Chapter 10: Carbohydrate Metabolism II: Aerobic Respiration

Chapter 10: Carbohydrate Metabolism II: Aerobic Respiration
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- During a myocardial infarction, the oxygen supply to an area of the heart is dramatically reduced, forcing the cardiac myocytes to switch to anaerobic metabolism. Under these conditions, which of the following enzymes would be activated by increased levels of intracellular AMP?
- Succinate dehydrogenase
- Phosphofructokinase-1
- Isocitrate dehydrogenase
- Pyruvate dehydrogenase
- A patient has been exposed to a toxic compound that increases the permeability of mitochondrial membranes to protons. Which of the following metabolic changes would be expected in this patient?
- Increased ATP levels
- Increased oxygen utilization
- Increased ATP synthase activity
- Decreased pyruvate dehydrogenase activity
- Which of the following INCORRECTLY pairs a metabolic process with its site of occurrence?
- Glycolysis—cytosol
- Citric acid cycle—outer mitochondrial membrane
- ATP phosphorylation—cytosol and mitochondria
- Electron transport chain—inner mitochondrial membrane
- Which of the following processes has the following net reaction?
2 acetyl-CoA + 6 NAD+ + 2 FAD + 2 GDP + 2 Pi + 6 H2O → 4 CO2 + 6 NADH + 2 FADH2+ 2 GTP + 6 H++ 2 CoA–SH
- Pyruvate decarboxylation
- Fermentation
- Tricarboxylic acid cycle
- Electron transport chain
- In glucose degradation under aerobic conditions:
- oxygen is the final electron acceptor.
- oxygen is necessary for all ATP synthesis.
- net water is consumed.
- the proton-motive force is necessary for all ATP synthesis.
- Fatty acids enter the catabolic pathway in the form of:
- glycerol.
- adipose tissue.
- acetyl-CoA.
- ketone bodies.
- All of the following methods are used to produce acetyl-CoA in cells EXCEPT for one. What is the exception?
- Decarboxylation of pyruvate
- Catabolism of ketogenic amino acids
- Reduction of fatty acids
- Metabolism of alcohol
- In which part of the cell is cytochrome c located?
- Mitochondrial matrix
- Outer mitochondrial membrane
- Inner mitochondrial membrane
- Cytosol
- Which of the following correctly shows the amount of ATP produced from the given high-energy carriers?
- FADH2 → 1 ATP
- FADH2 → 1.5 ATP
- NADH → 3 ATP
- NADH → 3.5 ATP
- Why is it preferable to cleave thioester links rather than typical ester links in aerobic metabolism?
- Oxygen must be conserved for the electron transport chain.
- Thioester hydrolysis has a higher energy yield.
- Typical ester hydrolysis cannot occur in vivo.
- Thioester cleavage requires more energy.
- Which enzyme converts GDP to GTP?
- Nucleosidediphosphate phosphatase
- Nucleosidediphosphate kinase
- Isocitrate dehydrogenase
- Pyruvate dehydrogenase
- Which of the following best explains why cytosolic NADH can yield potentially less ATP than mitochondrial NADH?
- Cytosolic NADH always loses energy when transferring electrons.
- Once NADH enters the matrix from the cytosol, it becomes FADH2.
- Electron transfer from cytosol to matrix can take more than one pathway.
- There is an energy cost for bringing cytosolic NADH into the matrix.
- In high doses, aspirin functions as a mitochondrial uncoupler. How would this affect glycogen stores?
- It causes depletion of glycogen stores.
- It has no effect on glycogen stores.
- It promotes additional storage of glucose as glycogen.
- Its effect on glycogen stores varies from cell to cell.
- Which complex does NOT contribute to the proton-motive force?
- Complex I
- Complex II
- Complex III
- Complex IV
- Which of the following directly provides the energy needed to form ATP in the mitochondrion?
- Electron transfer in the electron transport chain
- An electrochemical proton gradient
- Oxidation of acetyl-CoA
- β-Oxidation of fatty acids
Answer Key
- B
- B
- B
- C
- A
- C
- C
- C
- B
- B
- B
- C
- A
- B
- B
Chapter 10: Carbohydrate Metabolism II: Aerobic Respiration
CHAPTER 10
CARBOHYDRATE METABOLISM II: AEROBIC RESPIRATION
In This Chapter
10.1 Acetyl-CoA
Methods of Forming Acetyl-CoA
10.2 Reactions of the Citric Acid Cycle
Key Reactions
Net Results and ATP Yield
Regulation
10.3 The Electron Transport Chain
Electron Flow and Complexes
The Proton-Motive Force
NADH Shuttles
10.4 Oxidative Phosphorylation
Chemiosmotic Coupling
Regulation
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 6% of all questions about biochemistry on the MCAT.
This chapter covers material from the following AAMC content category:
1D: Principles of bioenergetics and fuel molecule metabolism
Introduction
Have you ever heard that eating peach pits is deadly? Before you start panicking about the snack you had during your study break, you should know that the accuracy of such a statement is debatable. While it is true that digesting peach pits can result in the formation of trace amounts of cyanide, the concentration is far too low to be clinically worrisome. Cyanide is a poison that binds irreversibly to cytochrome a/a3, a protein located in the electron transport chain of the mitochondria. Why can this be deadly? Blocking the electron transport chain (ETC) inhibits aerobic respiration from yielding the ATP the body requires to function properly. Cyanide poisoning leaves cells unable to utilize oxygen for aerobic respiration because it blocks oxygen from binding to the ETC. Therefore, symptoms resemble those of tissue hypoxia: perceived difficulty breathing, general weakness, and, in higher doses, cardiac arrest followed by death within minutes.
But what about the metabolic pathways described in Chapter 9 of MCAT Biochemistry Review—don’t they produce energy without oxygen? While glycolysis does not depend on oxygen, it only yields a net 2 ATP per molecule of glucose, which is not nearly enough to maintain the body’s energy requirements. This brings us to two of the most tested topics on the MCAT: the citric acid cycle and oxidative phosphorylation.
In this chapter, we’ll take a close look at what’s gained when the products of glycolysis and other derivatives of metabolic pathways enter the citric acid cycle. We’ll also look at how this process is regulated with regard to the substrates, products, and reactions involved. Lastly, we’ll observe what happens when this cycle’s products undergo oxidative phosphorylation, with particular emphasis on how the electron transport chain facilitates the process and the ATP that is yielded.
10.1 Acetyl-CoA
LEARNING OBJECTIVES
After Chapter 10.1, you will be able to:
- Detail four potential energy sources for the synthesis of acetyl-CoA
- Identify the major inputs and outputs through the pyruvate dehydrogenase complex
The citric acid cycle, also called the Krebs cycle or the tricarboxylic acid (TCA) cycle, occurs in the mitochondria. The main function of this cycle is the oxidation of acetyl-CoA to CO2 and H2O. In addition, the cycle produces the high-energy electron-carrying molecules NADH and FADH2. Acetyl-CoA can be obtained from the metabolism of carbohydrates, fatty acids, and amino acids, making it a key molecule in the crossroads of many metabolic pathways and a highly testable compound.
Methods of Forming Acetyl-CoA
Recall from Chapter 9 of MCAT Biochemistry Review that after glucose undergoes glycolysis, its product, pyruvate, enters the mitochondrion via active transport and is oxidized and decarboxylated. These reactions are catalyzed by a multienzyme complex called the pyruvate dehydrogenase complex, which is located in the mitochondrial matrix. As we take a deeper look at the enzymes that make up this complex, as well as the substrates and products of their reactions, it is helpful to follow the carbons in the molecules. For example, the three-carbon pyruvate is cleaved into a two-carbon acetyl group and carbon dioxide. This reaction is irreversible, which explains why glucose cannot be formed directly from acetyl-CoA. In mammals, pyruvate dehydrogenase complex is made up of five enzymes: pyruvate dehydrogenase (PDH), dihydrolipoyl transacetylase,dihydrolipoyl dehydrogenase,pyruvate dehydrogenase kinase, and pyruvate dehydrogenase phosphatase. While the first three work in concert to convert pyruvate to acetyl-CoA, the latter two regulate the actions of PDH. Figure 10.1 shows the overall reaction for the conversion of pyruvate to acetyl-CoA. The reaction is exergonic
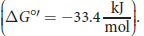
The complex is inhibited by an accumulation of acetyl-CoA and NADH that can occur if the electron transport chain is not properly functioning or is inhibited.

Figure 10.1 Overall Reaction of Pyruvate Dehydrogenase Complex
MCAT EXPERTISE
Similar to the gluco–/glyco– terminology in Chapter 9 of MCAT Biochemistry Review, it is critical to keep straight the various enzymes containing pyruvate:pyruvate dehydrogenase (PDH), its two regulators (PDH kinase andPDH phosphatase), and pyruvate carboxylase, an enzyme in gluconeogenesis.
Note that coenzyme A (CoA) is written as CoA–SH in the reaction above. This is because CoA is a thiol, containing an –SH group. When acetyl-CoA forms, it does so via covalent attachment of the acetyl group to the –SH group, resulting in the formation of a thioester, which contains sulfur instead of the typical oxygen ester –OR. The formation of a thioester rather than a typical ester is worth noting because of the high-energy properties of thioesters. That is to say, when a thioester undergoes a reaction such as hydrolysis, a significant amount of energy will be released. This can be enough to drive other reactions forward, like the citric acid cycle. The pyruvate dehydrogenase complex enzymes needed to catalyze acetyl-CoA formation are listed below in sequential order, and the mechanism is shown in Figure 10.2.
- Pyruvate dehydrogenase (PDH): Pyruvate is oxidized, yielding CO2, while the remaining two-carbon molecule binds covalently to thiamine pyrophosphate (vitamin B1, TPP). TPP is a coenzyme held by noncovalent interactions to PDH. Mg2+ is also required.
- Dihydrolipoyl transacetylase: The two-carbon molecule bonded to TPP is oxidized and transferred to lipoic acid, a coenzyme that is covalently bonded to the enzyme. Lipoic acid’s disulfide group acts as an oxidizing agent, creating the acetyl group. The acetyl group is now bonded to lipoic acid via thioester linkage. After this, dihydrolipoyl transacetylase catalyzes the CoA–SH interaction with the newly formed thioester link, causing transfer of an acetyl group to form acetyl-CoA. Lipoic acid is left in its reduced form.
- Dihydrolipoyl dehydrogenase: Flavin adenine dinucleotide (FAD) is used as a coenzyme in order to reoxidize lipoic acid, allowing lipoic acid to facilitate acetyl-CoA formation in future reactions. As lipoic acid is reoxidized, FAD is reduced to FADH2. In subsequent reactions, this FADH2 is reoxidized to FAD, while NAD+ is reduced to NADH.
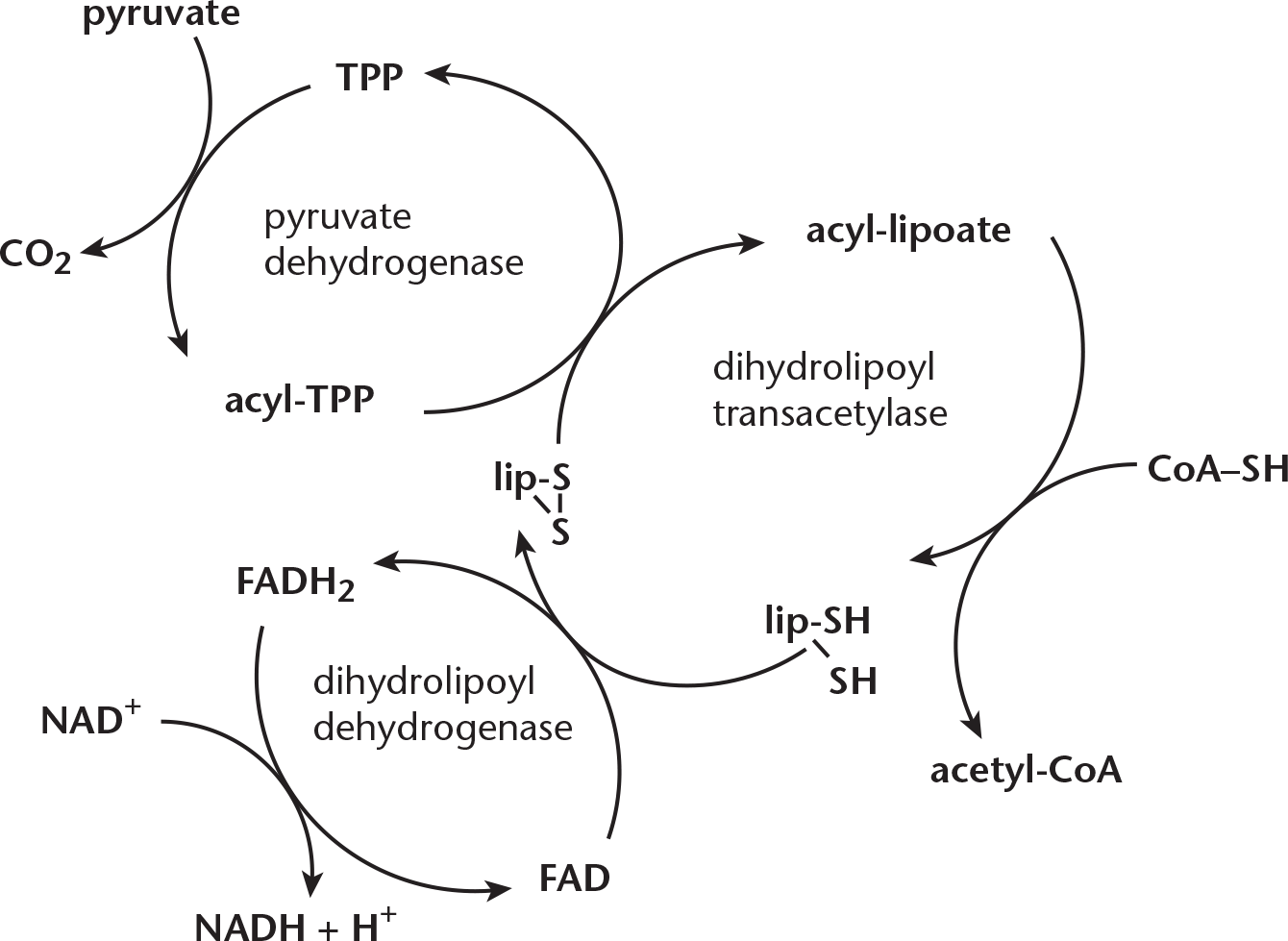
Figure 10.2 Mechanism of Pyruvate Dehydrogenase
REAL WORLD
In studies of pathologies that affect the central cholinergic system such as Alzheimer’s disease, Huntington’s disease, and even alcoholism, a decrease in glucose metabolism and oxidative phosphorylation has been observed in the brain. Ongoing research will hopefully determine if the resulting lack of acetyl-CoA could be a cause of the disease or a result of the disease. With decreased amounts of acetyl-CoA, not only is energy production a concern, but also the production of the neurotransmitter acetylcholine.
While glycolysis is a heavily reviewed and heavily tested contributor to the production of acetyl-CoA, other pathways are capable of forming acetyl-CoA. These pathways act on fatty acids, ketogenic amino acids, ketone bodies, and alcohol. Descriptions of these pathways are provided below. The ultimate production of acetyl-CoA allows all of these pathways to culminate in the final common pathway of the citric acid cycle.
- Fatty acid oxidation (***β*-oxidation):In the cytosol, a process calledactivation** causes a thioester bond to form between carboxyl groups of fatty acids and CoA–SH. Because this activated fatty acyl-CoA cannot cross the inner mitochondrial membrane, the fatty acyl group is transferred to carnitine via a transesterification reaction, as shown in Figure 10.3. Carnitine is a molecule that can cross the inner membrane with a fatty acyl group in tow. Once acyl-carnitine crosses the inner membrane; it transfers the fatty acyl group to a mitochondrial CoA–SH via another transesterification reaction. In other words, carnitine’s function is merely to carry the acyl group from a cytosolic CoA–SH to a mitochondrial CoA–SH. Once acyl-CoA is formed in the matrix, β-oxidation can occur, which removes two-carbon fragments from the carboxyl end.
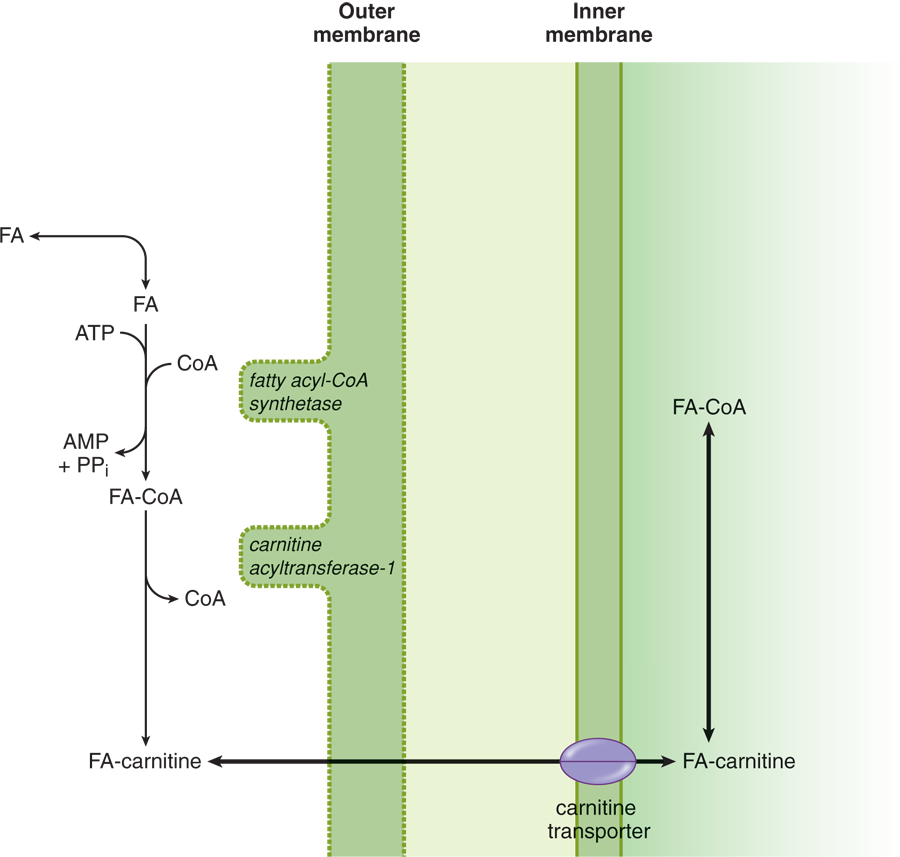
Figure 10.3 Fatty Acid Activation and Transport
- Amino acid catabolism: Certain amino acids can be used to form acetyl-CoA. These amino acids must lose their amino group via transamination; their carbon skeletons can then form ketone bodies. These amino acids are termed ketogenic for that reason. The conversion of ketone bodies to acetyl-CoA is mentioned below.
- Ketones: Although acetyl-CoA is typically used to produce ketones when the pyruvate dehydrogenase complex is inhibited, the reverse reaction can occur as well.
- Alcohol: When alcohol is consumed in moderate amounts, the enzymes alcohol dehydrogenase and acetaldehyde dehydrogenase convert it to acetyl-CoA. However, this reaction is accompanied by NADH buildup, which inhibits the Krebs cycle. Therefore, the acetyl-CoA formed through this process is used primarily to synthesize fatty acids.
BRIDGE
Once formed, mitochondrial acyl-CoA can undergo β-oxidation. This process is discussed in Chapter 11 of MCAT Biochemistry Review.
REAL WORLD
While the brain normally uses glucose for energy, under conditions such as starvation, ketone bodies can become the brain’s major source of energy.
MCAT CONCEPT CHECK 10.1:
Before you move on, assess your understanding of the material with these questions.
- What is the overall reaction of the pyruvate dehydrogenase complex?
___________________________________
- What other molecules can be used to make acetyl-CoA, and how does the body perform this conversion for each?
Molecule Mechanism of Conversion to Acetyl-CoA
10.2 Reactions of the Citric Acid Cycle
LEARNING OBJECTIVES
After Chapter 10.2, you will be able to:
- Explain the purpose of the citric acid cycle, including major inputs/outputs
- Identify the importance of key enzymes within the citric acid cycle
- Recall the inhibitors and activators of the enzymes citrate synthase, isocitrate dehydrogenase, and alpha-ketoglutarate dehydrogenase complex
The citric acid cycle takes place in the mitochondrial matrix and begins with the coupling of a molecule of acetyl-CoA to a molecule of oxaloacetate. While parts of this molecule are oxidized to carbon dioxide and both energy (GTP) and energy carriers (NADH and FADH2) are produced, the other substrates and products of the cycle are reused over and over again. Although oxygen is not directly required in the cycle, the pathway will not occur anaerobically. This is because NADH and FADH2 will accumulate if oxygen is not available for the electron transport chain and will inhibit the cycle. As we look at the individual reactions that take place during the citric acid cycle, it cannot be overemphasized that this process is exactly what it’s called: a cycle, not just a series of reactions. An overview of the cycle is provided in Figure 10.4, and we’ll take a deeper look at those steps below.
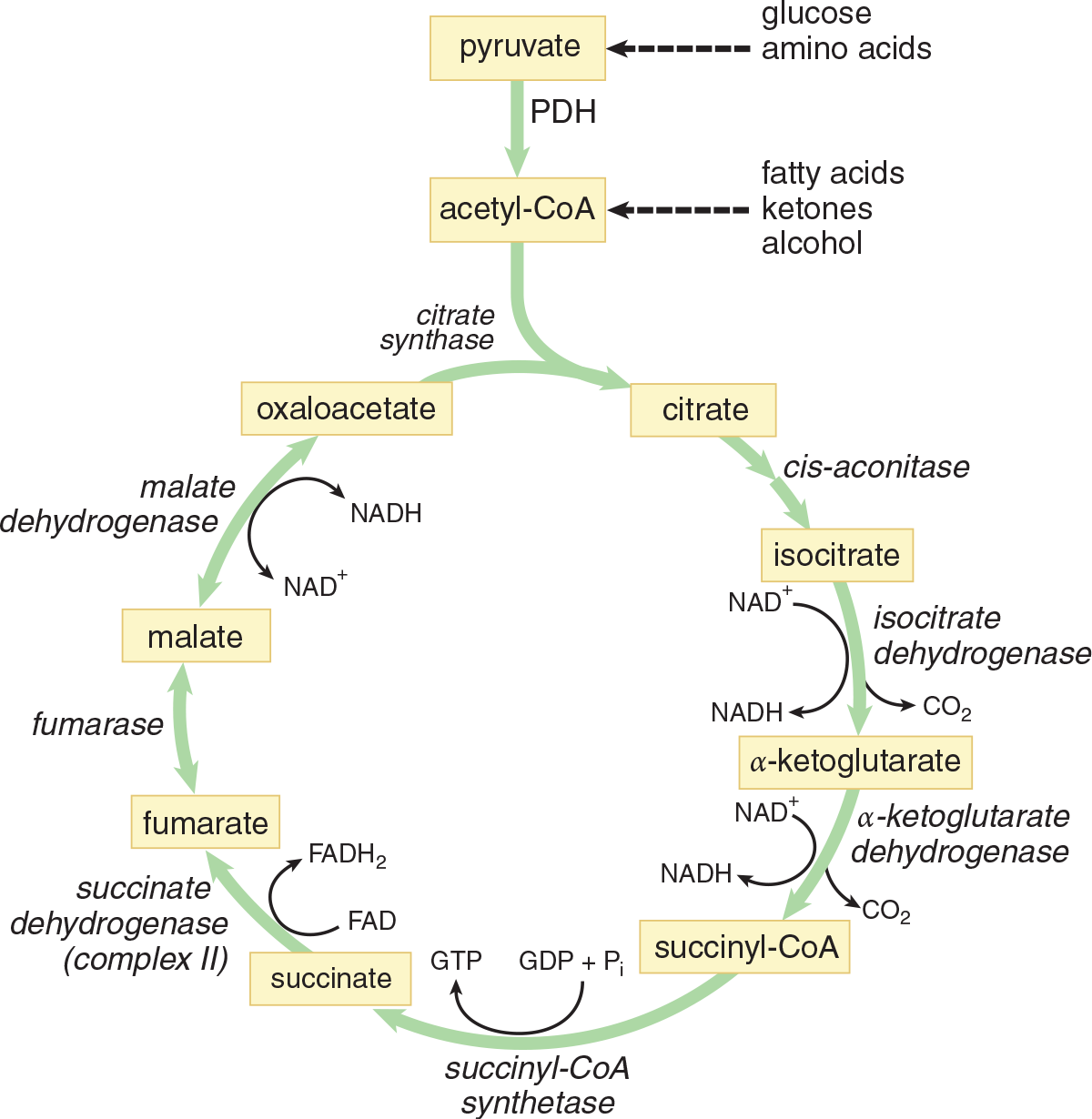
Figure 10.4 The Citric Acid Cycle
Key Reactions
Step 1—Citrate Formation: First, acetyl-CoA and oxaloacetate undergo a condensation reaction to form citryl-CoA, an intermediate. Then, the hydrolysis of citryl-CoA yields citrate and CoA–SH. This reaction is catalyzed by citrate synthase. As discussed in Chapter 2 of MCAT Biochemistry Review, synthases are enzymes that form new covalent bonds without needing significant energy. This second part of this step energetically favors the formation of citrate and helps the cycle revolve in the forward direction. This reaction can be seen in Figure 10.5.
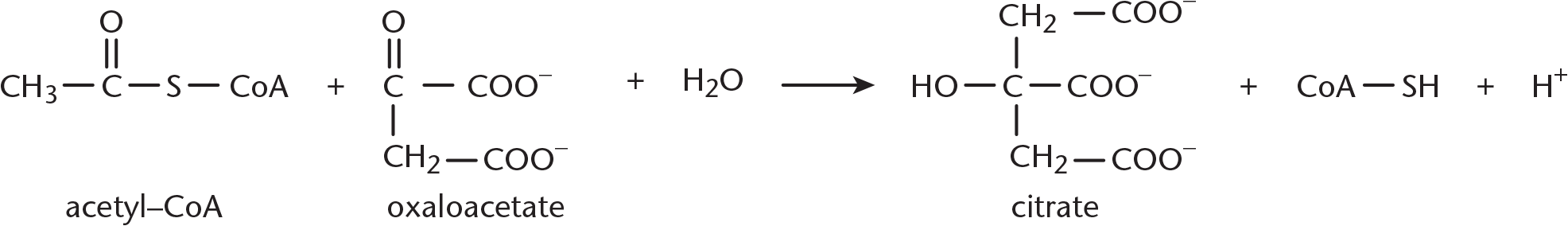
Figure 10.5 Citrate Formation
Step 2—Citrate Isomerized to Isocitrate: Achiral citrate is isomerized to one of four possible isomers of isocitrate. First, citrate binds at three points to the enzyme aconitase. Then water is lost from citrate, yielding cis-aconitate. Finally, water is added back to form isocitrate. The enzyme is a metalloprotein that requires Fe2+. In Figure 10.6, you can see that this results in a switching of a hydrogen and a hydroxyl group. Overall, this step is necessary to facilitate the subsequent oxidative decarboxylation.
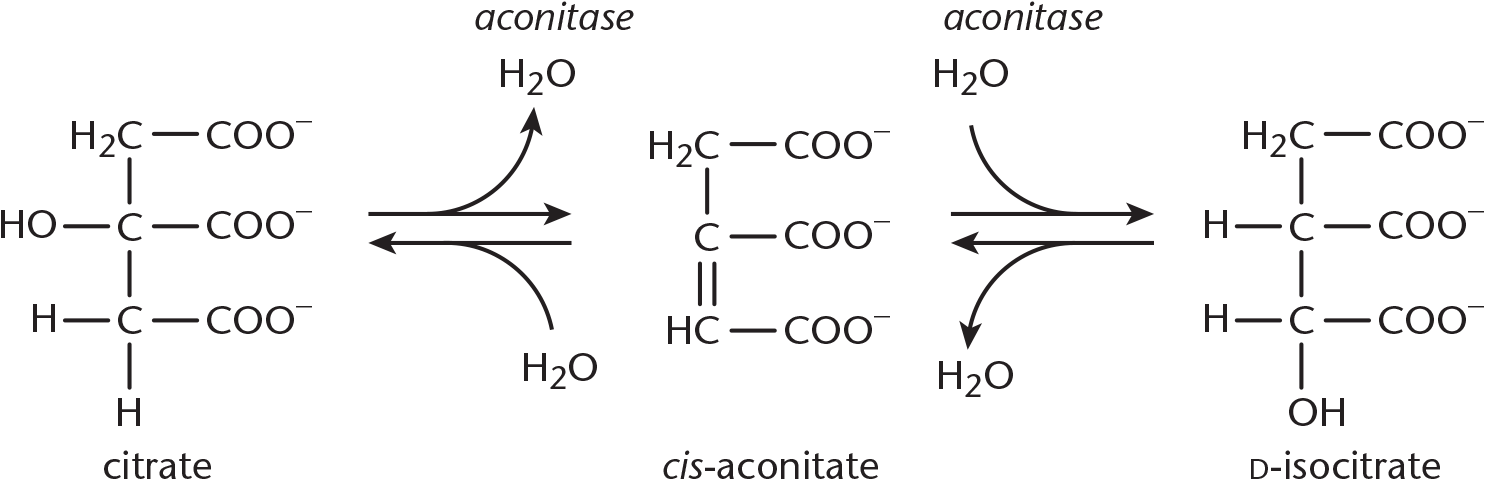
Figure 10.6 Citrate Isomerized to Isocitrate
**Step 3—α-Ketoglutarate and CO**2 Formation: Isocitrate is first oxidized to oxalosuccinate by isocitrate dehydrogenase. Then oxalosuccinate is decarboxylated to produce *α**-ketoglutarate* and CO2, as shown in Figure 10.7. This is a very important step to know for Test Day because isocitrate dehydrogenase is the rate-limiting enzyme of the citric acid cycle. The first of the two carbons from the cycle is lost here. This is also the first NADH produced from intermediates in the cycle.
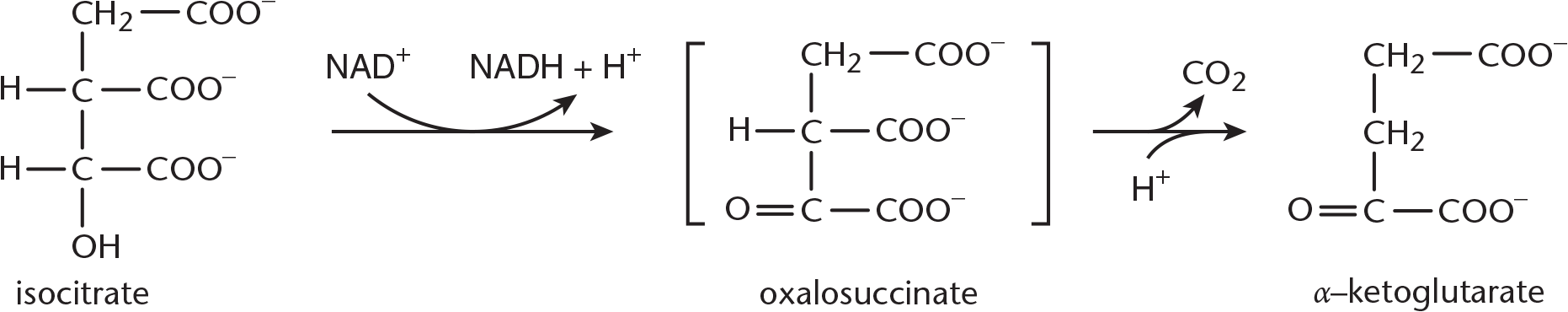
Figure 10.7 α-Ketoglutarate and CO2 Formation
Step 4—Succinyl-CoA and CO2 Formation: These reactions are carried out by the *α**-ketoglutarate dehydrogenase complex, which is similar in mechanism, cofactors, and coenzymes to the pyruvate dehydrogenase (PDH) complex. In the formation of succinyl-CoA,α*-ketoglutarate and CoA come together and produce a molecule of carbon dioxide, as shown in Figure 10.8. This carbon dioxide represents the second and last carbon lost from the cycle. Reducing NAD+ produces another NADH.
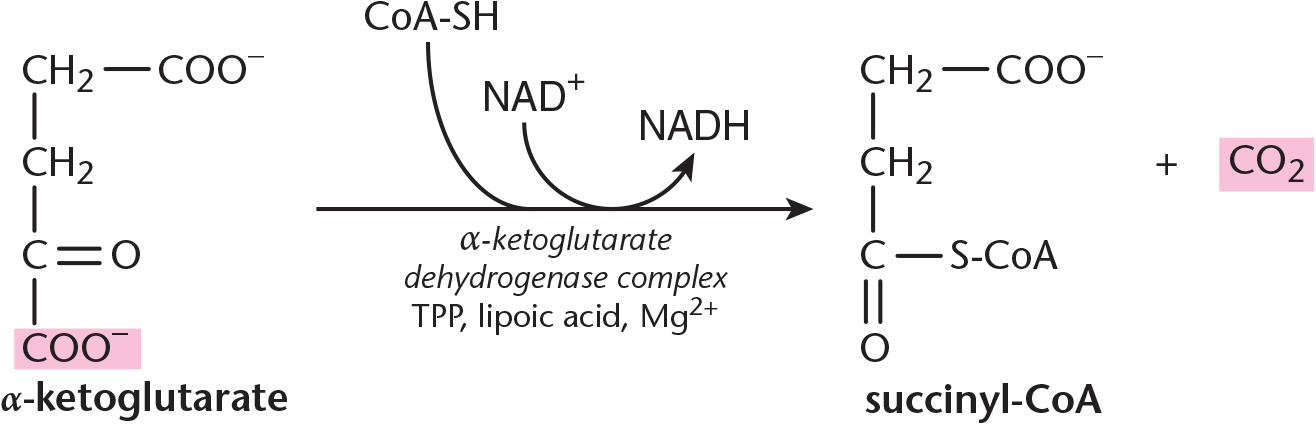
Figure 10.8 Succinyl-CoA and CO2 Formation
KEY CONCEPT
Dehydrogenases are a subtype of oxidoreductases (enzymes that catalyze an oxidation–reduction reaction). Dehydrogenases transfer a hydride ion (H–) to an electron acceptor, usually NAD+ or FAD. Therefore, whenever you see dehydrogenase in aerobic metabolism, be on the lookout for a high-energy electron carrier being formed!
Step 5—Succinate Formation: Hydrolysis of the thioester bond on succinyl-CoA yields succinate and CoA–SH, and is coupled to the phosphorylation of GDP to GTP. This reaction is catalyzed by succinyl-CoA synthetase, as shown in Figure 10.9. Synthetases, unlike synthases, create new covalent bonds with energy input. Recall the earlier discussion about thioester bonds with regard to acetyl-CoA: they’re unique in that their hydrolysis is accompanied by a significant release of energy. Therefore, phosphorylation of GDP to GTP is driven by the energy released by thioester hydrolysis. Once GTP is formed, an enzyme called nucleosidediphosphate kinase catalyzes phosphate transfer from GTP to ADP, thus producing ATP. Note that this is the only time in the entire citric acid cycle that ATP is produced directly; ATP production occurs predominantly within the electron transport chain.
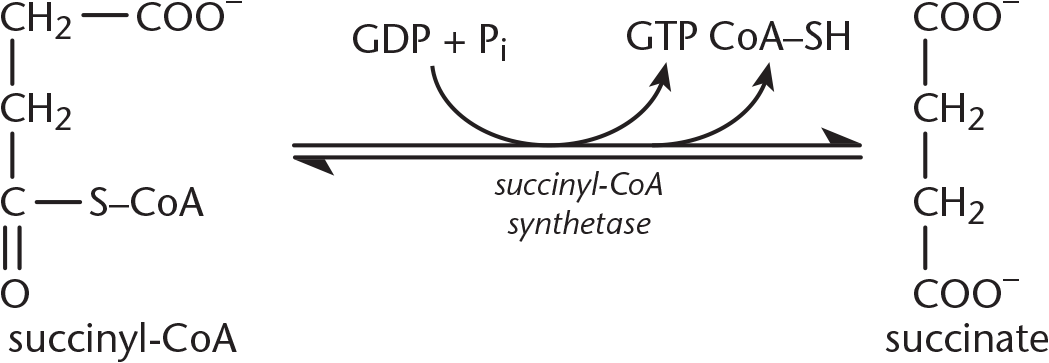
Figure 10.9 Succinate Formation
KEY CONCEPT
Citrate synthase doesn’t require energy input in order to form covalent bonds, but succinyl-CoA synthetase certainly does. Pay careful attention to enzyme names: little things can add up to careless mistakes on Test Day otherwise!
Step 6—Fumarate Formation: This is the only step of the citric acid cycle that doesn’t take place in the mitochondrial matrix; instead, it occurs on the inner membrane. Let’s look at why: succinate undergoes oxidation to yield fumarate. This reaction is catalyzed by succinate dehydrogenase. Succinate dehydrogenase is considered a flavoprotein because it is covalently bonded to FAD, the electron acceptor in this reaction. This enzyme is an integral protein on the inner mitochondrial membrane. As succinate is oxidized to fumarate, FAD is reduced to FADH2. Each molecule of FADH2 then passes the electrons it carries to the electron transport chain, which eventually leads to the production of 1.5 ATP (unlike NADH, which will give rise to 2.5 ATP). FAD is the electron acceptor in this reaction because the reducing power of succinate is not great enough to reduce NAD+.
Step 7—Malate Formation: The enzyme fumarase catalyzes the hydrolysis of the alkene bond in fumarate, thereby giving rise to malate. Although two enantiomeric forms are possible, only L-malate forms in this reaction.
MNEMONIC
Substrates of Citric Acid Cycle: Please, Can I Keep Selling Seashells For Money, Officer?
- Pyruvate
- Citrate
- Isocitrate
- α-Ketoglutarate
- Succinyl-CoA
- Succinate
- Fumarate
- Malate
- Oxaloacetate
Step 8—Oxaloacetate Formed Anew: The enzyme malate dehydrogenase catalyzes the oxidation of malate to oxaloacetate. A third and final molecule of NAD+ is reduced to NADH. The newly formed oxaloacetate is ready to take part in another turn of the citric acid cycle, and we’ve gained all of the high-energy electron carriers possible from one turn of the cycle. The last steps of the citric acid cycle—from succinate to oxaloacetate—are shown in Figure 10.10.
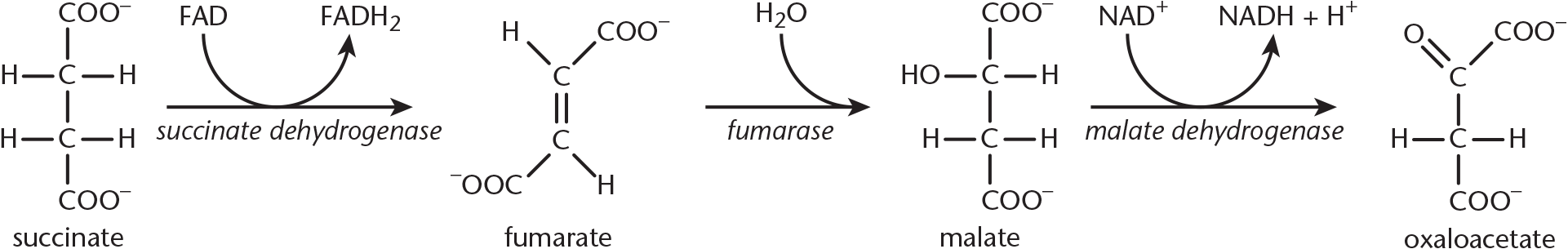
Figure 10.10 The Final Steps of the Citric Acid Cycle
Net Results and ATP Yield
Now let’s take a step back and see what our net yield is from the steps we just took. Starting with the pyruvate dehydrogenase complex, recall that the products of this reaction include one acetyl-CoA and one NADH. In the citric acid cycle, steps 3, 4, and 8 each produce one NADH, while step 6 forms one FADH2. Step 5 yields one GTP, which can be converted to ATP. Two carbons leave the cycle in the form of CO2. Each NADH can be converted to approximately 2.5 ATP, while each FADH2 molecule can yield about 1.5 ATP. The total amount of chemical energy harvested per pyruvate is listed below.
Pyruvate Dehydrogenase Complex:
pyruvate + CoA–SH + NAD+ → acetyl-CoA + NADH + CO2 + H+
Citric Acid Cycle:
acetyl-CoA + 3 NAD+ + FAD + GDP + Pi + 2 H2O → 2 CO2 + CoA–SH + 3 NADH + 3 H+ + FADH2 + GTP
ATP Production:
- 4 NADH → 10 ATP (2.5 ATP per NADH)
- 1 FADH2 → 1.5 ATP (1.5 ATP per FADH2)
- 1 GTP → 1 ATP
- Total: 12.5 ATP per pyruvate = 25 ATP per glucose
Glycolysis yields two ATP and two NADH, providing another seven molecules of ATP; thus, the net yield of ATP for one glucose molecule from glycolysis through oxidative phosphorylation is 30–32 ATP. Note that the efficiency of glycolysis varies slightly from cell to cell, so there is a range of ATP yield from one molecule of glucose.
Regulation
Let’s say it’s Test Day, and you see the following question: Which of the following is an inhibitor of isocitrate dehydrogenase? Before you start to panic, take a step back and use critical thinking. Where have we heard of isocitrate dehydrogenase—or merely isocitrate—before? The Krebs cycle. By knowing this, you can already make a fair attempt at such a question. Because energy (ATP) and energy carriers (NADH and FADH2) are products of this process, it makes sense that these molecules would have a negative feedback effect on the citric acid cycle. Always consider the big picture when faced with questions like these. For now, we’ll outline how regulation occurs throughout the citric acid cycle; look for the recurring theme that energy products inhibit energy production processes.
Pyruvate Dehydrogenase Complex Regulation
Even upstream from its actual starting point, the citric acid cycle can be regulated. The mechanism by which this happens is phosphorylation of PDH, which is facilitated by the enzyme pyruvate dehydrogenase kinase. Thus, whenever levels of ATP rise, phosphorylating PDH inhibits acetyl-CoA production. Conversely, the pyruvate dehydrogenase complex is reactivated by the enzyme pyruvate dehydrogenase phosphatase in response to high levels of ADP. By removing a phosphate from PDH, pyruvate dehydrogenase phosphatase is able to reactivate acetyl-CoA production. Acetyl-CoA also has a negative feedback effect on its own production. When using alternative fuel sources such as fats, the acetyl-CoA production is sufficient to make it redundant to continue producing acetyl-CoA from carbohydrate metabolism—that’s part of why eating a high-fat meal fills you up so quickly! ATP and NADH, as markers of the cell being satisfied energetically, also inhibit PDH.
Control Points of the Citric Acid Cycle
There are three essential checkpoints that regulate the citric acid cycle from within, and allosteric activators and inhibitors regulate all of them. The details of these mechanisms are outlined below and in Figure 10.11.
- Citrate synthase: ATP and NADH function as allosteric inhibitors of citrate synthase, which makes sense because both are products (indirect and direct, respectively) of the enzyme. Citrate also allosterically inhibits citrate synthase directly, as does succinyl-CoA.
- Isocitrate dehydrogenase: As we discussed in the beginning of this section, this enzyme that catalyzes the citric acid cycle is likely to be inhibited by energy products: ATP and NADH. Conversely, ADP and NAD+ function as allosteric activators for the enzyme and enhance its affinity for substrates.
- ***α*-Ketoglutarate dehydrogenase complex**: Once again, the reaction products of succinyl-CoA and NADH function as inhibitors of this enzyme complex. ATP is also inhibitory and slows the rate of the cycle when the cell has high levels of ATP. The complex is stimulated by ADP and calcium ions.
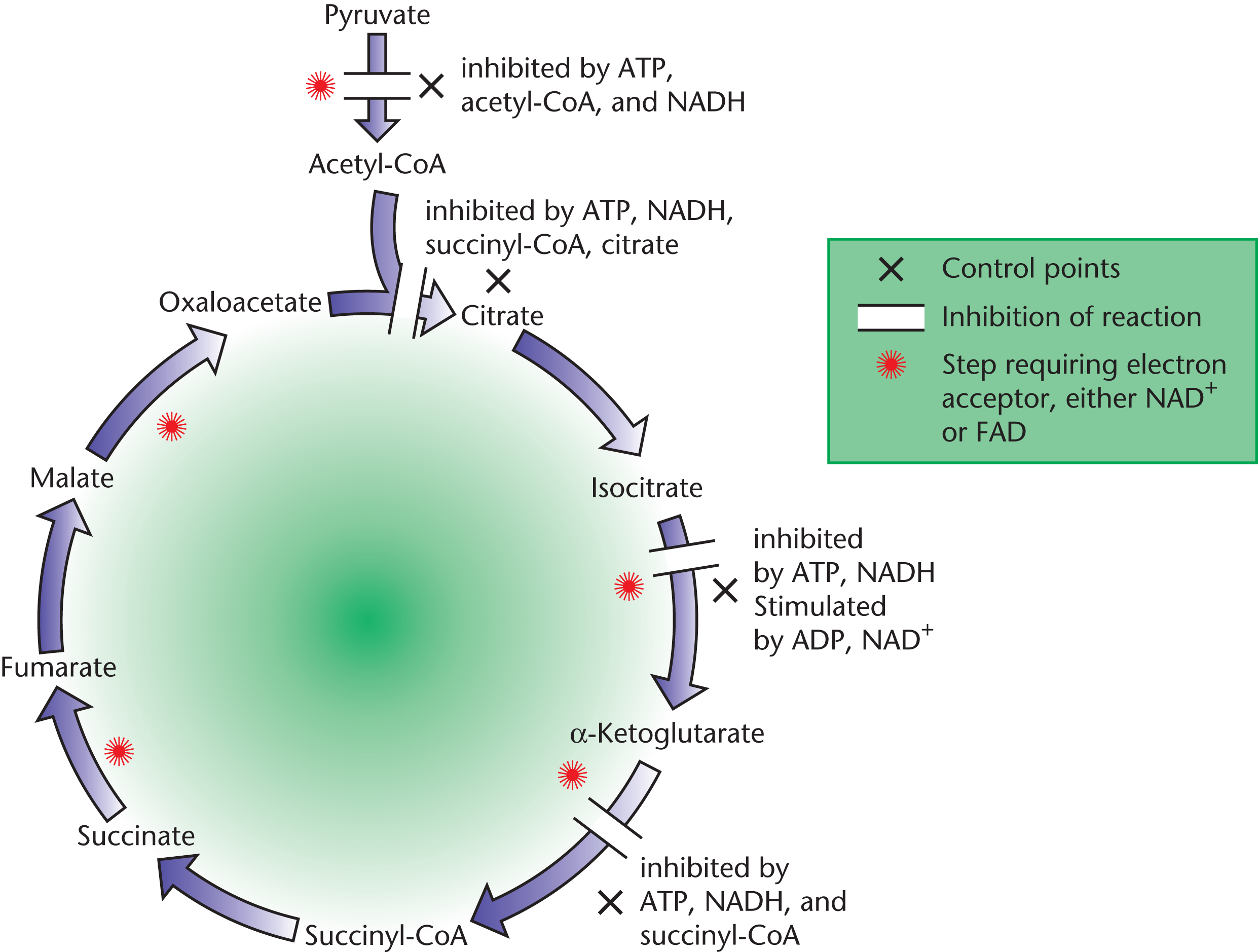
Figure 10.11 Checkpoints and Regulation of the Citric Acid Cycle
Note that high levels of ATP and NADH inhibit the citric acid cycle, while high levels of ADP and NAD+ promote it. This isn’t a coincidence! When energy is being consumed in large amounts, more and more ATP is converted to ADP and NADH is converted to NAD+. It is therefore the ATP/ADP ratio and NADH/NAD+ ratio that help determine whether the citric acid cycle will be inhibited or activated. During a metabolically active state, ADP and NAD+ levels should rise as ATP and NADH levels decline, thus inducing activation at all the various checkpoints described above, replacing the energy used up by active tissues.
MCAT CONCEPT CHECK 10.2:
Before you move on, assess your understanding of the material with these questions.
- What is the purpose of all the reactions that collectively make up the citric acid cycle?
_________________________________
- What enzyme catalyzes the rate-limiting step of the citric acid cycle?
__________________________________
- What are the three main sites of regulation within the citric acid cycle? What molecules inhibit and activate the three main checkpoints?
Checkpoints Inhibitors Activators
10.3 The Electron Transport Chain
LEARNING OBJECTIVES
After Chapter 10.3, you will be able to:
- Connect the reactions of the electron transport chain to the generation of ATP
- Distinguish between the two shuttle mechanisms for NADH transport into the mitochondrion
- Recall the inputs, outputs, and major components of the four complexes of the electron transport chain:
The electron transport chain is the final common pathway that utilizes the harvested electrons from different fuels in the body. It is important to make the distinction that it is not the flow of electrons but the proton gradient it generates that ultimately produces ATP. Aerobic metabolism is the most efficient way of generating energy in living systems, and the mitochondrion is the reason why. In eukaryotes, the aerobic components of respiration are executed in mitochondria, while anaerobic processes such as glycolysis and fermentation occur in the cytosol. Looking at Figure 10.12, notice how the components of the mitochondria are critical in the harvesting of energy. The citric acid cycle takes place in the mitochondrial matrix. The assemblies needed to complete oxidative phosphorylation are housed adjacent to the matrix in the inner membrane of the mitochondria. The inner mitochondrial membrane is assembled into folds called cristae, which maximize surface area. It is the inner mitochondrial membrane that will be essential for generating ATP using the proton-motive force, an electrochemical proton gradient generated by the complexes of the electron transport chain.
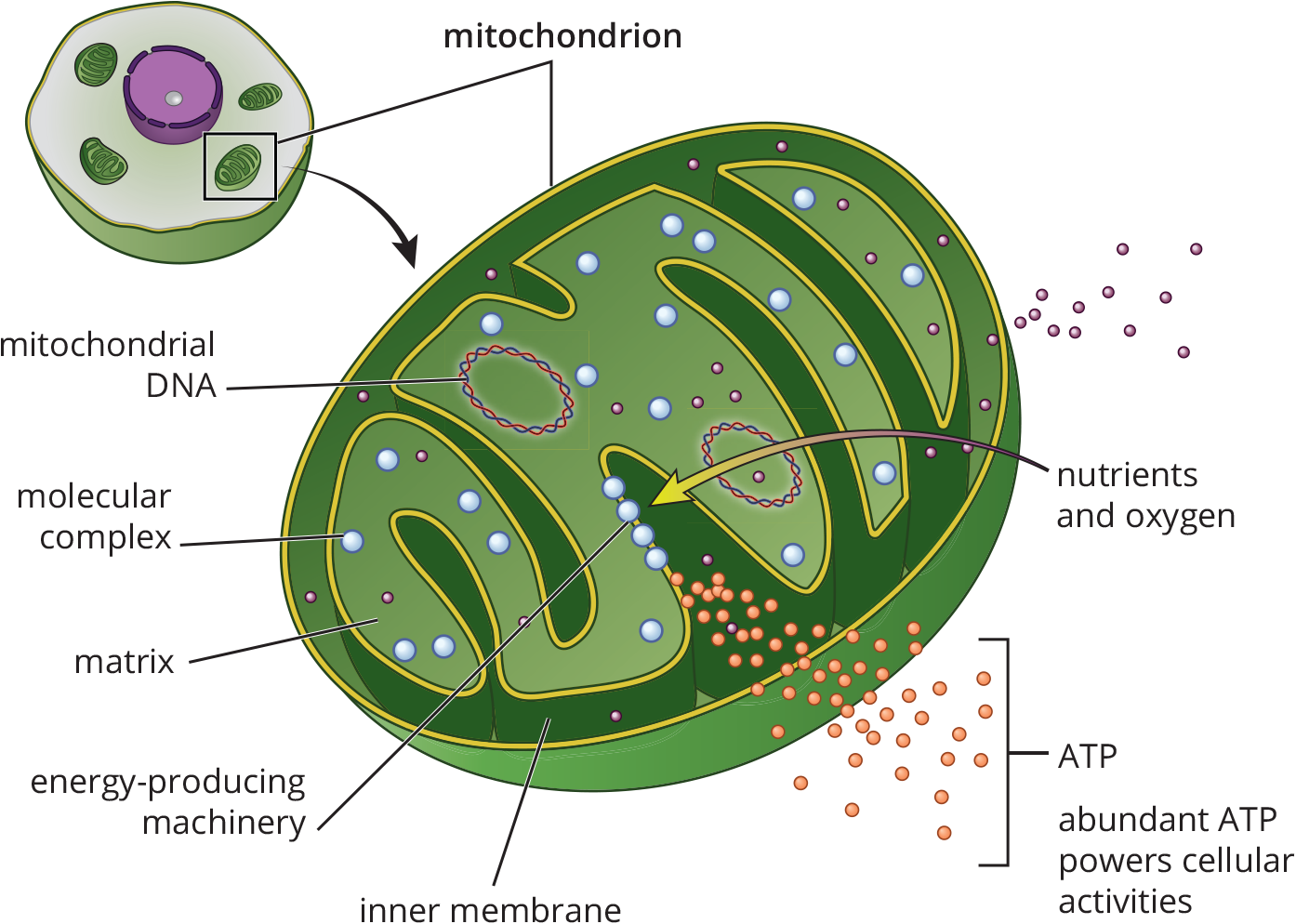
Figure 10.12 Mitochondrial Structure
The final step in aerobic respiration is actually two steps: electron transport along the inner mitochondrial membrane and the generation of ATP via ADP phosphorylation. While these two processes are actually separate entities, they are very much coupled, so explaining these steps together makes a great deal of sense. The electron-rich molecules NADH and FADH2 are formed as byproducts at earlier steps in respiration. They transfer their electrons to carrier proteins located along the inner mitochondrial membrane. Finally, these electrons are given to oxygen in the form of hydride ions (H–) and water is formed. While this is happening, energy released from transporting electrons facilitates proton transport at three specific locations in the chain. Protons are moved from the mitochondrial matrix into the intermembrane space of the mitochondria, thereby creating a greater concentration gradient of hydrogen ions that can be used to drive ATP production.
Electron Flow and Complexes
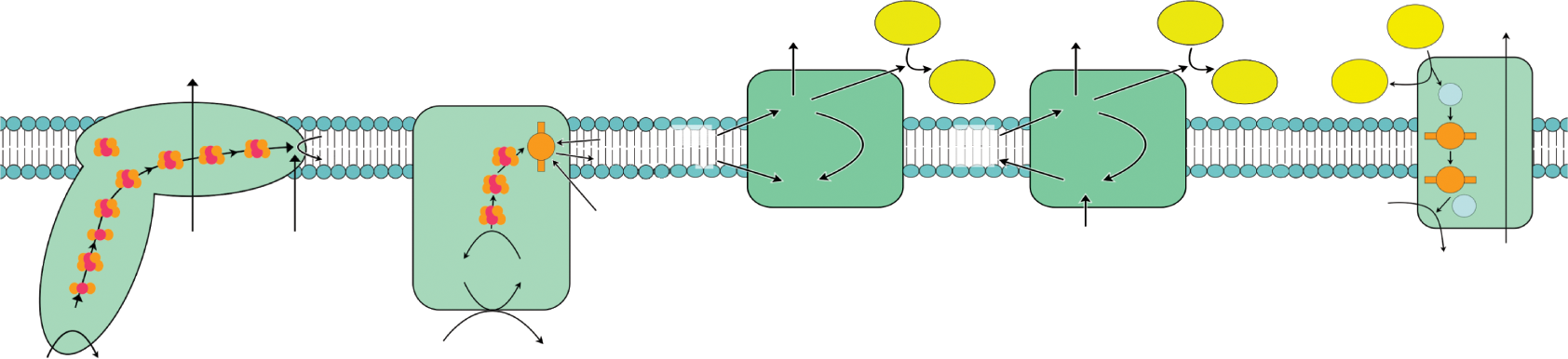
The formation of ATP is endergonic and electron transport is an exergonic pathway. By coupling these reactions, the energy yielded by one reaction can fuel the other. In order for energy to be harnessed via electron transport reactions, the proteins along the inner membrane must transfer the electrons donated by NADH and FADH2 in a specific order and direction. The physical property that determines the direction of electron flow is reduction potential. Recall from Chapter 12 of MCAT General Chemistry Review that if you pair two molecules with different reduction potentials, the molecule with the higher potential will be reduced, while the other molecule will become oxidized. The electron transport chain is therefore nothing more than a series of oxidations and reductions that occur via the same mechanism. NADH is a good electron donor, and the high reduction potential of oxygen makes it a great final acceptor in the electron transport chain. The organizational structure of the membrane-bound complexes that make up the transport chain is diagrammed in Figure 10.13 and further detailed below.
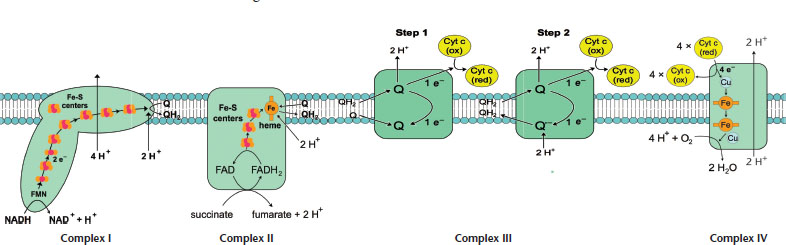
Figure 10.13. Respiratory Complexes on the Inner Mitochondrial Membrane Steps 1 and 2 of Complex III are drawn as two separate steps here for clarity; however, the same CoQH2-cytochrome c oxidoreductase complex is used for both steps.
-
Complex I (NADH-CoQ oxidoreductase): The transfer of electrons from NADH to coenzyme Q (CoQ) is catalyzed in this first complex. This complex has over 20 subunits, but the two highlighted here include a protein that has an iron–sulfur cluster and a flavoprotein that oxidizes NADH. The flavoprotein has a coenzyme called flavin mononucleotide (FMN) covalently bonded to it. FMN is quite similar in structure to FAD, flavin adenine dinucleotide. The first step in the reaction involves NADH transferring its electrons over to FMN, thereby becoming oxidized to NAD+ as FMN is reduced to FMNH2. Next, the flavoprotein becomes reoxidized while the iron–sulfur subunit is reduced. Finally, the reduced iron–sulfur subunit donates the electrons it received from FMNH2 to coenzyme Q (also called ubiquinone). Coenzyme Q becomes CoQH2. This first complex is one of three sites where proton pumping occurs, as four protons are moved to the intermembrane space.
- NADH + H+ + FMN → NAD+ + FMNH2
- FMNH2 + 2 Fe–Soxidized → FMN + 2 Fe–Sreduced + 2 H+
- 2 Fe–Sreduced + CoQ + 2 H+ → 2 Fe–Soxidized + CoQH2
The net effect is passing high-energy electrons from NADH to CoQ to form CoQH2:
- NADH + H+ + CoQ → NAD+ + CoQH2
-
Complex II (Succinate-CoQ oxidoreductase): Just like Complex I, Complex II transfers electrons to coenzyme Q. While Complex I received electrons from NADH, Complex II actually receives electrons from succinate. Remember that succinate is a citric acid cycle intermediate, and that it is oxidized to fumarate upon interacting with FAD. FAD is covalently bonded to Complex II, and once succinate is oxidized, it’s converted to FADH2. After this, FADH2 gets reoxidized to FAD as it reduces an iron–sulfur protein. The final step reoxidizes the iron–sulfur protein as coenzyme Q is reduced. Because succinate dehydrogenase was responsible for oxidizing succinate to fumarate in the citric acid cycle, it makes sense that succinate dehydrogenase is also a part of Complex II. It should be noted that no hydrogen pumping occurs here to contribute to the proton gradient.
- succinate + FAD → fumarate + FADH2
- FADH2 + Fe–Soxidized → FAD + Fe–Sreduced
- Fe–Sreduced + CoQ + 2 H+ → Fe–Soxidized + CoQH2
REAL WORLD
Unlike iron in cytochromes, iron in the heme group of hemoglobin always remains as Fe2+ during the transport of oxygen through the bloodstream under normal conditions.
The net effect is passing high-energy electrons from succinate to CoQ to form CoQH2:
- succinate + CoQ + 2 H+ → fumarate + CoQH2
-
Complex III (CoQH2***-cytochromecoxidoreductase***): Also called cytochrome reductase, this complex facilitates the transfer of electrons from coenzyme Q to cytochrome c in a few steps. Though Complex III is drawn as two separate complexes in Figure 10.13 to illustrate the sequential reactions that occur within the complex, both of these steps are occurring within the same complex, using the same coenzyme Q. The overall reaction is written below. The following steps involve the oxidation and reduction of cytochromes: proteins with heme groups in which iron is reduced to Fe2+ and reoxidized to Fe3+.
CoQ + 2 cytochrome c [with Fe2+] + 2 H+
- CoQH2 + 2 cytochrome c [with Fe3+] →
In the transfer of electrons from iron, only one electron is transferred per reaction, but because coenzyme Q has two electrons to transfer, two cytochrome c molecules will be needed. Complex III’s main contribution to the proton-motive force is via the Q cycle. In the Q cycle, two electrons are shuttled from a molecule of ubiquinol (CoQH2) near the intermembrane space to a molecule of ubiquinone (CoQ) near the mitochondrial matrix. Another two electrons are attached to heme moieties, reducing two molecules of cytochrome c. A carrier containing iron and sulfur assists this process. In shuttling these electrons, four protons are also displaced to the intermembrane space; therefore, the Q cycle continues to increase the gradient of the proton-motive force across the inner mitochondrial membrane.
BRIDGE
Ubiquinone can be created from its corresponding phenol by oxidation and represents an example of a quinone (2,5-cyclohexadiene-1,4-diones). These fascinating compounds are explored in Chapter 5 of MCAT Organic Chemistry Review.
KEY CONCEPT
Both coenzyme Q and cytochrome c aren’t technically part of the complexes we’re describing. However, because both are able to move freely in the inner mitochondrial membrane, this degree of mobility allows these carriers to transfer electrons by physically interacting with the next component of the transport chain.
-
Complex IV (***cytochromecoxidase***): This complex facilitates the culminating step of the electron transport chain: transfer of electrons from cytochrome c to oxygen, the final electron acceptor. This complex includes subunits of cytochrome a, cytochrome a3, and Cu2+ ions. Together, cytochromes a and a3 make up cytochrome oxidase. Through a series of redox reactions, cytochrome oxidase gets oxidized as oxygen, becomes reduced, and forms water. This is the final location on the transport chain where proton pumping occurs, as two protons are moved across the membrane. The role proton pumping plays in ATP synthesis is an essential one that we will describe in detail next. The overall reaction is:
4 cytochrome c [with Fe3+] + 2 H2O
- 4 cytochrome c [with Fe2+] + 4 H+ + O2 →
KEY CONCEPT
Cyanide, mentioned in the introduction to this chapter, is an inhibitor of cytochrome subunits a and a3. The cyanide anion is able to attach to the iron group and prevent the transfer of electrons. Tissues that rely heavily on aerobic respiration such as the heart and the central nervous system can be greatly impacted.
BIOCHEMISTRY GUIDED EXAMPLE WITH EXPERT THINKING
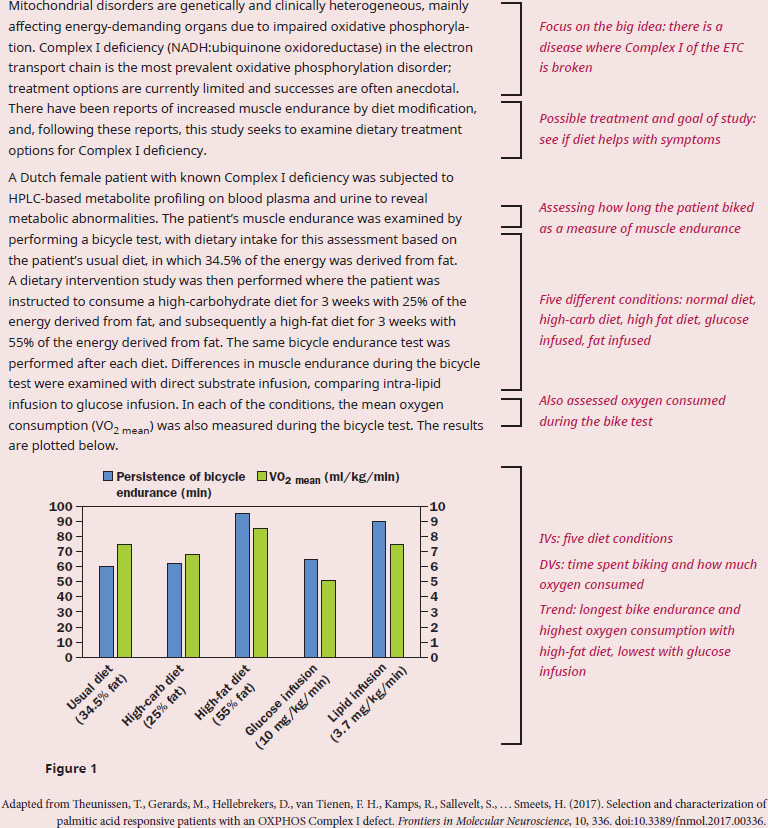
According to the data, what is the best treatment option for the patient's Complex I deficiency and why?
The question stem starts with “according to the data”, so we need to ensure that we have a firm understanding of the passage presenting the context for that data. The experimental context introduced in paragraph 1 is a malfunction of Complex I in the ETC, and the hypothesis centers on dietary treatment that can help increase muscle endurance. We can recall from our content background that the ETC functions to transfer electrons from NADH and FADH2 to electron acceptor, thereby creating a proton gradient to fuel the production of ATP. The coupling of the ETC to ATP synthase is termed oxidative phosphorylation and requires the availability of oxygen, as oxygen is the final electron acceptor in the ETC. Since ATP is required for muscle contraction, and oxygen is an essential component of the ETC, both high endurance and high oxygen consumption would indicate a high level of aerobic respiration.
The question asks us about the mechanism behind the best treatment option, so we must now analyze the data to see which treatment was best. According to the graph, the high-fat diet allowed the patient to endure the bicycle test the longest, and have the highest level of oxygen consumption. Therefore, the best treatment option is a diet with a high proportion of fat. This conclusion is supported by the fact that lipid infusion also gave higher values compared to glucose infusion, though not as much as the high-fat diet. The next aspect of the question to address is to examine the “why?”—why does ingesting high-fat work this way? Again, using prior knowledge, recall that metabolism of fats generates acetyl-CoA, and acetyl-CoA can then enter the citric acid cycle to produce NADH and FADH2. Complex I receives electrons from NADH, and Complex II receives electrons from FADH2, so in this patient with a Complex I disorder, only the production of FADH2 is fueling aerobic respiration. So, why is there such a large difference between the high-fat and high-carbohydrate diets, especially considering the fact that metabolism of glucose will also eventually generate acetyl-CoA? Glycolysis (break down of glucose into pyruvate) only generates NADH, so the ratio of FADH2/NADH is higher in fat metabolism vs. carbohydrate metabolism, leading to more efficient generation of energy by funneling more electron carriers to Complex II.
In summary, the best treatment for the patient's Complex I deficiency is the high-fat diet, due to increased funneling of electron carriers to Complex II.
The Proton-Motive Force
Let’s take a step back and look at the proton gradient that formed as electrons were passed along the ETC. As [H+] increases in the intermembrane space, two things happen simultaneously: pH drops in the intermembrane space, and the voltage difference between the intermembrane space and matrix increases due to proton pumping. Together, these two changes contribute to what is referred to as an electrochemical gradient: a gradient that has both chemical and electrostatic properties. Because it is based on protons, we often refer to the electrochemical gradient across the inner mitochondrial membrane as the proton-motive force. Any electrochemical gradient stores energy, and it will be the responsibility of ATP synthase to harness this energy to form ATP from ADP and an inorganic phosphate.
NADH Shuttles
As we look at the net ATP yield per glucose, note that a range exists between 30 and 32. This is because efficiency of aerobic respiration varies between cells. This variable efficiency is caused by the fact that cytosolic NADH formed through glycolysis cannot directly cross into the mitochondrial matrix. Because it cannot contribute its electrons to the transport chain directly, it must find alternate means of transportation referred to as shuttle mechanisms. A shuttle mechanism transfers the high-energy electrons of NADH to a carrier that can cross the inner mitochondrial membrane. Depending on which of the two shuttle mechanisms NADH participates in, either 1.5 or 2.5 ATP will end up being produced. Let’s take a look at the two mechanisms:
- Glycerol 3-phosphate shuttle: The cytosol contains one isoform of glycerol-3-phosphate dehydrogenase, which oxidizes cytosolic NADH to NAD+ while forming glycerol 3-phosphate from dihydroxyacetone phosphate (DHAP). On the outer face of the inner mitochondrial membrane, there exists another isoform of glycerol-3-phosphate dehydrogenase that is FAD-dependent. This mitochondrial FAD is the oxidizing agent, and ends up being reduced to FADH2. Once reduced, FADH2 proceeds to transfer its electrons to the ETC via Complex II, thus generating 1.5 ATP for every molecule of cytosolic NADH that participates in this pathway, which is shown in Figure 10.14.
- Malate–aspartate shuttle: Cytosolic oxaloacetate, which cannot pass through the inner mitochondrial membrane, is reduced to malate, which can. This is accomplished by cytosolic malate dehydrogenase. Accompanying this reduction is the oxidation of cytosolic NADH to NAD+. Once malate crosses into the matrix, mitochondrial malate dehydrogenase reverses the reaction to form mitochondrial NADH. Now that NADH is in the matrix, it can pass along its electrons to the ETC via Complex I and generate 2.5 ATP per molecule of NADH. Recycling the malate requires oxidation to oxaloacetate, which can be transaminated to form aspartate. Aspartate crosses into the cytosol, and can be reconverted to oxaloacetate to restart the cycle, as shown in Figure 10.15.
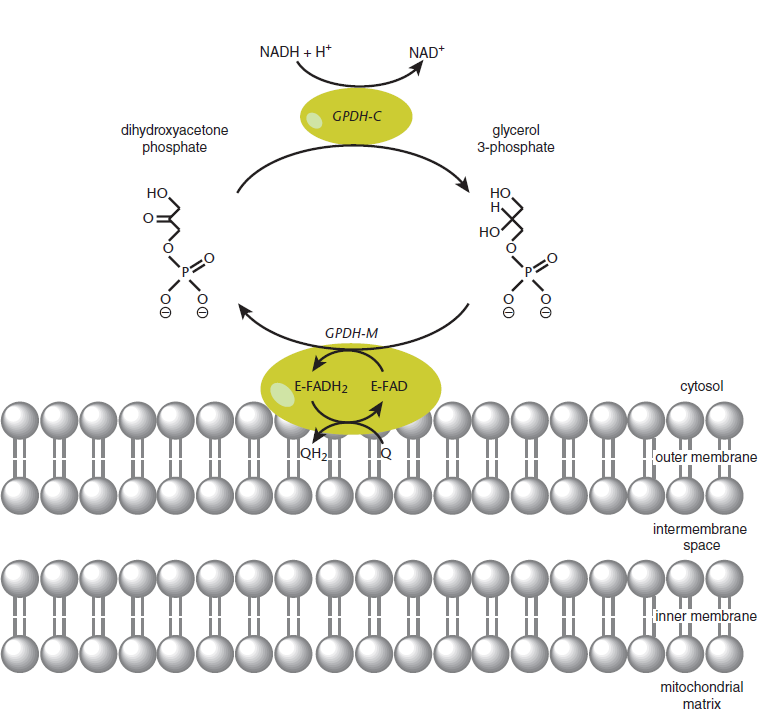
Figure 10.14 Glycerol-3-Phosphate Shuttle
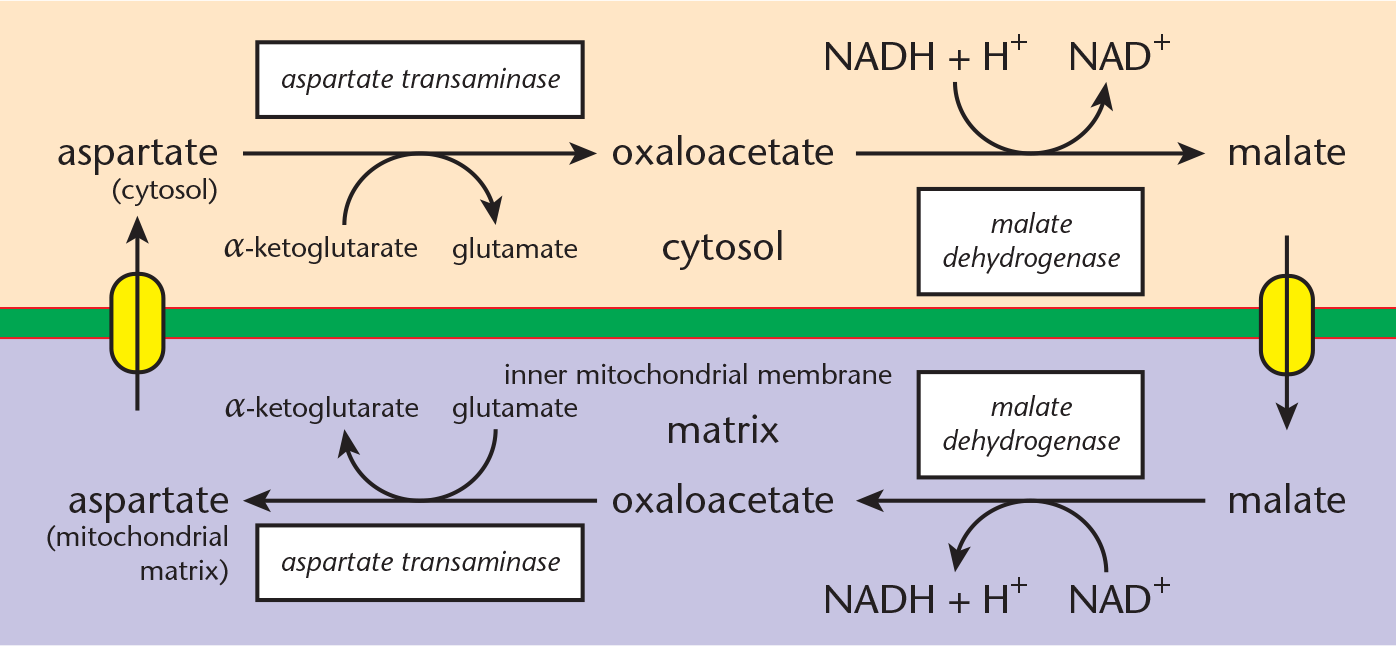
Figure 10.15 Malate–Aspartate Shuttle
BRIDGE
Glycerol 3-phosphate is an important link between lipid metabolism, discussed in Chapter 11 of MCAT Biochemistry Review, and glycolysis, discussed in Chapter 9. Its ability to be converted to DHAP, an intermediate of glycolysis, means that the glycerol of triacylglycerols can be shunted into glycolysis for energy.
MCAT CONCEPT CHECK 10.3:
Before you move on, assess your understanding of the material with these questions.
- Which complex(es) are associated with each of the following? (circle all that apply)
- Pumping a proton into the intermembrane space
I II III IV
- Acquiring electrons from NADH
I II III IV
- Acquiring electrons from FADH2
I II III IV
- Having the highest reduction potential
I II III IV
- What role does the electron transport chain play in the generation of ATP?
_______________________________
- Based on its needs, which of the two shuttle mechanisms is cardiac muscle most likely to utilize? Why?
________________________________
10.4 Oxidative Phosphorylation
LEARNING OBJECTIVES
After Chapter 10.4, you will be able to:
- Compare and contrast the ETC and oxidative phosphorylation
- Explain why the ETC generates more ATP than the direct reduction of oxygen by NADH
We have arrived at the payout site of aerobic respiration: ATP synthesis. Knowing the nuances of ATP synthesis is an absolute must by Test Day. The link between electron transport and ATP synthesis starts with a protein complex called ATP synthase, which spans the entire inner mitochondrial membrane and protrudes into the matrix.
REAL WORLD
A small fraction—only 13 of approximately 100 polypeptides–that are necessary for oxidative phosphorylation are encoded by mitochondrial DNA. The significance of this fact is that mitochondrial DNA has a mutation rate nearly ten times higher than that of nuclear DNA.
Chemiosmotic Coupling
The proton-motive force interacts with the portion of ATP synthase that spans the membrane, which is called the F0 portion. F0 functions as an ion channel, so protons travel through F0 along their gradient back into the matrix. As this happens, a process called chemiosmotic coupling allows the chemical energy of the gradient to be harnessed as a means of phosphorylating ADP, thus forming ATP. In other words, the ETC generates a high concentration of protons in the intermembrane space; the protons then flow through the F0 ion channel of ATP synthase back into the matrix. As this happens, the other portion of ATP synthase, which is called the F1 portion, utilizes the energy released from this electrochemical gradient to phosphorylate ADP to ATP, as demonstrated in Figure 10.16. The specific mechanism by which ADP is actually phosphorylated is still a matter of debate.
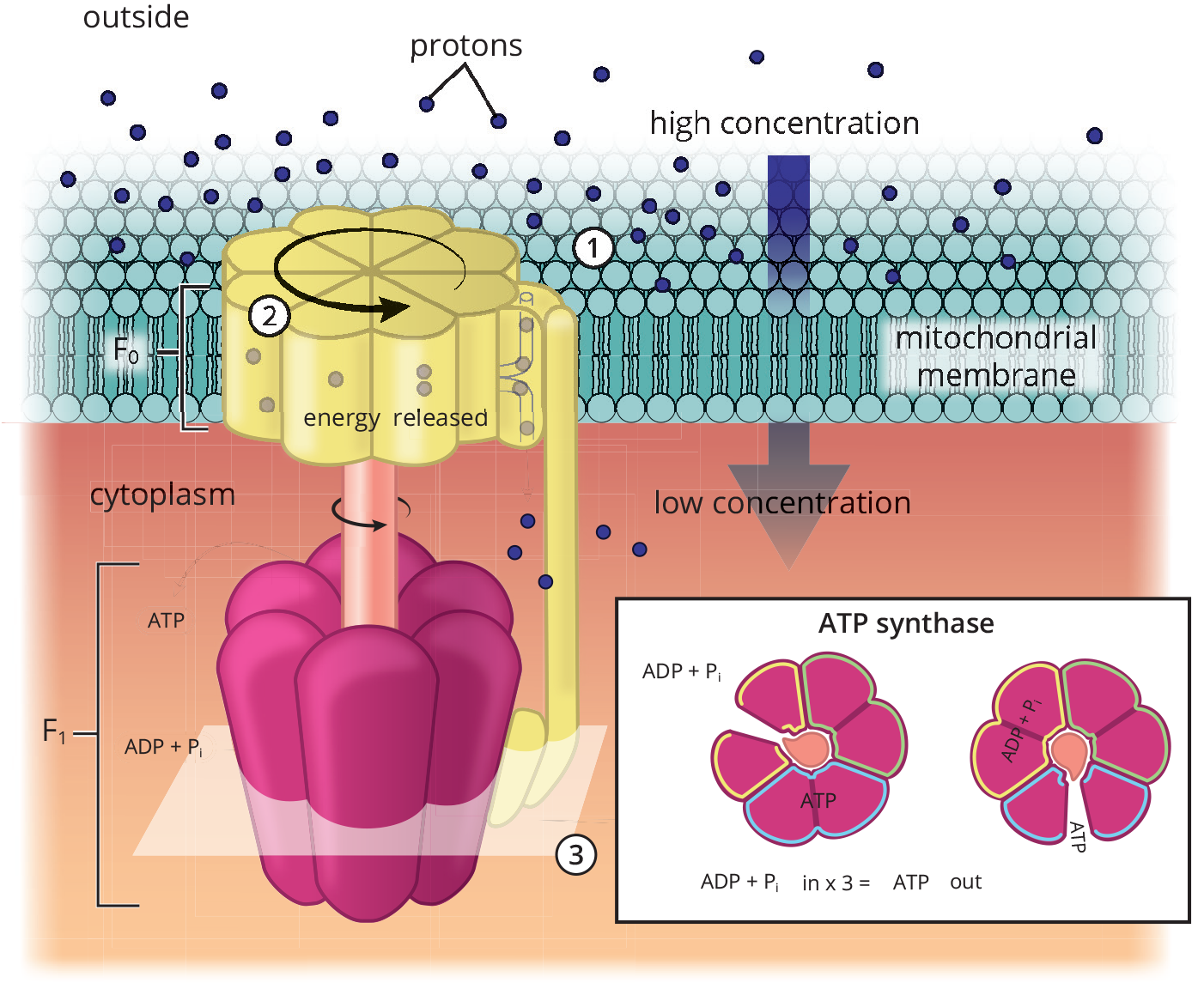
Figure 10.16 ATP Synthase Reaction ATP synthase generates ATP from ADP and inorganic phosphate by allowing high-energy protons to move down the concentration gradient created by the electron transport chain.
Chemiosmotic coupling describes a direct relationship between the proton gradient and ATP synthesis. It is the predominant mechanism accepted in the scientific community when describing oxidative phosphorylation. However, another mechanism called conformational coupling suggests that the relationship between the proton gradient and ATP synthesis is indirect. Instead, ATP is released by the synthase as a result of conformational change caused by the gradient. In this mechanism, the F1 portion of ATP synthase is reminiscent of a turbine, spinning within a stationary compartment to facilitate the harnessing of gradient energy for chemical bonding.
MCAT EXPERTISE
When tackling complex mechanisms such as chemiosmotic coupling on Test Day, it’s easy to make mistakes such as interpreting a pH drop to be a [H+] drop instead of a rise in proton concentration. Always read actively to avoid such mistakes.
So we now know how we generate ATP, but how much energy was required to do so? When the proton-motive force is dissipated through the F0 portion of ATP synthase, the free energy change of the reaction, ΔG°′, is
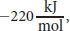
a highly exergonic reaction. This makes sense because phosphorylating ADP to form ATP is an endergonic process. So, by coupling these reactions, the energy harnessed from one reaction can drive another.
REAL WORLD
Uncouplers are compounds that prevent ATP synthesis without affecting the ETC, thus greatly decreasing the efficiency of the ETC/oxidative phosphorylation pathway. Because ADP builds up and ATP synthesis decreases, the body responds to this perceived lack of energy by increasing O2 consumption and NADH oxidation. The energy produced from the transport of electrons is released as heat. An example would be the fever experienced with toxic levels of salicylates, including aspirin.
Regulation
Because the citric acid cycle provides the electron-rich molecules that feed into the ETC, it should come as no surprise that the rates of oxidative phosphorylation and the citric acid cycle are closely coordinated. Always think of O2 and ADP as the key regulators of oxidative phosphorylation. If O2 is limited, the rate of oxidative phosphorylation decreases, and the concentrations of NADH and FADH2 increase. The accumulation of NADH, in turn, inhibits the citric acid cycle. The coordinated regulation of these pathways is known as respiratory control. In the presence of adequate O2, the rate of oxidative phosphorylation is dependent on the availability of ADP. The concentrations of ADP and ATP are reciprocally related; an accumulation of ADP is accompanied by a decrease in ATP and the amount of energy available to the cell. Therefore, ADP accumulation signals the need for ATP synthesis. ADP allosterically activates isocitrate dehydrogenase, thereby increasing the rate of the citric acid cycle and the production of NADH and FADH2. The elevated levels of these reduced coenzymes, in turn, increase the rate of electron transport and ATP synthesis.
MCAT CONCEPT CHECK 10.4:
Before you move on, assess your understanding of the material with these questions.
- What is the difference between the ETC and oxidative phosphorylation? What links the two?
__________________________________
- The ΔG° of NADH reducing oxygen directly is significantly greater than any individual step along the electron transport chain. If this is the case, why does transferring electrons along the ETC generate more ATP than direct reduction of oxygen by NADH?
__________________________________
Conclusion
Both topics discussed in this chapter—the citric acid cycle and oxidative phosphorylation—take place in the mitochondria. In the mitochondrial matrix, the citric acid cycle completely oxidizes acetyl-CoA to carbon dioxide. While this happens, energy is conserved via reduction reactions, forming high-energy electron carriers such as FADH2 and NADH. ATP is also indirectly formed via GTP synthesis. These electron-rich carriers then transfer their electrons to the electron transport chain, which is located along the inner mitochondrial membrane. A series of oxidation–reduction reactions occurs in specific complexes until oxygen, the final electron acceptor, gets reduced and forms H2O. This electrical pathway generates an electrochemical proton gradient that is harnessed by ATP synthase to generate ATP. The link between these two processes is highlighted by the fact that control of the citric acid cycle is NADH-dependent. When NADH accumulates, isocitrate dehydrogenase inhibition occurs, thus stopping both the citric acid cycle and electron transport chain.
It is worth noting that, while glycolysis is a major source of acetyl-CoA for the citric acid cycle, fatty acids also serve as an important source. In the next chapter, we turn our attention to the metabolism of two other types of biomolecules: lipids and amino acids.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Acetyl-CoA
- Acetyl-CoA contains a high-energy thioester bond that can be used to drive other reactions when hydrolysis occurs.
- It can be formed from pyruvate via pyruvate dehydrogenase complex, a five-enzyme complex in the mitochondrial matrix that forms—and is also inhibited by—acetyl-CoA and NADH.
- Pyruvate dehydrogenase (PDH) oxidizes pyruvate, creating CO2; it requires thiamine pyrophosphate (vitamin B1, TPP) and Mg2+.
- Dihydrolipoyl transacetylase oxidizes the remaining two-carbon molecule using lipoic acid, and transfers the resulting acetyl group to CoA, forming acetyl-CoA.
- Dihydrolipoyl dehydrogenase uses FAD to reoxidize lipoic acid, forming FADH2. This FADH2 can later transfer electrons to NAD+, forming NADH that can feed into the electron transport chain.
- Pyruvate dehydrogenase kinase phosphorylates PDH when ATP or acetyl-CoA levels are high, turning it off.
- Pyruvate dehydrogenase phosphatase dephosphorylates PDH when ADP levels are high, turning it on.
- Acetyl-CoA can be formed from fatty acids, which enter the mitochondria using carriers.
- The fatty acid couples with CoA in the cytosol to form fatty acyl-CoA, which moves to the intermembrane space.
- The acyl (fatty acid) group is transferred to carnitine to form acyl-carnitine, which crosses the inner membrane.
- The acyl group is transferred to a mitochondrial CoA to re-form fatty acyl-CoA, which can undergo β-oxidation to form acetyl-CoA.
- Acetyl-CoA can be formed from the carbon skeletons of ketogenic amino acids, ketone bodies, and alcohol.
Reactions of the Citric Acid Cycle
- The citric acid cycle takes place in the mitochondrial matrix.
- Its main purpose is to oxidize carbons in intermediates to CO2 and generate high-energy electron carriers (NADH and FADH2) and GTP.
- Key enzymes and reactions:
- Citrate synthase couples acetyl-CoA to oxaloacetate and then hydrolyzes the resulting product, forming citrate and CoA–SH. This enzyme is regulated by negative feedback from ATP, NADH, succinyl-CoA, and citrate.
- Aconitase isomerizes citrate to isocitrate.
- Isocitrate dehydrogenase oxidizes and decarboxylates isocitrate to form α-ketoglutarate. This enzyme generates the first CO2 and first NADH of the cycle. As the rate-limiting step of the citric acid cycle, it is heavily regulated: ATP and NADH are inhibitors; ADP and NAD+ are activators.
- α-Ketoglutarate dehydrogenase complex acts similarly to PDH complex, metabolizing α-ketoglutarate to form succinyl-CoA. This enzyme generates the second CO2 and second NADH of the cycle. It is inhibited by ATP, NADH, and succinyl-CoA; it is activated by ADP and Ca2+.
- Succinyl-CoA synthetase hydrolyzes the thioester bond in succinyl-CoA to form succinate and CoA–SH. This enzyme generates the one GTP generated in the cycle.
- Succinate dehydrogenase oxidizes succinate to form fumarate. This flavoprotein is anchored to the inner mitochondrial membrane because it requires FAD, which is reduced to form the one FADH2 generated in the cycle.
- Fumarase hydrolyzes the alkene bond of fumarate, forming malate.
- Malate dehydrogenase oxidizes malate to oxaloacetate. This enzyme generates the third and final NADH of the cycle.
The Electron Transport Chain
- The electron transport chain takes place on the matrix-facing surface of the inner mitochondrial membrane.
- NADH donates electrons to the chain, which are passed from one complex to the next. As the ETC progresses, reduction potentials increase until oxygen, which has the highest reduction potential, receives the electrons.
- Complex I (NADH-CoQ oxidoreductase) uses an iron–sulfur cluster to transfer electrons from NADH to flavin mononucleotide (FMN), and then to coenzyme Q (CoQ), forming CoQH2. Four protons are translocated by Complex I.
- Complex II (Succinate-CoQ oxidoreductase) uses an iron–sulfur cluster to transfer electrons from succinate to FAD, and then to CoQ, forming CoQH2. No proton pumping occurs at Complex II.
- Complex III (CoQH2**-cytochrome c oxidoreductase**) uses an iron–sulfur cluster to transfer electrons from CoQH2 to heme, forming cytochrome c as part of the Q cycle. Four protons are translocated by Complex III.
- Complex IV (**cytochrome c oxidase**) uses cytochromes and Cu2+ to transfer electrons in the form of hydride ions (H–) from cytochrome c to oxygen, forming water. Two protons are translocated by Complex IV.
- NADH cannot cross the inner mitochondrial membrane. Therefore, one of two available shuttle mechanisms to transfer electrons in the mitochondrial matrix must be used.
- In the glycerol 3-phosphate shuttle, electrons are transferred from NADH to dihydroxyacetone phosphate (DHAP), forming glycerol 3-phosphate. These electrons can then be transferred to mitochondrial FAD, forming FADH2.
- In the malate–aspartate shuttle, electrons are transferred from NADH to oxaloacetate, forming malate. Malate can then cross the inner mitochondrial membrane and transfer the electrons to mitochondrial NAD+, forming NADH.
Oxidative Phosphorylation
- The proton-motive force is the electrochemical gradient generated by the electron transport chain across the inner mitochondrial membrane. The intermembrane space has a higher concentration of protons than the matrix; this gradient stores energy, which can be used to form ATP via chemiosmotic coupling.
- ATP synthase is the enzyme responsible for generating ATP from ADP and an inorganic phosphate (Pi).
- The F0 portion is an ion channel, allowing protons to flow down the gradient from the intermembrane space to the matrix.
- The F1 portion uses the energy released by the gradient to phosphorylate ADP into ATP.
- The following is a summary of the energy yield of the various carbohydrate metabolism processes:
- Glycolysis generates 2 NADH and 2 ATP.
- Pyruvate dehydrogenase generates 1 NADH per molecule of pyruvate. Because each glucose forms two molecules of pyruvate, this complex produces a net of 2 NADH.
- The citric acid cycle generates 3 NADH, 1 FADH2, and 1 GTP (6 NADH, 2 FADH2, and 2 GTP per molecule of glucose).
- Each NADH yields 2.5 ATP; 10 NADH form 25 ATP.
- Each FADH2 yields 1.5 ATP; 2 FADH2 form 3 ATP.
- GTP are converted to ATP.
- 2 ATP from glycolysis + 2 ATP (GTP) from the citric acid cycle + 25 ATP from NADH + 3 ATP from FADH2 = 32 ATP per molecule of glucose (optimal). Inefficiencies of the system and variability between cells make 30–32 ATP/glucose the commonly accepted range for energy yield.
ANSWERS TO CONCEPT CHECKS
**10.1**
-
Pyruvate + CoA–SH + NAD+ → acetyl-CoA + CO2 + NADH + H+
-
Molecule Mechanism of Conversion to Acetyl-CoA Fatty acids Shuttle acyl group from cytosolic CoA–SH to mitochondrial CoA–SH via carnitine; then undergo β-oxidation
Ketogenic amino acids Transaminate to lose nitrogen; convert carbon skeleton into ketone body, which can be converted into acetyl-CoA
Ketones Reverse of ketone body formation
Alcohol Alcohol dehydrogenase and acetaldehyde dehydrogenase convert alcohol into acetyl-CoA
**10.2**
- Complete oxidation of carbons in intermediates to CO2 so that reduction reactions can be coupled with CO2 formation, thus forming energy carriers such as NADH and FADH2 for the electron transport chain.
-
Isocitrate dehydrogenase
-
Checkpoints Inhibitors Activators Citrate synthase ATP, NADH, succinyl-CoA, citrate None
Isocitrate dehydrogenase ATP, NADH ADP, NAD+
***α*-Ketoglutarate complex** ATP, NADH, succinyl-CoA ADP, Ca2+
**10.3**
-
- Pumping a proton into the intermembrane space: I, III, and IV
- Acquiring electrons from NADH: I
- Acquiring electrons from FADH2: II
- Having the highest reduction potential: IV (reduction potentials increase along the ETC)
- The electron transport chain generates the proton-motive force, an electrochemical gradient across the inner mitochondrial membrane, which provides the energy for ATP synthase to function.
- The malate–aspartate shuttle. Because this mechanism is the more efficient one, it makes sense for a highly aerobic organ such as the heart to utilize it in order to maximize its ATP yield.
**10.4**
- The ETC is made up of the physical set of intermembrane proteins located on the inner mitochondrial matrix, and they undergo oxidation–reduction reactions as they transfer electrons to oxygen, the final electron acceptor. As electrons are transferred, a proton-motive force is generated in the intermembrane space. Oxidative phosphorylation is the process by which ATP is generated via harnessing the proton gradient, and it utilizes ATP synthase to do so.
- By splitting up electron transfer into several complexes, enough energy is released to facilitate the creation of a proton gradient at many locations, rather than just one. The greater the proton gradient is, the greater the ATP generation will be. Direct reduction of oxygen by NADH would release a significant amount of energy to the environment, resulting in inefficient electron transport.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. B
Phosphofructokinase-1 (PFK-1), which catalyzes the rate-limiting step of glycolysis, is the only enzyme listed here that functions under anaerobic conditions. The other enzymes are all involved in the oxygen-requiring processes discussed in this chapter. Succinate dehydrogenase, (A), appears in both the citric acid cycle and as part of Complex II of the electron transport chain. Isocitrate dehydrogenase, (C), catalyzes the rate-limiting step of the citric acid cycle. Pyruvate dehydrogenase, (D), is one of the five enzymes that make up the pyruvate dehydrogenase complex.
2. B
The increased permeability of the inner mitochondrial membrane allows the proton-motive force to be dissipated through locations besides the F0 portion of ATP synthase. Therefore, ATP synthase is less active and is forming less ATP, invalidating (A) and (C). The body will attempt to regenerate the proton-motive force by increasing fuel catabolism, eliminating (D). This increase in fuel use requires more oxygen utilization in the electron transport chain.
3. B
The citric acid cycle takes place in the mitochondrial matrix, not the outer mitochondrial membrane. While most citric acid cycle enzymes are located within the matrix, succinate dehydrogenase is located on the inner mitochondrial membrane.
4. C
It is not necessary to have all the net reactions memorized for each metabolic process to answer this question; all we need is to identify a few key reactants and products. In this case, we start with acetyl-CoA and end with CoA–SH. We also notice that in this reaction, NAD+and FAD are reduced to NADH and FADH2, and that CO2 is formed. The only metabolic process in which all of the above reactions would occur is the citric acid cycle, also called the tricarboxylic acid (TCA) or Krebs cycle.
5. A
This question is testing our general knowledge of cellular respiration. Notice that all types of cellular respiration (aerobic and anaerobic) start with the degradation of glucose by glycolysis. In aerobic respiration, oxygen is the final electron acceptor, and water is therefore produced at the end of the electron transport chain. While oxygen is needed for aerobic respiration in order to produce the optimal 32 molecules of ATP per glucose, it is not the only method by which ATP is produced. Glycolysis still provides 2 ATP per glucose without the need for oxygen, thus making (B) and (D) incorrect. Water, mentioned in (C), is produced in aerobic metabolism, not consumed.
6. C
Fat molecules stored in adipose tissue can be hydrolyzed by lipases to fatty acids and glycerol. While glycerol can be converted into glyceraldehyde 3-phosphate, a glycolytic intermediate, a fatty acid must first be activated in the cytoplasm by coupling the fatty acid to CoA–SH, forming fatty acyl-CoA. The fatty acid is then transferred to a molecule of carnitine, which can carry it across the inner mitochondrial membrane. Once inside, the fatty acid is transferred to a mitochondrial CoA–SH, re-forming fatty acyl-CoA. Through fatty acid oxidation, this fatty acyl-CoA can become acetyl-CoA, which enters the citric acid cycle.
7. C
Based on the wording of this question, three choices are used to produce acetyl-CoA and one is not. As part of aerobic metabolism, decarboxylation of pyruvate by the pyruvate dehydrogenase complex yields acetyl-CoA, NADH, and CO2. Other sources of acetyl-CoA include amino acid catabolism, beta oxidation of fatty acids, and alcohol metabolism. Fatty acids are produced via reduction of acetyl-CoA and cannot be further reduced, making (C) correct.
8. C
Cytochrome c carries electrons from CoQH2-cytochrome c oxidoreductase (Complex III) to cytochrome c oxidase (Complex IV) as part of the electron transport chain. The ETC takes place on the inner mitochondrial membrane.
9. B
During oxidative phosphorylation, energy is harvested from the energy carriers FADH2 and NADH in order to form ATP. One molecule of mitochondrial FADH2 is oxidized to produce 1.5 molecules of ATP. Similarly, one molecule of mitochondrial NADH is oxidized to produce 2.5 molecules of ATP in the electron transport chain.
10. B
Thioester links release a great deal of energy when hydrolyzed, making them well-suited as respiration reaction drivers. They are particularly useful because they release more energy than typical ester cleavage. It is thioester formation, not hydrolysis, that requires a great deal of energy, making (D) incorrect.
11. B
The conversion of GDP to GTP is a phosphorylation reaction, in which a phosphate group is added to a molecule. Such reactions are catalyzed by kinases. Nomenclature is helpful here, as nucleosidediphosphate kinase is the only enzyme that contains kinase in its name.
12. C
The wording of these answer choices is critical. The electrons from cytosolic NADH can enter the mitochondrion through one of two shuttle mechanisms: the glycerol 3-phosphate shuttle, which ultimately moves these electrons to mitochondrial FAD, and the malate–aspartate shuttle, which ultimately moves these electrons to mitochondrial NAD+. If the electrons are transferred using the malate–aspartate shuttle, then no energy is lost, making (A) and (D) incorrect. NADH cannot enter the matrix directly, making (B) incorrect. It is the fact that electrons can use more than one pathway—one of which loses energy that could be used for ATP synthesis—that accounts for the potentially decreased yield of ATP from cytosolic NADH.
13. A
Uncouplers inhibit ATP synthesis without affecting the electron transport chain. Because the body must burn more fuel to maintain the proton-motive force, glycogen stores will be mobilized to feed into glycolysis, then the TCA, and finally oxidative phosphorylation.
14. B
Complex II is the only complex of the ETC that does not contribute to the proton gradient. Complexes I and III each add four protons to the gradient; Complex IV adds two protons to the gradient.
15. B
While all of the other answers contribute to energy production, it is the electrochemical gradient (proton-motive force) that directly drives the phosphorylation of ATP by the F1 portion of ATP synthase.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 2
- Enzymes
- Biochemistry Chapter 4
- Carbohydrate Structure and Function
- Biochemistry Chapter 9
- Carbohydrate Metabolism I
- Biochemistry Chapter 11
- Lipid and Amino Acid Metabolism
- Biochemistry Chapter 12
- Bioenergetics and Regulation of Metabolism
- General Chemistry Chapter 12
- Electrochemistry