Chapter 5: Lipid Structure and Function

Chapter 5: Lipid Structure and Function
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- Which of the following is NOT a type of glycolipid?
- Cerebroside
- Globoside
- Ganglioside
- Sphingomyelin
- During saponification:
- triacylglycerols undergo a condensation reaction.
- triacylglycerols undergo ester hydrolysis.
- fatty acid salts are produced using a strong acid.
- fatty acid salts are bound to albumin.
- Which of the following best describes the structure of steroids?
- Three cyclopentane rings, one cyclohexane ring
- Three cyclohexane rings, one cyclopentane ring
- Four carbon rings, differing in structure for each steroid
- Three cyclic carbon rings and a functional group
- Soap bubbles form because fatty acid salts organize into:
- lysosomes.
- micelles.
- phospholipid bilayers.
- hydrogen bonds.
- Steroid hormones are steroids that:
- have specific high-affinity receptors.
- travel in the bloodstream from endocrine glands to distant sites.
- affect gene transcription by binding directly to DNA.
- I only
- III only
- I and II only
- I and III only
- Why are triacylglycerols used in the human body for energy storage?
- They are highly hydrated and therefore can store lots of energy.
- They always have short fatty acid chains for easy access by metabolic enzymes.
- The carbon atoms of the fatty acid chains are highly reduced and therefore yield more energy upon oxidation.
- Polysaccharides, which would actually be a better energy storage form, would dissolve in the body.
- Which of the following is correct about fat-soluble vitamins?
- Vitamin E is important for calcium regulation.
- Vitamin D protects against cancer because it is a biological antioxidant.
- Vitamin K is necessary for the posttranslational introduction of calcium-binding sites.
- Vitamin A is metabolized to retinal, which is important for sight.
- III only
- I and II only
- III and IV only
- II, III, and IV only
- Which of the following is true of amphipathic molecules?
- They form protective spheres in any solvent, with hydrophobic molecules interior and hydrophilic molecules exterior.
- They have two fatty acid chains and a polar head group.
- They are important to the formation of the phospholipid bilayer and soap bubbles.
- They have a glycerol base.
- Which of the following is/are true about sphingolipids?
- They are all phospholipids.
- They all contain a sphingosine backbone.
- They can have either phosphodiester or glycosidic linkages to their polar head groups.
- I only
- III only
- II and III only
- I, II, and III
- Which of the following statements about saturation is FALSE?
- It can describe the number of double or triple bonds in a fatty acid tail.
- It determines at least one of the properties of membranes.
- More saturated fatty acids make for a more fluid solution.
- Fully saturated fatty acids have only single bonds.
- Which of the following is true about glycerophospholipids?
- Glycerophospholipids can sometimes be sphingolipids, depending on the bonds in their head groups.
- Glycerophospholipids are merely a subset of phospholipids.
- Glycerophospholipids are used in the ABO blood typing system.
- Glycerophospholipids have one glycerol, one polar head group, and one fatty acid tail.
- Which of the following statements about terpenes is FALSE?
- Terpenes are strongly scented molecules that sometimes serve protective functions.
- Terpenes are steroid precursors.
- A triterpene is made of three isoprene moieties, and therefore has 15 carbons.
- Terpenes are made by plants and insects.
- Which of the following is true about cholesterol?
- Cholesterol always increases membrane fluidity in cells.
- Cholesterol is a steroid hormone precursor.
- Cholesterol is a precursor for vitamin A, which is produced in the skin.
- Cholesterol interacts only with the hydrophobic tails of phospholipids.
- Which of the following statements regarding prostaglandins is FALSE?
- Prostaglandins regulate the synthesis of cAMP.
- Prostaglandin synthesis is inhibited by NSAIDs.
- Prostaglandins affect pain, inflammation, and smooth muscle function.
- Prostaglandins are endocrine hormones, like steroid hormones.
- Which of the statements regarding waxes is FALSE?
- Waxes generally have melting points above room temperature.
- Waxes are produced only in plants and insects and therefore must be consumed by humans.
- Waxes protect against dehydration and parasites.
- Waxes are esters of long-chain fatty acids and long-chain alcohols.
Answer Key
- D
- B
- B
- B
- C
- C
- C
- C
- B
- C
- B
- C
- B
- D
- B
Chapter 5: Lipid Structure and Function
CHAPTER 5
LIPID STRUCTURE AND FUNCTION
In This Chapter
5.1 Structural Lipids
Phospholipids
Glycerophospholipids
Sphingolipids
Waxes
5.2 Signaling Lipids
Terpenes and Terpenoids
Steroids
Prostaglandins
Fat-Soluble Vitamins
5.3 Energy Storage
Triacylglycerols
Free Fatty Acids and Saponification
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 3% of all questions about biochemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1D: Principles of bioenergetics and fuel molecule metabolism
3A: Structure and functions of the nervous and endocrine systems, and ways in which these systems coordinate the organ systems
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
What do beer, human eyes, and sperm whales have in common? For all of these, lipids play a key role in some of their most interesting characteristics. The taste and smell of hops comes from lipids called terpenes. The ability of the human eye to respond to light (and therefore see) relies heavily on retinal, a lipid derived from vitamin A. Sperm whales use an enormous reservoir of spermaceti to dive to a depth of up to three kilometers to hunt giant squid. Spermaceti is a lipid with a density that changes dramatically with temperature—effectively allowing sperm whales to adjust their density with depth so that they can stay submerged without having to constantly fight buoyancy.
Lipids, as a class, are characterized by insolubility in water and solubility in nonpolar organic solvents. Aside from this shared feature, lipids diverge dramatically in their structural organization and biological functions, serving vital structural, signaling, and energy storage roles. In this chapter, we will explore the structural and functional characteristics of each of the major categories of lipids tested on the MCAT.
As structural building blocks, we will investigate phospholipids and sterols, which make up vesicles, liposomes, and membranes. When it comes to signaling, we will note that lipids serve multiple roles, from enzyme cofactors to light-absorbing pigments, and from intracellular messengers to hormones. Finally, we will see that lipids are the workhorse of energy storage, giving the most “bang” for the metabolic “buck” by weight.
5.1 Structural Lipids
LEARNING OBJECTIVES
After Chapter 5.1, you will be able to:
- Recall the major structural traits of phospholipids, glycerophospholipids, and sphingolipids
- Recognize how membrane lipid structure contributes to overall membrane structure and function
- Distinguish between the three types of sphingolipids
- Identify the polar and nonpolar regions of lipids:
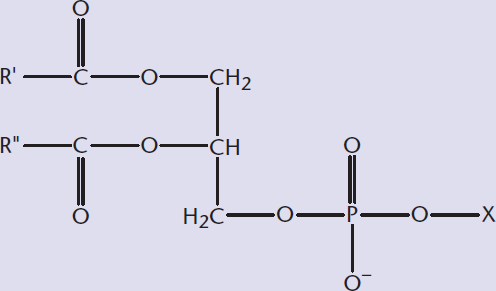
Lipids are the major component of the phospholipid bilayer, one of the most important structural parts of the cell. The unique ability of phospholipids to form a bilayer allows our cells to function as they do, separating the cell interior from the surrounding environment. We will first take a close look at the structure and role of phospholipids, glycerophospholipids, and sphingolipids. Finally, we will review the gross structural characteristics of the unique class called waxes.
Each of the membrane components is an amphipathic molecule, meaning that it has both hydrophilic and hydrophobic regions. For these membrane lipids, the polar head is the hydrophilic region, whereas the fatty acid tails are the hydrophobic region. When placed in aqueous solution, these molecules spontaneously form structures that allow the hydrophobic regions to group internally while the hydrophilic regions interact with water. This leads to the formation of various structures, including liposomes, micelles, and the phospholipid bilayer, shown in Figure 5.1.

Figure 5.1 Membrane Lipids Form Various Structures in Aqueous Solutions
Phospholipids
Phospholipids contain the following elements: a phosphate and alcohol that comprise the polar head group, joined to a hydrophobic fatty acid tail by phosphodiester linkages. One or more fatty acids are attached to a backbone to form the hydrophobic tail region. Phospholipids can be further classified according to the backbone on which the molecule is built. For example, glycerol, a three-carbon alcohol, forms phosphoglycerides or glycerophospholipids, and sphingolipids have a sphingosine backbone. One important thing to note, however, is that not all sphingolipids are phospholipids, as described later in this chapter.
BRIDGE
Although phospholipids are indeed the largest component of the phospholipid bilayer, nonphospholipids like glycolipids also play a role—and can be an important part of processes like cell recognition and signaling, discussed in Chapter 3 of MCAT Biochemistry Review.
One thing that these lipids do all share in common is a tail composed of long-chain fatty acids. These hydrocarbon chains vary by their degree of saturation and length. These two properties determine how the overall molecule will behave. Fully saturated fatty acid tails will have only single bonds; the carbon atom is considered saturated when it is bonded to four other atoms, with no π bonds. Saturated fatty acids, such as those in butter, have greater van der Waals forces and a more stable overall structure. Therefore, they form solids at room temperature. An unsaturated fatty acid includes one or more double bonds. Double bonds introduce kinks into the fatty acid chain, which makes it difficult for them to stack and solidify. This contrast with phospholipids that have saturated fatty acid tails is depicted in Figure 5.2. As a consequence of this difficulty stacking, unsaturated fats—like olive oil—tend to be liquids at room temperature. The same rules apply in the phospholipid bilayer: phospholipids with unsaturated fatty acid tails make up more fluid regions of the phospholipid bilayer. Phospholipids, glycerophospholipids, and sphingolipids can have any of a variety of fatty acid tails and also different head groups, which determine their properties at the surface of the cell membrane. The next two sections—glycerophospholipids and sphingolipids—focus on the various polar head groups that different phospholipids may have.
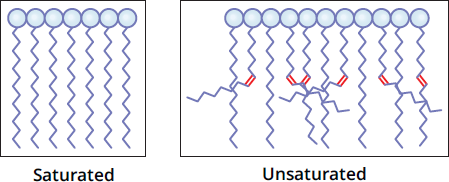
Figure 5.2 Phospholipids with Saturated and Unsaturated Fatty Acid Tails
KEY CONCEPT
Lipid properties—for all categories of lipids—are determined by the degree of saturation in fatty acid chains and the functional groups to which the fatty acid chains are bonded.
Glycerophospholipids
As mentioned in the last section, glycerophospholipids are all phospholipids; yet, not all phospholipids are glycerophospholipids! Glycerophospholipids (or phosphoglycerides) are specifically those phospholipids that contain a glycerol backbone bonded by ester linkages to two fatty acids and by a phosphodiester linkage to a highly polar head group, as shown in Figure 5.3. Because the head group determines the membrane surface properties, glycerophospholipids are named according to their head group. For example, phosphatidylcholine is the name of a glycerophospholipid with a choline head group, and phosphatidylethanolamine is one with an ethanolamine head group. The head group can be positively charged, negatively charged, or neutral. The membrane surface properties of these molecules make them very important to cell recognition, signaling, and binding. Within each subtype, the fatty acid chains can vary in length and saturation, resulting in an astounding variety of functions that are the focus of active scientific research.
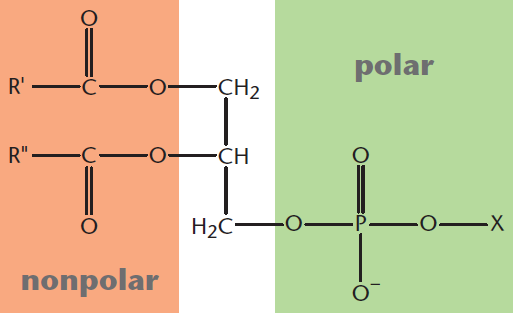
Figure 5.3 Structure of a Glycerophospholipid X denotes the head group connected to the glycerol backbone by a phosphodiester linkage.
Sphingolipids
Blood typing makes it possible to give life-saving blood transfusions without risking potentially fatal acute hemolytic reactions. The ABO blood typing system is based on cell-surface antigens on red blood cells. These cell-surface antigens are some of the most well-known sphingolipids. Like glycerophospholipids, sphingolipids are also sites of biological recognition at the cell surface and can be bonded to various head groups and fatty acids.
REAL WORLD
Although experiments in blood transfusion are recorded as early as the 17th century, blood typing wasn’t developed until the 20th century. With the advent of the ABO and Rh factor blood typing systems, blood transfusions could now be successfully administered to patients with hemophilia (a clotting disorder that causes significant bleeding), during surgery, and in a number of other applications.
Sphingolipids have a sphingosine or sphingoid (sphingosine-like) backbone, as opposed to the glycerol backbone of glycerophospholipids. These molecules also have long-chain, nonpolar fatty acid tails and polar head groups. Many sphingolipids are also phospholipids because they contain a phosphodiester linkage. However, other sphingolipids contain glycosidic linkages to sugars; any lipid linked to a sugar can be termed a glycolipid. Sphingolipids are divided into four major subclasses, differing by their head group.
The simplest sphingolipid is ceramide, which has a single hydrogen atom as its head group.
Sphingomyelins are the major class of sphingolipids that are also phospholipids (sphingophospholipids). These molecules have either phosphocholine or phosphoethanolamine as a head group, and thus contain a phosphodiester bond. Sphingomyelin head groups have no net charge. As the name implies, sphingomyelins are major components in the plasma membranes of cells producing myelin (oligodendrocytes and Schwann cells), the insulating sheath for axons.
REAL WORLD
Sphingolipid accumulation is associated with numerous pathological conditions. Sphingomyelins found in the myelin sheath help in signal transduction. Accumulation of sphingomyelin, resulting from lack of the enzyme sphingomyelinase, can result in Niemann–Pick disease. Symptoms can include intellectual disability and seizures. Sulfatides are sulfated cerebrosides associated with Alzheimer’s disease.
Sphingolipids with head groups composed of sugars bonded by glycosidic linkages are considered glycolipids, as mentioned above, or, more specifically, glycosphingolipids. These molecules are not phospholipids because they contain no phosphodiester linkage. Glycosphingolipids are found mainly on the outer surface of the plasma membrane and can be further classified as cerebrosides or globosides. Cerebrosides have a single sugar, whereas globosides have two or more. These molecules are also referred to as neutral glycolipids because they have no net charge at physiological pH.
The final group is composed of the most complex sphingolipids. Gangliosides are glycolipids that have polar head groups composed of oligosaccharides with one or more N-acetylneuraminic acid (NANA; also called sialic acid) molecules at the terminus and a negative charge. These molecules are also considered glycolipids because they have a glycosidic linkage and no phosphate group. Gangliosides play a major role in cell interaction, recognition, and signal transduction.
MNEMONIC
Gangliosides are the “gangly” sphingolipids, with the most complex structure and functional groups (oligosaccharides and NANA) in all directions.
A summary of the different types of sphingolipids is provided in Figure 5.4.
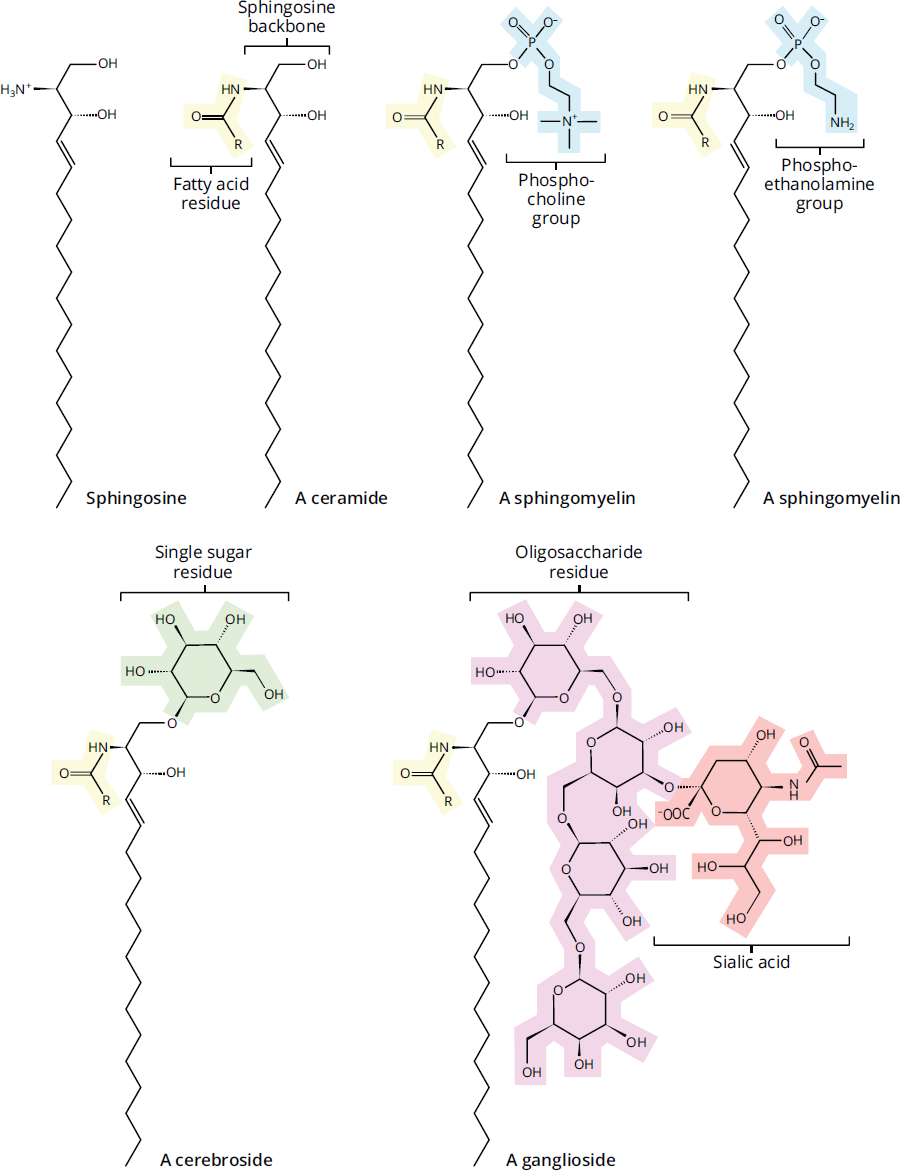
Figure 5.4 Types of Sphingolipids
Waxes
Waxes are esters created from long-chain fatty acids and long-chain alcohols. As one might expect, they form pliable solids at room temperature (what we generally think of as wax). Biologically, they function as protection for both plants and animals. In plants, waxes are secreted as a surface coating to prevent excessive evaporation and to protect against parasites. In animals, waxes are secreted to prevent dehydration, as a water-repellant to keep skin and feathers dry, and as lubricant. For example, carnauba wax is made from the leaves of the Copernicia prunifera palm and is used to coat candies and wax cars. Bees secrete waxes to construct shelter, as shown in Figure 5.5.

Figure 5.5 Honeycomb Structure Made from Beeswax The solid and plastic nature of waxes, which contain esters with long alkyl chains, permits their use for structure building.
MCAT CONCEPT CHECK 5.1:
Before you move on, assess your understanding of the material with these questions.
- Which components of membrane lipids contribute to their structural role in membranes? Which components contribute to function?
- Structure:
________________________________________
- Function:
________________________________________
- What is the difference between a sphingolipid that is also a phospholipid and one that is NOT?
________________________________________
- Name the three main types of sphingolipids and their characteristics.
Type Phospholipid or Glycolipid? Functional Group(s)
- What would happen if an amphipathic molecule were placed in a nonpolar solvent rather than an aqueous solution?
________________________________________
5.2 Signaling Lipids
LEARNING OBJECTIVES
After Chapter 5.2, you will be able to:
- Recall the structural features of terpenes, steroids, and prostaglandins
- Differentiate steroids from steroid hormones
- Connect prostaglandins to the symptoms associated with their presence, such as pain and inflammation
- Explain the importance of fat-soluble vitamins: A, D, E, and K
In addition to passive roles in structure, lipids also perform active roles in cellular signaling and as coenzymes. Lipids serve as coenzymes in the electron transport chain and in glycosylation reactions. Lipids also function as hormones that transmit signals over long distances and as intracellular messengers responding to extracellular signals. Certain special lipids with conjugated double bonds absorb light, which is extremely important for vision; others act as pigments in plants and animals. Here, we will focus on three important categories of signaling lipids: steroids, prostaglandins, and fat-soluble vitamins, as well as important precursors like terpenes.
BRIDGE
Remember from Chapter 2 of MCAT Biochemistry Review that a coenzyme is an organic, nonprotein factor bound to an enzyme and required for its normal activity.
Terpenes and Terpenoids
Before we delve into the details of downstream lipid signaling molecules, we must first turn our attention to terpenes. These odiferous chemicals are the metabolic precursors to steroids and other lipid signaling molecules, and have varied independent functions. Terpenes are a class of lipids built from isoprene (C5H8) moieties and share a common structural pattern with carbons grouped in multiples of five, as shown in Figure 5.6.

Figure 5.6 Isoprene
Terpenes are produced mainly by plants and also by some insects. They are generally strongly scented. In some cases, these pungent chemicals are part of the plant or insect’s protective mechanism. The strong smell of turpentine, a derivative of resin, comes from the monoterpenes that are resin’s major components; terpenes actually get their name from their original discovery in turpentine. Terpenes are also the primary components of much more pleasant-smelling essential oils extracted from plants.
Terpenes are grouped according to the number of isoprene units present; a single terpene unit contains two isoprene units. Monoterpenes (C10H16), which are abundant in both essential oils and turpentine as described above, contain two isoprene units. Sesquiterpenes (sesqui– meaning one-and-a-half) contain three isoprene units, and diterpenes contain four. Vitamin A, which will be discussed later in this chapter, is a diterpene from which retinal, a visual pigment vital for sight, is derived. Triterpenes, with six isoprene units, can be converted to cholesterol and various steroids, also discussed later in this chapter. Carotenoids, like β-carotene and lutein, are tetraterpenes and have eight isoprene units. Natural rubber has isoprene chains between 1000 and 5000 units long and is therefore considered a polyterpene.
Terpenoids, also sometimes referred to as isoprenoids, are derivatives of terpenes that have undergone oxygenation or rearrangement of the carbon skeleton. These compounds are further modified, as are terpenes, by the addition of an extensive variety of functional groups. Terpenoids share similar characteristics with terpenes in terms of both biological precursor function and aromatic properties, contributing to steroid biosynthesis, as well as the scents of cinnamon, eucalyptus, camphor, turmeric, and numerous other compounds. Terpenoids are named in an analogous fashion, with diterpenoids deriving from four isoprene units and so on. Terpenes and terpenoids are precursor molecules that feed into various biosynthesis pathways that produce important products, including steroids, which have widespread effects on biological function, and vitamin A, which is vital to sight.
Steroids
The term steroid probably brings to mind muscle–bound body builders or home run–hitting professional athletes. In science, and on the MCAT, steroid refers not just to the infamously abused anabolic steroids, but also to a broader class of molecules defined by their structure.
Structurally, steroids, shown in Figure 5.7, are metabolic derivatives of terpenes and are very different from the lipids mentioned earlier in this chapter in both structure and function. Steroids are characterized by having four cycloalkane rings fused together: three cyclohexane and one cyclopentane. Steroid functionality is determined by the oxidation status of these rings, as well as the functional groups they carry. It is important to note that the large number of carbons and hydrogens make steroids nonpolar, like the other lipids mentioned.
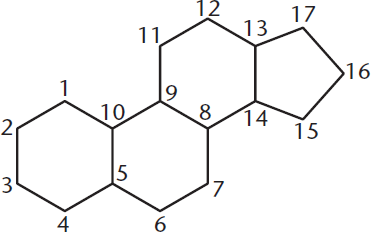
Figure 5.7 Common Steroid Structure
Keep in mind the terminology difference between steroids and steroid hormones. Steroid refers to a group defined by a particular chemical structure, demonstrated above. Steroid hormones are steroids that act as hormones, meaning that they are secreted by endocrine glands into the bloodstream and then travel on protein carriers to distant sites, where they can bind to specific high-affinity receptors and alter gene expression levels. Steroid hormones are potent biological signals that regulate gene expression and metabolism, affecting a wide variety of biological systems even at low concentrations. Some important steroid hormones include testosterone, various estrogens, cortisol, and aldosterone, which are discussed in Chapter 5 of *MCAT Biology** Review*. Plants, like animals, also use steroids as signaling molecules.
BRIDGE
Hormone types, including steroid hormones, are the focus of Chapter 5 of Kaplan MCAT Biology Review.
Cholesterol, shown in Figure 5.8, is a steroid of primary importance. Cholesterol is a major component of the phospholipid bilayer and is responsible for mediating membrane fluidity. Cholesterol, like a phospholipid, is an amphipathic molecule containing both hydrophilic and hydrophobic components. Interactions with both the hydrophobic tails and hydrophilic heads of phospholipids allows cholesterol to maintain relatively constant fluidity in cell membranes. At low temperatures, it keeps the cell membrane from solidifying; at high temperatures, it holds the membrane intact and prevents it from becoming too permeable. Cholesterol also serves as a precursor to many important molecules, including steroid hormones, bile acids, and vitamin D.
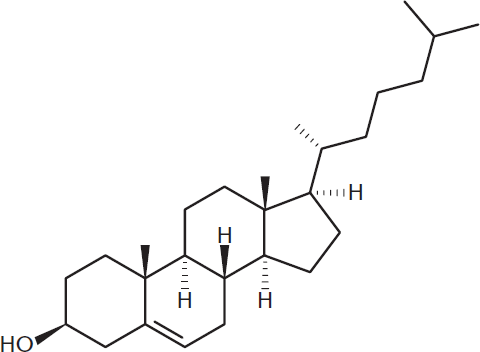
Figure 5.8 Cholesterol
REAL WORLD
Cholesterol can be produced de novo or absorbed from dietary sources. Hypercholesterolemia, or high cholesterol, is a condition that is strongly correlated with heart disease, in which the combination of these two cholesterol sources is excessive. Accumulation of cholesterol in arterial walls results in atherosclerosis, a hardening of the wall.
Prostaglandins
Prostaglandins acquired their name because they were first thought to be produced by the prostate gland, but have since been determined to be produced by almost all cells in the body. These 20-carbon molecules are unsaturated carboxylic acids derived from arachidonic acid and contain one five-carbon ring. They act as paracrine or autocrine signaling molecules. In many tissues, the biological function of prostaglandins is to regulate the synthesis of cyclic adenosine monophosphate (cAMP), which is a ubiquitous intracellular messenger. In turn, cAMP mediates the actions of many other hormones. Downstream effects of prostaglandins include powerful effects on smooth muscle function, influence over the sleep–wake cycle, and the elevation of body temperature associated with fever and pain. Nonsteroidal anti-inflammatory drugs (NSAIDs) like aspirin inhibit the enzyme cyclooxygenase (COX), which aids in the production of prostaglandins.
Fat-Soluble Vitamins
A vitamin is an essential nutrient that cannot be adequately synthesized by the body and therefore must be consumed in the diet. Vitamins are commonly divided into water-soluble and lipid-soluble categories. Lipid-soluble vitamins can accumulate in stored fat, whereas excess water-soluble vitamins are excreted through the urine. The fat-soluble vitamins include A, D, E, and K. Each of these has important and varied functions.
Vitamin A
Vitamin A, or carotene, is an unsaturated hydrocarbon that is important in vision, growth and development, and immune function. The most significant metabolite of vitamin A is the aldehyde form, retinal, which is a component of the light-sensing molecular system in the human eye. Retinol, the storage form of vitamin A, is also oxidized to retinoic acid, a hormone that regulates gene expression during epithelial development.
MNEMONIC
One way to remember carotene is to remember that carrots are high in vitamin A, which is why eating carrots is colloquially suggested to improve vision. To remember that vitamin D regulates calcium, remember that it is frequently added to milk in order to aid in the absorption of calcium.
Vitamin D
Vitamin D, or cholecalciferol, can be consumed or formed in a UV light–driven reaction in the skin. In the liver and kidneys, vitamin D is converted to calcitriol (1,25-(OH)2D3), the biologically active form of vitamin D. Calcitriol increases calcium and phosphate uptake in the intestines, which promotes bone production. A lack of vitamin D can result in rickets, a condition seen in children and characterized by underdeveloped, curved long bones as well as impeded growth.
Vitamin E
Vitamin E characterizes a group of closely related lipids called tocopherols and tocotrienols. These are characterized by a substituted aromatic ring with a long isoprenoid side chain and are characteristically hydrophobic. Tocopherols are biological antioxidants. The aromatic ring reacts with free radicals, destroying them. This, in turn, prevents oxidative damage, an important contributor to the development of cancer and aging.
Vitamin K
Vitamin K is actually a group of compounds, including phylloquinone (K1) and the menaquinones (K2). Vitamin K is vital to the posttranslational modifications required to form prothrombin, an important clotting factor in the blood. The aromatic ring of vitamin K undergoes a cycle of oxidation and reduction during the formation of prothrombin. Vitamin K is also required to introduce calcium-binding sites on several calcium-dependent proteins.
MNEMONIC
Vitamin K is for Koagulation.
REAL WORLD
The anticoagulant warfarin works by blocking the recycling of vitamin K, causing a deficiency or lowering of the active amount of vitamin K. Therefore, patients taking warfarin are recommended to stay away from food containing high amounts of vitamin K, such as green leafy vegetables.
MCAT CONCEPT CHECK 5.2:
Before you move on, assess your understanding of the material with these questions.
- How many carbons are in a diterpene?
________________________________________
- What is the difference between a steroid and a steroid hormone?
________________________________________
- NSAIDs block prostaglandin production in order to reduce pain and inflammation. What do prostaglandins do to bring about these symptoms?
________________________________________
- What are the names and functions of the four fat-soluble vitamins?
Name Function
5.3 Energy Storage
LEARNING OBJECTIVES
After Chapter 5.3, you will be able to:
- Explain why energy is more optimally stored as fat than as sugar
- Recall the structure and function of triacylglycerols
- Predict the products of saponification reactions:
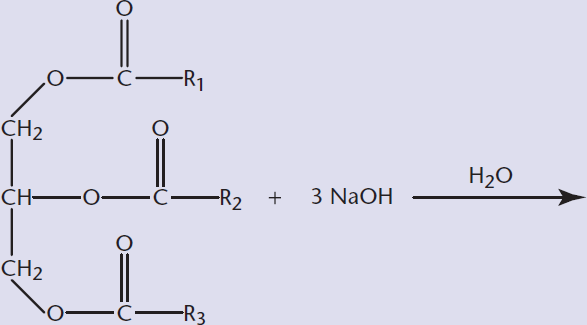
Triacylglycerols are a class of lipids specifically used for energy storage. From the body’s point of view, lipids in general are a fantastic way to store energy. This is true for two major reasons. First, the carbon atoms of fatty acids are more reduced than those of sugars, which contain numerous alcohol groups. The result of this is that the oxidation of triacylglycerols yields twice the amount of energy per gram as carbohydrates, making this a far more energy-dense storage mechanism compared to polysaccharides like glycogen. Second, triacylglycerols are hydrophobic. They do not draw in water and do not require hydration for stability. This helps decrease their weight, especially in comparison to hydrophilic polysaccharides. One final perk for vertebrates surviving in colder temperatures (like penguins, polar bears, and arctic explorers) is that the layer of lipids serves a dual purpose of energy storage and insulation—it helps to retain body heat so that less energy is required to maintain a constant internal temperature.
Triacylglycerols
Triacylglycerols, also called triglycerides, are composed of three fatty acids bonded by ester linkages to glycerol, as shown in Figure 5.9. For most naturally occurring triacylglycerols, it is rare for all three fatty acids to be the same.
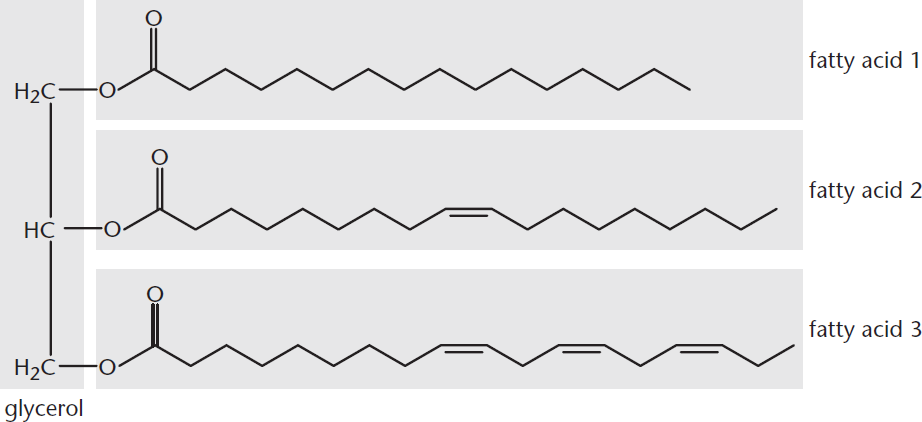
Figure 5.9 Triacylglycerol Structure The fatty acids used here are palmitic acid, oleic acid, andα-linolenic acid.
Overall, these compounds are nonpolar and hydrophobic. This contributes to their insolubility in water, as the polar hydroxyl groups of the glycerol component and the polar carboxylates of fatty acids are bonded together, decreasing their polarity.
Triacylglycerol deposits can be observed in cells as oily droplets in the cytosol. These serve as depots of metabolic fuel that can be recruited when the cell needs additional energy to divide or survive when other fuel supplies are low. Special cells in animals, known as adipocytes, store large amounts of fat and are found primarily under the skin, around mammary glands, and in the abdominal cavity. In plants, triacylglycerol deposits are also found in seeds as oils. Triacylglycerols travel bidirectionally in the bloodstream between the liver and adipose tissue. The physical characteristics of triacylglycerols are primarily determined by the saturation (or unsaturation) of the fatty acid chains that make them up, much like phospholipids.
REAL WORLD
The two main methods of energy storage in the body are as triacylglycerols in adipose tissue or as carbohydrates in glycogen. Each method has its advantages and disadvantages. For example, glycogen offers access to metabolic energy in a faster water-soluble form; however, because of its low energy density, glycogen can only provide energy for a bit less than one day. In contrast, an individual who is moderately obese with 15 to 20 kg of stored triacylglycerols in adipocytes could draw upon fat stores for months, but it takes more time to mobilize this energy.
Free Fatty Acids and Saponification
Free fatty acids are unesterified fatty acids with a free carboxylate group. In the body, these circulate in the blood bonded noncovalently to serum albumin. Fatty acids also make up what we know as soap, which can be produced through a process called saponification.
Saponification is the ester hydrolysis of triacylglycerols using a strong base. Traditionally, the base that is used is lye, the common name for sodium or potassium hydroxide. The result is the basic cleavage of the fatty acid, leaving the sodium salt of the fatty acid and glycerol, as shown in Figure 5.10. The fatty acid salt is what we know as soap.
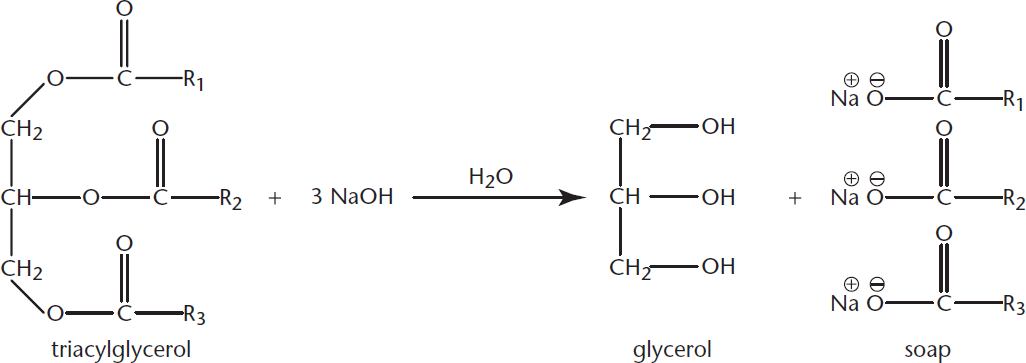
Figure 5.10 Saponification Ester hydrolysis of a triacylglycerol using lye (NaOH)
REAL WORLD
Saponification also occurs naturally, although more slowly, in corpses and oil paintings, as the triacylglycerols are hydrolyzed by naturally occurring bases. In corpses, the result of this process is known as adipocere, or grave wax.
Soaps can act as surfactants. A surfactant lowers the surface tension at the surface of a liquid, serving as a detergent or emulsifier. This is important to how soap works. If we try to combine an aqueous solution and oil, as with vinegar and olive oil in salad dressing, these solutions will remain in separate phases. If we were to add a soap, however, the two phases would appear to combine into a single phase, forming a colloid. This occurs because of the formation of micelles: tiny aggregates of soap with the hydrophobic tails turned inward and the hydrophilic heads turned outward, thereby shielding the hydrophobic lipid tails and allowing for overall solvation, as shown in Figure 5.11. We saw these earlier when we discussed membrane lipids.

Figure 5.11 Cross-Section of a Micelle Micelles organize in aqueous solution by forcing hydrophobic tails to the interior, allowing the hydrophilic heads to interact with water in the environment.
Nonpolar compounds can dissolve in the hydrophobic interior of the water-soluble micelle, meaning that our cleaning agents can dissolve both water-soluble and water-insoluble messes and then wash them all away together. Micelles are also important in the body for the absorption of fat-soluble vitamins (A, D, E, and K) and complicated lipids such as lecithins. Fatty acids and bile salts secreted by the gallbladder form micelles that can increase the surface area available for lipolytic enzymes.
BIOCHEMISTRY GUIDED EXAMPLE WITH EXPERT THINKING
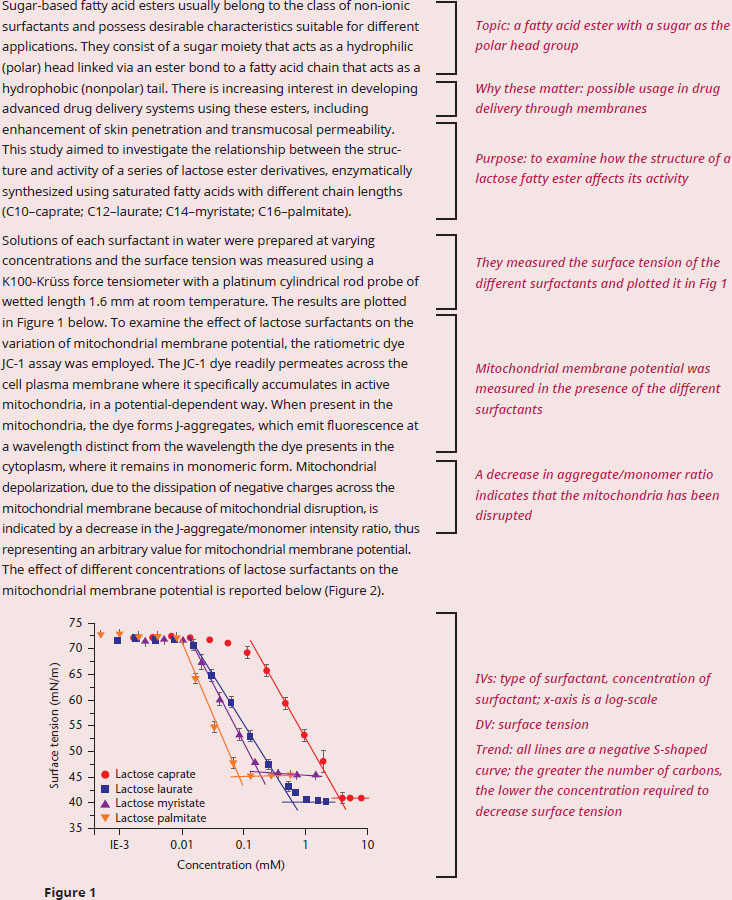
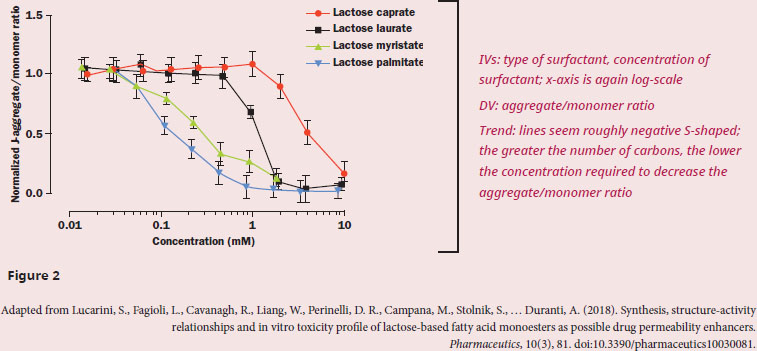
If the researchers were to examine cell viability in future experiments, which of the four surfactants would be expected to be the most cytotoxic?
The question asks us to predict the outcome of a future experiment, which indicates that we need to understand the results of the current experiment in order to predict future events. In the first paragraph, we see that the purpose of the passage is to discuss how these surfactants work as a function of the number of carbons attached to the sugar head group. From our outside knowledge we want to recall that surfactants have polar head groups, fatty acid tails, and often form micelles, allowing for nonpolar compounds (like many drugs!) to dissolve in the hydrophobic interior, while the surface-exposed portion of the micelle is water-soluble. Since the question specifically asks about the cytotoxic effects of the surfactants, we can mostly ignore Figure 1, as information about surface tension would not tell us if the surfactant was cytotoxic. Quickly assessing whether or not a figure is useful is an important skill for Test Day: not all of the figures will be valuable in answering every question. The measured variable in Figure 2 is the J-aggregate/monomer ratio, and the passage tells us that a drop in this ratio indicates that the potential across the mitochondrial membrane has been disrupted. We know from our content background that a proton gradient must be built across the inner mitochondrial membrane in order for ATP to be synthesized, and without that proton gradient the cell will quickly die due to a lack of energy. It’s possible that the amphiphilic nature of the surfactant is “poking” holes into the inner mitochondrial membrane, allowing for the proton gradient to dissipate without moving through ATP synthase.
Figure 2 shows that increasing carbon number in the fatty acid tail corresponds with decreasing concentration required to dissipate the mitochondrial potential. Thus, we can predict that a longer fatty acid tail will be linked with greater cytotoxic activity. Specifically, we would expect lactose palmitate to be the most cytotoxic of the four surfactants studied in the current experiment.
MCAT CONCEPT CHECK 5.3:
Before you move on, assess your understanding of the material with these questions.
- How does the human body store spare energy? Why doesn’t the human body store most energy as sugar?
________________________________________
- Describe the structure and function of triacylglycerols.
________________________________________
- What bonds are broken during saponification?
________________________________________
- Why does soap appear to dissolve in water, and how is this fact important to cleaning?
________________________________________
Conclusion
In this chapter, we examined the myriad biological functions performed by lipids. We first learned the structural functions of lipids, looking at the phospholipids that are the primary component of the phospholipid bilayer and other membrane lipids. Making our way through terpenes, we looked at the structure and function of signaling lipids, examining steroid hormones in particular. We looked into the fat-soluble vitamins and their downstream functions in the body. Finally, we summarized energy storage in the form of triacylglycerols and applied our acid–base chemistry knowledge to the formation of soap. In the next chapter, we turn our attention to the final class of biomolecules: nucleic acids.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Structural Lipids
- Lipids are insoluble in water and soluble in nonpolar organic solvents.
- Phospholipids are amphipathic and form the bilayer of biological membranes.
- They contain a hydrophilic (polar) head group and hydrophobic (nonpolar) tails.
- The head group is attached by a phosphodiester linkage and, because it interacts with the environment, determines the function of the phospholipid.
- The saturation of the fatty acid tails determines the fluidity of the membrane; saturated fatty acids are less fluid than unsaturated ones. Fatty acids form most of the structural thickness of the phospholipid bilayer.
- Glycerophospholipids are phospholipids that contain a glycerol backbone.
- Sphingolipids contain a sphingosine or sphingoid backbone.
- Many (but not all) sphingolipids are also phospholipids, containing a phosphodiester bond; these are termed sphingophospholipids.
- Sphingomyelins are the major class of sphingophospholipids and contain a phosphatidylcholine or phosphatidylethanolamine head group. They are a major component of the myelin sheath.
- Glycosphingolipids are attached to sugar moieties instead of a phosphate group. Cerebrosides have one sugar connected to sphingosine; globosides have two or more.
- Gangliosides contain oligosaccharides with at least one terminal N-acetylneuraminic acid (NANA; also called sialic acid).
- Waxes contain long-chain fatty acids esterified to long-chain alcohols. They are used as protection against evaporation and parasites in plants and animals.
Signaling Lipids
- Terpenes are odiferous steroid precursors made from isoprene, a five-carbon molecule.
- One terpene unit (a monoterpene) contains two isoprene units.
- Terpenoids are derived from terpenes via oxygenation or backbone rearrangement. They have similar odorous characteristics.
- Steroids contain three cyclohexane rings and one cyclopentane ring. Their oxidation state and functional groups may vary.
- Steroid hormones have high-affinity receptors, work at low concentrations, and affect gene expression and metabolism.
- Cholesterol is a steroid important to membrane fluidity and stability; it serves as a precursor to a host of other molecules.
- Prostaglandins are autocrine and paracrine signaling molecules that regulate cAMP levels. They have powerful effects on smooth muscle contraction, body temperature, the sleep–wake cycle, fever, and pain.
- The fat-soluble vitamins include vitamins A, D, E, and K.
- Vitamin A (carotene) is metabolized to retinal for vision and retinoic acid for gene expression in epithelial development.
- Vitamin D (cholecalciferol) is metabolized to calcitriol in the kidneys and regulates calcium and phosphorus homeostasis in the intestines (increasing calcium and phosphate absorption), promoting bone formation. A deficiency of vitamin D causes rickets.
- Vitamin E (tocopherols) act as biological antioxidants. Their aromatic rings destroy free radicals, preventing oxidative damage.
- Vitamin K (phylloquinone and menaquinones) is important for formation of prothrombin, a clotting factor. It performs posttranslational modifications on a number of proteins, creating calcium-binding sites.
Energy Storage
- Triacylglycerols (triglycerides) are the preferred method of storing energy for long-term use.
- They contain one glycerol attached to three fatty acids by ester bonds. The fatty acids usually vary within the same triacylglycerol.
- The carbon atoms in lipids are more reduced than carbohydrates, giving twice as much energy per gram during oxidation.
- Triacylglycerols are very hydrophobic, so they are not hydrated by body water and do not carry additional water weight.
- Animal cells specifically used for storage of large triacylglycerol deposits are called adipocytes.
- Free fatty acids are unesterified fatty acids that travel in the bloodstream. Salts of free fatty acids are soaps and can be synthesized in saponification.
- Saponification is the ester hydrolysis of triacylglycerols using a strong base, like sodium or potassium hydroxide.
- Soaps act as surfactants, forming micelles. A micelle can dissolve a lipid-soluble molecule in its fatty acid core, and washes away with water because of its shell of carboxylate head groups.
ANSWERS TO CONCEPT CHECKS
5.1
- Membrane lipids are amphipathic: they have hydrophilic heads and hydrophobic tails, allowing for the formation of bilayers in aqueous solution. The fatty acid tails form the bulk of the phospholipid bilayer, and play a predominantly structural role. On the other hand, the functional differences between membrane lipids are determined by the polar head group, due to its constant exposure to the exterior environment of the phospholipid bilayer (remember, this can be either the inside or outside of the cell). The degree of unsaturation of fatty acid tails can also play a small role in function.
- The difference is the bond between the sphingosine backbone and the head group. When this is a phosphodiester bond, it’s a phospholipid (note the phospho– prefixes). Nonphospholipid sphingolipids include glycolipids, which contain a glycosidic linkage to a sugar.
-
Type Phospholipid or Glycolipid? Functional Group(s) Sphingomyelin Phospholipid Phosphatidylethanolamine/phosphatidylcholine
Glycosphingolipid Glycolipid Sugars (mono- or polysaccharide)
Ganglioside Glycolipid Oligosaccharides and N-acetylneuraminic acid (NANA)
- In a nonpolar solvent, we would see the opposite of what happens in a polar solvent like water: the hydrophilic, polar part of the molecules would be sequestered inside, while the nonpolar, hydrophobic part of the molecules would be found on the exterior and exposed to the solvent.
**5.2**
- A diterpene has 20 carbon molecules in its backbone. One terpene unit is made from two isoprene units, each of which has five carbons.
- A steroid is defined by its structure: it includes three cyclohexane rings and a cyclopentane ring. A steroid hormone is a molecule within this class that also functions as a hormone, meaning that it travels in the bloodstream, is active at low concentrations, has high-affinity receptors, and affects gene expression and metabolism.
- Prostaglandins regulate the synthesis of cAMP, which is involved in many pathways, including those that drive pain and inflammation.
-
Name Function
A (carotene) As retinal: vision; as retinoic acid: epithelial development
D (cholecalciferol) As calcitriol: calcium and phosphate regulation
E (tocopherols) Antioxidants, using aromatic ring
K (phylloquinone and menaquinones) Posttranslational modification of prothrombin, addition of calcium-binding sites on many proteins
**5.3**
- The human body stores energy as glycogen and triacylglycerols. Triacylglycerols are preferred because their carbons are more reduced, resulting in a larger amount of energy yield per unit weight. In addition, due to their hydrophobic nature, triacylglycerols do not need to carry extra weight from hydration.
- Triacylglycerols, also called triglycerides, are composed of a glycerol backbone esterified to three fatty acids. They are used for energy storage.
- The ester bonds of triacylglycerols are broken to form a glycerol molecule and the salts of fatty acids (soap).
- Soap appears to dissolve in water because amphipathic free fatty acid salts form micelles, with hydrophobic fatty acid tails toward the center and carboxylate groups facing outward toward the water. Fat-soluble particles can then dissolve inside micelles in the soap–water solution and wash away. Water-soluble compounds can freely dissolve in the water.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
Glycolipids contain sugar moieties connected to their backbone. Sphingomyelin is not a glycolipid, but rather a phospholipid. This class can either have phosphatidylcholine or phosphatidylethanolamine as a head group and therefore contains a phosphodiester, not glycosidic, bond.
2. B
Saponification is the ester hydrolysis of triacylglycerol using a strong base like sodium or potassium hydroxide to form glycerol and fatty acid salts. This is not a condensation reaction, as in (A), but a cleavage reaction. Fatty acids do travel in the body bonded to serum albumin, as in (D), but that is unrelated to the process of saponification.
3. B
The basic backbone of steroid structure contains three cyclohexane rings and one cyclopentane ring. Although the oxidation status of these rings varies for different steroids, the overall structure does not, as in (C).
4. B
Fatty acid salt micelles are responsible for the formation of soap bubbles. While phospholipids can form bilayers, as in (C), the fatty acids in soap are free fatty acids, not phospholipids.
5. C
Steroid hormones are produced in endocrine glands and travel in the bloodstream to bind high-affinity receptors in the nucleus. The hormone’s receptor binds to DNA as part of the hormone–receptor complex, but the hormone itself does not.
6. C
Triacylglycerols are highly hydrophobic and therefore not highly hydrated (which would add extra weight from the water of hydration, taking away from the energy density of these molecules), eliminating (A). The fatty acid chains produce twice as much energy as polysaccharides during oxidation because they are highly reduced. The fatty acid chains vary in length and saturation.
7. C
Vitamin A is metabolized to retinal, which is important for sight. Vitamin D is metabolized to calcitriol, which is important for calcium regulation. Vitamin E is made up of tocopherols, which are biological antioxidants. Vitamin K is necessary for the introduction of calcium binding sites, such as during the posttranslational modification of prothrombin.
8. C
Phospholipids are amphipathic, as are fatty acid salts. Although amphipathic molecules take spherical forms with hydrophobic molecules interior in aqueous solution, as in (A), the opposite would be true in a nonpolar solvent. (B) describes phospholipids and sphingolipids, and (D) describes triacylglycerols and phospholipids; both groups do not include fatty acid salts.
9. B
Sphingolipids can either have a phosphodiester bond, and therefore be phospholipids, or have a glycosidic linkage and therefore be glycolipids. Not all sphingolipids have a sphingosine backbone, as in statement II; some have related (sphingoid) compounds as backbones instead.
10. C
More saturated fatty acids make for a less fluid solution. This is because they can pack more tightly and form more noncovalent bonds, resulting in more energy being needed to disrupt the overall structure.
11. B
Glycerophospholipids are a subset of phospholipids, as are sphingomyelins. Glycerophospholipids are never sphingolipids because they contain a glycerol backbone (rather than sphingosine or a sphingoid backbone), eliminating (A). Sphingolipids are used in the ABO blood typing system, eliminating (C). Glycerophospholipids have a polar head group, glycerol, and two fatty acid tails, not one, as in (D).
12. C
A triterpene is made of six isoprene moieties (remember, one terpene unit contains two isoprene units), and therefore has a 30-carbon backbone.
13. B
Cholesterol is a steroid hormone precursor that has variable effects on membrane fluidity depending on temperature, eliminating (A). It interacts with both the hydrophobic tails and the hydrophilic heads of membrane lipids, nullifying (D). It is also a precursor for vitamin D (not vitamin A), which can be produced in the skin in a UV-driven reaction, eliminating (C).
14. D
Prostaglandins are paracrine or autocrine signaling molecules, not endocrine—they affect regions close to where they are produced, rather than affecting the entire body. Think of the swelling that happens when you bash your knee into your desk: your knee will swell, become discolored, and possibly bruise. Luckily, however, your entire body won’t swell as well.
15. B
Waxes are also produced in animals for similar protective functions. Cerumen, or earwax, is a prime example in humans.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 2
- Enzymes
- Biochemistry Chapter 8
- Biological Membranes
- Biochemistry Chapter 11
- Lipid and Amino Acid Metabolism
- Biology Chapter 5
- The Endocrine System
- Biology Chapter 9
- The Digestive System
- Organic Chemistry Chapter 8
- Carboxylic Acids