Chapter 3: Bonding and Chemical Interactions

Chapter 3: Bonding and Chemical Interactions
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
What is the character of the bonds in carbon dioxide?
- Ionic
- Polar covalent
- Nonpolar covalent
- Coordinate covalent
-
Which of the following molecules contains the oxygen atoms with the most negative average formal charge?
- H2O
- CO32–
- O3
- CH2O
-
Which of the following elements does NOT break the octet rule?
- Be
- S
- Na
- P
-
Which of the following correctly ranks the compounds below by ascending boiling point?
- Acetone
- KCl
- Kr
- Isopropyl alcohol
- I < II < IV < III
- III < IV < I < II
- II < IV < I < III
- III < I < IV < II
-
Both CO32– and ClF3 have three atoms bonded to a central atom. What is the best explanation for why CO32– has trigonal planar electronic geometry, while ClF3 has trigonal bipyramidal electronic geometry?
- CO32– has multiple resonance structures, while ClF3 does not.
- CO32– has a charge of −2, while ClF3 has no charge.
- ClF3 has lone pairs on its central atom, while CO32– has none.
- CO32– has lone pairs on its central atom, while ClF3 has none.
-
Which of the following elements, when paired together, would form ionic bonds?
- Mg
- Cl
- O
- H
- II and IV
- I and II
- II and III
- III and IV
-
Despite the fact that both C2H2 and HCN contain triple bonds, the lengths of these triple bonds are not equal. Which of the following is the best explanation for this finding?
- In C2H2, the bond is shorter because it is between atoms of the same element.
- The two molecules have different resonance structures.
- Carbon is more electronegative than hydrogen.
- Nitrogen is more electronegative than carbon.
-
Which of the following is the best explanation of the phenomenon of hydrogen bonding?
- Hydrogen has a strong affinity for holding onto valence electrons.
- Hydrogen can only hold two valence electrons.
- Electronegative atoms disproportionately carry shared electron pairs when bonded to hydrogen.
- Hydrogen bonds have ionic character.
-
Which of the following best describes the number and character of the bonds in an ammonium cation?
- Three polar covalent bonds
- Four polar covalent bonds, of which none are coordinate covalent bonds
- Four polar covalent bonds, of which one is a coordinate covalent bond
- Four polar covalent bonds, of which two are coordinate covalent bonds
-
Although the octet rule dictates much of molecular structure, some atoms can violate the octet rule by being surrounded by more than eight electrons. Which of the following is the best explanation for why some atoms can exceed the octet?
- Atoms that exceed the octet already have eight electrons in their outermost electron shell.
- Atoms that exceed the octet only do so when bonding with transition metals.
- Atoms that exceed the octet can do so because they have d-orbitals in which extra electrons can reside.
- Some atoms can exceed the octet because they are highly electronegative.
-
Which of the following types of intermolecular forces provides the most accurate explanation for why noble gases can liquefy?
- Hydrogen bonding
- Ion–dipole interactions
- Dispersion forces
- Dipole–dipole interactions
-
In the structure shown, which atom(s) have the most positive charge?
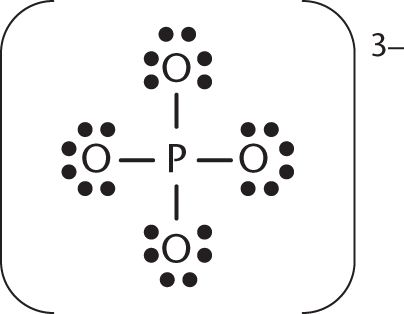
- The phosphorus atom has the most positive charge.
- All atoms share the charge equally.
- The four oxygen atoms share the highest charge.
- The oxygen atom at the peak of the trigonal pyramidal geometry has the most positive charge.
-
Which of the following is the best name for the new bond formed in the reaction shown?
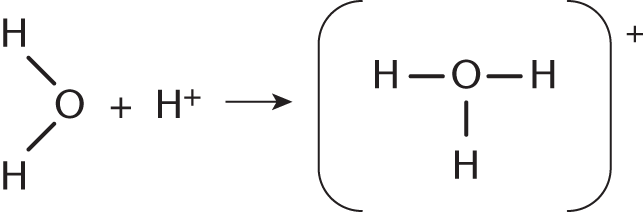
- Nonpolar covalent bond
- Ionic bond
- Coordinate covalent bond
- Hydrogen bond
-
Both BF3 and NH3 have three atoms bonded to the central atom. Which of the following is the best explanation for why the geometry of these two molecules is different?
- BF3 has three bonded atoms and no lone pairs, which makes its geometry trigonal pyramidal.
- NH3 is nonpolar, while BF3 is polar.
- NH3 has three bonded atoms and one lone pair, which makes its geometry trigonal pyramidal.
- BF3 is nonpolar, while NH3 is polar.
-
Which of the following compounds has the highest melting point?
- Sucrose
- NaCl
- Glycerol
- H2O
Answer Key
- B
- B
- C
- D
- C
- B
- D
- C
- C
- C
- C
- A
- C
- C
- B
Chapter 3: Bonding and Chemical Interactions
CHAPTER 3
BONDING AND CHEMICAL INTERACTIONS
In This Chapter
3.1 Bonding
The Octet Rule
Types of Bonds
3.2 Ionic Bonds 3.3 Covalent Bonds
Properties of Covalent Compounds
Coordinate Covalent Bonds
Covalent Bond Notation
Geometry and Polarity
Atomic and Molecular Orbitals
3.4 Intermolecular Forces
London Dispersion Forces
Dipole–Dipole Interactions
Hydrogen Bonds
Concept Summary
CHAPTER PROFILE
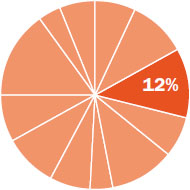
The content in this chapter should be relevant to about 12% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
4C:Electrochemistry and electrical circuits and their elements
5B: Nature of molecules and intermolecular interactions
Introduction
The Maillard reaction is one of the most important chemical processes that occurs while cooking. The reaction mechanism itself is one with which you are closely familiar from your studies of organic chemistry: a nucleophilic reaction between the amino terminus of the peptide chain of a protein and the carbonyl functionality of a sugar to form an N-substituted glycosylamine. This compound undergoes a complex series of rearrangements and other reactions to produce a set of compounds that gives cooked food its pleasing color and delectable flavor. This reaction is especially important for browning meat.
When the surface of the meat comes into contact with the hot surface of a pan or grill, the proteins and sugars on the meat’s exterior begin interacting via the Maillard reaction. The pan must be sufficiently hot to bring the exterior of the meat to a temperature of 155 °C (310°F), the optimal temperature for the reaction to occur. So how does a grill master achieve the impossible: generating very high heat for the exterior but not overcooking the interior? The answer lies, in part, in drying the meat. When meat that has a lot of water on its exterior surface hits the hot pan, the first process that takes place is the boiling of the water. Boiling is a phase change from liquid to gas and occurs at a constant temperature; water’s boiling point is 100 °C (212°F). Because this temperature is considerably lower than that necessary for the Maillard reaction, no browning will occur and the flavor compounds will not form. The lesson here is: if you want a tasty steak, always dry the surface of your meat!
Of course, the real lesson is the topic of discussion for this chapter: bonding and chemical interactions. We will not address complex chemical bonding, such as that which takes place in the Maillard reaction, in this chapter. Rather, this chapter will address the basics of chemical bonding and interactions. Here, we will investigate the nature and behavior of covalent and ionic bonds. We will also review a system—Lewis structures—by which bonding electrons are accounted for, and we will address the main principles of valence shell electron pair repulsion (VSEPR) theory. Finally, we will recount the various modes of interaction between molecules: intermolecular forces.
3.1 Bonding
LEARNING OBJECTIVES
After Chapter 3.1, you will be able to:
- Compare and contrast ionic and covalent compounds
- Identify elements that do not obey the octet rule
- Apply periodic trends to determine whether a covalent bond is polar or nonpolar:
The atoms of most elements, except for a few noble gases, can combine to form molecules. The atoms within these molecules are held together by strong attractive forces called chemical bonds, which are formed via the interaction of the valence electrons of the combining atoms. The chemical and physical properties of the resulting compound are usually very different from those of the constituent elements. For example, elemental sodium, an alkali metal, is so reactive that it can actually produce fire when reacting with water because the reaction is highly exothermic. Diatomic chlorine gas is so toxic that it was used for chemical warfare during World War I. However, when sodium and chlorine react, the biologically important compound NaCl (table salt) is produced.
The Octet Rule
How do atoms join together to form compounds? In the example above, how do the sodium and the chlorine atoms form sodium chloride? For many molecules, the constituent atoms bond according to the octet rule, which states that an atom tends to bond with other atoms so that it has eight electrons in its outermost shell, thereby forming a stable electron configuration similar to that of the noble gases. An example of an octet configuration is shown for the noble gas argon (Ar) in Figure 3.1.
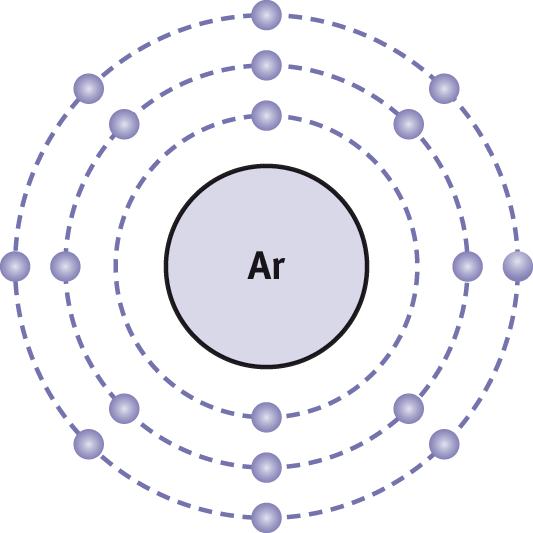
Figure 3.1. Electron Configuration of Argon As a noble gas, argon has a complete octet in its valence shell.
However, this is more of a “rule of thumb,” because there are more elements that can be exceptions to the rule than those that follow the rule. These “exceptional” elements include hydrogen, which can only have two valence electrons (achieving the configuration of helium); lithium and beryllium, which bond to attain two and four valence electrons, respectively; boron, which bonds to attain six valence electrons; and all elements in period 3 and greater, which can expand the valence shell to include more than eight electrons by incorporating d-orbitals. For example, in certain compounds, chlorine can form seven covalent bonds, thereby holding 14 electrons in its valence shell.
KEY CONCEPT
The octet rule is the desire of all atoms to achieve noble gas configuration. However, keep in mind that there are many exceptions to this rule.
A simple way to remember all the exceptions is as follows:
- Incomplete octet: These elements are stable with fewer than 8 electrons in their valence shell and include hydrogen (stable with 2 electrons), helium (2), lithium (2), beryllium (4), and boron (6).
- Expanded octet: Any element in period 3 and greater can hold more than 8 electrons, including phosphorus (10), sulfur (12), chlorine (14), and many others.
- Odd numbers of electrons: Any molecule with an odd number of valence electrons cannot distribute those electrons to give eight to each atom; for example, nitric oxide (NO) has eleven valence electrons.
Another way to remember the exceptions is to remember the common elements that almost always abide by the octet rule: carbon, nitrogen, oxygen, fluorine, sodium, and magnesium. Note that nonmetals gain electrons and metals lose electrons to achieve their respective complete octets.
Types of Bonds
We classify chemical bonds into two distinct types: ionic and covalent.
In ionic bonding, one or more electrons from an atom with a low ionization energy, typically a metal, are transferred to an atom with a high electron affinity, typically a nonmetal. An illustration of such a process is shown for our initial example, sodium chloride, in Figure 3.2.
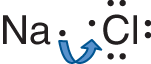
Figure 3.2. Formation of an Ionic Bond Sodium (Na) has a low ionization energy, easily releasing an electron; chlorine (Cl) has a high electron affinity, easily absorbing that electron into its valence shell. In this example, both atoms achieve an octet formation.
The resulting electrostatic attraction between opposite charges is what holds the ions together. This is the nature of the bond in sodium chloride, where the positively charged sodium cation is electrostatically attracted to the negatively charged chloride anion. It is important to note that this type of electrostatic attraction creates lattice structures consisting of repeating rows of cations and anions, rather than individual molecular bonds, as shown in Figure 3.3.

Figure 3.3. Crystal Lattice Structure of Sodium Chloride Sodium = purple; chloride = green
In covalent bonding, an electron pair is shared between two atoms, typically nonmetals, that have relatively similar values of electronegativity. The degree to which the pair of electrons is shared equally or unequally between the two atoms determines the degree of polarity in the covalent bond. For example, if the electron pair is shared equally, the covalent bond is nonpolar; and if the pair is shared unequally, the bond is polar. If both of the shared electrons are contributed by only one of the two atoms, the bond is called coordinate covalent.
BRIDGE
Electronegativity, discussed in Chapter 2 of MCAT General Chemistry Review, is a property that addresses how an individual atom acts within a bond and will help us understand the formation of molecules from atoms with different electronegativities.
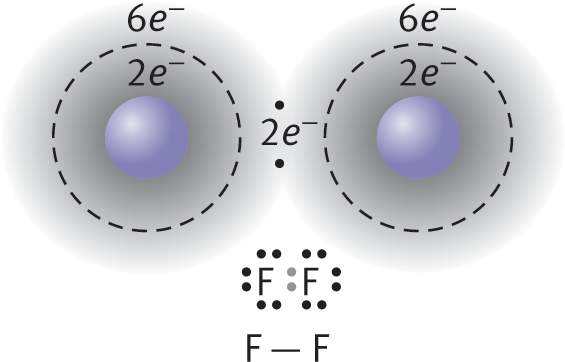
An example of nonpolar covalent bonding is shown for diatomic fluorine in Figure 3.4. Each atom has seven electrons in its valence shell, and by sharing one of these electrons from each atom, they can each form an octet. Unlike ionic crystal lattices, covalent compounds consist of individually bonded molecules.
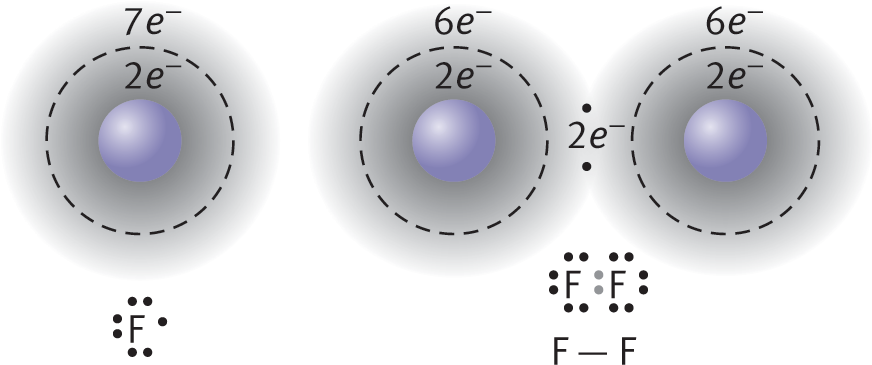
Figure 3.4. Formation of a Covalent Bond Fluorine (F) has seven valence electrons; by sharing one electron from each atom, both fluorine atoms achieve an octet formation.
MCAT CONCEPT CHECK 3.1:
Before you move on, assess your understanding of the material with these questions.
-
Describe the atomic differences between ionic and covalent compounds:
- Ionic: ________________________________
- Covalent: ____________________________
-
List three elements that do not follow the octet rule and explain why:
Element Why It Violates the Octet Rule
-
Which periodic trend determines whether a covalent bond is polar or nonpolar?
_______________________________________
3.2 Ionic Bonds
LEARNING OBJECTIVES
After Chapter 3.2, you will be able to:
- Explain why ionic bonds are commonly formed between metals and nonmetals
- Recall the major characteristics of ionic compounds
Ionic bonds form between atoms that have significantly different electronegativities. The atom that loses the electrons becomes a cation, and the atom that gains electrons becomes an anion. The ionic bond is the result of an electrostatic force of attraction between the opposite charges of these ions. Electrons are not shared in an ionic bond. For this electron transfer to occur, the difference in electronegativity must be greater than 1.7 on the Pauling scale.
MNEMONIC
- MeTals lose electrons to become caTions = posiTive (+) ions
- Nonmetals gain electrons to become aNions = Negative (–) ions
The MCAT won’t expect you to memorize the Pauling scale, but recognize that ionic bonds are generally formed between a metal and a nonmetal. For example, alkali and alkaline earth metals of Groups IA and IIA (Groups 1 and 2, respectively) readily form ionic bonds with the halogens of Group VIIA (Group 17). The atoms of the active metals loosely hold onto their electrons, whereas the halogens are more likely to gain an electron to complete their valence shell. The differences in bonding behavior for these classes of elements, and their differences in electronegativity values (ΔEN), explain the formation of ionic compounds such as cesium chloride (ΔEN = 2.3), potassium iodide (ΔEN = 1.7), and sodium fluoride (ΔEN = 3.1).
Ionic compounds have characteristic physical properties that you should recognize for Test Day. Because of the strength of the electrostatic force between the ionic constituents of the compound, ionic compounds have very high melting and boiling points. For example, the melting point of sodium chloride is 801 °C. Many ionic compounds dissolve readily in water and other polar solvents and, in the molten or aqueous state, are good conductors of electricity. In the solid state, the ionic constituents of the compound form a crystalline lattice consisting of repeating positive and negative ions, as shown earlier in Figure 3.3. With this arrangement, the attractive forces between oppositely charged ions are maximized, and the repulsive forces between ions of like charge are minimized.
MCAT CONCEPT CHECK 3.2:
Before you move on, assess your understanding of the material with these questions.
-
Why do ionic bonds tend to form between metals and nonmetals?
_______________________________________
_______________________________________
-
Describe five characteristics of ionic compounds.
- ___________________________
- ___________________________
- ___________________________
- ___________________________
- ___________________________
3.3 Covalent Bonds
LEARNING OBJECTIVES
After Chapter 3.3, you will be able to:
- Explain the relationship between bond strength, bond length, and bond energy
- Identify the values of ΔEN for which polar covalent, nonpolar covalent, and ionic bonds will form
- Predict the molecular geometry of a molecule given its formula
- Draw Lewis dot structures for simple molecules, including resonance structures, such as:
When two or more atoms with similar electronegativities interact, the energy required to form ions through the complete transfer of one or more electrons is greater than the energy that would be released upon the formation of an ionic bond. That is, when two atoms of similar tendency to attract electrons form a compound, it is energetically unfavorable to create ions. Rather than transferring electrons to form octets, the atoms share electrons. The bonding force between the atoms is not ionic; instead, there is an attraction that each electron in the shared pair has for the two positive nuclei of the bonded atoms.
MCAT EXPERTISE
Think of bonds as a tug-of-war between two atoms. When the difference in electronegativity is great (more than 1.7), then the “stronger” atom wins all of the electrons and becomes the anion. When the electronegativity values are relatively similar, then we have a stalemate, or a covalent bond with mostly equal sharing of electrons.
Covalent compounds contain discrete molecular units with relatively weak intermolecular interactions. As a result, compounds like carbon dioxide (CO2) tend to have lower melting and boiling points. In addition, because they do not break down into constituent ions, they are poor conductors of electricity in the liquid state or in aqueous solutions.
Properties of Covalent Compounds
The formation of one covalent bond may not be sufficient to fill the valence shell for a given atom. Thus, many atoms can form bonds with more than one other atom, and most atoms can form multiple bonds with other atoms. Two atoms sharing one, two, or three pairs of electrons are said to be joined by a single, double, or triple covalent bond, respectively. The number of shared electron pairs between two atoms is called the bond order; hence, a single bond has a bond order of one, a double bond has a bond order of two, and a triple bond has a bond order of three. There are three important characteristics of a covalent bond to explain: bond length, bond energy, and polarity.
Bond Length
Bond length is the average distance between the two nuclei of atoms in a bond. As the number of shared electron pairs increases, the two atoms are pulled closer together, resulting in a decrease in bond length. Thus, for a given pair of atoms, a triple bond is shorter than a double bond, which is shorter than a single bond.
KEY CONCEPT
You will see this inverse relationship between bond length and strength in both organic and general chemistry.
Bond Length Bond Strength
C–C longest weakest
C=C medium medium
C≡C shortest strongest
Know this relationship on Test Day and you’ll earn quick points!
Bond Energy
Bond energy is the energy required to break a bond by separating its components into their isolated, gaseous atomic states. The greater the number of pairs of electrons shared between the atomic nuclei, the more energy is required to break the bonds holding the atoms together. Thus, triple bonds have the greatest bond energy, and single bonds have the lowest bond energy. We will discuss bond energy and calculations involving bond enthalpy in Chapter 7 of MCAT General Chemistry Review. By convention, the greater the bond energy is, the stronger the bond.
Polarity
Polarity occurs when two atoms have a relative difference in electronegativities. When these atoms come together in covalent bonds, they must negotiate the degree to which the electron pairs will be shared. The atom with the higher electronegativity gets the larger share of the electron density. A polar bond creates a dipole, with the positive end of the dipole at the less electronegative atom and the negative end at the more electronegative atom, as shown in Figure 3.5.
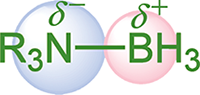
Figure 3.5. Polar Covalent Bond in an Amine Borane Nitrogen takes on a partial negative charge (δ–), boron takes on a partial positive charge (δ+).
When atoms that have identical or nearly identical electronegativities share electron pairs, they do so with equal distribution of the electrons. This is called a nonpolar covalent bond, and there is no separation of charge across the bond. Note that only atoms of the same element have exactly the same electronegativity, and therefore only diatomic molecules contain bonds with a purely equal distribution of electrons. The seven common diatomic molecules are H2, N2, O2, F2, Cl2, Br2, and I2. At the same time, many bonds are close to nonpolar. Any bond between atoms with a difference in electronegativity less than 0.5 is generally considered nonpolar.
MNEMONIC
Here’s a quick way to remember the naturally occurring diatomic elements on the periodic table: they form the number 7 on the periodic table (except for H), there are 7 of them, and most of them are in Group VIIA: H2, N2, O2, F2, Cl2, Br2, I2.
Polar Covalent Bond
Atoms that differ moderately in their electronegativities will share electrons unevenly, resulting in polar covalent bonds. While the difference in their electronegativities (between 0.5 and 1.7) is not enough to result in the formation of an ionic bond, it is sufficient to cause a separation of charge across the bond. This results in the more electronegative element acquiring a greater portion of the electron density, taking on a partial negative charge (δ–), and the less electronegative element acquiring a smaller portion of the electron density, taking on a partial positive charge (δ+). For instance, the covalent bond in HCl is polar because the two atoms have a moderate difference in electronegativity (ΔEN = 0.9). In this bond, the chlorine atom gains a partial negative charge, and the hydrogen atom gains a partial positive charge. The difference in charge between the atoms is indicated by an arrow crossed at its tail end (giving the appearance of a “plus” sign) and pointing toward the negative end, as shown in Figure 3.6.
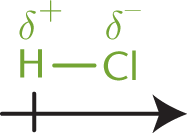
Figure 3.6. Dipole Moment of HCl
MCAT EXPERTISE
The range of electronegativities for nonpolar bonds is roughly 0 to 0.5. Polar bonds are found from 0.5 to 1.7, and ionic bonds are at 1.7 and above. Some chemistry courses allude to a grey area from 1.7 to 2.0. For the MCAT, if a molecule in this range has a metal and nonmetal, then it is effectively ionic; otherwise, it is polar covalent.
A molecule that has such a separation of positive and negative charges is called a polar molecule. The dipole moment of the polar bond or polar molecule is a vector quantity given by the equation:
p = qd Equation 3.1
where p is the dipole moment, q is the magnitude of the charge, and d is the displacement vector separating the two partial charges. The dipole moment vector, represented by an arrow pointing from the positive to the negative charge, is measured in Debye units (coulomb–meters).
Coordinate Covalent Bonds
In a coordinate covalent bond, both of the shared electrons originated on the same atom. Generally, this means that a lone pair of one atom attacked another atom with an unhybridized p-orbital to form a bond, as shown in Figure 3.7. Once such a bond forms, however, it is indistinguishable from any other covalent bond. The distinction is only helpful for keeping track of the valence electrons and formal charges. Coordinate covalent bonds are typically found in Lewis acid–base reactions, described in Chapter 10 of MCAT General Chemistry Review. A Lewis acid is any compound that will accept a lone pair of electrons, while a Lewis base is any compound that will donate a pair of electrons to form a covalent bond.
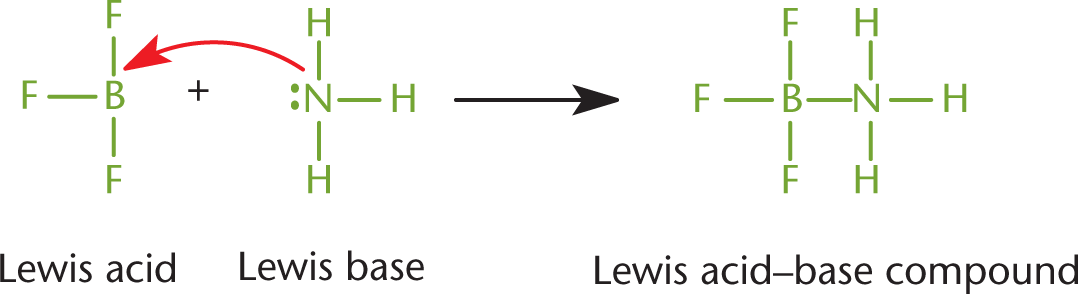
Figure 3.7. Coordinate Covalent Bond
BRIDGE
The chemistry that creates coordinate covalent bonds appears in many forms. These reactions can be called nucleophile–electrophile reactions, described in Chapter 4 of MCAT Organic Chemistry Review; Lewis acid–base reactions, described in Chapter 10 of MCAT General Chemistry Review, or complexation reactions, described in Chapter 9 of MCAT General Chemistry Review.
Here, NH3 donates a pair of electrons to form a coordinate covalent bond; thus, it acts as a Lewis base. At the same time, BF3 accepts this pair of electrons to form the coordinate covalent bond; thus, it acts as a Lewis acid.
Covalent Bond Notation
The electrons involved in a covalent bond are in the valence shell and are bonding electrons, while those electrons in the valence shell that are not involved in covalent bonds are nonbonding electrons. The unshared electron pairs are also known as lone pairs because they are associated only with one atomic nucleus. Because atoms can bond with other atoms in many different combinations, the Lewis structure system of notation was developed to keep track of the bonded and nonbonded electron pairs.
Think of Lewis structures as a bookkeeping method for electrons. The number of valence electrons attributed to a particular atom in the Lewis structure of a molecule is not necessarily the same as the number of valence electrons in the neutral atom. This difference accounts for the formal charge of an atom in a Lewis structure. Often, more than one Lewis structure can be drawn for a molecule. If the possible Lewis structures differ in their bond connectivity or arrangement, then the Lewis structures represent different possible compounds. However, if the Lewis structures show the same bond connectivity and differ only in the arrangement of the electron pairs, then these structures represent different resonance forms of a single compound. Note that Lewis structures do not represent the actual or even theoretical geometry of a real compound. Their usefulness lies in showing the different possible ways in which atoms may be combined to form different compounds or resonance forms of a single compound.
When more than one arrangement can be made, one can assess the likelihood of each arrangement by checking the formal charges on the atoms in each arrangement. The arrangement that minimizes the number and magnitude of formal charges is usually the most stable arrangement of the compound.
Lewis Structures
A Lewis structure, or Lewis dot diagram, is the chemical symbol of an element surrounded by dots, each representing one of the s or p valence electrons of the atom. The Lewis symbols of the elements in the second period of the periodic table are shown in Table 3.1.
Table 3.1. Lewis Symbols for Period 2 Elements

Lithium

Nitrogen

Beryllium

Oxygen

Boron

Fluorine

Carbon

Neon
KEY CONCEPT
In drawing Lewis dot structures, remember that some atoms can expand their octets by utilizing the d-orbitals in their outer shell. This will only take place with atoms in period 3 or greater.
MCAT EXPERTISE
The number of dots in Lewis Structure notation comes from group numbers. Lithium is in Group IA and therefore has one electron (dot). Carbon is in Group IVA and has four dots.
Just as a Lewis symbol is used to represent the distribution of valence electrons in an atom, it can also be used to represent the distribution of valence electrons in a molecule. For example, the Lewis symbol for a fluoride ion, F–, is
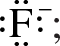
the Lewis structure of the diatomic molecule F2 is

. Certain rules must be followed in assigning a Lewis structure to a molecule. The steps for drawing a Lewis structure are outlined here, using HCN as an example.
- Draw out the backbone of the compound—that is, the arrangement of atoms. In general, the least electronegative atom is the central atom. Hydrogen (always) and the halogens F, Cl, Br, and I (usually) occupy a terminal position.
In HCN, H must occupy an end position. Of the remaining two atoms, C is the least electronegative and, therefore, occupies the central position. Therefore, the skeletal structure is as follows:
H – C – N
- Count all the valence electrons of the atoms. The number of valence electrons of the molecule is the sum of the valence electrons of all atoms present:
- H has 1 valence electron
- C has 4 valence electrons
- N has 5 valence electrons; therefore,
- HCN has a total of 10 valence electrons.
- Draw single bonds between the central atom and the atoms surrounding it. Each single bond corresponds to a pair of electrons:
H : C : N
- Complete the octets of all atoms bonded to the central atom, using the remaining valence electrons left to be assigned. Recall that H is an exception to the octet rule because it can only have two valence electrons. In this example, H already has two valence electrons from its bond with C.
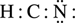
- Place any extra electrons on the central atom. If the central atom has less than an octet, try to write double or triple bonds between the central and surrounding atoms using the lone pairs on the surrounding atoms.
The HCN structure above does not satisfy the octet rule for C because C only has four valence electrons. Therefore, two lone electron pairs from the N atom must be moved to form two more bonds with C, creating a triple bond between C and N. To make it easier to visualize, bonding electron pairs are represented as lines. You should be familiar with both dot and line notation for bonds.
H – C ≡ N:
Now, the octet rule is satisfied for all three atoms; C and N have eight valence electrons, and H has two valence electrons.
Formal Charge
To determine if a Lewis structure is representative of the actual arrangement of atoms in a compound, one must calculate the formal charge of each atom. In doing so, assume a perfectly equal sharing of all bonded electron pairs, regardless of actual differences in electronegativity. In other words, assume that each electron pair is split evenly between the two nuclei in the bond. The difference between the number of electrons assigned to an atom in a Lewis structure and the number of electrons normally found in that atom’s valence shell is the formal charge. A simple equation you can use to calculate formal charge is:
formal charge = V − N nonbonding − 1 2 N bonding
Equation 3.2
where V is the normal number of electrons in the atom’s valence shell, Nnonbonding is the number of nonbonding electrons, and Nbonding is the number of bonding electrons (double the number of bonds because each bond has two electrons). The charge of an ion or compound is equal to the sum of the formal charges of the individual atoms comprising the ion or compound.
MNEMONIC
A less formal way to calculate formal charge is with the formula:
Formal charge = valence electrons — dots — sticks
Where a “dot” refers to a lone electron and a “stick” refers to a bond.
Example: Calculate the formal charge on the central N atom of [NH4]+.
Solution: The Lewis structure of [NH4]+ is:
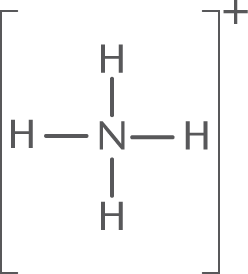
Nitrogen is in Group VA; thus, it has five valence electrons. In [NH4]+, N has four bonds (eight bonding electrons and zero nonbonding electrons).
- Thus, V = 5; Nbonding = 8; Nnonbonding = 0
- Formal charge = 5 − 0 − 1 2 ( 8 ) = + 1
- Thus, the formal charge on the N atom in [NH4]+ is +1.
One can also use logic to determine formal charge. As drawn, N has four bonds. Assuming equal sharing of the electrons in the bonds, this means N has four valence electrons. In its normal state, N has five valence electrons. Thus, nitrogen has one fewer electron than its normal state, and has a +1 charge.
Let us offer a brief note of explanation on the difference between formal charge and oxidation number: formal charge underestimates the effect of electronegativity differences, whereas oxidation numbers overestimate the effect of electronegativity differences, assuming that the more electronegative atom has a 100 percent share of the bonding electron pair. For example, in a molecule of CO2 (carbon dioxide), the formal charge on each of the atoms is 0, but the oxidation number of each of the oxygen atoms is −2 and of the carbon is +4. In reality, the distribution of electron density between the carbon and oxygen atoms lies somewhere between the extremes predicted by the formal charges and the oxidation states.
Resonance
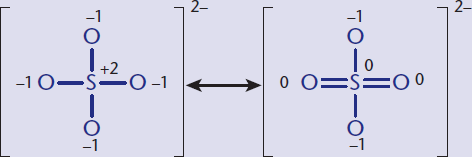
As suggested earlier, it may be possible to draw two or more Lewis structures that demonstrate the same arrangement of atoms but that differ in the specific placement of the electrons. These are called resonance structures and are represented with a double-headed arrow between them. The actual electronic distribution in the compound is a hybrid, or composite, of all of the possible resonance structures. For example, SO2 has three resonance structures, as shown in Figure 3.8.
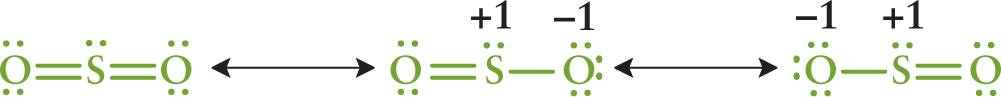
Figure 3.8 Resonance Structures for SO2 The double-headed arrows indicate that these molecules are involved in a resonance hybrid.
BRIDGE
Resonance is an important topic in both general and organic chemistry. It allows for greater stability, delocalizing electrons and charges over what is known as a π (pi) system. Resonance in organic molecules is discussed in Chapter 3 of MCAT Organic Chemistry Review.
The nature of the bonds within the actual compound is a hybrid of these three structures. If one were to evaluate the spectral data, it would indicate that the two S–O bonds are identical and equivalent. This phenomenon is known as resonance, and the actual structure of the compound is called the resonance hybrid.
The first resonance structure in Figure 3.8 is significantly more stable than the other two structures. Consequently, it is the major contributor to the resonance hybrid. In general, the more stable the structure, the more it contributes to the character of the resonance hybrid. In Figure 3.8, the minor contributors contain formal charges, indicating decreased stability. One can use formal charge to assess the stability of resonance structures according to the following guidelines:
- A Lewis structure with small or no formal charges is preferred over a Lewis structure with large formal charges.
- A Lewis structure with less separation between opposite charges is preferred over a Lewis structure with a large separation of opposite charges.
- A Lewis structure in which negative formal charges are placed on more electronegative atoms is more stable than one in which the negative formal charges are placed on less electronegative atoms.
Example: Write the resonance structures for [NCO]–.
Solution:
- C is the least electronegative of the three given atoms. Therefore, the C atom occupies the central position in the skeletal structure of [NCO]–:
N – C – O
C has 4 valence electrons; O has 6 valence electrons; and the species has one negative charge. Total valence electrons = 5 + 4 + 6 + 1 = 16
- N has 5 valence electrons;
- Draw single bonds between the central C atom and the surrounding atoms, N and O. Draw a pair of electrons to represent each bond.
N : C : O
- Complete the octets of N and O with the remaining 12 electrons.

- The C octet is incomplete. There are three ways in which double and triple bonds can be formed to complete the C octet: two lone pairs from the O atom can be used to form a triple bond between the C and O atoms:

Or one lone electron pair can be taken from both O and N to form two double bonds, one between N and C, the other between O and C:

Or two lone electron pairs can be taken from the N atom to form a triple bond between the C and N atoms:

All three are resonance structures of [NCO]–
- Assign formal charges to each atom of each resonance structure.
The most stable structure is this:

because the charges are minimized, and the negative formal charge is on the most electronegative atom, O.
Exceptions to the Octet Rule
As stated previously, the octet rule has many exceptions. In addition to hydrogen, helium, lithium, beryllium, and boron, which are exceptions because they cannot or do not usually reach the octet, all elements in or beyond the third period may be exceptions because they can take on more than eight electrons in their valence shells. These electrons can be placed into orbitals of the d subshell, and as a result, atoms of these elements can form more than four bonds. On Test Day, don’t automatically discount a Lewis structure with a central atom that has more than four bonds—the testmakers may be testing your ability to recognize that many atoms can expand their valence shells beyond the octet.
Consider the sulfate ion, SO42−. In the Lewis structure for the sulfate ion, giving the sulfur 12 valence electrons permits three of the five atoms to be assigned a formal charge of zero. The sulfate ion can be drawn in at least six resonance forms, many of which have two double bonds attached to a different combination of oxygen atoms. Figure 3.9 shows two of the possible forms.
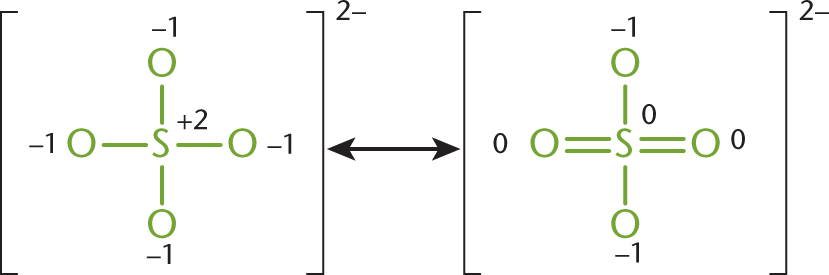
Figure 3.9. Two Different Resonance Forms of the Sulfate Ion
MCAT EXPERTISE
Don’t be surprised if you can draw more than two resonance structures for a molecule or ion. Becoming proficient at drawing and, more importantly, recognizing resonance structures will save you time on Test Day.
Geometry and Polarity
Because Lewis dot structures do not suggest or reflect the actual geometric arrangement of atoms in a compound, we need another system to provide this information. One such system is known as the valence shell electron pair repulsion (VSEPR) theory.
Valence Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory uses Lewis dot structures to predict the molecular geometry of covalently bonded molecules. It states that the three-dimensional arrangement of atoms surrounding a central atom is determined by the repulsions between bonding and nonbonding electron pairs in the valence shell of the central atom. These electron pairs arrange themselves as far apart as possible, thereby minimizing repulsive forces. The following steps are used to predict the geometrical structure of a molecule using the VSEPR theory:
- Draw the Lewis dot structure of the molecule.
- Count the total number of bonding and nonbonding electron pairs in the valence shell of the central atom.
- Arrange the electron pairs around the central atom so that they are as far apart as possible. For example, the compound AX2 has the Lewis structure X : A : X. The A atom has two bonding electron pairs in its valence shell. To position these electron pairs as far apart as possible, their geometric structure should be linear:
X – A – X
A summary of electronic geometries as predicted by VSEPR theory is shown in Table 3.2.
Table 3.2. VSEPR Theory This table lists the five most common electronic configurations of molecules.
REGIONS OF ELECTRON DENSITY EXAMPLE GEOMETRIC ARRANGEMENT OF ELECTRON PAIRS AROUND THE CENTRAL ATOM SHAPE ANGLE BETWEEN ELECTRON PAIRS
2 BeCl2 X – A – X linear 180°
3 BH3
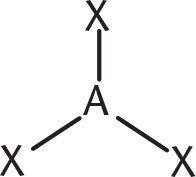
trigonal planar 120°
4 CH4
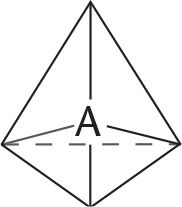
tetrahedral 109.5°
5 PCl5
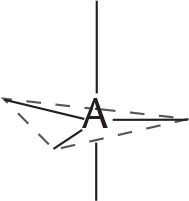
trigonal bipyramidal 90°, 120°, 180°
6 SF6
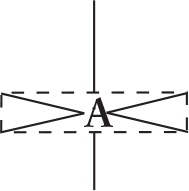
octahedral 90°, 180°
MCAT EXPERTISE
According to the AAMC’s official content lists, you need to be prepared to draw and identify structural formulas for molecules involving H, C, N, O, F, S, P, Si, and Cl. Rather than memorizing these elements, however, just be familiar with the process of creating a Lewis diagram for any element and predicting its three-dimensional shape from VSEPR theory.
Example: Predict the molecular geometry of NH3.
Solution:
- The Lewis structure of NH3 is:
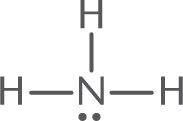
- The central atom, N, has three bonding electron pairs and one nonbonding electron pair, for a total of four electron pairs.
- The four electron pairs will be farthest apart when they occupy the corners of a tetrahedron. Because one of the four electron pairs is a lone pair, the observed molecular geometry is trigonal pyramidal, shown below.

In describing the shape of a molecule, only the arrangement of atoms (not electrons) is considered. Even though the electron pairs are arranged tetrahedrally, the shape of NH3 is pyramidal. It is not trigonal planar because the lone pair repels the three bonding electron pairs, causing them to move as far apart as possible.
KEY CONCEPT
The shapes from Table 3.2 refer to electronic geometry, which is different from molecular geometry. In the worked example, notice that the ammonia molecule has a tetrahedral electronic structure, but is considered to have a molecular structure that is trigonal pyramidal.
Example: Predict the geometry of CO2.
Solution: The Lewis structure of CO2 is
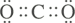
The double bond behaves just like a single bond for the purposes of predicting molecular shape. This compound has two groups of electrons around the carbon. According to the VSEPR theory, the two sets of electrons will orient themselves 180° apart, on opposite sides of the carbon atom, minimizing electron repulsion. Therefore, the molecular structure of CO2 is linear:

One subtlety that the MCAT loves to test is the difference between electronic geometry and molecular geometry. Electronic geometry describes the spatial arrangement of all pairs of electrons around the central atom, including both the bonding and the lone pairs. In contrast, the molecular geometry describes the spatial arrangement of only the bonding pairs of electrons. The coordination number, which is the number of atoms that surround and are bonded to a central atom, is the relevant factor when determining molecular geometry. For example, consider that CH4 (methane), NH3 (ammonia), and H2O all have the same electronic geometry: in each compound, four pairs of electrons surround the central atom. This is tetrahedral electronic geometry. However, because each molecule has a different coordination number, they have different molecular geometries. In molecular geometry, methane has tetrahedral geometry, ammonia has trigonal pyramidal geometry, and water is identified as angular or bent.
MCAT EXPERTISE
CH4, NH3, and H2O all have a tetrahedral electronic geometry, but differ in their molecular shapes:
CH4 is tetrahedral, NH3 is pyramidal, and H2O is bent or angular.
The distinction is important, and the MCAT will primarily focus on molecular geometry. However, there is one important implication of electronic geometry: the determination of the ideal bond angle. Tetrahedral electronic geometry, for example, is associated with an ideal bond angle of 109.5°; however, nonbonding pairs are able to exert more repulsion than bonding pairs because these electrons reside closer to the nucleus. Thus, the angle in ammonia is closer to 107°, and the angle in water is 104.5°.
Polarity of Molecules
When two atoms of different electronegativities bond covalently, sharing one or more pairs of electrons, the resulting bond is polar, with the more electronegative atom possessing the greater share of the electron density. However, the presence of bond dipoles does not necessarily result in a molecular dipole; that is, an overall separation of charge across the molecule. We must first consider the molecular geometry and the vector addition of the bond dipoles based upon that molecular geometry. A compound with nonpolar bonds is always nonpolar. However, a compound with polar bonds may be polar or nonpolar, depending upon the spatial orientation of the polar bonds in the molecule. If the compound has a molecular geometry such that the bond dipole moments cancel each other out (that is, if the vector sum is zero), then the result is a nonpolar compound. For example, CCl4 (carbon tetrachloride) has four polar C–Cl bonds, but because the molecular geometry of carbon tetrachloride is tetrahedral, the four bond dipoles point to the vertices of the tetrahedron and, therefore, cancel each other out, resulting in a nonpolar compound, as shown in Figure 3.10.
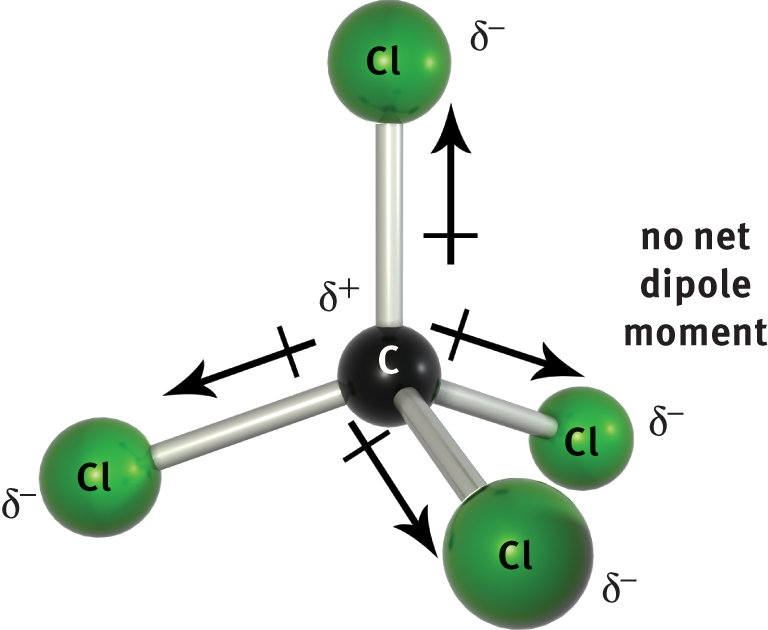
Figure 3.10. CCl4 is a Nonpolar Compound with Four Polar Bonds
However, when the molecular geometry is arranged such that the bond dipoles do not cancel each other out, the molecule will have a net dipole moment and will therefore be polar. For instance, the O–H bonds in H2O are polar, with each hydrogen atom assuming a partial positive charge and the oxygen assuming a partial negative charge. Recall that the molecular geometry of water is angular (bent). Therefore, the vector summation of the bond dipoles results in a molecular dipole moment from the partially positive hydrogen end to the partially negative oxygen end, as illustrated in Figure 3.11.
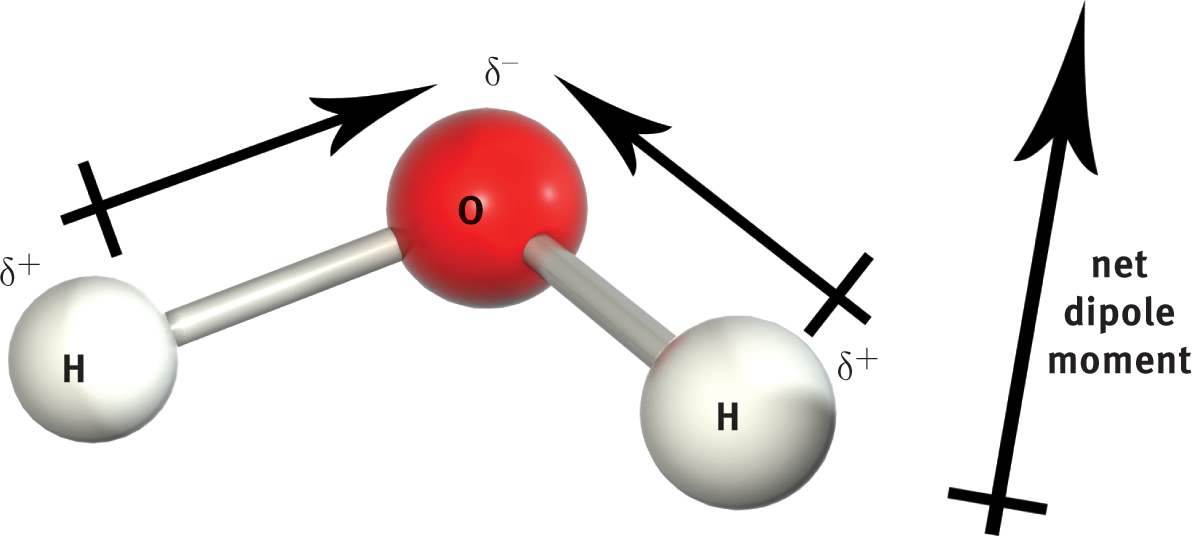
Figure 3.11. H2O is a Polar Molecule with Two Polar Bonds
MCAT EXPERTISE
Be careful! If you spot a polar bond in a molecule, the molecule can be either polar or nonpolar. On the contrary, when you see only nonpolar bonds in a molecule, the structure must be nonpolar. When in doubt, draw out relevant structures on your scratch paper on Test Day.
Atomic and Molecular Orbitals
To finish the discussion of covalent bonds, we need to address the concept of atomic and molecular orbitals. Recall the model of the atom as a dense, positively charged nucleus surrounded by a cloud of electrons organized into orbitals (regions in space surrounding the nucleus within which there are certain probabilities of finding an electron). The four quantum numbers describe the energy and position of an electron in an atom. While the principal quantum number, n, indicates the average energy level of the shell, the azimuthal quantum number, l, describes the subshells within each principal energy level. When l = 0, this indicates the s subshell, which has one orbital that is spherical in shape. The 1s-orbital (n = 1, l = 0, ml = 0) is plotted in Figure 3.12.
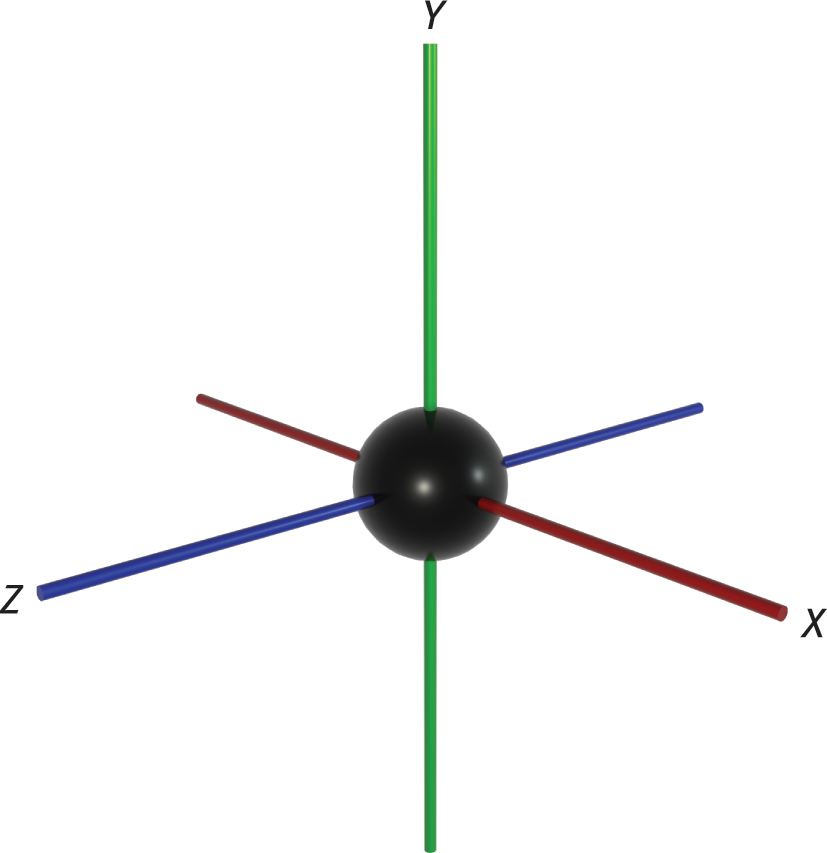
Figure 3.12. 1s-Orbital
BRIDGE
Quantum Numbers (Chapter 1 of MCAT General Chemistry Review) revisited:
For any value of n, there are n values of l (0 → n – 1).
- l = 0 → s
- l = 1 → p
- l = 2 → d
- l = 3 → f
For any value of l, there are 2l + 1 values of ml (number of orbitals); values range from –l to l.
When l = 1, this indicates the p subshell, which has three orbitals shaped like barbells along the x-, y-, and z-axes at right angles to each other. The 2p-orbitals (n = 2, l = 1, ml = −1, 0, and +1) are plotted in Figure 3.13.
Although well beyond the scope of the MCAT, mathematical analysis of the wave functions of the orbitals is used to determine and assign plus and minus signs to each lobe of the p-orbitals. The shapes of the five d-orbitals and the seven f-orbitals are more complex and do not need to be memorized for the MCAT.
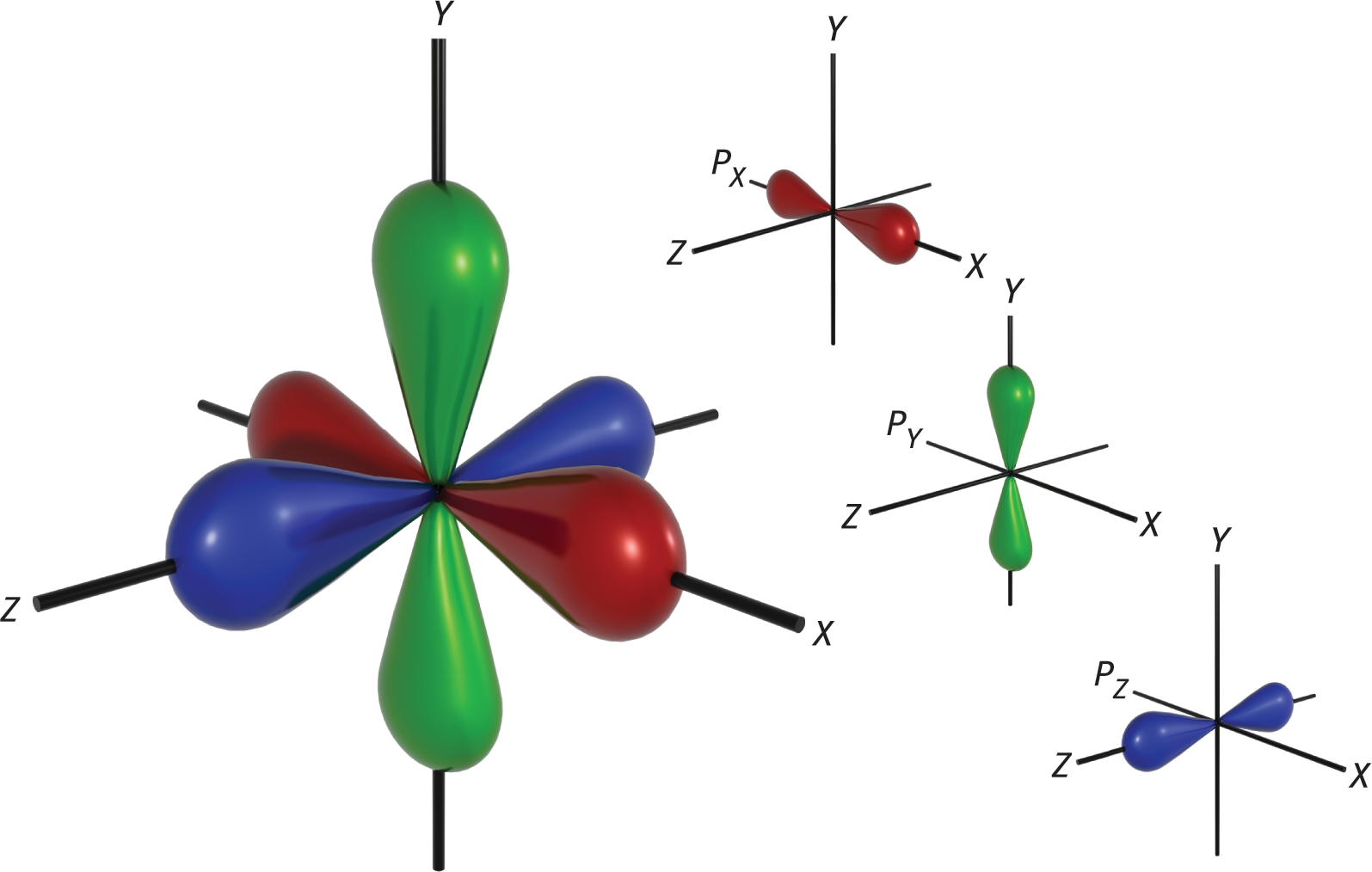
Figure 3.13. p-Orbitals on the x-, y-, and z-Axes
When two atoms bond to form a compound, the atomic orbitals interact to form a molecular orbital that describes the probability of finding the bonding electrons in a given space. Molecular orbitals are obtained by combining the wave functions of the atomic orbitals. Qualitatively, the overlap of two atomic orbitals describes this molecular orbital. If the signs of the two atomic orbitals are the same, a bonding orbital forms. If the signs are different, an antibonding orbital forms.
Two different patterns of overlap are observed in the formation of molecular bonds. When orbitals overlap head-to-head, the resulting bond is a sigma (σ) bond. σ bonds allow for free rotation about their axes because the electron density of the bonding orbital is a single linear accumulation between the atomic nuclei.
When the orbitals overlap in such a way that there are two parallel electron cloud densities, a pi(π) bond is formed. π bonds do not allow for free rotation because the electron densities of the orbitals are parallel and cannot be twisted in such a way that allows continuous overlapping of the clouds of electron densities.
MCAT CONCEPT CHECK 3.3:
Before you move on, assess your understanding of the material with these questions.
-
Describe the relationship between bond strength, bond length, and bond energy.
____________________________
-
For what values of ΔEN will a nonpolar covalent bond form? Polar covalent? Ionic?
- Nonpolar covalent: ________________________________
- Polar covalent: ________________________________
- Ionic: ____________________________
-
Draw a Lewis dot structure for the carbonate ion (CO32–) and its two other resonance structures.
_______________________________________
-
Predict the molecular geometries of the following molecules:
- PCl5: ________________________________
- MgF2: ________________________________
- AlF3: ________________________________
- UBr6: ________________________________
- SiH4: _________________________________
3.4 Intermolecular Forces
LEARNING OBJECTIVES
After Chapter 3.4, you will be able to:
- Order the intermolecular forces from strongest to weakest
- Describe what occurs during dipole-dipole, hydrogen bonding, and London dispersion force interactions
- Predict what intermolecular forces are possible for given interacting molecules:
Atoms and compounds participate in weak electrostatic interactions. The strength of these intermolecular forces can impact certain physical properties, such as melting and boiling points. The weakest of the intermolecular interactions are the dispersion forces, also known as London forces. Next are the dipole–dipole interactions, which are of intermediate strength. Finally, we have the strongest type of interaction, the hydrogen bond, which is a misnomer because there is no actual sharing or transfer of electrons. We must keep in mind, however, that even hydrogen bonds, the strongest of these interactions, only have about 10 percent of the strength of a covalent bond. Therefore, these electrostatic interactions can be overcome with small or moderate amounts of energy.
BRIDGE
These intermolecular forces are the bonding forces that keep a substance together in its solid or liquid state and determine whether two substances are miscible or immiscible in solution. Solutions and solubility are discussed in Chapter 9 of MCAT General Chemistry Review.
London Dispersion Forces
The bonding electrons in nonpolar covalent bonds may appear to be shared equally between two atoms, but at any point in time, they will be located randomly throughout the orbital. In a given moment, the electron density may be unequally distributed between the two atoms. This results in a rapid polarization and counterpolarization of the electron cloud and the formation of short-lived dipole moments. Subsequently, these dipoles interact with the electron clouds of neighboring compounds, inducing the formation of more dipoles. The momentarily negative end of one molecule will cause the closest region in any neighboring molecule to become temporarily positive itself. This causes the other end of the neighboring molecule to become temporarily negative, which in turn induces other molecules to become temporarily polarized, and the cycle begins again. The attractive or repulsive interactions of these short-lived and rapidly shifting dipoles are known as London dispersion forces, a type of van der Waals force.
Dispersion forces are the weakest of all of the intermolecular interactions because they are the result of induced dipoles that change and shift moment to moment. They do not extend over long distances and are, therefore, significant only when molecules are in close proximity. The strength of the London force also depends on the degree and ease by which the molecules can be polarized—that is, how easily the electrons can be shifted around. Large molecules are more easily polarizable than comparable smaller molecules and thus possess greater dispersion forces.
REAL WORLD
While dispersion forces (a type of van der Waals force) are the weakest of the intermolecular attractions, when there are millions of these interactions there is an amazing power of adhesion. This is demonstrated by geckos’ feet; the animal’s ability to climb smooth, vertical, and even inverted surfaces is due to dispersion forces.
Despite their weak nature, don’t underestimate the importance of dispersion forces. If it weren’t for them, the noble gases would not liquefy at any temperature because no other intermolecular forces exist between the noble gas atoms. The low temperatures at which noble gases liquefy are indicative of the very small magnitude of the dispersion forces between the atoms.
Dipole–Dipole Interactions
Polar molecules tend to orient themselves in such a way that the oppositely charged ends of the respective molecular dipoles are closest to each other: the positive region of one molecule is close to the negative region of another molecule. This arrangement is energetically favorable because an attractive electrostatic force is formed between the two molecules. This attractive force is denoted by dashed lines in most molecular notations and indicates a temporary bonding interaction, as shown in Figure 3.14.
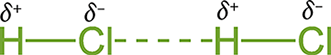
Figure 3.14. Dipole–Dipole Interactions in HCl
Dipole–dipole interactions are present in the solid and liquid phases but become negligible in the gas phase because of the significantly increased distance between gas particles. Polar species tend to have higher melting and boiling points than nonpolar species of comparable molecular weight due to these interactions. Realize that London forces and dipole–dipole interactions are different not in kind but in duration. Both are electrostatic forces between opposite partial charges; the difference is only in the transience or permanence of the molecular dipole.
BRIDGE
In organic chemistry, carbonyl groups possess distinct dipoles that facilitate nucleophilic attacks. This is the focus of almost all of the reactions in Chapters 6 to 9 of MCAT Organic Chemistry Review.
Hydrogen Bonds
Hydrogen bonds are a favorite topic on the MCAT. A hydrogen bond is a specific, unusually strong form of dipole–dipole interaction that may be intra- or intermolecular. Hydrogen bonds are not actually bonds—there is no sharing or transferring of electrons between two atoms. When hydrogen is bonded to one of three highly electronegative atoms—nitrogen, oxygen, or fluorine—the hydrogen atom carries only a small amount of the electron density in the covalent bond.
The hydrogen atom essentially acts as a naked proton. The positively charged hydrogen atom interacts with the partial negative charge of fluorine, oxygen, or nitrogen on nearby molecules. Substances that display hydrogen bonding tend to have unusually high boiling points compared to compounds of similar molecular weights that do not exhibit hydrogen bonding. The difference derives from the energy required to break the hydrogen bonds. Hydrogen bonding, shown in Figure 3.15, is particularly important in the behavior of water, alcohols, amines, and carboxylic acids.
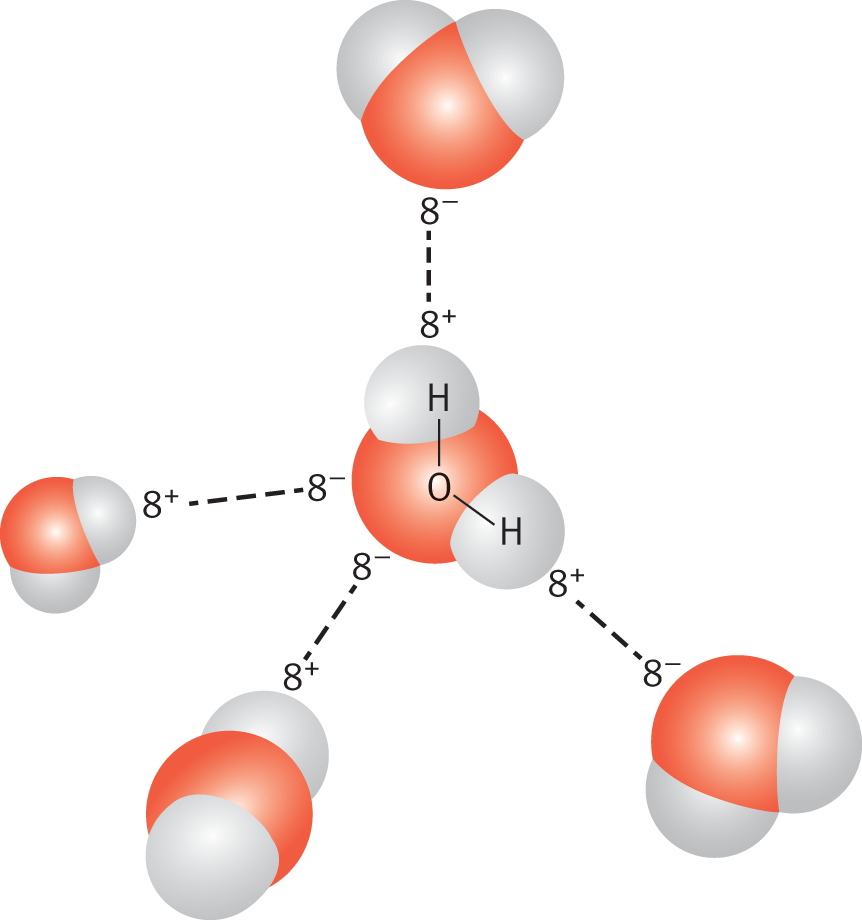
Figure 3.15. Hydrogen Bonding in Water
MNEMONIC
Hydrogen bonds: Pick up the FON (phone):
Hydrogen bonds exist in molecules containing a hydrogen bonded to Fluorine, Oxygen, or Nitrogen.
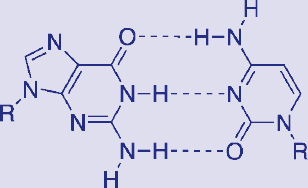
Many biochemical molecules, such as nucleotides, have different regions that are stabilized by hydrogen bonding, as shown in Figure 3.16. It is not an overstatement to say that, were it not for water’s ability to form hydrogen bonds and exist in the liquid state at room temperature, we would not exist (at least not in the form we recognize as “human”).
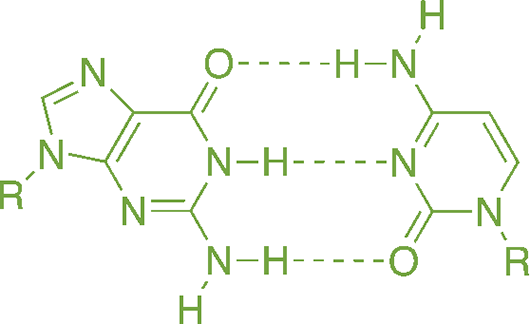
Figure 3.16. Hydrogen Bonding between Guanine and Cytosine
MCAT CONCEPT CHECK 3.4:
Before you move on, assess your understanding of the material with these questions.
-
Rank the major intermolecular forces from strongest to weakest:
- ____________________
- ____________________
- ____________________
-
Describe what occurs during dipole–dipole interactions.
______________________________________
-
In order to exhibit hydrogen bonding, what must be true of a given molecule?
_______________________________________
Conclusion
This chapter built on our knowledge of the atom and the trends demonstrated by the elements in the periodic table to explain the different ways by which atoms partner together to form compounds, either by exchanging electrons to form ions, which are then held together by electrostatic attractions between opposite charges; or by sharing electrons to form covalent bonds. We discussed the nature and characteristics of covalent bonds, noting their relative lengths and energies, as well as polarities. A review of Lewis dot structures and VSEPR theory will prepare you for predicting likely bond arrangements, resonance structures, and molecular geometries. Finally, we compared the relative strengths of the most important intermolecular electrostatic interactions, noting that even the strongest of these—hydrogen bonding—is still much weaker than an actual covalent bond. The next time you’re “browning” some of your food in a pan or the oven, take a moment to consider what’s happening at the atomic and molecular level. It’s not just cooking; it’s science!
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Bonding
- Chemical bonds can be ionic or covalent.
- Elements will form bonds to attain a noble gas-like electron configuration.
- The octet rule states that elements will be most stable with eight valence electrons. However, there are many exceptions to this rule:
- Elements with an incomplete octet are stable with fewer than eight electrons and include H, He, Li, Be, and B.
- Elements with an expanded octet are stable with more than eight electrons and include all elements in period 3 or greater.
- Compounds with an odd number of electrons cannot have eight electrons on each element.
Ionic Bonds
- An ionic bond is formed via the transfer of one or more electrons from an element with a relatively low ionization energy to an element with a relatively high electron affinity.
- Ionic bonds occur between elements with large differences in electronegativity (ΔEN > 1.7), usually between metals and nonmetals.
- A positively charged ion is called a cation. A negatively charged ion is called an anion.
- The resulting electrostatic attraction between the ions causes them to remain in close proximity, forming the bond.
- Ionic compounds form crystalline lattices—large, organized arrays of ions.
- Ionic compounds have unique physical and chemical properties.
- Ionic compounds tend to dissociate in water and other polar solvents.
- Ionic solids tend to have high melting points.
Covalent Bonds
- Acovalent bond is formed via the sharing of electrons between two elements of similar electronegativities.
- Bond order refers to whether a covalent bond is a single bond, double bond, or triple bond. As bond order increases, bond strength increases, bond energy increases, and bond length decreases.
- Covalent bonds can be categorized as nonpolar or polar based on the nature of the elements involved.
- Nonpolar bonds result in molecules in which both atoms have exactly the same electronegativity; some bonds are considered nonpolar when there is a very small difference in electronegativity between the atoms (ΔEN < 0.5), even though they are technically slightly polar.
- Polar bonds form when there is a significant difference in electronegativities (ΔEN = 0.5 to 1.7), but not enough to transfer electrons and form an ionic bond. In a polar bond, the more electronegative element takes on a partial negative charge, and the less electronegative element takes on a partial positive charge.
- Coordinate covalent bonds result when a single atom provides both bonding electrons while the other atom does not contribute any; coordinate covalent bonds are most often found in Lewis acid–base chemistry.
- Lewis dot symbols are a chemical representation of an atom’s valence electrons.
- Drawing a complete Lewis dot structure requires a balance of valence, bonding, and nonbonding electrons in a molecule or ion.
- Formal charges exist when an atom is surrounded by more or fewer valence electrons than it has in its neutral state (assuming equal sharing of electrons in a bond).
- For any molecule with a π (pi) system of electrons, resonance structures exist; these represent all of the possible configurations of electrons—stable and unstable—that contribute to the overall structure.
- The valence shell electron pair repulsion (VSEPR) theory predicts the three-dimensional molecular geometry of covalently bonded molecules. In this theory, electrons—whether bonding or nonbonding—arrange themselves to be as far apart as possible from each other in three-dimensional space, leading to characteristic geometries.
- Nonbonding electrons exert more repulsion than bonding electrons because they reside closer to the nucleus.
- Electronic geometry refers to the position of all electrons in a molecule, whether bonding or nonbonding. Molecular geometry refers to the position of only the bonding pairs of electrons in a molecule.
- The polarity of molecules is dependent on the dipole moment of each bond and the sum of the dipole moments in a molecular structure.
- All polar molecules contain polar bonds.
- Nonpolar molecules may contain nonpolar bonds, or polar bonds with dipole moments that cancel each other.
- σ and π bonds describe the patterns of overlap observed when molecular bonds are formed.
- Sigma(σ)bonds are the result of head-to-head overlap.
- Pi(π) bonds are the result of the overlap of two parallel electron cloud densities.
Intermolecular Forces
- Intermolecular forces are electrostatic attractions between molecules. They are significantly weaker than covalent bonds (which are weaker than ionic bonds).
- London dispersion forces are the weakest interactions, but are present in all atoms and molecules. As the size of the atom or structure increases, so does the corresponding London dispersion force.
- Dipole–dipole interactions, which occur between the oppositely charged ends of polar molecules, are stronger than London forces; these interactions are evident in the solid and liquid phases but negligible in the gas phase due to the distance between particles.
- Hydrogen bonds are a specialized subset of dipole–dipole interactions involved in intra- and intermolecular attraction; hydrogen bonding occurs when hydrogen is bonded to one of three very electronegative atoms—fluorine, oxygen, or nitrogen.
ANSWERS TO CONCEPT CHECKS
**3.1**
- Ionic bonds form between ions and involve gain or loss of electrons. Covalent bonds occur when electrons are shared between atoms.
- Any three examples that form incomplete octets (H, He, Li, Be, B) or expanded octets (Period 3 and greater) are acceptable.
- The polarity in a covalent bond is determined by differences in electronegativity between the two atoms involved.
**3.2**
- Metals lose electrons because they have low ionization energies, while nonmetals gain electrons because they have high electron affinities. These processes are complementary, leading to the formation of an ionic bond.
- Some characteristics of ionic compounds include high melting and boiling points due to electrostatic attractions, solubility of ions in water due to interactions with polar solvents, good conductors of heat and electricity, crystal lattice arrangement to minimize repulsive forces, and large electronegativity differences between ions, among other possible answers.
**3.3**
- Bond strength is defined by the electrostatic attraction between nuclei and electrons; multiple bonds (higher bond order) increases strength. Bond length is a consequence of these attractions. The stronger the bond, the shorter it is. Bond energy is the minimum amount of energy needed to break a bond. The stronger the bond, the higher the bond energy.
- Nonpolar covalent bonds form with ΔEN = 0 to 0.5. Polar covalent bonds form with ΔEN = 0.5 to 1.7. Ionic bonds form with ΔEN = 1.7 or higher.
-

- PCl5: trigonal bipyramidal, MgF2: linear, AlF3: trigonal planar, UBr6: octahedral, SiH4: tetrahedral
**3.4**
- Hydrogen bonding > dipole–dipole interactions > dispersion (London) forces
- A dipole consists of a segment of a molecule with partial positive and partial negative regions. The positive end of one molecule is attracted to the negative end of another molecule, and vice-versa.
- To experience hydrogen bonding, a molecule must contain a hydrogen bonded to a very electronegative atom (nitrogen, oxygen, or fluorine).
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. B
Carbon dioxide, CO2, has a double bond between carbon and each of its oxygen atoms. In polar covalent bonds, the difference in electronegativity between the bonded atoms is great enough to cause electrons to move disproportionately toward the more electronegative atom; hence the bonds are polar. But the electronegativity difference is not great enough to completely transfer electrons to the more electronegative atom; hence the bonds are covalent. Because oxygen has somewhat greater electronegativity compared to carbon, the bonds of carbon dioxide are polar covalent.
2. B
To answer this question, one must understand the contribution of resonance structures to average formal charge. In (B), there are three possible resonance structures. Each of the three oxygen atoms carries a formal charge of −1 in two out of the three structures. This averages to approximately − 2 3 charge on each oxygen atom, which is more negative than in the other answer choices. Both water and formaldehyde, (A) and (D), have no formal charge on the oxygen. Ozone, (C), has a − 1 2 on two of the three oxygens and a +1 charge on the central oxygen.
3. C
This question is asking for an element that reliably follows the octet rule. There are only a few elements that do not break the octet rule, including carbon, nitrogen, oxygen, fluorine, sodium, and magnesium. Thus, (C) is correct. To eliminate (A), (B), and (D), observe that the remaining elements can have other valences; Be is stable with a valence of four electrons, while both S and P can expand their octets.
4. D
The key to answering this question is to understand the types of intermolecular forces that exist in each of these molecules because larger intermolecular forces correspond to higher boiling points. Kr is a noble gas with a full octet, so the only intermolecular forces present are London dispersion forces, the weakest type of intermolecular forces. Acetone and isopropyl alcohol are both polar, so both have dipole–dipole interactions, which are stronger than dispersion forces. However, isopropyl alcohol can also form hydrogen bonds, increasing its boiling point. Finally, the strongest interactions are ionic bonds, which exist in potassium chloride.
5. C
The central carbon in carbonate has no lone pairs. It has three resonance structures, each of which involves a double bond between carbon and one of the three oxygens. Having made four bonds, carbon has no further orbitals for bonding or to carry lone pairs. This makes carbonate’s geometry trigonal planar. Alternatively, ClF3 also has three bonds; however, chloride still maintains two extra lone pairs. These lone pairs each inhabit one orbital, meaning that the central chloride must organize five items about itself: three bonds to fluorides and two lone pairs. The best configuration for maximizing the distance between all of these groups is trigonal bipyramidal. (A) and (B) are true statements but do not account for the difference in geometry.
6. B
Ionic bonds are formed between elements with large differences in electronegativity, i.e. between metals and nonmetals. Mg is a metal, whereas Cl, O, and H are all nonmetals. Since (B) contains a metal and nonmetal, it is correct. (A), (C), and (D) all describe covalent bonds. While HCl does ionize in aqueous solution, it exists as a polar covalent compound in the gaseous state.
7. D
Bond lengths decrease as the bond order increases, and they also decrease with larger differences in electronegativity. In this case, because both C2H2 and HCN have triple bonds, we cannot compare the bond lengths based on bond order. We must then rely on other periodic trends. The bond length decreases when moving to the right along the periodic table’s rows because more electronegative atoms have shorter atomic radii. The nitrogen in HCN is likely to hold its electrons closer, or in a shorter radius, than the carbons in C2H2.
8. C
Electronegative atoms bonded to hydrogen disproportionately pull covalently bonded electrons toward themselves, which leaves hydrogen with a partial positive character. That partial positive charge is attracted to nearby negative or partial negative charges, such as those on other electronegative atoms.
9. C
First recall that ammonium is NH4+, while ammonia is NH3. Ammonium is formed by the association of NH3, an uncharged molecule with a lone pair on the nitrogen, with a positively charged hydrogen cation. In other words, NH3 is a Lewis base, while H+ is a Lewis acid. This type of bonding between a Lewis acid and base is a coordinate covalent bond.
10. C
All atoms in the third period or greater have d-orbitals, which can hold an additional 10 electrons. The typical “octet” electrons reside in s- and p-orbitals, but elements in period 3 or higher can place electrons into these d-orbitals.
11. C
All of the listed types of forces describe interactions between different types of molecules. However, noble gases are entirely uncharged and do not have polar covalent bonds, ionic bonds, or dipole moments. Therefore, the only intermolecular forces experienced by noble gases are London dispersion forces. Although these interactions are small in magnitude, they are necessary for condensation into a liquid.
12. A
In this Lewis diagram, the phosphate molecule has an overall formal charge of −3. The four oxygen atoms would each be assigned a formal charge of −1. Given the overall charge of −3 and the −1 charge on each oxygen, the phosphorus must have a formal charge of +1.
13. C
The reaction in this question shows a water molecule, which has two lone pairs of electrons on the central oxygen, combining with a free hydrogen cation. The resulting molecule, H3O+ has formed a new bond between H+ and H2O. This bond is created via the sharing of one of oxygen’s lone pairs with the free H+ ion. This represents the donation of a shared pair of electrons from a Lewis base (H2O) to a Lewis acid (H+, electron acceptor). This type of bond is called a coordinate covalent bond.
14. C
NH3 has three hydrogen atoms bonded to the central nitrogen, which also has a lone pair. These four groups—three atoms, one lone pair—lead NH3 to assume tetrahedral electronic geometry yet trigonal pyramidal molecular geometry. The nitrogen in ammonia is sp3-hybridized. By hybridizing all three p-orbitals and the one s-orbital, four groups are arranged about the central atom, maximizing the distances between the groups to minimize the energy of the molecule with a tetrahedral configuration. In contrast, BF3 has three atoms and no lone pairs, resulting in sp2-hybridization. Its shape is called trigonal planar.
15. B
When a compound melts, the interactions between molecules must be broken to a sufficient extent to enable movement. In NaCl, these interactions are ionic. On the other hand, solids made of molecules of sucrose, glycerol, or water are held together by hydrogen bonds. Due to the greater electrostatic attraction between the full charges of ionic bonds, the ionic bonds of NaCl are significantly stronger than hydrogen bonds of other molecules. Thus, NaCl has the highest melting point, consistent with (B).
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(3.1) Dipole moment: p = qd
(3.2) Formal charge: V − N nonbonding − 1 2 N bonding
SHARED CONCEPTS
General Chemistry Chapter 1
Atomic Structure
General Chemistry Chapter 2
The Periodic Table
General Chemistry Chapter 4
Compounds and Stoichiometry
Organic Chemistry Chapter 3
Bonding
Organic Chemistry Chapter 4
Analyzing Organic Reactions
Physics and Math Chapter 5
Electrostatics and Magnetism