Chapter 8: Biological Membranes

Chapter 8: Biological Membranes
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- A student is trying to determine the type of membrane transport occurring in a cell. The student finds that the molecule to be transported is very large and polar, and when transported across the membrane, no energy is required. Which of the following is the most likely mechanism of transport?
- Active transport
- Simple diffusion
- Facilitated diffusion
- Exocytosis
- A researcher treats a solution containing animal cells with ouabain, a poisonous substance that interferes with the sodium–potassium ATPase embedded in the cell membrane, and the cell lyses as a result. Which of the following statements best describes ouabain’s effects?
- Treatment with ouabain results in high levels of extracellular calcium.
- Treatment with ouabain results in high levels of extracellular potassium and sodium.
- Treatment with ouabain increases intracellular concentrations of sodium.
- Treatment with ouabain decreases intracellular concentrations of sodium.
- Resting membrane potential depends on:
- the differential distribution of ions across the membrane.
- active transport processes.
- selective permeability of the phospholipid bilayer.
- I only
- I and III only
- II and III only
- I, II, and III
- Which of the following is NOT a function of the cell membrane?
- Cytoskeletal attachment
- Protein synthesis
- Transport regulation
- Second messenger reservoir
- The dynamic properties of molecules in the cell membrane are most rapid in:
- phospholipids moving within the plane of the membrane.
- phospholipids moving between the layers of the membrane.
- proteins moving within the plane of the membrane.
- proteins exiting the cell through exocytosis.
- Which lipid type is LEAST likely to contribute to membrane fluidity?
- Unsaturated glycerophospholipids
- trans glycerophospholipids
- Cholesterol
- Unsaturated sphingolipids
- A membrane receptor is most likely to be a(n):
- embedded protein with catalytic activity.
- transmembrane protein with sequestration activity.
- membrane-associated protein with sequestration activity.
- transmembrane protein with catalytic activity.
- Which of the following is NOT a cell–cell junction in animals?
- Desmosomes
- Gap junctions
- Plasmodesmata
- Tight junctions
- Which of the following is true of diffusion and osmosis?
- Diffusion and osmosis rely on the concentration gradient of only the compound of interest.
- Diffusion and osmosis rely on the concentration gradient of all compounds in a cell.
- Diffusion and osmosis will proceed in the same direction if there is only one solute.
- Diffusion and osmosis cannot occur simultaneously.
- The bulk movement of liquid into a cell through vesicular infoldings is known as:
- phagocytosis.
- pinocytosis.
- exocytosis.
- drinking.
- Which of the following is LEAST likely to be the resting membrane potential of a cell?
- –70 mV
- –55 mV
- 0 mV
- +35 mV
- How does the inner mitochondrial membrane differ from the outer mitochondrial membrane?
- The inner mitochondrial membrane is more permeable and lacks cholesterol.
- The inner mitochondrial membrane is less permeable and lacks cholesterol.
- The inner mitochondrial membrane is more permeable and has cholesterol.
- The inner mitochondrial membrane is less permeable and has cholesterol.
- For most cells, the extracellular calcium concentration is around 10,000 times higher than the intracellular calcium concentration. What is the membrane potential established by this electrochemical gradient?
- –123 mV
- –61.5 mV
- +61.5 mV
- +123 mV
- Which of the following statements conflicts with the fluid mosaic model?
- The cell membrane is static in structure.
- Membrane components can be derived from multiple biomolecules.
- Hydrophobic interactions stabilize the lipid bilayer.
- Proteins are asymmetrically distributed within the cell membrane.
- Which of the following is a sphingolipid?
- Lecithin
- Phosphatidylinositol
- Cholesterol
- Ganglioside
Answer Key
- C
- C
- D
- B
- A
- B
- D
- C
- A
- B
- C
- B
- D
- A
- D
Chapter 8: Biological Membranes
CHAPTER 8
BIOLOGICAL MEMBRANES
In This Chapter
8.1 Fluid Mosaic Model
General Membrane Structure and Function
Membrane Dynamics
8.2 Membrane Components
Lipids
Proteins
Carbohydrates
Membrane Receptors
Cell–Cell Junctions
8.3 Membrane Transport
Concentration Gradients
Passive Transport
Active Transport
Endocytosis and Exocytosis
8.4 Specialized Membranes
Membrane Potential
Mitochondrial Membranes
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 10% of all questions about biochemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
2A: Assemblies of molecules, cells, and groups of cells within single cellular and multicellular organisms
3A: Structure and functions of the nervous and endocrine systems, and ways in which these systems coordinate the organ systems
Introduction
Biological membranes are a stunning combination of opposites and contrasts. They are exceptionally thin, structurally bland, and relatively straightforward to describe. Yet, they define the borders of cells, tissues, and organelles; carry out a significant number of the biological functions within cells; and are an unending source of scientific inquiry and discovery. The most commonly tested biological membrane on the MCAT is the plasma membrane. At first, the plasma membrane seems like it’s only a shell—just a barrier that defines the cell. But the plasma membrane plays roles in signaling, entry of nutrients and expulsion of waste, cell recognition, transport of materials between tissues, and even electronic functions.
Cell membranes have both a stretchy, flexible component (phospholipids) and an abundance of stabilizing molecules (cholesterol and protein) to make sure that everything remains intact. In this chapter, we will examine the general function, composition, and transport properties of biological membranes. We will conclude by taking a look at a few specialized cell membranes within the body, in addition to specific membrane properties.
8.1 Fluid Mosaic Model
LEARNING OBJECTIVES
After Chapter 8.1, you will be able to:
- Describe the functions of flippases and lipid rafts
- Order a given list of membrane components from least to most abundant
The cell (plasma) membrane is often described as a semipermeable phospholipid bilayer. This phrase alone describes both the function and structure of the cell membrane: as a semipermeable barrier, it chooses which particles can enter and leave the cell at any point in time. This selectivity is mediated not only by the various channels and carriers that poke holes in the membrane, but also by the membrane itself. Composed primarily of two layers of phospholipids, the cell membrane permits fat-soluble compounds to cross easily, while larger and water-soluble compounds must seek alternative entry. The cell membrane is illustrated in Figure 8.1; the theory that underlies the structure and function of the cell membrane is referred to as the fluid mosaic model.
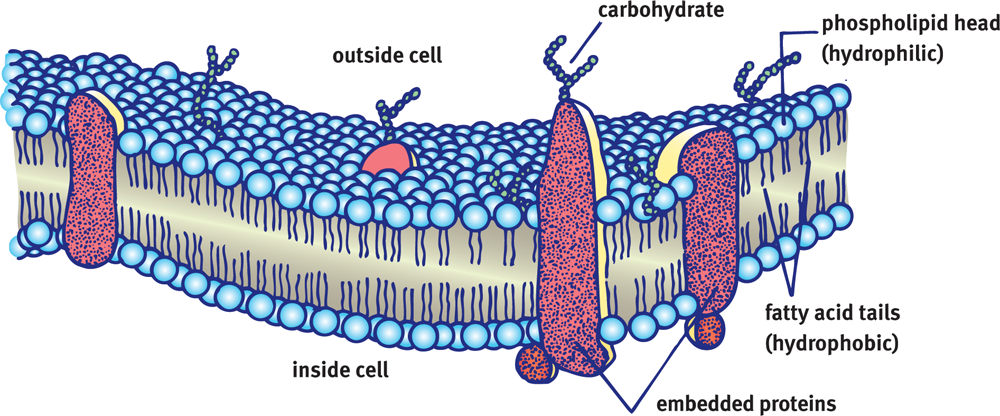
Figure 8.1 Cell Membrane The cell membrane is a phospholipid bilayer that regulates movement of solutes into and out of the cell.
General Membrane Structure and Function
The phospholipid bilayer also includes proteins and distinct signaling areas within lipid rafts. Carbohydrates associated with membrane-bound proteins create a glycoprotein coat. The cell wall of plants, bacteria, and fungi contain higher levels of carbohydrates.
The main function of the cell membrane is to protect the interior of the cell from the external environment. Cellular membranes selectively regulate traffic into and out of the cell and are involved in both intracellular and intercellular communication and transport. Cell membranes also contain proteins embedded within the lipid bilayer that act as cellular receptors during signal transduction. These proteins play an important role in regulating and maintaining overall cellular activity.
Membrane Dynamics
The cell membrane functions as a stable semisolid barrier between the cytoplasm and the environment, but it is in a constant state of flux on the molecular level. Phospholipids move rapidly in the plane of the membrane through simple diffusion. This can be seen when fusing two membranes that have been tagged with different labels; the tags will migrate with their associated lipids until both types are rapidly intermixed. Lipid rafts are collections of similar lipids with or without associated proteins that serve as attachment points for other biomolecules; these rafts often serve roles in signaling. Both lipid rafts and proteins also travel within the plane of the membrane, but more slowly. Lipids can also move between the membrane layers, but this is energetically unfavorable because the polar head group of the phospholipid must be forced through the nonpolar tail region in the interior of the membrane. Specialized enzymes called flippases assist in the transition or “flip” between layers. Figure 8.2 illustrates a lipid raft and the role of flippases.
REAL WORLD
Many antidepressants increase levels of neurotransmitters in the brain, but the effects take longer to appear than the changes in neurochemistry. The reason for this delay is that the nervous system must still upregulate its postsynaptic receptors to respond to the new levels of neurotransmitter.
Dynamic changes in the concentrations of various membrane proteins are mediated by gene regulation, endocytic activity, and protein insertion. Many cells, particularly those involved in biosignaling processes, can up- or downregulate the number of specific cellular receptors on their surface in order to meet cellular requirements.
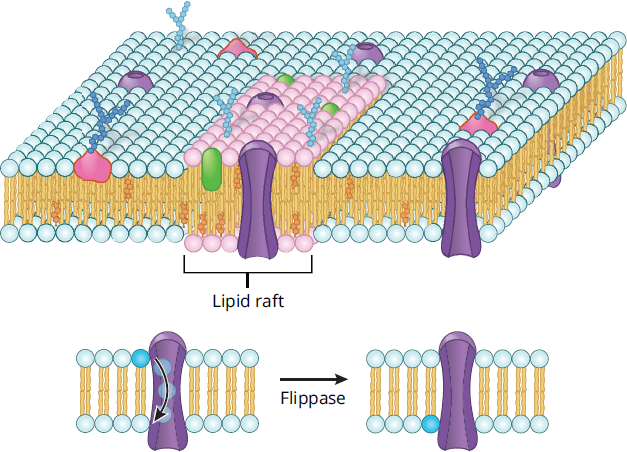
Figure 8.2 Lipid Rafts and Flippases Lipid rafts and flippases aid in signaling at the cell membrane.
MCAT CONCEPT CHECK 8.1:
Before you move on, assess your understanding of the material with these questions.
- Describe the role of flippases and lipid rafts in biological membranes.
- Flippases: ____________________________________
- Lipid rafts: ____________________________________
- List the following membrane components in order from most plentiful to least plentiful: carbohydrates, lipids, proteins, nucleic acids.
______ > ____ > ____ > ______
8.2 Membrane Components
LEARNING OBJECTIVES
After Chapter 8.2, you will be able to:
- Describe the role of cholesterol in cell membranes
- Define the three classes of membrane proteins: transmembrane, embedded, and membrane-associated proteins
- Differentiate between gap junctions, tight junctions, desmosomes, and hemidesmosomes
- Identify level of saturation, as well as the hydrophilic and hydrophobic portions of a phospholipid:
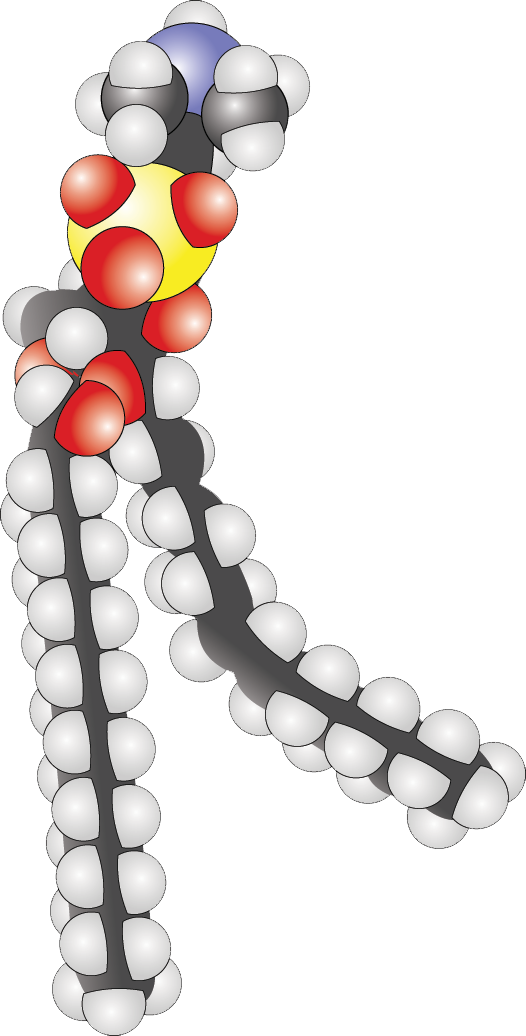
While the fluid mosaic model outlines the general composition of the membrane, the MCAT expects us to have a stronger grasp of the specifics, especially as it pertains to lipids and proteins.
Lipids
The cell membrane is composed predominantly of lipids with some associated proteins and carbohydrates. At times, the cell membrane as a whole will be referred to as a phospholipid bilayer, as it is the primary component of this barrier around the cell. Within the cell membrane, there are a large number of phospholipids with very few free fatty acids. In addition, steroid molecules and cholesterol, which lend fluidity to the membrane, and waxes, which provide membrane stability, help to maintain the structural integrity of the cell. While the structural details of these lipids were discussed in detail in Chapter 5 of MCAT Biochemistry Review, we will briefly describe their key points here.
Fatty Acids and Triacylglycerols
Fatty acids are carboxylic acids that contain a hydrocarbon chain and terminal carboxyl group. Triacylglycerols, also referred to as triglycerides, are storage lipids involved in human metabolic processes. They contain three fatty acid chains esterified to a glycerol molecule. Fatty acid chains can be saturated or unsaturated. Unsaturated fatty acids are regarded as “healthier” fats because they tend to have one or more double bonds and exist in liquid form at room temperature; in the plasma membrane, these characteristics impart fluidity to the membrane. Humans can only synthesize a few of the unsaturated fatty acids; the rest come from essential fatty acids in the diet that are transported as triacylglycerols from the intestine inside chylomicrons. Two important essential fatty acids for humans are *α**-linolenic acidandlinoleic acid*. Saturated fatty acids are the main components of animal fats and tend to exist as solids at room temperature. Saturated fats are found in processed foods and are considered less healthy. As shown in Figure 8.3, when incorporated into phospholipid membranes, saturated fatty acids decrease the overall membrane fluidity.
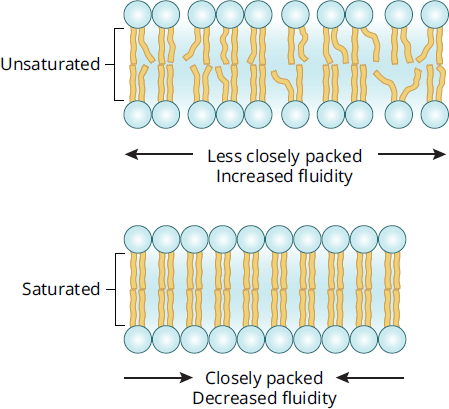
Figure 8.3 Effects of Fatty Acid Saturation on Membrane Fluidity
REAL WORLD
Trans fats, which result from the partial hydrogenation of some unsaturated fatty acids, have been banned from certain stores and cities because of their health risks. Part of the health concern is due to their ability to lower membrane fluidity, in addition to the tendency of trans fats to accumulate and form plaques in blood vessels.
Phospholipids
By substituting one of the fatty acid chains of triacylglycerol with a phosphate group, a polar head group joins the nonpolar tails, forming a glycerophospholipid, commonly called a phospholipid. Phospholipids spontaneously assemble into micelles (small monolayer vesicles) or liposomes (bilayered vesicles) due to hydrophobic interactions. Glycerophospholipids are used for membrane synthesis and can produce a hydrophilic surface layer on lipoproteins such as very-low-density lipoprotein (VLDL), a lipid transporter. In addition, phospholipids are the primary component of cell membranes. Phospholipids serve not only structural roles, but can also serve as second messengers in signal transduction. The phosphate group also provides an attachment point for water-soluble groups, such as choline (phosphatidylcholine, also known as lecithin) or inositol (phosphatidylinositol). A comparison of triacylglycerols and glycerophospholipids is shown in Figure 8.4.
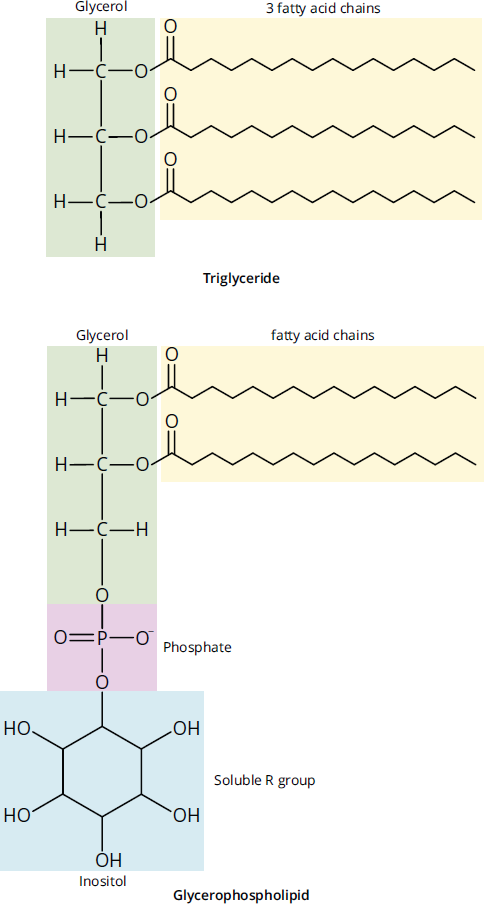
Figure 8.4 Triacylglycerol and Glycerophospholipid (Phosphatidylinositol)
Sphingolipids
Sphingolipids are also important constituents of cell membranes. Although sphingolipids do not contain glycerol, they are similar in structure to glycerophospholipids, in that they contain a hydrophilic region and two fatty acid–derived hydrophobic tails. The various classes of sphingolipids shown in Figure 8.5 differ primarily in the identity of their hydrophilic regions. Classes of sphingolipids and their hydrophilic groups include ceramide, sphingomyelins, cerebrosides, and gangliosides.
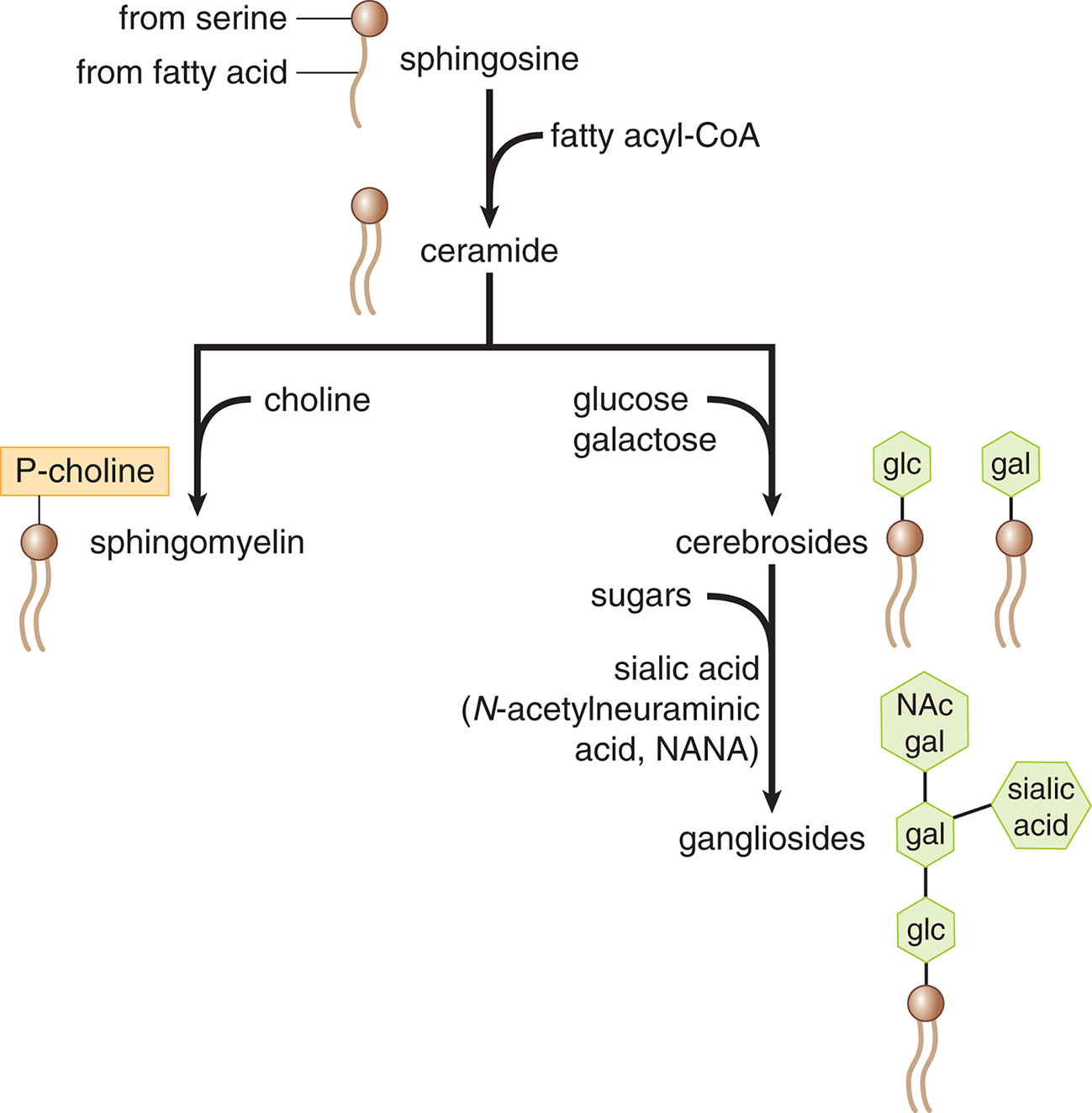
Figure 8.5 Types of Sphingolipids Sphingolipids are sequentially modified to form each of the biologically necessary molecules in the class.
MCAT EXPERTISE
The ratio of certain sphingolipids to glycerophospholipids can help to identify particular membranes within the cell, but memorizing this information is unnecessary for Test Day. Where small details like this are important, they will be provided in a passage.
Cholesterol and Steroids
Cholesterol is associated with a number of negative health effects and receives a lot of negative press; however, it is also a very important molecule in our cells. Cholesterol not only regulates membrane fluidity, but it is also necessary in the synthesis of all steroids, which are derived from cholesterol.
The structure of cholesterol is similar to that of phospholipids in that cholesterol contains both a hydrophilic and hydrophobic region. Membrane stability is derived from interactions with both the hydrophilic and hydrophobic regions that make up the phospholipid bilayer. While cholesterol stabilizes adjacent phospholipids, it also occupies space between them. This prevents the formation of crystal structures in the membrane, increasing fluidity at lower temperatures. At high temperatures, cholesterol has the opposite effect: by limiting movement of phospholipids within the bilayer, it decreases fluidity and helps hold the membrane intact. The temperature effects of cholesterol on the phospholipid bilayer are shown in Figure 8.6. By mass, cholesterol composes about 20 percent of the cell membrane; by mole fraction, it makes up about half. This large ratio of cholesterol to phospholipid ensures that the membrane remains fluid.
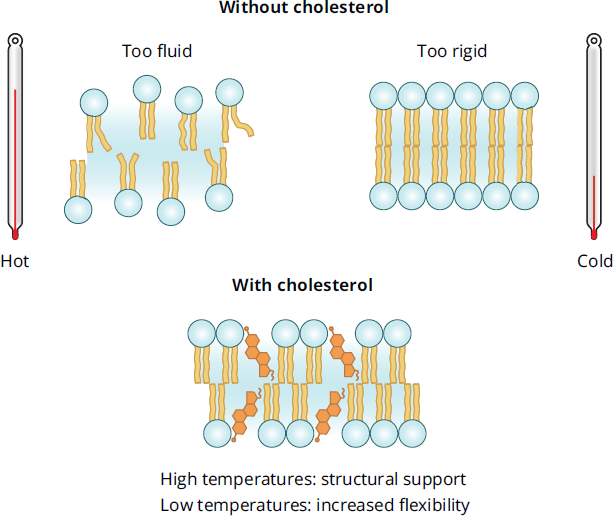
Figure 8.6 Stabilizing Effects of Cholesterol on Membranes
Waxes
Waxes are a class of lipids that are extremely hydrophobic and are rarely found in the cell membranes of animals, but are sometimes found in the cell membranes of plants. A wax is composed of a long-chain fatty acid and a long-chain alcohol, which contribute to the high melting point of these substances. When present within the cell membrane, waxes can provide both stability and rigidity within the nonpolar tail region only. Most waxes serve an extracellular function in protection or waterproofing.
Proteins
The fluid mosaic model also accounts for the presence of three types of membrane proteins, as shown in Figure 8.7. Transmembrane proteins pass completely through the lipid bilayer. Embedded proteins, on the other hand, are associated with only the interior (cytoplasmic) or exterior (extracellular) surface of the cell membrane. Together, transmembrane and embedded proteins are considered integral proteins because of their association with the interior of the plasma membrane, which is usually assisted by one or more membrane-associated domains that are partially hydrophobic. Membrane-associated (peripheral) proteins may be bound through electrostatic interactions with the lipid bilayer, especially at lipid rafts, or to other transmembrane or embedded proteins, like the G proteins found in G protein-coupled receptors. Transporters, channels, and receptors are generally transmembrane proteins.
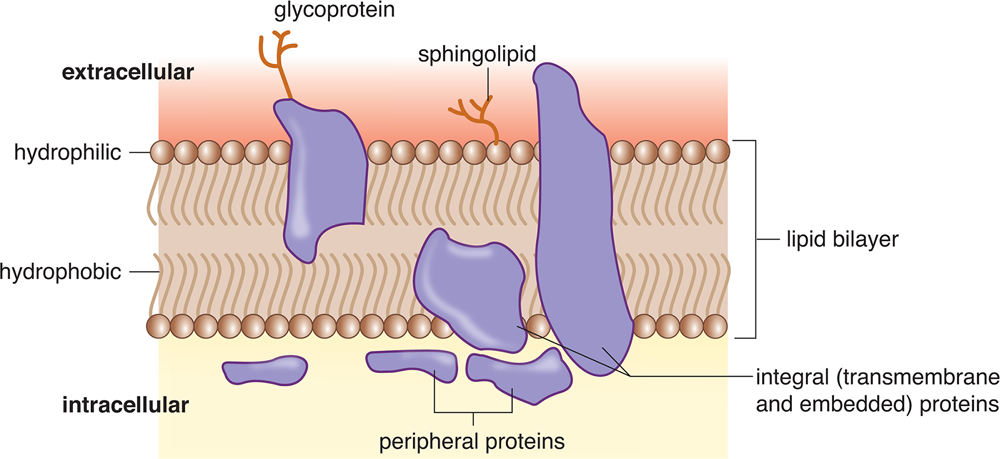
Figure 8.7 Plasma Membrane Proteins
Carbohydrates
Carbohydrates are generally attached to protein molecules on the extracellular surface of cells. Because carbohydrates are generally hydrophilic, interactions between glycoproteins and water can form a coat around the cell, as shown in Figure 8.8. In addition, carbohydrates can act as signaling and recognition molecules. For example, blood group (ABO) antigens on red blood cells are sphingolipids that differ only in their carbohydrate sequence. Our immune systems and some pathogens take advantage of these membrane carbohydrates and membrane proteins to target particular cells.
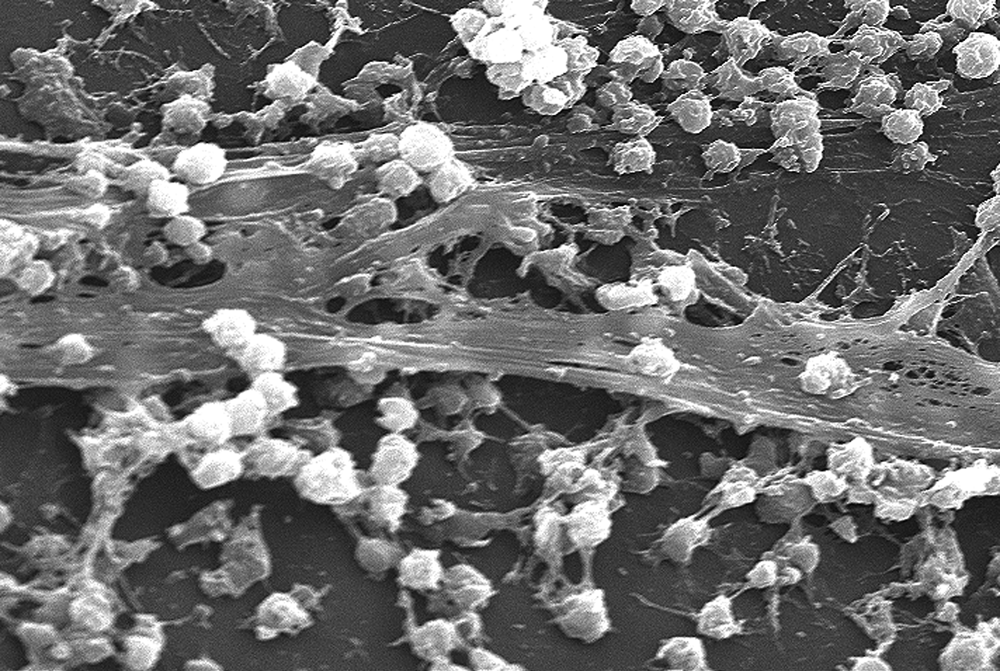
Figure 8.8 Extracellular Membrane-Associated Carbohydrates Staphylococcus aureus bacteria embedded in bands of extracellular polysaccharides and glycolipids, forming a biofilm
Membrane Receptors
Some of the transporters for facilitated diffusion and active transport can be activated or deactivated by membrane receptors, which tend to be transmembrane proteins. For example, ligand-gated ion channels are membrane receptors that open a channel in response to the binding of a specific ligand. Other membrane receptors participate in biosignaling; for example, G protein-coupled receptors are involved in several different signal transduction cascades. Membrane receptors are generally proteins, although there are some carbohydrate and lipid receptors, especially in viruses.
BRIDGE
Biosignaling is a major function of the cell membrane. Receptors and signal cascades are covered in more detail in Chapter 3 of MCAT Biochemistry Review.
Cell–Cell Junctions
Cells within tissues can form a cohesive layer via intercellular junctions. These junctions provide direct pathways of communication between neighboring cells or between cells and the extracellular matrix. Cell–cell junctions are generally comprised of cell adhesion molecules (CAM), which are proteins that allow cells to recognize each other and contribute to proper cell differentiation and development.
Gap Junctions
Gap junctions allow for direct cell–cell communication and are often found in small bunches together. Gap junctions are also called connexons and are formed by the alignment and interaction of pores composed of six molecules of connexin, as shown in Figure 8.9. They permit movement of water and some solutes directly between cells. Proteins are generally not transferred through gap junctions.
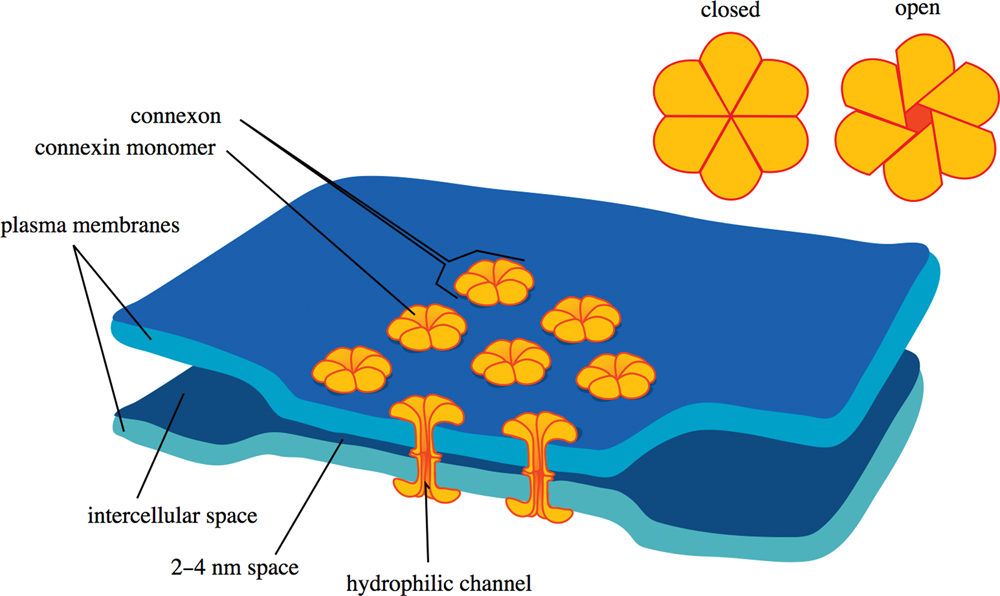
Figure 8.9 Gap Junction A connexon (gap junction) is composed of six monomers of connexin and permits travel of solutes between cells.
Tight Junctions
Tight junctions prevent solutes from leaking into the space between cells via a paracellular route. Tight junctions are found in epithelial cells and function as a physical link between the cells as they form a single layer of tissue. Tight junctions can limit permeability enough to create a transepithelial voltage difference based on differing concentrations of ions on either side of the epithelium. To be effective, tight junctions must form a continuous band around the cell; otherwise, fluid could leak through spaces between tight junctions.
MNEMONIC
Tight junctions form a water tight seal, preventing paracellular transport of water and solutes.
BRIDGE
Tight junctions are found in the lining of renal tubules, where they restrict passage of solutes and water without cellular control. Nephrons are discussed in Chapter 10 of MCAT Biology Review.
Desmosomes
Desmosomes bind adjacent cells by anchoring to their cytoskeletons. Desmosomes are formed by interactions between transmembrane proteins associated with intermediate filaments inside adjacent cells, as shown in Figure 8.10. Desmosomes are primarily found at the interface between two layers of epithelial tissue. Hemidesmosomes have a similar function, but their main function is to attach epithelial cells to underlying structures, especially the basement membrane.

Figure 8.10 Desmosomes Between Adjacent Cells
BIOCHEMISTRY GUIDED EXAMPLE WITH EXPERT THINKING
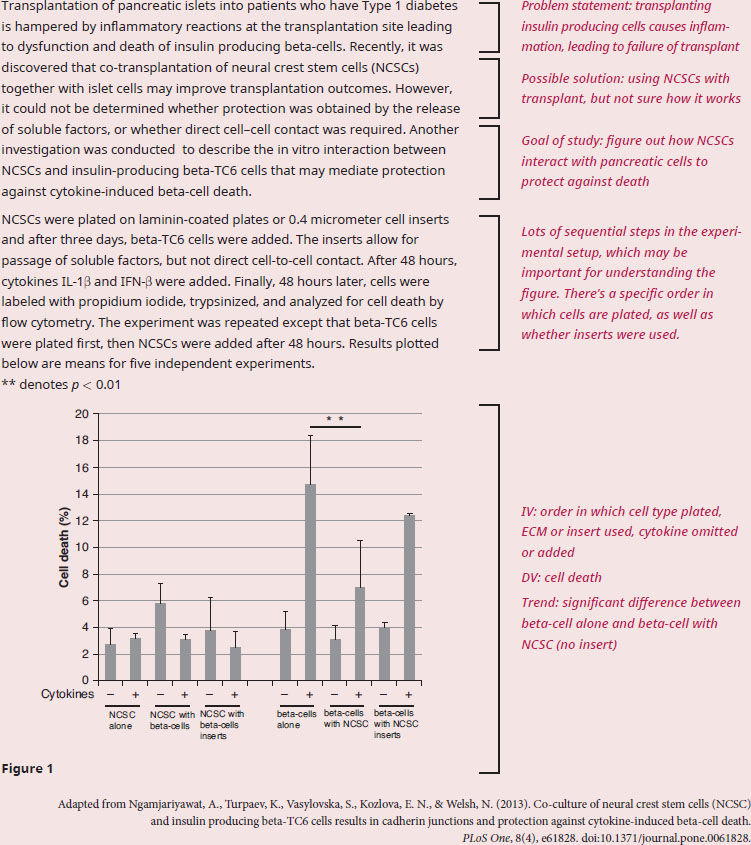
Based on this experiment, are NCSCs protecting beta islet cells from cytokine-induced cell death through direct contact, or through indirect contact?
Since the question requires data from the figure, we need to have a solid understanding of the results . The experimental question is asking whether beta-cells require direct contact with NCSCs, or whether the NCSCs are secreting soluble factors that are mediating protection from cytokine-mediated cell death. We'll need to refer to the passage for more information on these phenomena and how they interact. In the introduction to the experiment we can see that we are learning about co-transplantation of NCSCs and beta-cells, and that the experiments are trying to determine why co-transplantation is more effective than transplanting beta-cells alone. The way this was tested in the experimental setup is through the usage of cell inserts, which we are told in paragraph 2 will allow for the passage of factors, but not cell-cell contact.
The results on the left (first 6 bars) correspond to where NCSCs are plated first, then beta-cells are added with and without inserts. All 6 bars are roughly equal and the addition of cytokines do not seem to induce additional cell death, meaning these results cannot be used to make any conclusions since the expected control response (beta-cells inducing cell death in the presence of cytokines) is not shown. Taking a look at the next six columns in the graph, we can see that these are the experiments with the beta-cells being plated first. NCSCs were added 48 hours later, either with or without insert. The graph tells us that in the presence of cytokines, beta-cells alone show a high percentage of cell death (the expected control response), but having the NCSCs without insert shows a significant drop in cell death. This effect goes away when the insert is reintroduced: beta-cells with the NCSC insert have a higher level of cell death, though it’s unclear whether that difference is significant.
Because the inserts obstruct cell-to-cell contact, we can conclude that direct contact between beta-cells and NCSCs is required for protection against cytokine-mediated cell death.
MCAT CONCEPT CHECK 8.2:
Before you move on, assess your understanding of the material with these questions.
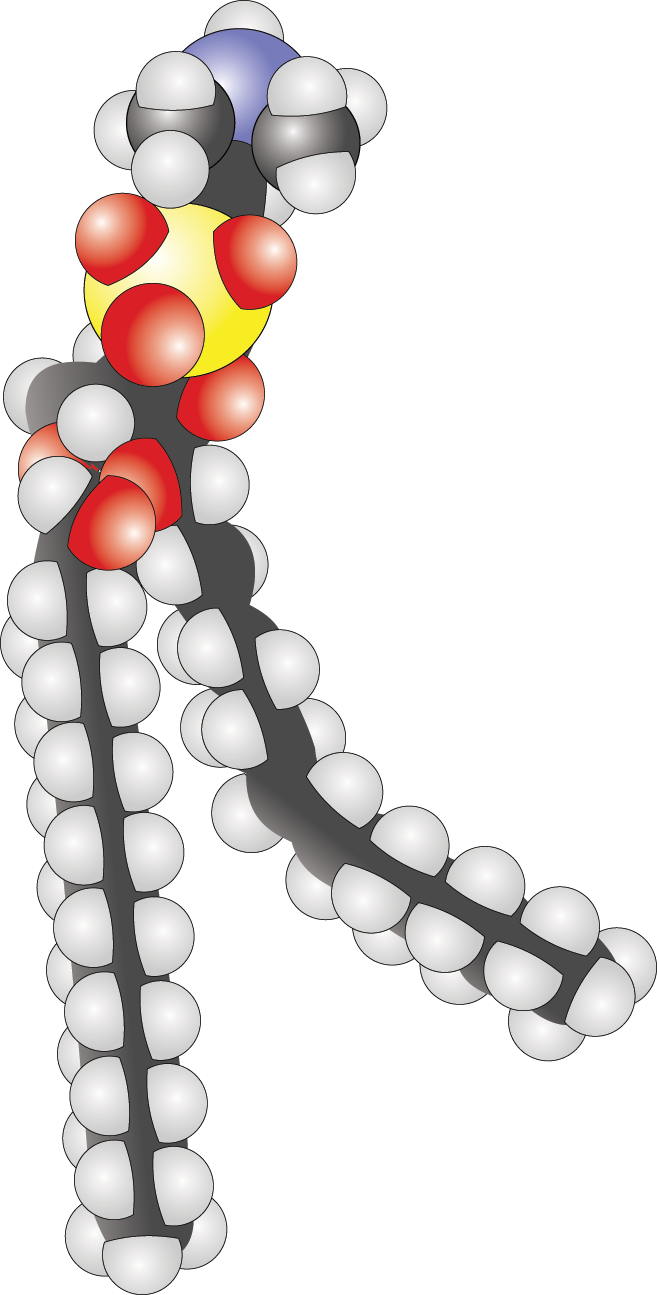
- In the following phospholipid, determine whether the fatty acids are saturated or unsaturated and label their hydrophobic and hydrophilic regions.
- How does cholesterol play a role in the fluidity and stability of the plasma membrane?
__________________________________
- What are the three classes of membrane proteins? How are they each most likely to function?
- _________________________________________
- _________________________________________
- _________________________________________
- Contrast gap junctions and tight junctions.
- Gap junctions: _____________________________________
- Tight junctions: _____________________________________
8.3 Membrane Transport
LEARNING OBJECTIVES
After Chapter 8.3, you will be able to:
- Explain the driving factors behind passive transport mechanisms
- Contrast symport and antiport mechanisms for active transport
- Relate osmotic pressure to the direction of osmosis:
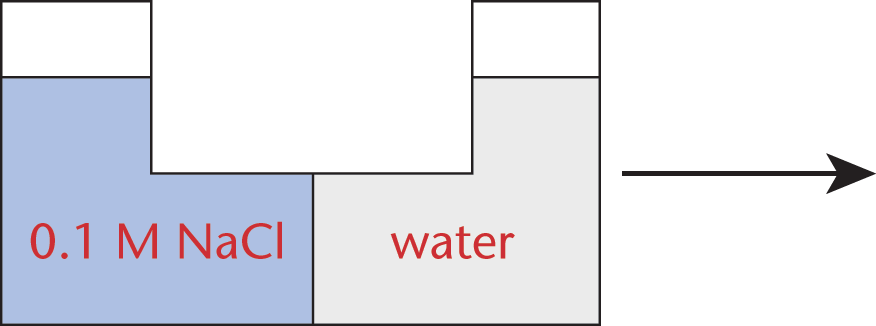
The cell membrane functions to control movement of substances into and out of the cell; however, it varies in its selectivity for different substances. Transport of small nonpolar molecules occurs rapidly through the cell membrane via diffusion, while ions and larger molecules require more specialized transport processes. The different membrane traffic processes are classified as either active or passive, and are driven by concentration gradients or intracellular energy stores.
Concentration Gradients
Transport processes can be classified as active or passive depending on their thermodynamics. Spontaneous processes that do not require energy (negative ΔG) proceed through passive transport, while those that are nonspontaneous and require energy (positive ΔG) proceed through active transport. Diffusion, facilitated diffusion, and osmosis generally increase in rate as temperature increases, while active transport may or may not be affected by temperature, depending on the enthalpy (ΔH) of the process. The primary thermodynamic motivator in most passive transport is an increase in entropy (ΔS).
MCAT EXPERTISE
An important point to keep in mind is that all transmembrane movement is based on concentration gradients, which are an MCAT favorite; understanding concentration gradients will net you points on Test Day. Remember that the gradient will tell us whether this process will be passive or active.
Passive Transport
Passive transport processes are those that do not require intracellular energy stores but rather utilize the concentration gradient to supply the energy for particles to move.
Simple Diffusion
The most basic of all membrane traffic processes is simple diffusion, in which substrates move down their concentration gradient directly across the membrane. Only particles that are freely permeable to the membrane are able to undergo simple diffusion. There is potential energy in a chemical gradient; some of this energy is dissipated as the gradient is utilized during simple diffusion. We can liken this process to a ball rolling down a hill: there is potential energy in the ball when it sits at the top of the hill, and as the ball spontaneously rolls down the hill, some of the energy is dissipated.
Osmosis
Osmosis is a specific kind of simple diffusion that concerns water; water will move from a region of lower solute concentration to one of higher solute concentration. That is, it will move from a region of higher water concentration (more dilute solution) down its gradient to a region of lower water concentration (more concentrated solution). Osmosis is important in several places, most notably when the solute itself is impermeable to the membrane. In such a case, water will move to try to bring solute concentrations to equimolarity, as shown in Figure 8.11. If the concentration of solutes inside the cell is higher than the surrounding solution, the solution is said to be hypotonic; such a solution will cause a cell to swell as water rushes in, sometimes to the point of bursting (lysing). A solution that is more concentrated than the cell is termed a hypertonic solution, and water will move out of the cell. If the solutions inside and outside are equimolar, they are said to be isotonic. A key point here is that isotonicity does not prevent movement; rather, it prevents the net movement of particles. Water molecules will continue to move; however, the cell will neither gain nor lose water overall.
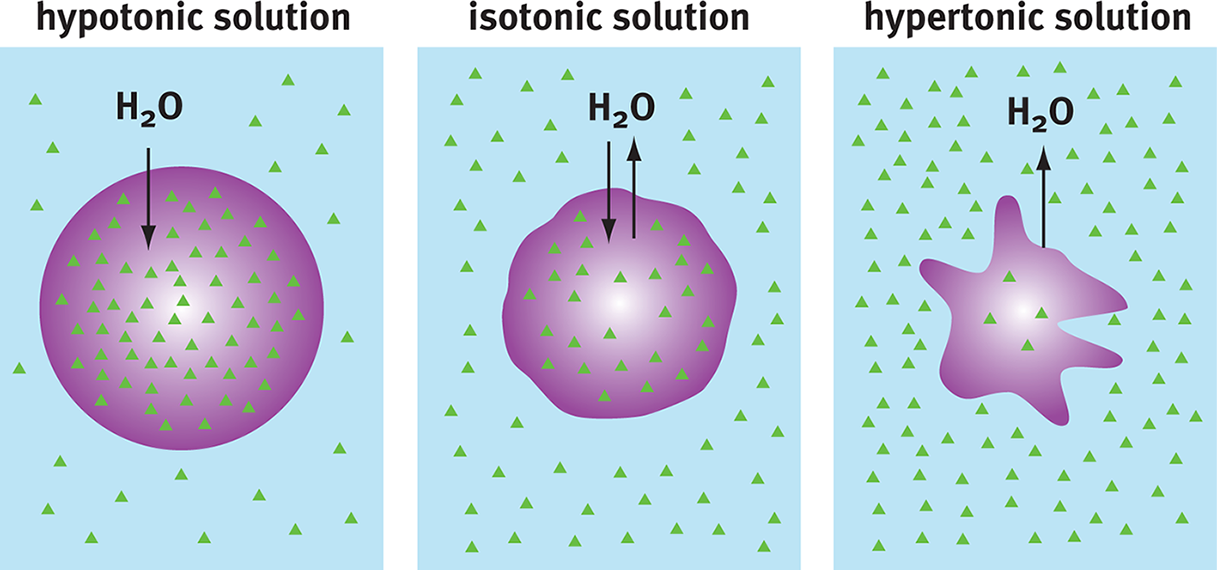
Figure 8.11 Osmosis Water moves from areas of low solute (high water) concentration to high solute (low water) concentration.
REAL WORLD
Osmolarity explains why pure water should never be given intravenously for resuscitation. Red blood cells have an osmolarity around 300
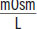
, while pure water has an osmolarity of 0
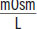
. Water would rush into the red blood cells, causing them to burst. To avoid this, saline or dextrose-containing solutions are used.
MNEMONIC
To remember that water flows into a cell placed in hypOtonic solution, imagine the cell swelling to form a giant letter O.
One method of quantifying the driving force behind osmosis is osmotic pressure. Osmotic pressure is a colligative property: a physical property of solutions that is dependent on the concentration of dissolved particles but not on the chemical identity of those dissolved particles. Other examples of colligative properties include vapor pressure depression (Raoult’s Law), boiling point elevation, and freezing point depression.
To illustrate osmotic pressure, consider a container separated into two compartments by a semipermeable membrane, just like the membranes in our cells. One compartment contains pure water, while the other contains water with dissolved solutes. The membrane allows water but not solutes to pass through. Because substances tend to flow, or diffuse, from higher to lower concentration (which results in an increase in entropy), water will diffuse from the compartment containing pure water into the compartment containing the water–solute mixture. This net flow will cause the water level in the compartment containing the solution to rise above the level in the compartment containing pure water, as shown in Figure 8.12.

Figure 8.12 Change in Water Level Due to Osmotic Pressure
Because the solute cannot pass through the membrane, the concentrations of solute in the two compartments can never be equal. However, the hydrostatic pressure exerted by the water level in the solute-containing compartment will eventually oppose the influx of water; thus, the water level will only rise to the point at which it exerts a sufficient pressure to counterbalance the tendency of water to flow across the membrane. This pressure, defined as the osmotic pressure (Π) of the solution, is given by the formula:
Π = iMRT
Equation 8.1
where M is the molarity of the solution, R is the ideal gas constant, T is the absolute temperature (in kelvins), i is the van ’t Hoff factor, which is simply the number of particles obtained from the molecule when in solution. For example, glucose remains one intact molecule, so iglucose = 1; sodium chloride becomes two ions (Na+ and Cl–), so iNaCl = 2. The equation clearly shows that osmotic pressure is directly proportional to the molarity of the solution. Thus, osmotic pressure, like all colligative properties, depends only on the presence and number of particles in solution, but not their actual identity.
In cells, the osmotic pressure is maintained against the cell membrane, rather than the force of gravity. If the osmotic pressure created by the solutes within a cell exceeds the pressure that the cell membrane can withstand, the cell will lyse. Generally, osmotic pressure is best thought of as a “sucking” pressure, drawing water into the cell in proportion to the concentration of the solution.
Facilitated Diffusion
Facilitated diffusion is simple diffusion for molecules that are impermeable to the membrane (large, polar, or charged); the energy barrier is too high for these molecules to cross freely. Facilitated diffusion requires integral membrane proteins to serve as transporters or channels for these substrates.
KEY CONCEPT
Unless otherwise specified, semipermeable membrane refers to a membrane governed by the same permeability rules as biological membranes: small, nonpolar, lipid-soluble particles (and water) can pass through freely, while large, polar, or charged particles cannot.
The classic examples of facilitated diffusion involve a carrier or channel protein. Carriers are only open to one side of the cell membrane at any given point. This model is similar to a revolving door because the substrate binds to the transport protein (walks in), remains in the transporter during a conformational change (spins), and then finally dissociates from the substrate-binding site of the transporter (walks out). Binding of the substrate molecule to the transporter protein induces a conformational change; for a brief time, the carrier is in the occluded state, in which the carrier is not open to either side of the phospholipid bilayer. In addition to carriers, channels are also viable transporters for facilitated diffusion. Channels may be in an open or closed conformation. In their open conformation, channels are exposed to both sides of the cell membrane and act like a tunnel for the particles to diffuse through, thereby permitting much more rapid transport kinetics. The activity of the three main types of ion channels is discussed in Chapter 3 of MCAT Biochemistry Review. Figure 8.13 shows the different types of passive transport, including simple diffusion and facilitated diffusion.
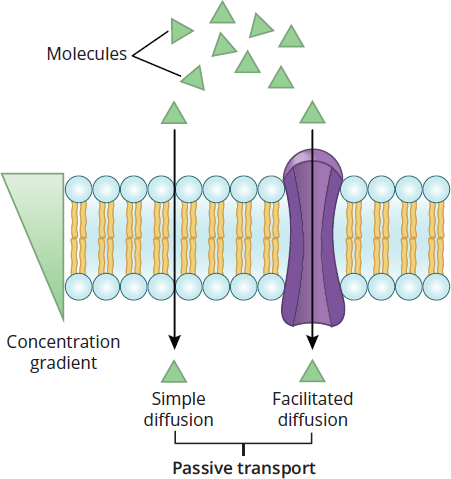
Figure 8.13 Passive Transport Processes The passive movement of solutes across the cell membrane is mediated by concentration gradients.
Active Transport
Active transport results in the net movement of a solute against its concentration gradient, just like rolling a ball uphill. Active transport always requires energy, but the source of this energy can vary. Primary active transport uses ATP or another energy molecule to directly power the transport of molecules across a membrane. Generally, primary active transport involves the use of a transmembrane ATPase. Secondary active transport, also known as coupled transport, also uses energy to transport particles across the membrane; however, in contrast to primary active transport, there is no direct coupling to ATP hydrolysis. Instead, secondary active transport harnesses the energy released by one particle going down its electrochemical gradient to drive a different particle up its gradient. When both particles flow the same direction across the membrane, it is termed symport. When the particles flow in opposite directions, it is called antiport. Active transport is important in many tissues. For instance, primary active transport maintains the membrane potential of neurons in the nervous system. The kidneys use secondary active transport, usually driven by sodium, to reabsorb and secrete various solutes into and out of the filtrate. Figure 8.14 shows active transport, while Table 8.1 summarizes how molecules move across membranes.
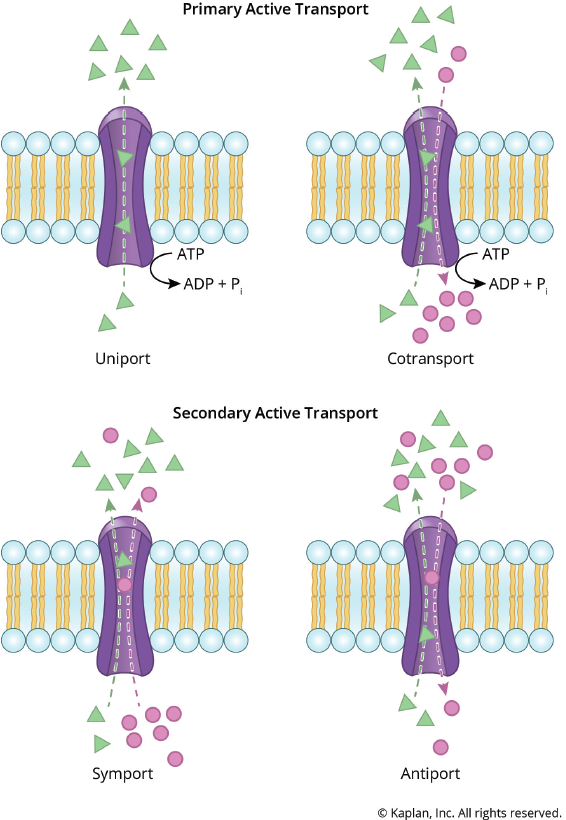
Figure 8.14 Active Transport Processes The movement of solutes against a gradient requires an input of energy.
Table 8.1. Membrane Transport Processes
SIMPLE DIFFUSION OSMOSIS FACILITATED DIFFUSION ACTIVE TRANSPORT
Concentration gradient of solute High → Low Low → High High → Low Low → High
Membrane protein required No No Yes Yes
Energy required No—this is a passive process No—this is a passive process No—this is a passive process Yes—this is an active process; requires energy
Example molecule(s) transported Small, nonpolar (O2, CO2) H2O Polar molecules (glucose) or ions (Na+, Cl−) Polar molecules or ions (Na+, Cl−, K+)
Endocytosis and Exocytosis
Endocytosis
Endocytosis occurs when the cell membrane invaginates and engulfs material to bring it into the cell. The material is encased in a vesicle, which is important because cells will sometimes ingest toxic substances. Pinocytosis is the endocytosis of fluids and dissolved particles, whereas phagocytosis is the ingestion of large solids such as bacteria. Substrate binding to specific receptors embedded within the plasma membrane will initiate the process of endocytosis. Invagination will then be initiated and carried out by vesicle-coating proteins, most notably clathrin.
Exocytosis
Exocytosis occurs when secretory vesicles fuse with the membrane, releasing material from inside the cell to the extracellular environment. Exocytosis is important in the nervous system and intercellular signaling. For instance, exocytosis of neurotransmitters from synaptic vesicles is a crucial aspect of neuron physiology. Both endo- and exocytosis are illustrated in Figure 8.15.

Figure 8.15 Endocytosis and Exocytosis
MCAT CONCEPT CHECK 8.3:
Before you move on, assess your understanding of the material with these questions.
- What is the primary thermodynamic factor responsible for passive transport?
____________________________________
- What is the relationship between osmotic pressure and the direction of osmosis through a semipermeable membrane?
_________________________________
- Compare the two types of active transport. What is the difference between symport and antiport?
____________________________________
8.4 Specialized Membranes
LEARNING OBJECTIVES
After Chapter 8.4, you will be able to:
- Identify the channels involved in maintenance of the resting membrane potential
- Calculate resting membrane potential using the Nernst equation
- Distinguish between the different regions of the mitochondrion
The membranes of most organelles are similar to the cell membrane in both composition and general characteristics; however, it is important to note that some membranes are specialized to accomplish specific functions. For instance, the sarcolemma of muscle cells must maintain a membrane potential for muscle contraction to occur. Membrane composition may also be altered slightly, especially in the case of mitochondria.
Membrane Potential
The impermeability of the cell membrane to ions and the selectivity of ion channels both lead to an electrochemical gradient between the exterior and interior of cells. The difference in electrical potential across cell membranes is called the membrane potential, Vm. The resting potential for most cells is between –40 and –80 mV, although the potential can rise as high as +35 mV during depolarization of the cell. Maintaining membrane potential requires energy because ions may passively diffuse through the cell membrane over time using leak channels; therefore, an ion transporter or pump such as the sodium–potassium pump (Na+/K+ ATPase) regulates the concentration of intracellular and extracellular sodium and potassium ions. Chloride ions also participate in establishing membrane potential. The Nernst equation can be used to determine the membrane potential from the intra- and extracellular concentrations of the various ions:
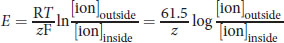
Equation 8.2
where R is the ideal gas constant, T is the temperature in kelvins, z is the charge of the ion, and F is the Faraday constant
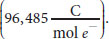
The simplification to 61.5 in the numerator assumes body temperature, 310 K. The Goldman–Hodgkin–Katz voltage equation flows from the Nernst equation, taking into account the relative contribution of each major ion to the membrane potential:
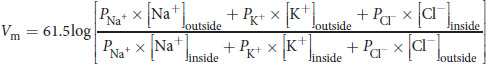
Equation 8.3
where P represents the permeability for the relevant ion. Note that chloride is inverted relative to the other ions because it carries a negative charge.
BRIDGE
The cell membrane is often compared to a capacitor because opposite charges are maintained on either side of the membrane. Capacitance is discussed in Chapter 6 of MCAT Physics and Math Review.
Sodium–Potassium Pump
There is a steady-state resting relationship between ion diffusion and the Na+/K+ ATPase. One of the main functions of the Na+/K+ ATPase is to maintain a low concentration of sodium ions and high concentration of potassium ions intracellularly by pumping three sodium ions out for every two potassium ions pumped in. This movement of ions removes one positive charge from the intracellular space of the cell, which maintains the negative resting potential of the cell. As mentioned before, the cell membrane also contains leak channels that allow ions, such as Na+ and K+, to passively diffuse into or out of the cell down their concentration gradients. Cell membranes are more permeable to K+ ions than Na+ ions at rest because there are more K+ leak channels than Na+ leak channels. The combination of Na+/K+ ATPase activity and leak channels together maintain a stable resting membrane potential.
Mitochondrial Membranes
Mitochondria are referred to as the “powerhouse” of the cell because of their ability to produce ATP by oxidative respiration. Mitochondria contain two membranes: the inner and outer mitochondrial membranes.
Outer Mitochondrial Membrane
The outer mitochondrial membrane is highly permeable due to many large pores that allow the passage of ions and small proteins. The outer membrane completely surrounds the inner mitochondrial membrane, with the presence of a small intermembrane space in between the two layers.
Inner Mitochondrial Membrane
The inner mitochondrial membrane has a much more restricted permeability compared to the outer mitochondrial membrane. Structurally, the inner mitochondrial membrane contains numerous infoldings, known as cristae, which increase the available surface area for the integral proteins associated with the membrane. These proteins, discussed in Chapter 10 of MCAT Biochemistry Review, are involved in the electron transport chain and ATP synthesis. The inner membrane also encloses the mitochondrial matrix, where the citric acid cycle produces high-energy electron carriers used in the electron transport chain. The inner mitochondrial membrane contains a very high level of cardiolipin and does not contain cholesterol.
MCAT CONCEPT CHECK 8.4:
Before you move on, assess your understanding of the material with these questions.
- How is the resting membrane potential maintained?
_________________________________
_________________________________
_________________________________
- Given the following data, calculate the resting membrane potential of this cell:
Ion Permeability (Relative) Intracellular Concentration Extracellular Concentration Na+ 0.05 14 mM 140 mM
K+ 1 120 mM 4 mM
Cl− 0 12 mM 120 mM
_________________________________
_________________________________
_________________________________
- What distinguishes the inner mitochondrial membrane from other biological membranes? What is the pH gradient between the cytoplasm and the intermembrane space?
_________________________________
_________________________________
_________________________________
Conclusion
Understanding biological membranes becomes increasingly important as you progress in your medical career. At this point, you should have a strong foundation of knowledge about the fluid mosaic model and how membranes exist dynamically. We’ve also covered the components of cell membranes, with a special emphasis on lipids and the phospholipid bilayer. We reviewed some basic physical properties of the cell, including cell–cell junctions. We also examined membrane transport, such as passive transport (simple diffusion, facilitated diffusion, and osmosis) and active transport, before briefly touching upon endocytosis and exocytosis. Finally, we reviewed specialized membranes within cells. Up to now, you have been exposed to each of the classes of molecules and some of their applications both experimentally and within the cell. This comprehensive review should provide you with a better understanding of what will be expected of you on Test Day and briefly introduce you to topics that you will learn more about in medical school.
The first seven chapters of MCAT Biochemistry Review focused on various types of biomolecules, their structures, and their functions. In this chapter, we applied this knowledge of biomolecules to make sense of biological membranes. In the remaining four chapters, we’ll turn our attention to the metabolic pathways by which the body builds, stores, and burns these biomolecules.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Fluid Mosaic Model
- The fluid mosaic model accounts for the presence of lipids, proteins, and carbohydrates in a dynamic, semisolid plasma membrane that surrounds cells.
- The plasma membrane contains proteins embedded within the phospholipid bilayer.
- The membrane is not static.
- Lipids move freely in the plane of the membrane and can assemble into lipid rafts.
- Flippases are specific membrane proteins that maintain the bidirectional transport of lipids between the layers of the phospholipid bilayer in cells.
- Proteins and carbohydrates may also move within the membrane, but are slowed by their relatively large size.
Membrane Components
- Lipids are the primary membrane component, both by mass and mole fraction.
- Triacylglycerols and free fatty acids act as phospholipid precursors and are found in low levels in the membrane.
- Glycerophospholipids replace one fatty acid with a phosphate group, which is often linked to other hydrophilic groups.
- Cholesterol is present in large amounts and contributes to membrane fluidity and stability.
- Waxes are present in very small amounts, if at all; they are most prevalent in plants and function in waterproofing and defense.
- Proteins located within the cell membrane act as transporters, cell adhesion molecules, and enzymes.
- Transmembrane proteins can have one or more hydrophobic domains and are most likely to function as receptors or channels.
- Embedded proteins are most likely part of a catalytic complex or involved in cellular communication.
- Membrane-associated proteins may act as recognition molecules or enzymes.
- Carbohydrates can form a protective glycoprotein coat and also function in cell recognition.
- Extracellular ligands can bind to membrane receptors, which function as channels or enzymes in second messenger pathways.
- Cell–cell junctions regulate transport intracellularly and intercellularly.
- Gap junctions allow for the rapid exchange of ions and other small molecules between adjacent cells.
- Tight junctions prevent paracellular transport, but do not provide intercellular transport.
- Desmosomes and hemidesmosomes anchor layers of epithelial tissue together.
Membrane Transport
- Concentration gradients help to determine appropriate membrane transport mechanisms in cells.
- Osmotic pressure, a colligative property, is the pressure applied to a pure solvent to prevent osmosis and is used to express the concentration of the solution.
- It is often better conceptualized as a “sucking” pressure in which a solution is drawing water in, proportional to its concentration.
- Passive transport does not require energy because the molecule is moving down its concentration gradient or from an area with higher concentration to an area with lower concentration.
- Simple diffusion does not require a transporter. Small, nonpolar molecules passively move from an area of high concentration to an area of low concentration until equilibrium is achieved.
- Osmosis describes the diffusion of water across a selectively permeable membrane.
- Facilitated diffusion uses transport proteins to move impermeable solutes across the cell membrane.
- Active transport requires energy in the form of ATP or an existing favorable ion gradient.
- Active transport may be primary or secondary depending on the energy source. Secondary active transport can be further classified as symport or antiport.
- Endocytosis and exocytosis are methods of engulfing material into cells or releasing material to the exterior of cells, both via the cell membrane. Pinocytosis is the ingestion of liquid into the cell in vesicles formed from the cell membrane and phagocytosis is the ingestion of larger, solid molecules.
Specialized Membranes
- The composition of cell membranes is fairly consistent; however, there are some cells that contain specialized membranes.
- Membrane potential is maintained by the sodium–potassium pump and leak channels.
- The electrical potential created by one ion can be calculated using the Nernst equation.
- The resting potential of a membrane at physiological temperature can be calculated using the Goldman–Hodgkin–Katz voltage equation, which is derived from the Nernst equation.
- The mitochondrial membrane differs from the cell membrane:
- The outer mitochondrial membrane is highly permeable to metabolic molecules and small proteins.
- The inner mitochondrial membrane surrounds the mitochondrial matrix, where the citric acid cycle produces electrons used in the electron transport chain and where many other enzymes important in cellular respiration are located. The inner mitochondrial membrane also does not contain cholesterol.
ANSWERS TO CONCEPT CHECKS
**8.1**
- Flippases are responsible for the movement of phospholipids between the layers of the plasma membrane because it is otherwise energetically unfavorable. Lipid rafts are aggregates of specific lipids in the membrane that function as attachment points for other biomolecules and play roles in signaling.
- Lipids, including phospholipids, cholesterol, and others, are most plentiful; proteins, including transmembrane proteins (channels and receptors), membrane-associated proteins, and embedded proteins, are next most plentiful; carbohydrates, including the glycoprotein coat and signaling molecules, are next; nucleic acids are essentially absent.
**8.2**
- The hydrophilic region is at the top of this diagram. While you need not be able to recognize it, the head group is phosphatidylcholine in this example. The hydrophobic region is at the bottom and is composed of two fatty acid tails. The tail on the left is saturated; the tail on the right is unsaturated, as evidenced by the kink in its chain.
- Cholesterol moderates membrane fluidity by interfering with the crystal structure of the cell membrane and occupying space between phospholipid molecules at low temperatures, and by restricting excessive movement of phospholipids at high temperatures. Cholesterol also provides stability by cross-linking adjacent phospholipids through interactions at the polar head group and hydrophobic interactions at the nearby fatty acid tail.
- Transmembrane proteins are most likely to serve as channels or receptors. Embedded membrane proteins are most likely to have catalytic activity linked to nearby enzymes. Membrane-associated (peripheral) proteins are most likely to be involved in signaling or are recognition molecules on the extracellular surface.
- Gap junctions allow for the intercellular transport of materials and do not prevent paracellular transport of materials. Tight junctions are not used for intercellular transport but do prevent paracellular transport. Gap junctions are in discontinuous bunches around the cell, while tight junctions form bands around the cell.
**8.3**
- The primary thermodynamic factor responsible for passive transport is entropy.
- As osmotic pressure increases, more water will tend to flow into the compartment to decrease solute concentration. Osmotic pressure is often considered a “sucking” pressure because water will move toward the compartment with the highest osmotic pressure.
- Primary active transport uses ATP as an energy source for the movement of molecules against their concentration gradient, while secondary active transport uses an electrochemical gradient to power the transport. Symport moves both particles in secondary active transport across the membrane in the same direction, while antiport moves particles across the cell membrane in opposite directions.
**8.4**
- The membrane potential, which results from a difference in the number of positive and negative charges on either side of the membrane, is maintained primarily by the sodium–potassium pump, which moves three sodium ions out of the cell for every two potassium ions pumped in, and to a minor extent by leak channels that allow the passive transport of ions.
-
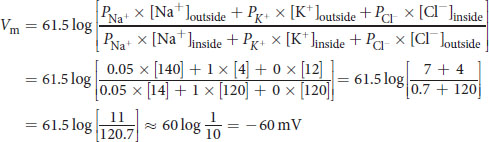
The exact value is – 64.0 mV
- The inner mitochondrial membrane lacks cholesterol, which differentiates it from most other biological membranes. There is no pH gradient between the cytoplasm and the intermembrane space because the outer mitochondrial membrane has such high permeability to biomolecules (the proton-motive force of the mitochondria is across the inner mitochondrial membrane, not the outer mitochondrial membrane).
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. C
We are asked to identify the type of transport that would allow a large, polar molecule to cross the membrane without any energy expenditure. This scenario describes facilitated diffusion, which uses a transport protein (or channel) to facilitate the movement of large, polar molecules across the nonpolar, hydrophobic membrane. Facilitated diffusion, like simple diffusion, does not require energy.
2. C
This question requires an understanding of osmosis and the action of the sodium–potassium pump. When a cell is placed in a hypertonic solution (a solution having a higher solute concentration than the cell), fluid will diffuse out of the cell and result in cell shrinkage. When a cell is placed in hypotonic solution (a solution having a lower solute concentration than the cell), fluid will diffuse from the solution into the cell, causing the cell to expand and possibly lyse. The sodium–potassium pump moves three sodium ions out of the cell for every two potassium ions it lets into the cell. Therefore, inhibition of the sodium–potassium pump by ouabain will cause a net increase in the sodium concentration inside the cell and water will diffuse in, causing the cell to swell and then lyse.
3. D
The polarization of the membrane at rest is the result of an uneven distribution of ions between the inside and outside of the cell. This difference is achieved through active pumping of ions (predominantly sodium and potassium) into and out of the cell and the selective permeability of the membrane, which allows only certain ions to cross.
4. B
Ribosomes are the site of protein synthesis within a cell and are not coupled to the cell membrane. The cell membrane functions as a site for cytoskeletal attachment, (A), through proteins and lipid rafts. Transport regulation, (C), is accomplished through channels, transporters, and selective permeability, while the phospholipids act as a reagent for second messenger formation, (D).
5. A
Movement of individual molecules in the cell membrane will be affected by size and polarity, just as with diffusion. Lipids are much smaller than proteins in the plasma membrane and will move more quickly. Lipids will move fastest within the plane of the cell membrane because the polar head group does not need to pass through the hydrophobic tail region in the same way that it would if it were moving between the membrane layers.
6. B
Compounds that contribute to membrane fluidity will lower the melting point or disrupt the crystal structure. Cholesterol, (C), and unsaturated lipids, (A) and (D), are known for these functions. transglycerophospholipids tend to increase the melting point of the membrane and therefore decrease membrane fluidity.
7. D
Membrane receptors must have both an extracellular and intracellular domain; therefore, they are considered transmembrane proteins. In order to initiate a second messenger cascade, they typically display enzymatic activity, although some may act strictly as channels.
8. C
Plasmodesmata are cell–cell junctions that are found in plants, not animals. Gap junctions, tight junctions, desmosomes, and hemidesmosomes are all found in animals, particularly in epithelia.
9. A
The movement of any molecule is dependent on multiple factors, including electric potential, membrane solubility, and concentration gradient. While the electric potential takes into account multiple molecules, the movement of any one solute or water by diffusion or osmosis is dependent only on the concentration gradient of that molecule.
10. B
The endocytosis (bulk uptake through vesicle formation) of fluid is known as pinocytosis. Phagocytosis, (A), is the endocytic intake of solids, while exocytosis, (C), is a method of releasing vesicular contents. Drinking, (D), does not apply on a cellular level.
11. C
Cell membranes are most likely to have a resting membrane potential that is nonzero because the resting membrane potential creates a state that is capable of responding to stimuli. Signaling molecules and channels would not be as useful with a membrane potential of zero. The values given in the answer choices correspond to different stages of the action potential, but the key information is that a resting potential of 0 mV does not maintain gradients for later activity.
12. B
The outer mitochondrial membrane is very permeable while the inner membrane is highly impermeable. The inner mitochondrial membrane is unique within the cell because it lacks cholesterol.
13. D
The Nernst equation relates the intra- and extracellular concentrations of an ion to the potential created by that gradient. At physiological temperature, it can be simplified to
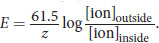
. For calcium, z= +2 (Ca2+) and the ratio of [ionoutside] to [ioninside] = 104. Plugging in, we get:
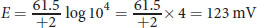
14. A
The fluid mosaic model accounts for a dynamic membrane. In this model, membrane components contain both fatty and carbohydrate-derived components, eliminating (B). Further, the membrane is stabilized by the hydrophobic interactions of both fatty acid tails and membrane proteins, which may be found on the cytosolic or extracellular side of the membrane, or may run directly through the membrane; thus, (C) and (D) are also eliminated.
15. D
Gangliosides, along with ceramide, sphingomyelin, and cerebrosides, are sphingolipids.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(8.1) Osmotic pressure: Π = iMRT
(8.2) Nernst equation:
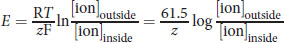
(8.3) Goldman–Hodgkin–Katz voltage equation:
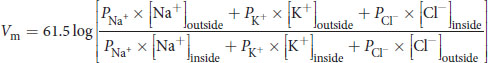
SHARED CONCEPTS
- Biochemistry Chapter 3
- Nonenzymatic Protein Function and Protein Analysis
- Biochemistry Chapter 5
- Lipid Structure and Function
- Biology Chapter 1
- The Cell
- Biology Chapter 10
- Homeostasis
- General Chemistry Chapter 9
- Solutions
- Physics and Math Chapter 5
- Electrostatics