Chapter 4: Carbohydrate Structure and Function

Chapter 4: Carbohydrate Structure and Function
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- When glucose is in a straight-chain formation, it:
- is an aldoketose.
- is a pentose.
- has five chiral carbons.
- is one of a group of 16 stereoisomers.
- All of the following are true of epimers EXCEPT:
- they differ in configuration about only one carbon.
- they usually have slightly different chemical and physical properties.
- they are diastereomers (with the exception of glyceraldehyde).
- they have equal but opposite optical activities.
- Aldonic acids are compounds that:
- can be oxidized, and therefore act as reducing agents.
- can be reduced, and therefore act as reducing agents.
- have been oxidized, and have acted as reducing agents.
- have been oxidized, and have acted as oxidizing agents.
- The formation of α-D-glucopyranose from β-D-glucopyranose is called:
- glycosidation.
- mutarotation.
- enantiomerization.
- racemization.
- Ketose sugars may have the ability to act as reducing sugars. Which process explains this?
- Ketose sugars undergo tautomerization.
- The ketone group is oxidized directly.
- Ketose sugars undergo anomerization.
- The ketone group is reduced directly.
- What is the product of the following reaction?
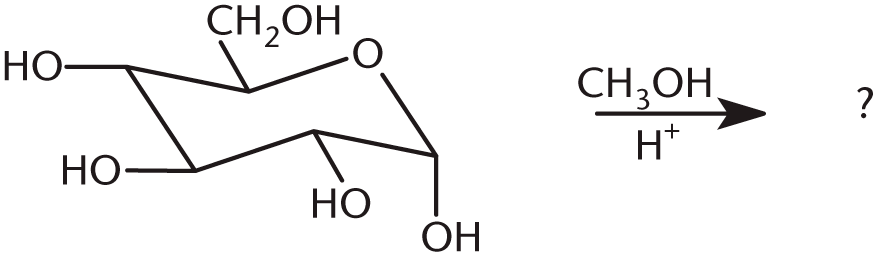
-
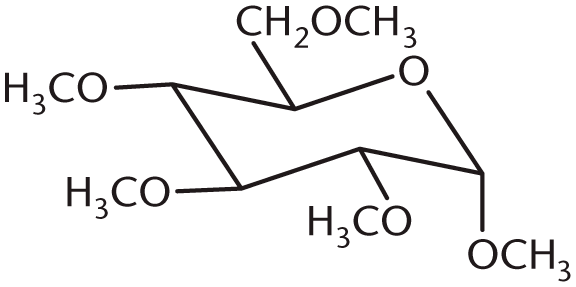
-
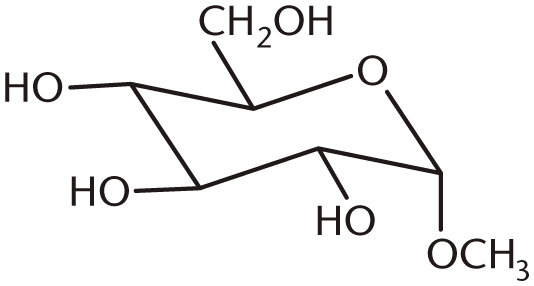
-
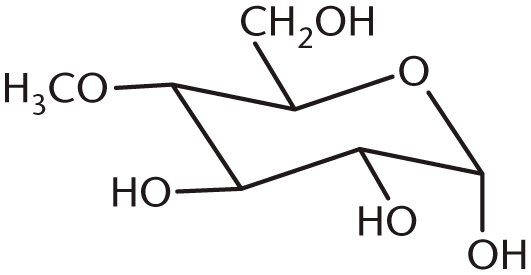
-
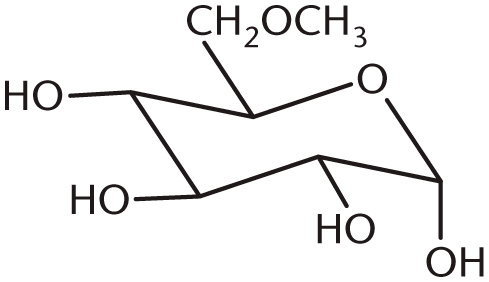
- Which of the following enzymes cleaves polysaccharide chains and yields maltose exclusively?
- α-Amylase
- β-Amylase
- Debranching enzyme
- Glycogen phosphorylase
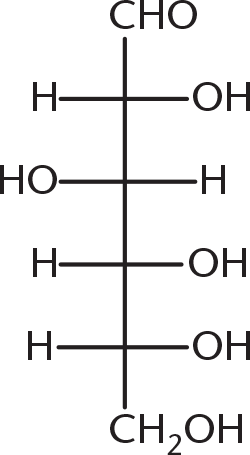
- When the following straight-chain Fischer projection is converted to a chair or ring conformation, its structure will be:
-
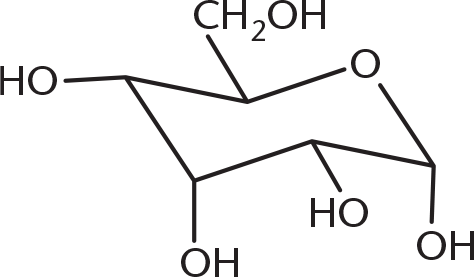
-
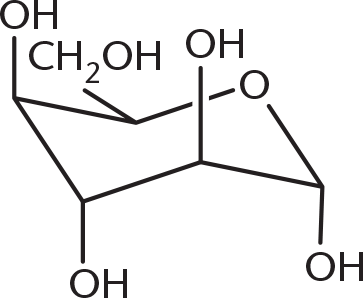
-
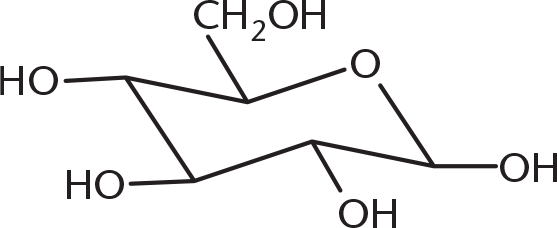
-
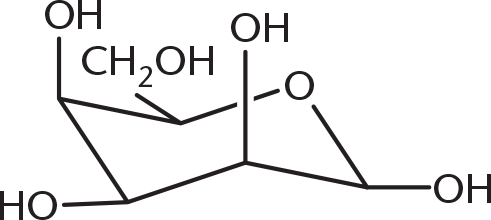
- Why is the α-anomer of D-glucose less likely to form than the β-anomer?
- The β-anomer is preferred for metabolism.
- The β-anomer undergoes less electron repulsion.
- The α-anomer is the more stable anomer.
- The α-anomer forms more in L-glucose.
- Which two polysaccharides share all of their glycosidic linkage types in common?
- Cellulose and amylopectin
- Amylose and glycogen
- Amylose and cellulose
- Glycogen and amylopectin
- Which of the following is digestible by humans and is made up of only one type of monosaccharide?
- Lactose
- Sucrose
- Maltose
- Cellobiose
- The reaction below is an example of one step in:
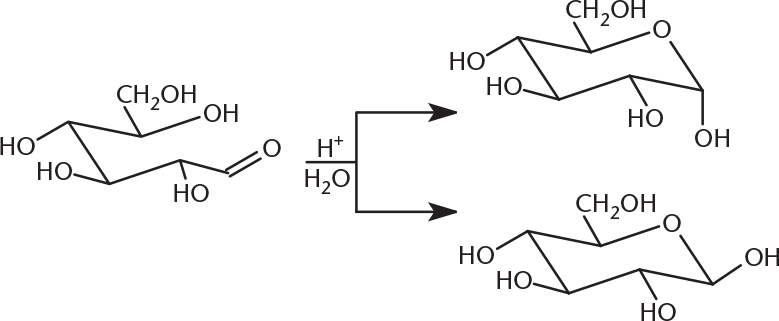
- aldehyde formation.
- hemiketal formation.
- mutarotation.
- glycosidic bond cleavage.
- Galactose is the C-4 epimer of glucose, the structure of which is shown below. Which of the following structures is galactose?
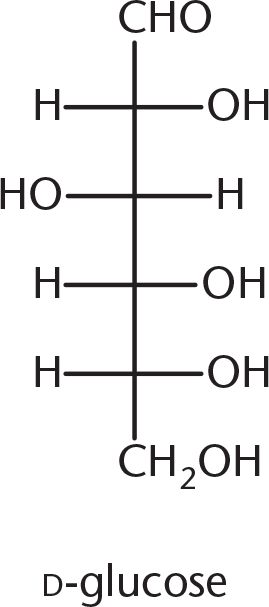
-
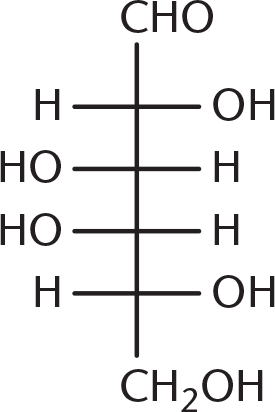
-
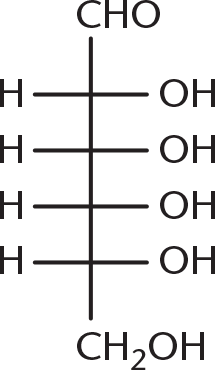
-
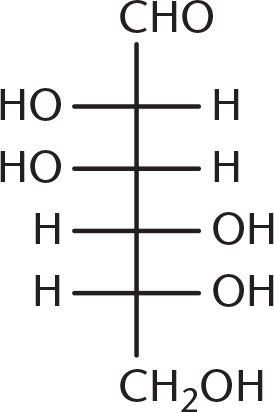
-
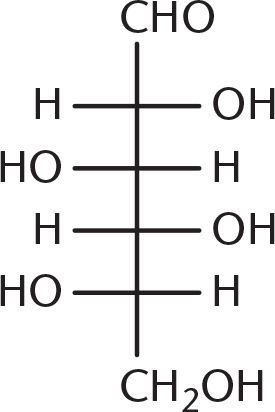
- Andersen’s disease (glycogen storage disease type IV) is a condition characterized by a deficiency in glycogen branching enzyme. Absence of this enzyme would be likely to cause all of the following effects EXCEPT:
- decreased glycogen solubility in human cells.
- slower action of glycogen phosphorylase.
- less storage of glucose in the body.
- glycogen devoid of α-1,4 linkages.
- The cyclic forms of monosaccharides are:
- hemiacetals.
- hemiketals.
- acetals.
- I only
- III only
- I and II only
- I, II, and III
Answer Key
- D
- D
- C
- B
- A
- B
- B
- C
- B
- D
- C
- C
- A
- D
- C
Chapter 4: Carbohydrate Structure and Function
CHAPTER 4
CARBOHYDRATE STRUCTURE AND FUNCTION
In This Chapter
**4.1 Carbohydrate Classification
**
Nomenclature
Stereochemistry
4.2 Cyclic Sugar Molecules
Hexose Conformations
Mutarotation
4.3 Monosaccharides
Oxidation and Reduction
Esterification
Glycoside Formation
4.4 Complex Carbohydrates
Disaccharides
Polysaccharides
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 4% of all questions about biochemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1D: Principles of bioenergetics and fuel molecule metabolism
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
Carbohydrates (or, as they are known colloquially, carbs) have experienced a tumultuous few decades in American culinary culture. Remember the food pyramid, which advised that we consume 6 to 11 servings of carbohydrates—in the form of bread, cereal, rice, and pasta—per day? Nowadays, we’re inundated with recommendations to cut down or even cut out carbs; still, carbohydrates make up most of the food and drink that continues to fill our refrigerators and cupboards. Whether or not this is healthy is debatable, as research on food choices continues. What is certain, however, is that carbohydrates are the most direct source of chemical energy for almost all organisms, ranging from single-celled protozoa to more complex organisms, such as plants and animals—including people.
From a molecular standpoint, carbohydrates used to be defined by the empirical formula Cn(H2O)n, but this definition is now considered antiquated. This formula only applies to simple monomeric sugars, which are also called monosaccharides. As simple sugars link to form complex sugars, water loss occurs, thus changing the empirical formula to Cn(H2O)m for complex sugars.
In this chapter, we’ll discuss how the various types of carbohydrates we utilize for metabolism are classified, the structures in which they exist, and the biochemical reactions they undergo.
4.1 Carbohydrate Classification
LEARNING OBJECTIVES
After Chapter 4.1, you will be able to:
- Recognize common features of sugar nomenclature
- Apply principles of stereoisomerism to sugar nomenclature and structure
- Distinguish enantiomers from epimers
- Classify and name a simple sugar based on its structure
Carbohydrates come in many types. They can be classified by the number of sugar moieties that make them up, the number of carbons in each sugar, the functional groups present on the molecule, and the stereochemistry of the sugar.
Nomenclature
Whenever we discuss carbohydrates (or anything else in biology), it makes sense to start with the most basic structural units, which are monosaccharides. The simplest monosaccharides contain three carbon atoms and are called trioses. Carbohydrates with four, five, and six carbon atoms are called tetroses, pentoses, and hexoses, respectively. Carbohydrates that contain an aldehyde group as their most oxidized functional group are called aldoses and those with a ketone group as their most oxidized functional group are called ketoses. Taken together, a six-carbon sugar with an aldehyde group would be called an aldohexose, while a five-carbon sugar with a ketone group would be called a ketopentose. The basic structure of a monosaccharide is illustrated by the simple sugar glyceraldehyde, which is an aldose as shown in Figure 4.1.
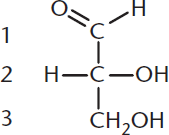
Figure 4.1 Glyceraldehyde The simplest aldose (an aldotriose)
MCAT EXPERTISE
The MCAT likes to present complex, novel molecules and then test you on the most basic information about them. Therefore, when dealing with carbohydrates on the exam, look for the functional groups we have seen before (aldehydes, ketones, and alcohols) and realize that they retain the same chemical properties that you already know.
Glyceraldehyde is a polyhydroxylated aldehyde, or as described above, an aldose (aldehyde sugar). The numbering of carbon atoms in a monosaccharide follows the rules described in Chapter 1 of MCAT Organic Chemistry Review. The carbonyl carbon is the most oxidized, and therefore will always have the lowest possible number. In an aldose, the aldehyde carbon will always be carbon number one (C-1). The aldehyde carbon can participate in glycosidic linkages; sugars acting as substituents via this linkage are called glycosyl residues.
The simplest ketone sugar (ketose) is dihydroxyacetone, shown in Figure 4.2. Again, the carbonyl carbon is the most oxidized; in this case, the lowest number it can be assigned is carbon number two (C-2). This is true, in fact, for most ketoses on the MCAT: the carbonyl carbon is C-2. Ketoses can also participate in glycosidic bonds at this carbon. Notice that on every monosaccharide, every carbon other than the carbonyl carbon will carry a hydroxyl group.
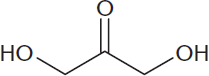
Figure 4.2 Dihydroxyacetone The simplest ketose (a ketotriose)
Common Names
On the MCAT, a few sugars are tested by referencing their common names, or names that do not necessarily follow the nomenclature rules listed above. You should be familiar with the names of the important monosaccharides listed in Figure 4.3.
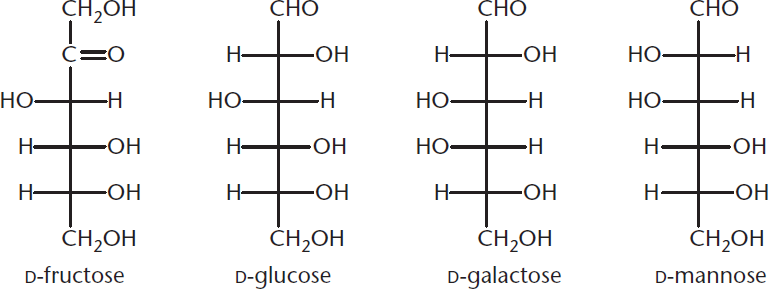
Figure 4.3 Common Names of Frequently Tested Sugars on the MCAT
BIOCHEMISTRY GUIDED EXAMPLE WITH EXPERT THINKING
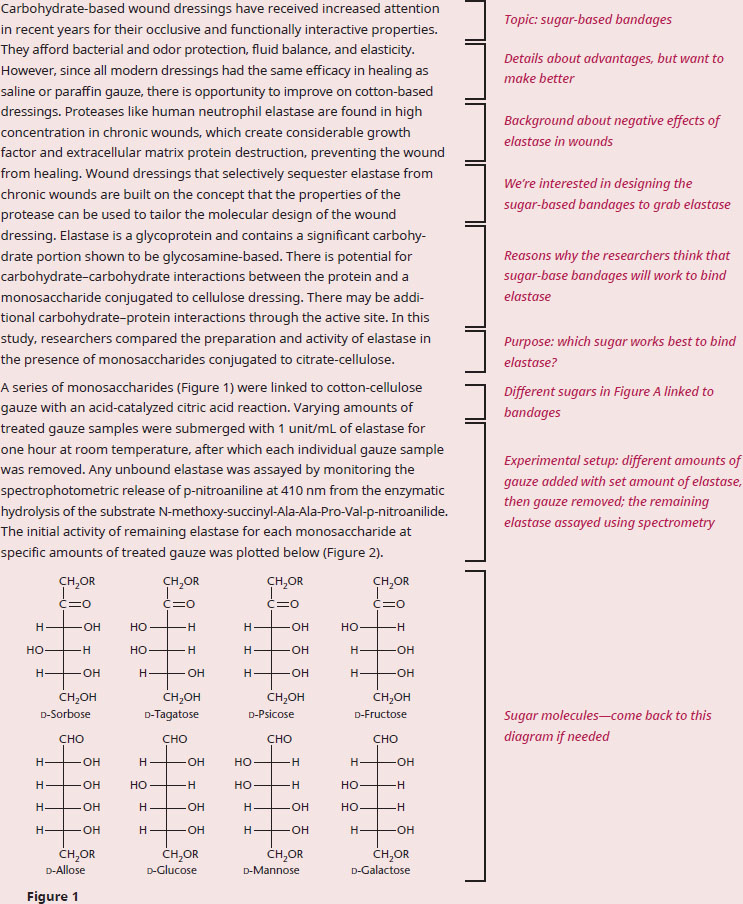
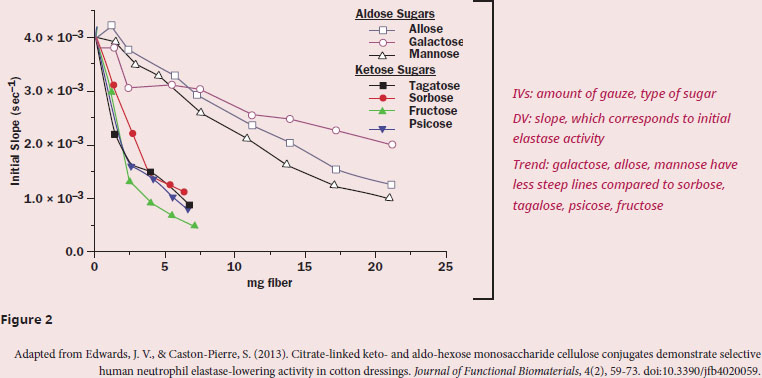
According to the data, which class of monosaccharide should be used in gauze to help prevent elastase-mediated wound damage?
The question asks us to select a type of monosaccharide to prevent elastase from doing damage, which means that it’s important to understand the purpose of the passage and what the data tells us. The first paragraph describes how advantageous bandages currently are, but that researchers are interested in improving them by selectively binding proteases that prevent healing, like elastase.
Let's take a look at the experimental setup in paragraph 2. Since the sugar-linked bandages are soaked in a solution with elastase and then removed, the assumption is that the better the sugar binds elastase, the less elastase there is left over in the solution when the researchers assay it, after the bandage is removed Therefore, the better the binding, the less activity there should be. The figure clearly has two sets of lines: galactose/allose/mannose have activity over a larger amount of fiber used, and, based on Figure 1, all three of these sugars are aldohexoses, which are sugars with an aldehyde at the C-1 position. Sorbose/tagatose/psicose/fructose, however, quickly drop in activity with increasing amounts of fiber, and, based on Figure 1, all four of these sugars are ketohexoses, which are sugars with a ketone at the C-2 position. To make the comparison a bit more clear, let's consider the activity for each set at 5 mg of fiber—ketohexoses have ~1.5×10–3 slope, and aldohexoses have ~3.3×10–3 slope. Since slope corresponds to activity, less activity is seen for ketohexoses with equivalent amounts of carbohydrate-bound bandage, therefore corresponding to more elastase binding.
Therefore, we can conclude that the researchers should investigate the usage of ketohexoses to help prevent elastase-mediated wound damage, since this class of sugar seems to bind elastase better than aldohexoses.
Stereochemistry
Optical isomers, also called stereoisomers, are compounds that have the same chemical formula; these molecules differ from one another only in terms of the spatial arrangement of their component atoms. A special type of isomerism exists between stereoisomers that are nonidentical, nonsuperimposable mirror images of each other. These molecules are called enantiomers. A chiral carbon atom is one that has four different groups attached to it; any molecule that contains chiral carbons and no internal planes of symmetry has an enantiomer.
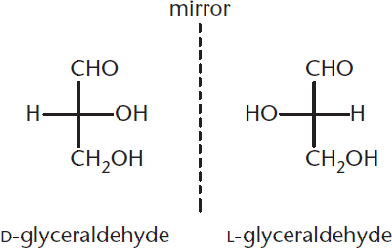
Figure 4.4 Enantiomers of Glyceraldehyde
BRIDGE
Isomerism is discussed in detail in Chapter 2 of *MCAT Organic Chemistry** Review*. The isomerism seen in carbohydrates is just an extension of that conversation; many sugars are related to each other as enantiomers or diastereomers.
Figure 4.4 illustrates the two enantiomers of glyceraldehyde: D- and L-glyceraldehyde. The particular three-dimensional arrangement of the groups attached to the chiral carbon determines the compound’s absolute configuration. While organic chemists use the newer (R) and (S) system when denoting absolute configuration, biochemists use the older D and L system. Notice in the figure above that D-glyceraldehyde and L-glyceraldehyde are mirror images of one another—this makes them enantiomers because they must have opposite absolute configurations. Because there is only one chiral carbon present, these are the only two stereoisomers that exist for glyceraldehyde. As the number of chiral carbons increases, so too does the number of possible stereoisomers because one compound may have many diastereomers. The number of possible stereoisomers of a compound can be calculated by:
Number of stereoisomers with common backbone = 2n
Equation 4.1
where n is the number of chiral carbons in the molecule.
KEY CONCEPT
The two systems for naming chiral carbons are not interchangeable! While some D-isomers are equivalent with (R), others are (S). When dealing with biomolecules like sugars, anticipate using the biochemists’ method of nomenclature: D and L.
KEY CONCEPT
When trying to figure out how many possible stereoisomers can exist for a multi-carbon compound, identify the number of chiral carbons (n) and plug into the formula 2n. For example, 21 = 2 stereoisomers and 22 = 4 stereoisomers.
Early in the twentieth century, scientists used glyceraldehyde to learn about the optical rotation of sugars. The results of this early study led to the d and l naming convention. D-Glyceraldehyde was later determined to exhibit a positive rotation (designated as D-(+)-glyceraldehyde), and L-glyceraldehyde a negative rotation (designated as L-(–)-glyceraldehyde). Note that the direction of rotation, (+) or (–), must be determined experimentally and cannot be determined from the D or L designation for the sugar.
KEY CONCEPT
D and L are based on the stereochemistry of glyceraldehyde. These are NOT directly related to the (+) and (–) designations denoting optical rotation.
On the MCAT, all monosaccharides are assigned the D or l configuration based on their relationship to glyceraldehyde. The Fischer projection is a simple two-dimensional drawing of stereoisomers. Recall that in a Fischer projection, the horizontal lines are wedges (out of the page), while vertical lines are dashes (into the page), as shown in Figure 4.5.

Figure 4.5 Fisher Projection Horizontal lines are wedges (out of the page); vertical lines are dashes (into the page).
Fischer projections allow scientists to identify different enantiomers. Using this system of structural representation, all D-sugars have the hydroxide of their highest-numbered chiral center on the right, and all L-sugars have that hydroxide on the left. Because D-glucose and L-glucose are enantiomers, this means that every chiral center in D-glucose has the opposite configuration of L-glucose, as shown in Figure 4.6.
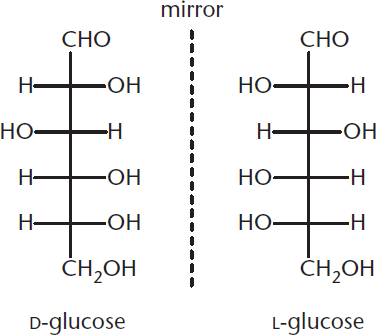
Figure 4.6 Enantiomers of Glucose
Make sure that you are familiar with these three types of stereoisomers:
- The same sugars, in different optical families, are enantiomers (such as D-glucose and L-glucose).
- Two sugars that are in the same family (both are either ketoses or aldoses, and have the same number of carbons) that are not identical and are not mirror images of each other are diastereomers.
- A special subtype of diastereomers are those that differ in configuration at exactly one chiral center. These are defined as epimers (such as D-ribose and D-arabinose, which only differ at C-2, as shown in Figure 4.7).
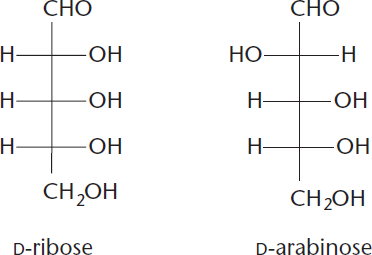
Figure 4.7 D-Ribose and D-Arabinose Are C-2 Epimers
A summary of these different types of isomers is provided in Figure 4.8, which shows four different stereoisomers of an aldotetrose, including two optical forms of erythrose and two optical forms of threose.
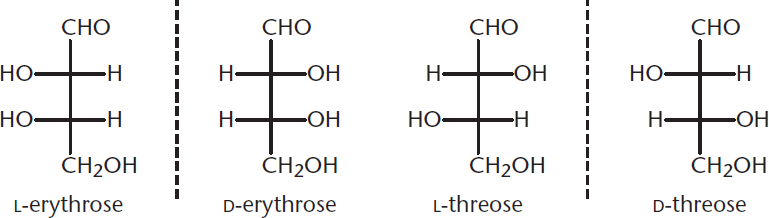
Figure 4.8 Four Stereoisomers of an Aldotetrose
Because D-erythrose and L-erythrose are nonsuperimposable mirror images of one another, they are enantiomers. On the other hand, while D-erythrose and D-threose are not mirror images of one another, they are still nonsuperimposable, which makes them diastereomers. Similarly, L-erythrose and D-threose are diastereomers. Because they differ in configuration at only one chiral center, they can also be defined as epimers of one another. Remember, a compound can have only one enantiomer (the left hand to its right hand, or vice versa), but may have multiple diastereomers, depending on how many (and which) chiral carbons are inverted between the two molecules.
MCAT CONCEPT CHECK 4.1:
Before you move on, assess your understanding of the material with these questions.
- What is the name for a five-carbon sugar with an aldehyde group? A six-carbon sugar with a ketone group?
________________________________________
- Draw all of the possible D-stereoisomers of glucose in Fischer projection form.
- Which of the diastereomers of glucose from the previous question are considered to be epimers of glucose? Enantiomers?
- Epimers:
________________________________________
- Enantiomers:
________________________________________
4.2 Cyclic Sugar Molecules
LEARNING OBJECTIVES
After Chapter 4.2, you will be able to:
- Convert between Haworth and Fischer projections
- Define and explain sugar-related concepts, including hemiacetal, hemiketal, pyranose, furanose, and anomeric carbon
- Predict the impact of mutarotation on conformation
- Identify the more stable anomer of a given sugar:
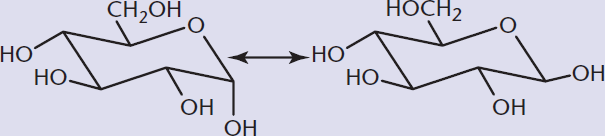
Monosaccharides contain both a hydroxyl group, which can serve as a nucleophile, and a carbonyl group, which is the most common electrophile on the MCAT. Therefore, they can undergo intramolecular reactions to form cyclic hemiacetals (from aldoses) and hemiketals (from ketoses). Due to ring strain, the only cyclic molecules that are stable in solution are six-membered pyranose rings or five-membered furanose rings. In fact, such sugars tend to exist predominantly in cyclic form. The hydroxyl group acts as the nucleophile during ring formation, so oxygen becomes a member of the ring structure. Regardless of whether hemiacetal or hemiketal is formed, the carbonyl carbon becomes chiral in this process, and is referred to as the anomeric carbon. Figure 4.9 demonstrates how the carbonyl containing C-1 and the hydroxyl group on C-5 of D-glucose undergo intramolecular hemiacetal formation. One of two ring forms can emerge during cyclization of a sugar molecule: α or β. Because these two molecules differ at the anomeric carbon, they are termed anomers of one another. In glucose, the α-anomer has the –OH group of C-1 trans to the –CH2OH substituent (axial and down), whereas the β-anomer has the –OH group of C-1 cis to the –CH2OH substituent (equatorial and up).
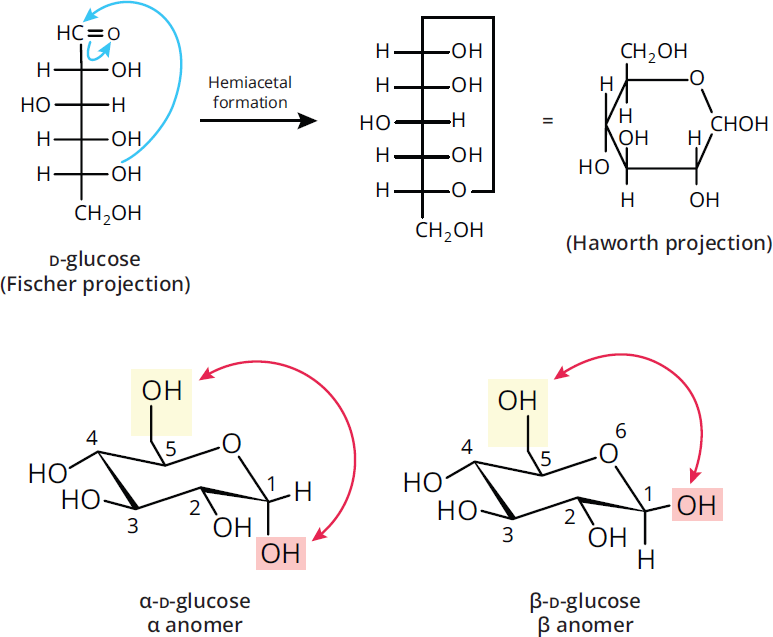
Figure 4.9 Cyclic Sugar Formation via Intramolecular Nucleophilic Addition Glucose forms a six-membered ring with two anomeric forms:α(left) andβ(right).
Hexose Conformations
Note how Figure 4.9 above has two kinds of projections for glucopyranose: the Haworth projection and the Fischer projection. The Haworth projection is a useful method for describing the three-dimensional conformations of cyclic structures. Haworth projections depict cyclic sugars as planar five- or six-membered rings with the top and bottom faces of the ring nearly perpendicular to the page. In reality the five-membered rings are very close to planar, but the pyranose rings adopt a chair-like configuration, and the substituents assume axial or equatorial positions to minimize steric hindrance. When we convert the monosaccharide from its straight-chain Fischer projection to the Haworth projection, any group on the right in the Fischer projection will point down.
Mutarotation
Exposing hemiacetal rings to water will cause them to spontaneously cycle between the open and closed form. Because the substituents on the single bond between C-1 and C-2 can rotate freely, either the α- or β-anomer can be formed, as demonstrated in Figure 4.10. This spontaneous change of configuration about C-1 is known as mutarotation, and occurs more rapidly when the reaction is catalyzed with an acid or base. Mutarotation results in a mixture that contains both α- and β-anomers at equilibrium concentrations (for glucose: 36% α, 64% β). In solution, the α-anomeric configuration is less favored because the hydroxyl group of the anomeric carbon is axial, adding to the steric strain of the molecule. In its solid state (not in solution), this preference can be mitigated by the anomeric effect, which helps stabilize the α-anomer, although this is outside the scope of the MCAT.
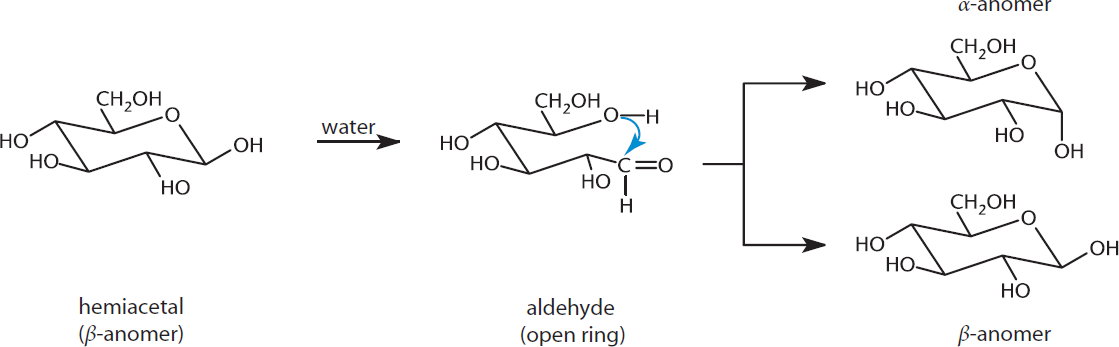
Figure 4.10 Mutarotation Interconversion between theα- andβ-anomers via ring opening and reclosing
MCAT CONCEPT CHECK 4.2:
Before you move on, assess your understanding of the material with these questions.
-
Explain the relationship between the carbonyl carbon, anomeric carbon, and the alpha and beta forms of a sugar molecule.
________________________________________
- Draw the less stable anomer of D-glucose in Haworth projection form.
- Draw the less stable anomer of D-glucose in chair configuration.
4.3 Monosaccharides
LEARNING OBJECTIVES
After Chapter 4.3, you will be able to:
- Predict the products of sugar reactions, including oxidation, reduction, esterification, and glycosidic linkage formation
- Contrast Tollens' reagent and Benedict's reagent
- Apply reactions of sugars to biological contexts
Monosaccharides contain alcohols and either aldehydes or ketones. As such, these functional groups undergo the same reactions that they do when present in other compounds. These include oxidation and reduction, esterification, and nucleophilic attack (creating glycosides).
Oxidation and Reduction
One of the most important biochemical reactions in the human body is the oxidation of carbohydrates in order to yield energy. As monosaccharides switch between anomeric configurations, the hemiacetal rings spend a short period of time in the open-chain aldehyde form. Just like other aldehydes, they can be oxidized to carboxylic acids; these oxidized aldoses are called aldonic acids. Because aldoses can be oxidized, they are considered reducing agents. Therefore, any monosaccharide with a hemiacetal ring is considered a reducing sugar. When the aldose in question is in ring form, oxidation yields a lactone instead—a cyclic ester with a carbonyl group persisting on the anomeric carbon, as shown in Figure 4.11. Lactones, such as vitamin C, play an essential role in the human body.
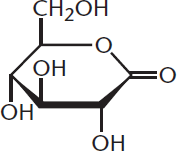
Figure 4.11 Lactone Contains a cyclic ester
Two standard reagents are used to detect the presence of reducing sugars: Tollens' reagent and Benedict’s reagent. Tollens' reagent must be freshly prepared, starting with silver nitrate (AgNO3), which is mixed with NaOH to produce silver oxide (Ag2O). Silver oxide is dissolved in ammonia to produce [Ag(NH3)2]+, the actual Tollens' reagent. Tollens' reagent is reduced to produce a silvery mirror when aldehydes are present. When Benedict’s reagent is used, the aldehyde group of an aldose is readily oxidized, indicated by a red precipitate of Cu2O, as demonstrated in Figure 4.12. To test specifically for glucose, one may utilize the enzyme glucose oxidase, which does not react with other reducing sugars. A more powerful oxidizing agent, such as dilute nitric acid, will oxidize both the aldehyde and the primary alcohol (on C-6) to carboxylic acids.
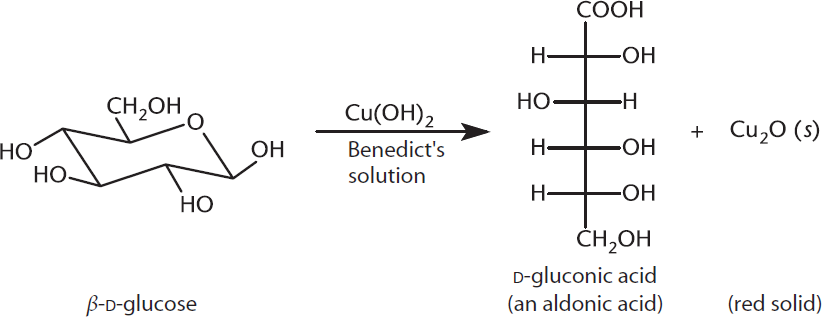
Figure 4.12 Positive Test for an Aldose Using Benedict’s Reagent Aldoses will react, forming copper(I) oxide; ketones may react more slowly.
An interesting phenomenon is that ketose sugars like fructose are also reducing sugars and give positive Tollens’ and Benedict’s tests. Although ketones cannot be oxidized directly to carboxylic acids, they can tautomerize to form aldoses under basic conditions, via keto–enol shifts. While in the aldose form, they can react with Tollens’ or Benedict’s reagents to form the carboxylic acid. Tautomerization refers to the rearrangement of bonds in a compound, usually by moving a hydrogen and forming a double bond. The tautomerization of fructose to glucose is shown in Figure 4.13. The ketone group picks up a hydrogen while the double bond is moved between two adjacent carbons, resulting in an enol: a compound with a double bond and an alcohol group. In this case, an enediol is the intermediate, which contains an alcohol on each of the two carbons. The enediol spontaneously rearranges to form glucose, an aldose.
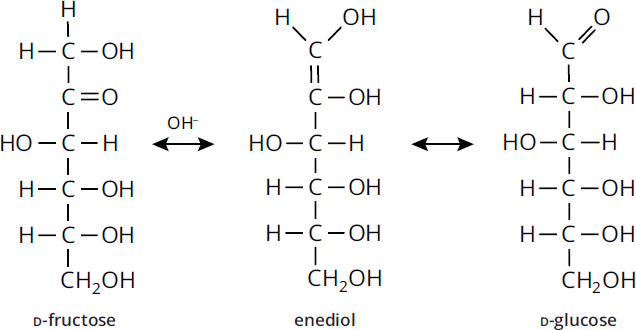
Figure 4.13 Tautomerization of Fructose to Glucose
Reduced sugars also play an essential role in human biochemistry. When the aldehyde group of an aldose is reduced to an alcohol, the compound is considered an alditol. A deoxy sugar, on the other hand, contains a hydrogen that replaces a hydroxyl group on the sugar. The most well-known of these sugars is D-2-deoxyribose, the carbohydrate found in DNA.
Esterification
Because carbohydrates have hydroxyl groups, they are able to participate in reactions with carboxylic acids and carboxylic acid derivatives to form esters, as shown in Figure 4.14.
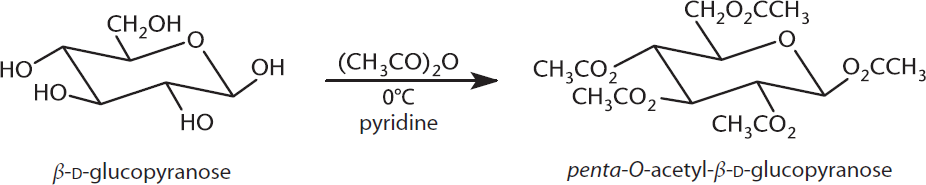
Figure 4.14 Esterification of Glucose Acetic anhydride used as carboxylic acid derivative
In the body, esterification is very similar to the phosphorylation of glucose, in which a phosphate ester is formed. Phosphorylation of glucose is an extremely important metabolic reaction of glycolysis in which a phosphate group is transferred from ATP to glucose, thus phosphorylating glucose while forming ADP, as shown in Figure 4.15. Hexokinase (or glucokinase, in the liver and pancreatic β-islet cells) catalyzes this reaction.
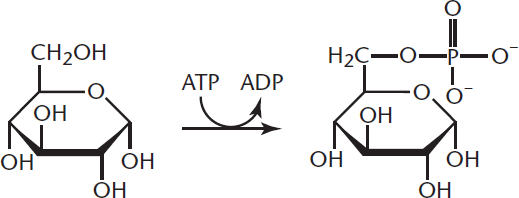
Figure 4.15 Phosphorylation of Glucose
BRIDGE
The action of hexokinase and glucokinase (as well as all the key glycolytic enzymes) is discussed in Chapter 9 of MCAT Biochemistry Review.
Glycoside Formation
Hemiacetals are the anomeric carbons that result when sugars undergo intramolecular reactions. Hemiacetals can react with alcohols to form acetals. The anomeric hydroxyl group is transformed into an alkoxy group, yielding a mixture of α- and β-acetals (with water as a leaving group). The resulting carbon–oxygen (C–O) bonds are called glycosidic bonds, and the acetals formed are glycosides. An example is the reaction of glucose with ethanol shown in Figure 4.16. Equivalent reactions happen with hemiketals, forming ketals.
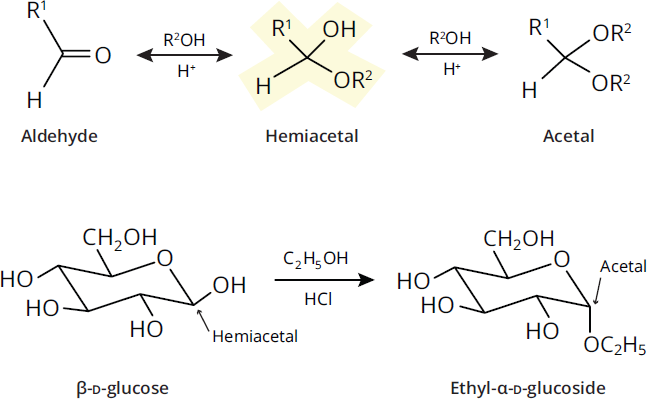
Figure 4.16 Glycosidic Linkage Formation Hemiacetal (or hemiketal) sugars react with alcohols under acidic conditions to form acetals (or ketals).
Disaccharides and polysaccharides form as a result of glycosidic bonds between monosaccharides. Glycosides derived from furanose rings are referred to as furanosides and those derived from pyranose rings are called pyranosides. Note that glycoside formation is a dehydration reaction; thus, breaking a glycosidic bond requires hydrolysis.
MCAT CONCEPT CHECK 4.3:
Before you move on, assess your understanding of the material with these questions.
- Explain the difference between esterification and glycoside formation.
________________________________________
- What purpose do Tollens' reagent and Benedict's reagent serve? How do they differ from each other?
________________________________________
- From a metabolic standpoint, does it make sense for carbohydrates to get oxidized or reduced? What is the purpose of this process?
________________________________________
4.4 Complex Carbohydrates
LEARNING OBJECTIVES
After Chapter 4.4, you will be able to:
- Compare starches, glycogen, and cellulose
- Predict the comparative solubility of different starch forms based on structure
- Recognize important biologically relevant disaccharides
Complex carbohydrates include all carbohydrates with at least two sugar molecules linked together (disaccharides, oligosaccharides, and polysaccharides).
Disaccharides
As discussed previously, monosaccharides react with alcohols to form acetals. Glycosidic bonds formed between hydroxyl groups of two monosaccharides result in the formation of a disaccharide, as shown in Figure 4.17.
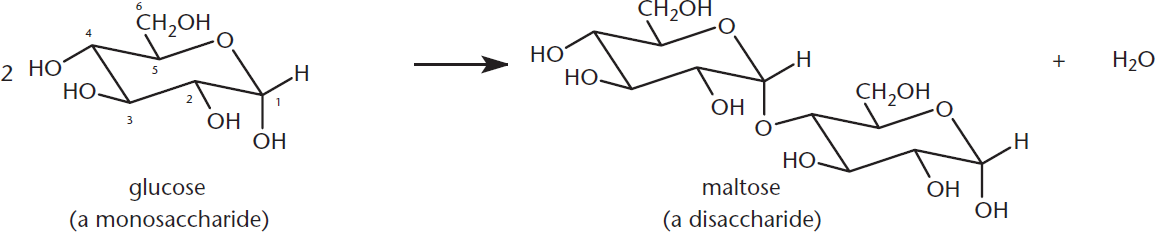
Figure 4.17 Disaccharide Formation The hydroxyl group on the anomeric carbon reacts with the hydroxyl of another sugar to form an acetal (or ketal) with a 1,2; 1,4; or 1,6 glycosidic linkage.
Formation of an α- or β-glycosidic linkage is nonspecific in that the anomeric carbon of a cyclic sugar can react with any hydroxyl group on any other sugar molecule. The linkages are named for the configuration of the anomeric carbon and the numbers of the hydroxyl-containing carbons involved in the linkage. For example, in an α-1,6 glycosidic bond formation between two D-glucose molecules, the α-anomeric carbon of the first glucose (C-1) attaches to C-6 of the second glucose. Note that the second glucose could be either the α- or β-anomer. In the event that a glycosidic bond is formed between two anomeric carbons, this must be specified in the name. For example, a bond formed between the anomeric carbons of two α-D-glucose molecules would be termed an α,α-1,1 linkage, as demonstrated in Figure 4.18.
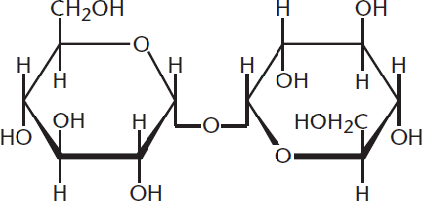
Figure 4.18 Trehalose Example of a disaccharide with anα,α-1,1 linkage between theα-anomeric carbons of two glucose molecules
Various combinations of monosaccharides linked by glycosidic bonds result in the formation of different disaccharides. For instance, two glucose molecules linked by an α-1,4 glycosidic bond is called maltose, while two glucose molecules joined by a β-1,4 linkage is called cellobiose.
Important Disaccharides
When discussing disaccharides in a real-world context, the most important of these sugars are sucrose, lactose, and maltose. These disaccharides are commonly produced in the cell by enzymatic activity. Their molecular structures and linkages are highlighted in Figure 4.19.
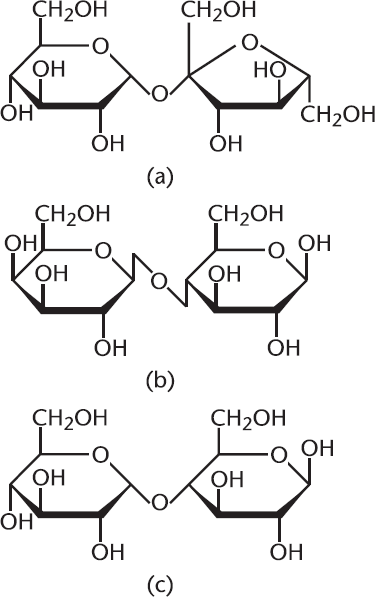
Figure 4.19 Important Disaccharides (a) Sucrose (glucose-α-1,2-fructose), (b) Lactose (galactose-β-1,4-glucose), (c) Maltose (glucose-α-1,4-glucose)
Polysaccharides
Polysaccharides are long chains of monosaccharides linked together by glycosidic bonds. While glucose is the most frequently encountered monosaccharide, it is not the only one. A polysaccharide composed entirely of glucose (or any other monosaccharide) is referred to as a homopolysaccharide, while a polymer made up of more than one type of monosaccharide is considered a heteropolysaccharide. The three most important biological polysaccharides are cellulose, starch, and glycogen. Although these three polysaccharides have different functions, they are all composed of the same monosaccharide, D-glucose. These polysaccharides differ in configuration about the anomeric carbon and the position of glycosidic bonds, resulting in notable biological differences.
Because glycosidic bonding can occur at multiple hydroxyl groups in a monosaccharide, polymer formation can either be linear or branched. Branching happens when an internal monosaccharide in a polymer chain forms at least two glycosidic bonds, allowing branch formation. We’ll take a closer look at how this is a key part of glycogen synthesis and glycogenolysis in Chapter 9 of MCAT Biochemistry Review.
Cellulose
Cellulose is the main structural component of plants. A homopolysaccharide, cellulose is a chain of β-D-glucose molecules linked by β-1,4 glycosidic bonds, with hydrogen bonds holding the actual polymer chains together for support. Humans are not able to digest cellulose because we lack the cellulase enzyme responsible for hydrolyzing cellulose to glucose monomers. Therefore, cellulose found in fruits and vegetables serves as a great source of fiber in our diet, drawing water into the gut. Cellulase is produced by some bacteria found in the digestive tract of certain animals, such as termites, cows, and goats, which enables them to digest cellulose. A portion of a cellulose chain can be seen in Figure 4.20 below.
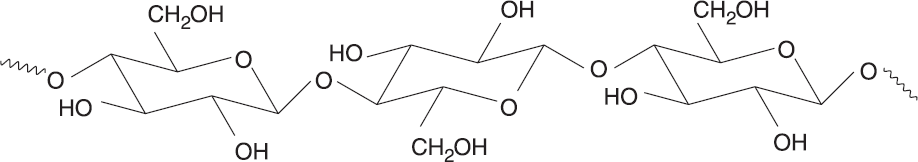
Figure 4.20 Cellulose Structure Cellulose is a polymer of 1,4-linkedβ-D-glucose.
Starches
Starches are polysaccharides that are more digestible by humans because they are linked α-D-glucose monomers. Plants predominantly store starch as amylose, a linear glucose polymer linked via α-1,4 glycosidic bonds. Another type of starch is amylopectin, which starts off with the same type of linkage that amylose exhibits, but also contains branches via α-1,6 glycosidic bonds. Iodine is a well-known reagent that tests for the presence of starch and does so by fitting inside the helix conformation amylose typically makes, forming a starch–iodine complex. The structure of amylose is depicted in Figure 4.21.
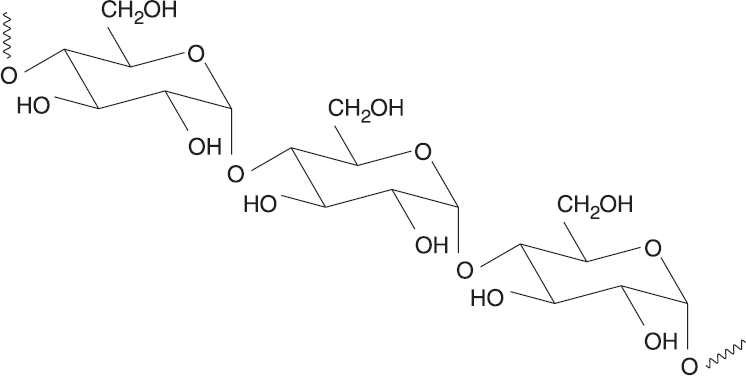
Figure 4.21 Starch Structure Starches are polymers of 1,4-linkedα-D-glucose.
BRIDGE
The contrast between cellulose and starch digestibility exemplifies the specificity of enzymes. A slight 109.5° rotation of the anomeric carbon to form β-linkages (instead of α-linkages) is enough to make the molecule indigestible by any enzymes in humans—even though they’re both D-glucose polymers! Enzyme specificity is discussed in Chapter 2 of MCAT Biochemistry Review.
Starches like amylose and amylopectin are broken down by enzymes in the body and are used as a source of energy. Amylose is degraded by α-amylase and β-amylase. β-Amylase cleaves amylose at the nonreducing end of the polymer (the end with acetal) to yield maltose, while α-amylase cleaves randomly along the chain to yield shorter polysaccharide chains, maltose, and glucose. Because amylopectin is highly branched, debranching enzymes help degrade the polysaccharide chain.
Glycogen
Glycogen is a carbohydrate storage unit in animals. It is similar to starch, except that it has more α-1,6 glycosidic bonds (approximately one for every 10 glucose molecules, while amylopectin has approximately one for every 25), which makes it a highly branched compound. This branching optimizes the energy efficiency of glycogen and makes it more soluble in solution, thereby allowing more glucose to be stored in the body. Also, its branching pattern allows enzymes that cleave glucose from glycogen, such as glycogen phosphorylase, to work on many sites within the molecule simultaneously. Glycogen phosphorylase functions by cleaving glucose from the nonreducing end of a glycogen branch and phosphorylating it, thereby producing glucose 1-phosphate, which plays an important role in metabolism.
REAL WORLD
Hers disease, also known as glycogen storage disease (GSD) type VI, is a condition characterized by a deficiency in liver glycogen phosphorylase. Patients with this disease cannot break down glycogen in their livers and therefore have hepatomegaly (a swollen liver). They may also have hypoglycemia (low blood sugar) between meals because they cannot use glycogen to maintain blood glucose concentrations.
MCAT CONCEPT CHECK 4.4:
Before you move on, assess your understanding of the material with these questions.
- Which of the two forms of starch is more soluble in solution. Why?
________________________________________
- Regarding glycogen and amylopectin, which of these two polymers should experience a higher rate of enzyme activity from enzymes that cleave side branches? Why?
________________________________________
Conclusion
This chapter examined, in depth, the unique characteristics of carbohydrates. Monosaccharides are the most basic form of carbohydrates, and in terms of human biochemistry, they typically range from three to seven carbon atoms. Classifying these monomers depends on the number of chiral centers present, which tells us the number of potential stereoisomers. Open-chain structures are most easily represented through Fischer projection diagrams; however, sugars tend to exist in ring form in biological systems, so Haworth projections are also used to depict three-dimensional structure. The most important reactions monosaccharides undergo are redox reactions, esterification, and glycoside formation—particularly when glycoside formation results in the formation of disaccharides. Polysaccharides are formed by glycosidic bonding of carbohydrates, and the polymers cellulose, starch, and glycogen are most commonly found in nature. Glycogen is the primary storage form of glucose in humans and other animals, and its unique branching structure allows for rapid access to these glucose stores.
The body has two primary energy-storage molecules: glycogen and triacylglycerols. Each has its own pros and cons—glycogen is more rapidly mobilized, but requires water of hydration, which increases its weight. Triacylglycerols serve as a long-term repository of energy, but take time to utilize. In the next chapter, we turn our attention to triacylglycerols, as well as the lipids used for structure and cell signaling.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Carbohydrate Classification
- Carbohydrates are organized by their number of carbon atoms and functional groups.
- Common names are also frequently used when referring to sugars, such as glucose, fructose, and galactose.
- Three-carbon sugars are trioses, four-carbon sugars are tetroses, and so on.
- Sugars with aldehydes as their most oxidized group are aldoses; sugars with ketones as their most oxidized group are ketoses.
- The nomenclature of all sugars is based on the D- and L-forms of glyceraldehyde. Sugars with the highest-numbered chiral carbon with the –OH group on the right (in a Fischer projection) are D-sugars; those with the –OH on the left are L-sugars. D- and L-forms of the same sugar are enantiomers.
- Diastereomers are nonsuperimposable configurations of molecules with similar connectivity. They differ at at least one—but not all—chiral carbons. These also include epimers and anomers.
- Epimers are a subtype of diastereomers that differ at exactly one chiral carbon.
- Anomers are a subtype of epimers that differ at the anomeric carbon.
Cyclic Sugar Molecules
- Cyclization describes the ring formation of carbohydrates from their straight-chain forms.
- When rings form, the anomeric carbon can take on either an α- or β-conformation.
- The anomeric carbon is the new chiral center formed in ring closure; it was the carbon containing the carbonyl in the straight-chain form.
- α-anomers have the –OH on the anomeric carbon trans to the free –CH2OH group.
- β-anomers have the –OH on the anomeric carbon cis to the free –CH2OH group.
- Haworth projections provide a good way to represent three-dimensional structure.
- Cyclic compounds can undergo mutarotation, in which they shift from one anomeric form to another with the straight-chain form as an intermediate.
Monosaccharides
- Monosaccharides are single carbohydrate units, with glucose as the most commonly observed monomer. They can undergo three main reactions: oxidation–reduction, esterification, and glycoside formation.
- Aldoses can be oxidized to aldonic acids and reduced to alditols.
- Sugars that can be oxidized are reducing agents (reducing sugars) themselves, and can be detected by reacting with Tollens' or Benedict’s reagents.
- Sugars with a –H replacing an –OH group are termed deoxy sugars.
- Sugars can react with carboxylic acids and their derivatives, forming esters (esterification). Phosphorylation is a similar reaction in which a phosphate ester is formed by transferring a phosphate group from ATP onto a sugar.
- Glycoside formation is the basis for building complex carbohydrates and requires the anomeric carbon to link to another sugar.
Complex Carbohydrates
- Disaccharides form as result of glycosidic bonding between two monosaccharide subunits; polysaccharides form by repeated monosaccharide or polysaccharide glycosidic bonding.
- Common disaccharides include sucrose (glucose-α-1,2-fructose), lactose (galactose-β-1,4-glucose), and maltose (glucose-α-1,4-glucose).
- Polysaccharides play various roles:
- Cellulose is the main structural component for plant cell walls and is a main source of fiber in the human diet.
- Starches (amylose and amylopectin) function as a main energy storage form for plants.
- Glycogen functions as a main energy storage form for animals.
ANSWERS TO CONCEPT CHECKS
4.1
- Aldopentose; ketohexose
-
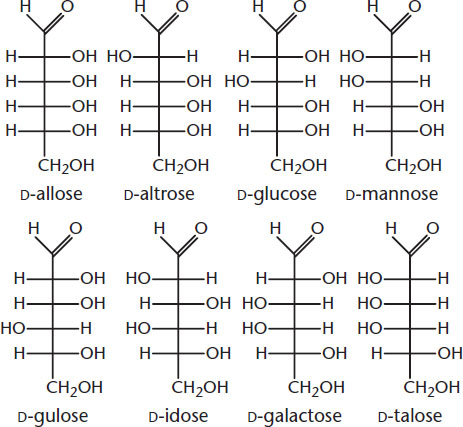
- D-Glucose’s epimers are D-mannose (C-2), D-allose (C-3), and D-galactose (C-4). None of the D-stereoisomers is an enantiomer for glucose; L-glucose is the enantiomer of D-glucose.
**4.2**
- During hemiacetal or hemiketal formation, the carbonyl carbon becomes chiral and is termed the anomeric carbon. The orientation of the –OH substituent on this carbon determines if the sugar molecule is the α- or β-anomer.
-
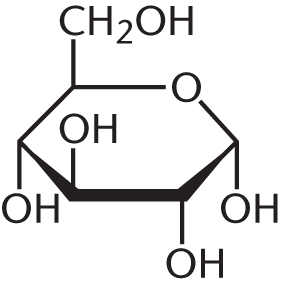
-
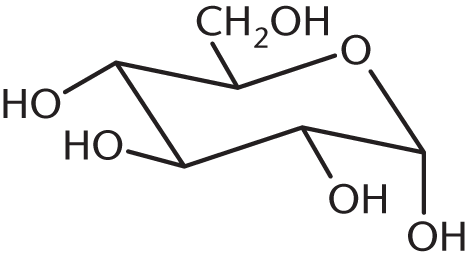
**4.3**
- Esterification is the reaction by which a hydroxyl group reacts with either a carboxylic acid or a carboxylic acid derivative to form an ester. Glycoside formation refers to the reaction between an alcohol and a hemiacetal (or hemiketal) group on a sugar to yield an alkoxy group.
- Tollens’ reagent and Benedict’s reagent are used to detect the presence of reducing sugars. Tollens’ reagent is reduced to produce a silvery mirror when aldehydes are present whereas Benedict’s reagent is indicated by a reddish precipitate of Cu2O.
- It makes sense for carbohydrates to become oxidized while reducing other groups. This is the case because aerobic metabolism requires reduced forms of electron carriers to facilitate processes such as oxidative phosphorylation. Because carbohydrates are a primary energy source, they are oxidized.
**4.4**
- Amylopectin is more soluble in solution than amylose because of its branched structure. The highly branched structure of amylopectin decreases intermolecular bonding between polysaccharide polymers and increases interaction with the surrounding solution.
- Glycogen has a higher rate of enzymatic branch cleavage because it contains significantly more branching than amylopectin.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
Glucose is an aldohexose, meaning that it has one aldehyde group and six carbons. Given this information, (A) and (B) can be eliminated. In aldose sugars, each nonterminal carbon is chiral. Therefore, glucose has four chiral centers, not five, as mentioned in (C). The number of stereoisomers possible for a chiral molecule is 2n, where n is the number of chiral carbons. Because glucose has four chiral centers, there are 24 = 16 possible stereoisomers.
2. D
Epimers are monosaccharide diastereomers that differ in their configuration about only one carbon. As with all diastereomers, epimers have different chemical and physical properties, and their optical activities have no relation to each other. Enantiomers have equal but opposite optical activities. Therefore, (D) is the only statement that does not apply to epimers.
3. C
Aldonic acids form after the aldehyde group on a reducing sugar reduces another compound, becoming oxidized in the process.
4. B
Mutarotation is the interconversion between anomers of a compound. Enantiomerization and racemization, (C) and (D), are related: enantiomerization is the formation of a mirror-image or optically inverted form of a compound, whereas racemization is moving a solution toward an equal concentration of both enantiomers. Glycosidation, (A), is the addition of a sugar to another compound.
5. A
Ketose sugars undergo tautomerization, a rearrangement of bonds, to undergo keto–enol shifts. This forms an aldose, which then allows them to act as reducing sugars. A ketone group alone cannot be oxidized. Anomerization, mentioned in (C), refers to ring closure of a monosaccharide, creating an anomeric carbon.
6. B
When glucose reacts with methanol under acid catalysis, the hemiacetal is converted to an acetal via replacement of the anomeric hydroxyl group with an alkoxy group. The result is a type of acetal known as a glycoside. This corresponds with (B). The other choices all show alkoxy groups on the wrong carbon, or too many carbons.
7. B
β-Amylase cleaves amylose at the nonreducing end of the polymer to yield maltose exclusively, while α-amylase, (A), cleaves amylose anywhere along the chain to yield short polysaccharides, maltose, and glucose. Debranching enzyme, (C), removes oligosaccharides from a branch in glycogen or starches, while glycogen phosphorylase, (D), yields glucose 1-phosphate.
8. C
Start by drawing out the Haworth projection. Recall that all the groups on the right in the Fischer projection will go on the bottom of the Haworth projection, and all the groups on the left will go on the top. Next, draw the chair structure, with the oxygen in the back right corner. Label the carbons in the ring 1 through 5, starting from the oxygen and moving clockwise around the ring. Now, draw in the lines for all the axial substituents, alternating above and below the ring. Remember to start on the anomeric C-1 carbon, where the axial substituent points down. Now start filling in the substituents. The substituent can be in either position on the anomeric carbon, so skip that one for now. The –OH groups on C-2 and C-4 should point downward while the –OH group on C-3 should point upward; (C), the β-anomer of D-glucose, is the only one that matches.
9. B
The hydroxyl group on the anomeric carbon of the β-anomer is equatorial, thereby creating less nonbonded strain than the α-anomer, which has the hydroxyl group of the anomeric carbon in axial position.
10. D
Glycogen and amylopectin are the only polysaccharide forms that demonstrate branching structure, making them most similar in terms of linkage. Both glycogen and amylopectin use α-1,4 and α-1,6 linkages. Cellulose uses β-1,4 linkages and amylose does not contain α-1,6 linkages.
11. C
While maltose and cellobiose both have the same glucose subunits, only maltose is digestible by humans because the β-glycosidic linkages in cellobiose cannot be cleaved in the human body.
12. C
In solution, the hemiacetal ring of glucose will break open spontaneously and then re-form. When the ring is broken, bond rotation occurs between C-1 and C-2 to produce either the α- or the β-anomer. The reaction given in this question depicts the mutarotation of glucose. (A) is incorrect because the reactant is an aldehyde, not the product. (B) is incorrect because a hemiketal has an –OH group, an –OR group, and two –R groups. In addition, hemiketals are formed from ketones, and our starting reactant is an aldehyde. Finally, (D) is incorrect because there is no glycosidic bond in the starting reactant.
13. A
Galactose is a diastereomer of glucose, with the stereochemistry at C-4 (counting from the aldehyde) reversed. Being able to identify C-4 is enough to answer this question, even without looking at the glucose molecule. Because (B), (C), and (D) have identical stereochemistry at C-4, they are incorrect.
14. D
In Andersen’s disease, glycogen is less branched than normal, thereby inducing lower solubility of glycogen. Branches reduce the interactions between adjacent chains of glycogen and encourage interactions with the aqueous environment. The smaller number of branches means that glycogen phosphorylase has fewer terminal glucose monomers on which to act, making enzyme activity slower than normal overall. Finally, without branches, the density of glucose monomers cannot be as high; therefore, the total glucose stored is lower than normal. Glycogen synthase is still functioning normally, so we would expect normal α-1,4 linkages in the glycogen of an individual with Andersen’s disease but few (if any) α-1,6 linkages.
15. C
Monosaccharides can exist as hemiacetals or hemiketals, depending on whether they are aldoses or ketoses. When a monosaccharide is in its cyclic form, the anomeric carbon is attached to the oxygen in the ring and a hydroxyl group. Hence, it is only a hemiacetal or hemiketal because an acetal or ketal would require the –OH group to be converted to another –OR group.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(4.1) Number of stereoisomers with common backbone= 2n
SHARED CONCEPTS
- Biochemistry Chapter 9
- Carbohydrate Metabolism I
- Biochemistry Chapter 10
- Carbohydrate Metabolism II
- Organic Chemistry Chapter 1
- Nomenclature
- Organic Chemistry Chapter 2
- Isomers
- Organic Chemistry Chapter 5
- Alcohols
- Organic Chemistry Chapter 6
- Aldehydes and Ketones I