Chapter 11: Oxidation–reduction Reactions

Chapter 11: Oxidation–reduction Reactions
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- Consider the following equation:
6 Na (s) + 2 NH3 (aq) → 2 Na3N (s) + 3 H2 (g)
Which species acts as an oxidizing agent?
- Na
- N in NH3
- H in NH3
- H2
-
How many electrons are involved in the following half-reaction after it is balanced?
Cr2O72− + H+ + e− → Cr2+ + H2O
- 2
- 8
- 12
- 16
-
Lithium aluminum hydride (LiAlH4) is often used in laboratories because of its tendency to donate a hydride ion. Which of the following roles would lithium aluminum hydride likely play in a reaction?
- Strong reducing agent only
- Strong oxidizing agent only
- Both a strong reducing agent and strong oxidizing agent
- Neither a strong reducing agent nor a strong oxidizing agent
-
What is the oxidation number of chlorine in NaClO?
- –1
- 0
- +1
- +2
-
The following electronic configurations represent elements in their neutral form. Which element is the strongest oxidizing agent?
- 1s22s22p63s23p64s2
- 1s22s22p63s23p64s23d5
- 1s22s22p63s23p64s23d104p1
- 1s22s22p63s23p64s23d104p5
-
Which of the following is the correct net ionic reaction for the reaction of copper with silver(I) nitrate?
- Cu + AgNO3 → Cu(NO3)2 + Ag
- Cu + 2 Ag+ + 2 NO3− → Cu2+ + 2 NO3− + 2 Ag
- 2 Ag+ + 2 NO3− → 2 NO3− + 2 Ag+
- Cu + 2 Ag+ → Cu2+ + 2 Ag
-
One way to test for the presence of iron in solution is by adding potassium thiocyanate to the solution. The product when this reagent reacts with iron is FeSCN2+, which creates a dark red color in solution via the following net ionic equation:
Fe3+ + SCN– → FeSCN2+
How many grams of iron sulfate would be needed to produce 2 moles of FeSCN2+?
- 110 g
- 220 g
- 400 g
- 500 g
-
During the assigning of oxidation numbers, which of the following elements would most likely be determined last?
- Ar
- F
- Sr
- Ir
-
As methanol is converted to methanal, and then methanoic acid, the oxidation number of the carbon:
- increases.
- decreases.
- increases, then decreases.
- decreases, then increases.
-
In the compound KH2PO4, which element has the highest oxidation number?
- K
- H
- P
- O
-
If a certain metal has multiple oxidation states, its acidity as an oxide generally increases as the oxidation state increases. Therefore, which of the following tungsten compounds is likely to be the strongest acid?
- WO2
- WO3
- W2O3
- W2O5
-
Consider the following steps in the reaction between oxalic acid and chlorine:
- Cl2 + H2O → HOCl + Cl– + H+
- H2C2O4 → H+ + HC2O4−
- HOCl + HC2O4− → H2O + Cl− + 2 CO2
Which of these steps, occurring in aqueous solution, is an example of a disproportionation reaction?
- I only
- III only
- I and III only
- I, II, and III
-
Potentiometry in an oxidation–reduction titration is analogous to performing an acid–base titration with a(n):
- acidic indicator.
- basic indicator.
- pH meter.
- oxidizing agent.
-
After balancing the following oxidation–reduction reaction, what is the sum of the stoichiometric coefficients of all of the reactants and products?
S8 (s) + NO3− (aq) → SO32− (aq) + NO (g)
- 4
- 50
- 91
- 115
-
An assay is performed to determine the gold content in a supply of crushed ore. One method for pulling gold out of ore is to react it in a concentrated cyanide (CN–) solution. The equation is provided below:
Au + NaCN + O2 + H2O → Na[Au(CN)2] + NaOH
An indicator is used during this reaction, and approximately 100 mL of a 2 M NaCN solution is used to reach the endpoint. How many moles of Au are present in the crushed ore?
- 0.01 mol
- 0.02 mol
- 0.10 mol
- 0.20 mol
Answer Key
- C
- B
- A
- C
- D
- D
- C
- D
- A
- C
- B
- A
- C
- D
- C
Chapter 11: Oxidation–reduction Reactions
CHAPTER 11
OXIDATION–REDUCTION REACTIONS
In This Chapter
11.1 Oxidation–Reduction Reactions
Oxidation and Reduction
Assigning Oxidation Numbers
Balancing Oxidation–Reduction Reactions
11.2 Net Ionic Equations
Overview
Disproportionation Reactions
Oxidation–Reduction Titrations
Concept Summary
CHAPTER PROFILE
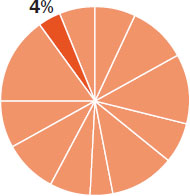
The content in this chapter should be relevant to about 4% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
4E:Atoms, nuclear decay, electronic structure, and atomic chemical behavior
5A:Unique nature of water and its solutions
Introduction
You’re on a night call in the emergency department (ED) when a 5-month-old infant patient’s chart appears on your screen. You click through the triage notes and read what the caregiver reports: poor sucking ability and loss of head control and motor skills. You’re puzzled by the findings and the previous ED admissions of lactic acidosis. You suspect diabetic ketoacidosis (DKA), liver, or kidney diseases—and possibly even poisoning—but nothing seems to fit. Minutes later, the child is brought into the examination room and does not stop crying. Over the course of an hour, another episode of lactic acidosis develops. The child is eventually admitted to the neonatal intensive care unit for long-term care.
Later, you ask the neonatologist about the patient. They point you to the charts and a genetic test performed shortly after birth. The child was diagnosed with Leigh disease, an extremely rare mitochondrial disorder. In Leigh disease, a number of key mitochondrial enzymes are disrupted and the process of oxidative phosphorylation is never achieved. Specifically, some of the most important enzymes that catalyze oxidation–reduction reactions, such as the pyruvate dehydrogenase complex and succinate dehydrogenase complex, are affected. When pyruvate cannot be oxidized to acetyl-CoA, it is instead fermented to lactic acid.
In biological systems, oxidation is coincident with the loss of electrons, sometimes in the form of hydrogen (dehydrogenation). The enzymes that catalyze these oxidations are called dehydrogenases. Many other macromolecules besides enzymes, such as vitamins, also carry out their functions by oxidizing or reducing other compounds. Iron in hemoglobin likewise undergoes rounds of oxidation and reduction as it carries oxygen from the lungs to tissues.
In this chapter, we focus our attention on the movement of electrons in chemical reactions. Such reactions are called oxidation–reduction (redox) reactions because they always occur in pairs. Oxidation–reduction reactions are particularly important because they tie into a number of topics in organic chemistry and biochemistry. In fact, Chapters 5 through 10 of MCAT Organic Chemistry Review and Chapters 9 through 11 of MCAT Biochemistry Review all touch on oxidation–reduction reactions in different sets of molecules.
11.1 Oxidation–Reduction Reactions
LEARNING OBJECTIVES
After Chapter 11.1, you will be able to:
- Separate a redox reaction into oxidation and reduction half-reactions
- Balance a redox reaction
- Identify the oxidizing agent, reducing agent, and relevant oxidation states for a given reaction: SnCl2 + PbCl4 → SnCl4 + PbCl2
Reactions that involve the transfer of electrons from one chemical species to another can be classified as oxidation–reduction (redox) reactions.
Oxidation and Reduction
The law of conservation of charge states that electrical charge can be neither created nor destroyed. Thus, an isolated loss or gain of electrons cannot occur; oxidation (loss of electrons) and reduction (gain of electrons) must occur simultaneously, resulting in an electron transfer called a redox reaction. An oxidizing agent causes another atom in a redox reaction to undergo oxidation and is itself reduced. A reducing agent causes the other atom to be reduced and is itself oxidized. There are various mnemonics to remember these terms, as highlighted in the sidebar.
MNEMONIC
Redox reactions: choose one of the mnemonics and stick with it!
- OIL RIG: Oxidation Is Loss of electrons, Reduction Is Gain of electrons.
- LEO the lion says GER: Loss of Electrons is Oxidation, Gain of Electrons is Reduction.
- LEORA says GEROA: Loss of Electrons is Oxidation (Reducing Agent), Gain of Electrons is Reduction (Oxidizing Agent).
Being familiar with some common oxidizing and reducing agents can save significant time on Test Day, especially in organic chemistry reactions. Some of the commonly used agents on the MCAT are listed in Table 11.1. Note that almost all oxidizing agents contain oxygen or another strongly electronegative element (such as a halogen). Reducing agents often contain metal ions or hydrides (H–).
Table 11.1. Common Oxidizing and Reducing Agents
OXIDIZING AGENTS REDUCING AGENTS
O2 CO
H2O2 C
F2, Cl2, Br2, I2 (halogens) B2H6
H2SO4 Sn2+ and other pure metals
HNO3 Hydrazine*
NaClO Zn(Hg)*
KMnO4Lindlar’s catalyst
CrO3, Na2Cr2O7NaBH4
Pyridinium chlorochromate (PCC)LiAlH4
NAD+, FADHNADH, FADH2
* These oxidizing agents and reducing agents are commonly seen in organic chemistry reactions. ** These and other biochemical redox reagents often act as energy carriers in biochemistry reactions.
Note that biochemical redox reagents such as NAD+ tend to act as both oxidizing and reducing agents at different times during metabolic pathways. As such, they act as mediators of energy transfer during many metabolic processes, as shown in Figure 11.1.
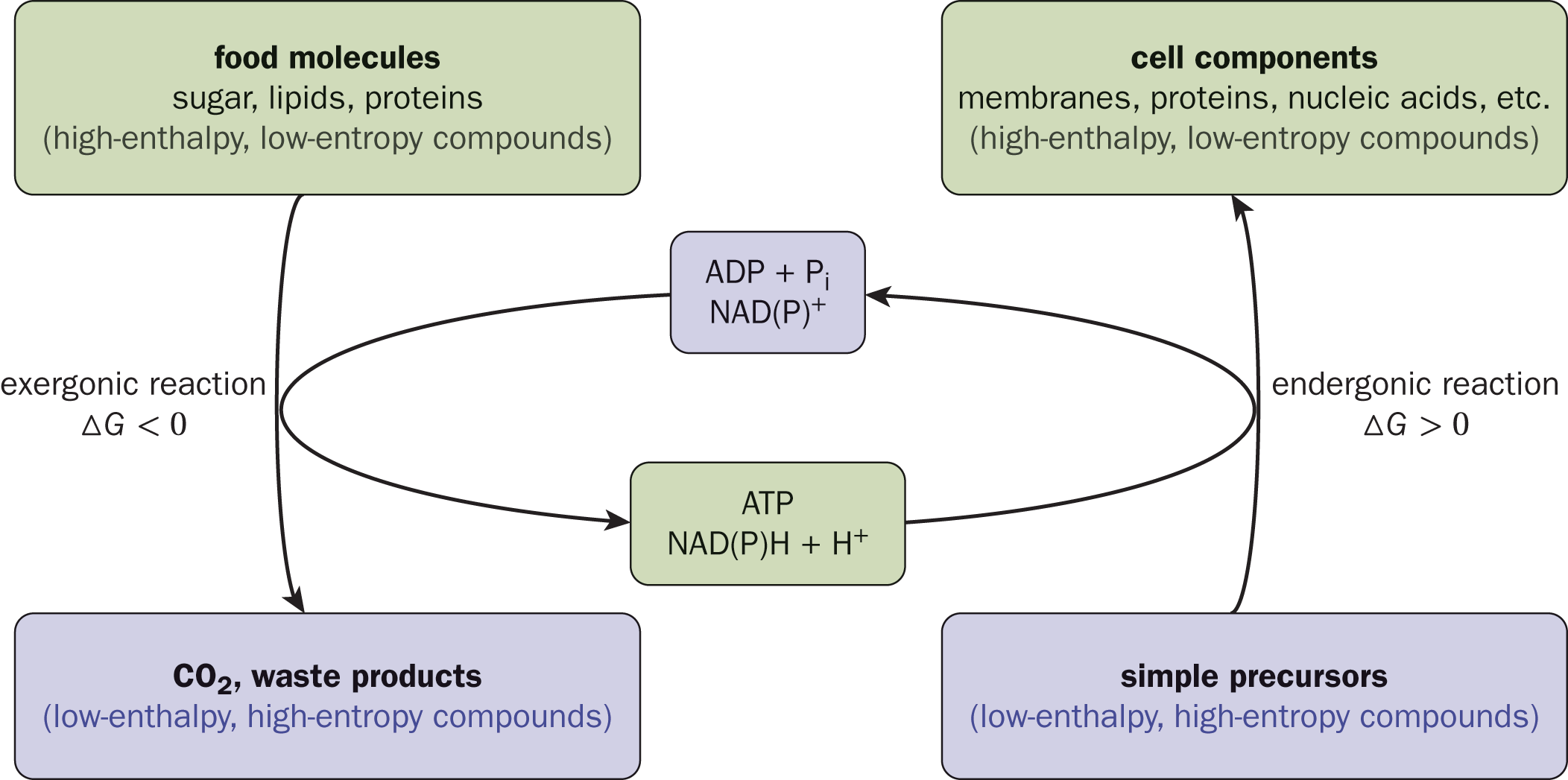
Figure 11.1. Oxidation and Reduction of Biochemical Compounds Serves as a Method of Energy Transfer
On a technical level, the term oxidizing agent or reducing agent is applied specifically to the atom that gains or loses electrons, respectively. However, many science texts will describe the compound as a whole (CrO3, rather than Cr6+) as the oxidizing or reducing agent.
Assigning Oxidation Numbers
It is important, of course, to know which atom is oxidized and which is reduced. Oxidation numbers are assigned to atoms in order to keep track of the redistribution of electrons during chemical reactions. Based on the oxidation numbers of the reactants and products, it is possible to determine how many electrons are gained or lost by each atom.
BRIDGE
In Chapter 3 of MCAT General Chemistry Review, we illustrated that metals form cations and nonmetals form anions. To form a cation, a metal must lose electrons. Therefore, metals like to get oxidized (lose electrons) and act as good reducing agents. Nonmetals, on the other hand, like to get reduced (gain electrons) and act as good oxidizing agents.
The oxidation number of an atom in a compound is assigned according to the following rules:
- The oxidation number of a free element is zero.For example, the atoms in N2, P4, S8, and He all have oxidation numbers of zero.
- The oxidation number for a monatomic ion is equal to the charge of the ion. For example, the oxidation numbers for Na+, Cu2+, Fe3+, Cl–, and N3– are +1, +2, +3, –1, and –3, respectively.
- The oxidation number of each Group IA element in a compound is+1.
- The oxidation number of each Group IIA element in a compound is+2.
- *The oxidation **number of each Group VIIA element in a compound is –1, except when combined with an element of higher electronegativity.* For example, in HCl, the oxidation number of Cl is –1; in HOCl, however, the oxidation number of Cl is +1.
- The oxidation number of hydrogen is usually+1; however, its oxidation number is–1in compounds with less electronegative elements (Groups IA and IIA). Hydrogen is +1 in HCl, but –1 in NaH.
- In most compounds, the oxidation number of oxygen is–2. The two exceptions are peroxides (O22−), for which the charge on each oxygen is –1, and compounds with more electronegative elements, such as OF2, in which oxygen has a +2 charge.
- The sum of the oxidation numbers of all the atoms present in a neutral compound is zero. The sum of the oxidation numbers of the atoms present in a polyatomic ion is equal to the charge of the ion.Thus, for (SO42−), the sum of the oxidation numbers must be –2.
MCAT EXPERTISE
Think of the oxidation number as the typical charge of an element based on its group number, metallicity, and general location in the periodic table.
KEY CONCEPT
The conventions of formula writing put cation first and anion second. Thus HCl implies H+, and NaH implies H–. Use the way the compound is written on the MCAT along with the periodic table to determine oxidation states.
MCAT EXPERTISE
Don’t forget that you can click on “Periodic table” to pull it up on Test Day. Note the trends for assigning oxidation numbers; these general rules will help reduce the need to memorize individual oxidation numbers. Be aware that the transition metals can take on multiple oxidation states and therefore multiple oxidation numbers.
Oxidation number is often confused with formal charge, discussed in Chapter 3 of MCAT General Chemistry Review. Both account for the perceived charge on an element, but do so in different ways. Oxidation number assumes unequal division of electrons in bonds, “awarding” the electrons to the more electronegative element. Formal charge, on the other hand, assumes equal division of electrons in bonds, “awarding” one electron to each atom in the bond. In reality, the distribution of electron density lies somewhere between these two extremes. The assigning of oxidation numbers can be seen in Figure 11.2.
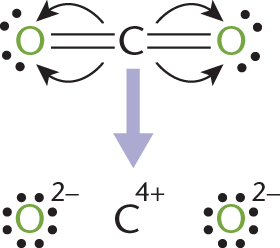
Figure 11.2. Assigning Oxidation Numbers to Carbon Dioxide
When assigning oxidation numbers, start with the known atoms (usually Groups I and II, halides, and oxygen) and use this information to determine the oxidation states of the other atoms. Keep in mind that most transition metals can take on multiple oxidation states. When transition metals are oxidized or reduced, the absorption and emission of light from a metal is altered such that different frequencies are absorbed. For this reason, changes of oxidation state in transition metals usually correspond to a color change.
Example: Assign oxidation numbers to the atoms in the following reaction to determine the oxidizing and reducing agents.
SnCl2 + PbCl4 → SnCl4 + PbCl2
Solution: All of these species are neutral, so the oxidation numbers of each compound must add up to zero. In SnCl2, tin must have an oxidation number of +2 because there are two chlorines present and each chlorine has an oxidation number of –1. Similarly, the oxidation number of Sn in SnCl4 is +4; the oxidation number of Pb is +4 in PbCl4 and +2 in PbCl2.
The oxidation number of Sn goes from +2 to +4; it loses electrons and thus is oxidized, making it the reducing agent. Because the oxidation number of Pb has decreased from +4 to +2, it gains electrons and is reduced, making it the oxidizing agent. The sum of the charges on both sides of the reaction is equal to zero, so charge has been conserved. Keep in mind that oxidation state also plays a role in nomenclature; the reactants in this reaction would be called tin(II) chloride and lead(IV) chloride.
Balancing Oxidation–Reduction Reactions
By assigning oxidation numbers to the reactants and products, one can determine how many moles of each species are required for conservation of charge and mass, which is necessary to balance the equation. To balance a redox reaction, both the net charge and the number of atoms must be equal on both sides of the equation. The most common method for balancing redox equations is the half-reaction method, also known as the ion–electron method, in which the equation is separated into two half-reactions—the oxidation part and the reduction part. Each half-reaction is balanced separately, and they are then added to give a balanced overall reaction.
KEY CONCEPT
Oxidizing agents oxidize other molecules, but are themselves reduced. Reducing agents reduce other molecules, but are themselves oxidized. If you determine one ion to be an oxidizing agent then the other must be a reducing agent.
Example: Balance this redox reaction using the half-reaction method:
MnO4− + l− → l2 + Mn2+
Step 1: Separate the two half-reactions. I − → I 2 MnO 4 − → Mn 2 +
Step 2: Balance the atoms of each half-reaction. First, balance all atoms except H and O. Next, in an acidic solution, add H2O to balance the O atoms and then add H+ to balance the H atoms. In a basic solution, use OH– and H2O to balance the O and H atoms. 2 I − → I 2 MnO 4 − + 8 H + → Mn 2 + + 4 H 2 O
Step 3: Balance the charges of each half-reaction. Add electrons as necessary to one side of the reaction so that the charges are equal on both sides. 2 I − → I 2 + 2 e − MnO 4 − + 8 H + + 5 e − → Mn 2 + + 4 H 2 O
Step 4: Both half-reactions must have the same number of electrons so that they cancel each other out in the next step. In this example, you need to multiply the oxidation half-reaction by 5 and the reduction half-reaction by 2. 10 I − → 5 I 2 + 10 e − 2 MnO 4 − + 16 H + + 10 e − → 2 Mn 2 + + 8 H 2 O
Step 5: Add the half-reactions, canceling out terms that appear on both sides of the reaction arrow.
2 MnO4− + 16 H+ + 10 I− → 2 Mn2+ + 5 I2 + 8 H2O
Step 6: Confirm that mass and charge are balanced. There is a +4 net charge on each side of the reaction equation, and the atoms are stoichiometrically balanced.
MCAT EXPERTISE
Methodical, step-by-step approaches like the half-reaction method are great for the MCAT. Usually, you will not have to go through all of these steps before you can narrow down your answer choices and may be able to find the correct answer partway through the problem with a little critical thinking.
MCAT CONCEPT CHECK 11.1:
Before you move on, assess your understanding of the material with these questions.
-
For each of the reactions below, identify the oxidation states of the relevant atoms, the oxidizing agent, and the reducing agent:
Reaction Oxidation Numbers Oxidizing Agent Reducing Agent
2 KI + H2 → 2 K + 2 HI
Al + BPO4 → B + AlPO4
-
Identify the oxidation and reduction half-reactions in the following redox reaction:
Zn + Cu2+ → Zn2+ + Cu
- Oxidation:________________________________
- Reduction:____________________________
-
Balance the following redox reaction using the half-reaction method:
Mg (s) + HNO3 (aq) → Mg2+ (aq) + NO (g)
11.2 Net Ionic Equations
LEARNING OBJECTIVES
After Chapter 11.2, you will be able to:
- Identify the element undergoing disproportionation and the oxidation state of the products for a given reaction
- Apply redox reaction principles to balance and solve application-style problems, such as ones that involve redox titrations
- Determine the net ionic equation for a reaction:
Zn (s) + CuSO4 (aq) → Cu (s) + ZnSO4 (aq)
When we discussed reaction types in Chapter 4 of *MCAT General Chemistry **Review*, we left out the rationale for why certain elements come together and others do not. Now that we have discussed oxidation–reduction reactions, it should be clearer that the gain and loss of electrons drives the formation of many compounds, especially ionic ones. Below, we will revisit many important reaction types and understand their basis in oxidation–reduction reactions.
Overview
In our discussion of acids and bases, we focused only on the presence of protons and hydroxide ions, with little concern for which species actually provided those ions. Similarly, in redox reactions, our focus is on the shifting of electrons more so than the identities of the ions themselves. Consider the following single-displacement reaction:
Zn (s) + CuSO4 (aq) → Cu (s) + ZnSO4 (aq)
If we split the various species into all of the ions present, we get the complete ionic equation:
Zn (s) + Cu2+ (aq) + SO42− (aq) → Cu (s) + Zn2+ (aq) + SO42− (aq)
Note that the sulfate ion is present on both sides of the equation in the same form; this ion is chemically inert during this reaction. In other words, the sulfate is not taking part in the overall reaction but simply remaining in the solution unchanged. We call such species spectator ions. Because the sulfate ion is not involved in the oxidation–reduction reaction, we can simplify the reaction to its net ionic equation, showing only the species that actually participate in the reaction:
Zn (s) + Cu2+ (aq) → Cu (s) + Zn2+ (aq)
When writing net ionic equations, all aqueous compounds should be split into their constituent ions. Solid salts, on the other hand, should be kept together as a single entity. Let’s return to some of the other reactions we have seen previously, including combination, decomposition, combustion, and double-displacement (metathesis) reactions.
MCAT EXPERTISE
Look for compounds such as polyatomic anions that retain their charge before and after reactions; these are usually spectator ions and will not be found in the net ionic equation.
Combination Reactions
In combination reactions, two or more species come together to form a product. For example:
H 0 2 ( g ) + F 2 0 ( g ) → 2 H + 1 F − 1 ( a q )
The relevant half-reactions would be:
H 2 → 2 H + + 2 e − F 2 + 2 e − → 2 F −
The net ionic equation is:
H2 + F2 → 2 H+ + 2 F–
In this reaction, molecular hydrogen acts as a reducing agent as it is oxidized from 0 to +1. Molecular fluorine is the oxidizing agent as it is reduced from 0 to –1. In this reaction, there is no spectator ion.
Decomposition Reactions
In decomposition reactions, one product breaks down into two or more species. For example:
( N − 3 H + 1 4 ) 2 Cr + 6 2 O − 2 7 ( a q ) → N 0 2 ( g ) + Cr + 3 2 O − 2 3 ( s ) + 4 H + 1 2 O − 2 ( g )
The relevant half-reactions would be:
2 NH 4 + → N 2 + 8 H + Cr 2 O 7 + 8 H + → Cr 2 O 3 + 4 H 2 O
The net ionic equation is:
2 NH4+ + Cr2O72− → N2 + Cr2O3 + 4 H2O
In this reaction, the nitrogen atom in the ammonium cation acts as a reducing agent as it is oxidized from –3 to 0. The chromium in the dichromate anion acts as the oxidizing agent as it is reduced from +6 to +3. In this reaction, there is no spectator ion. Note that the net ionic equation is not significantly different from the original balanced equation.
Combustion Reactions
In combustion reactions, a fuel (usually a hydrocarbon) is mixed with an oxidant (usually oxygen), forming carbon dioxide and water. For example:
C − 4 H + 1 4 ( g ) + 2 O 0 2 ( g ) → C + 4 O − 2 2 ( g ) + 2 H + 1 2 O − 2 ( l )
The relevant half-reactions would be:
CH 4 + 2 H 2 O → CO 2 + 8 H + + 8 e − 2 O 2 + 8 H + + 8 e − → 4 H 2 O
The net ionic equation is identical to the overall balanced equation because there are no spectator ions and no aqueous species:
CH4 + 2 O2 → CO2 + 2 H2O
Combustion reactions can have complex half-reactions, depending on the type of fuel used. In this instance, carbon in methane is the reducing agent as it is oxidized from –4 to +4. Molecular oxygen is the oxidizing agent as it is reduced from 0 to –2.
Double-Displacement (Metathesis) Reactions
Double-displacement or metathesis reactions involve the switching of counterions. Because all ions generally retain their oxidation state, these are not usually oxidation–reduction reactions. For example:
Ag + 1 NO 3 ︷ − 1 ( a q ) + H + 1 Cl − 1 ( a q ) → H + 1 NO 3 ︷ − 1 ( a q ) + Ag + 1 Cl − 1 ( s )
Because all species retain the same oxidation numbers, this is not considered oxidation–reduction. The net ionic reaction would be:
Ag+ + Cl– → AgCl
The nitrate anion and hydrogen cation both act as spectator ions in this reaction.
In double-displacement reactions where both reactants and both products are aqueous, there is no net ionic reaction. For example:
Na + 1 NO 3 ︷ − 1 ( a q ) + H + 1 Cl − 1 ( a q ) → H + 1 NO 3 ︷ − 1 ( a q ) + Na + 1 Cl − 1 ( a q )
This reaction is not an oxidation–reduction reaction because no species change their oxidation states. Further, because all species are aqueous, the complete ionic reaction is:
Na+ + NO3− + H+ + Cl− → H+ + NO3− + Na+ + Cl−
Because all of the ions appear on both sides of the reaction, there is no net ionic reaction.
Disproportionation Reactions
Disproportionation (or dismutation) is a specific type of redox reaction in which an element undergoes both oxidation and reduction in producing its products. Many biological enzymes utilize a disproportionation mechanism. An example of such a reaction is the catalysis of peroxides by catalase, an enzyme found in peroxisomes. Catalase is a critical biological enzyme used to protect cells from excessive oxidation by free radicals or reactive oxygen species. The activity of catalase can be seen when disinfecting a wound with hydrogen peroxide:
2 H + 1 2 O − 1 2 ( a q ) → catalase 2 H + 1 2 O − 2 ( l ) + O 0 2 ( g )
As can be seen from this reaction, oxygen is disproportioned between water and molecular oxygen. In hydrogen peroxide, each oxygen has an oxidation state of –1 (the peroxide ion has a charge of –2 overall). In water, oxygen has an oxidation state of –2, and in molecular oxygen, it has an oxidation state of 0. Therefore, the oxygen is both reduced and oxidized in this reaction.
Another related biological disproportionation mechanism is that of the enzyme superoxide dismutase. As the name implies, a dismutase catalyzes dismutation. This enzyme disproportions oxygen free radicals in the reaction
2 O 2 · − − 1 2 + 2 H + + 1 → H + 1 2 O − 1 2 + O 0 2
where peroxide and oxygen are the disproportioned products. The oxidation state of oxygen in the free radical is − 1 2 (a negative charge divided over two oxygen atoms), and it is reduced to –1 in the peroxide and oxidized to 0 in molecular oxygen.
Biochemical disproportionation reactions—and oxidation–reduction reactions in biological systems in general—are usually accomplished by enzymes. Structurally, these enzymes often have metals such as Cu and Zn in their active sites that act as reducing agents, as shown in Figure 11.3.
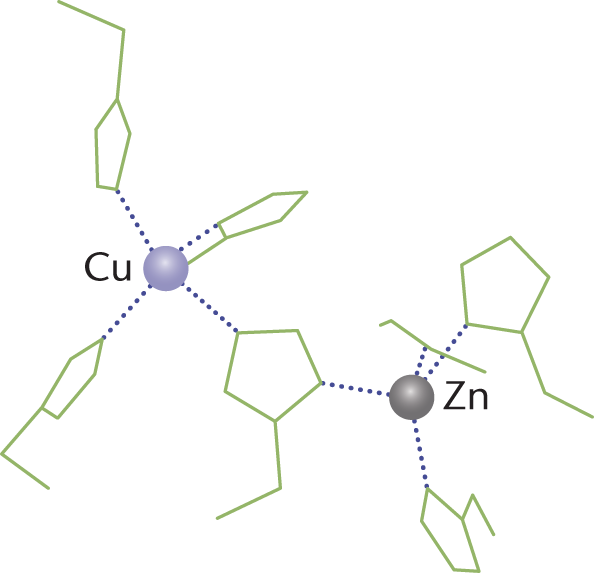
Figure 11.3. Active Site of Superoxide Dismutase Cu and Zn atoms act as reducing agents, losing electrons during catalysis. The atoms are stabilized in position by histidine residues.
Oxidation–Reduction Titrations
Oxidation–reduction titrations are similar in setup to acid–base titrations, but the focus is different. Whereas acid–base titrations follow the movement of protons, as discussed in Chapter 10 of MCAT General Chemistry Review, redox titrations follow the transfer of charge (as electrons) to reach the equivalence point. Redox titrations can utilize indicators that change color at a particular voltage (emf) value. Some common indicators are listed in Table 11.2. As for acid–base titrations, it is not necessary to memorize these indicators, but rather to understand their utility.
Table 11.2. Common Indicators for Oxidation–Reduction (Redox) Titrations
INDICATOR VOLTAGE OF COLOR CHANGE OXIDIZED FORM REDUCED FORM
Bipyridine metal complexes About +1 V Colorless (Ru), Cyan (Fe) Yellow (Ru), Red (Fe)
Diphenylamine +0.76 V Violet Colorless
Safranin +0.24 /–0.29 V* Red-Violet Colorless
* Safranin is unique in that its color change is not only voltage dependent, but also pH dependent.
One prototypical redox titration involves the use of starch indicators to identify iodine complexes. This specific redox titration is called an iodimetric titration because it relies on the titration of free iodine radicals. The presence of iodine is initially determined by a dark solution in the presence of starch, and at the endpoint of the titration, a colorless solution develops. A common general chemistry laboratory experiment involves the standardization of a thiosulfate solution using iodimetry, as described in the example below.
Example: A group of students prepares to standardize a Na2S2O3 solution. 32 mL of the Na2S2O3 solution is titrated into 50 mL of a 0.01 M KIO3 solution to reach the equivalence point. They first titrate the KIO3 solution until it loses color, then add a starch indicator until the reaction is complete. The reaction proceeds in these two steps: IO 3 − + I − + H + → I 3 − + H 2 O I 3 − + S 2 O 3 2 − → I − + S 4 O 6 2 −
Determine the concentration of the sodium thiosulfate solution at the beginning of the experiment.
Solution: The titration is performed in two steps. In the first step, the iodate is converted into triiodide anions. The initial titration creates a colorless l3− solution in water, which then requires a starch indicator for the remainder of the titration.
In the second step, the triiodide ions are then reduced in the presence of thiosulfate to determine its concentration. Note that the reactions are unbalanced. It would be tempting to balance the first equation as:
IO3− + 2 I− + 6 H+ → I3− + 3 H2O
However, this reaction has a charge of +3 on the reactants side and –1 on the products side. In oxidation–reduction chemistry, we must balance not only for stoichiometry but also for charge. The correct balanced equations are: IO 3 − + 8 I − + 6 H + → 3 I 3 − + 3 H 2 O I 3 − + 2 S 2 O 3 2 − → 3 I − + S 4 O 6 2 −
Keeping in mind that each iodate is used to make three triiodide anions, and each triiodide anion interacts with two thiosulfate anions, the mole ratio of thiosulfate to iodate is six to one: IO 3 − + 8 I − + 6 H + → 3 I 3 − + 3 H 2 O 3 I 3 − + 6 S 2 O 3 2 − → 9 I − + 3 S 4 O 6 2 −
Then, use stoichiometry to determine the molarity of the thiosulfate solution from the 50 mL potassium iodate solution. 0.01 M IO 3 − [ 0.05 L ] [ 6 mol S 2 O 3 2 − 1 mol IO 3 − ] = 3 × 10 − 3 mol
Now, we can use the volume to find the molarity.
M S 2 O 3 2 − = 3 × 10 − 3 mol 32 mL [ 1 L 1000 mL ] = 3 × 10 − 3 32 × 10 − 3 ≈ 3 30 = 0.1 M (actual = 0 .094 M )
Potentiometric titration is a form of redox titration where no indicator is used. Instead, the electrical potential difference (voltage) is measured using a voltmeter. As a redox titration progresses, its voltage changes; this is analogous to following an acid–base titration with a pH meter instead of a color indicator.
MCAT CONCEPT CHECK 11.2:
Before you move on, assess your understanding of the material with these questions.
-
Write the net ionic equations for the reactions below:
- CuNO3 (aq) + NaCl (aq) → CuCl (s) + NaNO3(aq) ________________________________
- Mg (s) + AlCl3 (aq) → Al (s) + MgCl2 (aq) ________________________
-
In each of the reactions below, which element undergoes disproportionation? What are that element’s oxidation states in the products?
3 Cl2 (g) + 6 NaOH (aq) → 5 NaCl (aq) + NaClO3 (aq) + 3 H2O (l)
S2O32− (aq) + 2 H+ (aq) → S (s) + SO2 (g) + H2O (l)
- Element undergoing disproportionation: ________________________________
- Oxidation states in products: ________________________________
- Element undergoing disproportionation: ________________________________
- Oxidation states in products: _____________________
-
A sample is assayed for lead by a redox titration with I3− (aq). A 10.00 g sample is crushed, dissolved in sulfuric acid, and passed over a reducing agent so that all the lead is in the form Pb2+. The Pb2+ (aq) is completely oxidized to Pb4+ by 32.60 mL of a 0.7 M solution of NaI3. The balanced equation for the reaction is:
I3− + (aq) + Pb2+ (aq) → Pb4+ (aq) + 3 I− (aq)
Calculate the mass of lead in the sample.
____________________________________
Conclusion
In this chapter, we covered the essential MCAT topic of oxidation–reduction reactions. We reviewed the rules for assigning oxidation numbers to help us keep track of the movement of electrons from the species that are oxidized (reducing agents) to the species that are reduced (oxidizing agents). We also covered the sequence of steps involved in balancing half-reactions, redox titrations, and disproportionation reactions.
In addition to understanding the fundamental chemical principles behind these reactions, you will begin to see these concepts resurface in MCAT Organic Chemistry Review and MCAT Biochemistry Review. Oxidation–reduction reactions are often used for energy transfer in biological systems, and any deficiencies in such systems are profoundly deleterious (such as metabolic, mitochondrial, and immunologic diseases). Our next chapter—the last of MCAT General Chemistry Review—brings the principles of oxidation–reduction reactions to their application in electrochemical cells. By the end of the next chapter, you will have reviewed all of the general chemistry knowledge required for Test Day!
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Oxidation–Reduction Reactions
- Oxidation is a loss of electrons, and reduction is a gain of electrons; the two are paired together in what is known as an oxidation–reduction (redox) reaction.
- An oxidizing agent facilitates the oxidation of another compound and is reduced itself in the process; a reducing agent facilitates the reduction of another compound and is itself oxidized in the process.
- Common oxidizing agents almost all contain oxygen or a similarly electronegative element.
- Common reducing agents often contain metal ions or hydrides (H–).
- To assign oxidation numbers, one must know the common oxidation states of the representative elements.
- Any free element or diatomic species has an oxidation number of zero.
- The oxidation number of a monatomic ion is equal to the charge of the ion.
- When in compounds, Group IA metals have an oxidation number of +1; Group IIA metals have an oxidation number of +2.
- When in compounds, Group VIIA elements have an oxidation number of –1 (unless combined with an element with higher electronegativity).
- The oxidation state of hydrogen is +1 unless it is paired with a less electronegative element, in which case it is –1.
- The oxidation state of oxygen is usually –2, except in peroxides (when its charge is –1) or in compounds with more electronegative elements.
- The sum of the oxidation numbers of all the atoms present in a compound is equal to the overall charge of that compound.
- When balancing redox reactions, the half-reaction method, also called the ion–electron method, is the most common.
- Separate the two half-reactions.
- Balance the atoms of each half-reaction. Start with all the elements besides H and O. In acidic solution, balance H and O using water and H+. In basic solution, balance H and O using water and OH–.
- Balance the charges of each half-reaction by adding electrons as necessary to one side of the reaction.
- Multiply the half-reactions as necessary to obtain the same number of electrons in both half-reactions.
- Add the half-reactions, canceling out terms on both sides of the reaction arrow.
- Confirm that the mass and charge are balanced.
Net Ionic Equations
- A complete ionic equation accounts for all of the ions present in a reaction. To write a complete ionic reaction, split all aqueous compounds into their relevant ions. Keep solid salts intact.
- Net ionic equations ignore spectator ions to focus only on the species that actually participate in the reaction. To obtain a net ionic reaction, subtract the ions appearing on both sides of the reaction, which are called spectator ions.
- For reactions that contain no aqueous salts, the net ionic equation is generally the same as the overall balanced reaction.
- For double displacement (metathesis) reactions that do not form a solid salt, there is no net ionic reaction because all ions remain in solution and do not change oxidation number.
- Disproportionation (dismutation) reactions are a type of redox reaction in which one element is both oxidized and reduced, forming at least two molecules containing the element with different oxidation states.
- Oxidation–reduction titrations are similar in methodology to acid–base titrations. These titrations follow transfer of charge.
- Indicators used in such titrations change color when certain voltages of solutions are achieved.
- Potentiometric titration is a form of redox titration in which a voltmeter or external cell measures the electromotive force (emf) of a solution. No indicator is used, and the equivalence point is determined by a sharp change in voltage.
ANSWERS TO CONCEPT CHECKS
**11.1**
-
Reaction Oxidation Numbers Oxidizing Agent Reducing Agent
2 KI + H2 → 2 K + 2 HI 2 K + 1 I − 1 + H 0 2 → 2 K 0 + 2 H + 1 I − 1 K+ (charge goes from +1 to 0) H2 (charge goes from 0 to +1)
Al + BPO4 → B + AlPO4 Al 0 + B + 3 PO 4 ︷ − 3 → B 0 + Al + 3 PO 4 ︷ − 3 B3+ (charge goes from +3 to 0) Al (charge goes from 0 to +3)
- Oxidation: Zn → Zn2+ + 2 e–
Reduction: Cu2+ + 2 e– → Cu
- 1. { Mg → Mg 2 + HNO 3 → NO 2. { Mg → Mg 2 + HNO 3 + 3 H + → NO + 2 H 2 O 3. { Mg → Mg 2 + + 2 e − HNO 3 + 3 H + + 3 e − → NO + 2 H 2 O 4. { 3 Mg → 3 Mg 2 + + 6 e − 2 HNO 3 + 6 H + + 6 e − → 2 NO + 4 H 2 O 5. 2 HNO 3 + 3 Mg + 6 H + → 2 NO + 3 Mg 2 + + 4 H 2 O
**11.2**
- Cu+ + Cl– → CuCl
3 Mg + 2 Al3+ → 3 Mg2+ + 2 Al (don’t forget to balance the reaction!)
- In the first reaction, chlorine undergoes disproportionation to have a –1 oxidation state in NaCl and a +5 oxidation state in NaClO3.
In the second reaction, sulfur undergoes disproportionation to have a 0 oxidation state in elemental sulfur and +4 oxidation state in SO2.
- 0.7 M I 3 − [ 32.60 × 10 − 3 L ] [ 1 mol Pb 2 + 1 mol I 3 − ] [ 207.2 g Pb 2 + 1 mol Pb 2 + ] ≈ 0.7 × 3 × 2 = 4.2 g (actual = 4 .73 g)
Note that question 3 also included the extraneous value 10.0 g, which is not needed to calculate the mass of lead produced.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. C
The oxidizing agent is the species that is reduced in any given equation. In this problem, six hydrogen atoms with +1 oxidation states in NH3 are reduced to three neutral H2 molecules.
2. B
First, balance the atoms in the equation:
Cr2O72− + 14 H+ → 2 Cr2+ + 7 H2O
Now, adjust the number of electrons to balance the charge. Currently, the left side has a charge of +12 (–2 from dichromate and +14 from protons). The right side has a charge of +4 (+2 from each chromium cation). To decrease the charge on the left side from +12 to +4, we should add 8 electrons:
Cr2O72− + 14 H+ + 8 e− → 2 Cr2+ + 7 H2O
3. A
Hydride ions are composed of a hydrogen nucleus with two electrons, thereby giving it a negative charge and a considerable tendency to donate electrons. LiAlH4 is therefore a strong reducing agent. Strong reducing agents tend to have metals or hydrides; strong oxidizing agents tend to have oxygen or a similarly electronegative element.
4. C
In NaClO (sodium hypochlorite), sodium carries its typical +1 charge, and oxygen carries its typical –2 charge. This means that the chlorine atom must carry a +1 charge in order to balance the overall charge of zero.
5. D
A strong oxidizing agent will be easily reduced, meaning that it will have a tendency to gain electrons. Atoms usually gain electrons if they are one or two electrons away from filling up their valence shell. (A) has a full 4s-orbital, meaning that it can only gain an electron if it gains an entire subshell. (B) has a stable, half-full 3d-orbital, so it is unlikely to pick up electrons unless it can gain five. (C) has only a single electron in the outer shell, which is more likely lost upon ionization. (D) would fill up its 4p-orbital by gaining one electron, so it is easily reduced.
6. D
A net ionic equation represents each of the aqueous ions comprising the reactants and products as individual ions, instead of combining them as formula units. Thus, (A) is not a net ionic reaction. The term net means that the correct answer does not include any spectator ions (ions that do not participate in the reaction). In this reaction, nitrate (NO3−) remains unchanged. Therefore, (B) and (C) are eliminated.
7. C
What you are shown is a net ionic equation. If two moles of FeSCN are created, two moles of Fe3+ must be used because the mole ratio is 1:1. Iron sulfate has the formula Fe2(SO4)3 because sulfate has a charge of –2 and iron has a charge of +3 (based on the net ionic equation). Therefore, one mole of iron sulfate is needed to make two moles of iron for the reaction. The molar mass of iron sulfate is
2 × 55.8 g mol + 3 × 32.1 g mol + 12 × 16.0 g mol = 399.9 g mol
This most closely matches answer (C). The most common error would be to calculate the amount of iron, which would be 111.6 g, (A).
8. D
When assigning oxidation numbers, one starts with elements of known oxidation state first, and determines the oxidation state of the other elements by deduction. As a noble gas, argon, (A), will always have an oxidation state of 0. As a Group VIIA element, fluorine, (B), will have an oxidation state of 0 (by itself) or –1 (in a compound). As a Group IIA element, strontium, (C), will have an oxidation state of 0 (by itself) or +2 (in a compound). Like most transition metals, iridium, (D), can have various oxidation states, ranging from –3 to +8. Therefore, one would have to determine the oxidation states of other atoms in an iridium-containing compound to determine iridium’s oxidation number.
9. A
The formula for methanol is H3COH, for methanal is HCHO, and for methanoic acid is HCOOH. If we assign oxidation numbers to carbon in each molecule, it starts at –2, then becomes 0, then becomes +2: H + 1 3 C − 2 O − 2 H + 1 → H + 1 C 0 H + 1 O − 2 → H + 1 C + 2 O − 2 O − 2 H + 1
In general, it is often easier to think of oxidation as a gain of bonds to oxygen (or a similarly electronegative element) or loss of bonds to hydrogen for organic compounds. Therefore, because the carbon is oxidized as one converts from an alcohol to an aldehyde to a carboxylic acid, the oxidation number must increase.
10. C
Start with the atoms that have oxidation states of which you are certain. Potassium is a Group IA metal, and therefore must have an oxidation state of +1. Hydrogen is almost always +1, unless it is paired with a less electronegative element (which is not the case here). Oxygen is generally –2. Because there are four oxygens, they create a total negative charge of –8 which is partially balanced by two hydrogens (+2) and potassium (+1). Therefore, phosphorus has a +5 charge, making it the highest oxidation state.
11. B
Recall that oxygen has an oxidation state of –2. Therefore, in tungsten(IV) oxide, (A), tungsten has an oxidation state of +4. In tungsten(VI) oxide, (B), it has an oxidation state of +6. In tungsten(III) oxide, (C), it is +3. In tungsten pentoxide, (D), it is +5.
12. A
Step I is a disproportionation reaction because chlorine starts with an oxidation state of 0 in the reactants and ends up with an oxidation state of +1 in HOCl and –1 as Cl–. In the other reactions, no element appears with different oxidation states in two different products. Therefore, only step I is a disproportionation reaction.
13. C
Potentiometry refers to carrying out an oxidation–reduction titration with a voltmeter present to get precise readings of the reaction’s electromotive force (emf) to determine the endpoint. This is analogous to using a pH meter in an acid–base titration because it uses technology to get precise readings for plotting a titration curve. Indicators, as in (A) and (B), can be used in both acid–base and redox titrations, but provide a qualitative (rather than quantitative) analysis of the titration. Oxidizing and reducing agents are used in redox titrations, not acid–base titrations, eliminating (D).
14. D
Utilize the method described earlier to balance this redox reaction. The balanced half-reactions are: S 8 + 24 H 2 O → 8 SO 3 2 − + 48 H + + 32 e − NO 3 − + 4 H + + 3 e − → NO + 2 H 2 O
To get equal numbers of electrons in each half-reaction, the oxidation half-reaction will have to be multiplied by 3, and the reduction half-reaction will have to be multiplied by 32: 3 S 8 + 72 H 2 O → 24 SO 3 2 − + 144 H + + 96 e − 32 NO 3 − + 128 H + + 96 e − → 32 NO + 64 H 2 O
This makes the overall reaction:
3 S8 + 32 NO3− + 8 H2O → 24 SO32− + 32 NO + 16 H+
The sum of the stoichiometric coefficients is therefore: 3 + 32 + 8 + 24 + 32 + 16 = 115.
15. C
First, balance the chemical equation:
4 Au + 8 NaCN + O2 + 2 H2O → 4 Na[Au(CN)2] + 4 NaOH
Now, determine the number of moles of NaCN used in the reaction: 0.1 L × 2 mol L = 0.2 mol NaCN
If 0.2 mol NaCN are used in the reaction, then 0.2 mol NaCN × 4 mol Au 8 mol NaCN = 0.1 mol Au is oxidized.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
Biochemistry Chapter 2
Enzymes
Biochemistry Chapter 10
Carbohydrate Metabolism II
Biology Chapter 7
The Cardiovascular System
General Chemistry Chapter 10
Acids and Bases
General Chemistry Chapter 12
Electrochemistry
Organic Chemistry Chapter 4
Analyzing Organic Reactions