Chapter 12: Electrochemistry

Chapter 12: Electrochemistry
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- Rusting occurs due to the oxidation–reduction reaction of iron with environmental oxygen:
4 Fe (s) + 3 O2 (g) → 2 Fe2O3 (s)
Some metals cannot react with oxygen in this fashion.
Which of the following best explains why iron can?
- Iron has a more positive reduction potential than those metals, making it more likely to donate electrons to oxygen.
- Iron has a more positive reduction potential than those metals, making it more likely to accept electrons from oxygen.
- Iron has a less positive reduction potential than those metals, making it more likely to donate electrons to oxygen.
- Iron has a less positive reduction potential than those metals, making it more likely to accept electrons from oxygen.
-
Given the following standard reduction potentials:
Zn 2 + + 2 e − → Zn E red ∘ = − 0.763 V Ag + + e − → Ag E red ∘ = + 0.337 V
What is the standard electromotive force of the following reaction?
Zn2+ + 2 Ag → 2 Ag+ + Zn
- –2.2 V
- –1.1 V
- +1.1 V
- +2.2 V
-
Consider the following data:
Hg 2 + + 2 e − → Hg E red ∘ = + 0.85 V Cu + + e − → Cu E red ∘ = + 0.52 V Zn 2 + + 2 e − → Zn E red ∘ = − 0.76 V Al 3 + + 3 e − → Al E red ∘ = − 1.66 V
The anode of a certain galvanic cell is composed of copper. Which of the metals from the data table can be used at the cathode, assuming equal concentrations of the two electrolyte solutions?
- Hg
- Cu
- Zn
- Al
-
An electrolytic cell is filled with water. Which of the following will move toward the cathode of such a cell?
- H+ ions
- O2– ions
- Electrons
- I only
- II only
- I and III only
- II and III only
-
If the value of E°cell is known, what other data is needed to calculate ΔG°?
- Equilibrium constant
- Reaction quotient
- Temperature of the system
- Half-reactions of the cells
-
Which of the following compounds is LEAST likely to be found in the salt bridge of a galvanic cell?
- NaCl
- SO3
- MgSO3
- NH4NO3
-
If the surface area of electrode material in an electrochemical cell is tripled, what else is necessarily tripled?
- E°cell
- Current
- Keq
- I only
- II only
- I and II only
- II and III only
-
Which of the following can alter the emf of an electrochemical cell?
- The mass of the electrodes
- The length of the wire connecting the half-cells
- The overall size of the battery
- The temperature of the solutions in the half-cells
-
Which of the following statements could be true about a Na–Cd cell, based on the information below?
Na + + e − → Na E red ∘ = − 2.71 V Cd 2 + + 2 e − → Cd E red ∘ = − 0.40 V
- It is a galvanic cell, and sodium is the cathode.
- It is an electrolytic cell, and cadmium is the anode.
- It is a galvanic cell, with E°cell = 3.11 V.
- It is an electrolytic cell, with E°cell = −3.11 V.
-
Which of the following expressions correctly describes the relationship between standard electromotive force and standard change in free energy?
- ΔG° = −nF(E°red,anode − E°red,cathode)
- E cell ∘ = n F R T ln K eq
- E cell ∘ = − R T n F ln K eq
- ΔG° = nF(E°red,anode − E°red,cathode)
-
Which of the following choices is indicative of a spontaneous reaction, assuming standard conditions?
- E°cell is negative.
- Q = Keq
- The cell is a concentration cell.
- Keq > 1
-
For a cell with the following half-reactions:
Anode: SO2 + 2 H2O → SO42− + 4 H+ + 2 e−
Cathode: Pd2+ + 2 e− → Pd
How would decreasing the pH of the solution inside the cell affect the electromotive force (emf)?
- The emf would decrease.
- The emf would remain the same.
- The emf would increase.
- The emf would become zero.
-
An electrolytic cell necessarily has:
- ΔS° > 0
- ΔG° < 0
- Keq < 1
- E°cell > 0
-
Which of the following is the best explanation for the fact that a larger mass of electrodes are required for lead–acid batteries, as compared to other batteries, to produce a certain current?
- The lead–acid electrolyte, sulfuric acid, is diprotic and incompletely dissociates in solution.
- The energy density of lead–acid electrodes is higher than that of other batteries.
- The electrolytes in other batteries less readily dissociate than those of lead–acid batteries.
- The energy density of lead–acid electrodes is lower than that of other batteries.
-
Which of the following best describes why over-charging a Ni–Cd battery is not detrimental?
- The energy density of a Ni–Cd battery is high, so it can store more charge than other batteries per its mass.
- The electrodes of a Ni–Cd battery can discharge through the circuit when they are fully charged.
- The Ni–Cd battery will stop accepting electrons from an outside source when its electrodes are recharged.
- Ni–Cd batteries have a high surge current and can dissipate the overcharge before damage can occur to electrodes.
Answer Key
- C
- B
- A
- C
- D
- B
- B
- D
- B
- D
- D
- A
- C
- D
- C
Chapter 12: Electrochemistry
CHAPTER 12
ELECTROCHEMISTRY
In This Chapter
12.1 Electrochemical Cells
Galvanic (Voltaic) Cells
Electrolytic Cells
Concentration Cells
Rechargeable Cells
Electrode Charge Designations
12.2 Cell Potentials
Reduction Potentials
The Electromotive Force
12.3 Electromotive Force and Thermodynamics
Gibbs Free Energy
Reaction Quotients
Equilibria
Concept Summary
CHAPTER PROFILE
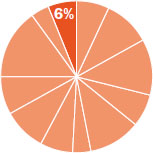
The content in this chapter should be relevant to about 6% of all questions about general chemistry on the MCAT
This chapter covers material from the following AAMC content categories:
1D: Principles of bioenergetics and fuel molecule metabolism
3A: Structure and functions of the nervous and endocrine systems and ways in which these systems coordinate the organ systems
4C: Electrochemistry and electrical circuits and their elements
5E: Principles of chemical thermodynamics and kinetics
Introduction
The mitochondria are powerhouses of energy. Their primary purpose is to manufacture a deliverable and usable form of energy. By now, you are well aware of the complex processes by which the potential energy in the chemical bonds of carbohydrates, amino acids, and lipids is converted into the potential energy of the phosphate bond in adenosine triphosphate (ATP). ATP is then delivered to different parts of the cell, where it is used to energize most of the processes essential to the maintenance of life.
The mitochondria generate tremendous amounts of ATP—in humans, the average daily turnover of ATP is more than 50 kilograms! Without a continuous supply and replenishment of ATP, we wouldn’t survive. ATP powers the contraction of our heart muscle and maintains the membrane potential essential for neurological function, among thousands of other essential roles. How do the mitochondria manufacture these packets of life-sustaining energy? Remember that mitochondria rely on their double-membrane structure to carry out the electron transport chain and oxidative phosphorylation. As such, mitochondria truly act as batteries of the cell. In fact, note the similarity between the proton-motive force of the mitochondria and the electromotive force of electrochemistry. Are these two terms the same thing or—at the very least—similar in nature?
Indeed, mitochondria and batteries do function in similar ways. Specifically, mitochondria function most similarly to concentration cells. In both concentration cells and mitochondria, a concentration gradient of ions between two separated compartments—connected to each other by some means of charge conduction—establishes an electrical potential difference (voltage). This voltage, called electromotive force in concentration cells and proton-motive force in the mitochondria, provides the drive to move charge from one compartment to the other, creating current. In the concentration cell, an oxidation–reduction reaction takes place, and electrons move in the direction that causes the concentration gradient to be dissipated. In the mitochondria, the charge buildup is in the form of a hydrogen ion (proton) gradient between the intermembrane space and the matrix. Embedded in the inner membrane is ATP synthase, which serves a dual role as a proton channel and a catalyst for the formation of the high-energy phosphate bond in ATP. As the hydrogen ions flow down their electrochemical gradient, energy is dissipated, and this energy is harnessed by ATP synthase to form ATP.
In this final chapter of MCAT General Chemistry Review, we will focus our attention on the study of various electrochemical cells. Utilizing our knowledge of oxidation–reduction reactions from Chapter 11, we will study how these principles can be applied to create different types of electrochemical cells, including galvanic (voltaic), electrolytic, and concentration cells. Regarding the thermodynamics of electrochemistry, we will focus on the significance of reduction potentials and examine the relationship between electromotive force, the equilibrium constant, and Gibbs free energy.
12.1 Electrochemical Cells
LEARNING OBJECTIVES
After Chapter 12.1, you will be able to:
- Distinguish between electrolytic and galvanic cells
- Describe electrolytic and galvanic cells
- Predict which electrode will act as the cathode or anode in an electrolytic or a galvanic cell
- Calculate ΔGand emf values for a given galvanic or electrolytic cell
- Apply the Nernst equation to electrochemical cell questions
Electrochemical cells are contained systems in which oxidation–reduction reactions occur. There are three fundamental types of electrochemical cells: galvanic cells (also known as voltaic cells), electrolytic cells, and concentration cells. In addition, there are specific commercial cells such as Ni–Cd batteries through which we can understand these fundamental models.
Galvanic cells and concentration cells house spontaneous reactions, whereas electrolytic cells contain nonspontaneous reactions. Remember that spontaneity is indicated by the change in Gibbs free energy, ΔG. All three types contain electrodes where oxidation and reduction take place. For all electrochemical cells, the electrode where oxidation occurs is called the anode, and the electrode where reduction occurs is called the cathode. Other descriptors of electrochemical cells include the electromotive force (emf), which corresponds to the voltage or electrical potential difference of the cell. If the emf is positive, the cell is able to release energy (ΔG < 0), which means it is spontaneous. If the emf is negative, the cell must absorb energy (ΔG > 0), which means it is nonspontaneous.
MNEMONIC
Electrodes in an electrochemical cell: AN OX and a RED CAT
The anode is the site of oxidation; reduction occurs at the cathode.
Furthermore, we can also state that, for all electrochemical cells, the movement of electrons is from anode to cathode, and the current (I) runs from cathode to anode. This point can be a point of confusion among students. In physics, it is typical to state that current is the direction of flow of a positive charge through a circuit; this model was first proposed by Ben Franklin and continues to be used among physicists. Modern chemists are interested in the flow of electrons, but may discuss the current (a theoretical flow of positive charge) as a proxy for the flow of electrons; the current and the flow of electrons are always of equal magnitude but in opposite directions.
KEY CONCEPT
Electrons move through an electrochemical cell opposite to the flow of current (I).
Last, it is important to note that all batteries are influenced by temperature changes. For instance, lead–acid batteries in cars, like most galvanic cells, tend to fail most in cold weather. The thermodynamic reasons behind this will be discussed later in this chapter.
Galvanic (Voltaic) Cells
All of the nonrechargeable batteries you own are galvanic cells, also called voltaic cells. Accordingly, because household batteries are used to supply energy to a flashlight or remote control, the reactions in these cells must be spontaneous. This means that the reaction’s free energy is decreasing (ΔG < 0) as the cell releases energy to the environment. By extension, if the free energy change is negative for these cells, their electromotive force (Ecell) must be positive; the free energy change and electromotive force always have opposite signs.
REAL WORLD
Galvanic cells are commonly used as batteries; to be worthwhile (that is, producing energy to power some device or appliance), these batteries must be spontaneous!
Let’s examine the inner workings of a galvanic (voltaic) cell. Two electrodes of distinct chemical identity are placed in separate compartments, which are called half-cells. The two electrodes are connected to each other by a conductive material, such as a copper wire. Along the wire, there may be other various components of a circuit, such as resistors or capacitors, but for now, we’ll focus on the battery itself.
Surrounding each of the electrodes is an aqueous electrolytesolution composed of cations and anions. As shown in the Daniell cell illustrated in Figure 12.1, the cations in the two half-cell solutions can be of the same element as the respective metal electrode. Connecting the two solutions is a structure called a salt bridge, which consists of an inert salt. When the electrodes are connected to each other by a conductive material, charge will begin to flow as the result of an oxidation–reduction reaction that is taking place between the two half-cells. The redox reaction in a galvanic cell is spontaneous, and therefore the change in Gibbs free energy for the reaction is negative (ΔG < 0). As the spontaneous reaction proceeds toward equilibrium, the movement of electrons results in a conversion of electrical potential energy into kinetic energy. By separating the reduction and oxidation half-reactions into two compartments, we are able to harness this energy and use it to do work by connecting various electrical devices into the circuit between the two electrodes.
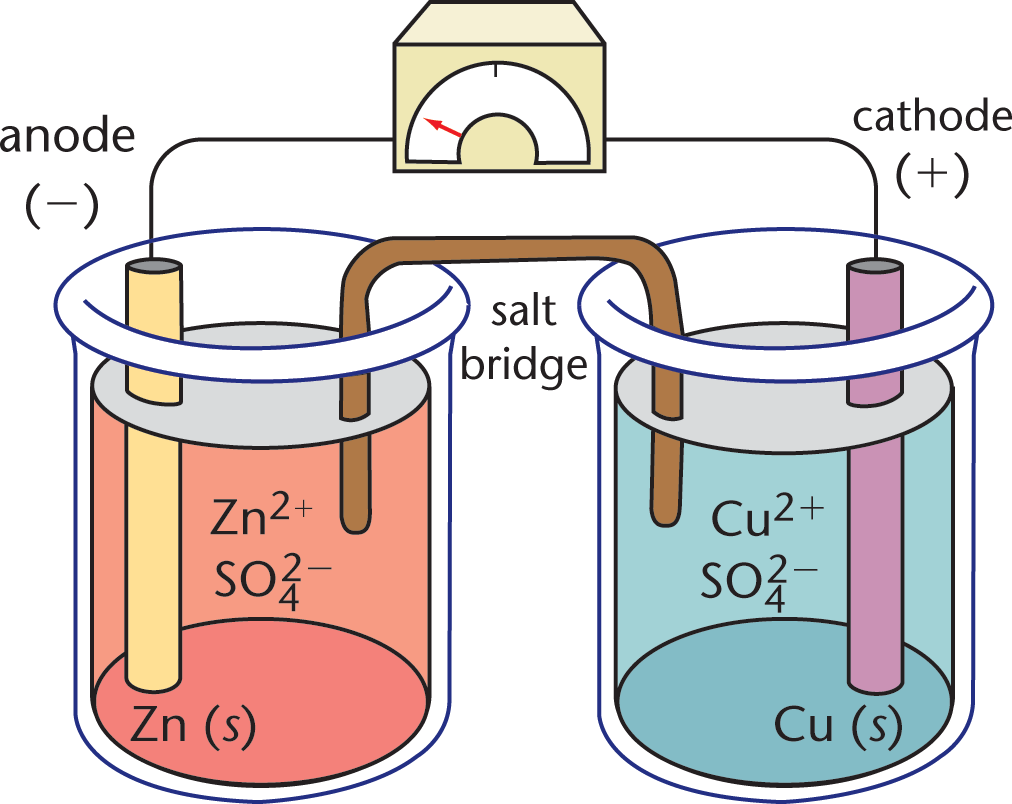
Figure 12.1. Daniell Cell In this galvanic cell, zinc is the anode and copper is the cathode; each electrode is bathed in an electrolyte solution containing its cation and sulfate.
In the Daniell cell, a zinc electrode is placed in an aqueous ZnSO4 solution, and a copper electrode is placed in an aqueous CuSO4 solution. The anode of this cell is the zinc bar where Zn (s) is oxidized to Zn2+ (aq). The cathode is the copper bar, and it is the site of the reduction of Cu2+ (aq) to Cu (s). The half-cell reactions are written as follows:
Zn ( s ) → Zn 2 + ( a q ) + 2 e − E red = − 0.762 V ( anode ) Cu 2 + ( a q ) + 2 e − → Cu ( s ) E red = + 0.340 V ( cathode )
The net reaction is
Zn ( s ) + Cu 2 + ( a q ) → Zn 2 + ( a q ) + Cu ( s ) E cell = + 1.102 V
We will discuss the calculation of cell potential in the next section. For now, appreciate that the calculation can be accomplished by knowing each half-reaction. If the two half-cells were not separated, the Cu2+ ions would react directly with the zinc bar, and no useful electrical work would be done. Because the solutions and electrodes are physically separated, they must be connected by a conductive material to complete the circuit.
However, if only a wire were provided for this electron flow, the reaction would soon stop because an excess positive charge would build up on the anode, and an excess negative charge would build up on the cathode. Eventually, the excessive charge accumulation would provide a countervoltage large enough to prevent the oxidation–reduction reaction from taking place, and the current would cease. This charge gradient is dissipated by the presence of a salt bridge, which permits the exchange of cations and anions. The salt bridge contains an inert electrolyte, usually KCl or NH4NO3, which contains ions that will not react with the electrodes or with the ions in solution. While the anions from the salt bridge (Cl–) diffuse into the solution on the anode side (ZnSO4) to balance out the charge of the newly created Zn2+ ions, the cations of the salt bridge (K+) flow into the solution on the cathode side (CuSO4) to balance out the charge of the sulfate ions left in solution when the Cu2+ ions are reduced to Cu and precipitate onto the electrode. This precipitation process onto the cathode itself can also be called plating or galvanization.
KEY CONCEPT
The purpose of the salt bridge is to exchange anions and cations to balance, or dissipate, newly generated charges.
During the course of the reaction, electrons flow from the zinc anode through the wire and to the copper cathode. A voltmeter can be connected to measure this electromotive force. As mentioned earlier, the anions (Cl–) flow externally from the salt bridge into the ZnSO4, and the cations (K+) flow externally from the salt bridge into the CuSO4. This flow depletes the salt bridge and, along with the finite quantity of Cu2+ in the solution, accounts for the relatively short lifespan of the cell.
MNEMONIC
Electron flow in an electrochemical cell: A → C (order in the alphabet)
Electrons flow from anode to cathode in all types of electrochemical cells.
A cell diagram is a shorthand notation representing the reactions in an electrochemical cell. A cell diagram for the Daniell cell is as follows:
Zn (s) | Zn2+ (1 M) || Cu2+ (1 M) | Cu (s)
The following rules are used in constructing a cell diagram:
- The reactants and products are always listed from left to right in this form: anode | anode solution (concentration) || cathode solution (concentration) | cathode
- A single vertical line indicates a phase boundary.
- A double vertical line indicates the presence of a salt bridge or some other type of barrier.
MCAT EXPERTISE
Recognize and understand the shorthand cell notation for electrochemical cells on Test Day. Passages frequently use this format rather than spelling out which reactions take place at the anode and cathode.
Electrolytic Cells
When comparing and contrasting galvanic and electrolytic cells, it is important to keep straight what remains consistent between the two types of cells and what differs. All types of electrochemical cells have a reduction reaction occurring at the cathode, an oxidation reaction occurring at the anode, a current flowing from cathode to anode, and electron flow from anode to cathode. However, electrolytic cells, in almost all of their characteristics and behavior, are otherwise the opposite of galvanic cells. Whereas galvanic cells house spontaneous oxidation–reduction reactions that generate electrical energy, electrolytic cells house nonspontaneous reactions that require the input of energy to proceed. Therefore, the change in free energy for an electrolytic cell is positive. This type of oxidation–reduction reaction driven by an external voltage source is called electrolysis, in which chemical compounds are decomposed. For example, electrolytic cells can be used to drive the nonspontaneous decomposition of water into oxygen and hydrogen gas. Another example, the electrolysis of molten NaCl, is illustrated in Figure 12.2.
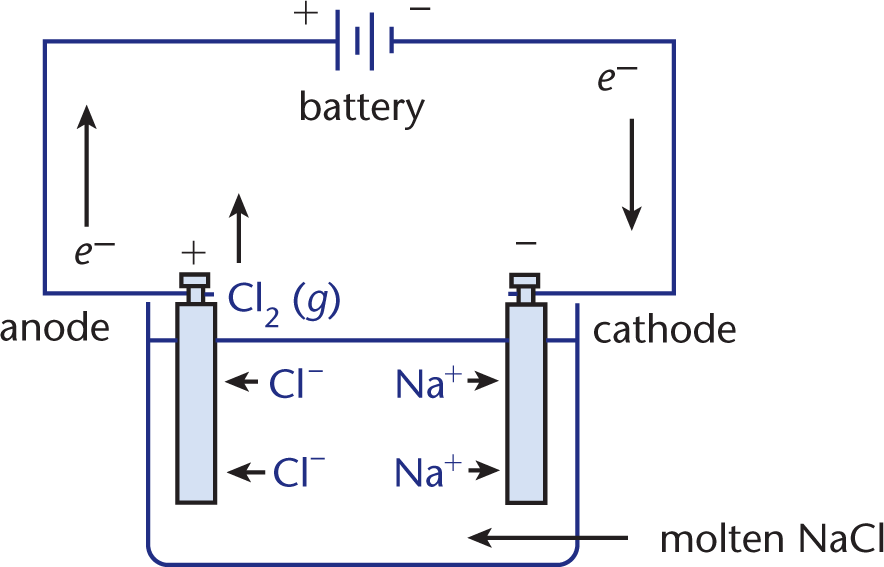
Figure 12.2. Electrolysis of Molten NaCl
KEY CONCEPT
Because electrolysis is nonspontaneous, the electrode (anode or cathode) can consist of any material so long as it can resist the high temperatures and corrosion of the process.
In this electrolytic cell, molten NaCl is decomposed into Cl2 (g) and Na (l). The external voltage source—a battery—supplies energy sufficient to drive the oxidation–reduction reaction in the direction that is thermodynamically unfavorable (nonspontaneous).
In this example, Na+ ions migrate toward the cathode, where they are reduced to Na (l). At the same time, Cl– ions migrate toward the anode, where they are oxidized to Cl2 (g). Notice that the half-reactions do not need to be separated into different compartments; this is because the desired reaction is nonspontaneous. Note that sodium is a liquid at the temperature of molten NaCl; it is also less dense than the molten salt and, thus, is easily removed as it floats to the top of the reaction vessel.
This cell is used in industry as the major means of sodium and chlorine production. You may wonder why one would do so much work to obtain pure sodium and chlorine. Remember that these elements are never found naturally in their elemental form because they are so reactive. Thus, to use elemental sodium or chlorine gas in a reaction, it must be manufactured through processes such as these.
Michael Faraday was the first to define certain quantitative principles governing the behavior of electrolytic cells. He theorized that the amount of chemical change induced in an electrolytic cell is directly proportional to the number of moles of electrons that are exchanged during the oxidation–reduction reaction. The number of moles exchanged can be determined from the balanced half-reaction. In general, for a reaction that involves the transfer of n electrons per atom M:
Mn+ + n e– → M (s)
Equation 12.1
KEY CONCEPT
Faraday’s laws state that the liberation of gas and deposition of elements on electrodes is directly proportional to the number of electrons being transferred during the oxidation–reduction reaction. Here, normality or gram equivalent weight is used. These observations are proxy measurements of the amount of current flowing in a circuit.
According to this equation, one mole of metal M (s) will logically be produced if n moles of electrons are supplied to one mole of Mn+. Additionally, the number of moles of electrons needed to produce a certain amount of M (s) can now be related to the measurable electrical property of charge. One electron carries a charge of 1.6 × 10–19 coulombs (C). The charge carried by one mole of electrons can be calculated by multiplying this number by Avogadro’s number, as follows:
[ 1.6 × 10 − 19 C electron ] [ 6.02 × 10 23 electrons 1 mol e − ] = 96 , 485 C mol e −
This number is called the Faraday constant, and one faraday (F) is equivalent to the amount of charge contained in one mole of electrons (1 F = 96,485 C) or one equivalent. On the MCAT, you should round up this number to 10 5 C mol e − to make calculations more manageable.
KEY CONCEPT
One faraday (F) is equivalent to the amount of charge contained in one mole of electrons (1 F = 96,485 C).
The electrodeposition equation summarizes this process and helps determine the number of moles of element being deposited on a plate:
mol M = I t n F
Equation 12.2
where mol M is the amount of metal ion being deposited at a specific electrode, I is current, t is time, n is the number of electron equivalents for a specific metal ion, and F is the Faraday constant. This equation can also be used to determine the amount of gas liberated during electrolysis.
MNEMONIC
Electrodeposition equation: Calculating Moles of Metal, It is Not Fun.
mol M = I t n F
Example: What mass of copper will be deposited in a Daniell cell if a current of 2 A flows through the cell for 3 hours?
Solution: We will use the equation mol M = I t n F
A Daniell cell uses a copper electrode in copper sulfate (CuSO4) solution. Because the oxidation state of copper in solution is +2, n = 2. Now we can plug into the equation. mol M = I t n F = ( 2 A)(3 hr) ( 3600 s hr ) ( 2 mol e − ) ( 96 , 485 C mol e − ) ≈ 3 × 3600 10 5 = 0.1 mol Cu (actual = 0.11 mol Cu )
Then, we must determine the actual mass of copper being deposited. 0.1 mol Cu should have a mass of 6.35 g because the molar mass of copper is 63.5 g mol (actual = 7.11 g).
Concentration Cells
A concentration cell is a special type of galvanic cell. Like all galvanic cells, it contains two half-cells connected by a conductive material, allowing a spontaneous oxidation–reduction reaction to proceed, which generates a current and delivers energy. The distinguishing characteristic of a concentration cell is in its design: the electrodes are chemically identical. For example, if both electrodes are copper metal, they have the same reduction potential. Therefore, current is generated as a function of a concentration gradient established between the two solutions surrounding the electrodes. The concentration gradient results in a potential difference between the two compartments and drives the movement of electrons in the direction that results in equilibration of the ion gradient. The current will stop when the concentrations of ionic species in the half-cells are equal. This implies that the voltage (V) or electromotive force of a concentration cell is zero when the concentrations are equal; the voltage, as a function of concentrations, can be calculated using the Nernst equation.
In a biological system, a concentration cell is best represented by the cell membrane of a neuron, as shown in Figure 12.3. Sodium and potassium cations, and chlorine anions, are exchanged as needed to produce an electrical potential. The actual value depends on both the concentrations and charges of the ions. In this way, a resting membrane potential (Vm) can be maintained. Disturbances of the resting membrane potential, if sufficiently large, may stimulate the firing of an action potential.

Figure 12.3. The Cell Membrane as an Example of a Concentration Cell The electrochemical gradient created by separation of ions across the cell membrane is analogous to a cell with two electrodes composed of the same material.
BRIDGE
The maintenance of a resting membrane potential is discussed in Chapter 8 of MCAT Biochemistry Review. The conduction of an action potential is discussed in Chapter 4 of MCAT Biology Review. The transfer of ions and electrons during an action potential produces biochemical work.
Rechargeable Cells
A rechargeable cell or rechargeable battery is one that can function as both a galvanic and electrolytic cell.
Lead–Acid Batteries
A lead–acid battery, also known as a lead storage battery, is a specific type of rechargeable battery. As a voltaic cell, when fully charged, it consists of two half-cells—a Pb anode and a porous PbO2 cathode, connected by a conductive material (concentrated 4 M H2SO4). When fully discharged, it consists of two PbSO4 electroplated lead electrodes with a dilute concentration of H2SO4, as shown in Figure 12.4.

Figure 12.4. Lead–Acid Battery When charged (a), the cell contains a Pb anode and PbO2 cathode; when discharged (b), both electrodes are coated with lead sulfate.
The oxidation half-reaction at the lead (negative) anode is:
Pb ( s ) + HSO 4 − ( a q ) → PbSO 4 ( s ) + H + ( a q ) + 2 e − E red ∘ = − 0.356 V
The reduction half-reaction at the lead(IV) oxide (positive) cathode is:
PbO 2 ( s ) + SO 4 2 − ( a q ) + 4 H + + 2 e − → PbSO 4 ( s ) + 2 H 2 O E red ∘ = 1.685 V
Both half-reactions cause the electrodes to plate with lead sulfate (PbSO4) and dilute the acid electrolyte when discharging. The lead anode is negatively charged and attracts the anionic bisulfate. The lead(IV) oxide cathode is a bit more complicated. This electrode is porous, which allows the electrolyte (sulfuric acid) to solvate the cathode into lead and oxide ions. Then, the hydrogen ions in solution react with the oxide ions to produce water, and the remaining sulfate ions react with the lead to produce the electroplated lead sulfate.
Overall, the net equation for a discharging lead–acid battery is:
Pb ( s ) + PbO 2 ( s ) + 2 H 2 SO 4 ( a q ) → 2 PbSO 4 ( s ) + 2 H 2 O E cell ∘ = 1.685 − ( − 0.356 ) = 2.041 V
When charging, the lead–acid cell is part of an electrolytic circuit. These equations and electrode charge designations are the opposite because an external source reverses the electroplating process and concentrates the acid solution—this external source is very evident when one uses jumper cables to restart a car.
Lead–acid batteries, as compared to other cells, have some of the lowest energy-to-weight ratios (otherwise known as energy density). Energy density is a measure of a battery’s ability to produce power as a function of its weight. Lead–acid batteries, therefore, require a heavier amount of battery material to produce a certain output as compared to other batteries.
Nickel–Cadmium Batteries
Nickel–cadmium batteries are also rechargeable cells. They consist of two half-cells made of solid cadmium (the anode) and nickel(III) oxide-hydroxide (the cathode) connected by a conductive material, typically potassium hydroxide (KOH). Most of us are familiar with AA and AAA cells made of Ni–Cd materials, inside of which the electrodes are layered and wrapped around in a cylinder, as shown in Figure 12.5.

Figure 12.5. A Nickel–Cadmium Battery (1) Metal casing, (2) salt bridge, (3) NiO(OH) cathode, (4) Cd anode
The oxidation half-reaction at the cadmium (negative) anode is:
Cd ( s ) + 2 OH − ( a q ) → Cd ( OH ) 2 ( s ) + 2 e − E red ∘ = − 0.86 V
The reduction half-reaction at the nickel oxide–hydroxide (positive) cathode is:
2 NiO ( OH ) ( s ) + 2 H 2 O + 2 e − → 2 Ni ( OH ) 2 ( s ) + 2 OH − E red ∘ = 0.49 V
Both half-reactions cause the electrodes to plate with their respective products. Overall, the net equation for a Ni–Cd battery is:
2 NiO ( OH ) ( s ) + Cd + 2 H 2 O → 2 Ni ( OH ) 2 ( s ) + Cd ( OH ) 2 ( s ) E cell ∘ = 0.49 − ( − 0.86 ) = 1.35 V
As in our previous example, charging reverses the electrolytic cell potentials. Some Ni–Cd designs are vented for this reason to allow for the release of built-up hydrogen and oxygen gas during electrolysis.
Ni–Cd batteries have a higher energy density than lead–acid batteries. The electrochemistry of the Ni–Cd half-reactions also tends to provide higher surge current. Surge currents are periods of large current (amperage) early in the discharge cycle. This is preferable in appliances such as remote controls that demand rapid responses. It is important to note that modern Ni–Cd batteries have largely been replaced by more efficient nickel–metal hydride (NiMH) batteries. These newer batteries have more energy density, are more cost effective, and are significantly less toxic. As the name suggests, in lieu of a pure metal anode, a metal hydride is used instead.
Electrode Charge Designations
In a galvanic cell, current is spontaneously generated as electrons are released by the oxidized species at the anode and travel through the conductive material to the cathode, where reduction takes place. Because the anode of a galvanic cell is the source of electrons, it is considered the negative electrode; the cathode is considered the positive electrode, as shown in Figure 12.1 previously. Electrons, therefore, move from negative (low electrical potential) to positive (high electrical potential), while the current—the flow of positive charge—is from positive (high electrical potential) to negative (low electrical potential).
MNEMONIC
In a galvanic cell, the anode is negative.
Conversely, the anode of an electrolytic cell is considered positive because it is attached to the positive pole of the external voltage source and attracts anions from the solution. The cathode of an electrolytic cell is considered negative because it is attached to the negative pole of the external voltage source and attracts cations from the solution.
KEY CONCEPT
In a galvanic cell, the anode is negative and the cathode is positive. In an electrolytic cell, the anode is positive and the cathode is negative. This is because an external source is used to reverse the charge of an electrolytic cell. However, in both types of cells, reduction occurs at the cathode, and oxidation occurs at the anode; cations are attracted to the cathode, and anions are attracted to the anode.
In spite of this difference in designating charge (sign), oxidation always takes place at the anode and reduction always takes place at the cathode in both types of cells; electrons always flow through the wire from the anode to the cathode and current flows from cathode to anode. Finally, note that—regardless of its charge designation—the cathode always attracts cations and the anode always attracts anions. In the Daniell cell, for example, the electrons created at the anode by the oxidation of elemental zinc travel through the wire to the copper half-cell. There, they attract copper(II) cations to the cathode, resulting in the reduction of the copper ions to elemental copper, and drawing cations out of the salt bridge into the compartment. The anode, having lost electrons, attracts anions from the salt bridge at the same time that zinc(II) ions formed by the oxidation process dissolve away from the anode.
REAL WORLD
Recognize that in any system in which batteries are placed, it is important to line up cathodes and anodes. Electronics tend to have (+) and (–) designations to line up the electrodes—think jumper cables, television remotes, and button batteries in watches.
This is an important rule to understand not only for electrochemistry in the Chemical and Physical Foundations of Biological Systems section on Test Day, but also for electrophoresis in both this section and Biological and Biochemical Foundations of Living Systems. Isoelectric focusing is a technique used to separate amino acids or polypeptides based on their isoelectric points (pI). The positively charged amino acids (protonated at the solution’s pH) will migrate toward the cathode; negatively charged amino acids (deprotonated at the solution’s pH) will migrate toward the anode. The technique of isoelectric focusing is discussed in detail in Chapter 3 of MCAT Biochemistry Review.
MNEMONIC
Anions are attracted to the anode. Cations are attracted to the cathode. This is true regardless of the type of cell (galvanic, electrolytic, or concentration cells).
MCAT CONCEPT CHECK 12.1:
Before you move on, assess your understanding of the material with these questions.
-
Circle which electrode each of the following statements describes in a galvanic (voltaic) cell:
Anode Cathode
- Site of oxidation:
Anode Cathode
- Electrons flow toward it:
Anode Cathode
- Current flows toward it:
Anode Cathode
- Has (–) designation:
Anode Cathode
- Attracts cations:
-
Circle which electrode each of the following statements describes in an electrolytic cell:
Anode Cathode
- Site of oxidation:
Anode Cathode
- Electrons flow toward it:
Anode Cathode
- Current flows toward it:
Anode Cathode
- Has (–) designation:
Anode Cathode
- Attracts cations:
-
Write the cell diagram for the discharging state of a lead–acid battery:
_______________________________________
-
Which type of cell has a positive ΔG? A positive Ecell?
- Positive ΔG: ________________________________
- Positive Ecell: ____________________________
-
How much current is required to produce 0.23 kg Na from a molten NaCl electrolytic cell that runs for 30 hours? Assume the cell is 100% efficient.
____________________________
-
Fill in the following chart to summarize electrode charge designations of batteries we have analyzed:
Battery State of Use Galvanic or Electrolytic Anode Material Anode Charge Cathode Material Cathode Charge
Ni—Cd Discharging
Ni—Cd Charging
Molten NaCl Discharging
Daniell cell Discharging
Lead–acid Charging
Lead–acid Discharging
12.2 Cell Potentials
LEARNING OBJECTIVES
After Chapter 12.2, you will be able to:
- Describe how standard reduction potentials are measured
- Explain the importance of the sign for electromotive force
- Determine whether a cell using a given reaction is galvanic or electrolytic
- Calculate the net E value for a redox reaction between two species:
Ag + + e − → Ag ( s ) E red ∘ = + 0.80 V Tl + + e − → Tl ( s ) E red ∘ = − 0.34 V
For galvanic cells, the direction of spontaneous movement of charge is from the anode, the site of oxidation, to the cathode, the site of reduction. This is simple enough to remember, but it begs the question: how do we determine which electrode species will be oxidized and which will be reduced? The relative tendencies of different chemical species to be reduced have been determined experimentally, using the tendency of the hydrogen ion (H+) to be reduced as an arbitrary zero reference point.
Reduction Potentials
A reduction potential is measured in volts (V) and defined relative to the standard hydrogen electrode (SHE), which is given a potential of 0 V by convention. The species in a reaction that will be oxidized or reduced can be determined from the reduction potential of each species, defined as the tendency of a species to gain electrons and to be reduced. Each species has its own intrinsic reduction potential; the more positive the potential, the greater the tendency to be reduced.
Standard reduction potential (***E*°red) is measured understandard conditions**: 25 °C (298 K), 1 atm pressure, and 1 M concentrations. The relative reactivities of different half-cells can be compared to predict the direction of electron flow. A more positive E°red means a greater relative tendency for reduction to occur, while a less positive E°red means a greater relative tendency for oxidation to occur.
KEY CONCEPT
A reduction potential is exactly what it sounds like. It tells us how likely a compound is to be reduced. The more positive the value, the more likely it is to be reduced—the more it wants to be reduced.
For galvanic cells, the electrode with the more positive reduction potential is the cathode, and the electrode with the less positive reduction potential is the anode. Because the species with a stronger tendency to gain electrons (that wants to gain electrons more) is actually doing so, the reaction is spontaneous and ΔG is negative. For electrolytic cells, the electrode with the more positive reduction potential is forced by the external voltage source to be oxidized and is, therefore, the anode. The electrode with the less positive reduction potential is forced to be reduced and is, therefore, the cathode. Because the movement of electrons is in the direction against the tendency or desires of the respective electrochemical species, the reaction is nonspontaneous and ΔG is positive.
Example: Given the following half-reactions and E°red values, determine which species would be oxidized and which would be reduced in a galvanic cell.
Ag + + e − → Ag ( s ) E red ∘ = + 0.80 V Tl + + e − → Tl ( s ) E red ∘ = − 0.34 V
Solution: E°red indicates the reduction potential, or the likelihood of a compound to be reduced via a given reaction. A positive E°red value indicates a spontaneous reduction, and a negative value indicates a non–spontaneous reduction. In a galvanic cell, Ag+ will be spontaneously reduced to Ag (s) and Tl (s) will be spontaneously oxidized to Tl+ because Ag+ has the more positive E°red and thus the more favorable reduction reaction. Therefore, the net ionic equation would be:
Ag+ + Tl (s) → Tl+ + Ag (s)
which is the sum of the two spontaneous half-reactions.
It should be noted that reduction and oxidation are opposite processes. Therefore, to obtain the oxidation potential of a given half-reaction, both the reduction half-reaction and the sign of the reduction potential are reversed. For instance, from the example above, the oxidation half-reaction and oxidation potential of Tl (s) are:
T1 ( s ) → T 1 + + e − E ox o = + 0.34 V
Note that, in the examples of batteries given above (lead–acid storage batteries and nickel–cadmium batteries), the oxidation half-reaction was given with the reduction potential of the reverse reaction. These two quantities have equal magnitudes but opposite signs. On the MCAT, reduction potentials are generally given rather than oxidation potentials. Therefore, all references in this book (with exception of the thallium example immediately above) are given using reduction potentials—not oxidation potentials.
The Electromotive Force
Standard reduction potentials are also used to calculate the standard electromotive force (emf or ***E*°cell**) of a reaction, which is the difference in potential (voltage) between two half-cells under standard conditions. The emf of a reaction is determined by calculating the difference in reduction potentials between the two half-cells:
E°cell = E°red,cathode − E°red,anode
Equation 12.3
When subtracting standard potentials, do not multiply them by the number of moles oxidized or reduced. This is because the potential of each electrode does not depend on the size of the electrode (the amount of material), but rather the identity of the material. The standard reduction potential of an electrode will not change unless the chemical identity of that electrode is changed.
KEY CONCEPT
If you need to multiply each half-reaction by a common denominator to cancel out electrons when coming up with the net ionic equation, do not multiply the reduction potential, E°red, by that number. That would indicate a change in the chemical identity of the electrode, which is not occurring.
Example: Given that the standard reduction potentials for Sm3+ and [RhCl6]3– are –2.41 V and +0.44 V, respectively, calculate the electromotive force of the following reaction:
Sm3+ + Rh + 6 Cl– → [RhCl6]3– + Sm
Solution: First, determine the oxidation and reduction half-reactions. As written, the Rh is oxidized, and the Sm3+ is reduced:
Sm 3 + + 3 e − → Sm Rh + 6 Cl − → [ RhCl 6 ] 3 − + 3 e −
Now, we simply take the difference between the samarium(III) reduction potential and the hexachlororhodate(III) reduction potential. We need not change the sign on the hexachlororhodate(III) reduction potential because we are subtracting it from that of samarium(III).
Using the equation provided, the emf can be calculated as: –2.41 V – (+0.44 V) = –2.85 V. The cell is thus electrolytic. If this were instead a galvanic cell the reaction would proceed spontaneously to the left, toward reactants, in which case the Sm would be oxidized while [RhCl6]3– would be reduced with an emf of +2.85 V.
MCAT CONCEPT CHECK 12.2:
Before you move on, assess your understanding of the material with these questions.
-
How are standard reduction potentials measured?
_______________________________________
-
If a cell’s electromotive force (emf) is denoted as a positive value, what does that mean? What if it is negative?
- Positive emf:_____________________________
- Negative emf:____________________________
-
Given the following reactions, determine whether the cell is galvanic or electrolytic:
- 2 Fe3+ (aq) + 2 Cl– (aq) → 2 Fe2+ (aq) + Cl2 (g) (E°cell = –0.59 V):_____________________
- 2 Fe3+ (aq) + 2 I– (aq) → 2 Fe2+ (aq) + I2 (aq) (E°cell = +0.25 V): _______________________
-
Given the two half-reactions below, what would be the spontaneous oxidation–reduction reaction between these two species?
Fe 3 + + 3 e − → Fe E red ∘ = − 0.036 V I 3 − + 2 e − → 3 I − E red ∘ = + 0.534 V
_____________________________________
_____________________________________
12.3 Electromotive Force and Thermodynamics
LEARNING OBJECTIVES
After Chapter 12.3, you will be able to:
- Apply the formula ΔG° = –RT ln Keq to calculations of Gibbs free energy or electromotive force
- Predict Ecell given reaction quotients and equilibrium constants
Throughout our discussion of electrochemistry and the different types of electrochemical cells, we have been making references to the spontaneity or nonspontaneity of the redox reactions housed in each of the different cell types. Let’s now look more formally at this topic by relating free energy to electromotive force (emf) and the concentrations of the oxidation–reduction reactants and products to the voltage of a cell at a given point in time.
Gibbs Free Energy
By now, you should be familiar with the thermodynamic criterion for determining the spontaneity of a reaction: the change in Gibbs free energy, ΔG. This is the change in the amount of energy available in a chemical system to do work. In an electrochemical cell, the work done is dependent on the number of coulombs of charge transferred and the energy available. Thus, ΔG° and emf are related as follows:
ΔG° = –nFE°cell
Equation 12.4
where ΔG° is the standard change in free energy, n is the number of moles of electrons exchanged, F is the Faraday constant, and E°cell is the standard emf of the cell. Keep in mind that, if the Faraday constant is expressed in coulombs ( J V ) , then ΔG° must be expressed in J, not kJ. Notice the similarity of this relationship to that expressed in the physics formula W= qΔV for the amount of work available or needed in the transport of a charge q across a potential difference ΔV: n× F is a charge, and E°cell is a voltage. This application in electrostatics is discussed in Chapter 5 of MCAT Physics and Math Review.
BRIDGE
Recall from Chapter 6 of MCAT General Chemistry Review that, if ΔG is positive, the reaction is nonspontaneous; if ΔG is negative, the reaction is spontaneous.
Note the significance of the negative sign on the right side of the equation. ΔG° and E°cell will always have opposite signs. Therefore, galvanic cells have negative ΔG° and positive E°cell values; electrolytic cells have positive ΔG° and negative E°cell values.
Example: Determine the standard change in free energy of a cell with the following net reaction. (Note: The standard reduction potential of iron(III) is +0.77 V; the standard reduction potential of molecular chlorine is +1.36 V.)
2 Fe3+ (aq) + 2 Cl– (aq) → 2 Fe2+ (aq) + Cl2 (g)
Solution: First, separate the reaction into the half-reactions:
2 Fe 3 + + 2 e − → 2 Fe 2 + 2 Cl − → Cl 2 + 2 e −
In this reaction, iron(III) is reduced and is the cathode, whereas Cl– is oxidized and is the anode. The reduction potential of chlorine is actually higher than that of iron(III); this means that the electrodes are serving the opposite role from their natural tendency, and the reaction is nonspontaneous. This is an electrolytic cell, and should have a negative emf value.
Now, determine the emf:
E°cell = E°red,cathode − E°red,anode = 0.77 V − 1.36 V = −0.59 V
Use the emf to determine the free energy change (note that as 2 electrons are transferred, n = 2):
Δ G ° = − n F E cell ∘ = − ( 2 mol e − ) ( 96 , 485 C mol e − ) ( − 0.59 V ) ≈ 2 × 10 5 × ( 0.6 ) = 1.2 × 10 5 J ( actual = 1.14 × 10 5 J )
The free energy change is about +120 kJ, which represents a non-spontaneous reaction.
Reaction Quotients
So far, we have considered the calculation of a cell’s emf only under standard conditions. However, electrochemical cells may have ionic concentrations that deviate from 1 M. Also, for the concentration cell, the concentrations of the ions in the two compartments must be different for there to be a measurable voltage and current. Concentration and the emf of a cell are related: emf varies with the changing concentrations of the species in the cell. When conditions deviate from standard conditions, one can use the Nernst equation:
E cell = E cell ∘ − R T n F ln Q
Equation 12.5
where Ecell is the emf of the cell under nonstandard conditions, E°cell is the emf of the cell under standard conditions, R is the ideal gas constant, T is the temperature in kelvin, n is the number of moles of electrons, F is the Faraday constant, and Q is the reaction quotient for the reaction at a given point in time. The following simplified version of the equation can be used, assuming T = 298 K:
E cell = E cell ∘ − 0.0592 n log Q
Equation 12.6
This simplified version of the equation brings together R, T (298 K), and F, and converts the natural logarithm to the base-ten logarithm to make calculations easier.
MCAT EXPERTISE
If the Nernst equation is needed on Test Day, stick with the log10 version because natural logarithm calculations get very tedious.
Remember that the reaction quotient, Q, for a general reaction aA + bB → cC + dD has the form:
Q = [ C] c [ D] d [ A] a [ B] b
Equation 12.7
Although the expression for the reaction quotient Q has two terms for the concentrations of reactants and two terms for the concentrations of products, remember that only the species in solution are included. When considering the case of the Daniell cell, for example, only the concentrations of zinc and copper ions are considered:
Zn ( s ) + Cu 2 + ( a q ) → Zn 2 + ( a q ) + Cu ( s ) Q = [ Zn 2 + ] [ Cu 2 + ]
The emf of a cell can be measured with a voltmeter. A potentiometer is a kind of voltmeter that draws no current and gives a more accurate reading of the difference in potential between two electrodes.
Example: Find the emf of a galvanic cell at 25 °C based on the following standard reduction potentials: Fe 2 + + 2 e − → Fe E red ∘ = − 0.44 V Cl 2 + 2 e − → 2 Cl − E red ∘ = + 1.36 V
In this cell, [Fe2+] = 0.01 M and [Cl–] = 0.1 M.
Solution: First, determine the standard cell potential. Because the chlorine half-reaction has a higher reduction potential, it will be the cathode. Iron will act as the anode. The standard cell potential is:
E°cell = E°red,cathode −E°red,anode = 1.36 V − (−0.44 V) = +1.80 V
Now, determine the net ionic equation. Remember that iron is being oxidized, so its reduction half-reaction in the question stem will have to be reversed. The net ionic equation is:
Fe + Cl2 → Fe2+ + 2 Cl–
From this equation, we can determine the value of the reaction quotient:
Q = [Fe2+][Cl–]2 = (0.01 M)(0.1 M)2 = 10–4
Now, plug into the Nernst equation, keeping in mind that two electrons are transferred (n = 2):
E cell = E cell ∘ − 0.0592 n log Q = 1.8 − 0.0592 2 log 10 − 4 = 1.8 + 4 × 0.0592 2 ≈ 1 . 8 + 2 × 0 . 06 = 1.92 V
In this case, the cell actually has a higher voltage than it normally would due to the concentrations of ions present.
BRIDGE
While a mathematically rigorous equation, the Nernst equation has a powerful use in biochemistry for calculating resting and depolarized membrane potentials based on concentrations of ions. Its more extended version, the Goldman–Hodgkin–Katz equation, is discussed in Chapter 8 of MCAT Biochemistry Review.Equation 8.2 in that chapter looks slightly different than Equation 12.6 here. This is because the temperature is different (310 K rather than 298 K) and the units are different (mV instead of V).
Equilibria
As discussed in Chapter 7 of MCAT General Chemistry Review, ΔG° can also be determined in another manner:
ΔG° = –RT ln Keq
Equation 12.8
where R is the ideal gas constant, T is the absolute temperature, and Keq is the equilibrium constant for the reaction.
Combining the two expressions that solve for standard free energy change, we see that
ΔG° = –nFE°cell = –RT ln Keq
or
nFE°cell = RT ln Keq
By extension, if the values for n, T, and Keq are known, then E°cell for the reaction is easily calculated. On the MCAT, you will not be expected to calculate natural logarithm values in your head. That being said, these equations can still be tested but in a conceptual way.
MCAT EXPERTISE
Whether it is log or ln, remember that a logarithm will be positive when equilibrium constants are greater than 1, negative when equilibrium constants are less than 1, and 0 when equilibrium constants are equal to 1.
Analysis of the equations shows us that, for redox reactions with equilibrium constants less than 1 (equilibrium state favors the reactants), the E°cell will be negative because the natural logarithm of any number between 0 and 1 is negative. These properties are characteristic of electrolytic cells, which house nonspontaneous oxidation–reduction reactions. Instead, if the equilibrium constant for the reaction is greater than 1 (equilibrium state favors the products), the E°cell will be positive because the natural logarithm of any number greater than 1 is positive. These properties are characteristic of galvanic cells, which house spontaneous oxidation–reduction reactions. If the equilibrium constant is equal to 1 (concentrations of the reactants and products are equal at equilibrium), the E°cell will be equal to zero. An easy way to remember this is that E°cell = 0 V for any concentration cell with equimolar concentrations in both half-cells because there is no net ionic equation (both half-cells contain the same ions).
KEY CONCEPT
If E°cell is positive, ln Keq is positive. This means that Keq must be greater than one and that the equilibrium lies to the right (products are favored).
Knowing the effects of concentration on equilibria, we can now derive the change in Gibbs free energy of an electrochemical cell with varying concentrations using the equation:
ΔG = ΔG° + RTln Q
Equation 12.9
where ΔG is the free energy change under nonstandard conditions, ΔG° is the free energy change under standard conditions (which can be determined from Equation 12.4 or Equation 12.8 above), R is the ideal gas constant, T is the temperature, and Q is the reaction quotient.
MCAT CONCEPT CHECK 12.3:
Before you move on, assess your understanding of the material with these questions.
-
Fill in the table to show the relationships between the equilibrium constant, Gibbs free energy, and electromotive force (emf), assuming standard conditions:
***Keq ΔG°: (+) or (−)? Reaction: Spontaneous or Nonspontaneous?E*°cell: (+) or (−)? 1.2 × 10–2 2 × 102 1**
-
Given the following reaction quotients and equilibrium constants, determine the direction of the reaction and the sign of Ecell:
***Q Keq Reaction Direction (Forward, Backward, or Equilibrium) Sign ofE*cell**
10–3 10–2
102 1.1
1 1
Conclusion
In this chapter, we covered the essential MCAT topic of electrochemistry. We reviewed the behavior of many different types of electrochemical cells. Galvanic cells rely on spontaneous oxidation–reduction reactions to produce current and supply energy. The concentration cell is a special type of galvanic cell for which the current is dependent on an ion concentration gradient rather than a difference in reduction potential between two chemically distinct electrodes. Electrolytic cells rely on external voltage sources to drive a nonspontaneous oxidation–reduction reaction called electrolysis. Finally, we considered the thermodynamics of the different cell types. Galvanic and concentration cells have positive electromotive forces (emf) and negative free energy changes, whereas electrolytic cells have negative electromotive forces and positive free energy changes.
In retrospect, the content you have learned in MCAT General Chemistry Review has numerous organic (biological) and inorganic applications. And as you prepare to be a physician, you must begin to understand and treat the individual as a sum of many intertwining systems and parts. Many body systems and parts rely on electrochemical cells: the heart is a self-paced electrochemical cell, the neurons of the brain and spinal cord are rechargeable concentration cells, and every cell that contains mitochondria (all cells except erythrocytes) rely on the proton-motive force across the inner mitochondrial membrane to function. Our discussion here of inorganic systems has value through analogy to many biological systems.
Without further delay, we want to offer you our heartiest congratulations for completing this final chapter of MCAT General Chemistry Review. The hard work, time, and energy you have invested in a careful and thorough review of the topics covered within the pages of this book will pay off on Test Day. We hope that we have been successful in meeting our goals in writing this Kaplan MCAT Review series: to assess the general concepts and principles essential to correctly and efficiently answer the general chemistry questions on the MCAT; to guide you in the development of critical thinking skills necessary for analyzing passages, question stems, and answer choices; and to provide holistic preparation for your Test Day experience. In addition to all of these, we aimed to relate the science to everyday life experiences and future experiences as a physician, demystify the concepts, and have some fun in the process. We are grateful for the opportunity to have been a part of your journey to success on the MCAT, and—beyond that—success in your medical education and future practice as the great physician you deserve to be!
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Electrochemical Cells
- An electrochemical cell describes any cell in which oxidation–reduction reactions take place. Certain characteristics are shared between all types of electrochemical cells.
- Electrodes are strips of metal or other conductive materials placed in an electrolyte solution.
- The anode is always the site of oxidation. It attracts anions.
- The cathode is always the site of reduction. It attracts cations.
- Electrons flow from the anode to the cathode.
- Current flows from the cathode to the anode.
- Cell diagrams are shorthand notation that represent the reactions taking place in an electrochemical cell.
- Cell diagrams are written from anode to cathode with electrolytes (the solution) in between.
- A vertical line represents a phase boundary, and a double vertical line represents a salt bridge or other physical boundary.
- Galvanic (voltaic) cells house spontaneous reactions (ΔG < 0) with a positive electromotive force.
- Electrolytic cells house nonspontaneous reactions (ΔG > 0) with a negative electromotive force. These nonspontaneous cells can be used to create useful products through electrolysis.
- Concentration cells are a specialized form of a galvanic cell in which both electrodes are made of the same material. Rather than a potential difference causing the movement of charge, it is the concentration gradient between the two solutions.
- The charge on an electrode is dependent on the type of electrochemical cell one is studying.
- For galvanic cells, the anode is negatively charged and the cathode is positively charged.
- For electrolytic cells, the anode is positively charged and the cathode is negatively charged.
- Rechargeable batteries are electrochemical cells that can experience charging (electrolytic) and discharging (galvanic) states. Rechargeable batteries are often ranked by energy density—the amount of energy a cell can produce relative to the mass of battery material.
- Lead–acid batteries, when discharging, consist of a Pb anode and a PbO2 cathode in a concentrated sulfuric acid solution. When charging, the PbSO4-plated electrodes are dissociated to restore the original Pb and PbO2 electrodes and concentrate the electrolyte. These cells have a low energy density.
- Nickel–cadmium batteries (Ni–Cd), when discharging, consist of a Cd anode and a NiO(OH) cathode in a concentrated KOH solution. When charging, the Ni(OH)2 and Cd(OH)2 plated electrodes are dissociated to restore the original Cd and NiO(OH) electrodes and concentrate the electrolyte. These cells have a higher energy density than lead–acid batteries.
- Nickel–metal hydride (NiMH) batteries have more or less replaced Ni–Cd batteries because they have higher energy density, are more cost effective, and are significantly less toxic.
- Surge current is an above-average current transiently released at the beginning of the discharge phase; it wanes rapidly until a stable current is achieved.
Cell Potentials
- A reduction potential quantifies the tendency for a species to gain electrons and be reduced. The higher the reduction potential, the more a given species wants to be reduced.
- Standard reduction potentials (E°red) are calculated by comparison to the standard hydrogen electrode (SHE) under the standard conditions of 298 K, 1 atm pressure, and 1 M concentrations.
- The standard hydrogen electrode has a standard reduction potential of 0 V.
- Standard electromotive force (E°cell) is the difference in standard reduction potential between the two half-cells.
- For galvanic cells, the difference of the reduction potentials of the two half-reactions is positive; for electrolytic cells, the difference of the reduction potentials of the two half-reactions is negative.
Electromotive Force and Thermodynamics
- Electromotive force and change in free energy always have opposite signs.
- When E°cell is positive, ΔG° is negative. This is the case in galvanic cells.
- When E°cell is negative, ΔG° is positive. This is the case in electrolytic cells.
- When E°cell is 0, ΔG° is 0. This is the case in concentration cells.
- The Nernst equation describes the relationship between the concentration of species in a solution under nonstandard conditions and the electromotive force.
- There exists a relationship between the equilibrium constant (Keq) and E°cell.
- When Keq (the ratio of products’ concentrations at equilibrium over reactants’, raised to their stoichiometric coefficients) is greater than 1, E°cell is positive.
- When Keq is less than 1, E°cell is negative.
- When Keq is equal to 1, E°cell is 0.
ANSWERS TO CONCEPT CHECKS
**12.1**
- In a galvanic cell, the anode is the site of oxidation, has current flowing toward it, and has a (–) designation. The cathode has electrons flowing toward it and attracts cations.
- In an electrolytic cell, the anode is the site of oxidation and has current flowing toward it. The cathode has electrons flowing toward it, has a (–) designation, and attracts cations.
- Pb (s) | H2SO4 (4 M) || H2SO4 (4 M) | PbO2 (s)
- Electrolytic cells are nonspontaneous and have a positive ΔG. Galvanic cells are spontaneous and have a negative ΔG; therefore, they have a positive Ecell.
- mol M = I t n F → I = ( mol M) n F t = ( 230 g 23 g mol ) ( 1 mol e − ) ( 96 , 485 C mol e – ) 30 hr ( 3600 s 1 hr ) ≈ 10 × 10 5 10 5 = 10 A (actual = 8 .93 A)
-
Battery State of Use Galvanic or Electrolytic Anode Material Anode Charge Cathode Material Cathode Charge
Ni—Cd Discharging Galvanic Cd Negative NiO(OH) Positive
Ni—Cd Charging Electrolytic Cd(OH)2 Positive Ni(OH)2 Negative
Molten NaCl Discharging Electrolytic Any Positive Any Negative
Daniell cell Discharging Galvanic Zn Negative Cu Positive
Lead–acid Charging Electrolytic PbSO4 Positive PbSO4 Negative
Lead–acid Discharging Galvanic Pb Negative PbO2 Positive
**12.2**
- A sample is measured by setting up a cell relative to a standard hydrogen electrode, which is given a reduction potential of 0 V by convention.
- A positive emf means the cell is spontaneous (galvanic); a negative emf means the cell is nonspontaneous (electrolytic).
- The first cell is electrolytic because it has a negative emf. The second cell is galvanic because it has a positive emf.
- The reduction potential of triiodide is higher than iron(III), so triiodide will be reduced and iron will be oxidized: 2 Fe + 3 I3− → 2 Fe3+ + 9 I− (E°cell = +0.57 V).
**12.3**
-
***Keq ΔG°: (+) or (−)? Reaction: Spontaneous or Nonspontaneous?E*°cell: (+) or (−)? 1.2 × 10–2** + Nonspontaneous –
2 × 102 – Spontaneous +
1 0 Not applicable—applies to any cell at equilibrium 0
Remember that ΔG° = –RT ln Keq; if Keq < 1, ln Keq < 0, and ΔG° > 0. If Keq > 1, ln Keq > 0, and ΔG° < 0. If Keq = 1, ln Keq = 0, and ΔG° = 0.
-
***Q Keq Reaction Direction (Forward, Backward, or Equilibrium) Sign ofE*cell**
10−3 10−2 Forward +
102 1.1 Backward –
1 1 Equilibrium 0
Note that these calculations do not assume standard conditions, unlike question 1.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. C
In the oxidation–reduction reaction of a metal with oxygen, the metal will be oxidized (donate electrons) and oxygen will be reduced (accept electrons). This fact allows us to immediately eliminate (B) and (D). A species with a higher reduction potential is more likely to be reduced, and a species with a lower reduction potential is more likely to be oxidized. Based on the information in the question, iron is oxidized more readily than those metals; this means that iron has a lower reduction potential.
2. B
To determine the standard electromotive force of a cell, simply subtract the standard reduction potentials of the two electrodes. In this case, the cathode is zinc because it is being reduced; the anode is silver because it is being oxidized. Thus,
E°cell = E°red,cathode − E°red,anode = − 0.763 − 0.337 = − 1.10 V
While we must multiply the silver half-reaction by two to balance electrons, the actual value for the reduction potential does not change. Remember that the standard reduction potential is determined by the identity of the electrode, not the amount of it present.
3. A
Oxidation occurs at the anode, and reduction occurs at the cathode. Because Cu is the anode, it must be oxidized. The reduction potential of the cathode cannot be less than that of the anode for a galvanic cell. Therefore, mercury, (A), must be the cathode. In a concentration cell, the same material is used as both the cathode and anode; however, this question assumes equal concentrations. If both electrolyte solutions have the same concentration, there will be no oxidation–reduction reaction and, therefore, no anode or cathode. This eliminates (B).
4. C
In an electrolytic cell, ionic compounds are broken up into their constituents; the cations (positively charged ions) migrate toward the cathode, and the anions (negatively charged ions) migrate toward the anode. In this case, the cations are H+ ions (protons), so option I is correct. Electrons flow from anode to cathode in all types of cells, meaning that option III is also correct. Option II is incorrect for two reasons. First, it is unlikely that the anions in any cell would be O2– rather than OH–. Second, and more significantly, these anions would flow to the anode, not the cathode.
5. D
This answer comes directly from the equation relating Gibbs free energy and E°cell. ΔG° = −nFE°cell, where n is the number of moles of electrons transferred and F is the Faraday constant, 96 , 485 C mol e − . To determine n, one must look at the balanced half-reactions occurring in the oxidation–reduction reaction.
6. B
Salt bridges contain inert electrolytes. Ionic compounds, such as (A), (C), and (D), are known to be strong electrolytes because they completely dissociate in solution. (B) cannot be considered an electrolyte because its atoms are covalently bonded and will not dissociate in aqueous solution. (B) and (C) may appear similar, but there is an important distinction to be made. (C) implies that Mg2+ and SO32− are the final, dissociated ionic constituents, while (B) implies that neutral SO3 would have to be dissolved in solution.
7. B
Potential, as measured by E°cell, is dependent only on the identity of the electrodes and not the amount present. Similarly, the equilibrium constant depends only on the identity of the electrolyte solutions and the temperature. However, as the electrode material is increased, the surface area participating in oxidation–reduction reactions is increased and more electrons are released, making statement II correct.
8. D
E°cell is dependent upon the change in free energy of the system through the equation RT ln Keq = nFE°cell. The temperature, T, appears in this equation; thus, a change in temperature will impact the E°cell.
9. B
If this were a galvanic cell, the species with the more positive reduction potential (cadmium) would be reduced. The cathode is always reduced in an electrochemical cell, so sodium could not be the cathode in such a galvanic cell, eliminating (A). Sodium would be the cathode in an electrolytic cell, however, which would make cadmium the anode. Thus, the answer is (B). Note that we do not have to determine E°cell because we already know the answer. However, the E°cell would be −2.71 − (−0.40) = −2.31 V for an electrolytic cell, and +2.31 V for a galvanic cell, eliminating (C) and (D).
10. D
There are only two equations involving standard change in free energy in electrochemical cells: ΔG° = −nFE°cell and ΔG° = –RT ln Keq. Substituting E°cell = E°red,cathode − E°red,anode into the first equation and distributing the negative sign gives (D). (A) would be the opposite of ΔG°. Setting the two equations equal to each other, we get RT ln Keq = nFE°cell. Solving for E°cell, we get E cell ∘ = R T n F ln K eq , which is the opposite of (B). (C) incorrectly solves the algebra.
11. D
A spontaneous electrochemical reaction has a negative ΔG. Using the equation ΔG° = –RT ln Keq, Keq > 1 would result in ln Keq > 0, which means ΔG° < 0. A negative electromotive force, (A), or equilibrium state, (B), would not correspond to a spontaneous reaction. Concentration cells can be spontaneous; however, if the concentration cell had reached equilibrium, it would cease to be a spontaneous reaction, eliminating (C). When an answer choice may be true, but does not have to be—it is the wrong answer on Test Day.
12. A
A change in pH has a direct correlation to the hydrogen ion (H+) concentration. Decreasing the pH increases the H+ concentration, which means the concentration of products has increased in the oxidation of sulfur dioxide. This means it would be harder to liberate electrons, thus decreasing the emf. One could also view this decrease in oxidation potential as an increase in reduction potential. If E°red,anode increases, then E°cell must decrease according to E°cell = E°red,cathode − E°red,anode.
13. C
An electrolytic cell is nonspontaneous. Therefore, the ΔG° must be positive and E°cell must be negative, eliminating (B) and (D). The change in entropy may be positive or negative, depending on the species involved, eliminating (A). According to the equation ΔG° = –RT ln Keq, Keq < 1 would result in ln Keq < 0, which means ΔG° > 0.
14. D
Compared to other cell types, lead–acid batteries have a characteristically low energy density, (D). While (A) is a true statement, the incomplete dissociation of sulfuric acid does not fully explain the low energy density of lead–acid batteries. (C) is likely to be an opposite; the more easily the electrodes dissociate, the easier it is to carry out oxidation–reduction reactions with them.
15. C
During the recharge cycle, Ni–Cd cells will accept current from an outside source until the Cd and NiO(OH) electrodes are pure; at this point, the reaction will stop because Cd(OH)2 runs out and no more electrons can be accepted. (A) and (B) are both true statements, but they fail to explain why overcharging the battery (continuing to try to run current into the battery even when the electrodes are reverted to their original state) is not a problem with Ni–Cd batteries. Finally, surge current refers to the initial burst of current seen in some batteries; once charged, the surge current will not increase even if the power source continues to be run because no additional charge will be stored on the electrodes, eliminating (D).
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(12.1) Moles of electrons transferred during reduction:Mn+ + n e– → M (s)
(12.2) Electrodeposition equation: mol M = I t n F
(12.3) Standard electromotive force of a cell:E°cell = E°red,cathode − E°red,anode
(12.4) Standard change in free energy from standard emf:ΔG° = −nFE°cell
(12.5) Nernst equation (full): E cell = E cell ∘ − R T n F ln Q
(12.6) Nernst equation (simplified): E cell = E cell ∘ − 0.0592 n log Q
(12.7) Reaction quotient: Q = [ C] c [ D] d [ A] a [ B] b
(12.8) Standard change in free energy from equilibrium constant: ΔG° = –RT ln Keq
(12.9) Free energy change (nonstandard conditions):ΔG = ΔG° + RTln Q
SHARED CONCEPTS
Biochemistry Chapter 3
Nonenzymatic Protein Function and Protein Analysis
Biochemistry Chapter 8
Biological Membranes
General Chemistry Chapter 7
Thermochemistry
General Chemistry Chapter 11
Oxidation–Reduction Reactions
Physics and Math Chapter 5
Electrostatics and Magnetism
Physics and Math Chapter 6
Circuits