elements in Groups IA and IIA (Groups 1 and 2), such as lithium and beryllium, have such low ionization energies that they are called the active metals. The active metals do not exist naturally in their neutral forms; they are always found in ionic compounds, minerals, or ores. The loss of one electron from the alkali metals (Group IA) or the loss of two electrons from the alkaline earth metals (Group IIA) results in the formation of a stable, filled valence shell. In contrast, the Group VIIA (Group 17) elements—the halogens—do not typically give up their electrons. In fact, in their ionic form, they are generally anions. The first ionization energies of the elements are shown in Figure 2.7.
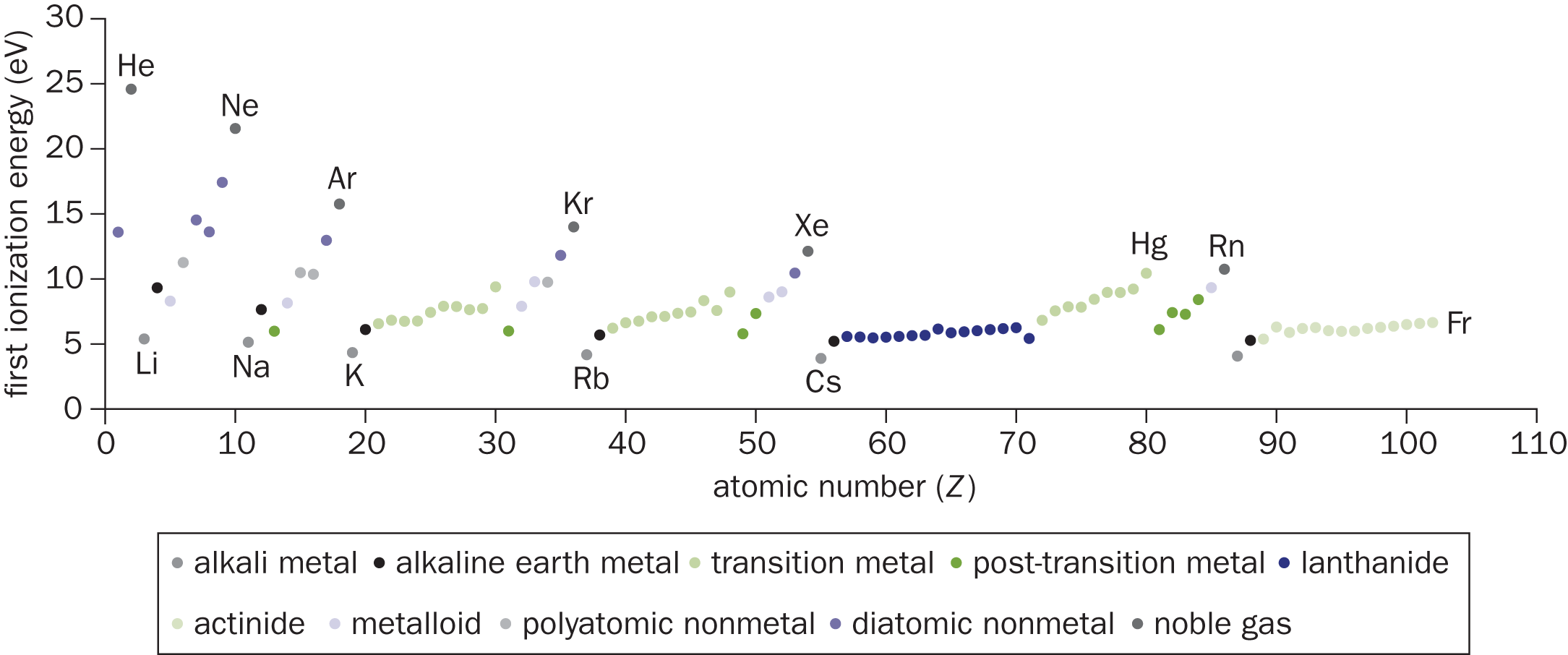
Figure 2.7. First Ionization Energies (in eV) of the Elements
MCAT EXPERTISE
First ionization energy (IE) will always be smaller than second IE, which will always be smaller than third IE. However, the degree to which the IE increases provides clues about the identity of the atom. If losing a certain number of electrons gives an element a noble gas-like electron configuration, then removing a subsequent electron will cost much more energy. For example:
- Mg2+ (g) → Mg3+ (g) + *e**–*
- third ionization energy = 7730 kJ mol
The values for second ionization energies are disproportionally larger for Group IA monovalent cations (like Na+) but generally not that much larger for Group IIA or subsequent monovalent cations (like Mg+). This is because removing one electron from a Group IA metal results in a noble gas-like electron configuration.
Group VIIIA (Group 18) elements, or noble or inert gases, are the least likely to give up electrons. They already have a stable electron configuration and are unwilling to disrupt that stability by giving up an electron. Therefore, noble gases are among the elements with the highest ionization energies.
Electron Affinity
Halogens are the most “greedy” group of elements on the periodic table when it comes to electrons. By acquiring one additional electron, a halogen is able to complete its octet and achieve a noble gas configuration. This exothermic process expels energy in the form of heat. Electron affinity refers to the energy dissipated by a gaseous species when it gains an electron. Note the electron affinity is essentially the opposite concept from ionization energy. Because this is an exothermic process, ΔHrxn has a negative sign; however, the electron affinity is reported as a positive number. This is because electron affinity refers to the energy dissipated: if 200 kJ mol of energy is released, Δ H rxn = − 200 kJ mol , and the electron affinity is 200 kJ mol . The stronger the electrostatic pull (the higher the Zeff) between the nucleus and the valence shell electrons, the greater the energy release will be when the atom gains the electron. Thus, electron affinity increases across a period from left to right. Because the valence shell is farther away from the nucleus as the principal quantum number increases, electron affinity decreases in a group from top to bottom. Groups IA and IIA (Groups 1 and 2) have very low electron affinities, preferring to give up electrons to achieve the octet configuration of the noble gas in the previous period. Conversely, Group VIIA (Group 17) elements have very high electron affinities because they need to gain only one electron to achieve the octet configuration of the noble gases (Group VIIIA or Group 18) in the same period. Although the noble gases would be predicted to have the highest electron affinities according to the trend, they actually have electron affinities on the order of zero because they already possess a stable octet and cannot readily accept an electron. Most metals also have low electron affinity values, as can be seen in Figure 2.8.
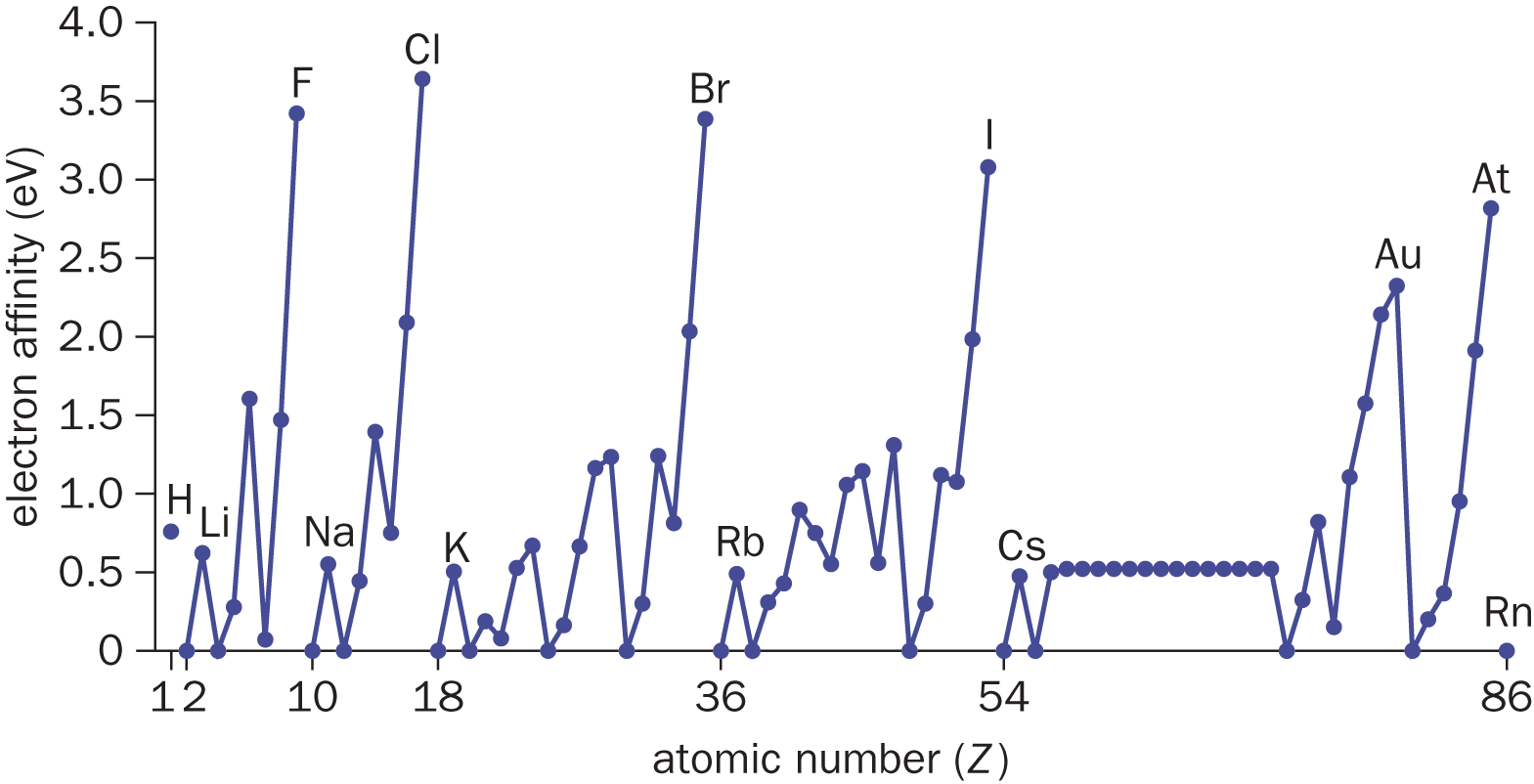
Figure 2.8. Electron Affinities (in eV) of the Elements
Electronegativity
Electronegativity is a measure of the attractive force that an atom will exert on an electron in a chemical bond. The greater the electronegativity of an atom, the more it attracts electrons within a bond. Electronegativity values are related to ionization energies: the lower the ionization energy, the lower the electronegativity; the higher the ionization energy, the higher the electronegativity. The first three noble gases are exceptions: despite their high ionization energies, these elements have negligible electronegativity because they do not often form bonds.
The electronegativity value is a relative measure, and there are different scales used to express it. The most common scale is the Pauling electronegativity scale, which ranges from 0.7 for cesium, the least electronegative (most electropositive) element, to 4.0 for fluorine, the most electronegative element. Electronegativity increases across a period from left to right and decreases in a group from top to bottom. Figure 2.9 shows the electronegativity values of the elements.
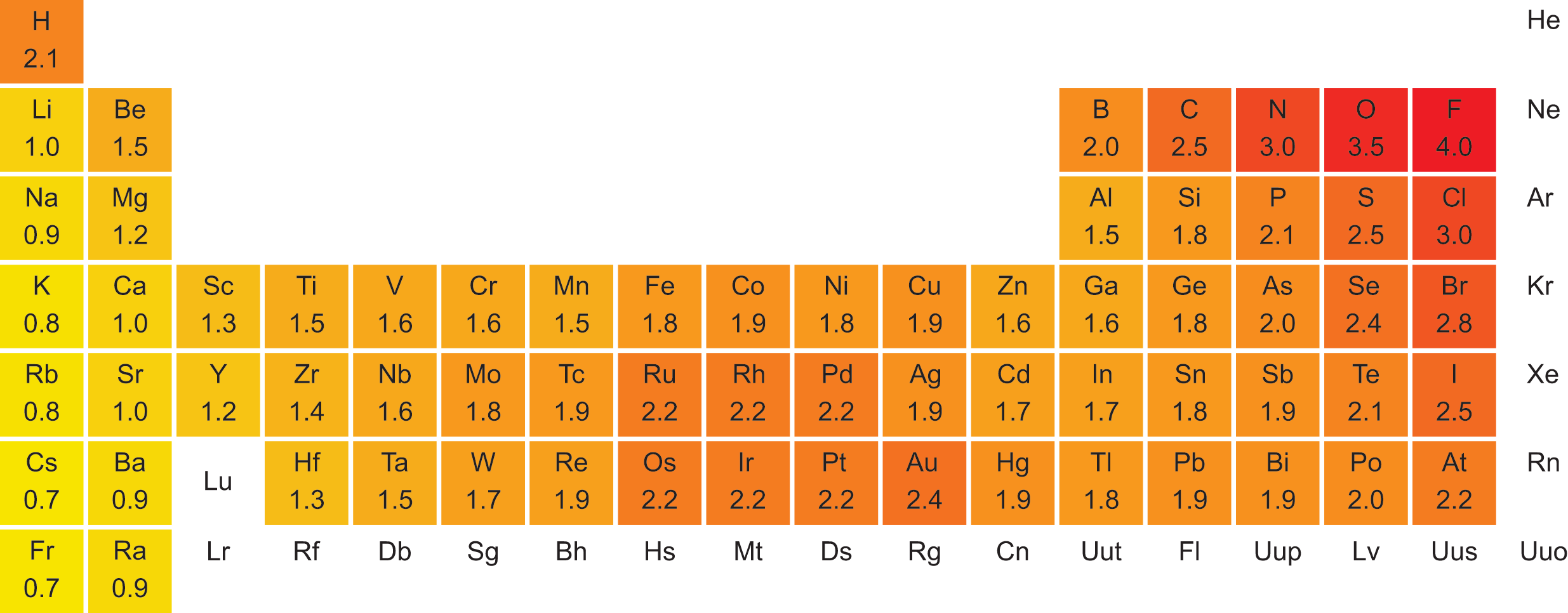
Figure 2.9. Pauling Electronegativity Values of the Elements
KEY CONCEPT
- Cs = largest, least electronegative
- F = smallest, most electronegative
The periodic trends are summarized together in Figure 2.10.
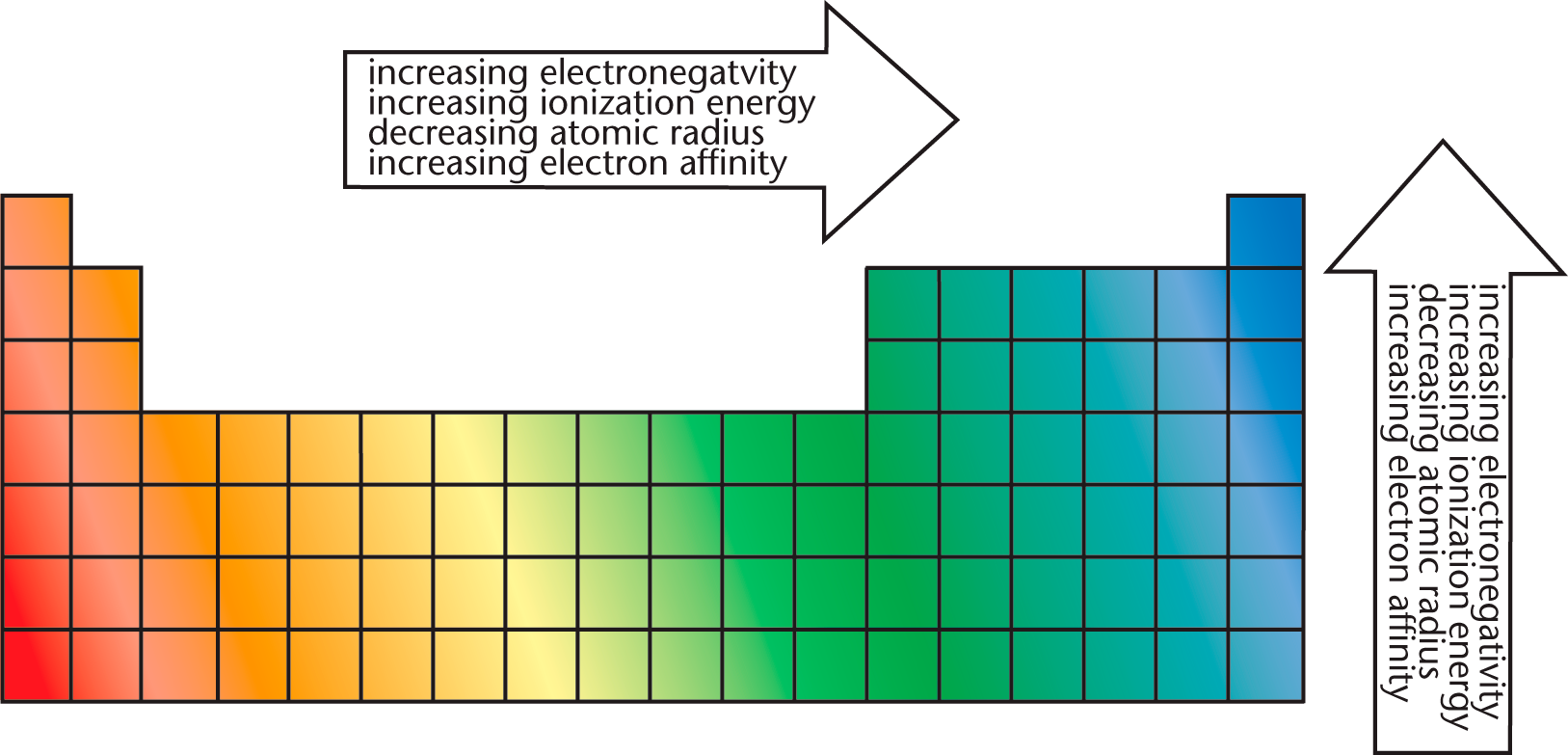
Figure 2.10. Periodic Trends
KEY CONCEPT
- Periodic Trends
- Left → Right
- Atomic radius ↓
- Ionization energy →
- Electron affinity →
- Electronegativity →
- Top → Bottom
- Atomic radius →
- Ionization energy ↓
- Electron affinity ↓
- Electronegativity ↓
Note: Atomic radius is always opposite the other trends. Ionic radius is variable.
MCAT CONCEPT CHECK 2.3:
Before you move on, assess your understanding of the material with these questions.
-
In each of the following pairs, which has the larger radius?
- F or F– _____________________
- K or K+ _____________________
-
Rank the following elements by decreasing first ionization energy: calcium (Ca), carbon (C), germanium (Ge), potassium (K)
- ____________________
- ____________________
- ____________________
- ____________________
-
Rank the following elements by increasing electron affinity: barium (Ba), copper (Cu), sulfur (S), yttrium (Y)
- ____________________
- ____________________
- ____________________
- ____________________
-
Rank the following elements by decreasing electronegativity: antimony (Sb), neon (Ne), oxygen (O), thallium (Tl)
- ____________________
- ____________________
- ____________________
- ____________________
-
Rank the following elements by increasing atomic radius: niobium (Nb), praseodymium (Pr), tantalum (Ta), xenon (Xe)
- ____________________
- ____________________
- ____________________
- ____________________
2.4 The Chemistry of Groups
LEARNING OBJECTIVES
After Chapter 2.4, you will be able to:
- Identify the groups on the periodic table by the properties they exhibit
- Connect periodic table groups 1, 2, 16, 17, 18, and 3–12 to their common names
What follows is a discussion of the major groups you are likely to encounter on the MCAT. While it is rare to be tested on every group, it is important to understand the overarching trends we have already discussed and how they relate across different groups.
Alkali Metals (IA)
The alkali metals (Group IA or Group 1) possess most of the classic physical properties of metals, except that their densities are lower than those of other metals (as described for lithium earlier in this chapter). The alkali metals have only one loosely bound electron in their outermost shells. Their Zeff values are very low, giving them the largest atomic radii of all the elements in their respective periods. This low Zeff value also explains the other trends: low ionization energies, low electron affinities, and low electronegativities. Alkali metals easily lose one electron to form univalent cations, and they react readily with nonmetals—especially the halogens—as in NaCl. Figure 2.11 illustrates the reaction of an alkali metal with water, a stereotypically violent reaction.

Figure 2.11. Reaction of Sodium with Water Group IA metals react violently with water, forming strong bases.
REAL WORLD
Due to their high reactivity with water and air, most alkali metals are stored in mineral oil.
Alkaline Earth Metals (IIA)
The alkaline earth metals (Group IIA or Group 2) also possess many properties characteristic of metals. They share most of the characteristics of the alkali metals, except that they have slightly higher effective nuclear charges and thus slightly smaller atomic radii. They have two electrons in their valence shell, both of which are easily removed to form divalent cations. Together, the alkali and alkaline earth metals are called the active metals because they are so reactive that they are not naturally found in their elemental (neutral) state.
Chalcogens (VIA)
The chalcogens (Group VIA or Group 16) are an eclectic group of nonmetals and metalloids. While not as reactive as the halogens, they are crucial for normal biological functions. They each have six electrons in their valence electron shell and, due to their proximity to the metalloids, generally have small atomic radii and large ionic radii. Oxygen is the most important element in this group for many reasons; it is one of the primary constituents of water, carbohydrates, and other biological molecules. Sulfur is also an important component of certain amino acids and vitamins. Selenium also is an important nutrient for microorganisms and has a role in protection from oxidative stress. The remainder of this group is primarily metallic and generally toxic to living organisms. It is important to note that, at high concentrations, many of these elements—no matter how biologically useful—can be toxic or damaging.
BRIDGE
Many of the molecules discussed in metabolism, covered in Chapters 9 through 12 of MCAT Biochemistry Review, utilize lighter nontoxic elements from the chalcogen group (oxygen and sulfur). Many of the heavier chalcogens are toxic metals.
Halogens (VIIA)
The halogens (Group VIIA or Group 17) are highly reactive nonmetals with seven valence electrons. These elements are desperate to complete their octets by gaining one additional electron. The physical properties of this group are variable. At standard conditions, the halogens range from gaseous (F2 and Cl2) to liquid (Br2) to solid (I2) forms. Their chemical reactivity is more uniform, and, due to their very high electronegativities and electron affinities, they are especially reactive toward the alkali and alkaline earth metals. Fluorine (F) has the highest electronegativity of all the elements. The halogens are so reactive that they are not naturally found in their elemental state but rather as ions (called halides) or diatomic molecules. Diatomic iodine at standard conditions can be seen in Figure 2.12.

Figure 2.12. Iodine in Standard State (Diatomic Iodine)
MCAT EXPERTISE
Halogens are frequently tested on the MCAT. Remember that they only need one more electron to have a noble gas-like electron configuration (full valence shell).
Noble Gases (VIIIA)
The noble gases (Group VIIIA or Group 18) are also known as inert gases because they have minimal chemical reactivity due to their filled valence shells. They have high ionization energies, little or no tendency to gain or lose electrons, and (for He, Ne, and Ar, at least), no measurable electronegativities. The noble gases have extremely low boiling points and exist as gases at room temperature. Noble gases have found a commercial niche as lighting sources, as seen in Figure 2.13, due to their lack of reactivity.
Figure 2.13. Noble Gases Used in “Neon” Signs
Transition Metals (B)
The transition elements (Groups IB to VIIIB or Groups 3 to 12) are considered to be metals and as such have low electron affinities, low ionization energies, and low electronegativities. These metals are very hard and have high melting and boiling points. They tend to be quite malleable and are good conductors due to the loosely held electrons that progressively fill the d-orbitals in their valence shells. One of the unique properties of the transition metals is that many of them can have different possible charged forms or oxidation states because they are capable of losing different numbers of electrons from the s- and d-orbitals in their valence shells. For instance, copper (Cu) can exist in either the +1 or the +2 oxidation state, and manganese (Mn) can exist in the +2, +3, +4, +6, or +7 oxidation state. Because of this ability to attain different positive oxidation states, transition metals form many different ionic compounds. These different oxidation states often correspond to different colors; solutions with transition metal-containing complexes are often vibrant, as shown in Figure 2.14.

Figure 2.14. Solutions of Transition Metal-Containing Compounds From left to right: cobalt(II) nitrate, Co(NO3)2 (red); potassium dichromate, K2Cr2O7 (orange); potassium chromate, K2CrO4 (yellow); nickel(II) chloride, NiCl2 (green); copper(II) sulfate, CuSO4 (blue); potassium permanganate, KMnO4 (violet)
These complex ions tend to associate in solution either with molecules of water (hydration complexes, such as CuSO4 · 5 H2O) or with nonmetals (such as [Co(NH3)6]Cl3). This ability to form complexes contributes to the variable solubility of certain transition metal-containing compounds. For example, AgCl is insoluble in water but quite soluble in aqueous ammonia due to the formation of the complex ion [Ag(NH3)2]+. The formation of complexes causes the d-orbitals to split into two energy sublevels. This enables many of the complexes to absorb certain frequencies of light—those containing the precise amount of energy required to raise electrons from the lower- to the higher-energy d-orbitals. The frequencies not absorbed (known as the subtraction frequencies) give the complexes their characteristic colors.
BRIDGE
Many transition metals act as cofactors for enzymes, including vanadium, chromium, manganese, iron, cobalt, nickel, copper, and zinc. Cofactors and coenzymes are discussed in Chapter 2 of MCAT Biochemistry Review.
This brings up an important point about the perception of color: when we perceive an object as a particular color, it is because that color is not absorbed—but rather reflected—by the object. If an object absorbs a given color of light and reflects all others, our brain mixes these subtraction frequencies and we perceive the complementary colorof the frequency that was absorbed. This is best illustrated with an example. Caroteneis a photosynthetic pigment that strongly absorbs blue light but reflects other colors. Thus, our brains interpret the color of carotene as the result of white light minus blue light, which is yellow light. The complementary colors are shown in Figure 2.15; while the MCAT is unlikely to ask you to name the complement of a given color, the relationship between complementary colors, as explained here, is fair game. It should also be noted that the manner in which colors mix in this scheme is distinctly different from mixing, say, paint colors. The differences between these two schemes, termed additive and subtractive color mixing, respectively, are outside the scope of the MCAT.

Figure 2.15. Red–Green–Blue (Additive) Color Wheel Each color is directly across the circle from its complementary color; commonly referenced complementary pairs include red/cyan, green/magenta, and blue/yellow.
MCAT CONCEPT CHECK 2.4:
Before you move on, assess your understanding of the material with this question.
-
For each of the properties listed below, write down the groups of the periodic table that exhibit those properties.
- High reactivity to water: ________________________________
- Six valence electrons: ________________________________
- Contain at least one metal: ________________________________
- Multiple oxidation states: ________________________________
- Negative oxidation states: ________________________________
- Possess a full octet in the neutral state: _____________________
Conclusion
Now that we have completed our review of the Periodic Table of the Elements, commit to understanding (not just memorizing) the trends of physical and chemical properties of the elements. They will help you quickly answer many questions on the MCAT. As you progress through the chapters of this book, a foundational understanding of the elements will help you develop a richer, more nuanced understanding of their general and particular behaviors. Topics in general chemistry that may have given you trouble in the past will be understandable from the perspective of the behaviors and characteristics that you have reviewed here.
More broadly, you will see a diverse array of elements from the groups we have discussed here that are critical or detrimental to biological function. In addition, you may begin to see why the human body utilizes certain elements for specific purposes, taking advantage of the periodic trends discussed here.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
The Periodic Table
- The Periodic Table of the Elements organizes the elements according to their atomic numbers and reveals a pattern of similar chemical and physical properties among elements.
- Rows are called periods and are based on the same principal energy level, n.
- Columns are called groups. Elements in the same group have the same valence shell electron configuration.
Types of Elements
- The elements on the periodic table belong to one of three types.
- Metals are shiny (lustrous), conduct electricity well, and are malleable and ductile. Metals are found on left side and middle of the periodic table.
- Nonmetals are dull, poor conductors of electricity, and are brittle. Nonmetals are found on right side of the periodic table.
- Metalloids possess characteristics of both metals and nonmetals and are found in a stair-step pattern starting with boron (B).
Periodic Properties of the Elements
- Effective nuclear charge (Zeff) is the net positive charge experienced by electrons in the valence shell and forms the foundation for all periodic trends.
- Zeff increases from left to right across a period, with little change in value from top to bottom in a group.
- Valence electrons become increasingly separated from the nucleus as the principal energy level, n, increases from top to bottom in a group.
- Atomic radius decreases from left to right across a period and increases from top to bottom in a group.
- Ionic radius is the size of a charged species. The largest nonmetallic ionic radii and the smallest metallic ionic radii exist at the metalloid boundary.
- Cations are generally smaller than their corresponding neutral atom.
- Anions are generally larger than their corresponding neutral atom.
- Ionization energy is the amount of energy necessary to remove an electron from the valence shell of a gaseous species; it increases from left to right across a period and decreases from top to bottom in a group.
- Electron affinity is the amount of energy released when a gaseous species gains an electron in its valence shell; it increases from left to right across a period and decreases from top to bottom in a group.
- Electronegativity is a measure of the attractive force of the nucleus for electrons within a bond; it increases from left to right across a period and decreases from top to bottom in a group.
The Chemistry of Groups
- Alkali metals typically take on an oxidation state of +1 and prefer to lose an electron to achieve a noble gas-like configuration; they and the alkaline earth metals are the most reactive of all metals.
- Alkaline earth metals take on an oxidation state of +2 and can lose two electrons to achieve noble gas-like configurations.
- Chalcogens take on oxidation states of −2 or +6 (depending on whether they are nonmetals or metals, respectively) in order to achieve noble gas configuration. They are very biologically important.
- Halogens typically take on an oxidation state of −1 and prefer to gain an electron to achieve noble gas-like configurations; these nonmetals have the highest electronegativities.
- Noble gases have a fully filled valence shell in their standard state and prefer not to give up or take on additional electrons; they have very high ionization energies and (for He, Ne, and Ar), virtually nonexistent electronegativities and electron affinities.
- Transition metals are unique because they take on multiple oxidation states, which explains their ability to form colorful complexes with nonmetals in solution and their utility in certain biological systems.
ANSWERS TO CONCEPT CHECKS
**2.1**
- The modern periodic table is arranged in order by atomic number.
-
Element A or B Element A or B Element A or B Element A or B Ag B Al A K A P A
Pb A Li A Pu B U B
Cu B Cf B Zn B B A
N A Np B O A He A
**2.2**
- Metals have luster. Nonmetals have poor conductivity. Metalloids exhibit brittleness but good conductivity. Any answers within each of these categories are acceptable.
-
Element Class Element Class Element Class Element Class Ag M Al M K M P NM
Pb M Li M Pu M U M
Cu M As MO Zn M B MO
Si MO Np M O NM He NM
**2.3**
- F–> F; K > K+. The ionic radii of anions are larger than the associated atomic radii, while the ionic radii of cations are smaller.
- Ionization energy: carbon > germanium > calcium > potassium
- Electron affinity: barium < yttrium < copper < sulfur
- Electronegativity: oxygen > antimony > thallium > neon
- Atomic radius: xenon < niobium < tantalum < praseodymium
**2.4**
-
- High reactivity to water: Groups 1 and 2
- Six valence electrons: Groups 6 and 16
- Contain at least one metal: Groups 1 through 15
- Multiple oxidation states: All groups; most notably Groups 3 through 12 (transition metals)
- Negative oxidation states: Almost all groups; most notably Groups 14 through 17 (nonmetals)
- Possess a full octet in the neutral state: Group 18
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. B
The periodic table is organized into periods (rows) and groups (columns). Groups (columns) are particularly significant because they represent sets of elements with the same valence electron configuration, which in turn will dictate many of the chemical properties of those elements. Although (A) is true, the fact that both ions are positively charged does not explain the similarity in chemical properties; most metals produce positively charged ions. (C) is not true because lithium and sodium are in the same group, not period. Finally, although lithium and sodium do have relatively low atomic weights, so do several other elements that do not share the same properties, eliminating (D).
2. A
As one moves from top to bottom in a group (column), extra electron shells accumulate, despite the fact that the valence configurations remain identical. These extra electron shells provide shielding between the positive nucleus and the outermost electrons, decreasing the electrostatic attraction and increasing the atomic radius. Because carbon and silicon are in the same group, and silicon is farther down in the group, silicon will have a larger atomic radius because of its extra electron shell.
3. B
This question requires knowledge of the trends of electronegativity within the Periodic Table. Electronegativity increases as one moves from left to right for the same reasons that effective nuclear charge increases. Electronegativity decreases as one moves down the Periodic Table because there are more electron shells separating the nucleus from the outermost electrons. In this question, chlorine is the furthest toward the top-right corner of the Periodic Table, making (B) the correct answer.
4. C
Ionization energy increases from left to right, so the first ionization energy of lithium is lower than that of beryllium. Second ionization energy is always larger than first ionization energy, so beryllium’s second ionization energy should be the highest value. This is because removing an additional electron from Be+ requires one to overcome a significantly larger electrostatic force.
5. B
Antimony (Sb) is on the right side of the periodic table, but not far right enough to be a nonmetal, (D). It certainly does not lie far enough to the right to fall in Group VIIA (Group 17), which would classify it as a halogen, (C). While sources have rarely classified antimony as a metal, (A), it is usually classified as a metalloid, (B).
6. C
Electronegativity describes how strong an attraction an element will have for electrons in a bond. A nucleus with a larger effective nuclear charge will have a higher electronegativity; Zeff increases toward the right side of a period. A stronger nuclear pull will also lead to increased first ionization energy, as the forces make it more difficult to remove an electron. The vertical arrow can be explained by the size of the atoms. As size decreases, the positive charge becomes more effective at attracting electrons in a chemical bond (higher electronegativity), and the energy required to remove an electron (ionization energy) increases.
7. C
All four descriptions of metals are true, but the most significant property that contributes to the ability of metals to conduct electricity is the fact that they have valence electrons that can move freely. Malleability, (A), is the ability to shape a material with a hammer, which does not play a role in conducting electricity. The low electronegativity and high melting points of metals, (B) and (D), also do not play a major role in the conduction of electricity.
8. B
This block represents the alkaline earth metals, which form divalent cations, or ions with a +2 charge. All of the elements in Group IIA have two electrons in their outermost s subshell. Because loss of these two electrons would leave a full octet as the outermost shell, becoming a divalent cation is a stable configuration for all of the alkaline earth metals. Although some of these elements might be great conductors, they are not as effective as the alkali metals, eliminating (A). (C) is also incorrect because, although forming a divalent cation is a stable configuration for the alkaline earth metals, the second ionization energy is still always higher than the first. Finally, (D) is incorrect because atomic radii increase when moving down a group of elements because the number of electron shells increases.
9. B
Iron is a transition metal. Transition metals can often form more than one ion. Iron, for example, can be Fe2+ or Fe3+. The transition metals, in these various oxidation states, can often form hydration complexes with water. Part of the significance of these complexes is that, when a transition metal can form a complex, its solubility within the related solvent will increase. The other ions given might dissolve readily in water, but because none of them are transition metals, they will not likely form complexes.
10. D
This question is simple if one recalls that periods refer to the rows in the periodic table, while groups or families refer to the columns. Within the same period, an additional valence electron is added with each step toward the right side of the table.
11. C
Phosphorous and arsenic are both found in group 5A, indicating that they have a similar number of valence electrons. Since the valence electron configuration is one of the main determinants of bonding, elements with similar valence states react similarly, supporting (C).
12. D
The correct answer to this question may be surprising because this question illustrates an important exception to a trend. Electron affinity generally increases as one moves up and to the right on the periodic table. Based on this trend alone, one might expect that the atom fluorine, depicted in (B), would be correct. However, the correct answer is chlorine, (D), even despite the fact that chlorine is below fluorine on the periodic table. The diagrams in the answer choices help make sense of this apparent exception: Fluorine and chlorine have the same valence electron configuration and have a similar amount of effective nuclear charge. However, when an electron is added to fluorine's valence shell, that negative charge is added to the second energy shell, which is quite small in physical size, meaning that the new electron is crowded into a small energy shell with seven other electrons. As a result, energy is still released when an electron is added to fluorine, but not as much energy as might be predicted by the general trend alone. By contrast, when an additional electron is added to a chlorine atom, that new electron adds to the third energy shell, so there is less repulsion among the valence electrons, leading to a slightly greater electron affinity for chlorine. Thus, chlorine, (D), has the highest electron affinity and is the right answer.
13. D
The effective nuclear charge refers to the strength with which the protons in the nucleus can pull on electrons. This phenomenon helps to explain electron affinity, electronegativity, and ionization energy. The effective nuclear charge can be calculated by subtracting the number of non-valence electrons from the number of protons. In (A) and(B), chlorine has 17 protons and 10 non-valence electrons, giving it a Zeff of 7. Next, elemental potassium, (C), has the lowest effective nuclear charge because it contains additional inner shells that shield its valence electron from the nucleus. With 19 protons and 18 non-valence electrons, it has a Zeff of 1. (D), ionic potassium, has a higher effective nuclear charge than any of the other options. It has the same electron configuration as Cl− (10 non-valence electrons) but contains two extra protons in its nucleus, and thus has a Zeff of 9
14. D
Ionic bonds are formed through unequal sharing of electrons. These bonds typically occur because the electron affinities of the two bonded atoms differ greatly. For example, the halogens have high electron affinities because adding a single electron to their valence shells would create full valence shells. In contrast, the alkaline earth metals have very low electron affinities and are more likely to be electron donors because the loss of two electrons would leave them with full valence shells. (A) states the opposite and is incorrect because the halogens have high electron affinity and the alkaline earth metals have low electron affinity. (B) is incorrect because equal sharing of electrons is a classic description of covalent bonding, not ionic. (C) is a true statement, but is not relevant to why ionic bonds form.
15. C
When n = 3, l = 0, 1, or 2. The highest value for l in this case is 2, which corresponds to the d subshell. Although the 3d block appears to be part of the fourth period, it still has the principal quantum number n = 3. In general, the subshells within an energy shell increase in energy as follows: s < p < d < f (although there is no 3f subshell).
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
General Chemistry Chapter 1
Atomic Structure
General Chemistry Chapter 3
Bonding and Chemical Interactions
General Chemistry Chapter 4
Compounds and Stoichiometry
Organic Chemistry Chapter 3
Bonding
Physics and Math Chapter 5
Electrostatics and Magnetism
Physics and Math Chapter 9
Atomic and Nuclear Phenomena