Chapter 1: Nomenclature

Chapter 1: Nomenclature
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what’s important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don’t worry though—skipping something now does not mean you’ll never study it. Later on in your prep, as you complete full-length tests, you’ll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- Which of the following lists the correct common names for ethanal, methanal, and ethanol, respectively?
- Acetaldehyde, formaldehyde, ethyl alcohol
- Ethyl alcohol, propionaldehyde, isopropyl alcohol
- Ethyl alcohol, formaldehyde, acetaldehyde
- Isopropyl alcohol, ethyl alcohol, formaldehyde
- Which of the following are considered terminal functional groups?
- Aldehydes
- Ketones
- Carboxylic acids
- I only
- III only
- I and III only
- I, II, and III
- If all prefixes were dropped, what would be the name of the parent root of this molecule?
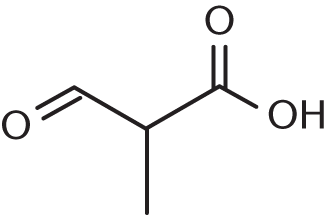
- Propanoate
- Propanol
- Propanoic acid
- Propanoic anhydride
- What is the highest-priority functional group in this molecule?
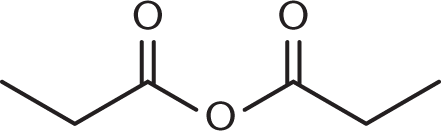
- Anhydride
- Carbonyl
- Ketone
- Alkyl chain
- The IUPAC name for the following structure ends in what suffix?
- –ol
- –one
- –oic acid
- –yne
- Which of the two possibilities below correctly numbers the carbon backbone of this molecule?
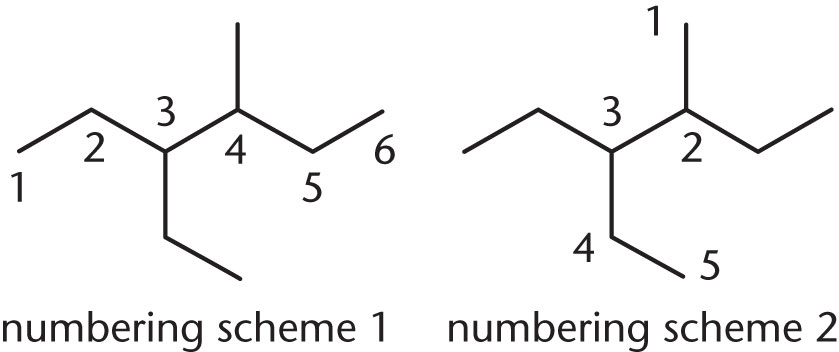
- Numbering scheme 1
- Numbering scheme 2
- Numbering schemes 1 and 2 are equivalent and correct.
- Numbering schemes 1 and 2 are equivalent and incorrect.
- What is the proper structure for 2,3-dihydroxybutanedioic acid (tartaric acid)?
-
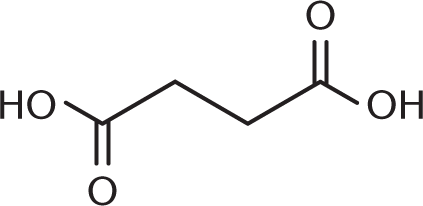
-
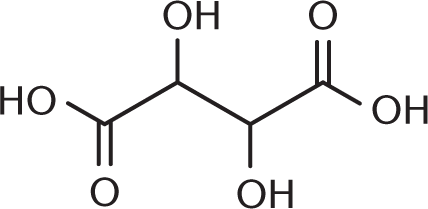
-
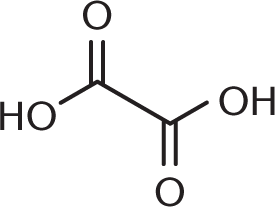
-
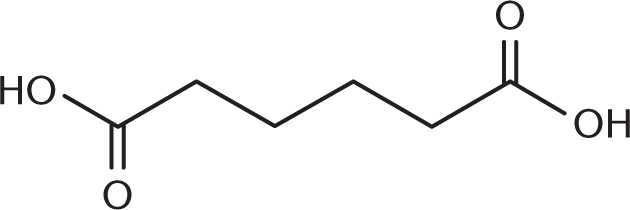
- The common names for the aldehydes and carboxylic acids that contain only one carbon start with which prefix?
- Para–
- Form–
- Meth–
- Acet–
- What is the IUPAC name for the following structure?
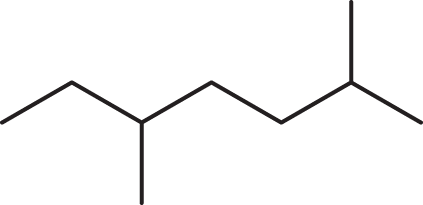
- 2,5-dimethylheptane
- 2-ethyl-5-methylhexane
- 3,6-dimethylheptane
- 5-ethyl-2-methylhexane
- What is the IUPAC name for the following structure?
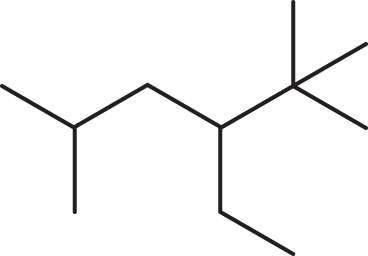
- 4-isopropyl-2-methylhexane
- 3-isopropyl-5-methylhexane
- 2,2,5-trimethyl-3-ethylhexane
- 3-ethyl-2,2,5-trimethylhexane
- The IUPAC name for the following structure starts with what prefix?
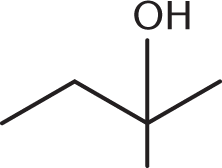
- 3-methyl-
- 2-methyl-
- 2-hydroxy-
- 3-hydroxy-
- NADH is a coenzyme that releases high-energy electrons into the electron transport chain. It is known as nicotinamide adenine dinucleotide or diphosphopyridine nucleotide. What functional groups exist in this molecule?
- Phosphate
- Amide
- Anhydride
- I only
- II only
- I and II only
- I, II, and III
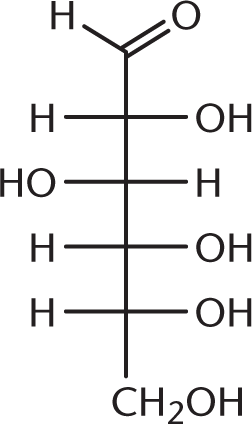
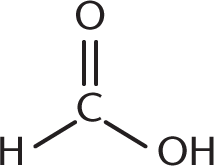
- Pyruvic acid, one of the end products of glycolysis, is commonly called acetylformic acid. Based on its common name, the structure of pyruvic acid must be:
-
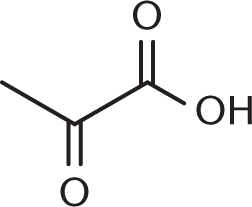
-
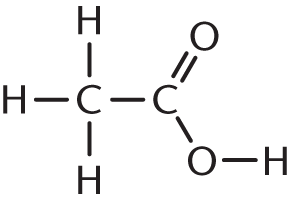
-
-
- Which of the following are common names for carboxylic acid derivatives?
- Acetic anhydride
- Formic acid
- Methyl formate
- I and II only
- I and III only
- II and III only
- I, II, and III
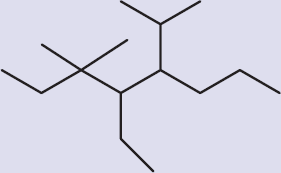
- Consider the name 2,3-diethylpentane. Based on the structure implied by this name, the correct IUPAC name for this molecule is:
- 2,3-diethylpentane.
- 1,2-diethylbutane.
- 3-ethyl-4-methylhexane.
- 3-methyl-4-ethylhexane.
Answer Key
- A
- C
- C
- A
- C
- A
- B
- B
- A
- D
- B
- C
- A
- B
- C
Chapter 1: Nomenclature
CHAPTER 1
NOMENCLATURE
In This Chapter
1.1 IUPAC Naming Conventions
Naming Steps
1.2 Hydrocarbons and Alcohols
Alkanes
Alkenes and Alkynes
Alcohols
1.3 Aldehydes and Ketones
Aldehydes
Ketones
1.4 Carboxylic Acids and Derivatives
Carboxylic Acids
Esters
Amides
Anhydrides
1.5 Summary of Functional Groups
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 4% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content category:
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
You walk into the pharmacy looking for something to take for a headache and find an entire aisle of drugs just for that purpose: Advil, Aleve, Motrin, Tylenol, ibuprofen, naproxen, acetaminophen, and aspirin. In this list, however, there are only four distinct drugs. In the United States, it is not uncommon for drugs to be known by both generic and brand names—and sometimes, multiple brands market the same medications. As a medical student, you’ll have to know both: while a doctor may order atorvastatin 40 mg qd, patients will tell you they’re taking Lipitor daily.
For doctors, a generic name is sufficiently unambiguous to specify a given compound, but this is not true within the pharmaceutical industry. Medications are usually large organic compounds with many functional groups and numerous chiral centers. Chemists needed to be able to describe such compounds, as well as innumerable others. Thus, within chemistry, a specific set of rules for naming and describing compounds was designed. In this chapter, we’ll examine the steps for naming a compound and then practice applying them to example compounds. By the end of the chapter, we’ll have discussed the most common functional groups for Test Day and how they relate to each other in the nomenclature hierarchy. Note that you may have learned nomenclature for a number of other compounds, including ethers, epoxides, amines, imines, sulfonic acids, and others in your organic chemistry courses; we have restricted the content of this chapter to only the functional groups you are expected to identify on Test Day.
MCAT EXPERTISE
According to the AAMC, the MCAT will not include standalone, nomenclature-only organic chemistry questions (such as “name this compound” questions). But as you probably remember from your own classes, nomenclature is the very foundation of the entire subject of organic chemistry, and you will lose points on Test Day if you don’t have nomenclature down cold.
1.1 IUPAC Naming Conventions
LEARNING OBJECTIVES
After Chapter 1.1, you will be able to:
- Identify the parent carbon chain in a complex molecule
- Describe how numbers are integrated into chemical nomenclature
- Apply the five steps of IUPAC nomenclature and name a molecule:
Nomenclature is one of the most important prerequisites for answering organic chemistry questions on Test Day; if you don’t know which chemical compound the question is asking about, it’s hard to get the answer right! That’s why it’s so important to understand both IUPAC and common nomenclature. Once you have a handle on these naming systems, you can easily translate question stems and focus on finding the correct answer. Let’s begin by examining IUPAC naming conventions before we highlight specific compounds and functional groups.
MCAT EXPERTISE
Nomenclature is often tested on the MCAT by providing the question stem and the answer choices in different formats. For example, the question stem may give you the IUPAC name of the reactant, and the answer choices may show product structures—leaving you to figure out both the structure of the reactant and the reaction taking place.
Naming Steps
The primary goal of the International Union of Pure and Applied Chemistry (IUPAC) naming system is to create an unambiguous relationship between the name and structure of a compound. With the conventions established by IUPAC, no two distinct compounds have the same name. The IUPAC naming system greatly simplifies chemical naming. Once we understand the rules, we can match names to structures with ease.
1. Identify the Longest Carbon Chain Containing the Highest-Order Functional Group
This will be called the parent chain and will be used to determine the root of the name. Keep in mind that if there are double or triple bonds between carbons, they must be considered when identifying the highest-order functional group. We’ll examine priorities of functional groups throughout this chapter, but keep in mind that the highest-priority functional group (with the most oxidized carbon) will provide the suffix. This step may sound easy, but be careful! The molecule may be drawn in such a way that the longest carbon chain is not immediately obvious. If there are two or more chains of equal length, then the more substituted chain gets priority as the parent chain. Figure 1.1 shows a hydrocarbon with the longest chain labeled.
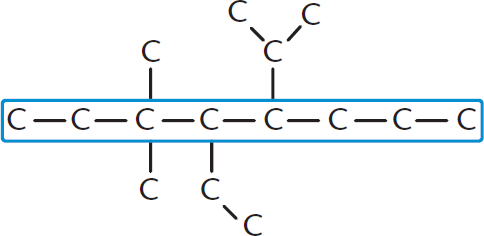
Figure 1.1. Finding the Longest Carbon Chain
2. Number the Chain
In order to appropriately name a compound, we need to number the carbon chain, as shown in Figure 1.2. As a convention, the carbon numbered 1 will be the one closest to the highest-priority functional group. If the functional groups all have the same priority, numbering the chain should make the numbers of the substituted carbons as low as possible.
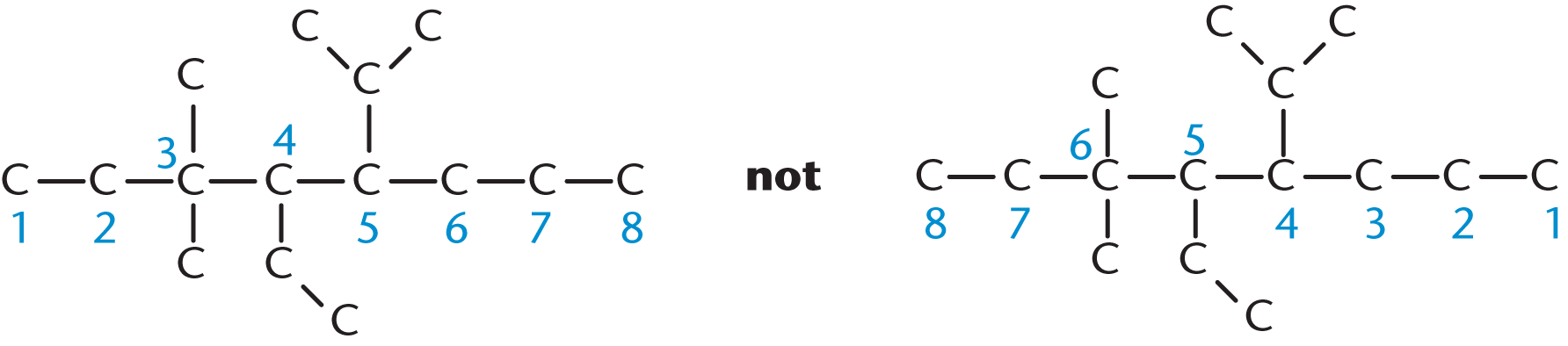
Figure 1.2. Numbering the Longest Carbon Chain The highest-priority functional group should have the lowest possible number; if all substituents have the same priority, as in this figure, make their numbers as low as possible.
After we have discussed the functional groups most commonly tested on the MCAT, we’ll review a table of those functional groups in order of priority. For now, keep in mind that the more oxidized the carbon is, the higher priority it has in the molecule. Oxidation state increases with more bonds to heteroatoms (atoms besides carbon and hydrogen, like oxygen, nitrogen, phosphorus, or halogens) and decreases with more bonds to hydrogen.
Just like straight chains, rings are numbered starting at the point of greatest substitution, continuing in the direction that gives the lowest numbers to the highest-priority functional groups. Somewhat counterintuitively, if there is a tie between assigning priority in a molecule with double and triple bonds, the double bond takes precedence.
3. Name the Substituents
Substituents are functional groups that are not part of the parent chain. A substituent’s name will be placed at the beginning of the compound name as a prefix, followed by the name of the longest chain. Remember that only the highest-priority functional group will determine the suffix for the compound and must be part of the parent chain.
Carbon chain substituents are named like alkanes, with the suffix –yl replacing –ane. The prefix n– that we see in Figure 1.3 on n-propyl simply indicates that this is “normal”—in other words, a straight-chain alkane. Because this prefix will not always be present, it is safe to assume that alkane substituents will be normal unless otherwise specified.
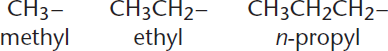
Figure 1.3. Common Normal Alkyl Substituents The bond on the right side of each substituent connects to the parent molecule.
In Figure 1.4, we see some examples of what alternative alkyl substituents may look like.
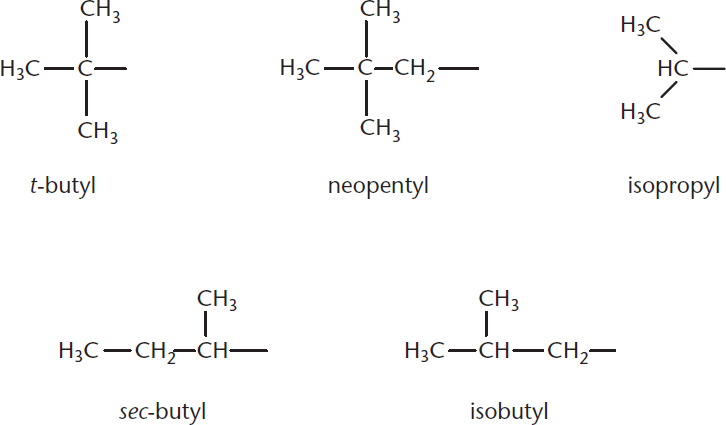
Figure 1.4. Common Alternative Alkyl Substituents The bond on the right side of each substituent connects to the parent molecule.
If there are multiple substituents of the same type, we use the prefixes di–, tri–, tetra–, and so on to indicate this fact. These prefixes are included directly before the substituent’s name.
4. Assign a Number to Each Substituent
Pair the substituents that you have named to the corresponding numbers in the parent chain. Multiple substituents of the same type will get both the di–, tri–, and tetra– prefixes that we have previously noted and also a carbon number designation—even if they are on the same carbon.
5. Complete the Name
Names always begin with the names of the substituents in alphabetical order, with each substituent preceded by its number. Note, however, that prefixes like di–, tri–, and tetra–, as well as the hyphenated prefixes like n– and tert– (or t–), are ignored while alphabetizing. Nonhyphenated roots that are part of the name, however, are included; these are modifiers like iso–, neo–, or cyclo–. Then the numbers are separated from each other with commas, and from words with hyphens. Finally, we finish the name with the name of the backbone chain, including the suffix for the functional group of highest priority. Figure 1.5 shows an example of an entire hydrocarbon named with IUPAC nomenclature.
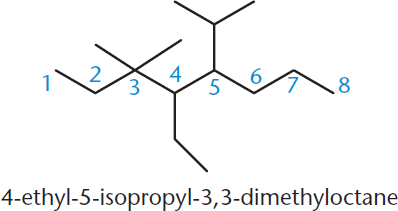
Figure 1.5. An Example of a Complete IUPAC Name
BRIDGE
Solutions to concept checks for a given chapter in MCAT Organic Chemistry Review can be found near the end of the chapter in which the concept check is located, following the Concept Summary for that chapter.
MCAT CONCEPT CHECK 1.1:
Before you move on, assess your understanding of the material with these questions.
- List the steps of IUPAC nomenclature:
- ____________________________
- ____________________________
- ____________________________
- ____________________________
- ____________________________
- Circle or highlight the parent chain in each of the following compounds:
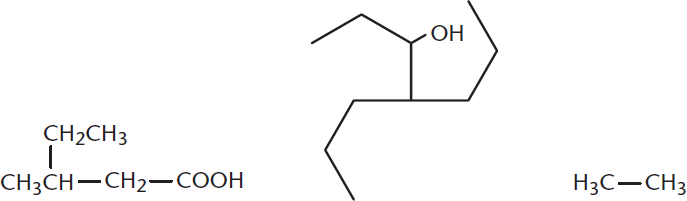
- Circle and name the substituents in the following molecule, then name the molecule.

1.2 Hydrocarbons and Alcohols
LEARNING OBJECTIVES
After Chapter 1.2, you will be able to:
- Predict the structure of a hydrocarbon or alcohol molecule when given a simple molecular formula, such as C9H20
- Differentiate between geminal and vicinal diols
- Recall common names of key compounds, such as 2-propanol
- Apply priority rules when naming molecules with multiple functional groups, such as:
Hydrocarbons are compounds that contain only carbon and hydrogen atoms. Alcohols, on the other hand, contain at least one –OH group, which lends them additional reactivity. In this section, we’ll explore the naming of hydrocarbons and alcohols.
Alkanes
Alkanes are simple hydrocarbon molecules with the formula CnH(2n+ 2). The names for the first four of these compounds are methane (one carbon), ethane (two carbons), propane (three carbons), and butane (four carbons). Alkanes with more than four carbons have a simpler naming pattern in which the name is the Greek root describing the number of carbons followed by –ane. From 5 to 12, these Greek roots are: pent–, hex–, hept–, oct–, non–, dec–, undec–, and dodec–. Some examples of alkanes are shown in Table 1.1.
Table 1.1 Examples of Alkanes
Number of Carbons Name Structure
1 methane

2 ethane

3 propane

4 butane

5 pentane

6 hexane
7 heptane
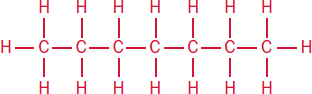
8 octane
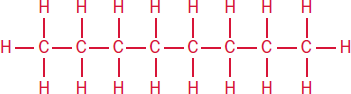
Halogens are common substituents on alkanes. Alkyl halides are indicated by a prefix: fluoro–, chloro–, bromo–, or iodo–.
Alkenes and Alkynes
The MCAT does not explicitly test reactions of alkenes or alkynes, but you may still see the suffixes –ene and –yne, which signify double and triple bonds, respectively. Keep in mind that many of these compounds will also have common names, and it is vital to know these common names as well. On Test Day, you are most likely to encounter double bonds in the context of unsaturated fatty acids or other biochemical compounds. The double or triple bond is named like a substituent and is indicated by the lower-numbered carbon involved in the bond. The number may precede the molecule name, as in 2-butene, or it may be placed near the suffix, as in but-2-ene; both are correct. If there are multiple multiple bonds, the numbering is generally separated from the suffix, as in 1,3-butadiene.
Alcohols
Alcohols are named by replacing the –e at the end of the name of the corresponding alkane with the suffix –ol. The chain is numbered so that the carbon attached to the hydroxyl group (–OH) gets the lowest possible number—even when there is a multiple bond present. The hydroxyl group takes precedence over multiple bonds because of the higher oxidation state of the carbon. If the alcohol is not the highest-priority functional group, then it is named as a hydroxyl substituent (hydroxy–). Figure 1.6 demonstrates a few alcohols and their IUPAC names.
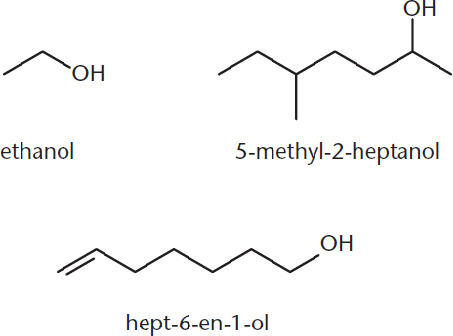
Figure 1.6. Naming Alcohols Alcohols are more oxidized than multiple bonds, so they take priority in nomenclature and are indicated with the suffix –ol.
Alcohols are often referred to by their common names, rather than their IUPAC names. In this version of naming, the name of the alkyl group is followed by the word alcohol. Examples include ethyl alcohol (rather than ethanol) and isopropyl alcohol (rather than 2-propanol.)
MCAT EXPERTISE
The MCAT may use the common names for some alcohols, as well as some of the molecules we will see later. For alcohols, these include ethyl alcohol and isopropyl alcohol—know what these common names refer to!
Alcohols with two hydroxyl groups are called diols or glycols and are indicated with the suffix –diol. The entire hydrocarbon name is preserved, and –diol is added. When naming diols, one must number each hydroxyl group. For example, ethane-1,2-diol is an ethane molecule that has a hydroxyl group on each carbon. This molecule is also known by its common name, ethylene glycol. Diols with hydroxyl groups on the same carbon are called geminal diols; diols with hydroxyl groups on adjacent carbons are called vicinal diols. Geminal diols, or hydrates, are not commonly seen because they spontaneously dehydrate (lose a water molecule) to produce carbonyl compounds with the functional group C=O.
MNEMONIC
Vicinal diols are in the vicinity of each other, on adjacent carbons. Geminal diols—like the Gemini twins—are paired on the same carbon.
MCAT CONCEPT CHECK 1.2:
Before you move on, assess your understanding of the material with these questions.
-
Fill in the correct names for the alkanes listed below. If more than one compound can be described with a given molecular formula, name the straight-chain alkane to which the formula refers and draw one alternative.
Molecular Formula IUPAC Name (Straight-Chain Alkane) Alternative Structure CH4 C2H6 C3H8 C4H10 C5H12 C6H14 C7H16 C8H18 C9H20 C10H22
-
In a molecule with two double bonds adjacent to each other and an alcohol, which functional group would take precedence in naming?
____________________
-
Is the following compound a geminal diol or a vicinal diol?
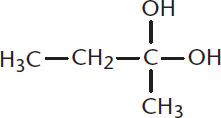
-
What are the common names for 2-propanol and ethanol?
- 2-Propanol:
__________________________________
- Ethanol:
___________________________________
1.3 Aldehydes and Ketones
LEARNING OBJECTIVES
After Chapter 1.3, you will be able to:
- Distinguish aldehydes from ketones
- Recall common names for methanal, ethanal, propanal, and propanone
- Determine the highest priority functional group in a complex molecule
- Apply appropriate prefixes and suffixes when naming molecules containing aldehyde and ketone groups
Aldehydes and ketones are two classes of molecules that contain a carbonyl group, which is a carbon double-bonded to an oxygen. Aldehydes and ketones differ in the placement of the carbonyl group: aldehydes are chain-terminating, meaning that they appear at the end of a parent chain, while ketones are found in the middle of carbon chains. Aldehydes and ketones do not have any leaving groups connected to the carbonyl carbon; they are only connected to alkyl chains or, in the case of aldehydes, hydrogen atoms. As we’ll examine later, carboxylic acids and their derivatives do contain leaving groups connected to the carbonyl carbon.
Aldehydes
Aldehydes have a carbonyl group found at the end of the carbon chain. Because this is a terminal functional group that takes precedence over many others, it is generally attached to carbon number 1. Aldehydes are named by replacing the –e of the parent alkane with the suffix –al. When the aldehyde is at position 1, as is usually the case, we do not need to include this number in the chemical name. Figure 1.7 shows the IUPAC nomenclature for two aldehydes.
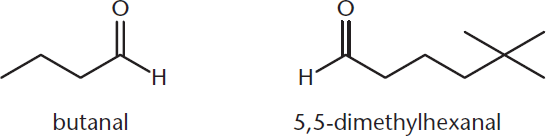
Figure 1.7. Naming Aldehydes The carbonyl group of the aldehyde usually does not receive a number in the name because it is a terminal functional group.
Methanal, ethanal, and propanal are referred to almost exclusively by their common names, formaldehyde, acetaldehyde, and propionaldehyde, rather than their IUPAC names. These molecules are shown in Figure 1.8.
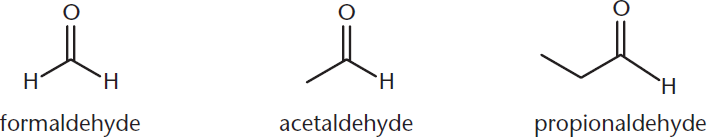
Figure 1.8. Common Names of Aldehydes
MCAT EXPERTISE
As is the case for alcohols, it is important to know both the common names and IUPAC names for common aldehydes and ketones. Make sure that you know what formaldehyde, acetaldehyde, and acetone are!
Ketones
Ketones contain a carbonyl group somewhere in the middle of the carbon chain. Because this is the case, we will always have to assign a number to the carbonyl carbon when naming ketones (except propanone, which must have the ketone on carbon 2 by default). Ketones are named by replacing the –e in the name of the parent alkane with the suffix –one. Just as when naming other compounds, be sure to give the carbonyl the lowest possible number if it is the highest-priority group.
BRIDGE
Sugars are classified as either aldoses (aldehyde sugars) or ketoses (ketone sugars). Understanding nomenclature can help you to identify the structure of a sugar, as well as some of its physical properties. Carbohydrate structure is discussed in Chapter 4 ofMCAT Biochemistry Review.
Ketones are commonly named by listing the alkyl groups in alphabetical order, followed by ketone, such as ethylmethylketone. Acetone is the smallest possible ketone molecule. Figure 1.9 includes IUPAC and common names for a number of ketones.
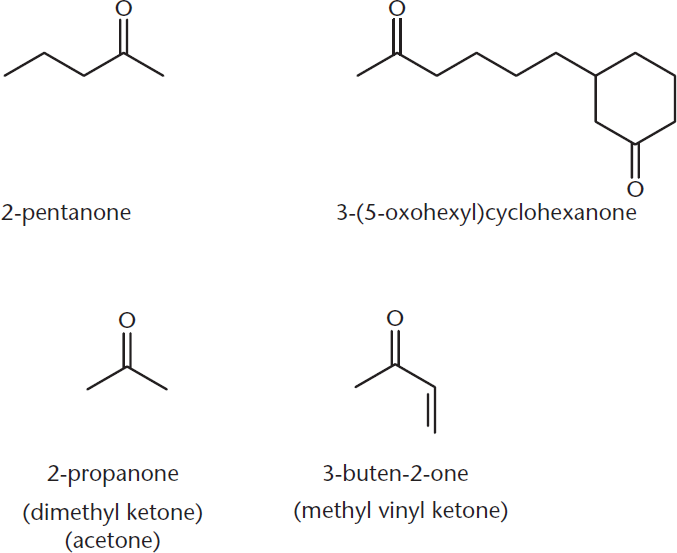
Figure 1.9. Naming Ketones Common names are included in parentheses.
In a more complex molecule with a higher-priority group that takes precedence over the carbonyl, we name aldehydes and ketones as substituents, using the prefix oxo–. This is in reference to the carbonyl oxygen and applies for both ketones and aldehydes. Sometimes ketones may also be indicated with the prefix keto–.
Another convention that you may see on the MCAT is naming carbons relative to the carbonyl group. By this convention, the carbon adjacent to the carbonyl carbon is indicated by alpha (α). Moving away from the carbonyl, the successive carbons are referred to as beta (β), gamma (γ), and delta (δ) carbons. This applies on both sides of the carbonyl in the same fashion, so the carbons on both sides of a ketone are considered alpha carbons. This will become important when we discuss α-hydrogen acidity in Chapter 7 of MCAT Organic Chemistry Review.
MCAT CONCEPT CHECK 1.3:
Before you move on, assess your understanding of the material with these questions.
-
What is the difference between an aldehyde and a ketone?
- Aldehyde:
________________________
- Ketone:
________________________
-
What suffixes are used for aldehydes and ketones; how are carbonyl groups named as a substituent?
- Aldehyde suffix: __________; substituent prefix: __________
- Ketone suffix: __________; substituent prefix: __________
-
Fill in the common names in the following chart.
IUPAC Name Common Name Methanal Ethanal Propanal Propanone
-
For a molecule with a double bond, an aldehyde, and an alcohol, which functional group would determine the suffix when naming?
_____________________________________
1.4 Carboxylic Acids and Derivatives
LEARNING OBJECTIVES
After Chapter 1.4, you will be able to:
- Name common carboxylic acid derivatives, including esters, amides, and anhydrides
- Differentiate between common carboxylic acid derivatives
Carboxylic Acids
Carboxylic acids contain both a carbonyl group (C=O) and a hydroxyl group (–OH) on a terminal carbon. Carboxylic acids, like aldehydes, are terminal functional groups; therefore, their associated carbon is usually numbered 1. This is the most oxidized functional group that appears on the MCAT, with three bonds to oxygen; only carbon dioxide, with four bonds to oxygen, contains a more oxidized carbon. Carboxylic acids are thus the highest-priority functional group in MCAT-tested nomenclature, and all other groups are named as substituents using prefixes. Carboxylic acids are named by replacing the –e at the end of the name of the parent alkane with the suffix –oic acid.
MCAT EXPERTISE
Did you notice that some of the common names are similar between aldehydes and carboxylic acids? Remembering that formaldehyde and formic acid both refer to molecules with methane as a parent alkane—and that acetaldehyde and acetic acid contain ethane as a parent alkane—will help consolidate this information.
Once again, the common names for carboxylic acids show up fairly often on the MCAT. Formic acid is the common name for methanoic acid; acetic acid is ethanoic acid; and propionic acid is propanoic acid. These compounds are shown in Figure 1.10. Be sure that you know both these common names and IUPAC names for Test Day.
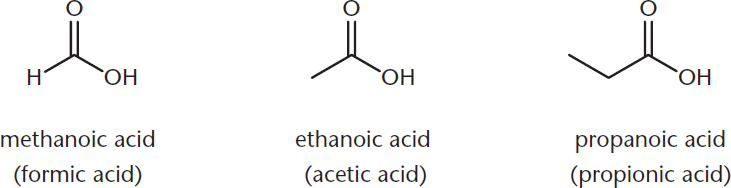
Figure 1.10. Naming Carboxylic Acids The carboxylic acid group does not receive a number in the name because it is a terminal functional group; common names are included in parentheses.
The carboxylic acid derivatives are the final category of functional groups. These include esters, amides, and anhydrides.
Esters
Esters are common carboxylic acid derivatives. In these compounds, the hydroxyl group (–OH) is replaced with an alkoxy group (–OR, where R is a hydrocarbon chain). Ester nomenclature is based on the naming conventions for carboxylic acids. The first term is the alkyl name of the esterifying group. Think of this first term as an adjective describing the ester, based on the identity of the alkyl (R) group. The second term is the name of the parent acid, with –oate replacing the –oic acid suffix. For example, methanoic acid (formic acid) would form butyl methanoate with exposure to butanol under appropriate reaction conditions. Examples of ester nomenclature are shown in Figure 1.11.
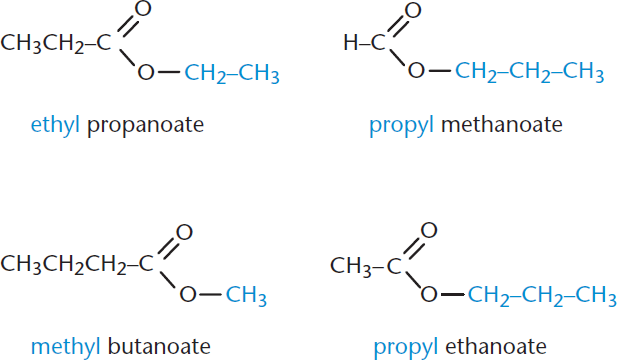
Figure 1.11. Naming Esters Groups bonded directly to the ester oxygen are named as substituents and are not numbered.
Amides
Another group of carboxylic acid derivatives includes amides. In an amide, the hydroxyl group is replaced by an amino group (nitrogen-containing group). These compounds can be more complex—the amino nitrogen can be bonded to zero, one, or two alkyl groups. Amides are named similarly to esters, except that the suffix becomes –amide. Substituents attached to the nitrogen atom are labeled with a capital N–, indicating that this group is bonded to the parent molecule via a nitrogen atom. These substituents are included as prefixes in the compound name and are not numbered. Several examples of amide nomenclature are included in Figure 1.12.
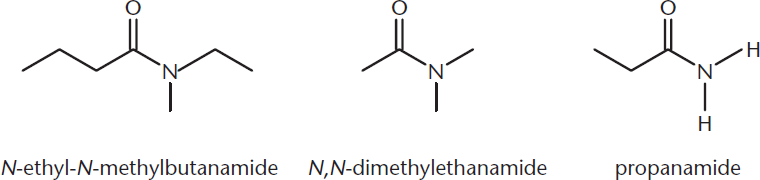
Figure 1.12. Naming Amides Groups bonded directly to the amide nitrogen are named as substituents with the prefix N–, and are not numbered.
Anhydrides
One final group of carboxylic acid derivatives is the anhydrides. In the formation of an anhydride from two carboxylic acid molecules, one water molecule is removed. Many anhydrides are cyclic, which may result from the intramolecular dehydration of a dicarboxylic acid. Anhydrides are named by replacing acid with anhydride in the name of the corresponding carboxylic acid if the anhydride is formed from only one type of carboxylic acid. If the anhydride is not symmetrical, both carboxylic acids are named (without the suffix acid) before anhydride is added to the name. Some examples of anhydrides are shown in Figure 1.13.
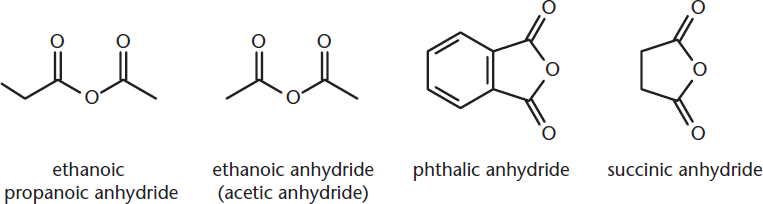
Figure 1.13. Naming Anhydrides Phthalic anhydride and succinic anhydride are given as examples of cyclic anhydrides; their names need not be memorized.
MNEMONIC
Because hydro– is a prefix meaning water,and an– is a prefix meaning not or without, we can remember that anhydrides have had water molecules removed during formation.
MCAT CONCEPT CHECK 1.4:
Before you move on, assess your understanding of the material with these questions.
-
What would be the names of the ester, amide, and anhydride derivatives of pentanoic acid? Assume that the R group on the ester is –CH3 and that the amide is unsubstituted.
- Ester:
________________________
- Amide:
________________________
- Anhydride:
________________________
-
Name the following compound:
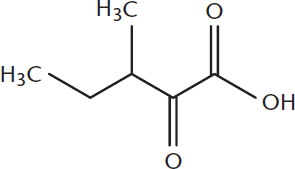
____________________________________
1.5 Summary of Functional Groups
LEARNING OBJECTIVES
After Chapter 1.5, you will be able to:
- Apply appropriate prefixes and suffixes for common organic functional groups
MCAT EXPERTISE
The "High-Yield" badge on this section indicates that the content is frequently tested on the MCAT.
Table 1.2 lists the functional groups that you will need to know for the MCAT in order of priority, with prefixes and suffixes. Carboxylic acids are the highest-priority functional group on the MCAT. In nomenclature, use the suffix if the functional group is the highest-priority group in the molecule; otherwise, name the group as a substituent using its prefix.
Table 1.2. Major Functional Groups
FUNCTIONAL GROUP PREFIX SUFFIX
Carboxylic acid carboxy– –oic acid
Anhydride alkanoyloxycarbonyl– anhydride
Ester alkoxycarbonyl– –oate
Amide carbamoyl– or amido– –amide
Aldehyde oxo– –al
Ketone oxo– or keto– –one
Alcohol hydroxy– –ol
Alkene* alkenyl– –ene
Alkyne* alkynyl– –yne
Alkane alkyl– –ane
*Note: Alkenes and alkynes are considered to be tied for priority except in cyclic compounds, where alkenes have higher priority.
KEY CONCEPT
Functional group priority is correlated with oxidation state. Carboxylic acids have the highest priority while alkanes have the lowest.
Conclusion
Now that we’ve worked through nomenclature, we should be able to navigate MCAT organic chemistry questions with ease and confidence. Remember that even if an MCAT question is asking about a reaction or laboratory technique, translating the name of a compound in the question stem may be a necessary step to get to the answer. We have also covered the important functional groups that will show up and have taken note of the order of priority for these groups when it comes to naming compounds. Remember, the common names can be just as important on the MCAT as the IUPAC names—so knowing both is key. Now that we know the language of organic chemistry, we will learn more about the properties of molecules in the next two chapters, and then will focus on reactions of functional groups and laboratory techniques in subsequent chapters of MCAT Organic Chemistry Review.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
IUPAC Naming Conventions
- The International Union of Pure and Applied Chemistry (IUPAC) has designated standards for naming chemical compounds. There are five steps in the process:
- First, find the longest carbon chain in the compound that contains the highest-priority functional group. This is called the parent chain.
- Second, number the chain in such a way that the highest-priority functional group receives the lowest possible number. This group will determine the suffix of the molecule.
- Third, name the substituents with a prefix. Multiple substituents of a single type receive another prefix denoting how many are present (di–, tri–, tetra–, and so on).
- Fourth, assign a number to each of the substituents depending on the carbon to which it is bonded.
- Finally, complete the name by alphabetizing the substituents and separating numbers from each other by commas and from words by hyphens.
Hydrocarbons and Alcohols
- Alkanes are hydrocarbons without any double or triple bonds. They have the general formula CnH(2n+ 2).
- Alkanes are named according to the number of carbons present followed by the suffix –ane.
- The first four alkanes are methane (CH4), ethane (C2H6), propane (C3H8), and butane (C4H10).
- Larger alkanes use the Greek root for the number (pentane, hexane, heptane, octane, and so on).
- Alkenes and alkynes contain double and triple bonds, respectively.
- Alkenes are named by substituting –ene for the suffix and numbering the double bond by its lower-numbered carbon. Alkynes substitute –yne with the same numbering.
- Alcohols contain a hydroxyl (–OH) group, which substitutes for one or more of the hydrogens in the hydrocarbon chain.
- Alcohols are named by substituting the suffix –ol or by using the prefix hydroxy– if a higher-priority group is present.
- Alcohols have higher priority than double or triple bonds and alkanes.
- Common names of alcohols include the name of the carbon chain followed by the word alcohol. For example, ethyl alcohol is the same compound as ethanol.
- Diols contain two hydroxyl groups. They are termed geminal if on the same carbon or vicinal if on adjacent carbons.
Aldehydes and Ketones
- Aldehydes and ketones contain a carbonyl group—a carbon double-bonded to an oxygen.
- Aldehydes have the carbonyl group on a terminal carbon that is also attached to a hydrogen atom.
- Aldehydes are named with the suffix –al, or by using the prefix oxo– if a higher-priority group is present.
- Common names of aldehydes include formaldehyde for methanal, acetaldehyde for ethanal, and propionaldehyde for propanal.
- Ketones have the carbonyl group on a nonterminal carbon.
- Ketones are named with the suffix –one and share the prefix oxo– if a higher-priority group is present. Ketones can also be indicated by the prefix keto–.
- The common names of ketones are constructed by naming the alkyl groups on either side alphabetically and adding ketone. For example, 2-butanone is called ethylmethylketone.
- Acetone is significant as the smallest ketone. Its IUPAC name is propanone.
- Carbonyl-containing compounds (aldehydes, ketones, carboxylic acids, and derivatives) also create a lettering scheme for carbons. The carbon adjacent to the carbonyl carbon is the ***α*-carbon**.
Carboxylic Acids and Derivatives
- Carboxylic acids are the highest-priority functional group because they contain three bonds to oxygen: one from a hydroxyl group and two from a carbonyl group.
- Carboxylic acids are always terminal, although their derivatives may occur within a molecule.
- Carboxylic acids are named with the suffix –oic acid. They are very rarely named as a prefix.
- Common names for carboxylic acids follow the trend for aldehydes. Formic acid is methanoic acid, acetic acid is ethanoic acid, and propionic acid is propanoic acid.
- Esters are carboxylic acid derivatives where –OH is replaced with –OR, an alkoxy group.
- Esters use the suffix –oate or the prefix alkoxycarbonyl–.
- Common names for esters are derived from the alcohol and the carboxylic acid used during synthesis.
- Amides replace the hydroxyl group of a carboxylic acid with an amino group that may or may not be substituted.
- Amides use the suffix –amide or the prefix carbamoyl– or amido–. Substituents attached to the amide nitrogen are designated with a capital N–.
- Anhydrides are formed from two carboxylic acids by dehydration. They may be symmetric (two of the same acid), asymmetric (two different acids), or cyclic (intramolecular reaction of a dicarboxylic acid).
- Anhydrides are named using the suffix anhydride in place of acid. If the anhydride is formed from more than one carboxylic acid, both are named in alphabetical order in the name before the word anhydride.
Summary of Functional Groups
- Functional groups are arranged in order of priority as follows: Carboxylic acid > anhydride > ester > amide > aldehyde > ketone > alcohol > alkene or alkyne > alkane.
ANSWERS TO CONCEPT CHECKS
1.1
- 1. Find the longest carbon chain in the compound with the highest-order functional group; 2. Number the chain; 3. Name the substituents; 4. Assign a number to each substituent; 5. Complete the name
-
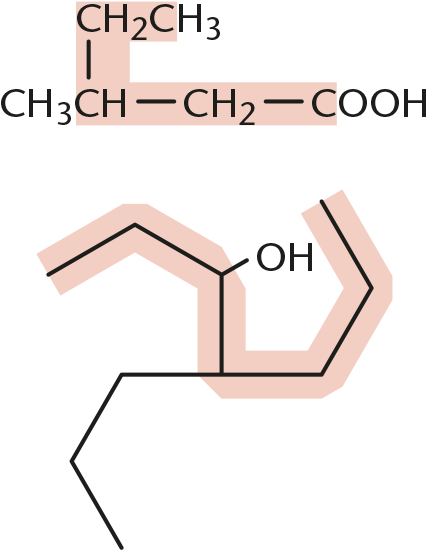
Note: There are two possible answers; however, the longest chain must include the hydroxyl group.

-
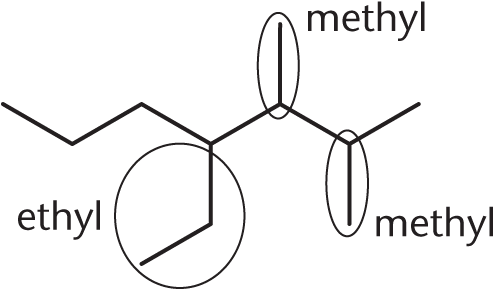
The question asks us to circle and name the substituents, but in order to determine the substituents it is necessary to identify the parent chain. So, begin by identifying the longest carbon chain that contains the highest-priority functional group. The molecule contains only alkyl functional groups, and the alkyl functional groups have the same priority, so the parent chain will be the longest continuous carbon chain (seven carbons). Once the parent chain is identified, the substituents can be circled and named as shown above.
Numbering the chain from right to left ensures that the substituents have the smallest possible locants (numbers). The substituents have already been named and numbered, so to complete the name, alphabetize the substituents (remembering that *d**i*– is ignored): 4-ethyl-2,3-dimethylheptane.
**1.2**
-
Molecular Formula IUPAC Name (Straight-Chain Alkane) Alternative Structure CH4 Methane No alternative structures
C2H6 Ethane No alternative structures
C3H8 Propane No alternative structures
C4H10 Butane See below
C5H12 Pentane See below
C6H14 Hexane See below
C7H16 Heptane See below
C8H18 Octane See below
C9H20 Nonane See below
C10H22 Decane See below
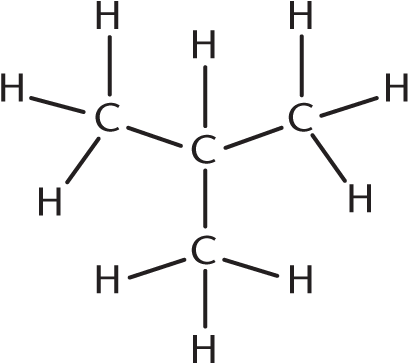
Butane and all hydrocarbons that are larger than butane may have a branched appearance, which shortens the parent chain. An example is isobutane, properly named methylpropane, shown here. Any branched hydrocarbon with the correct number of carbons and no multiple bonds or rings is correct.
- The alcohol would take precedence because the carbon to which it is attached has a higher oxidation state.
- Diols are alcohols with two hydroxyl groups. In a geminal diol, these hydroxyl groups are on the same carbon (gemini derives from the Latin for "paired, twins"). In a vicinal diol, the hydroxyls are in the vicinity of each other—on adjacent carbons (vicinus derives from the Latin for “neighbor”). Thus, the compound shown is a geminal diol.
- Isopropyl alcohol and ethyl alcohol, respectively.
**1.3**
- An aldehyde has a carbonyl group at the end of the chain. A ketone has a carbonyl group somewhere in the middle of the carbon chain. Another way to think of this is that the carbonyl carbon of an aldehyde has at least one bond to a hydrogen atom, whereas the carbonyl carbon of a ketone is always bonded to two other carbons.
- Aldehydes are referred to with the suffix –al, while ketones are given the suffix –one. Carbonyl groups of both aldehydes and ketones are labeled as oxo– substituents (ketones may also be called keto– substituents).
-
IUPAC Name Common Name Methanal Formaldehyde
Ethanal Acetaldehyde
Propanal Propionaldehyde
Propanone Acetone
- Ketones and aldehydes both take precedence over both alcohols and hydrocarbon chains, and the functional group that is the highest priority determines the suffix. Because the aldehyde is chain-terminating and therefore on carbon number 1, the aldehyde would determine the suffix when naming this compound.
**1.4**
- The ester derivative would be methyl pentanoate. The amide would be pentanamide. The anhydride would be pentanoic anhydride.
- 3-methyl-2-oxopentanoic acid
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. A
The common name of ethanal is acetaldehyde, the common name of methanal is formaldehyde, and the common name of ethanol is ethyl alcohol. Isopropyl alcohol is the common name of 2-propanol. Propionaldehyde is the common name of propanal.
2. C
Aldehydes and carboxylic acids are characterized by their positions at the ends of carbon backbones and are thus considered terminal groups. As a result, the carbons to which they are attached are usually designated carbon 1. Ketones are internal by definition because there must be a carbon on either side of the carbonyl.
3. C
The highest-priority functional group in this molecule is the carboxyl group, so this will be a component of the backbone and provides the suffix of the molecule. This molecule is 2-methyl-3-oxopropanoic acid.
4. A
This molecule features an anhydride. The only other groups are hydrocarbon chains, which will provide part of the name of the parent root. Keep in mind that when a carbonyl group is present with a leaving group, the larger functional group (carboxylic acid, anhydride, ester, amide) takes priority over the carbonyl group alone. This molecule is propanoic anhydride.
5. C
Among the functional groups presented, carboxylic acids have the highest priority, and their compounds end with an –oic acid suffix. (A) denotes an alcohol, (B) a ketone, and(D) an alkyne, all of which have lower priorities than carboxylic acids. The MCAT does not test nomenclature of halides or ethers, but note that these must have lower priority than a carboxylic acid because they are less oxidized groups.
6. A
This molecule is 3-ethyl-4-methylhexane, not 2,3-diethylpentane. When naming alkanes, one must locate the longest carbon chain (6 carbons rather than 5 carbons), and the numbering system should give the alkyl groups the lowest possible numbers.
7. B
We know from the IUPAC name that we have a butanedioic acid backbone—in other words, a four-carbon backbone with carboxylic acids at either end. Adding the hydroxyl groups at carbons 2 and 3 then yields the correct structure.
8. B
Form– is a prefix shared by the common names of methanoic acid (formic acid) and methanal (formaldehyde).
9. A
The first task in naming a compound is identifying the longest carbon chain. In this case, the longest chain has seven carbons, so the parent alkane ends in –heptane. (B) and (D) can therefore be eliminated. Then, we must make sure that the carbons are numbered so that the substituents’ position numbers are as small as possible. This compound has two methyl groups; minimizing their position numbers requires us to number the chain from right to left. These methyl groups are attached to carbons 2 and 5, so the correct IUPAC name is 2,5-dimethylheptane. (C) is incorrect because the position numbers of the substituents are not minimized.
10. D
We begin by finding the longest carbon chain; because there are no non-alkyl groups, we don’t need to worry about any other groups’ priorities. We then number our carbons such that the lowest possible combination of numbers is given to the various substituents. Then substituents are organized alphabetically, not numerically—eliminating (C).
11. B
We know right away that (C) and (D) will be incorrect because a hydroxyl group is of higher priority than a methyl group. We also know that we will number the carbon chain so that the hydroxyl group receives the lowest possible position. Therefore, this molecule is 2-methyl-2-butanol, which starts with the prefix 2-methyl.
12. C
The suffix –amide in nicotinamide indicates that this compound contains an amide functional group. The prefix diphospho– indicates that there are two phosphate groups as well. Even if we did not know the prefix phospho– from this chapter, we should recognize that nucleotides, mentioned in the name of the compound, contain a sugar, a phosphate group, and a nitrogenous base. The structure of NAD+, the oxidized form of NADH, is shown below.
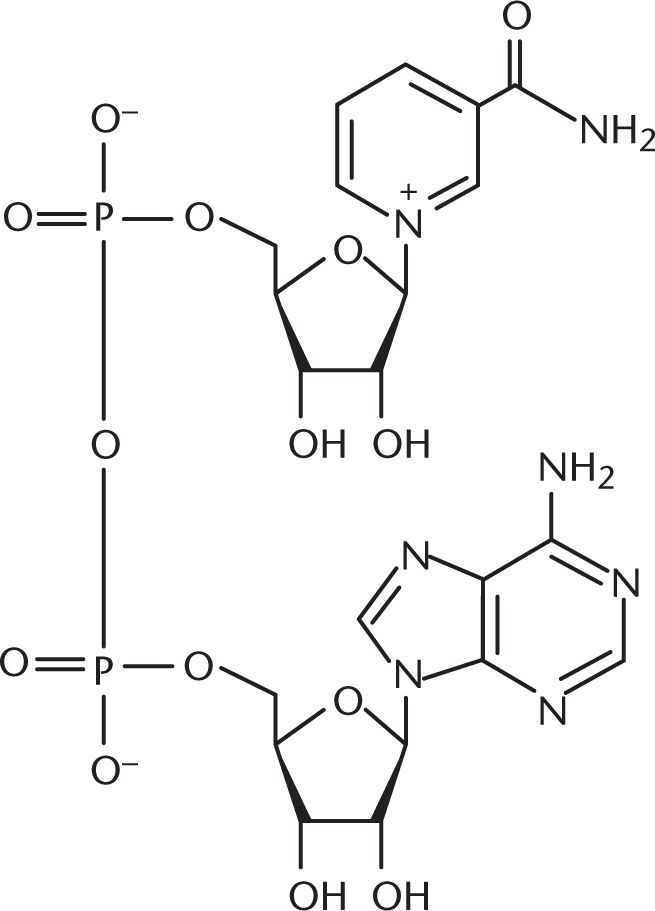
13. A
We can use the name acetylformic acid to figure out what our functional groups are. The prefix acet– refers to a two-carbon unit with one carbon in a carbonyl group—think of acetic acid, acetic anhydride, or acetaldehyde. The carbonyl carbon is the point of attachment to another functional group. Formic acid is a single-carbon carboxylic acid. Therefore, acetylformic acid is an acetyl group directly attached to formic acid, as shown in (A). (B) shows acetic acid, or vinegar; (C) shows glucose; and (D) shows formic acid.
14. B
Acetic anhydride is the common name for ethanoic anhydride. Methyl formate is the common name for methyl methanoate; we can infer this from the common root form– and the ester suffix –oate (which is sometimes shortened to –ate for pronunciation purposes). Formic acid is the common name for methanoic acid, but this is a carboxylic acid—not a derivative.
15. C
Draw out the molecule, and you will see that the longest carbon chain with the substituents at the lowest possible carbon numbers is actually different from the one chosen in the original name. The correct IUPAC name for this molecule is 3-ethyl-4-methylhexane.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Organic Chemistry Chapter 4
- Analyzing Organic Reactions
- Organic Chemistry Chapter 5
- Alcohols
- Organic Chemistry Chapter 6
- Aldehydes and Ketones I
- Organic Chemistry Chapter 8
- Carboxylic Acids
- Organic Chemistry Chapter 9
- Carboxylic Acid Derivatives
- Organic Chemistry Chapter 10
- Nitrogen- and Phosphorus-Containing Compounds