Chapter 12: Separations and Purifications

Chapter 12: Separations and Purifications
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Fractional distillation under atmospheric pressure would most likely be used to separate which of the following compounds?
- Methylene chloride (boiling point of 40 °C) and water (boiling point of 10 °C)
- Ethyl acetate (boiling point of 77 °C) and ethanol (boiling point of 80 °C)
- Aniline (boiling point of 184 °C) and benzyl alcohol (boiling point of 205 °C)
- Aniline (boiling point of 184 °C) and water (boiling point of 100 °C)
-
Which of the following compounds would be most effective in extracting benzoic acid from a diethyl ether solution?
- Tetrahydrofuran
- Aqueous hydrochloric acid
- Aqueous sodium hydroxide
- Water
-
Which of the following would be the best procedure for extracting acetaldehyde from an aqueous solution?
- A single extraction with 100 mL of ether
- Two successive extractions with 50 mL portions of ether
- Three successive extractions with 33.3 mL portions of ether
- Four successive extractions with 25 mL portions of ether
Questions 4 and 5 refer to the following table: Compound Retardation Factor in Ether
Benzyl alcohol 0.10
Benzyl acetate 0.26
p-Nitrophenol 0.23
1-Naphthalenemethanol 0.40
-
What would be the effect on the Rf values if thin-layer chromatography (TLC) were run with hexane rather than ether as the eluent?
- No effect
- Increase tenfold
- Double
- Decrease
-
If these compounds were separated by column chromatography with ether on silica gel, which would elute first?
- Benzyl alcohol
- Benzyl acetate
- p-Nitrophenol
- 1-Naphthalenemethanol
-
Four compounds, I, II, III, and IV, are separated by chromatographic techniques. Compound III is the most polar, II the least polar, and I and IV have intermediate polarity. The solvent system is 85:15 ethanol:methylene chloride. Which spot on the card below likely belongs to compound III?
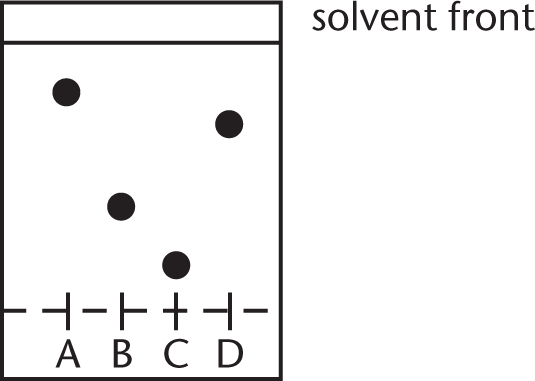
- A
- B
- C
- D
-
Suppose an extraction with methylene chloride
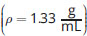
is performed, with the desired compound initially in brine
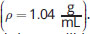
In a separatory funnel, which layer will be the organic layer?
- The top layer
- The bottom layer
- No layers are observed; methylene chloride and brine are miscible.
- More information is needed to answer the question.
-
Silica gel is often used in thin-layer chromatography. What property does silica gel probably possess that makes it useful for this purpose?
- Acidity
- Polarity
- Specifically sized pores
- Aqueous solubility
-
A mixture of sand, benzoic acid, and naphthalene in ether is best separated by:
- filtration, followed by acidic extraction, followed by recrystallization.
- filtration, followed by basic extraction, followed by evaporation.
- extraction, followed by distillation, followed by gas chromatography.
- filtration, followed by size-exclusion column chromatography, followed by extraction.
-
Simple distillation could be used to separate which of the following compounds?
- Toluene (boiling point of 111 °C) and water (boiling point of 100 °C)
- Naphthalene (boiling point of 218 °C) and butyric acid (boiling point of 163 °C)
- Propionaldehyde (boiling point of 50 °C) and acetic acid (boiling point of 119 °C)
- Benzene (boiling point of 80 °C) and isopropyl alcohol (boiling point of 83 °C)
-
In order to separate a biological effector from solution, which chromatographic technique would be the most effective?
- Thin-layer chromatography
- Ion-exchange chromatography
- Affinity chromatography
- Size-exclusion chromatography
-
Given a solution of insulin (molecular weight = 5.8 kD) and titin (molecular weight = 3816 kD), which chromatographic technique would be the most effective for separating out usable molecules of titin?
- Thin-layer chromatography
- Ion-exchange chromatography
- Affinity chromatography
- Size-exclusion chromatography
-
The gas eluent in gas chromatography and the liquid eluent in paper chromatography are examples of which component of these systems?
- Stationary phase
- Mobile phase
- Column
- Fraction
-
During gravity filtration, a student forgets to heat the solution before running it through the filter. After capturing the filtrate, the student analyzes the sample via infrared (IR) spectroscopy and finds none of the desired product in the filtrate. What likely occurred to the student’s product?
- The product degraded because of a prolonged filtration time.
- The product evaporated with collection of the filtrate.
- The product precipitated and is present in the residue.
- The product was dissolved in the solvent.
-
Lactoferrin, a milk protein, is a valuable antimicrobial agent that is extracted from pasteurized, defatted milk utilizing a column containing a charged resin. This is an example of which of the following chromatographic techniques?
- Thin-layer chromatography
- Ion-exchange chromatography
- Affinity chromatography
- Size-exclusion chromatography
Answer Key
- B
- C
- D
- D
- D
- A
- B
- B
- B
- C
- C
- D
- B
- C
- B
Chapter 12: Separations and Purifications
CHAPTER 12
SEPARATIONS AND PURIFICATIONS
In This Chapter
12.1 Solubility-Based Methods
Extraction
Other Methods
12.2 Distillation
Simple Distillation
Vacuum Distillation
Fractional Distillation
12.3 Chromatography
Thin-Layer and Paper Chromatography
Column Chromatography
Gas Chromatography
High-Performance Liquid Chromatography
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 17% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content category:
5C: Separation and purification methods
Introduction
We’ve spent a lot of time discussing how to theoretically get various products from a range of reagents. However, as we’re sure you’ve learned in your organic chemistry labs, chemistry isn’t as straightforward in the real world as it is on paper. Much of the time spent in the lab is dedicated to the isolation and purification of the desired product after the reaction has occurred. Throughout this chapter, we will discuss several techniques for this. Good news, though—there isn’t a lab practical on the MCAT, of course! All you need to understand is when these techniques are used and how they work.
MCAT EXPERTISE
Purification and separation techniques are procedures that exploit different physical properties. These show up on the MCAT to test your knowledge of the basic principles of each of these techniques.
12.1 Solubility-Based Methods
LEARNING OBJECTIVES
After Chapter 12.1, you will be able to:
- Recall the conditions required for two solvents to be used together in an extraction
- Explain why repetition is important in extraction procedures
- Predict whether a given solute is more likely to dissolve in the aqueous or organic layer within a separatory funnel
One of the simplest ways to separate out a desired product is through extraction, the transfer of a dissolved compound (the desired product) from a starting solvent into a solvent in which the product is more soluble. Extraction is based on the fundamental concept that like dissolves like. This principle tells us that a polar substance will dissolve best in polar solvents, and a nonpolar substance will dissolve best in nonpolar solvents. These characteristics can be taken advantage of in order to extract only the desired product, leaving most of the impurities behind in the first solvent.
KEY CONCEPT
Like dissolves like is a fundamental concept on the MCAT. Remember that polar substances will associate with other polar substances, and nonpolar with nonpolar.
Extraction
When we perform extractions, it is important to make sure that the two solvents are immiscible, meaning that they form two layers that do not mix, like water and oil. The two layers are temporarily mixed by shaking so that solute can pass from one solvent to the other. For example, in a solution of isobutyric acid and diethyl ether, shown in Figure 12.1, we can extract the isobutyric acid with water. Isobutyric acid, with its polar carboxyl group, is more soluble in a polar solvent like water than in a nonpolar solvent like ether. When the two solvents are mixed together, isobutyric acid will transfer to the water layer, which is called the aqueous phase (layer). The nonpolar ether layer is called the organic phase (layer).
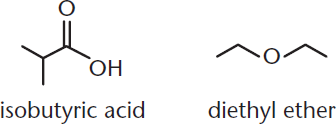
Figure 12.1. Isobutyric Acid and Diethyl Ether Isobutyric acid is more polar than diethyl ether and can exhibit hydrogen bonding, so it will congregate in the aqueous layer; diethyl ether will remain in the organic layer.
MCAT EXPERTISE
Think of the organic and aqueous layers as being like the oil and water in salad dressings: you can shake the mixture to increase their interaction, but ultimately they will separate again.
After the two layers are mixed together, how do we then get the desired product out? The water (aqueous) and ether (organic) phases will separate on their own, given time to do so. In order to isolate these two phases, we use a piece of equipment called a separatory funnel, as shown in Figure 12.2. Gravitational forces cause the denser layer to sink to the bottom of the funnel, where it can then be removed by turning the stopcock at the bottom. It is more common for the organic layer to be on top, although the opposite can also occur. Remember that the position of the layers is determined by their relative densities.
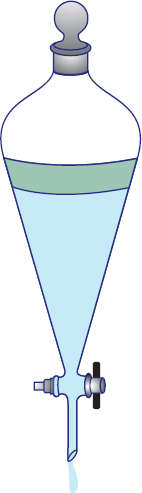
Figure 12.2. Separatory Funnel Used to separate solvents based on their relative densities; the denser solvent is always drained first.
KEY CONCEPT
Extraction depends on the rules of solubility and like dissolves like. Remember the three intermolecular forces that affect solubility:
- Hydrogen bonding: Compounds that can do this, such as alcohols or acids, will move most easily into the aqueous layer
- Dipole–dipole interactions: These compounds are less likely to move into the aqueous layer
- Van der Waals (London) forces: With only these interactions, compounds are least likely to move into the aqueous layer
In this example, we’ll assume that the aqueous layer is more dense and settles to the bottom of the separatory funnel. Once we drain the aqueous layer from the separatory funnel, we repeat the extraction several times. Additional water is added to the separatory funnel, it is shaken and allowed to settle, and the aqueous layer is once again drained off. This is done in order to extract as much of the isobutyric acid from the ether layer as possible because it does not completely transfer with the first extraction. Multiple extractions with fresh water are more effective for obtaining the most product, rather than a single extraction with a larger volume of water. You can imagine this as analogous to laundering dirty clothes several times, rather than laundering them with more water—the cleaner each volume of water is, the less dirt is likely to be left on the clothes afterward.
BRIDGE
You can use the properties of acids and bases to your advantage in extraction:
HA + base → A– + H–base+
When the acid dissociates, the anion formed will be more soluble in the aqueous layer than the original protonated acid because it is charged. Thus, adding a base will help to extract an acid into the aqueous phase.
Once the desired product has been isolated in the solvent, we can obtain the product alone by evaporating the solvent, usually by using a rotary evaporator (rotovap).
Another way to take advantage of solubility properties is to perform the reverse of the extraction we just described in order to remove unwanted impurities. In this case, a small amount of solvent is used to extract and remove impurities, rather than the compound of interest. This process is called a wash.
Other Methods
In addition to extraction, filtration and recrystallization make use of solubility characteristics to separate compounds from a mixture.
Filtration
Filtration isolates a solid from a liquid. In the chemistry lab, one pours a liquid–solid mixture onto a paper filter that allows only the solvent to pass through, much like a coffee filter. At the end of filtration, one is left with the solid, called the residue, and the flask full of liquid that passed through the filter, known as the filtrate.
Filtration can be modified depending on whether the substance of interest is the solid or is dissolved in the filtrate. Gravity filtration, in which the solvent’s own weight pulls it through the filter, is more commonly used when the product of interest is in the filtrate. Hot solvent is generally used to keep the product dissolved in liquid. Vacuum filtration, in which the solvent is forced through the filter by a vacuum connected to the flask, is more often used when the solid is the desired product.
Recrystallization
Recrystallization is a method for further purifying crystals in solution. In this process, we dissolve our product in a minimum amount of hot solvent and let it recrystallize as it cools. The solvent chosen for this process should be one in which the product is soluble only at high temperatures. Thus, when the solution cools, only the desired product will recrystallize out of solution, excluding the impurities.
MCAT CONCEPT CHECK 12.1:
Before you move on, assess your understanding of the material with these questions.
-
What must be true about the two solvents used for an extraction to work?
________________________
-
When doing an extraction, would it be better to do three extractions with 10 mL of solvent, or one extraction with 30 mL?
________________________
-
Would acid dissolve better in an aqueous acid or aqueous base? Why?
________________________
12.2 Distillation
LEARNING OBJECTIVES
After Chapter 12.2, you will be able to:
- Differentiate between the separatory capabilities of simple, vacuum, and fractional distillation
- Recall the conditions in which distillation is a helpful separatory technique
- Select the best distillation technique for a given solute mixture
Extraction requires two solvents that are immiscible in order to separate the product. But what happens when the product itself is a liquid that is soluble in the solvent? This is where distillation comes in handy. Distillation takes advantage of differences in the boiling point to separate two liquids by evaporation and condensation. The liquid with the lower boiling point will vaporize first, and the vapors will rise up the distillation column to condense in a water-cooled condenser. This condensate then drips down into a vessel. The end product is called the distillate. The heating temperature is kept low so that the liquid with the higher boiling point will not be able to boil and therefore will remain liquid in the initial container. This is the process that is used to make liquor at a distillery. Because ethanol boils at a lower temperature than water, we can use distillation to make beverages with high ethanol contents.
Simple Distillation
Simple distillation, as the name indicates, is the least complex version of distillation. It proceeds precisely as described above. This technique should only be used to separate liquids that boil below 150 °C and have at least a 25 °C difference in boiling points. These restrictions prevent the temperature from becoming so high that the compounds degrade and provide a large enough difference in boiling points that the second compound won’t accidentally boil off into the distillate. The apparatus for this technique consists of a distilling flask containing the combined liquid solution, a distillation column consisting of a thermometer and a condenser, and a receiving flask to collect the distillate. The setup is the same as that shown in Figure 12.3, sans the vacuum adapter. Sometimes an additional piece of equipment, such as a boiling chip, ebulliator, or magnetic stirrer, will be introduced to break surface tension and prevent superheating. Superheating occurs when a liquid is heated to a temperature above its boiling point without vaporization. Superheating situations occur when gas bubbles within a liquid are unable to overcome the combination of atmospheric pressure and surface tension.
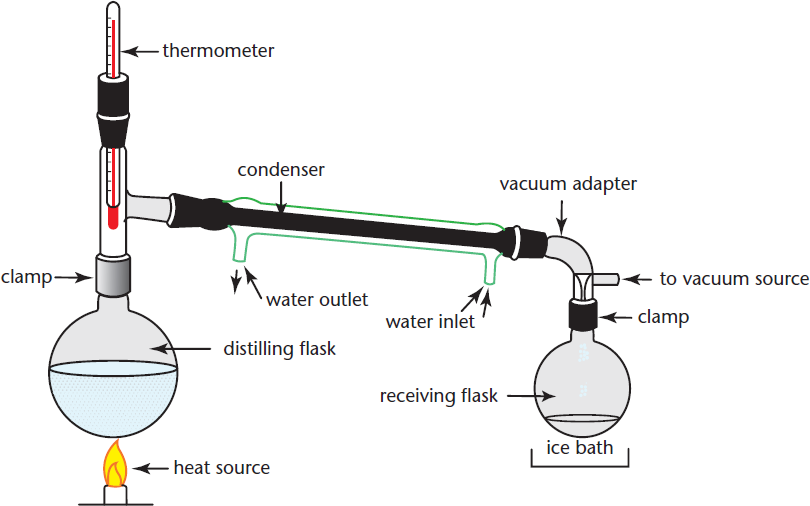
Figure 12.3. Vacuum Distillation The initial solution is placed in the heated distilling flask, where the components of the solution with the lowest boiling points will vaporize first. The vapor then condenses in the water-cooled condenser, and this distillate drips into the receiving flask.
Vacuum Distillation
We use vacuum distillation whenever we want to distill a liquid with a boiling point over 150 °C. By using a vacuum, we lower the ambient pressure, thereby decreasing the temperature that the liquid must reach in order to have sufficient vapor pressure to boil. This allows us to distill compounds with higher boiling points at lower temperatures so that we do not have to worry about degrading the product. The apparatus for vacuum distillation is shown above in Figure 12.3.
BRIDGE
Remember from Chapter 7 of MCAT General Chemistry Review that liquids boil when their vapor pressure equals ambient pressure. In vacuum distillation, we lower the ambient pressure so that the liquid can boil at lower temperatures.
Fractional Distillation
To separate two liquids with similar boiling points (less than 25 °C apart), we use fractional distillation. In this technique, a fractionation column connects the distillation flask to the condenser, as shown in Figure 12.4. A fractionation column is a column in which the surface area is increased by the inclusion of inert objects like glass beads or steel wool. As the vapor rises up the column, it condenses on these surfaces and refluxes back down until rising heat causes it to evaporate again, only to condense again higher in the column. Each time the condensate evaporates, the vapor consists of a higher proportion of the compound with the lower boiling point. By the time the top of the column is reached, only the desired product drips down to the receiving flask.
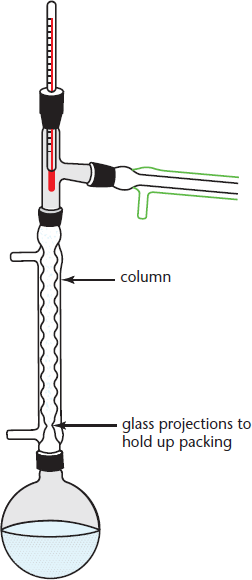
Figure 12.4. Fractional Distillation With increased surface area in the distillation column, the distillate has more places to condense on its way up the column. This allows for more refined separation of liquids with fairly close boiling points.
MCAT CONCEPT CHECK 12.2:
Before you move on, assess your understanding of the material with these questions.
-
Distillation separates compounds based on what property?
________________________
-
If we are given a solution of ether, with a boiling point of 308 K, and methylene chloride, with a boiling point of 313 K, which type of distillation should be used to separate them?
________________________
-
If we are given a solution of bromobenzene, with a boiling point of 156 °C, and camphor, with a boiling point of 204 °C, which type of distillation should be used to separate them?
________________________
12.3 Chromatography
LEARNING OBJECTIVES
After Chapter 12.3, you will be able to:
- Identify the properties that can be used to separate compounds through chromatography
- Differentiate between thin-layer, paper, column, gas, and high-performance liquid chromatography
- Describe the three main specialized types of columns used in column chromatography, and their methods for separation
- Predict the best chromatography method for a given mixture
Chromatography is another tool that uses physical and chemical properties to separate and identify compounds from a complex mixture. In all forms of chromatography discussed here, the concept is identical: the more similar a compound is to its surroundings (whether by polarity, charge, or other characteristics), the more it will stick to and move slowly through its surroundings.
KEY CONCEPT
Chromatography separates compounds based on how strongly they adhere to the solid, or stationary, phase (or in other words, how easily they come off into the mobile phase).
The process begins by placing the sample onto a solid medium called the stationary phase, or adsorbent. We then run the mobile phase, usually a liquid (or a gas in gas chromatography) through the stationary phase. This will displace (elute) the sample and carry it through the stationary phase. Depending on the characteristics of the substances in the sample and the polarity of the mobile phase, it will adhere to the stationary phase with differing strengths, causing the different substances to migrate at different speeds. This is called partitioning, and it represents an equilibrium between the two phases. Different compounds will have different partitioning coefficients and will elute at different rates. This results in separation within the stationary phase, allowing us to isolate each substance individually.
There are many different media that can be used as the stationary phase, each one exploiting different properties that allow us to separate out the desired compound. On the MCAT, the property most commonly used is polarity. For instance, thin-layer chromatography (TLC), which we will shortly discuss, uses silica gel, a highly polar substance, as its stationary phase. Cellulose, another polar substance, may also be used. This means that any polar compound will adhere well to the gel and thus move through (elute) slowly. When using column chromatography, size and charge both have a role in how quickly a compound moves through the stationary phase. Chromatography can use even strong interactions, such as antibody–ligand binding.
As mentioned earlier, chromatography is based on the speed at which compounds move through media. In practice, however, we will measure either how far each substance travels in a given amount of time (such as in TLC) or how long it takes to elute (as in column or gas chromatography).
The types of chromatography that we will discuss include thin-layer and paper chromatography, column chromatography, gas chromatography (also called gas–liquid chromatography), and high-performance liquid chromatography, or HPLC.
Thin-Layer and Paper Chromatography
Thin-layer chromatography and paper chromatography are extremely similar techniques, varying only in the medium used for the stationary phase. For thin-layer chromatography, a thin layer of silica gel or alumina adherent to an inert carrier sheet is used. For paper chromatography, as the name suggests, the medium used is paper, which is composed of cellulose.
For these techniques, the sample that we want to separate is placed directly onto the adsorbent itself; this is called spotting because we apply a small, well-defined spot of the sample directly onto the silica or paper plate. The plate is then developed, which involves placing the adsorbent upright in a developing chamber, usually a beaker with a lid or a wide-mouthed jar. At the bottom of this jar is a shallow pool of solvent, called the eluent. The spots of the sample must be above the level of the solvent, or else they will dissolve into the pool of solvent rather than running up the plate. When set up correctly, the solvent will creep up the plate by capillary action, carrying the various compounds in the sample with it at varying rates. When the solvent front nears the top of the plate, the plate is removed from the chamber and allowed to dry.
As mentioned before, TLC is often done with silica gel, which is polar and hydrophilic. The mobile phase, on the other hand, is usually an organic solvent of weak to moderate polarity, so it doesn’t bind well to the gel. Because of this, nonpolar compounds dissolve in the organic solvent and move quickly as the solvent moves up the plate, whereas the more polar molecules stick to the gel. Thus, the more nonpolar the sample is, the further up the plate it will move, as shown in Figure 12.5.
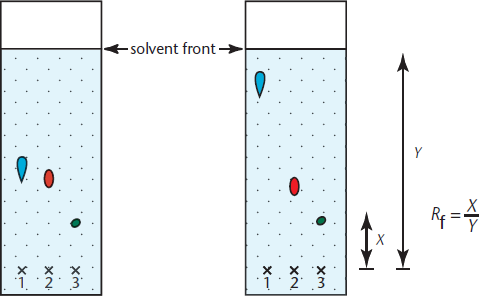
Figure 12.5. Thin-Layer Chromatography Samples are placed at the “X” marks. As the nonpolar solvent moves up the plate via capillary action, the samples that are nonpolar move further up the plate along with the solvent, while the samples that are polar do not move as far.
Reverse-phase chromatography is the exact opposite. In this technique, the stationary phase used is nonpolar, so polar molecules move up the plate quickly, while nonpolar molecules stick more tightly to the stationary phase.
The spots of individual compounds are usually white, which makes them difficult or impossible to see on the white paper or TLC plate. To get around this problem, the developed plate can be placed under ultraviolet light, which will show any compounds that are ultraviolet-sensitive. Alternatively, iodine, phosphomolybdic acid, or vanillin can be used to stain the spots, although this will destroy the compounds such that they cannot be recovered.
When TLC is performed, compounds are generally identified using the **retardation factor (Rf)**, which is relatively constant for a particular compound in a given solvent. The Rf is calculated using the equation:
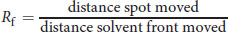
Equation 12.1
Because its value is relatively constant, the Rf value can be used to identify unknown compounds.
This technique is most frequently performed on a small scale to identify unknown compounds. It can also be used on a larger scale as a means of purification, a technique called preparative TLC. As the large plate develops, the larger spot of sample splits into bands of individual compounds, which can then be scraped off and washed to yield pure compounds.
Column Chromatography
The principles behind column chromatography are the same as for thin-layer chromatography, although there are some differences. First, column chromatography uses an entire column filled with silica or aluminum beads as an adsorbent, allowing for much greater separation. The setup for this is shown in Figure 12.6. In addition, thin-layer chromatography uses capillary action to move the solvent up the plate, whereas column chromatography uses gravity to move the solvent and compounds down the column. To speed up the process, one can force the solvent through the column using gas pressure, a technique called flash column chromatography. In column chromatography, the solvent polarity can also be changed to help elute the desired compound.
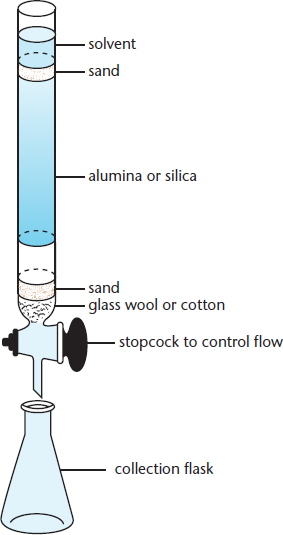
Figure 12.6. Column Chromatography The sample is added to the top of the column, and a solvent is poured over it. The more similar the sample is to the mobile phase, the faster it elutes; the more similar it is to the stationary phase, the more slowly it will elute (if at all).
Eventually, the solvent drips out of the end of the column, and the different fractions that leave the column can be collected over time. Each fraction will contain different compounds. After collection, the solvent can be evaporated, leaving behind the compounds of interest. Column chromatography is particularly useful in biochemistry because it can be used to separate and collect macromolecules such as proteins or nucleic acids. There are several techniques that can be used to isolate specific materials, which are described in the following paragraphs, as well as in Chapter 3 of MCAT Biochemistry Review.
Ion-Exchange Chromatography
In ion-exchange chromatography, the beads in the column are coated with charged substances so that they attract or bind compounds that have an opposite charge. For instance, a positively charged compound will attract and hold a negatively charged backbone of DNA or protein as it passes through the column, either increasing its retention time or retaining it completely. After all other compounds have moved through the column, a salt gradient is used to elute the charged molecules that have stuck to the column.
Size-Exclusion Chromatography
In size-exclusion chromatography, the beads used in the column contain tiny pores of varying sizes. These tiny pores allow small compounds to enter the beads, thus slowing them down. Large compounds can’t fit into the pores, so they will move around them and travel through the column faster. It is important to remember that in this type of chromatography, the small compounds are slowed down and retained longer—which may be counterintuitive. The size of the pores may be varied so that molecules with different molecular weights can be fractionated. A common approach in protein purification is to use an ion-exchange column followed by a size-exclusion column.
Affinity Chromatography
In affinity chromatography, a protein of interest is bound by creating a column with high affinity for that protein. This can be accomplished by coating beads with a receptor that binds the protein or a specific antibody to the protein; in either case, the protein is retained in the column. Common stationary phase molecules include nickel, which is used in the separation of genetically engineered proteins with histidine tags, antibodies or antigens, and enzyme substrate analogues, which mimic the natural substrate for an enzyme of interest. Once the protein is retained in the column, it can be eluted by washing the column with a free receptor (or target or antibody), which will compete with the bead-bound receptor and ultimately free the protein from the column. Eluents can also be created with a varying pH or salinity level that disrupts the bonds between the ligand and the protein of interest. The only drawback of the elution step is that the recovered substance can be bound to the eluent. If, for example, the eluent was an inhibitor of an enzyme, it could be difficult to remove.
Gas Chromatography
Gas chromatography (GC) is another method that can be used for qualitative separation. GC, also known as vapor-phase chromatography (VPC), is similar to the other types of chromatography and is shown in Figure 12.7. The main conceptual difference is that the eluent is a gas (usually helium or nitrogen) instead of a liquid. The adsorbent is a crushed metal or polymer inside a 30-foot column. This column is coiled and kept inside an oven to control its temperature. The mixture is then injected into the column and vaporized. The gaseous compounds travel through the column at different rates because they adhere to the adsorbent in the column to different degrees and will separate in space by the time they reach the end of the column. The injected compounds must be volatile: low melting-point, sublimable solids or vaporizable liquids. The compounds are registered by a detector, which records them as a peak on a chart.
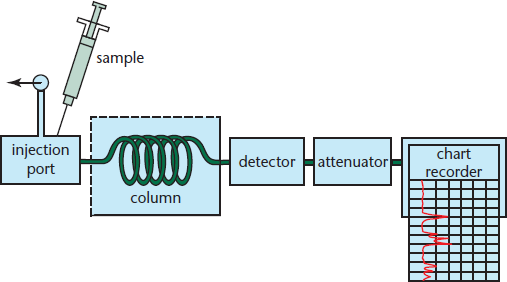
Figure 12.7. Gas Chromatography The sample is injected into the column and moves with the gaseous mobile phase through a stationary liquid or solid phase; a computer identifies the sample components.
It is common to separate molecules using GC and then to inject the pure molecules into a mass spectrometer for molecular weight determination. Mass spectrometry involves the ionization and fragmentation of compounds; these fragments are then run through a magnetic field, which separates them by mass-to-charge ratio. The total molecular weight can thus be determined, or the relative concentrations of the different fragments can be calculated and compared against reference values to identify the compound.
High-Performance Liquid Chromatography
High-performance liquid chromatography (HPLC) was previously called high-pressure liquid chromatography. As the name suggests, the eluent is a liquid, and it travels through a column of a defined composition. There is a variety of stationary phases that can be chosen depending on the target molecule and the quantity of material that needs to be purified. This is fairly similar to column chromatography because the various compounds in solution will react differently with the adsorbent material. In the past, very high pressures were used, but recent advances allow for much lower pressures—hence the change in name. In HPLC, a small sample is injected into the column, and separation occurs as it flows through. The compounds pass through a detector and are collected as the solvent flows out of the end of the apparatus. The interface is similar to that used for GC because the entire process is computerized, but it uses liquid under pressure instead of gas. Because the whole process is under computer control, sophisticated solvent gradients as well as temperature can be applied to the column to help resolve the various compounds in the sample—hence the higher performance of HPLC over regular column chromatography.
MCAT CONCEPT CHECK 12.3:
Before you move on, assess your understanding of the material with these questions.
-
What properties of molecules do thin-layer chromatography, paper chromatography, and standard column chromatography take advantage of to separate compounds?
________________________
-
What are the three specialized types of column chromatography? What does each use in order to separate the sample?
Type of Column Chromatography Method for Separating Sample
-
In what way is gas chromatography distinct from all of the other techniques we have discussed?
________________________
-
What is the major historical distinction between HPLC and column chromatography? What is the major distinction now?
- Historical distinction:
________________________
- Current distinction:______________________________
Conclusion
Don’t forget, the MCAT won’t ask you to get into your lab coat and extract the product of a reaction! As long as you understand the principles governing these techniques and when you should apply them, you’ll be in great shape. Remember that separation and purification techniques exploit physical properties of compounds, such as polarity, solubility, size and shape, and charge, to obtain a purified product. These properties can be traced back to intermolecular forces or properties of the molecules themselves. Having a variety of tools and methods to separate and collect a purified product is essential in practical organic chemistry, and choosing the proper techniques often requires knowledge and consideration of the desired product. When you look at the bigger picture, these methods may be easier to conceptualize than to actually apply in the lab!
Despite the subject’s compelling relevance to everyday life, college organic chemistry often terrifies and alienates its students. The MCAT, on the other hand, doesn’t ask you to memorize tables of reactants or regurgitate hundreds of named reactions from scratch. Instead, the MCAT asks you to look at the bigger picture, to know trends, and to participate in the logic of chemistry. We hope that studying for the MCAT has given you a chance to rediscover organic chemistry—to focus on the how and the why, instead of the what. Organic chemistry, like the MCAT as a whole, should be seen not as an obstacle but as an opportunity. So, work hard, have some fun along the way, and keep thinking about where you’re heading—you can almost feel that white coat.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Solubility-Based Methods
- Extraction combines two immiscible liquids, one of which easily dissolves the compound of interest.
- The polar (water) layer is called the aqueous phase and dissolves compounds with hydrogen bonding or polarity.
- The nonpolar layer is called the organic phase and dissolves nonpolar compounds.
- Extraction is carried out in a separatory funnel. One phase is collected, and the solvent is then evaporated.
- Acid–base properties can be used to increase solubility.
- A wash is the reverse of extraction, in which a small amount of solvent that dissolves impurities is run over the compound of interest.
- Filtration isolates a solid (residue) from a liquid (filtrate).
- Gravity filtration is used when the product of interest is in the filtrate. Hot solvent is used to maintain solubility.
- Vacuum filtration is used when the product of interest is the solid. A vacuum is connected to the flask to pull the solvent through more quickly.
- In recrystallization, the product is dissolved in a minimum amount of hot solvent. If the impurities are more soluble, the crystals will reform while the flask cools, excluding the impurities.
Distillation
- Distillation separates liquids according to differences in their boiling points; the liquid with the lowest boiling point vaporizes first and is collected as the distillate.
- Simple distillation can be used if the boiling points are under 150 °C and are at least 25 °C apart.
- Vacuum distillation should be used if the boiling points are over 150 °C to prevent degradation of the product.
- Fractional distillation should be used if the boiling points are less than 25 °C apart because it allows more refined separation of liquids by boiling point.
Chromatography
- All forms of chromatography use two phases to separate compounds based on physical or chemical properties.
- The stationary phase or adsorbent is usually a polar solid.
- The mobile phase runs through the stationary phase and is usually a liquid or gas. This elutes the sample through the stationary phase.
- Compounds with higher affinity for the stationary phase have smaller retardation factors and take longer to pass through, if at all; compounds with higher affinity for the mobile phase elute through more quickly. Compounds, therefore, get separated from each other, which is called partitioning.
- Thin-layer and paper chromatography are used to identify a sample.
- The stationary phase is a polar material, such as silica, alumina, or paper.
- The mobile phase is a nonpolar solvent, which climbs the card through capillary action.
- The card is spotted and developed; Rf values can be calculated and compared to reference values.
- Reverse-phase chromatography uses a nonpolar card with a polar solvent.
- Column chromatography utilizes polarity, size, or affinity to separate compounds based on their physical or chemical properties.
- The stationary phase is a column containing silica or alumina beads.
- The mobile phase is a nonpolar solvent, which travels through the column by gravity.
- In ion-exchange chromatography, the beads are coated with charged substances to bind compounds with opposite charge.
- In size-exclusion chromatography, the beads have small pores which trap smaller compounds and allow larger compounds to travel through faster.
- In affinity chromatography, the column is made to have high affinity for a compound by coating the beads with a receptor or antibody to the compound.
- Gas chromatography separates vaporizable compounds according to how well they adhere to the adsorbent in the column.
- The stationary phase is a coil of crushed metal or a polymer.
- The mobile phase is a nonreactive gas.
- Gas chromatography may be combined in sequence with mass spectrometry, which ionizes and fragments molecules and passes these fragments through a magnetic field to determine molecular weight or structure.
- High-performance liquid chromatography (HPLC) is similar to column chromatography but uses sophisticated computer-mediated solvent and temperature gradients. It is used if the sample size is small or if forces such as capillary action will affect results. It was formerly called high-pressure liquid chromatography.
ANSWERS TO CONCEPT CHECKS
**12.1**
- The two solvents must be immiscible and must have different polarity or acid–base properties that allow a compound of interest to dissolve more easily in one than the other.
- It is better to do three washes with 10 mL than to do one with 30 mL; more of the compound of interest would be extracted with multiple sequential extractions than one large one.
- Acid dissolves better in an aqueous base because it will dissociate to form the conjugate base and, being more highly charged, will become more soluble. Note that like dissolves like applies to polarity; acids and bases dissolve more easily in solutions with the opposite acid–base characteristics.
**12.2**
- Distillation takes advantage of differences in boiling points in order to separate solutions of miscible liquids.
- A solution of ether and methylene chloride, which have very close boiling points, can be separated by using fractional distillation.
- Vacuum distillation would be the best technique to separate two chemicals with such high boiling points because the decreased ambient pressure will allow them to boil at a lower temperature.
**12.3**
- Each of these methods separates compounds using charge and polarity.
-
Type of Column Chromatography Method for Separating Sample Ion-exchange The column is given a charge, which attracts molecules with the opposite charge.
Size-exclusion Small pores are used; smaller molecules are trapped, while larger molecules pass through the column.
Affinity Specific receptors or antibodies can trap the target in the column; the target must then be washed out using other solutions.
- As the name suggests, gas chromatography is simply the same technique of mobile and stationary phases performed with a gaseous eluent (instead of liquid). The stationary phase is usually a crushed metal or polymer.
- Historically, HPLC was performed at high pressures, whereas column chromatography uses gravity to pull the solution through the column. Now, HPLC is performed with sophisticated and variable solvent and temperature gradients, allowing for much more specific separation of compounds than column chromatography; high pressures are no longer required.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. B
Fractional distillation is the most effective procedure for separating two liquids that boil within a few degrees of each other. Ethyl acetate and ethanol boil well within 25 °C of each other and thus would be good candidates for fractional distillation. Fractional distillation could also be used for the liquids in (C), but would require lower pressures because of their high boiling points.
2. C
By extracting with sodium hydroxide, benzoic acid will be converted to its sodium salt, sodium benzoate. Sodium benzoate, unlike its conjugate acid, will dissolve in an aqueous solution. The aqueous layer simply has to be acidified afterward to retrieve benzoic acid. (A) is incorrect because diethyl ether and tetrahydrofuran are both nonpolar and are miscible. Hydrochloric acid will not transform benzoic acid into a soluble salt, so (B) is incorrect. Finally, (D) is incorrect because protonated benzoic acid has limited solubility in water.
3. D
It is more effective to perform four successive extractions with small amounts of ether than to perform one extraction with a large amount of ether.
4. D
Hexane is less polar than ether and, therefore, is less likely to displace polar compounds adsorbed to the silica gel. This would decrease the distance these polar compounds would travel, decreasing Rf values.
5. D
In column chromatography, as in TLC, the less polar compound travels most rapidly. This means that 1-naphthalenemethanol, with the highest Rf value, would travel most rapidly and would be the first to elute from the column.
6. A
This is an example of reverse-phase chromatography. The solvent system is polar, which means that the most polar compound will travel the furthest up the card, resulting in the largest Rf. This gives compound III the largest Rf, which corresponds to spot A.
7. B
Because methylene chloride is denser than brine (salt water), the organic layer will settle at the bottom of the funnel. Methylene chloride is not soluble in water, eliminating (C).
8. B
Silica gels are polar. Polarity is used to selectively attract specific solutes within a nonpolar solvent phase. Although silica gels have other properties, this is the most important to TLC.
9. B
In this question, three substances must be separated using a combination of techniques. The first step should be the most obvious: remove the sand by filtration. The remaining compounds—benzoic acid and naphthalene—are still dissolved in ether. If the solution is extracted with an aqueous base, the benzoate anion is formed and becomes dissolved in the aqueous layer, while naphthalene, a nonpolar compound, remains in the ether. Finally, evaporation of the ether will yield purified naphthalene.
10. C
This is the only option that would be effectively separated by a simple distillation. (B) would require vacuum distillation because the boiling points are over 150 °C. In (A) and (D), the boiling points are within 25 °C of each other and would therefore require fractional distillation in order to be separated.
11. C
Affinity chromatography, using the target for the biological effector or a specific antibody, would work best in this case. It will specifically bind the protein of interest and keep it in the column.
12. D
Because this solution is composed of a much larger molecule and a much smaller molecule, size-exclusion chromatography would effectively remove the smaller insulin molecule into the fraction retained in the column and allow the titin to be eluted. Affinity chromatography, (C), could also be used, but comes with a risk of rendering the titin unusable; the eluent run through an affinity chromatography column often binds to the target molecule.
13. B
Each of these is the mobile phase of the system, in which the solutes are dissolved and move. The stationary phase in gas chromatography is usually a crushed metal or polymer; the stationary phase in paper chromatography is paper.
14. C
Warm or hot solvent is generally used in gravity filtration to keep the desired product soluble. This allows the product to remain in the filtrate, which can then be collected. In this case, the student likely used solvent that was too cold, and the product crystallized out. The product should be present in the residue.
15. B
Because the lactoferrin proteins are likely to be charged, as is the resin described in the question, this is an example of ion-exchange chromatography. The charged protein molecules will stick to the column, while the remainder of the milk washes through and can later be washed off of the column and collected.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
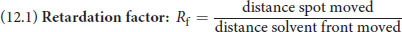
SHARED CONCEPTS
- Biochemistry Chapter 3
- Nonenzymatic Protein Function and Protein Analysis
- General Chemistry Chapter 7
- Thermochemistry
- General Chemistry Chapter 8
- The Gas Phase
- General Chemistry Chapter 9
- Solutions
- General Chemistry Chapter 10
- Acids and Bases
- Organic Chemistry Chapter 11
- Spectroscopy