Chapter 9: Carbohydrate Metabolism I: Glycolysis, Glycogen, Gluconeogenesis, and the Pentose Phosphate Pathway

Chapter 9: Carbohydrate Metabolism I: Glycolysis, Glycogen, Gluconeogenesis, and the Pentose Phosphate Pathway
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- Which of the following transporters is used by cells in the liver to store excess glucose and by beta cells in the pancreas as a glucose sensor?
- GLUT 1
- GLUT 2
- GLUT 3
- GLUT 4
- Which of the following organs does NOT require a constant supply of glucose from the blood for energy during a fast?
- Red blood cells
- Brain
- Pancreas
- Liver
- When insulin is released, it acts to increase the absorption of glucose into skeletal muscle predominantly through which of the following transporters?
- GLUT 1
- GLUT 2
- GLUT 3
- GLUT 4
- After an overnight fast, which of the following enzymes would be expected to have little, if any, physiological activity?
- Malate dehydrogenase
- Glucokinase
- α-Ketoglutarate dehydrogenase
- Phosphofructokinase-1
- Which of the following enzymes is NOT used to trap sugar in the cell?
- Hexokinase
- Galactokinase
- Phosphofructokinase
- Fructokinase
- When fatty acid β-oxidation predominates in the liver, mitochondrial pyruvate is most likely to be:
- carboxylated to phosphoenolpyruvate for entry into gluconeogenesis.
- oxidatively decarboxylated to acetyl-CoA for oxidation in the citric acid cycle.
- carboxylated to oxaloacetate for entry into gluconeogenesis.
- reduced to lactate in the process of fermentation.
- A biopsy is done on a child with an enlarged liver and shows accumulation of glycogen granules with single glucose residues remaining at the branch points near the periphery of the granule. The most likely genetic defect is in the gene encoding:
- α-1,4 phosphorylase (glycogen phosphorylase).
- α-1,4:α-1,6 transferase (branching enzyme).
- α-1,4:α-1,4 transferase (part of debranching enzyme complex).
- α-1,6 glucosidase (part of debranching enzyme complex).
- An investigator is measuring the activity of various enzymes involved in reactions of intermediary metabolism. One of the enzymes has greatly decreased activity compared to reference values. The buffer of the assay contains citrate. Which of the following enzymes will most likely be directly affected by the use of citrate?
- Fructose-2,6-bisphosphatase
- Isocitrate dehydrogenase
- Phosphofructokinase-1
- Pyruvate carboxylase
- After a brief period of intense exercise, the activity of muscle pyruvate dehydrogenase is greatly increased. This increased activity is most likely due to:
- decreased ADP.
- increased acetyl-CoA.
- increased NADH/NAD+ ratio.
- increased pyruvate concentration.
- The metabolism of which of the following sugars would be LEAST affected by the inhibition of the enzyme phosphofructokinase-1?
- Lactose
- Fructose
- Glucose
- Galactose
- A patient is given antibiotics to treat a urinary tract infection and develops an episode of red blood cell lysis. Further studies show weakness of the plasma membrane and Heinz bodies (collections of oxidized hemoglobin). Which of the following enzymes is most likely defective in this patient?
- Fructose-1,6-bisphosphatase
- Glucose-6-phosphate dehydrogenase
- Hexokinase
- Pyruvate kinase
- The unique enzymes of gluconeogenesis are used to circumvent specific irreversible steps of glycolysis. Which of the following correctly pairs an enzyme from glycolysis with its corresponding enzyme(s) used in gluconeogenesis?
- Phosphofructokinase-1 / fructose-1,6-bisphosphatase
- Pyruvate dehydrogenase / pyruvate carboxylase and phosphoenolpyruvate carboxykinase
- Hexokinase / glucokinase
- Pyruvate kinase / glucose-6-phosphatase
- After an overnight fast, which of the following processes would be expected to occur at an elevated rate compared with the well-fed state?
- Glycolysis
- Glycogenolysis
- Glycogenesis
- Glycerol synthesis
- Andersen disease is caused by a mutation in branching enzyme and is associated with cirrhosis. Scientists theorize the cause is likely an immune response against abnormal glycogen. Which of the following best describes the ratio of glycogen α-1,4 to α-1,6 linkages in individuals with this disorder?
- Elevated
- Decreased
- No difference
- Cannot determine
- Each of the following catalyzes a rate-limiting step of a carbohydrate metabolism pathway EXCEPT:
- hexokinase.
- glycogen synthase.
- glucose-6-phosphate dehydrogenase.
- fructose-1,6-bisphosphatase.
Answer Key
- B
- D
- D
- B
- C
- C
- D
- C
- D
- B
- B
- A
- B
- A
- A
Chapter 9: Carbohydrate Metabolism I: Glycolysis, Glycogen, Gluconeogenesis, and the Pentose Phosphate Pathway
CHAPTER 9
CARBOHYDRATE METABOLISM I: GLYCOLYSIS, GLYCOGEN, GLUCONEOGENESIS, AND THE PENTOSE PHOSPHATE PATHWAY
In This Chapter
9.1 Glucose Transport 9.2 Glycolysis
Important Enzymes of Glycolysis
Fermentation
Important Intermediates of Glycolysis
Irreversible Enzymes
Glycolysis in Erythrocytes
9.3 Other Monosaccharides
Galactose Metabolism
Fructose Metabolism
9.4 Pyruvate Dehydrogenase 9.5 Glycogenesis and Glycogenolysis
Glycogenesis
Glycogenolysis
Glycogen Storage Diseases
9.6 Gluconeogenesis
Important Enzymes of Gluconeogenesis
9.7 The Pentose Phosphate Pathway
Functions of NADPH
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 5% of all questions about biochemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1D: Principles of bioenergetics and fuel molecule metabolism
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
Maintaining a constant blood glucose concentration around
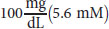
is of the utmost importance in the body: high blood sugar causes long-term damage to the retina, kidney, blood vessels, and nerves, while low blood sugar can cause autonomic disturbances, seizures, and even coma. Without the ability to take in glucose constantly, the body must find ways to store and release glucose as it is needed. And given the variety of food we eat on a daily basis, the body must find ways to use all of the various carbohydrates it takes in.
There’s a complex interplay between the neurological, endocrine, digestive, and excretory systems to maintain this blood glucose concentration, much of which is discussed in Chapter 12 of MCAT Biochemistry Review. In this chapter, we’ll take a look at the metabolic pathways that involve glucose: the methods by which our bodies digest glucose and other monosaccharides, store and release glucose for energy, generate glucose from other biomolecules, and use glucose to create some of the coenzymes and substrates needed for biosynthesis.
This chapter is the first of four that focus on metabolism in MCAT Biochemistry Review. Here, we focus on metabolic processes of glucose that do not require oxygen; in Chapter 10, we’ll turn our focus to the processes that only occur under aerobic conditions. In Chapter 11, we’ll explore the metabolism of lipids and amino acids. Finally, in Chapter 12, we’ll bring all of metabolism together with a focus on bioenergetics and the regulation of metabolism overall.
9.1 Glucose Transport
LEARNING OBJECTIVES
After Chapter 9.1, you will be able to:
- List the locations and functions of the GLUT 2 and GLUT 4 glucose transport proteins
- Predict how increased blood glucose levels will impact GLUT 2 and GLUT 4 activity
Glucose entry into most cells is driven by concentration and is independent of sodium, unlike absorption from the digestive tract. Normal glucose concentration in peripheral blood is 5.6 mM (normal range: 4–6 mM). There are four glucose transporters, called GLUT 1 through GLUT 4. GLUT 2 and GLUT 4 are the most significant of these because they are located only in specific cells and are highly regulated.
GLUT 2 is a low-affinity transporter in hepatocytes and pancreatic cells. After a meal, blood traveling through the hepatic portal vein from the intestine is rich in glucose. GLUT 2 captures the excess glucose primarily for storage. When the glucose concentration drops below the Km for the transporter, much of the remainder bypasses the liver and enters the peripheral circulation. The Km of GLUT 2 is quite high (~15 mM). This means that the liver will pick up glucose in proportion to its concentration in the blood (first-order kinetics). In other words, the liver will pick up excess glucose and store it preferentially after a meal, when blood glucose levels are high. In the β-islet cells of the pancreas, GLUT 2, along with the glycolytic enzyme glucokinase, serves as the glucose sensor for insulin release.
BRIDGE
The Km is the concentration of substrate when an enzyme is active at half of its maximum velocity (Vmax). The lower the Km, the higher the enzyme’s affinity for the substrate. See Chapter 2 of MCAT Biochemistry Review for more on Michaelis–Menten enzyme kinetics.
GLUT 4 is in adipose tissue and muscle and responds to the glucose concentration in peripheral blood. The rate of glucose transport in these two tissues is increased by insulin, which stimulates the movement of additional GLUT 4 transporters to the membrane by a mechanism involving exocytosis, as shown in Figure 9.1. The Km of GLUT 4 is close to the normal glucose levels in blood (~5 mM). This means that the transporter is saturated when blood glucose levels are just a bit higher than normal. When a person has high blood sugar concentrations, these transporters will still permit only a constant rate of glucose influx because they will be saturated (zero-order kinetics). Then how can cells with GLUT 4 transporters increase their intake of glucose? By increasing the number of GLUT 4 transporters on their surface.
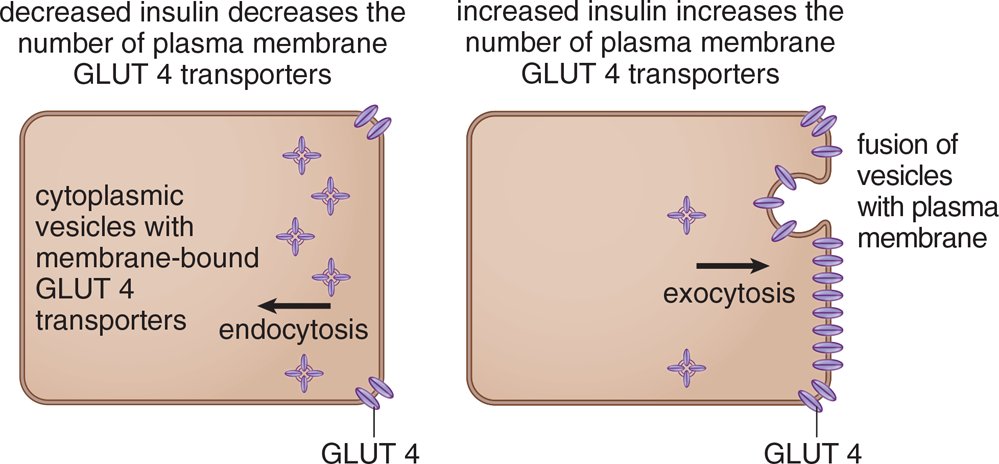
Figure 9.1 Insulin Regulation of Glucose Transport in Muscle and Adipose Cells
REAL WORLD
Diabetes mellitus is caused by a disruption of the insulin/GLUT 4 mechanism. In type 1 diabetes, insulin is absent and cannot stimulate the insulin receptor. In type 2 diabetes, the receptor becomes insensitive to insulin and fails to bring GLUT 4 transporters to the cell surface. In both cases, blood glucose rises, leading to immediate symptoms (increased urination, increased thirst, ketoacidosis) and long-term symptoms (blindness, heart attacks, strokes, nerve damage).
Although basal levels of transport occur in all cells independently of insulin, the transport rate increases in adipose tissue and muscle when insulin levels rise. Muscle stores excess glucose as glycogen, and adipose tissue requires glucose to form dihydroxyacetone phosphate (DHAP), which is converted to glycerol phosphate to store incoming fatty acids as triacylglycerols.
MCAT CONCEPT CHECK 9.1:
Before you move on, assess your understanding of the material with these questions.
- Compare and contrast GLUT 2 and GLUT 4:
**GLUT 2 GLUT 4 Important tissues Km Saturated at normal glucose levels? Responsive to insulin?**
- How does insulin promote glucose entry into cells?
________________________________________
BIOCHEMISTRY GUIDED EXAMPLE WITH EXPERT THINKING
According to the data, do the results support the usage of glycolytic inhibitors like 3BrPA to preferentially target malignant cells? Why or why not?
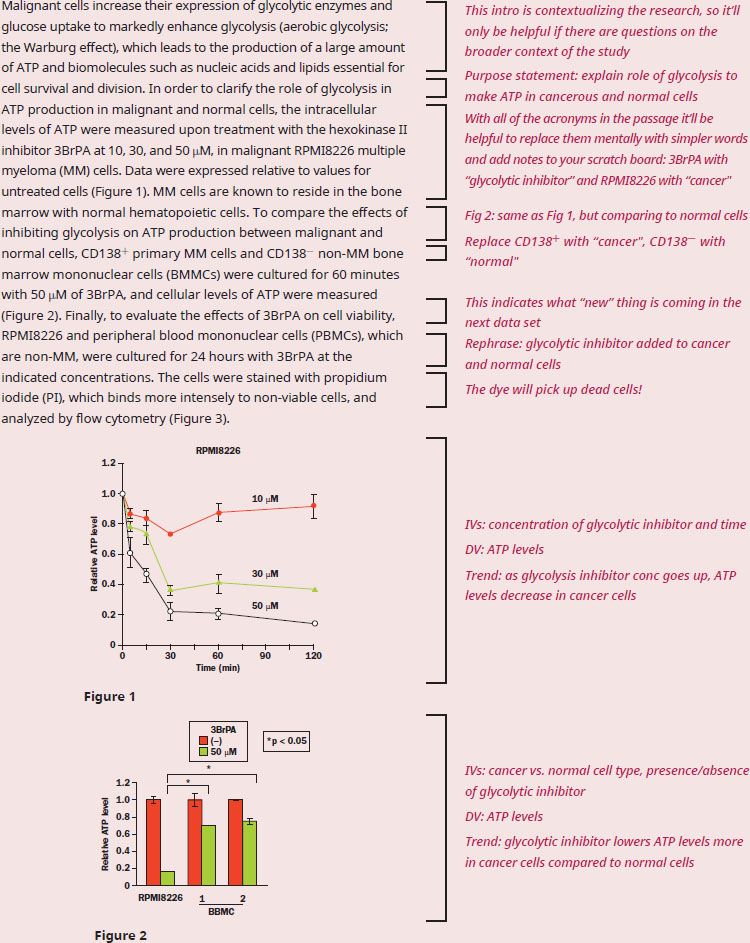
This question is going to require us to understand what this experiment is testing and how the results are gathered, so let's start by making sure we understand the passage fully. A quick scan of this passage reveals multiple challenges: lots of acronyms, unfamiliar jargon and experimental techniques, and four experimental graphs. While each piece of data will be distinct, they will all be centered around supporting the experiment’s purpose. The second sentence states that the overall purpose of this passage is to explore how cancer cells primarily use glycolysis, and how this preference can be used to specifically target tumors.
In Figure 1, we can see what's happening to ATP levels over time in multiple myeloma cells treated with hexokinase inhibitor. Looking at the data, we see that over time, the bigger the dose (written above the lines) of inhibitor, the more the ATP level in the cancer cells drops. If glycolysis is the primary way cancer cells produce ATP, which is what the passage stated in the first sentence, then the results in Figure 1 can be said to support this hypothesis.
Figure 2 shows a comparison of ATP production in the presence and absence of the inhibitor to cancer cells (RPMI8226 and CD138+) and normal cells (CD138–). While there is a drop in ATP when treated with the inhibitor in all the cell types, there is a statistical difference in the ATP levels of cancer vs. normal cells when the inhibitor is present. The brackets at the top of this figure indicate a statistically significant difference between the cancer cell line with the inhibitor and normal cell lines with the inhibitor. These kinds of statistical significance indications in a graph often point us toward where we should be heading to solve passage-based questions on the MCAT. This is the first piece of evidence that shows how inhibiting glycolysis has a bigger effect on cancer cells compared to normal, because we saw a statistically significant difference between those groups.
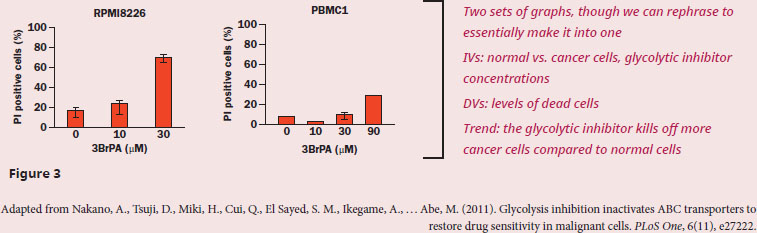
However, the most impactful proof comes in Figure 3. The cells have been treated with PI, which the passage tells us will stain non-viable (dead) cells more intensely. The cancer cells have progressively more dead cells (indicated by higher bars) with increasing amounts of the glycolytic inhibitor, which we can see in the first graph of Figure 3, while the normal cells (the second graph of Figure 3) show far fewer dead cells even at the highest dose. To directly compare, one can estimate that ~70% of cancer cells are PI positive with 30 μM 3BrPA, whereas ~10% of nonmalignant cells are PI positive with the same concentration of 3BRPA.
Because the results in Figure 3 tell us that glycolytic inhibitor 3BrPA is far more effective in killing cancerous vs. noncancerous cell lines, the findings are supportive of the idea that 3BrPA could possibly be used to target and treat cancerous cells.
9.2 Glycolysis
LEARNING OBJECTIVES
After Chapter 9.2, you will be able to:
- Recall the key steps, key intermediates, reactants, products, and key enzymes of glycolysis
- Explain the function and mechanism of lactate fermentation
- Explain the unique effects of glycolysis on hemoglobin and erythrocytes
- Recall function and regulatory mechanisms when given an enzyme of glycolysis, such as phosphoglycerate kinase
All cells can carry out glycolysis. In a few tissues, most importantly red blood cells, glycolysis represents the only energy-yielding pathway available because red blood cells lack mitochondria, which are required for the citric acid cycle, electron transport chain, oxidative phosphorylation, and fatty acid metabolism (β-oxidation). Glucose is the major monosaccharide that enters the pathway, but others such as galactose and fructose can also feed into it.
BRIDGE
Red blood cells extrude their mitochondria during development, as discussed in Chapter 7 of MCAT Biology Review. This helps them carry out their function (carrying oxygen) in two ways:
- Maximizing volume available for hemoglobin, the primary oxygen-carrying protein
- Stopping the red blood cell from utilizing the oxygen it’s supposed to be carrying to oxygen-depleted bodily tissues
Glycolysis is a cytoplasmic pathway that converts glucose into two pyruvatemolecules, releasing a modest amount of energy captured in two substrate-level phosphorylations and one oxidation reaction. If a cell has mitochondria and oxygen, the energy carriers produced in glycolysis (NADH) can feed into the aerobic respiration pathway to generate energy for the cell. If either mitochondria or oxygen is lacking (such as in erythrocytes or exercising skeletal muscle, respectively), glycolysis may occur anaerobically, although some of the available energy is lost.
Glycolysis also provides intermediates for other pathways. In the liver, glycolysis is part of the process by which excess glucose is converted to fatty acids for storage.
Important Enzymes of Glycolysis
While glycolysis contains many different steps, as illustrated in Figure 9.2, the MCAT predominantly tests on the enzymes that are highly regulated or that serve an important energetic function. Therefore, we’ll focus our attention on five of these enzymes.
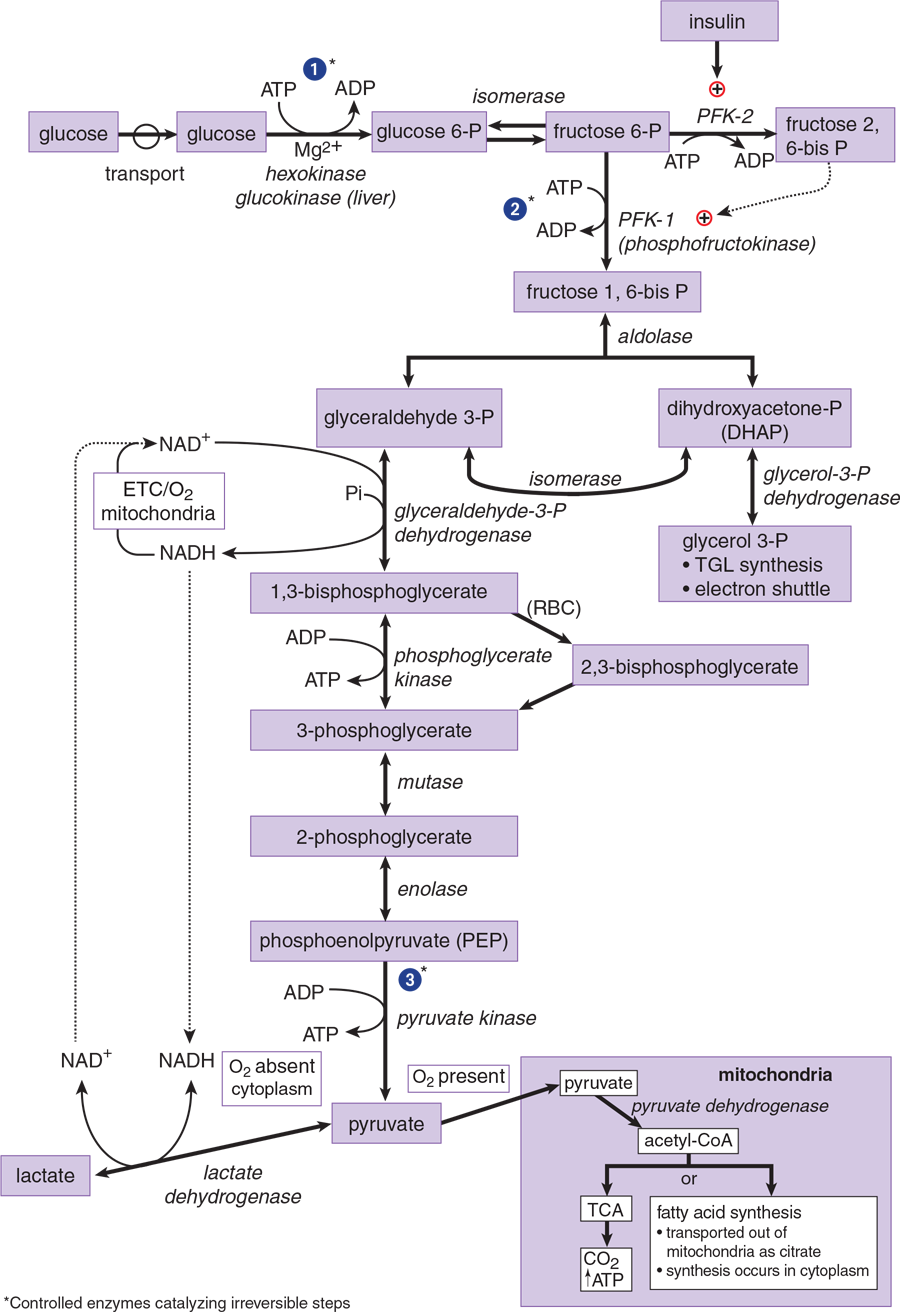
Figure 9.2 Glycolysis
REAL WORLD
Because glycolysis is necessary in every cell of the body, there are no known diseases caused by the complete absence of any enzyme in glycolysis; in other words, being unable to carry out glycolysis is incompatible with life. Partial enzyme defects are also rare, but include pyruvate kinase deficiency.
Hexokinase and Glucokinase
The first steps in glucose metabolism in any cell are transport across the membrane and phosphorylation by kinase enzymes inside the cell to prevent glucose from leaving via the transporter. Remember from Chapter 2 of MCAT Biochemistry Review that kinases attach a phosphate group from ATP to their substrates. Glucose enters the cell by facilitated diffusion or active transport; in either case, these kinases convert glucose to glucose 6-phosphate. Because the GLUT transporters are specific for glucose (not phosphorylated glucose), the glucose gets “trapped” inside the cell and cannot leak out. Hexokinase is widely distributed in tissues and is inhibited by its product, glucose 6-phosphate. Glucokinase is found only in liver cells and pancreatic β-islet cells; in the liver, glucokinase is induced by insulin. Table 9.1 identifies the differences between these enzymes. These coincide with the differences between the glucose transporters in these tissues.
Table 9.1. Comparison of Hexokinase and Glucokinase
HEXOKINASE GLUCOKINASE
Present in most tissues Present in hepatocytes and pancreatic β-islet cells (along with GLUT 2, acts as the glucose sensor)
Low Km (reaches maximum velocity at low [glucose]) High Km (acts on glucose proportionally to its concentration)
Inhibited by glucose 6-phosphate Induced by insulin in hepatocytes
KEY CONCEPT
Of all the enzymes the MCAT is most likely to test you on, the rate-limiting enzymes for each process are at the top of the list:
- Glycolysis: phosphofructokinase-1
- Fermentation: lactate dehydrogenase
- Glycogenesis: glycogen synthase
- Glycogenolysis: glycogen phosphorylase
- Gluconeogenesis: fructose-1,6-bisphosphatase
- Pentose Phosphate Pathway: *glucose**-6-phosphate dehydrogenase*
Phosphofructokinases (PFK-1 and PFK-2)
Phosphofructokinase-1 (PFK-1) is the rate-limiting enzyme and main control point in glycolysis. In this reaction, fructose 6-phosphate is phosphorylated to fructose 1,6-bisphosphate using ATP. PFK-1 is inhibited by ATP and citrate, and activated by AMP. This makes sense because the cell should turn off glycolysis when it has sufficient energy (high ATP) and turn on glycolysis when it needs energy (high AMP). Citrate is an intermediate of the citric acid cycle, so high levels of citrate also imply that the cell is producing sufficient energy.
Insulin stimulates and glucagon inhibits PFK-1 in hepatocytes by an indirect mechanism involving PFK-2 and fructose 2,6-bisphosphate, as shown in Figure 9.2. Insulin activates phosphofructokinase-2 (PFK-2), which converts a tiny amount of fructose 6-phosphate to fructose 2,6-bisphosphate (F2,6-BP). F2,6-BP activates PFK-1. On the other hand, glucagon inhibits PFK-2, lowering F2,6-BP and thereby inhibiting PFK-1. PFK-2 is found mostly in the liver. By activating PFK-1, it allows these cells to override the inhibition caused by ATP so that glycolysis can continue, even when the cell is energetically satisfied. The metabolites of glycolysis can thus be fed into the production of glycogen, fatty acids, and other storage molecules rather than just being burned to produce ATP.
Glyceraldehyde-3-Phosphate Dehydrogenase
Glyceraldehyde-3-phosphate dehydrogenase catalyzes an oxidation and addition of inorganic phosphate (Pi) to its substrate, glyceraldehyde 3-phosphate. This results in the production of a high-energy intermediate 1,3-bisphosphoglycerate and the reduction of NAD+ to NADH. If glycolysis is aerobic, the NADH can be oxidized by the mitochondrial electron transport chain, providing energy for ATP synthesis by oxidative phosphorylation.
BRIDGE
In Chapter 11 of MCAT General Chemistry Review, we learn that oxidation is loss of electrons and reduction is gain of electrons. While this is true with biomolecules, it may be easier to think of oxidation as increasing bonds to oxygen or other heteroatoms (atoms besides C and H) and reduction as increasing bonds to hydrogen, as discussed in Chapter 4 of MCAT Organic Chemistry Review. Thus, the conversion of NAD+ to NADH is a reduction reaction.
3-Phosphoglycerate Kinase
3-Phosphoglycerate kinase transfers the high-energy phosphate from 1,3-bisphosphoglycerate to ADP, forming ATP and 3-phosphoglycerate. This type of reaction, in which ADP is directly phosphorylated to ATP using a high-energy intermediate, is referred to as substrate-level phosphorylation. In contrast to oxidative phosphorylation in mitochondria, substrate-level phosphorylations are not dependent on oxygen, and are the only means of ATP generation in an anaerobic tissue.
Pyruvate Kinase
The last enzyme in aerobic glycolysis, pyruvate kinase catalyzes a substrate-level phosphorylation of ADP using the high-energy substrate phosphoenolpyruvate (PEP). Pyruvate kinase is activated by fructose 1,6-bisphosphate from the PFK-1 reaction. This is referred to as feed-forward activation, meaning that the product of an earlier reaction of glycolysis (fructose 1,6-bisphosphate) stimulates, or prepares, a later reaction in glycolysis (by activating pyruvate kinase).
Fermentation
In the absence of oxygen, fermentation will occur. The key fermentation enzyme in mammalian cells is lactate dehydrogenase, which converts pyruvate into lactate and concurrently oxidizes NADH to NAD+. Without oxygen, glycolysis would eventually come to a halt and stop any production of ATP when all the available NAD+ had been reduced to NADH. Under aerobic conditions, NADH produced by glycolysis donates its electrons to the ETC and is reconverted to NAD+, replenishing the pool of NAD+ needed by glyceraldehyde-3-phosphate dehydrogenase (GAPDH) within the glycolysis pathway. However, under anaerobic conditions, the ETC ceases to function and NADH is unable to donate its electrons to regenerate NAD+. By reducing pyruvate to lactate and oxidizing NADH to NAD+, lactate dehydrogenase prevents this potential problem from developing and allows glycolysis to continue. There is no net loss of carbon in this process: pyruvate and lactate are both three-carbon molecules. In yeast cells, fermentation is the conversion of pyruvate (three carbons) to ethanol (two carbons) and carbon dioxide (one carbon). While the end products are different, the result of both mammalian and yeast fermentation is the same: replenishing NAD+.
In aerobic tissues, lactate does not normally form in significant amounts. However, when oxygenation is poor, such as during strenuous exercise, most cellular ATP is generated by anaerobic glycolysis, and lactate production increases. When oxygen once again becomes available, some of this lactate can be converted back into pyruvate. However, most of the lactate will enter into the Cori cycle, also called the lactic acid cycle, a metabolic pathway shown in Figure 9.3. In this pathway, lactate produced by muscles during anaerobic respiration is transported to the liver and used in gluconeogenesis to create glucose, which is then returned to the muscles. The muscle cells then break down the glucose, recreating lactate, which can then undergo the same cycle again.
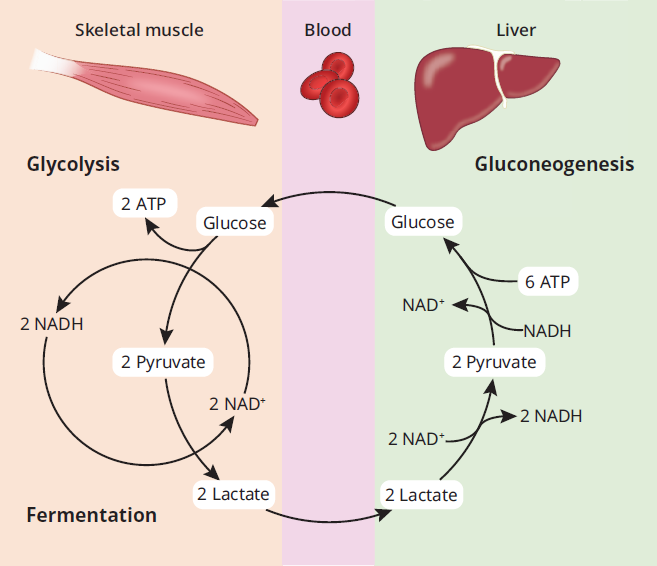
Figure 9.3 The Cori Cycle Lactate from muscles is used by the liver to create glucose, which is then used and metabolized back into lactate in the muscles.
Important Intermediates of Glycolysis
Glycolysis serves as a crossroads for a number of metabolic processes; the intermediates of glycolysis are often used to link different pathways during both catabolism and anabolism. Three of these intermediates are worth highlighting:
- Dihydroxyacetone phosphate (DHAP) is used in hepatic and adipose tissue for triacylglycerol synthesis. DHAP is formed from fructose 1,6-bisphosphate. It can be isomerized to glycerol 3-phosphate, which can then be converted to glycerol, the backbone of triacylglycerols.
- 1,3-Bisphosphoglycerate (1,3-BPG) and phosphoenolpyruvate (PEP) are high-energy intermediates used to generate ATP by substrate-level phosphorylation. This is the only ATP gained in anaerobic respiration.
Irreversible Enzymes
Three enzymes in the pathway catalyze reactions that are irreversible. This keeps the pathway moving in only one direction. However, the liver must be able to generate new glucose from other biomolecules through gluconeogenesis, which is essentially the reverse of glycolysis. Because of the irreversible enzymes of glycolysis, different reactions, and therefore different enzymes, must be used at these three points:
- Glucokinase or hexokinase
- PFK-1
- Pyruvate kinase
MNEMONIC
Irreversible steps of glycolysis:
How Glycolysis Pushes Forward the Process: Kinases.
- Hexokinase
- Glucokinase
- PFK-1
- Pyruvate Kinase
Glycolysis in Erythrocytes
In erythrocytes (red blood cells), anaerobic glycolysis represents the only pathway for ATP production, yielding a net 2 ATP per glucose.
BRIDGE
Adaptation to high altitudes (low pO2) involves:
- Increased respiration
- Increased oxygen affinity for hemoglobin (initial)
- Increased rate of glycolysis
- Increased [2,3-BPG] in RBC (over a 12–24 hour period)
- Normalized oxygen affinity for hemoglobin restored by the increased level of 2,3-BPG
- Increased hemoglobin (over days to weeks)
Gas exchange is discussed in Chapter 6 of MCAT Biology Review, and effects on hemoglobin are discussed in Chapter 7 of MCAT Biology Review.
Red blood cells have bisphosphoglycerate mutase, which produces 2,3-bisphosphoglycerate (2,3-BPG) from 1,3-BPG in glycolysis. Remember that mutases are enzymes that move a functional group from one place in a molecule to another; in this case, the phosphate is moved from the 1-position to the 2-position. 2,3-BPG binds allosterically to the β-chains of hemoglobin A (HbA) and decreases its affinity for oxygen. This effect of 2,3-BPG is seen in the oxygen dissociation curve for HbA, shown in Figure 9.4. The rightward shift in the curve is sufficient to allow unloading of oxygen in tissues, but still allows 100 percent saturation in the lungs. An abnormal increase in erythrocyte 2,3-BPG might shift the curve far enough so that HbA is not fully saturated in the lungs.
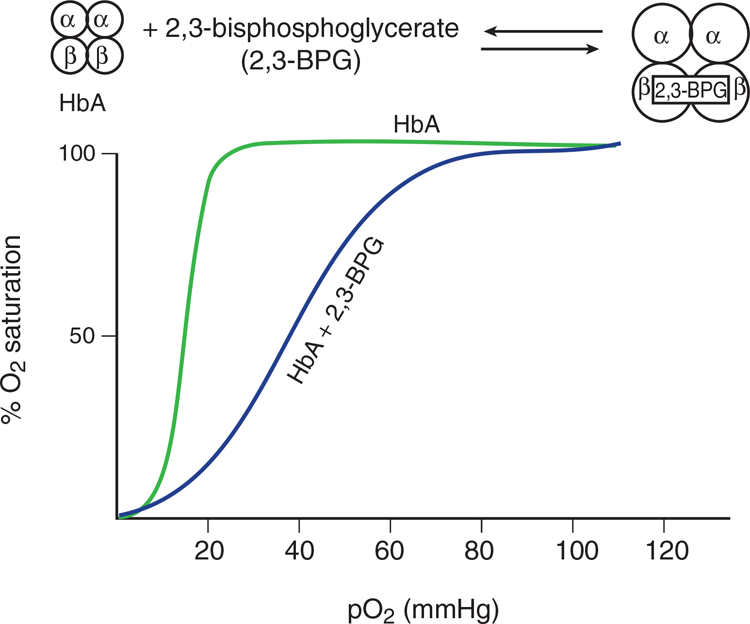
Figure 9.4 Effect of 2,3-Bisphosphoglycerate on Hemoglobin A
BRIDGE
Remember the other physiological changes that promote a right shift of the oxygen dissociation curve (the Bohr effect), discussed in Chapter 7 of MCAT Biology Review:
- High 2,3-BPG
- Low pH
- High [H+]
- High pCO2
These all occur during exercise, giving the mnemonic: “Exercise is the right thing to do.”
Although 2,3-BPG binds to HbA, it does not bind well to fetal hemoglobin (HbF), with the result that HbF has a higher affinity for oxygen than maternal HbA. This allows transplacental passage of oxygen from mother to fetus.
MCAT CONCEPT CHECK 9.2:
Before you move on, assess your understanding of the material with these questions.
- What are the function and key regulators of the following enzymes? Which ones are reversible?
Hexokinase
__________________________________________________
- Function:
__________________________________________________
- Regulation:
__________________________________________________
- Reversible?
Glucokinase
__________________________________________________
- Function:
__________________________________________________
- Regulation:
__________________________________________________
- Reversible?
Phosphofructokinase-1 (PFK-1)
__________________________________________________
- Function:
__________________________________________________
- Regulation:
__________________________________________________
- Reversible?
Glyceraldehyde-3-phosphate dehydrogenase
__________________________________________________
- Function:
__________________________________________________
- Reversible?
3-phosphoglycerate kinase
__________________________________________________
- Function:
__________________________________________________
- Reversible?
Pyruvate kinase
__________________________________________________
- Function:
__________________________________________________
- Regulation:
__________________________________________________
- Reversible?
__________________________________________________
- Why must pyruvate undergo fermentation for glycolysis to continue?
__________________________________________________
- Why is it necessary that fetal hemoglobin does not bind 2,3-BPG?
9.3 Other Monosaccharides
LEARNING OBJECTIVES
After Chapter 9.3, you will be able to:
- Explain the importance of trapping a sugar in the cell and linking its metabolism with the glycolysis pathway
- Recognize the key enzymes, reactants, and products of galactose and fructose metabolism
While glucose represents the primary monosaccharide used by cells, other monosaccharides such as galactose and fructose can also contribute to ATP production by feeding into glycolysis or other metabolic processes. These monosaccharides are tested far less frequently than glucose on the MCAT, but are included here to compare and contrast their metabolism with glycolysis. In particular, notice the similarities between Figure 9.2 (glycolysis) and Figures 9.5 (galactose metabolism) and 9.6 (fructose metabolism).
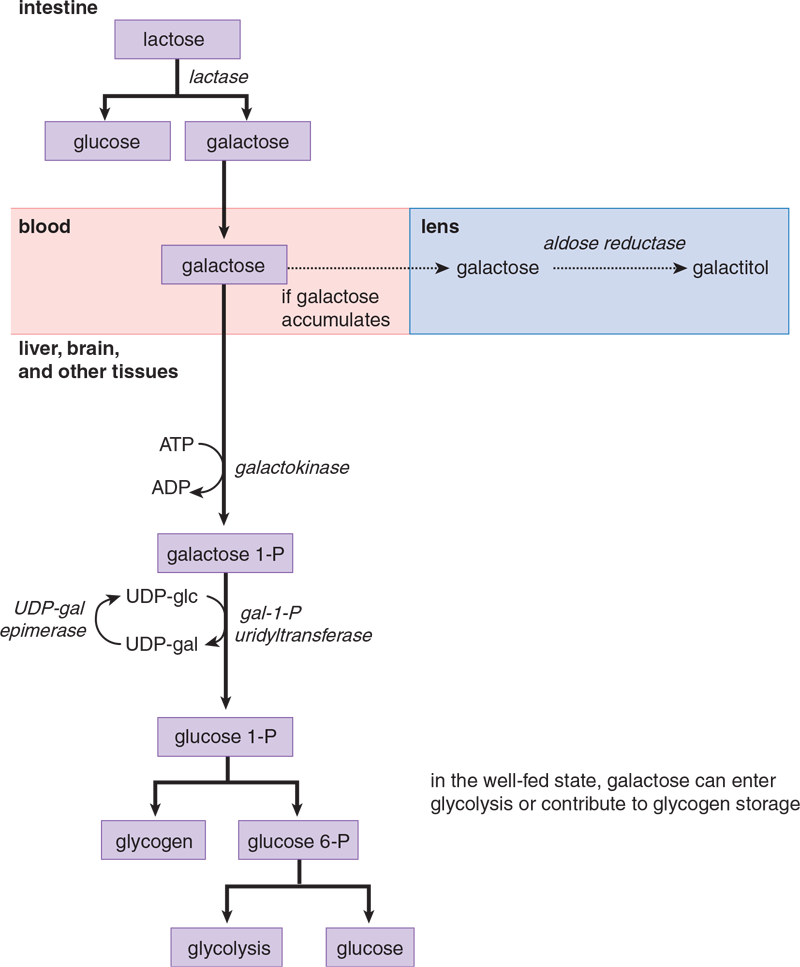
Figure 9.5 Galactose Metabolism
REAL WORLD
Primary lactose intolerance is caused by a hereditary deficiency of lactase. Secondary lactose intolerance can be precipitated at any age by gastrointestinal disturbances that cause damage to the intestinal lining, where lactase is found.
Common symptoms of lactose intolerance include vomiting, bloating, explosive and watery diarrhea, cramps, and dehydration. The symptoms can be attributed to bacterial fermentation of lactose, which produces a mixture of CH4, H2, and small organic acids. The acids are osmotically active and result in the movement of water into the intestinal lumen.
Galactose Metabolism
An important source of galactose in the diet is the disaccharide lactose present in milk. Lactose is hydrolyzed to galactose and glucose by lactase, which is a brush-border enzyme of the duodenum. Along with other monosaccharides, galactose reaches the liver through the hepatic portal vein. Once transported into tissues, galactose is phosphorylated by galactokinase, trapping it in the cell. The resulting galactose 1-phosphate is converted to glucose 1-phosphate by galactose-1-phosphate uridyltransferase and an epimerase. Epimerases are enzymes that catalyze the conversion of one sugar epimer to another; remember from Chapter 4 of MCAT Biochemistry Review that epimers are diastereomers that differ at exactly one chiral carbon. The pathway is shown in Figure 9.5; important enzymes to remember are:
- Galactokinase
- Galactose-1-phosphate uridyltransferase
REAL WORLD
Genetic deficiencies of galactokinase or galactose-1-phosphate uridyltransferase lead to galactosemia. Cataracts are a characteristic finding, which result from the conversion of excess galactose in the blood to galactitol in the lens of the eye by aldose reductase. Galactitol is a polyol (a carbon chain with many alcohol groups) and, as such, is hydrophilic. Accumulation of galactitol in the lens causes osmotic damage and cataracts.
Deficiency of galactose-1-phosphate uridyltransferase is more severe because, in addition to causing galactosemia, it leads to galactose 1-phosphate getting stuck intracellularly in the liver, brain, and other tissues and not diffusing out.
Fructose Metabolism
Fructose is found in honey and fruit and as part of the disaccharide sucrose (common table sugar). Sucrose is hydrolyzed by the duodenal brush-border enzyme sucrase, and the resulting monosaccharides, glucose and fructose, are absorbed into the hepatic portal vein. The liver phosphorylates fructose using fructokinase to trap it in the cell. The resulting fructose 1-phosphate is then cleaved into glyceraldehyde and DHAP by aldolase B. Smaller amounts are metabolized in renal proximal tubules. The pathway is shown in Figure 9.6.
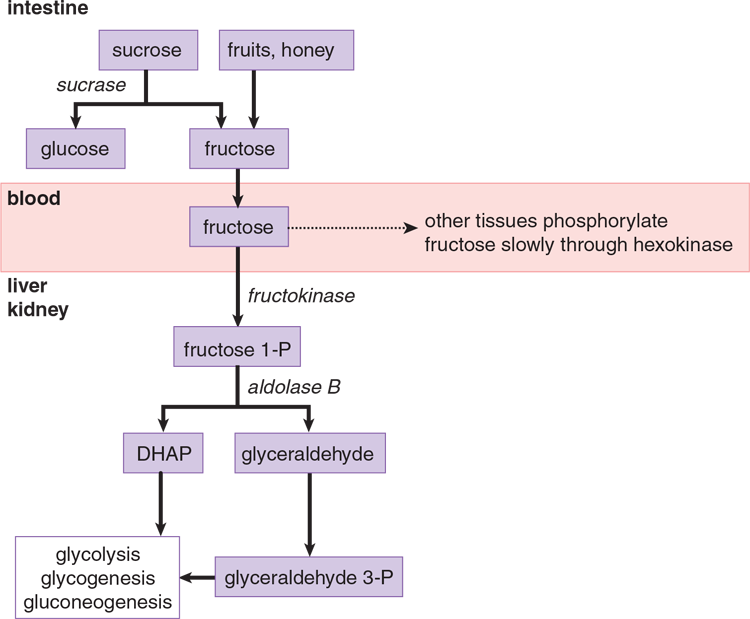
Figure 9.6 Fructose Metabolism
KEY CONCEPT
Because dihydroxyacetone phosphate (DHAP) and glyceraldehyde, the products of fructose metabolism, are downstream from the key regulatory and rate-limiting enzyme of glycolysis (PFK-1), a high-fructose drink supplies a quick source of energy in both aerobic and anaerobic cells.
MCAT CONCEPT CHECK 9.3:
Before you move on, assess your understanding of the material with these questions.
- Which enzyme is responsible for trapping galactose in the cell? What enzyme in galactose metabolism results in a product that can feed directly into glycolysis, linking the two pathways?
__________________________________________________
- “Trapping” enzyme:
__________________________________________________
- “Linking” enzyme:
- Which enzyme is responsible for trapping fructose in the cell? What enzyme in fructose metabolism results in a product that can feed directly into glycolysis, linking the two pathways?
__________________________________________________
- “Trapping” enzyme:
__________________________________________________
- “Linking” enzyme:
9.4 Pyruvate Dehydrogenase
LEARNING OBJECTIVES
After Chapter 9.4, you will be able to:
- Recall the reactants and products of the pyruvate dehydrogenase complex
- Describe the relationship between acetyl-CoA levels and PDH activity
Pyruvate from aerobic glycolysis enters mitochondria, where it may be converted to acetyl-CoA for entry into the citric acid cycle if ATP is needed, or for fatty acid synthesis if sufficient ATP is present. The pyruvate dehydrogenase complex (PDH) reaction, shown in Figure 9.7, is irreversible and cannot be used to convert acetyl-CoA to pyruvate or to glucose. Pyruvate dehydrogenase in the liver is activated by insulin, whereas in the nervous system, the enzyme is not responsive to hormones. This makes sense because high insulin levels signal to the liver that the individual is in a well-fed state; thus, the liver should not only burn glucose for energy, but shift the fatty acid equilibrium toward production and storage, rather than oxidation (fatty acid synthesis, discussed in Chapter 11 of MCAT Biochemistry Review, starts from citrate produced in the citric acid cycle).
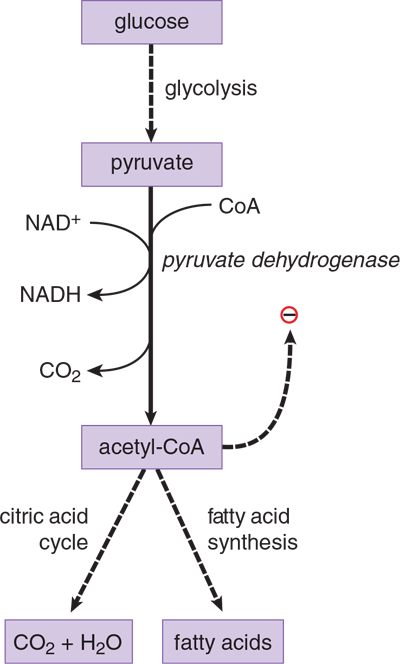
Figure 9.7 Pyruvate Dehydrogenase
Pyruvate dehydrogenase is actually a complex of enzymes carrying out multiple reactions in succession. The details of each of these reactions are covered in Chapter 10 of MCAT Biochemistry Review, but an overview of the enzyme is provided here because it represents one of three possible fates of pyruvate: conversion to acetyl-CoA by PDH, conversion to lactate by lactate dehydrogenase, or conversion to oxaloacetate by pyruvate carboxylase.
REAL WORLD
A deficiency in thiamine (vitamin B1) can result in:
- Beriberi, which is characterized by congestive heart failure or nerve damage.
- Wernicke–Korsakoff syndrome, which is characterized by difficulty walking, uncoordinated eye movements, confusion, and memory disturbances.
Giving glucose to an individual with thiamine deficiency can lead to severe lactic acidosis and other metabolic derangements because pyruvate cannot be converted into acetyl-CoA without the vitamin. This is why thiamine must be given before an infusion of glucose in individuals suspected to have thiamine deficiency (such as alcoholics).
This large complex requires multiple cofactors and coenzymes, including thiamine pyrophosphate, lipoic acid, CoA, FAD, and NAD+. Insufficient amounts of any of these cofactors or coenzymes can result in metabolic derangements.
Pyruvate dehydrogenase is inhibited by its product acetyl-CoA. This control is important in several contexts and should be considered along with pyruvate carboxylase, the other mitochondrial enzyme that uses pyruvate (introduced in gluconeogenesis, later in this chapter). Essentially, the buildup of acetyl-CoA (which happens during β-oxidation) causes a shift in metabolism: pyruvate is no longer converted into acetyl-CoA (to enter the citric acid cycle), but rather into oxaloacetate (to enter gluconeogenesis).
MCAT CONCEPT CHECK 9.4:
Before you move on, assess your understanding of the material with these questions.
- What are the reactants of the pyruvate dehydrogenase complex? What are the products?
__________________________________________________
- Reactants:
__________________________________________________
- Products:
__________________________________________________
- How does acetyl-CoA affect PDH complex activity? Why?
9.5 Glycogenesis and Glycogenolysis
LEARNING OBJECTIVES
After Chapter 9.5, you will be able to:
- Recall the key enzymes, reactants, and products in glycogenesis and glycogenolysis
- Describe the features of glycogen storage diseases
- Recognize the structural features of glycogen and the major glycosidic links within a glycogen granule:

Glycogen, a branched polymer of glucose, represents a storage form of glucose. Glycogen synthesis and degradation occur primarily in liver and skeletal muscle, although other tissues store smaller quantities. Glycogen is stored in the cytoplasm as granules. Each granule has a central protein core with polyglucose chains radiating outward to form a sphere, as shown in Figure 9.8. Glycogen granules composed entirely of linear chains have the highest density of glucose near the core. If the chains are branched, the glucose density is highest at the periphery of the granule, allowing more rapid release of glucose on demand.

Figure 9.8 A Glycogen Granule
KEY CONCEPT
The glycogen in the liver and in skeletal muscle serve two quite different roles. Liver glycogen is broken down to maintain a constant level of glucose in the blood; muscle glycogen is broken down to provide glucose to the muscle during vigorous exercise.
Glycogen stored in the liver is a source of glucose that is mobilized between meals to prevent low blood sugar, whereas muscle glycogen is stored as an energy reserve for muscle contraction.
While our focus is on human metabolism, it is worth mentioning that plants also store excess glucose in long α-linked chains of glucose called starch, as seen in Figure 9.9.

Figure 9.9 Potatoes and Potato Starch
Glycogenesis
Glycogenesis is the synthesis of glycogen granules. It begins with a core protein called glycogenin. As shown in Figure 9.10, glucose addition to a granule begins with glucose 6-phosphate, which is converted to glucose 1-phosphate. This glucose 1-phosphate is then activated by coupling to a molecule of uridine diphosphate (UDP), which permits its integration into the glycogen chain by glycogen synthase. This activation occurs when glucose 1-phosphate interacts with uridine triphosphate (UTP), forming UDP-glucose and a pyrophosphate (PPi).
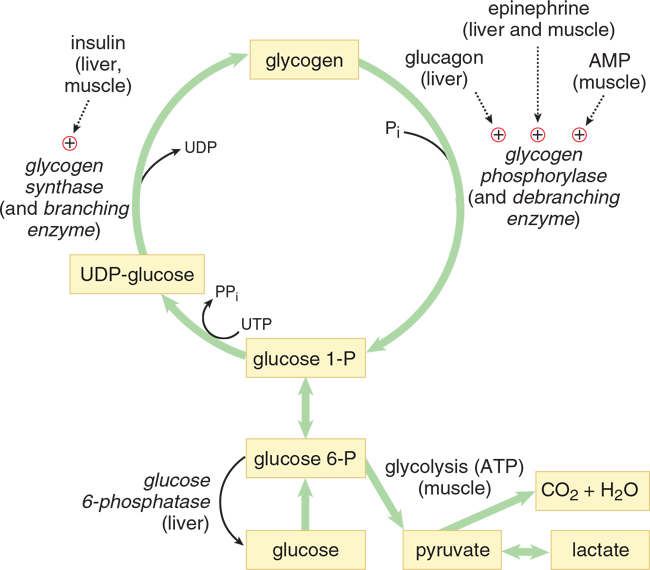
Figure 9.10 Glycogen Metabolism
Glycogen Synthase
Glycogen synthase is the rate-limiting enzyme of glycogen synthesis and forms the α-1,4 glycosidic bond found in the linear glucose chains of the granule. It is stimulated by glucose 6-phosphate and insulin. It is inhibited by epinephrine and glucagon through a protein kinase cascade that phosphorylates and inactivates the enzyme.
Branching Enzyme (Glycosyl *α*-1,4:*α*-1,6 Transferase)
Branching enzyme is responsible for introducing α-1,6-linked branches into the granule as it grows. The steps of glycogen synthesis and the process by which branches are introduced are shown schematically in Figure 9.11. Branching enzyme:
- Hydrolyzes one of the α-1,4 bonds to release a block of oligoglucose (a few glucose molecules bonded together in a chain), which is then moved and added in a slightly different location.
- Forms an α-1,6 bond to create a branch.
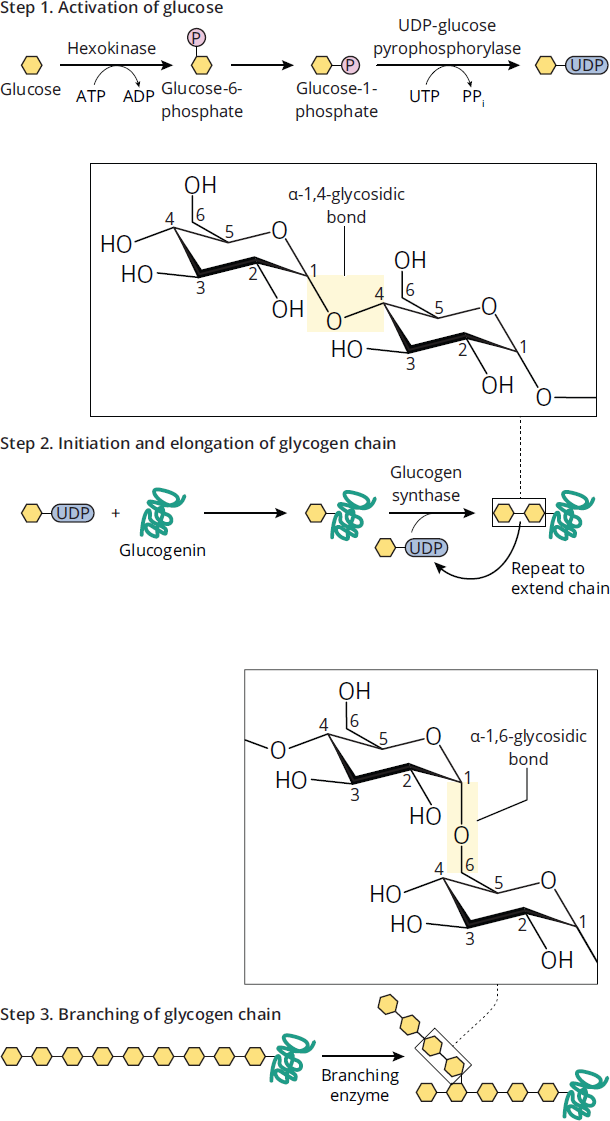
Figure 9.11 Glycogenesis
MNEMONIC
α-1,4 keeps the same branch moving “4 ward”; α-1,6 (one-six) “puts a branch in the mix.”
Glycogenolysis
The rate-limiting enzyme of glycogenolysis, the process of breaking down glycogen, is glycogen phosphorylase. In contrast to a hydrolase, a phosphorylase breaks bonds using an inorganic phosphate instead of water. The glucose 1-phosphate formed by glycogen phosphorylase is converted to glucose 6-phosphate by the same mutase used in glycogen synthesis, as shown in Figure 9.10.
MCAT EXPERTISE
Under the pressure of Test Day, it can be easy to misread words. When given a passage or question about carbohydrate metabolism, be sure you focus so you can distinguish between glycolysis, glycogenesis, glycogenolysis, and gluconeogenesis.
Glycogen Phosphorylase
Glycogen phosphorylase breaks α-1,4 glycosidic bonds, releasing glucose 1-phosphate from the periphery of the granule. It cannot break α-1,6 bonds and therefore stops when it nears the outermost branch points. Glycogen phosphorylase is activated by glucagon in the liver, so that glucose can be provided for the rest of the body. In skeletal muscle, it is activated by AMP and epinephrine, which signal that the muscle is active and requires more glucose. It is inhibited by ATP.
Debranching Enzyme (Glucosyl *α*-1,4:*α*-1,4 Transferase and *α*-1,6 Glucosidase)
Debranching enzyme is a two-enzyme complex that deconstructs the branches in glycogen that have been exposed by glycogen phosphorylase. The two-step process by which this occurs is diagrammed in Figure 9.12. Debranching enzyme:
- Breaks an α-1,4 bond adjacent to the branch point and moves the small oligoglucose chain that is released to the exposed end of the other chain.
- Forms a new α-1,4 bond.
- Hydrolyzes the α-1,6 bond, releasing the single residue at the branch point as free glucose. This represents the only free glucose produced directly in glycogenolysis (as opposed to the glucose produced from glucose 1-phosphate, which must be converted by a mutase to glucose 6-phosphate before it can be converted to glucose via the enzyme glucose-6-phosphatase).
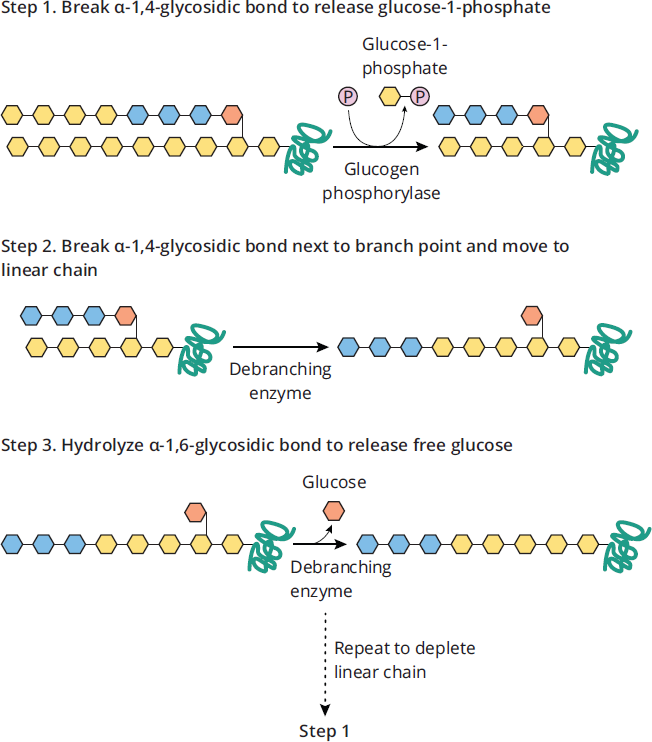
Figure 9.12 Debranching Enzyme
KEY CONCEPT
Debranching enzyme is actually made up of two enzymes with different functions: one moves the terminal end of a glycogen chain to the branch point (α-1,4:α-1,4 transferase), and one removes the glucose monomer actually present at the branch point (α-1,6 glucosidase).
Glycogen Storage Diseases
There are a number of genetic deficiencies that can impact the metabolism of glycogen. The clinical features of a metabolic glycogen defect depend on a few important factors: which enzyme is affected, the degree to which that enzyme’s activity is decreased, and which isoform of the enzyme is affected. Isoforms are slightly different versions of the same protein; in the case of glycogen enzymes, there are often different isoforms of the enzymes in the liver and muscle. These deficiencies are termed glycogen storage diseases because all are characterized by accumulation or lack of glycogen in one or more tissues.
REAL WORLD
The most common glycogen storage disease is von Gierke’s disease, a defect in glucose-6-phosphatase. Because this enzyme is also the last step of gluconeogenesis, this process is also affected, leading to periods of extremely low blood sugar between meals. These patients therefore need continuous feeding with carbohydrates to maintain blood sugar. With the buildup of glucose 6-phosphate in liver cells, the liver enlarges and is damaged over time.
MCAT CONCEPT CHECK 9.5:
Before you move on, assess your understanding of the material with these questions.
- What is the structure of glycogen? What types of glycosidic links exist in a glycogen granule?
__________________________________________________
- What are the two main enzymes of glycogenesis, and what does each accomplish?
- __________________________________________________
- __________________________________________________
- What are the two main enzymes of glycogenolysis, and what does each accomplish?
- __________________________________________________
- __________________________________________________
9.6 Gluconeogenesis
LEARNING OBJECTIVES
After Chapter 9.6, you will be able to:
- Recognize the conditions that favor gluconeogenesis
- Recall the four enzymes unique to gluconeogenesis, and relate them to the enzymes that catalyze the opposite process in glycolysis
- Detail the regulatory role of acetyl-CoA in the metabolism of pyruvate
The liver maintains glucose levels in blood during fasting through either glycogenolysis or gluconeogenesis. The kidney can also carry out gluconeogenesis, although its contribution is much smaller. These pathways are promoted by glucagon and epinephrine, which act to raise blood sugar levels, and are inhibited by insulin, which acts to lower blood sugar levels. During fasting, glycogen reserves drop dramatically in the first 12 hours, during which time gluconeogenesis increases. After 24 hours, it represents the sole source of glucose. Important substrates for gluconeogenesis are:
- Glycerol 3-phosphate (from stored fats, or triacylglycerols, in adipose tissue)
- Lactate (from anaerobic glycolysis)
- Glucogenic amino acids (from muscle proteins)
KEY CONCEPT
Insulin acts to lower blood sugar levels; the counterregulatory hormones, which include glucagon, epinephrine, cortisol, and growth hormone, act to raise blood sugar levels by stimulating glycogenolysis and gluconeogenesis. The regulation of metabolism is discussed in Chapter 12 of MCAT Biochemistry Review.
The last item of this list merits some explaining. Amino acids can be subclassified as glucogenic, ketogenic, or both. Glucogenic amino acids (all except leucine and lysine) can be converted into intermediates that feed into gluconeogenesis, while ketogenic amino acids can be converted into ketone bodies, which can be used as an alternative fuel, particularly during periods of prolonged starvation. See Chapter 11 of MCAT Biochemistry Review for more information on amino acid and protein metabolism.
BRIDGE
Amino acids and proteins are extremely important topics for the MCAT. Check out Chapter 1 of MCAT Biochemistry Review for more information on amino acids, peptides, and proteins.
Dietary fructose and galactose can also be converted to glucose in the liver, as described earlier in this chapter.
In humans, while glucose is converted into acetyl-CoA through glycolysis and pyruvate dehydrogenase, it is not possible to convert acetyl-CoA back to glucose. Because most fatty acids are metabolized solely to acetyl-CoA, they are not a major source of glucose either. One minor exception is fatty acids with an odd number of carbon atoms (for example, fatty acid tails containing 17 carbons), which yield a small amount of propionyl-CoA, which is glucogenic.
The pathway of gluconeogenesis is diagrammed in Figure 9.13. Each of the important gluconeogenic intermediates—lactate, alanine, and glycerol 3-phosphate—have enzymes that convert them into glycolytic intermediates.
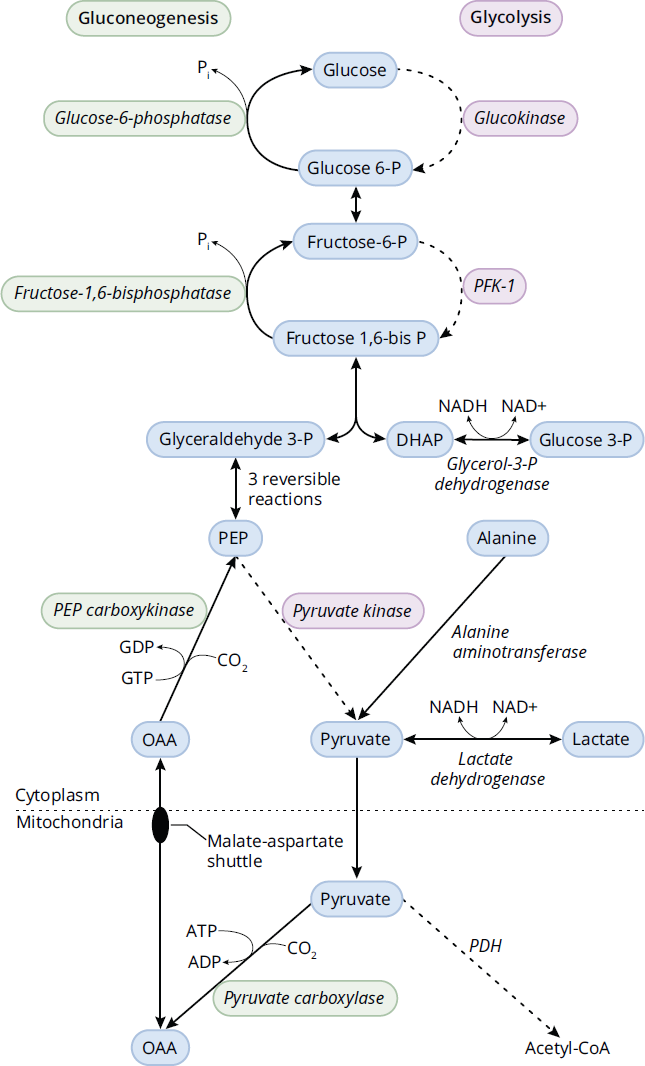
Figure 9.13 Gluconeogenesis
Lactate is converted to pyruvate by lactate dehydrogenase. Alanine is converted to pyruvate by alanine aminotransferase. Glycerol 3-phosphate is converted to dihydroxyacetone phosphate (DHAP) by glycerol-3-phosphate dehydrogenase.
Important Enzymes of Gluconeogenesis
Most steps in gluconeogenesis represent a reversal of glycolysis and have thus been omitted from the diagram. However, the four important enzymes to know are those required to catalyze reactions that circumvent the irreversible steps of glycolysis in the liver (those catalyzed by glucokinase, phosphofructokinase-1, and pyruvate kinase).
Pyruvate Carboxylase
Pyruvate carboxylase is a mitochondrial enzyme that is activated by acetyl-CoA (from β-oxidation). The product, oxaloacetate (OAA), is a citric acid cycle intermediate and cannot leave the mitochondrion. Rather, it is reduced to malate, which can leave the mitochondrion via the malate–aspartate shuttle, which is described in Chapter 10 of MCAT Biochemistry Review. Once in the cytoplasm, malate is oxidized to OAA. The fact that acetyl-CoA activates pyruvate carboxylase is an important point. Acetyl-CoA inhibits pyruvate dehydrogenase because a high level of acetyl-CoA implies that the cell is energetically satisfied and need not run the citric acid cycle in the forward direction; in other words, the cell should stop burning glucose. Rather, pyruvate will be shunted through pyruvate carboxylase to help generate additional glucose through gluconeogenesis. Note that the source of acetyl-CoA is not from glycolysis and pyruvate dehydrogenase in this case, but from fatty acids. Thus, to produce glucose in the liver during gluconeogenesis, fatty acids must be burned to provide this energy, stop the forward flow of the citric acid cycle, and produce massive amounts of OAA that can eventually lead to glucose production for the rest of the body.
KEY CONCEPT
Because glycolysis contains three irreversible steps (those catalyzed by hexokinase, phosphofructokinase-1, and pyruvate kinase), different enzymes must exist in gluconeogenesis to allow the body to revert pyruvate to glucose.
Phosphoenolpyruvate Carboxykinase (PEPCK)
Phosphoenolpyruvate carboxykinase (PEPCK) in the cytoplasm is induced by glucagon and cortisol, which generally act to raise blood sugar levels. It converts OAA to phosphoenolpyruvate (PEP) in a reaction that requires GTP. PEP continues in the pathway to fructose 1,6-bisphosphate. Thus, the combination of pyruvate carboxylase and PEPCK are used to circumvent the action of pyruvate kinase by converting pyruvate back into PEP.
Fructose-1,6-Bisphosphatase
Fructose-1,6-bisphosphatase in the cytoplasm is a key control point of gluconeogenesis and represents the rate-limiting step of the process. It reverses the action of phosphofructokinase-1, the rate-limiting step of glycolysis, by removing phosphate from fructose 1,6-bisphosphate to produce fructose 6-phosphate. A common pattern to note is that phosphatases oppose kinases. Fructose-1,6-bisphosphatase is activated by ATP and inhibited by AMP and fructose 2,6-bisphosphate. This should make sense: high levels of ATP imply that a cell is energetically satisfied enough to produce glucose for the rest of the body, whereas high levels of AMP imply that a cell needs energy and cannot afford to produce energy for the rest of the body before satisfying its own requirements. Fructose 2,6-bisphosphate (F2,6-BP) is sometimes thought of as a marker for satisfactory energy levels in liver cells. It helps these cells override the inhibition of phosphofructokinase-1 that occurs when high levels of acetyl-CoA are formed, signaling to the liver cell that it should shift its function from burning to storing fuel. F2,6-BP, produced by PFK-2, controls both gluconeogenesis and glycolysis (in the liver). Recall from the earlier discussion of this enzyme and Figure 9.2 that PFK-2 is activated by insulin and inhibited by glucagon. Thus, glucagon will lower F2,6-BP and stimulate gluconeogenesis, whereas insulin will increase F2,6-BP and inhibit gluconeogenesis.
Glucose-6-Phosphatase
Glucose-6-phosphatase is found only in the lumen of the endoplasmic reticulum in liver cells. Glucose 6-phosphate is transported into the ER, and free glucose is transported back into the cytoplasm, from where it can diffuse out of the cell using GLUT transporters. The absence of glucose-6-phosphatase in skeletal muscle means that muscle glycogen cannot serve as a source of blood glucose and rather is for use only within the muscle. Glucose-6-phosphatase is used to circumvent glucokinase and hexokinase, which convert glucose to glucose 6-phosphate.
KEY CONCEPT
Because gluconeogenesis requires acetyl-CoA to occur (to inhibit pyruvate dehydrogenase and stimulate pyruvate carboxylase), gluconeogenesis is inextricably linked to fatty acid oxidation. The source of acetyl-CoA cannot be glycolysis because this would just burn the glucose that is being generated in gluconeogenesis.
Although alanine is the major glucogenic amino acid, almost all amino acids are also glucogenic. Most of these are converted by individual pathways to citric acid cycle intermediates, then to malate, following the same path from there to glucose.
It is important to note that glucose produced by hepatic (liver-based) gluconeogenesis does not represent an energy source for the liver. Gluconeogenesis requires expenditure of ATP that is provided by β-oxidation of fatty acids. Therefore, as mentioned above, hepatic gluconeogenesis is always dependent on β-oxidation of fatty acids in the liver. During periods of low blood sugar, adipose tissue releases these fatty acids by breaking down triacylglycerols to glycerol (which can also be converted to the gluconeogenic intermediate DHAP) and free fatty acids.
REAL WORLD
Because red blood cells lack mitochondria, they cannot carry out aerobic metabolism. Rather, pyruvate is converted to lactic acid to regenerate NAD+. However, lactate is acidic; it must be removed from the bloodstream to avoid acidifying the blood. Red blood cells deliver this lactate to the liver, where it can be converted back into pyruvate and, through gluconeogenesis, become glucose for the red blood cells to use. This is known as the Cori cycle: glucose is converted to lactate in red blood cells, and lactate is converted to glucose in liver cells.
Although the acetyl-CoA from fatty acids cannot be converted into glucose, it can be converted into ketone bodies as an alternative fuel for cells, including the brain. Extended periods of low blood sugar are thus usually accompanied by high levels of ketones in the blood. Ketone bodies can be thought of as a transportable form of acetyl-CoA that is primarily utilized in periods of extended starvation.
MCAT CONCEPT CHECK 9.6:
Before you move on, assess your understanding of the material with these questions.
- Under what physiological conditions should the body carry out gluconeogenesis?
__________________________________________________
- What are the four enzymes unique to gluconeogenesis? Which irreversible glycolytic enzymes do they replace?
Gluconeogenic Enzyme Replaces
- How does acetyl-CoA shift the metabolism of pyruvate?
__________________________________________________
- Given that the glycogen storage disorder von Gierke's disease affects the last enzyme of gluconeogenesis, predict the associated metabolic derangement that occurs.
__________________________________________________
9.7 The Pentose Phosphate Pathway
LEARNING OBJECTIVES
After Chapter 9.7, you will be able to:
- Identify the two major products of the pentose phosphate pathway
- Explain the three primary functions of NADPH in cellular respiration
The pentose phosphate pathway (PPP), also known as the hexose monophosphate (HMP) shunt, occurs in the cytoplasm of all cells, where it serves two major functions: production of NADPH and serving as a source of ribose 5-phosphate for nucleotide synthesis.
An abbreviated diagram of the pathway is shown in Figure 9.14. The first part of the PPP begins with glucose 6-phosphate, ends with ribulose 5-phosphate, and is irreversible. This part produces NADPH and involves the important rate-limiting enzyme glucose-6-phosphate dehydrogenase (G6PD). G6PD is induced by insulin because the abundance of sugar entering the cell under insulin stimulation will be shunted into both fuel utilization pathways (glycolysis and aerobic respiration), as well as fuel storage pathways (fatty acid synthesis, glycogenesis, and the PPP). The shunt is also inhibited by its product, NADPH, and is activated by one of its reactants, NADP+.
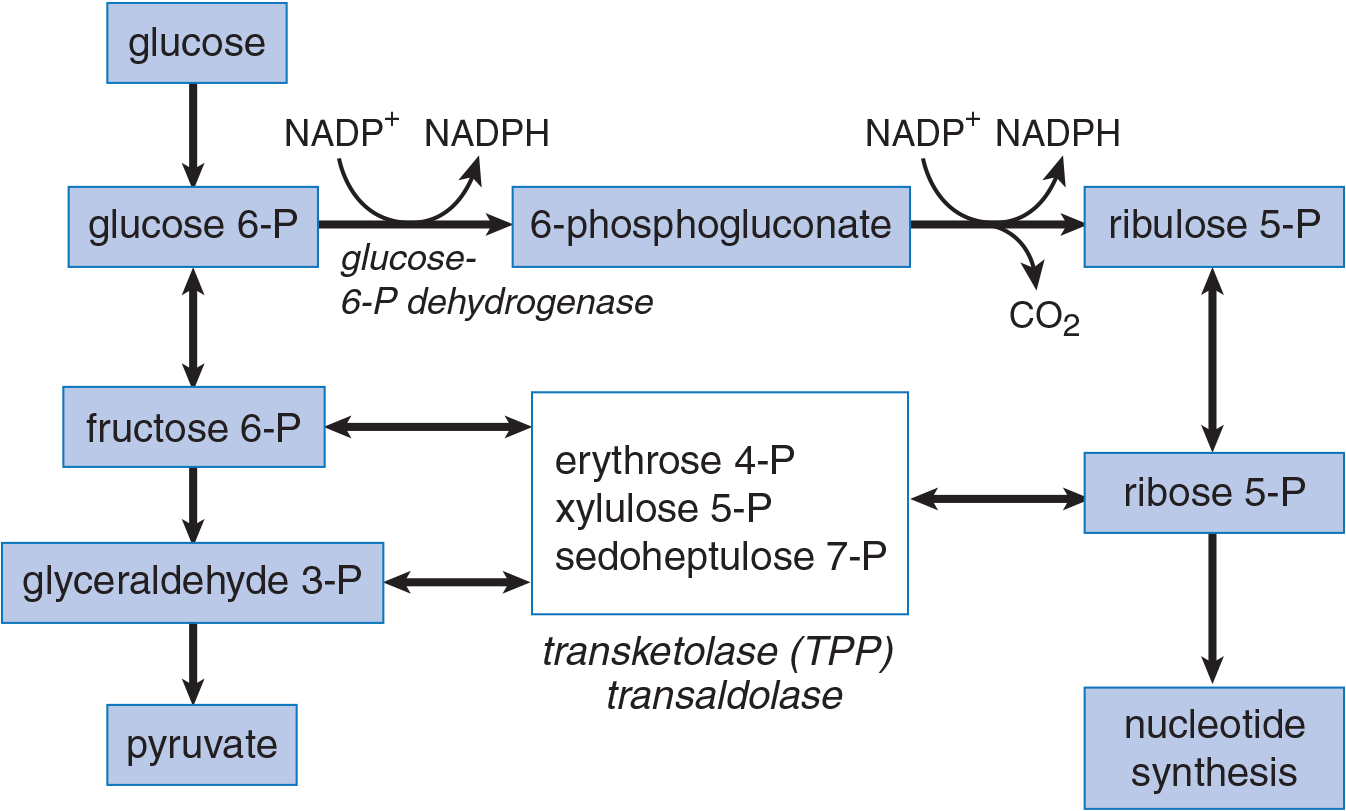
Figure 9.14 The Pentose Phosphate Pathway
REAL WORLD
G6PD deficiency is an X-linked disorder and is the most common inherited enzyme defect in the world. Because the PPP is critically important in maintaining levels of glutathione, which helps break down peroxides, these individuals are susceptible to oxidative stress, especially in red blood cells, which carry a large concentration of oxygen. Ingestion of certain oxidizing compounds (especially particular antibiotics and antimalarial medications) or infections can lead to high concentrations of reactive oxygen species, which cause red blood cell lysis. It is hypothesized that the defect evolved because it provides some resistance to malaria infection. G6PD deficiency has also been called favism because fava beans are a highly oxidizing food that will also cause hemolysis in these individuals.
The second part of the pathway, beginning with ribulose 5-phosphate, represents a series of reversible reactions that produce an equilibrated pool of sugars for biosynthesis, including ribose 5-phosphate for nucleotide synthesis. Because fructose 6-phosphate and glyceraldehyde 3-phosphate are among the sugars produced, intermediates can feed back into glycolysis; conversely, pentoses can be made from glycolytic intermediates without going through the G6PD reaction. These interconversions are primarily accomplished by the enzymes transketolase and transaldolase.
BRIDGE
The ribulose 5-phosphate created in the PPP is isomerized to ribose 5-phosphate, the backbone of nucleic acids. When coupled to a nitrogenous base, it forms a nucleotide that can be integrated into RNA. Make sure to review RNA synthesis (transcription; Chapter 7 of MCAT Biochemistry Review), as well as DNA synthesis (Chapter 6 of MCAT Biochemistry Review), because these are highly tested topics on the MCAT.
Functions of NADPH
While their names appear similar, NADPH and NADH are not the same thing. In the cell, NAD+ acts as a high-energy electron acceptor from a number of biochemical reactions. It thus can be thought of as a potent oxidizing agent because it helps another molecule be oxidized (and thus is reduced itself during the process). The NADH produced from this reduction of NAD+ can then feed into the electron transport chain to indirectly produce ATP.
KEY CONCEPT
NADPH and NADH are not the same thing. NAD+ is an energy carrier; NADPH is used in biosynthesis, in the immune system, and to help prevent oxidative damage.
Conversely, NADPH primarily acts as an electron donor in a number of biochemical reactions. It thus can be thought of as a potent reducing agent because it helps other molecules be reduced (and thus is oxidized itself during the process). Cells require NADPH for a variety of functions, including:
- Biosynthesis, mainly of fatty acids and cholesterol
- Assisting in cellular bleach production in certain white blood cells, thereby contributing to bactericidal activity
- Maintenance of a supply of reduced glutathione to protect against reactive oxygen species (acting as the body’s natural antioxidant)
This last function is important in protecting cells from free radical oxidative damage caused by peroxides. Hydrogen peroxide, H2O2, is produced as a byproduct in aerobic metabolism, and can break apart to form hydroxide radicals, OH•. Free radicals can attack lipids, including those in the phospholipids of the membrane. When oxidized, these lipids lose their function and can weaken the membrane, causing cell lysis. This is especially true in red blood cells, which contain high levels of oxygen, which, when oxidized by other free radicals, becomes the superoxide radical O2•. Free radicals can also damage DNA, potentially causing cancer. Glutathione is a reducing agent that can help reverse radical formation before damage is done to the cell.
MCAT CONCEPT CHECK 9.7:
Before you move on, assess your understanding of the material with these questions.
- What are the two major metabolic products of the pentose phosphate pathway (PPP)?
- __________________________________________________
- __________________________________________________
- What are three primary functions of NADPH?
- __________________________________________________
- __________________________________________________
- __________________________________________________
Conclusion
This chapter is critically important in your studying for the MCAT. The processes of carbohydrate metabolism that do not require oxygen are heavily tested, as is their integration. The body has evolved in such a way that we can use, store, or create fuel 24 hours a day, depending on the demands of the internal and external environment. We can turn on pathways when we need them and turn them off when we don’t. And the regulation of these pathways makes sense: for example, acetyl-CoA—a downstream product of glycolysis—can turn off the process of glycolysis and allow us to either store extra sugar as other biomolecules or generate sugar anew if we need it. Return to this chapter repeatedly during your studies to maximize points on metabolism on Test Day. In the next chapter, we’ll turn our attention to the oxygen-requiring carbohydrate metabolism processes, including the citric acid cycle, the electron transport chain (ETC), and oxidative phosphorylation.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Glucose Transport
- GLUT 2 is found in the liver (for glucose storage) and pancreatic β-islet cells (as part of the glucose sensor). It has a high Km.
- GLUT 4 is found in adipose tissue and muscle and is stimulated by insulin. It has a low Km.
Glycolysis
- Glycolysis occurs in the cytoplasm of all cells and does not require oxygen. It yields 2 ATP per molecule of glucose.
- Important glycolytic enzymes include:
- Glucokinase, which converts glucose to glucose 6-phosphate. It is present in the pancreatic β-islet cells as part of the glucose sensor and is responsive to insulin in the liver.
- Hexokinase, which converts glucose to glucose 6-phosphate in peripheral tissues.
- Phosphofructokinase-1 (PFK-1), which phosphorylates fructose 6-phosphate to fructose 1,6-bisphosphate in the rate-limiting step of glycolysis. PFK-1 is activated by AMP and fructose 2,6-bisphosphate (F2,6-BP) and is inhibited by ATP and citrate.
- Phosphofructokinase-2 (PFK-2), which produces the F2,6-BP that activates PFK-1. It is activated by insulin and inhibited by glucagon.
- Glyceraldehyde-3-phosphate dehydrogenase produces NADH, which can feed into the electron transport chain.
- 3-phosphoglycerate kinase and pyruvate kinase each perform substrate-level phosphorylation, placing an inorganic phosphate (Pi) onto ADP to form ATP.
- The enzymes that catalyze irreversible reactions are glucokinase/hexokinase, PFK-1, and pyruvate kinase.
- The NADH produced in glycolysis is oxidized by the mitochondrial electron transport chain when oxygen is present.
- If oxygen or mitochondria are absent, the NADH produced in glycolysis is oxidized by cytoplasmic lactate dehydrogenase. Examples include red blood cells, skeletal muscle (during short, intense bursts of exercise), and any cell deprived of oxygen.
Other Monosaccharides
- Galactose comes from lactose in milk. It is trapped in the cell by galactokinase, and converted to glucose 1-phosphate via galactose-1-phosphate uridyltransferase and an epimerase.
- Fructose comes from honey, fruit, and sucrose (common table sugar). It is trapped in the cell by fructokinase, and then cleaved by aldolase B to form glyceraldehyde and DHAP.
Pyruvate Dehydrogenase
- Pyruvate dehydrogenase refers to a complex of enzymes that convert pyruvate to acetyl-CoA.
- It is stimulated by insulin and inhibited by acetyl-CoA.
Glycogenesis and Glycogenolysis
- Glycogenesis (glycogen synthesis) is the production of glycogen using two main enzymes:
- Glycogen synthase, which creates α-1,4 glycosidic links between glucose molecules. It is activated by insulin in liver and muscle.
- Branching enzyme, which moves a block of oligoglucose from one chain and adds it to the growing glycogen as a new branch using an α-1,6 glycosidic link.
- Glycogenolysis is the breakdown of glycogen using two main enzymes:
- Glycogen phosphorylase, which removes single glucose 1-phosphate molecules by breaking α-1,4 glycosidic links. In the liver, it is activated by glucagon to prevent low blood sugar; in exercising skeletal muscle, it is activated by epinephrine and AMP to provide glucose for the muscle itself.
- Debranching enzyme, which moves a block of oligoglucose from one branch and connects it to the chain using an α-1,4 glycosidic link. It also removes the branchpoint, which is connected via an α-1,6 glycosidic link, releasing a free glucose molecule.
Gluconeogenesis
- Gluconeogenesis occurs in both the cytoplasm and mitochondria, predominantly in the liver. There is a small contribution from the kidneys.
- Most of gluconeogenesis is simply the reverse of glycolysis, using the same enzymes. The three irreversible steps of glycolysis must be bypassed by different enzymes:
- Pyruvate carboxylase converts pyruvate into oxaloacetate, which is converted to phosphoenolpyruvate by phosphoenolpyruvate carboxykinase (PEPCK). Together, these two enzymes bypass pyruvate kinase. Pyruvate carboxylase is activated by acetyl-CoA from β-oxidation; PEPCK is activated by glucagon and cortisol.
- Fructose-1,6-bisphosphatase converts fructose 1,6-bisphosphate to fructose 6-phosphate, bypassing phosphofructokinase-1. This is the rate-limiting step of gluconeogenesis. It is activated by ATP directly and glucagon indirectly (via decreased levels of fructose 2,6-bisphosphate). It is inhibited by AMP directly and insulin indirectly (via increased levels of fructose 2,6-bisphosphate).
- Glucose-6-phosphatase converts glucose 6-phosphate to free glucose, bypassing glucokinase. It is found only in the endoplasmic reticulum of the liver.
The Pentose Phosphate Pathway
- The pentose phosphate pathway (PPP), also known as the hexose monophosphate (HMP) shunt, occurs in the cytoplasm of most cells, generating NADPH and sugars for biosynthesis (derived from ribulose 5-phosphate).
- The rate-limiting enzyme is glucose-6-phosphate dehydrogenase, which is activated by NADP+ and insulin and inhibited by NADPH.
ANSWERS TO CONCEPT CHECKS
**9.1**
-
GLUT 2 GLUT 4 Important tissues Liver, pancreas Adipose tissue, muscle
***K*m** High (~15 mM) Low (~5 mM)
Saturated at normal glucose levels? No—cannot be saturated under normal physiological conditions Yes—saturated when glucose levels are only slightly above 5 mM
Responsive to insulin? No (but serves as glucose sensor to cause release of insulin in pancreatic β-cells) Yes
- GLUT 4 is saturated when glucose levels are only slightly above 5 mM, so glucose entry can only be increased by increasing the number of transporters. Insulin promotes the fusion of vesicles containing preformed GLUT 4 with the cell membrane.
**9.2**
-
- Hexokinase phosphorylates glucose to form glucose 6-phosphate, “trapping” glucose in the cell. It is inhibited by glucose 6-phosphate. It is irreversible.
- Glucokinase also phosphorylates and “traps” glucose in liver and pancreatic cells, and works with GLUT 2 as part of the glucose sensor in β-islet cells. In liver cells, it is induced by insulin. It is irreversible.
- PFK-1 catalyzes the rate-limiting step of glycolysis, phosphorylating fructose 6-phosphate to fructose 1,6-bisphosphate using ATP. It is inhibited by ATP, citrate, and glucagon. It is activated by AMP, fructose 2,6-bisphosphate, and insulin. It is irreversible.
- Glyceraldehyde-3-phosphate dehydrogenase generates NADH while phosphorylating glyceraldehyde 3-phosphate to 1,3-bisphosphoglycerate. It is reversible.
- 3-Phosphoglycerate kinase performs a substrate-level phosphorylation, transferring a phosphate from 1,3-bisphosphoglycerate to ADP, forming ATP and 3-phosphoglycerate. It is reversible.
- Pyruvate kinase performs another substrate-level phosphorylation, transferring a phosphate from phosphoenolpyruvate (PEP) to ADP, forming ATP and pyruvate. It is activated by fructose 1,6-bisphosphate. It is irreversible.
- Fermentation must occur to regenerate NAD+, which is in limited supply in cells. Fermentation generates no ATP or energy carriers; it merely regenerates the coenzymes needed in glycolysis.
- The binding of 2,3-BPG decreases hemoglobin’s affinity for oxygen. Fetal hemoglobin must be able to “steal” oxygen from maternal hemoglobin at the placental interface; therefore, it would be disadvantageous to lower its affinity for oxygen.
**9.3**
- Galactose is phosphorylated by galactokinase, trapping it in the cell. Galactose-1-phosphate uridyltransferase produces glucose 1-phosphate, a glycolytic intermediate, thus linking the pathways.
- Fructose is phosphorylated by fructokinase, trapping it in the cell (with a small contribution from hexokinase). Aldolase B produces dihydroxyacetone phosphate (DHAP) and glyceraldehyde (which can be phosphorylated to form glyceraldehyde 3-phosphate), which are glycolytic intermediates, thus linking the pathways.
**9.4**
- Pyruvate, NAD+, and CoA are the reactants of the PDH complex. Acetyl-CoA, NADH, and CO2 are the products.
- Acetyl-CoA inhibits the PDH complex. As a product of the enzyme complex, a buildup of acetyl-CoA from either the citric acid cycle or fatty acid oxidation signals that the cell is energetically satisfied and that the production of acetyl-CoA should be slowed or stopped. Pyruvate can then be used to form other products, such as oxaloacetate for use in gluconeogenesis.
**9.5**
- Glycogen is made up of a core protein of glycogenin with linear chains of glucose emanating out from the center, connected by α-1,4 glycosidic links. Some of these chains are branched, which requires α-1,6 glycosidic links.
- Glycogen synthase attaches the glucose molecule from UDP-glucose to the growing glycogen chain, forming an α-1,4 link in the process. Branching enzyme creates a branch by breaking an α-1,4 link in the growing chain and moving a block of oligoglucose to another location in the glycogen granule. The oligoglucose is then attached with an α-1,6 link.
- Glycogen phosphorylase removes a glucose molecule from glycogen using a phosphate, breaking the α-1,4 link and creating glucose 1-phosphate. Debranching enzyme moves all of the glucose from a branch to a longer glycogen chain by breaking an α-1,4 link and forming a new α-1,4 link to the longer chain. The branchpoint is left behind; this is removed by breaking the α-1,6 link to form a free molecule of glucose.
**9.6**
- Gluconeogenesis occurs when an individual has been fasting for >12 hours. To carry out gluconeogenesis, hepatic (and renal) cells must have enough energy to drive the process of glucose creation, which requires sufficient fat stores to undergo β-oxidation.
-
Gluconeogenic Enzyme Replaces
Pyruvate carboxylase Pyruvate kinase
Phosphoenolpyruvate carboxykinase (PEPCK) Pyruvate kinase
Fructose-1,6-bisphosphatase Phosphofructokinase-1
Glucose-6-phosphatase Glucokinase
- Acetyl-CoA inhibits pyruvate dehydrogenase complex while activating pyruvate carboxylase. The net effect is to shift from burning pyruvate in the citric acid cycle to creating new glucose molecules for the rest of the body. The acetyl-CoA for this regulation comes predominantly from β-oxidation, not glycolysis.
- The last enzyme in gluconeogenesis is glucose-6-phosphatase so patients with von Gierke's disease are unable to perform gluconeogenesis in addition to glycogenolysis. This means patients will be unable to produce glucose during periods of fasting (resulting in hypoglycemia). Furthermore, given a blocker in the gluconeogenic pathway, a buildup of intermediates (including lactate resulting in lactic acidosis) would also be expected.
**9.7**
- The two major metabolic products of the pentose phosphate pathway are ribose 5-phosphate and NADPH.
- NADPH is involved in lipid biosynthesis, bactericidal bleach formation in certain white blood cells, and maintenance of glutathione stores to protect against reactive oxygen species.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. B
The GLUT 2 receptor, (B), has a high Km and is expressed in hepatocytes of the liver. When the concentration of glucose in the blood is high, glucose is transported into the liver with first order kinetics. Also, in the pancreas, GLUT 2 and glucokinase serve as a glucose sensor. These observations together support (B) as the right answer.
2. D
The liver, like all cells, needs a constant supply of glucose; however, it is able to produce its own glucose through gluconeogenesis (cells in the kidney can also complete low levels of gluconeogenesis). The other cells listed here are absolutely dependent on a glucose source from the blood for energy, although they may also use other fuels in addition to glucose. For example, the brain can utilize ketone bodies during lengthy periods of starvation; however, it still requires at least some glucose for proper function.
3. D
GLUT is an abbreviation for glucose transporter and describes a family of sugar transporters with varying distributions and activities. GLUT 4 is found in adipose tissue and muscle, and mediates insulin-stimulated glucose uptake; in fact, it is the only insulin-responsive glucose transporter. Insulin acts via its receptor to translocate GLUT 4 to the plasma membrane. GLUT 4 in skeletal muscle is also stimulated by exercise through an insulin-independent pathway.
4. B
After an overnight fast, the liver is producing glucose and glucokinase activity would be insignificant. Glucokinase is used to trap extra glucose in liver cells as part of a storage mechanism; with low blood glucose, liver cells would be generating new glucose, not storing it. It is also in the pancreas, where it serves as a glucose sensor; if glucose levels are low, it has little activity in this tissue as well. Malate dehydrogenase, (A), and α-ketoglutarate dehydrogenase, (C), are citric acid cycle enzymes. Phosphofructokinase-1, (D), is a glycolytic enzyme. Other enzymes used in glycolysis, the citric acid cycle, or gluconeogenesis, such as phosphofructokinase-1, would be expected to maintain normal activity after an overnight fast, using glucose derived from glycogen or gluconeogenesis, rather than orally ingested glucose.
5. C
This question asks for an enzyme whose role is not to trap sugar in the cell. Phosphofructokinase (C) is the rate-limiting enzyme of glycolysis. Its role is to phosphorylate fructose-6-phosphate to fructose-1,6-bisphosphate during the third step of glycolysis. Importantly, the substrate of phosphofructokinase, i.e. fructose-6-phosphate, has already been once phosphorylated before reaching phosphofructokinase. Therefore, fructose-6-phosphate is already trapped in the cell before even reaching phosphofructokinase. For this reason, it cannot be said that phosphofructokinase is used to trap sugar in the cell. By contrast, (A), (B), and (D), can be eliminated because hexokinase phosphorylates glucose, galactokinase phosphorylates galactose, and fructokinase phosphorylates fructose, each reaction trapping the previously unphosphorylated sugar in the cell.
6. C
Pyruvate is converted primarily into three main intermediates: acetyl-CoA, (B), for the citric acid cycle (via pyruvate dehydrogenase complex); lactate, (D), during fermentation (via lactate dehydrogenase); or oxaloacetate, (C), for gluconeogenesis (via pyruvate carboxylase). High levels of acetyl-CoA, which is produced during β-oxidation, will inhibit pyruvate dehydrogenase and shift the citric acid cycle to run in the reverse direction, producing oxaloacetate for gluconeogenesis. Acetyl-CoA also stimulates pyruvate carboxylase directly.
7. D
The pattern described for this child’s glycogen demonstrates appropriate production: there are long chains of glucose monomers, implying that glycogen synthase works. There are also branch points, implying that branching enzyme, (B) works. During glycogenolysis, it seems that the child is able to remove individual glucose monomers and process glycogen down to the branch point itself, which requires glycogen phosphorylase, (A), and α-1,4:α-1,4 transferase, (C). The metabolic problem here is removing the final glucose at the branch point, which is an α-1,6 (not α-1,4) link. This requires (D), α-1,6 glucosidase.
8. C
Citrate is produced by citrate synthase from acetyl-CoA and oxaloacetate. This reaction takes place in the mitochondria. When the citric acid cycle slows down, citrate accumulates. In the cytosol, it acts as a negative allosteric regulator of phosphofructokinase-1, the enzyme that catalyzes the rate-limiting step of glycolysis.
9. D
In most biochemical pathways, only a few enzymatic reactions are under regulatory control. These often occur either at the beginning of pathways or at pathway branch points. The pyruvate dehydrogenase (PDH) complex controls the link between glycolysis and the citric acid cycle, and decarboxylates pyruvate (the end product of glycolysis) with production of NADH and acetyl-CoA (the substrate for the citric acid cycle). After intense exercise, one would expect PDH to be highly active to generate ATP. ADP levels, (A), should be high because ATP was just burned by the muscle. Acetyl-CoA, (B), is an inhibitor of PDH, causing a shift of pyruvate into the gluconeogenic pathway. A high NADH/NAD+ ratio, (C), would imply that the cell is already energetically satisfied and not in need of energy, which would not be expected in intensely exercising muscle.
10. B
In this question, the right answer will be the molecule whose metabolism will be the least impacted by the inhibition of phosphofructokinase-1 (PFK-1). The enzyme PFK-1 catalyzes the rate-limiting step of glycolysis. So sugars that enter the glycolytic pathway after this rate-limiting step, i.e. downstream of PFK-1, would be least impacted by inhibition of PFK-1. Given this criterion, recall the metabolic pathway of fructose, choice (B), which is phosphorylated and then cleaved into glyceraldehyde and DHAP, both of which enter glycolysis downstream of PFK-1. Therefore the metabolism of fructose does not depend on PFK-1, and would therefore be least impacted by the inhibition of PFK-1, making (B) the correct answer. By contrast, glucose, (C), is metabolised using the usual glycolytic pathway, and would therefore be impacted by inhibition of PFK-1. Similarly, galactose, (D), is phosphorylated and subsequently isomerized into glucose-6-phosphate, which then follows the usual pathway and would therefore be impacted by inhibition of PFK-1. And lactose, (A), is cleaved by lactase into glucose and galactose. For these reasons, (A), (B), and (D) can be eliminated.
11. B
Based on the question stem, we can infer that the antibiotics must have been an oxidative stress on the patient (antibiotics, antimalarial medications, infections, certain foods like fava beans, and other common exposures can induce an oxidative stress). The pentose phosphate pathway is responsible for generating NADPH, which is used to reduce glutathione, one of the natural antioxidants present in the body. In individuals with glucose-6-phosphate dehydrogenase (G6PD) deficiency, NADPH cannot be produced at sufficient levels, and oxidative stresses lead to cell membrane and protein (hemoglobin) damage. Note that you do not need to actually know the disease to answer this question; merely knowing that the enzyme must be from the pentose phosphate pathway, which is involved in mitigating oxidative stress, is sufficient.
12. A
The irreversible enzymes in glycolysis are hexokinase (or glucokinase in liver and pancreatic β-cells), phosphofructokinase-1, and pyruvate kinase. Pyruvate dehydrogenase is not considered a glycolytic enzyme because it requires the mitochondria to function. The list below shows the correct pairing of glycolytic enzymes with gluconeogenic enzymes:
- Hexokinase or glucokinase / glucose-6-phosphatase
- Phosphofructokinase-1 / fructose-1,6-bisphosphatase
- Pyruvate kinase / pyruvate carboxylase and phosphoenol-pyruvate carboxykinase (PEPCK)
13. B
After a fast, the liver must contribute glucose into the bloodstream through two main processes: glycogenolysis (early to intermediate fasting) and gluconeogenesis (intermediate to late fasting). The other processes would continue at normal basal levels or have decreased activity after a fast.
14. A
A deficiency in branching enzyme would result in chains of unbranched glycogen. Since glycogen synthase is not affected, the chain length would be normal, indicating a normal abundance of α-1,4 linkages. However, a loss of branching would correspond to a decrease in α-1,6, linkages. Thus, the ratio of α-1,4 to α-1,6 would increase as division by a smaller denominator yields a larger value, making (A) the correct answer.
15. A
Hexokinase catalyzes an important irreversible step of glycolysis, but it is not the rate-limiting step. Phosphofructokinase-1 catalyzes the rate-limiting step of glycolysis. Glycogen synthase, (B), catalyzes the rate-limiting step of glycogenesis; glucose-6-phosphate dehydrogenase, (C), catalyzes the rate-limiting step of the pentose phosphate pathway; and fructose-1,6-bisphosphatase, >(D), catalyzes the rate-limiting step of gluconeogenesis.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 4
- Carbohydrate Structure and Function
- Biochemistry Chapter 10
- Carbohydrate Metabolism II
- Biochemistry Chapter 12
- Bioenergetics and Regulation of Metabolism
- Biology Chapter 7
- The Cardiovascular System
- General Chemistry Chapter 5
- Chemical Kinetics
- General Chemistry Chapter 11
- Oxidation–Reduction Reactions