Chapter 2: Isomers

Chapter 2: Isomers
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of the following does NOT show optical activity?
- (R)-2-butanol
- (S)-2-butanol
- A solution containing 1 M (R)-2-butanol and 2 M (S)-2-butanol
- A solution containing 2 M (R)-2-butanol and 2 M (S)-2-butanol
-
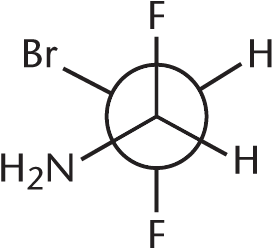
How many stereoisomers exist for the following aldehyde?
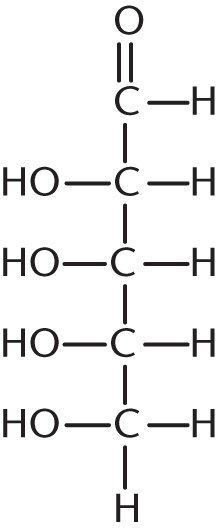
- 2
- 8
- 9
- 16
-
Which of the following compounds is optically inactive?
-
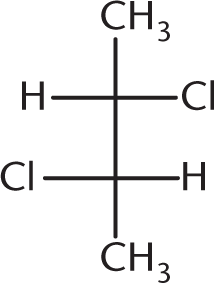
-
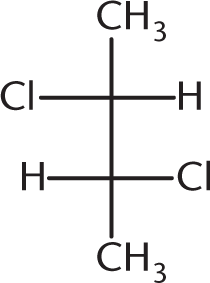
-
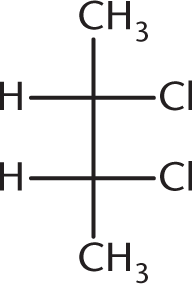
-
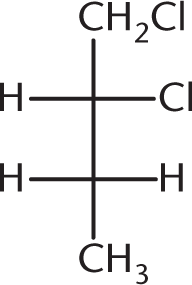
-
Cholesterol, shown below, contains how many chiral centers?
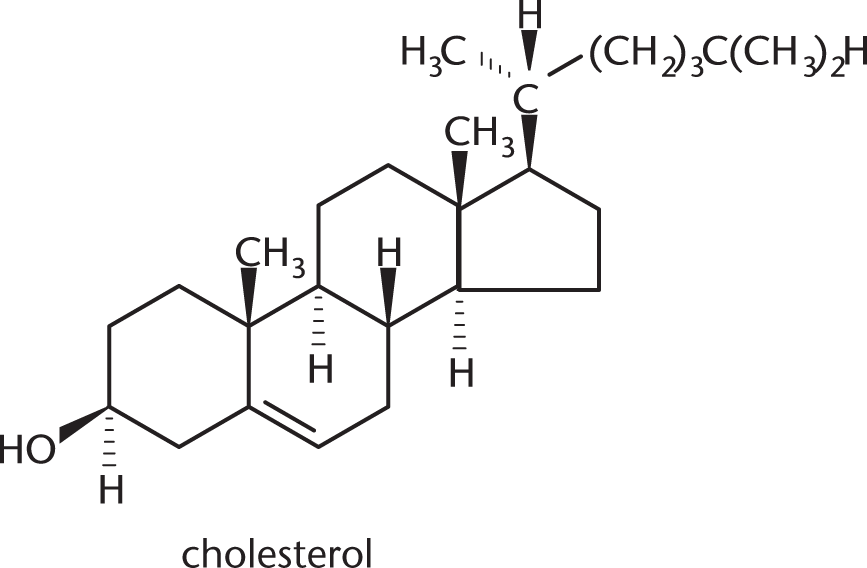
- 5
- 7
- 8
- 9
-
Which isomer of the following compound is the most stable?
-
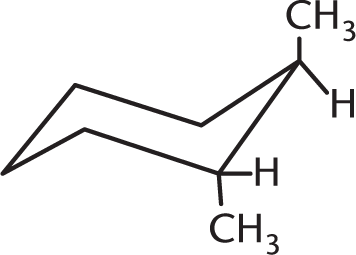
-
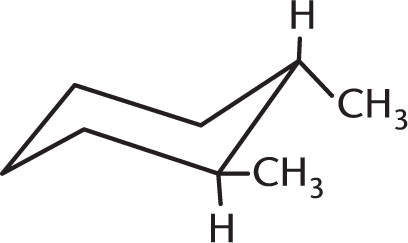
-
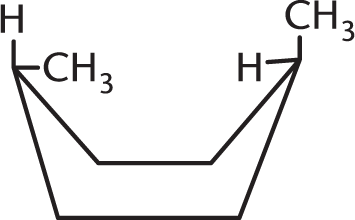
- They are all equally stable.
-
The following reaction results in:
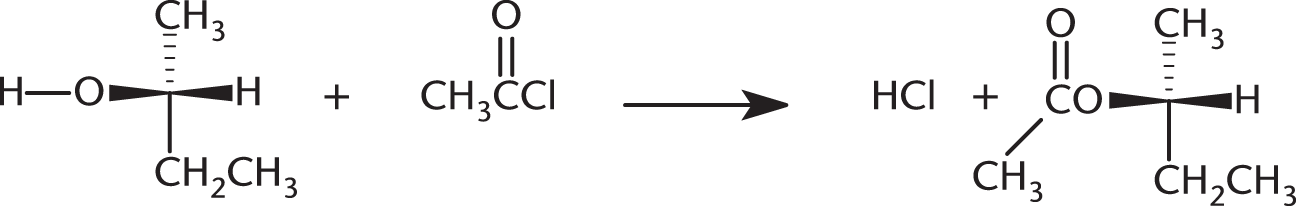
- retention of relative configuration and a change in the absolute configuration.
- a change in the relative and absolute configurations.
- retention of the relative and absolute configurations.
- retention of the absolute configuration and a change in the relative configuration.
-
The following molecules are considered to be:

- enantiomers.
- diastereomers.
- meso compounds.
- structural isomers.
-
(+)-Glyceraldehyde and (–)-glyceraldehyde refer to the (R) and (S) forms of 2,3-dihydroxypropanal, respectively. These molecules are considered:
- enantiomers.
- diastereomers.
- meso compounds.
- structural isomers.
-
Consider (E)-2-butene and (Z)-2-butene. This is a pair of what type(s) of isomers?
- Cis–trans isomers
- Diastereomers
- Enantiomers
- I only
- II only
- I and II only
- I and III only
-
3-methylpentane and hexane are related in that they are:
- enantiomers.
- diastereomers.
- constitutional isomers.
- conformational isomers.
-
(R)-2-chloro-(S)-3-bromobutane and (S)-2-chloro-(S)-3-bromobutane are:
- enantiomers.
- diastereomers.
- meso compounds.
- the same molecule.
-
A scientist takes a
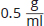
solution of an unknown pure dextrorotatory organic molecule and places it in a test tube with a diameter of 1 cm. He observes that a plane of polarized light is rotated 12° under these conditions. What is the specific rotation of this molecule?
- –240°
- –24°
- +24°
- +240°
-
Omeprazole is a proton pump inhibitor commonly used in gastroesophageal reflux disease. When omeprazole, a racemic mixture, went off-patent, pharmaceutical companies began to manufacture esomeprazole, the (S)-enantiomer of omeprazole, by itself. Given 1 M solutions of omeprazole and esomeprazole, which solution(s) would likely exhibit optical activity?
- Omeprazole only
- Esomeprazole only
- Both omeprazole and esomeprazole
- Neither omeprazole nor esomeprazole
-
(2R,3S)-2,3-dihydroxybutanedioic acid and (2S,3R)-2,3-dihydroxybutanedioic acid are:
- meso compounds.
- the same molecule.
- enantiomers.
- I only
- III only
- I and II only
- I and III only
-
If the methyl groups of butane are 120° apart, as seen in a Newman projection, this molecule is in its:
- highest-energy gauche form.
- lowest-energy staggered form.
- middle-energy eclipsed form.
- highest-energy eclipsed form.
Answer Key
- D
- B
- C
- C
- B
- C
- A
- A
- C
- C
- B
- D
- B
- C
- C
Chapter 2: Isomers
CHAPTER 2
ISOMERS
In This Chapter
2.1 Structural Isomers 2.2 Stereoisomers
Conformational Isomers
Configurational Isomers
2.3 Relative and Absolute Configurations
(E) and (Z) Forms
(R) and (S) Forms
Fischer Projections
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 12% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1D: Principles of bioenergetics and fuel molecule metabolism
5B: Nature of molecules and intermolecular interactions
5C: Separation and purification methods
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
An important way that we distinguish between molecules is by identifying isomers of the same compound—those that have the same molecular formula but different structures. Keep in mind that isomerism describes a relationship; just as there must be at least two children to have siblings, two molecules can be isomers to each other, but no molecule can be an isomer by itself. Throughout this chapter, we will learn how to identify these relationships and describe the similarities and differences between isomers. Figure 2.1 shows the isomer classes that we will learn more about and how they are related—feel free to come back to this figure as a reference after you have read through the more detailed explanations.
KEY CONCEPT
Isomers have the same molecular formula, but different structures.
2.1 Structural Isomers
LEARNING OBJECTIVES
After Chapter 2.1, you will be able to:
- Describe the shared and unique properties of structural isomers
- Explain what physical and chemical properties are
- Identify structural isomers
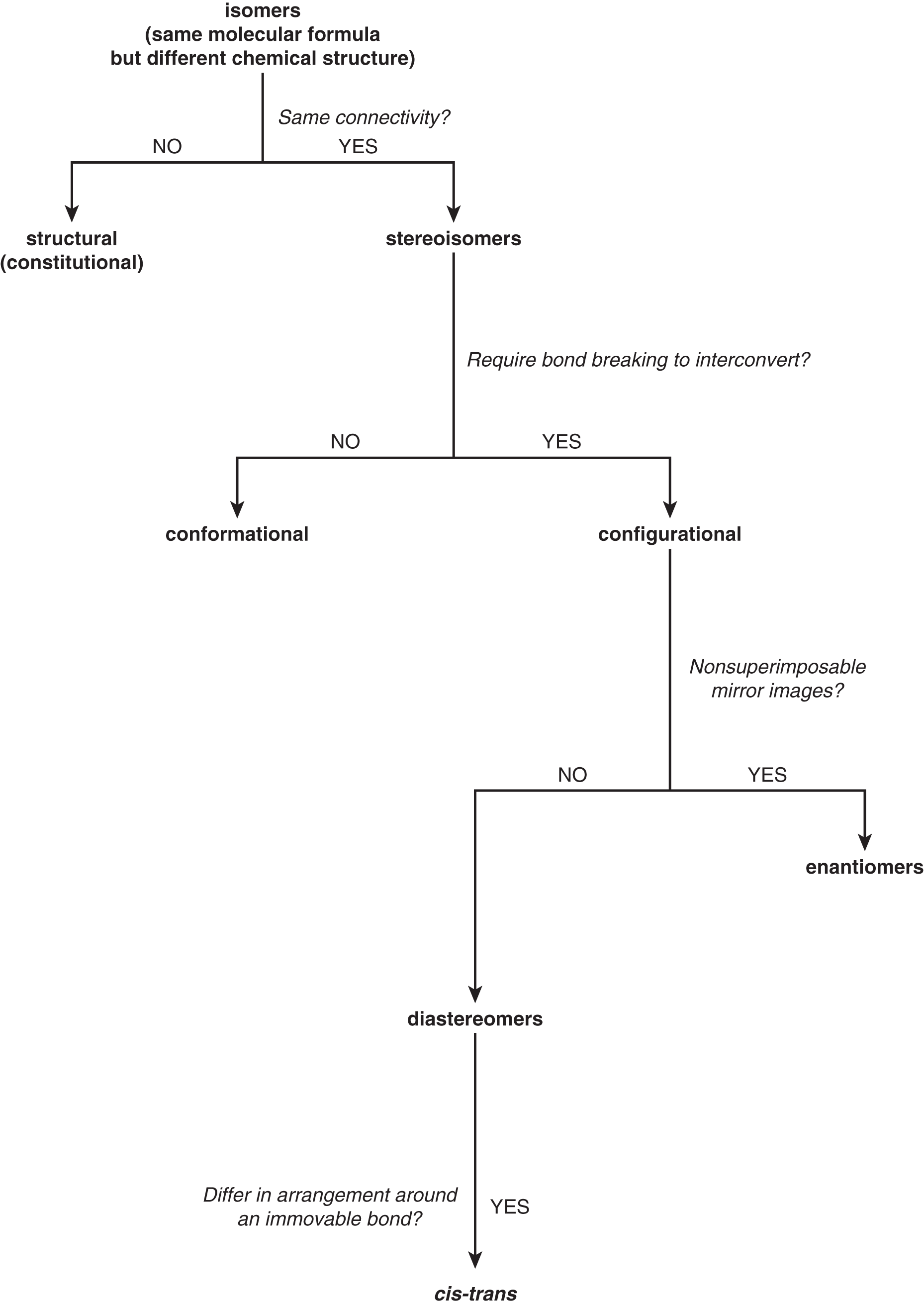
Figure 2.1. Flowchart of Isomer Relationships
Structural isomers are the least similar of all isomers. In fact, the only thing that structural isomers (also called constitutional isomers) share is their molecular formula, meaning that their molecular weights must be the same. Aside from this similarity, structural isomers are widely varied, with different chemical and physical properties. For example, five different structural isomers of C6H14 are shown in Figure 2.2. Each of these molecules looks completely different but has the same number of carbon and hydrogen atoms.
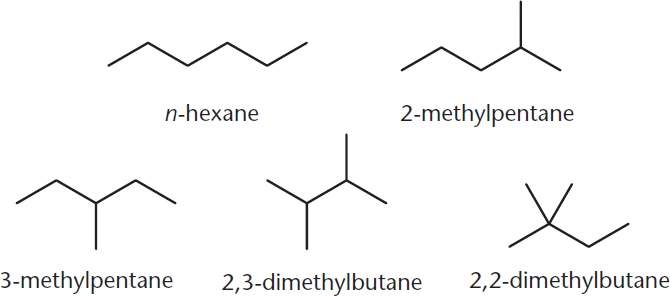
Figure 2.2. Structural (Constitutional) Isomers of C6H14
Physical and chemical properties are prime MCAT material and are often tested in the context of isomerism. Physical properties are characteristics of processes that don’t change the composition of matter, such as melting point, boiling point, solubility, odor, color, and density. Chemical properties have to do with the reactivity of the molecule with other molecules and result in changes in chemical composition. In organic chemistry, the chemical properties of a compound are generally dictated by the functional groups in the molecule.
KEY CONCEPT
Physical properties: no change in composition of matter; examples include melting point, boiling point, solubility, odor, color, density.
Chemical properties: reactivity of molecule, resulting in change in composition; generally attributable to functional groups in the molecule.
MCAT CONCEPT CHECK 2.1:
Before you move on, assess your understanding of the material with these questions.
-
What property or properties do structural isomers have in common?
________________________
-
Of the compounds cyclopropanol, 2-propanol, acetone, and prop-2-ene-1-ol, which are structural isomers of each other?
________________________
-
What are physical properties? Give three examples of physical properties.
-
________________________
-
________________________
-
________________________
- What are chemical properties?
________________________
2.2 Stereoisomers
LEARNING OBJECTIVES
After Chapter 2.2, you will be able to:
- Differentiate between conformational and configurational isomers
- Distinguish enantiomers from diastereomers
- Identify enantiomers, diastereomers, and meso compounds
- Convert between Newman and 3D molecular projections
Like structural isomers—and all isomers, for that matter—stereoisomers have the same chemical formula. Unlike structural isomers, however, stereoisomers also share the same atomic connectivity. In other words, they have the same structural backbone. Stereoisomers differ in how these atoms are arranged in space (their wedge-and-dash pattern), and all isomers that are not structural isomers fall under this category. The largest distinction within this class is between conformational and configurational isomers. Conformational isomers or conformers differ in rotation around single (σ) bonds; configurationalisomers can be interconverted only by breaking bonds.
Conformational Isomers
Of all of the isomers, conformational isomers are the most similar. Conformational isomers are, in fact, the same molecule, only at different points in their natural rotation around single (σ) bonds.
While double bonds hold molecules in a specific position (as explained with cis–trans isomers later), single bonds are free to rotate. Conformational isomers arise from the fact that varying degrees of rotation around single bonds can create different levels of strain. These conformations are easy to see when the molecule is depicted in a Newman projection, in which the molecule is visualized along a line extending through a carbon–carbon bond axis. The classic example for demonstrating conformational isomerism in a straight chain is butane, which is shown in Figure 2.3.
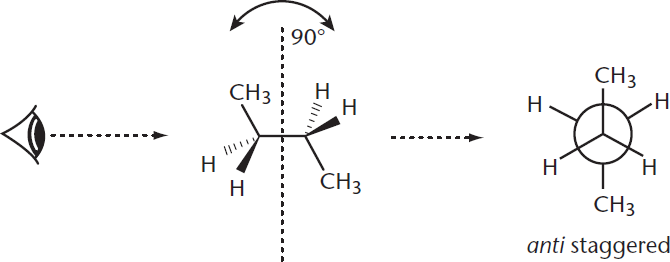
Figure 2.3. Newman Projection of Butane Depiction of different atoms’ positions from the point of view of the C-2 to C-3 bond axis.
Straight-Chain Conformations
For butane, the most stable conformation occurs when the two methyl groups (containing C-1 and C-4) are oriented 180° away from each other. In this position, there is minimal steric repulsion between the atoms’ electron clouds because they are as far apart as they can possibly be. Thus, the atoms are “happiest” and in their lowest-energy state. Because there is no overlap of atoms along the line of sight (besides C-2 and C-3), the molecule is said to be in a staggered conformation. Specifically, it is called the anti conformation because the two largest groups are antiperiplanar (in the same plane, but on opposite sides) to each other. This is the most energetically favorable type of staggered conformation. The other type of staggered conformation, called gauche, occurs when the two largest groups are 60° apart.
MNEMONIC
It’s gauche (unsophisticated or awkward) for one methyl group to stand too close to another group. Groups are eclipsed when they are completely in line with one another—just like a solar or lunar eclipse.
To convert from the anti to the gauche conformation, the molecule must pass through an eclipsed conformation in which the two methyl groups are 120° apart and overlap with the hydrogen atoms on the adjacent carbon. When the two methyl groups directly overlap each other with 0° separation, the molecule is said to be totally eclipsed and is in its highest-energy state. Totally eclipsed conformations are the least favorable energetically because the two largest groups are synperiplanar (in the same plane, on the same side). The different staggered and eclipsed conformations are demonstrated in Figures 2.3 and 2.4. For compounds larger than butane, the name of the conformation is decided by the relative positions of the two largest substituents about a given carbon–carbon bond.
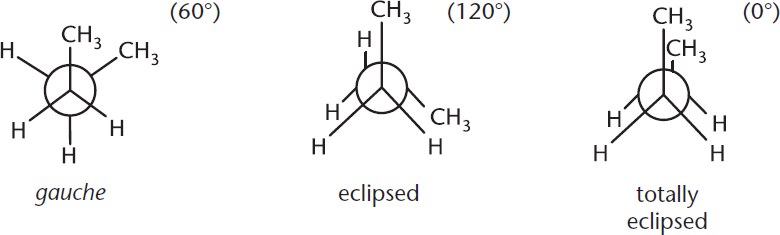
Figure 2.4. Stability of Straight-Chain Conformational Isomers Degree measurements indicate the angle between the two largest substituents about the carbon–carbon bond.
Figure 2.5 shows the plot of potential energy vs. degree of rotation about the bond between C-2 and C-3 in butane. It shows the relative minima and maxima of potential energy of the molecule throughout its various conformations. Remember that every molecule wants to be in the lowest energy state possible, so the higher the energy, the less time the molecule will spend in that energetically unfavorable state.
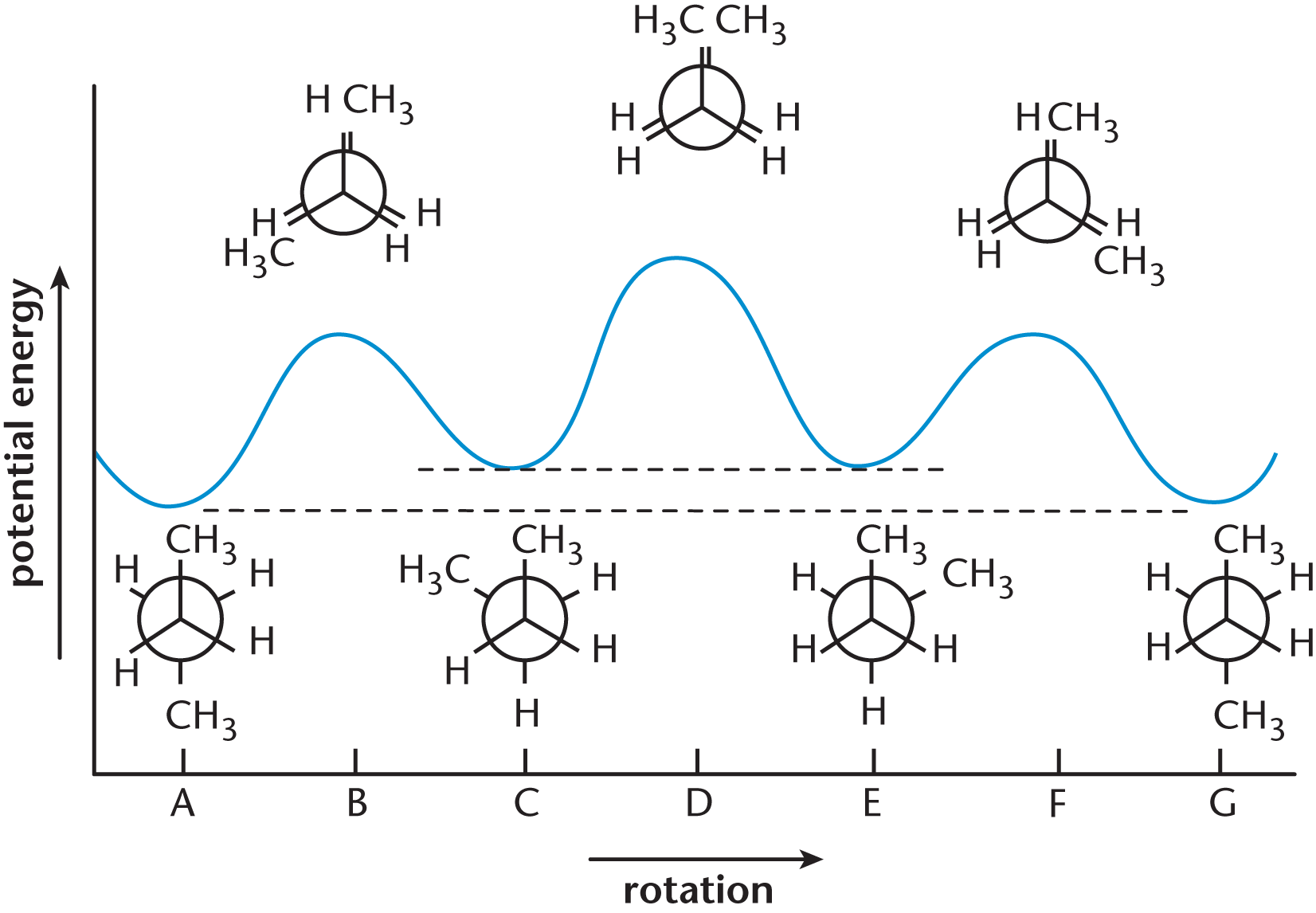
Figure 2.5. Potential Energy vs. Degree of Rotation about the C-2 to C-3 Bond in Butane
KEY CONCEPT
Notice that the anti staggered isomer (A and G) has the lowest energy, whereas the totally eclipsed isomer (D) has the highest energy.
These conformational interconversion barriers are small
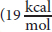
between anti staggered butane and totally eclipsed butane) and are easily overcome at room temperature. Nevertheless, at very low temperatures, conformational interconversions are dramatically slow. If the molecules do not possess sufficient energy to cross the energy barrier, they may not rotate at all (as happens to all molecules at absolute zero).
Cyclic Conformations
Cycloalkanes can be either fairly stable compounds or fairly unstable—depending on ring strain. Ring strain arises from three factors: angle strain, torsional strain, and nonbonded strain (sometimes referred to as steric strain). Angle strain results when bond angles deviate from their ideal values by being stretched or compressed. Torsional strain results when cyclic molecules must assume conformations that have eclipsed or gauche interactions. Nonbonded strain (van der Waals repulsion) results when nonadjacent atoms or groups compete for the same space. Nonbonded strain is the dominant source of steric strain in the flagpole interactions of the cyclohexane boat conformation. To alleviate the strain, cycloalkanes attempt to adopt various nonplanar conformations. Cyclobutane puckers into a slight “V” shape; cyclopentane adopts what is called an envelope conformation; and cyclohexane (the one you will undoubtedly see the most on the MCAT) exists mainly in three conformations called the chair, boat, and **twist-orskew-boat** forms. These cycloalkanes are shown in Figure 2.6.
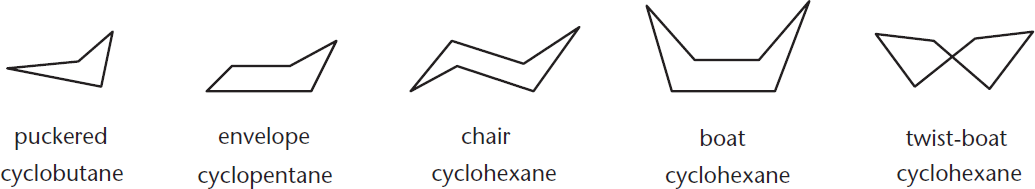
Figure 2.6. Conformations of Cycloalkanes
The most stable conformation of cyclohexane is the chair conformation, which minimizes all three types of strain. The hydrogen atoms that are perpendicular to the plane of the ring (sticking up or down) are called axial, and those parallel (sticking out) are called equatorial. The axial–equatorial orientations alternate around the ring; that is, if the wedge on C-1 is an axial group, the dash on C-2 will also be axial, the wedge on C-3 will be axial, and so on.
Cyclohexane can undergo a chair flip in which one chair form is converted to the other. In this process, the cyclohexane molecule briefly passes through a fourth conformation called the half-chair conformation. After the chair flip, all axial groups become equatorial and all equatorial groups become axial. All dashes remain dashes, and all wedges remain wedges. This interconversion can be slowed if a bulky group is attached to the ring; tert-butyl groups are classic examples of bulky groups on the MCAT. For substituted rings, the bulkiest group will favor the equatorial position to reduce nonbonded strain (flagpole interactions) with axial groups in the molecule, as shown in Figure 2.7.
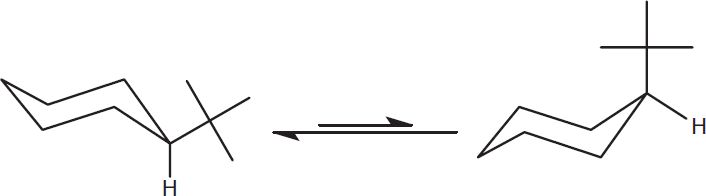
Figure 2.7. Axial and Equatorial Positions in Cyclohexane During a chair flip, axial components become equatorial and vice versa. However, components pointing “up” (wedge) remain up and components pointing “down” (dash) remain down.
In rings with more than one substituent, the preferred chair form is determined by the larger group, which will prefer the equatorial position. These rings also have associated nomenclature. If both groups are located on the same side of the ring, the molecule is called cis; if they are on opposite sides of the ring, it is called trans, as shown in Figure 2.8. These same terms are used for molecules with double bonds, as explained later in this chapter.
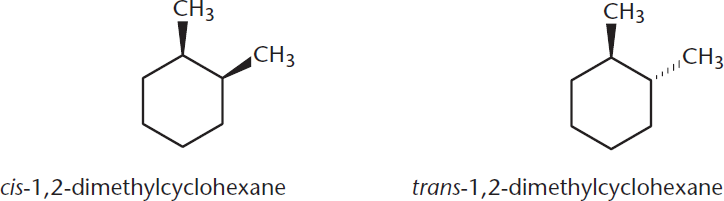
Figure 2.8. Nomenclature of Rings with Multiple Substituents
Configurational Isomers
Unlike conformational isomers that interconvert by simple bond rotation, configurational isomers can only change from one form to another by breaking and reforming covalent bonds. The two categories of configurational isomers are enantiomers and diastereomers. Both enantiomers and diastereomers can also be considered optical isomers because the different spatial arrangement of groups in these molecules affects the rotation of plane-polarized light.
Chirality
An object is considered chiral if its mirror image cannot be superimposed on the original object; this implies that the molecule lacks an internal plane of symmetry. Chirality can also be thought of as handedness. In fact, one of the easiest visualizations of chirality is to think of a pair of hands, as shown in Figure 2.9. Although essentially identical, the left hand will not be able to fit into a right-handed glove. Achiral objects have mirror images that can be superimposed; for example, a fork is identical to its mirror image and is therefore achiral.
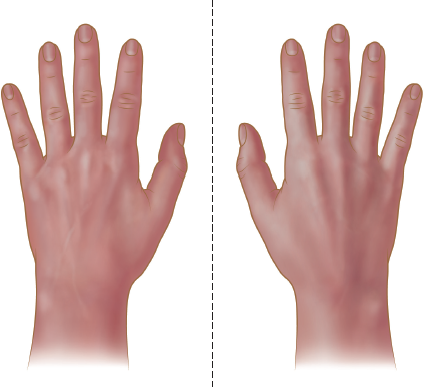
Figure 2.9. Hands as Examples of Chiral Structures Each hand has a nonsuperimposable mirror image.
KEY CONCEPT
Chirality = handedness
On the MCAT, you will often see this concept tested when there is a carbon atom with four different substituents. This carbon will be an asymmetrical core of optical activity and is known as a chiral center. As mentioned earlier, chiral centers lack a plane of symmetry. For example, the C-1 carbon atom in 1-bromo-1-chloroethane has four different substituents. As shown in Figure 2.10, this molecule is chiral because it is not superimposable on its mirror image.
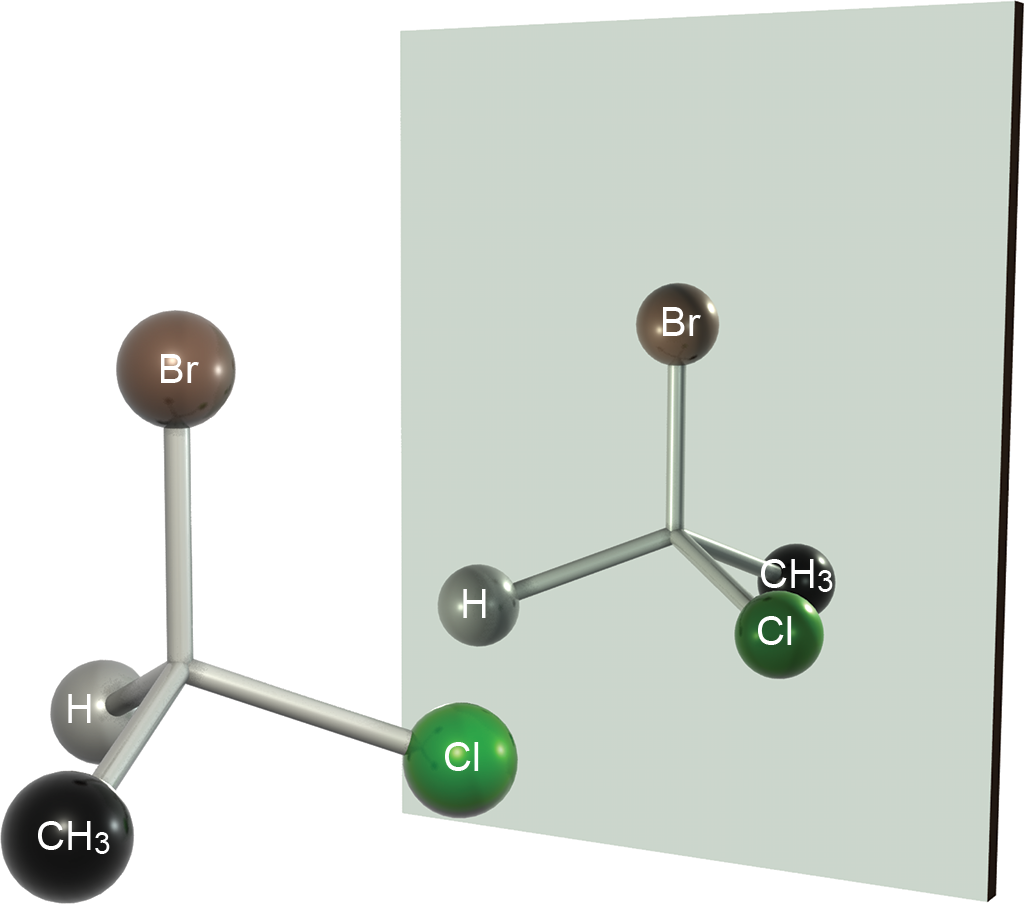
Figure 2.10. Enantiomers of 1-Bromo-1-Chloroethane
MCAT EXPERTISE
Whenever you see a carbon with four different substituents, think chirality.
Two molecules that are nonsuperimposable mirror images of each other are called enantiomers. Molecules may also be related as diastereomers. These molecules are chiral and share the same connectivity but are not mirror images of each other. This is because they differ at some (but not all) of their multiple chiral centers.
Alternatively, a carbon atom with only three different substituents, such as 1,1-dibromoethane, has a plane of symmetry and is therefore achiral. A simple 180° rotation around a vertical axis, as shown in Figure 2.11, allows the compound to be superimposed upon its mirror image.
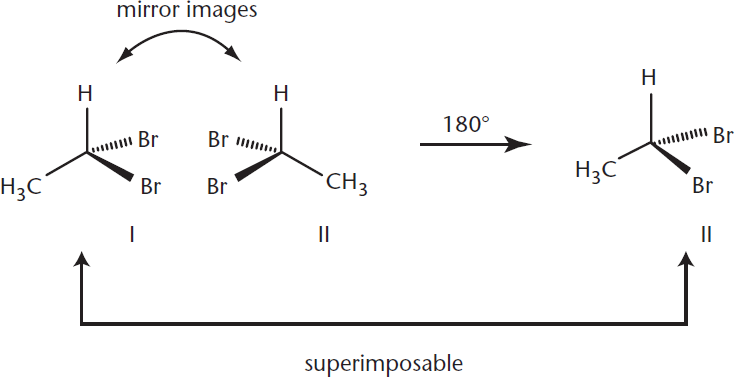
Figure 2.11. Rotation of an Achiral Molecule
Enantiomers
Enantiomers (nonsuperimposable mirror images) have the same connectivity but opposite configurations at every chiral center in the molecule. Enantiomers have identical physical and chemical properties with two notable exceptions: optical activity and reactions in chiral environments.
KEY CONCEPT
Enantiomers have nearly identical physical properties and chemical properties, but they rotate plane-polarized light in opposite directions and react differently in chiral environments.
A compound is optically active if it has the ability to rotate plane-polarized light. Ordinary light is unpolarized, which means that it consists of waves vibrating in all possible planes perpendicular to its direction of propagation. A polarizer allows light waves oscillating only in a particular direction to pass through, producing plane-polarized light, as shown in Figure 2.12.
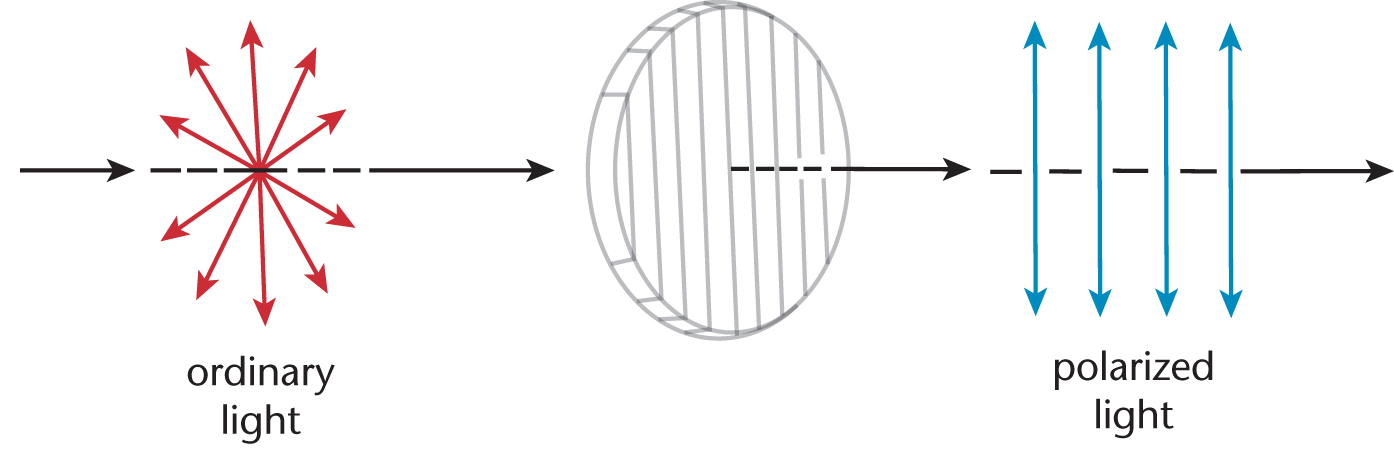
Figure 2.12. Polarizer
BRIDGE
While rotation of plane-polarized light can be tested in organic chemistry questions, the polarization of light itself is fair game as a physics question. Be sure to review light polarization, discussed in Chapter 8 of MCAT Physics and Math Review.
Optical activity refers to the rotation of this plane-polarized light by a chiral molecule. At the molecular level, one enantiomer will rotate plane-polarized light to the same magnitude but in the opposite direction of its mirror image (assuming concentration and path lengths are equal). A compound that rotates the plane of polarized light to the right, or clockwise, is dextrorotatory (d-) and is labeled (+). A compound that rotates light toward the left, or counterclockwise, is levorotatory (l-) and is labeled (–). The direction of rotation cannot be determined from the structure of a molecule and must be determined experimentally. That is, it is not related to the absolute configuration of the molecule.
KEY CONCEPT
The system for labeling optical activity always uses d- or (+) to refer to clockwise rotation of plane-polarized light, while l- and (–) always go together and refer to counterclockwise rotation of plane-polarized light. Do not confuse this with D- or L- labels on carbohydrates or amino acids, which are based on the absolute configuration of glyceraldehyde. (R) and (S) also refer to absolute configuration, which is determined by structure. Optical activity does not consistently align with the other systems.
The amount of rotation depends on the number of molecules that a light wave encounters. This depends on two factors: the concentration of the optically active compound and the length of the tube through which the light passes. Chemists have set standard conditions of
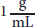
for concentration and 1 dm (10 cm) for length to compare the optical activities of different compounds. Rotations measured at different concentrations and tube lengths can be converted to a standardized specific rotation using the following equation:
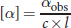
Equation 2.1
where [α] is specific rotation in degrees, αobs is the observed rotation in degrees, c is the concentration in
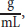
and l is the path length in dm.
When both (+) and (–) enantiomers are present in equal concentrations, they form a racemic mixture. In these solutions, the rotations cancel each other out, and no optical activity is observed. If enantiomerism is analogous to handedness, racemic mixtures are the equivalent of ambidexterity. These solutions possess no handedness overall and will not rotate plane-polarized light.
KEY CONCEPT
A racemic mixture displays no optical activity.
The fact that enantiomers have identical physical and chemical properties prompts a question about racemic mixtures: How can one separate the mixture into its two constituent isomers? The answer lies in the relationship between enantiomers and diastereomers. Reacting two enantiomers with a single enantiomer of another compound will, by definition, lead to two diastereomers. Imagine, for example, two enantiomers that contain only one chiral carbon; these compounds could be labeled (+) and (–). If each is reacted with only the (+) enantiomer of another compound, two products would result: (+,+) and (–,+). Because these two products differ at some—but not all—chiral centers, they are necessarily diastereomers. Diastereomers have different physical properties, as we will explore momentarily. These differences enable one to separate these products by common laboratory techniques such as crystallization, filtration, distillation, and others. Once separated, these diastereomers can be reacted to regenerate the original enantiomers.
Diastereomers
Diastereomers are non-mirror-image configurational isomers. Diastereomers occur when a molecule has two or more stereogenic centers and differs at some, but not all, of these centers. The term diastereomer encompasses any stereoisomer that is not an enantiomer. For any molecule with n chiral centers, there are 2n possible stereoisomers. Thus, if a compound has two chiral carbon atoms, it has a maximum of four possible stereoisomers, as shown in Figure 2.13.
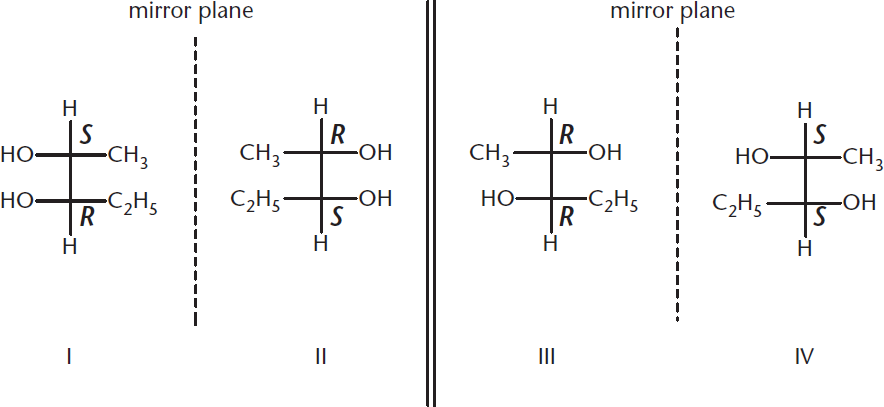
Figure 2.13. 2n Possible Stereoisomers (n = chiral centers) Four stereoisomers with two chiral centers; enantiomers = I/II and III/IV pairs, and all other combinations are diastereomers.
In this image, one can see that I and II are mirror images of each other and are therefore enantiomers of each other. Similarly, III and IV are enantiomers. However, I and III are not. These are stereoisomers that are not mirror images and are thus diastereomers. Notice that other combinations of non-mirror-image stereoisomers are also diastereomers: I and IV, II and III, and II and IV.
Diastereomers have different chemical properties. However, they might behave similarly in particular reactions because they have the same functional groups. Because they have different arrangements in space, they will consistently have different physical properties. Diastereomers will also rotate plane-polarized light; however, knowing the specific rotation of one diastereomer gives no indication of the specific rotation of another diastereomer. This is a stark contrast from enantiomers, which will always have equal-magnitude rotations in opposite directions.
*Cis–Trans* Isomers
Cis–transisomers (formerly called geometric isomers) are a specific subtype of diastereomers in which substituents differ in their position around an immovable bond, such as a double bond, or around a ring structure, such as a cycloalkane in which the rotation of bonds is greatly restricted. In simple compounds with only one substituent on either side of the immovable bond, we use the terms cis and trans. If two substituents are on the same side of the immovable bond, the molecule is considered cis. If they are on opposite sides, it is considered trans, as shown in Figure 2.8 earlier. For more complicated compounds with polysubstituted double bonds, (E)/(Z) nomenclature is used instead, as described in the next section.
MCAT EXPERTISE
While the MCAT is up-to-date with science, it is still possible to see older terms for some concepts on Test Day. Thus, it’s important to know not only the current name, cis–trans isomers, but also the older name, geometric isomers.
*Meso* Compounds
For a molecule to have optical activity, it must not only have chiral centers within it, but must also lack a plane of symmetry. Thus, if a plane of symmetry exists, the molecule is not optically active even if it possesses chiral centers. This plane of symmetry can occur either through the chiral center or between chiral centers. A molecule with chiral centers that has an internal plane of symmetry is called a mesocompound, an example of which is shown in Figure 2.14.
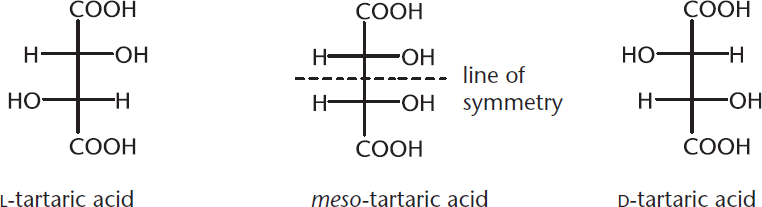
Figure 2.14. Example of a Meso Compound
As shown in this image, D- and L-tartaric acid are both optically active, but meso-tartaric acid has a plane of symmetry and is not optically active. This means that even though meso-tartaric acid has two chiral carbon atoms, the molecule as a whole does not display optical activity. Meso compounds are essentially the molecular equivalent of a racemic mixture.
KEY CONCEPT
Meso compounds are made up of two halves that are mirror images. Thus, as a whole they are not optically active.
MCAT CONCEPT CHECK 2.2:
Before you move on, assess your understanding of the material with these questions.
-
What is the difference between a conformational and a configurational isomer?
- Conformational:
________________________
- Configurational:
________________________
-
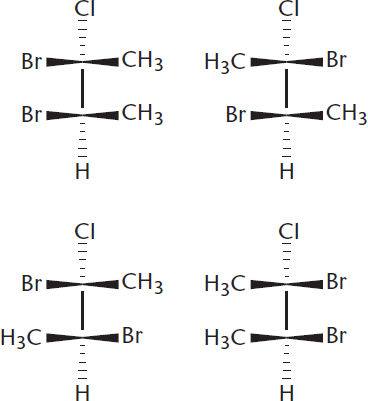
Consider the six pairs that the following four molecules can make. Which pairs are enantiomers? Diastereomers?
-
What is a meso compound?
________________________
-
Complete the Newman projection for the following compound:
2.3 Relative and Absolute Configurations
LEARNING OBJECTIVES
After Chapter 2.3, you will be able to:
- Name cis, trans, E, and Z molecules using appropriate nomenclature
- Apply Cahn–Ingold–Prelog priority rules to molecules with up to four substituents
The configuration of a stereoisomer refers to the spatial arrangement of the atoms or groups in the molecule. The relative configuration of a chiral molecule is its configuration in relation to another chiral molecule (often through chemical interconversion). We can use the relative configuration to determine whether molecules are enantiomers, diastereomers, or the same molecule. On the other hand, the absolute conformation of a chiral molecule describes the exact spatial arrangement of these atoms or groups, independent of other molecules.
(*E*) and (*Z*) Forms
(E) and (Z) nomenclature is used for compounds with oplysubstituted double bonds. Recall that simpler double-bond-containing compounds can use the cis–trans system. To determine the (E)/(Z) designation, one starts by identifying the highest-priority substituent attached to each double-bonded carbon. Using the Cahn–Ingold–Prelog priority rules, priority is assigned based on the atom bonded to the double-bonded carbons: the higher the atomic number, the higher the priority. If the atomic numbers are equal, priority is determined by the next atoms outward; again, whichever group contains the atom with the highest atomic number is given top priority. If a tie remains, the atoms in this group are compared one-by-one in descending atomic number order until the tie is broken. The alkene is named (Z) (German: zusammen, “together”) if the two highest-priority substituents on each carbon are on the same side of the double bond and (E) (entgegen, “opposite”) if they are on opposite sides, as shown in Figure 2.15.
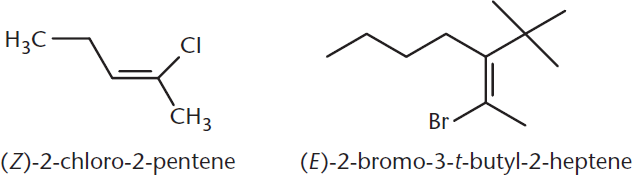
Figure 2.15. (E) and (Z) Designations of Alkenes
MNEMONIC
Z = “z”ame side; E = “e”pposite side
(*R*) and (*S*) Forms
(R) and (S) nomenclature is used for chiral (stereogenic) centers in molecules. We go through a set sequence to determine this absolute configuration:
Step 1: Assign Priority
Using the Cahn–Ingold–Prelog priority rules described earlier, assign priority to the four substituents, looking only at the atoms directly attached to the chiral center. Once again, higher atomic number takes priority over lower atomic number. If the atomic numbers are equal, priority is determined by the combination of the atoms attached to these atoms; if there is a double bond, it is counted as two individual bonds to that atom. If a tie is encountered, work outward from the stereocenter until the tie is broken. An example is shown in Figure 2.16.
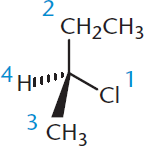
Figure 2.16. Applying the Cahn–Ingold–Prelog Priority Rules to Determine Absolute Configuration Assign priority by the highest atomic number.
KEY CONCEPT
When assigning priority, look only at the first atom attached to the chiral carbon, not at the group as a whole. The higher the atomic number of this first atom, the higher the priority—this same system is used to determine priority for both (E) and (Z) forms, as well as (R) and (S) forms.
Step 2 (Classic Version): Arrange in Space
Orient the molecule in three-dimensional space so that the atom with the lowest priority (usually a hydrogen atom) is at the back of the molecule. Another way to think of this is to arrange the point of view so that the line of sight proceeds down the bond from the asymmetrical carbon atom (the chiral center) to the substituent with lowest priority. The three substituents with higher priority should then radiate out from the central carbon, coming out of the page, as shown in Figure 2.17.
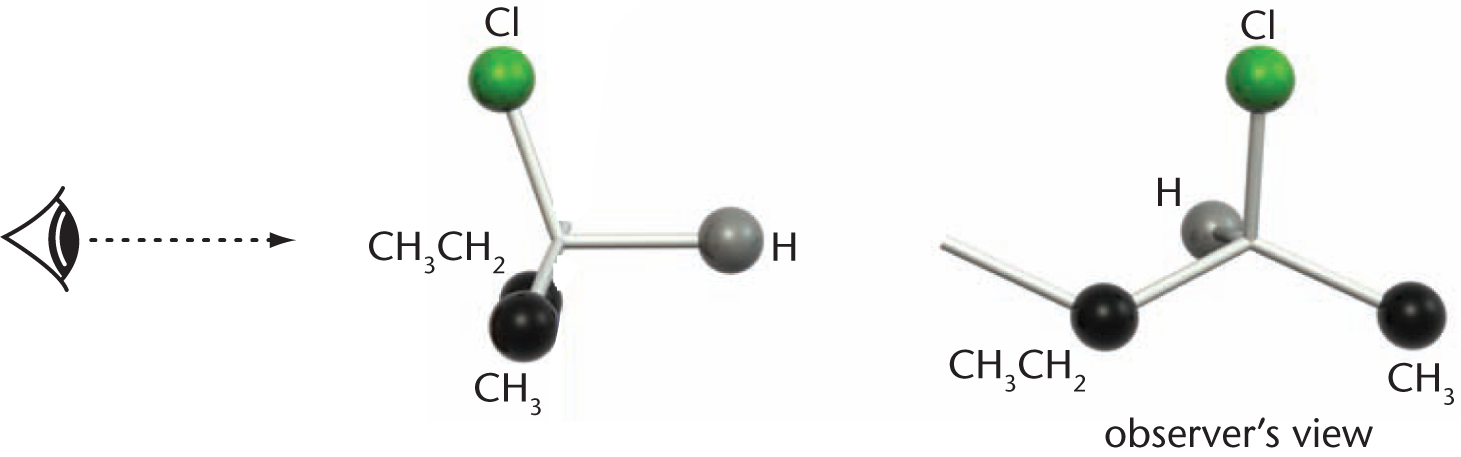
Figure 2.17. Placing the Lowest-Priority Group in the Back
Step 2 (Modified Version): Invert the Stereochemistry
If it is difficult to visualize rotating three-dimensional structures, one can simplify this process by remembering one simple rule: any time two groups are switched on a chiral carbon, the stereochemistry is inverted. By this logic, we can simply switch the lowest-priority group with the group at the back of the molecule (the substituent projecting into the page). We can then proceed to Step 3, keeping in mind that we have now changed the molecule to the opposite configuration. Therefore, if we use this modified step, we need to remember to switch our final answer (either (R) to (S), or (S) to (R)). This is a strategy we’ll commonly use on Fischer diagrams, as described below.
Step 3: Draw a Circle
Now, imagine drawing a circle connecting the substituents from number 1 to 2 to 3. Pay no attention to the lowest-priority group; it can be skipped because it projects directly into the page. If the circle is drawn counterclockwise, the asymmetric atom is called (S) (Latin: sinister, “left”). If it is clockwise, it is called (R) (rectus, “right”), as shown in Figure 2.18. Remember to correct the stereochemistry if the modified version of Step 2 was used.
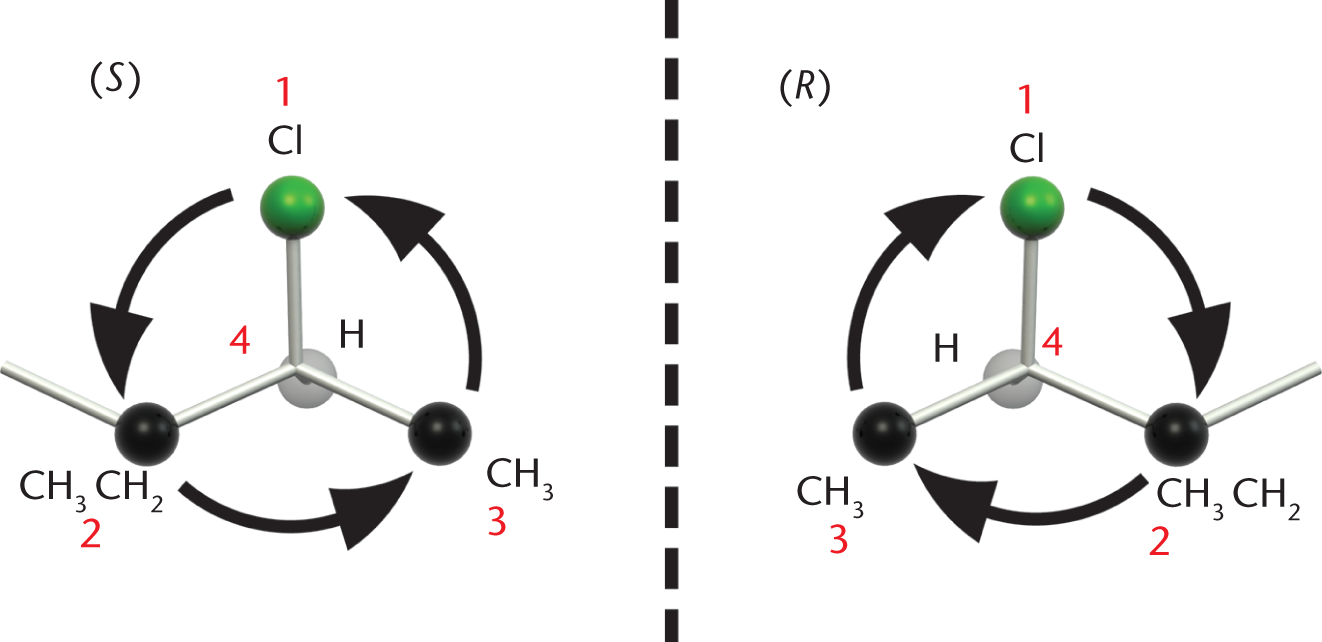
Figure 2.18. Drawing a Circle to Determine Absolute Configuration Counterclockwise = (S); clockwise = (R)
MNEMONIC
A clockwise arrangement is like turning a steering wheel clockwise, which makes a car turn Right—so the chirality at that center is (R).
Step 4: Write the Name
Once the (R)/(S) designation has been determined, the name can be written out. (R) and (S) are put in parentheses and separated from the rest of the name by a hyphen. If we have a compound with more than one chiral center, location is specified by a number preceding the R or S within the parentheses and without a hyphen.
KEY CONCEPT
To determine the absolute configuration at a chiral center:
- Assign priority by atomic number
- Arrange the molecule with the lowest-priority substituent in the back (or invert the stereochemistry by switching two substituents)
- Draw a circle around the molecule from highest to lowest priority (1 to 2 to 3)
- Clockwise = (R); counterclockwise = (S)
Fischer Projections
On the MCAT, one way to represent three-dimensional molecules is by a Fischer projection. In this system, horizontal lines indicate bonds that project out from the plane of the page (wedges), whereas vertical lines indicate bonds going into the plane of the page (dashes). The point of intersection of the lines represents a carbon atom.
To determine configurations using Fischer projections, we follow the same rules listed above. Once again, we have to make sure that the lowest-priority group projects into the page. A benefit of Fischer projections is that the lowest-priority group can be on the top or bottom of the molecule and still project into the page.
Another advantage is that we can manipulate Fischer projections without changing the compound. As mentioned before, switching two substituents around a chiral carbon will invert the stereochemistry ((R) to (S), or (S) to (R)). Rotating a Fischer projection in the plane of the page by 90° will also invert the stereochemistry of the molecule. By extension, interchanging any two pairs of substituents will revert the compound back to its original stereochemistry, and rotating a Fischer projection in the plane of the page by 180° will also retain the stereochemistry of the molecule. These manipulations are shown in Figure 2.19.
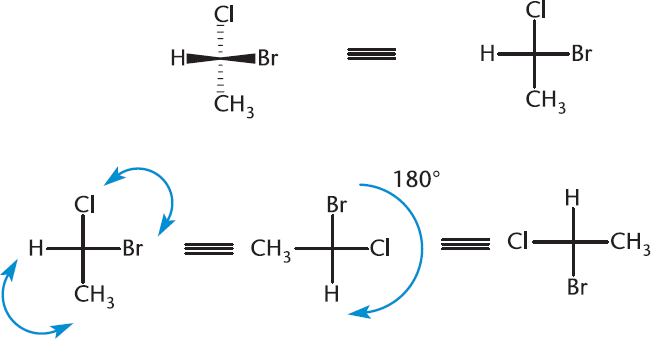
Figure 2.19. Manipulations of Fischer Projections
Again, determining the (R)/(S) designation of a Fischer projection of a compound follows the same rules as described previously. But what if our lowest-priority group is pointing to the side and, as such, pointing out of the page? Just as before, we’ve got a couple of different tricks to help determine the right stereochemistry.
Option 1: Make 0 Switches
Go ahead and determine the order of substituents as normal, drawing a circle from 1 to 2 to 3. Remember, number 4 doesn’t count, so just skip right over it when determining the order. Then, obtain the (R)/(S) designation. The true designation will be the opposite of what you just obtained.
Option 2: Make 1 Switch
Swap the lowest-priority group with one of the groups on the vertical axis. Obtain the (R)/(S) designation and, once again, the true designation will be the opposite of what you just found.
Option 3: Make 2 Switches
In this method, start with option 2, moving the lowest-priority group into the correct position. Then, switch the other two groups as well. Because we made two switches, this molecule will have the same designation as the initial molecule. This is the same as holding one substituent in place and rotating the other three in order.
MCAT EXPERTISE
Determine which option you prefer for Fischer projection (R)/(S) designation and stick with it. It’s more efficient to have a consistent method than to use all three interchangeably.
MCAT CONCEPT CHECK 2.3:
Before you move on, assess your understanding of the material with these questions.
-
How is priority assigned under the Cahn–Ingold–Prelog priority rules?
________________________
-
Name the following compound using E/Z nomenclature:

________________________
-
For each of the Fischer projection manipulations listed below, is stereochemistry retained or inverted?
- Switching a pair of substituents: ____________
- Switching two pairs of substituents: _________
- Rotating the molecule 90°: _______________
- Rotating the molecule 180°: _____________
Conclusion
Throughout this chapter, we’ve seen just how many different molecules can be derived from the same molecular formula. This information is going to be essential on the MCAT—not only for questions on isomerism itself but on every single organic chemistry question you encounter. Most of the compounds we come across will have different possible isomers, and you need to be prepared to differentiate among them to find the one and only correct answer. In the next chapter, we’ll explore how organic molecules are held together through discussions of hybridized orbitals and resonance.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Structural Isomers
- Structural isomers share only a molecular formula.
- They have different physical and chemical properties.
Stereoisomers
- Conformational isomers differ by rotation around a single (σ) bond.
- Staggered conformations have groups 60° apart, as seen in a Newman projection. In ***antistaggeredmolecules, the two largest groups are 180° apart, and strain is minimized. Ingauche* staggered** molecules, the two largest groups are 60° apart.
- Eclipsed conformations have groups directly in front of each other as seen in a Newman projection. In totally eclipsed conformations, the two largest groups are directly in front of each other and strain is maximized.
- The strain in cyclic molecules comes from angle strain (created by stretching or compressing angles from their normal size), torsional strain (from eclipsing conformations), and nonbonded strain (from interactions between substituents attached to nonadjacent carbons). Cyclic molecules will usually adopt nonplanar shapes to minimize this strain.
- Substituents attached to cyclohexane can be classified as axial (sticking up or down from the plane of the molecule) or equatorial (in the plane of the molecule). Axial substituents create more nonbonded strain.
- In cyclohexane molecules with multiple substituents, the largest substituent will usually take the equatorial position to minimize strain.
- Configurational isomers can only be interchanged by breaking and reforming bonds.
- Enantiomers are nonsuperimposable mirror images and thus have opposite stereochemistry at every chiral carbon. They have the same chemical and physical properties except for rotation of plane-polarized light and reactions in a chiral environment.
- Optical activity refers to the ability of a molecule to rotate plane-polarized light: d- or (+) molecules rotate light to the right; l- or (–) molecules rotate light to the left.
- Racemic mixtures, with equal concentrations of two enantiomers, will not be optically active because the two enantiomers’ rotations cancel each other out.
- Mesocompounds, with an internal plane of symmetry, will also be optically inactive because the two sides of the molecule cancel each other out.
- Diastereomers are non-mirror-image stereoisomers. They differ at some, but not all, chiral centers. They have different chemical and physical properties.
- Cis–trans isomers are a subtype of diastereomers in which groups differ in position about an immovable bond (such as a double bond or in a cycloalkane).
- Chiral centers have four different groups attached to the central carbon.
Relative and Absolute Configurations
- Relative configuration gives the stereochemistry of a compound in comparison to another molecule.
- Absolute configuration gives the stereochemistry of a compound without having to compare to other molecules.
- Absolute configuration uses the Cahn–Ingold–Prelog priority rules, in which priority is given by looking at the atoms connected to the chiral carbon or double-bonded carbons; whichever has the highest atomic number gets highest priority. If there is a tie, one moves outward from the chiral carbon or double-bonded carbon until the tie is broken.
- An alkene is (Z) if the highest-priority substituents are on the same side of the double bond and (E) if on opposite sides.
- A stereocenter’s configuration is determined by putting the lowest priority group in the back and drawing a circle from group 1 to 2 to 3 in descending priority. If this circle is clockwise, the stereocenter is (R); if it is counterclockwise, the stereocenter is (S).
- Vertical lines in Fischer diagrams go into the plane of the page (dashes); horizontal lines come out of the plane of the page (wedges).
- Switching one pair of substituents in a Fischer diagram inverts the stereochemistry of the chiral center. Switching two pairs retains the stereochemistry.
- Rotating a Fischer diagram 90° inverts the stereochemistry of the chiral center. Rotating 180° retains the stereochemistry.
ANSWERS TO CONCEPT CHECKS
**2.1**
- Structural isomers share a molecular formula, and not necessarily anything else.
- Cyclopropanol, acetone, and prop-2-ene-1-ol are all structural isomers of each other with the chemical formula C3H6O. 2-Propanol has the chemical formula C3H8O.
- Physical properties are aspects of a compound that do not play a role in changing chemical composition. Examples include melting point, boiling point, solubility, odor, color, and density.
- Chemical properties are aspects of a compound that change chemical composition; in organic chemistry, chemical properties are usually dictated by the reactivity of various functional groups.
**2.2**
- Conformational isomers are stereoisomers with the same molecular connectivity at different points of rotation around a single bond. Configurational isomers are stereoisomers with differing molecular connectivity.
- Enantiomers are nonsuperimposable mirror images. That means the molecules must be mirror images that are different from one another (superimposable mirror images represent the same object). The molecules on the top-left and bottom-right are nonsuperimposable mirror images and therefore enantiomers. The same is true for the top-right and bottom-left. All other combinations are diastereomeric because the pairs differ at some, but not all, stereocenters.
- A meso compound contains chiral centers but also has an internal plane of symmetry. This means that the molecule is overall achiral and will not rotate plane-polarized light.
-
**2.3**
- Priority is assigned by atomic number: the atom connected to the stereocenter or double-bonded carbon with the highest atomic number gets highest priority. If there is a tie, one works outward from the stereocenter or double-bonded carbon until the tie is broken.
- The highest-priority functional group is an alkene, and the longest carbon chain that contains the double bond is five carbons long. So, the root will be pent– and the suffix will be –ene. There are three substituents: a chlorine and two methyls. Number the chain to give the double bond the lowest possible number, in this case from left to right. Bringing it all together gives 1-chloro-2,3-dimethyl-1-pentene. However, there are two possible configurations around the double bond. To determine (E)/(Z) designation, start by identifying the highest-priority substituents attached to each double-bonded carbon. The chlorine and butyl groups are the highest-priority substituents and are on opposite sides of the double bond (in addition to being on opposite ends of the double bond), so this molecule is (E)-1-chloro-2,3-dimethyl-1-pentene.
- Switching a pair of substituents inverts stereochemistry; switching two pairs retains it. Rotating the molecule 90° inverts stereochemistry; rotating 180° retains it.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
This is a racemic mixture of 2-butanol because it consists of equimolar amounts of (R)-2-butanol and (S)-2-butanol. The (R)-2-butanol molecule rotates the plane of polarized light in one direction, and the (S)-2-butanol rotates it by the same angle but in the opposite direction; as a result, no net rotation of polarized light is observed.
2. B
The maximum number of stereoisomers of a compound equals 2n, where n is the number of chiral carbons in the compound. In this molecule, C-1 (the aldehydic carbon) is not chiral, nor is C-5 (because it is attached to two hydrogen atoms). Therefore, with three chiral centers, there are 23 = 8 stereoisomers.
3. C
This answer choice is an example of a meso compound—a compound that contains chiral centers but has an internal plane of symmetry:
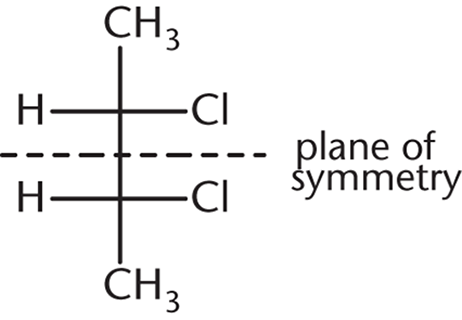
Owing to this internal plane of symmetry, the molecule is achiral and, hence, optically inactive. (A) and (B) are enantiomers of each other and will certainly show optical activity on their own. (D), because it contains a chiral carbon and no internal plane of symmetry, is optically active as well.
4. C
To be considered a chiral center, a carbon must have four different substituents. There are eight stereocenters in this molecule, which are marked below with asterisks.
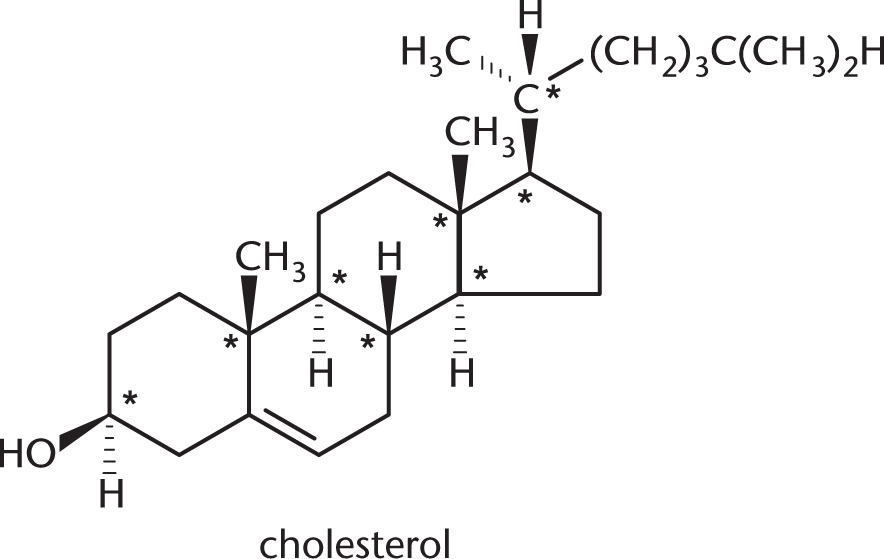
The other carbons are not chiral for various reasons. Many are bonded to two hydrogens; others participate in double bonds, which count as two bonds to the same atom.
5. B
This molecule is a chair conformation in which the two equatorial methyl groups are trans to each other. Because the axial methyl hydrogens do not compete for the same space as the hydrogens attached to the ring, this conformation ensures the least amount of steric strain. (A) would be less stable because the diaxial methyl group hydrogens are closer to the hydrogens on the ring, causing greater steric strain. (C) is incorrect because it is in the more unstable boat conformation.
6. C
The relative configuration is retained because the bonds of the stereocenter are not broken; thus, the positions of groups around the chiral carbon are maintained. The absolute configuration is also retained because both the reactant and product are (R).
7. A
These compounds are nonsuperimposable mirror images. To make analysis a bit easier, we can rotate structure II 180° to look like structure III. Structures I and III more clearly have opposite stereochemistry at every chiral center, meaning that they are enantiomers.
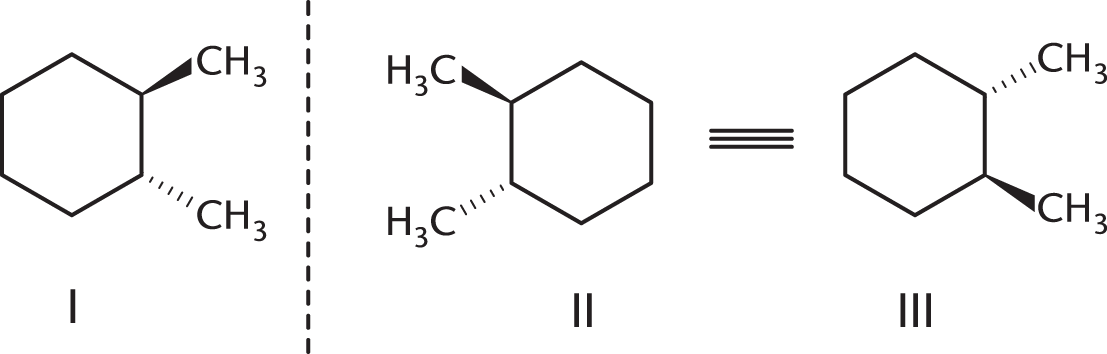
(B) is incorrect because diastereomers are stereoisomers that are not mirror images of each other. (C) is incorrect because meso compounds must contain a plane of symmetry, which neither of these molecules has. (D) is incorrect because structural isomers are compounds with the same molecular formula but different atomic connections. The connectivity in these two molecules is the same, which means that they are stereoisomers, not structural isomers.
8. A
(+)-Glyceraldehyde and (–)-glyceraldehyde, or (R)- and (S)-2,3-dihydroxypropanal, are enantiomers. Enantiomers are nonsuperimposable mirror images. Each has only one chiral center (C-2), which has opposite absolute configuration in these two molecules.
9. C
(E)-2-butene can also be called trans-2-butene; (Z)-2-butene can also be called cis-2-butene. As such, they are cis–trans isomers. Remember that cis–trans isomers are a subtype of diastereomers in which the position of substituents differs about an immovable bond. Diastereomers are molecules that are non-mirror-image stereoisomers (molecules with the same atomic connectivity). These are not enantiomers because they are not mirror images of each other.
10. C
Because they have the same molecular formula but different atomic connectivity, 3-methylpentane and hexane are constitutional isomers.
11. B
These two molecules are stereoisomers of one another, but are not nonsuperimposable mirror images. Therefore, they are diastereomers. Note that these molecules differ by at least one, but not all, chiral carbons.
12. D
Remember that the equation for specific rotation is
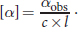
. In this example, αobs is +12° (remember that dextrorotatory, or clockwise, rotation is considered positive),
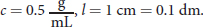
Remember that path length is always measured in decimeters when calculating specific rotation. Therefore, the specific rotation can be calculated as:
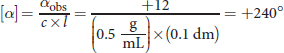
13. B
Racemic mixtures like omeprazole contain equimolar amounts of two enantiomers and thus have no observed optical activity. Each of the two enantiomers causes rotation in opposite directions, so their effects cancel out. Esomeprazole only contains one of the two enantiomers and thus should cause rotation of plane-polarized light.
14. C
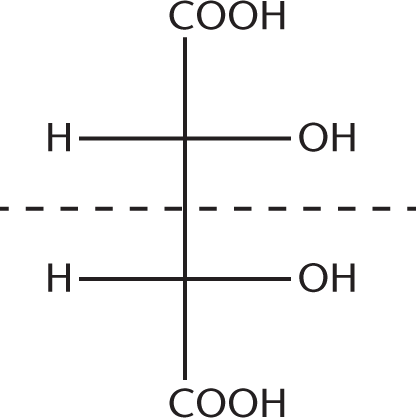
Draw out these structures. The two names describe the same molecule, which also happens to be a meso compound because it contains a plane of symmetry. These compounds are not enantiomers because they are superimposable mirror images of one another, not nonsuperimposable mirror images. These compounds are better termed meso-2,3-dihydroxybutanedioic acid:
15. C
In butane, the position at which the two methyl groups are 120° apart is an eclipsed conformation. This has a moderate amount of energy, although not as high as a totally eclipsed conformation in which the two methyl groups are 0° apart.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
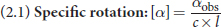
SHARED CONCEPTS
- Biochemistry Chapter 1
- Amino Acids, Peptides, and Proteins
- Biochemistry Chapter 4
- Carbohydrate Structure and Function
- General Chemistry Chapter 3
- Bonding and Chemical Interactions
- Organic Chemistry Chapter 3
- Bonding
- Organic Chemistry Chapter 4
- Analyzing Organic Reactions
- Physics and Math Chapter 8
- Light and Optics