Chapter 4: Compounds and Stoichiometry

Chapter 4: Compounds and Stoichiometry
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of the following best describes ionic compounds?
- Ionic compounds are formed from molecules containing two or more atoms.
- Ionic compounds are formed of charged particles and are measured by molecular weight.
- Ionic compounds are formed of charged particles that share electrons equally.
- Ionic compounds are three-dimensional arrays of charged particles.
-
Which of the following compounds has a formula weight between 74 and 75 grams per mole?
- KCl
- C4H10O
- MgCl2
- BF3
-
Which of the following is the gram equivalent weight of H2SO4 with respect to protons?
- 49.1 g
- 98.1 g
- 147.1 g
- 196.2 g
-
Which of the following molecules CANNOT be expressed by the empirical formula CH?
- Benzene
- Ethyne
-
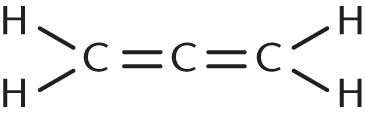
-
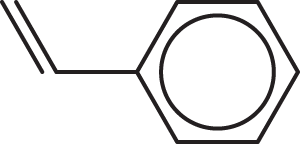
-
In which of the following compounds is the percent composition of carbon by mass closest to 62 percent?
- Acetone
- Ethanol
- Propane
- Methanol
-
What is the most specific characterization of the reaction shown?
Ca(OH)2 (aq) + H2SO4 (aq) → CaSO4 (aq) + H2O (l)
- Single-displacement
- Neutralization
- Double-displacement
- Oxidation–reduction
-
In the reaction shown, if 39.05 g of Na2S are reacted with 85.5 g of AgNO3, how much of the excess reagent will be left over once the reaction has gone to completion?
Na2S + 2 AgNO3 → Ag2S + 2 NaNO3
- 19.5 g Na2S
- 26.0 g Na2S
- 41.4 g AgNO3
- 74.3 g AgNO3
-
Using a given mass of KClO3, how would one calculate the mass of oxygen produced in the following reaction, assuming it goes to completion?
2 KClO3 → 2 KCl + 3 O2
- ( grams KClO 3 consumed ) ( 3 moles O 2 ) ( molar mass O 2 ) ( molar mass KCl O 3 ) ( 2 moles KCl O 3 )
- (grams KClO 3 consumed) ( molar mass O 2 ) ( molar mass KClO 3 ) ( 2 moles KClO 3 )
- ( molar mass KClO 3 ) ( 2 moles KClO 3 ) (grams KClO 3 consumed) ( molar mass O 2 )
- (grams KClO 3 consumed)(3 moles O 2 ) (molar mass KClO 3 )(2 moles KClO 3 )(molar mass O 2 )
-
Aluminum metal can be used to remove tarnish from silver when the two solid metals are placed in water, according to the following reaction:
3 AgO + 2 Al → 3 Ag + Al2O3
This reaction is a:
- double-displacement reaction.
- single-displacement reaction.
- oxidation–reduction reaction.
- combination reaction.
- II only
- IV only
- I and III only
- II and III only
-
Which of the following types of reactions generally have the same number of reactants and products?
- Double-displacement reactions
- Single-displacement reactions
- Combination reactions
- I only
- II only
- I and II only
- II and III only
-
Diethyl zinc is a very unstable compound. When exposed to air, it reacts with oxygen to produce zinc oxide, carbon dioxide, and water. The unbalanced reaction is shown below.
Z n ( C 2 H 5 ) 2 + O 2 → Z n O + C O 2 + H 2 O
What is the coefficient for carbon dioxide in the balanced reaction?
- 4
- 5
- 6
- 7
- In the process of photosynthesis, carbon dioxide and water combine with energy to form glucose and oxygen, according to the following equation:
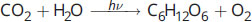
What is the theoretical yield of glucose if 30 grams of water are reacted with excess carbon dioxide and energy, according to the equation above?
- 30.0 g
- 50.0 g
- 300.1 g
- 1801 g
-
In the following reaction:
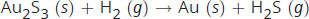
If 2 moles of Au2S3 (s) is reacted with 5 moles of hydrogen gas, what is the limiting reagent?
- Au2S3 (s)
- H2 (g)
- Au (s)
- H2S (g)
-
Which of the following would make the strongest electrolytic solution?
- A nonpolar covalent compound with significant solubility.
- An ionic compound composed of one cation with +3 charge and three anions with −1 charge.
- A polar covalent compound with a small dissociation constant.
- An ionic compound composed of two cations with +1 charge and one anion with −2 charge.
-
Carbon disulfide and nitrous oxide, when ignited, react exothermically to produce elemental sulfur, nitrogen, and carbon dioxide. Since this reaction produces a “woof” sound, it is sometimes referred to as the Barking Dog reaction. Which of the following represents the balanced chemical reaction?
- 8 N2O + 4 CS2 → S8 + 4 CO2 + 8 N2
- 6 N2O + 4 CS2 → S8 + 4 CO2 + 6 N2
- 8 N2O + 2 CS2 → S8 + 2 CO2 + 8 N2
- 7 N2O + 5 CS2 → S8 + 4 CO2 + 7 N2
Answer Key
- D
- A
- A
- C
- A
- B
- A
- A
- D
- C
- A
- B
- B
- B
- A
Chapter 4: Compounds and Stoichiometry
CHAPTER 4
COMPOUNDS AND STOICHIOMETRY
In This Chapter
4.1 Molecules and Moles
Molecular Weight
Mole
Equivalent Weight
4.2 Representation of Compounds
Law of Constant Composition
Empirical and Molecular Formulas
Percent Composition
4.3 Types of Chemical Reactions
Combination Reactions
Decomposition Reactions
Combustion Reactions
Single-Displacement Reactions
Double-Displacement Reactions
Neutralization Reactions
4.4 Balancing Chemical Equations
4.5 Applications of Stoichiometry
Limiting Reagent
Yield
4.6 Ions
Cations and Anions
Ion Charges
Electrolytes
Concept Summary
CHAPTER PROFILE
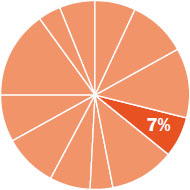
The content in this chapter should be relevant to about 7% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
4C: Electrochemistry and electrical circuits and their elements
4E:Atoms, nuclear decay, electronic structure, and atomic chemical behavior
5A: Unique nature of water and its solutions
Introduction
Oh—what is that smell? It smells like rancid almonds. Then you notice a few green bugs whose backs give the impression of a shield. Stink bugs! A stink bug “stinks” because it produces a highly concentrated solution of volatile compounds that we perceive as malodorous, noxious, and irritating. Interestingly enough, the primary compounds in the stink bug’s stink bomb are hydrogen cyanide—a highly toxic compound that inhibits cytochromecoxidase, thereby blocking aerobic respiration—and benzaldehyde. Like many other aromatic compounds, benzaldehyde vaporizes at room temperature and reaches the olfactory system as gas particles. Benzaldehyde is also the key ingredient in artificial almond extract. At low concentrations, it produces a pleasant aroma of toasted almonds. However, at high concentrations, its odor is that of rotten almonds, and it is a noxious irritant to the skin, eyes, and respiratory tract.
Benzaldehyde is a compound composed of seven carbon atoms, six hydrogen atoms, and one oxygen atom. One mole of benzaldehyde has a mass of approximately 106 grams. It can react with other atoms or compounds to form new compounds—pure substances composed of two or more elements in a fixed proportion. Compounds can be broken down by chemical means to produce their constituent elements or other compounds. They are characterized by describing their physical and chemical properties.
This chapter focuses on compounds and their reactions. It reviews the various ways in which compounds are represented, using empirical and molecular formulas and percent composition. There is a brief overview of the major classes of chemical reactions, which we will examine more closely in subsequent chapters, and finally, there is a recap of the steps involved in balancing chemical equations with a particular focus on identifying limiting reagents and calculating reaction yields.
4.1 Molecules and Moles
LEARNING OBJECTIVES
After Chapter 4.1, you will be able to:
- Calculate the molar mass of a given substance, such as AgCN
- Calculate the number of moles of a molecule given its mass in grams
- Compare the number of molecules in two different compounds given their gram weights and molecular formulas
- Determine the normality of a solution
A molecule is a combination of two or more atoms held together by covalent bonds. Molecules are the smallest units of compounds that display their identifying properties. Molecules can be composed of two or more atoms of the same element (such as N2 and O2) or may be composed of atoms of different elements, as in CO2 (carbon dioxide), SOCl2 (thionyl chloride), and C6H5CHO (benzaldehyde). Because reactions usually involve a very large number of molecules—far too many to count individually—we usually measure amounts of compounds in terms of moles or grams, using molar mass to interconvert between these units.
Ionic compounds do not form true molecules because of the way in which the oppositely charged ions arrange themselves in the solid state. As solids, they can be considered as nearly infinite three-dimensional arrays of the charged particles that comprise the compound. As described in Chapter 3 of MCAT General Chemistry Review, solid NaCl is a coordinated lattice in which each of the Na+ ions is surrounded by Cl– ions and each of the Cl– ions is surrounded by Na+ ions. This makes it rather difficult to clearly define a sodium chloride molecule, and the term formula unit, representing the empirical formula of the compound, is used instead. Because no molecule actually exists, molecular weight becomes meaningless, and the term formula weight is used instead.
BRIDGE
Ionic compounds form from combinations of elements with large electronegativity differences, such as sodium and chlorine. Molecular compounds form from elements of similar electronegativity, such as carbon with oxygen. The difference between ionic and covalent bonds is discussed in Chapter 3 of MCAT General Chemistry Review.
Molecular Weight
Remember that the term atomic weight is a misnomer because it is actually a weighted average of the masses of the naturally occurring isotopes of an element, not their weights. The same applies here to our discussion of molecularweight. Molecular weight, then, is simply the sum of the atomic weights of all the atoms in a molecule, and its units are atomic mass units (amu) per molecule. Similarly, the formula weight of an ionic compound is found by adding up the atomic weights of the constituent ions according to its empirical formula, and its units are also amu per molecule.
Example: What is the molecular weight of SOCl2?
Solution: To find the molecular weight of SOCl2, add together the atomic weights of each of the atoms.
1 S: 1 × 32.1 amu = 32.1 amu
1 O: 1 × 16.0 amu = 16.0 amu
2 Cl: 2 × 35.5 amu = 71.0 amu
Total molecular weight = 119.1 amu per molecule
Mole
Amole is a quantity of any substance (atoms, molecules, dollar bills, kittens—anything) equal to the number of particles that are found in 12 grams of carbon-12 ( 6 12 C ). This number of particles is defined as Avogadro’s number (NA), 6.022 × 1023 mol–1. One mole of a compound has a mass in grams equal to the molecular or formula weight of the compound in amu. For example, one molecule of H2CO3 (carbonic acid) has a mass of 62 amu; one mole of the compound has a mass of 62 grams. The mass of one mole of a compound is called its molar mass and is usually expressed in g mol . The term molecular weight is sometimes used incorrectly to imply molar mass; remember, molecular weight is measured in amu molecule , not g mol .
KEY CONCEPT
Remember that Avogadro’s number (and the mole) are just units of convenience, like the dozen is a convenient unit for eggs.
The formula for determining the number of moles of a sample substance is:
Moles = Mass of sample (g) Molar mass ( g mol )
Equation 4.1
This equation is often used in stoichiometry and titration problems.
Example: How many moles are in 9.53 g of MgCl2?
Solution: First, find the molar mass of MgCl2. ( 1 × 24.3 g mol ) + ( 2 × 35.5 g mol ) = 95.3 g mol
Now, solve for the number of moles.
9 .53 g 95 .3 g mol = 0.10 mol MgCl 2
Equivalent Weight
Equivalent weight and the related concept of equivalents are a source of confusion for many students. Part of the problem may be the context in which equivalents and equivalent weights are usually discussed: acid–base reactions, oxidation–reduction reactions, and precipitation reactions, all three of which can be sources of confusion and anxiety on their own. Therefore, let’s start with a more basic discussion of equivalents.
Often, certain elements or compounds can act more potently than others in performing certain reactions. For example, one mole of HCl has the ability to donate one mole of hydrogen ions (H+) in solution, but one mole of H2SO4 has the ability to donate two moles of hydrogen ions, and one mole of H3PO4 has the ability to donate three moles of hydrogen ions. To gather one mole of hydrogen ions for a particular acid–base reaction, we could use one mole of HCl, a half-mole of H2SO4, or one-third of a mole of H3PO4. Or, consider the difference between Na and Mg: one mole of sodium has the ability to donate one mole of electrons, while one mole of magnesium has the ability to donate two moles of electrons. This provides context for the concept of equivalents: How many moles of the thing we are interested in (protons, hydroxide ions, electrons, or ions) will one mole of a given compound produce? Sodium will donate one mole of electrons (one equivalent), but magnesium will donate two moles of electrons (two equivalents).
BRIDGE
The idea of equivalents is related to the concept of normality, which is explained in the discussion of acids and bases in Chapter 10 of *MCAT General** Chemistry Review*.
So far, this discussion has been focused on the mole-to-mole relationship between, say, an acid compound and the hydrogen ions it donates. However, sometimes we need to work in units of mass rather than moles. Just as one mole of HCl will donate one mole of hydrogen ions, a certain mass of HCl (about 36.5 g) will also donate one equivalent of hydrogen ions. This amount of a compound, measured in grams, that produces one equivalent of the particle of interest is called the gram equivalent weight and can be calculated from:
Gram equivalent weight = Molar mass n
Equation 4.2
where n is the number of particles of interest produced or consumed per molecule of the compound in the reaction. For example, one would need 31 grams of H2CO3 ( molar mass = 62 g mol ) to produce one equivalent of hydrogen ions because each molecule of H2CO3 can donate two hydrogen ions (n = 2). Simply put, the equivalent weight of a compound is the mass that provides one mole of the particle of interest.
MCAT EXPERTISE
Whenever confronting a stoichiometry problem, always look for normality by identifying an equivalent unit (protons, hydroxide ions, electrons, ions) and then multiplying it by the number of moles or molar concentration to find the normal concentration.
If the amount of a compound in a reaction is known and we need to determine how many equivalents are present, use the equation:
Equivalents = Mass of compound ( g ) Gram equivalent weight ( g )
Equation 4.3
Finally, we can now introduce the measurement of normality. Normality (N) is a measure of concentration, given in the units equivalents L . On the MCAT, it is most commonly used for hydrogen ion concentration. Thus, a 1 N solution of acid contains a concentration of hydrogen ions equal to 1 mole per liter; a 2 N solution of acid contains a concentration of hydrogen ions equal to 2 moles per liter. The actual concentration of the acidic compound may be the same or different from the normality because different compounds are able to donate different numbers of hydrogen ions. In a 1 N HCl solution, the molarity of HCl is 1 M because HCl is a monoprotic acid; in a 1 N H2CO3 solution, the molarity of H2CO3 is 0.5 M because H2CO3 is a diprotic acid. Note that normality calculations always assume that a reaction will proceed to completion; while carbonic acid does not fully dissociate in solution, it can be reacted with enough base for each molecule to give up both of its protons. The conversion from normality to molarity of a given solute is:
Molarity = Normality n
Equation 4.4
where n is the number of protons, hydroxide ions, electrons, or ions produced or consumed by the solute.
Figure 4.1 shows the titration of the diprotic acid H2CO3 with a base. The x-axis indicates that two equivalents of base are needed to neutralize both protons of this acid.
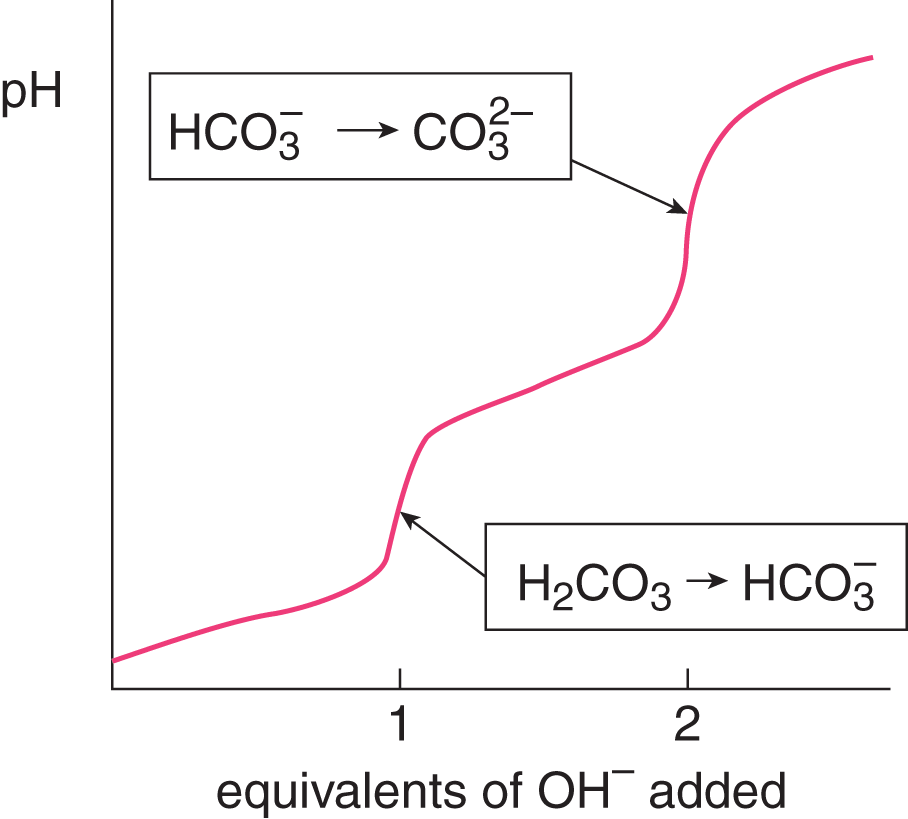
Figure 4.1. Titration of Carbonic Acid with a Base Carbonic acid is diprotic, so two equivalents of base are required to neutralize both protons of the acid.
There is a real benefit to working with equivalents and normality because it allows a direct comparison of the quantities of the entity we are most interested in. In an acid–base reaction, we care about the hydrogen or hydroxide ions; where the ions come from is not really the primary concern. It is convenient to be able to say that one equivalent of acid (hydrogen ions) will neutralize one equivalent of base (hydroxide ions), but the same could not necessarily be said if we were dealing with moles of acidic compounds and moles of basic compounds. For example, one mole of HCl will not completely neutralize one mole of Ca(OH)2 because one mole of HCl will donate one equivalent of acid, but Ca(OH)2 will donate two equivalents of base.
Example: What is the gram equivalent weight (GEW) of sulfuric acid?
Solution: First, find the molar mass of H2SO4.
( 2 × 1.0 g mol H ) + ( 1 × 32.1 g mol S ) + ( 4 × 16.0 g mol O ) = 98.1 g mol H 2 SO 4
Next, identify the equivalents: protons (H+), because these are transferred in acid–base reactions. The number of protons in sulfuric acid (n) is 2.
Now, calculate the gram equivalent weight.
Gram equivalent weight = Molar mass n GEW = 98.1 g mol H 2 SO 4 2 mol H + mol H 2 SO 4 = 49.05 g mol H +
KEY CONCEPT
In acid–base chemistry, the gram equivalent weight represents the mass of acid that yields one mole of protons, or the mass of base that yields one mole of hydroxide ions.
Example: What is the normality of a 2 M Mg(OH)2 solution?
Solution: First, identify the number of equivalents (n). There are two hydroxide ions (OH–) for each molecule of Mg(OH)2, which is the equivalent of interest because magnesium hydroxide is a base.
Then, calculate the normality. Normality = molarity × n = 2 M × 2 equiv OH − mol Mg(OH) 2 = 4 N Mg ( OH ) 2
MCAT CONCEPT CHECK 4.1:
Before you move on, assess your understanding of the material with these questions.
-
Calculate the molar masses of the following substances:
- NaBr: ________________________________
- SrCl2: ________________________________
- C6H12O6: ____________________________
-
Calculate the number of moles in 100 g of each of the following substances:
- NaBr: ________________________________
- SrCl2: ________________________________
- C6H12O6: ____________________________
-
How do the number of molecules in 18 g of H2O compare to the number of formula units in 58.5 g of NaCl?
_______________________________________
-
Determine the normality of the following solutions: (Note: The species of interest is H+.)
- 0.25 M H3PO4: __________________________________
- 95 g PO43− in 100 mL solution: __________________________________
4.2 Representation of Compounds
LEARNING OBJECTIVES
After Chapter 4.2, you will be able to:
- Recall the similarities and differences between molecular and empirical formulas
- Calculate the percent composition by mass of a compound, such as C6H12O6
- Determine the empirical formula of a compound given its percent composition by mass
There are different ways of representing compounds and their constituent atoms. We’ve already reviewed a couple of these systems in Chapter 3 of MCAT General Chemistry Review: Lewis dot structures and VSEPR theory. In organic chemistry, it is common to encounter skeletal representations of compounds, called structuralformulas, that show the various bonds between the constituent atoms of a compound. Inorganic (general) chemistry typically represents compounds by showing the constituent atoms without representing the actual bond connectivity or atomic arrangement. For example, the formula C6H12O6 (glucose) tells us that this particular compound consists of six atoms of carbon, twelve atoms of hydrogen, and six atoms of oxygen, but there is no indication of how the different atoms are arranged or how many bonds exist between each of the atoms.
BRIDGE
Many of these representations are discussed in more detail in Chapter 2 of MCAT Organic Chemistry Review. Understanding the theory behind such representations will help convert between different projections and representations with ease.
Law of Constant Composition
The law of constant compositionstates that any pure sample of a given compound will contain the same elements in an identical mass ratio. For example, every sample of water will contain two hydrogen atoms for every one oxygen atom, or—in terms of mass—for every one gram of hydrogen, there will be eight grams of oxygen.
REAL WORLD
Even biologically important molecules, such as water and amino acids on Earth are, by composition, the same anywhere else in the universe, even though densities and other physical properties may differ.
Empirical and Molecular Formulas
There are two ways to express the formula of a compound. The empirical formula gives the simplest whole-number ratio of the elements in the compound. The molecular formula gives the exact number of atoms of each element in the compound and is a multiple of the empirical formula. For example, the empirical formula for benzene is CH, while the molecular formula is C6H6. For some compounds, the empirical and molecular formulas are identical, as is the case for H2O. As previously discussed, ionic compounds, such as NaCl or CaCO3, will only have empirical formulas.
BRIDGE
An empirical formula of CH2O is indicative of a monosaccharide. Common monosaccharides include glucose, fructose, and galactose. The structures of these monosaccharides—and of carbohydrates in general—are discussed in Chapter 4 of MCAT Biochemistry Review.
Percent Composition
The percent composition of an element (by mass) is the percent of a specific compound that is made up of a given element. To determine the percent composition of an element in a compound, the following formula is used:
Percent composition = Mass of element in formula Molar mass × 100 %
Equation 4.5
One can calculate the percent composition of an element by using either the empirical or the molecular formula. It is also possible to determine the molecular formula given both the percent compositions and molar mass of a compound. The following examples demonstrate such calculations.
MCAT EXPERTISE
Percent composition is a common way for stoichiometry to be tested on the MCAT. Practice these problems to build up speed and efficiency for Test Day.
Example: What is the percent composition of chromium in K2Cr2O7?
Solution: The molar mass of K2Cr2O7 is:
( 2 × 39.1 g mol ) + ( 2 × 52.0 g mol ) + ( 7 × 16.0 g mol ) ≈ ( 2 × 40 ) + ( 2 × 50 ) + ( 7 × 16 ) = 292 g mol ( actual value = 294.2 g mol )
Calculate the percent composition of Cr:
Percent composition = 2 × 52 .0 g mol 294 .2 g mol × 100 % ≈ 2 × 50 300 × 100 % = 100 300 × 100 % = 33 % (actual value = 35.4 %)
Example: What are the empirical and molecular formulas of a carbohydrate that contains 40.9% carbon, 4.58% hydrogen, and 54.52% oxygen and has a molar mass of 264 g mol ?
Method One: First, determine the number of moles of each element in the compound by assuming a 100-gram sample; this converts the percentage of each element present directly into grams of that element. Then convert grams to moles:
moles C = 40 .9 g 12 g mol ≈ 3.4 mol moles H = 4.58 g 1 g mol ≈ 4.6 mol moles O = 54.52 g 16 g mol ≈ 3.4 mol
Next, find the simplest whole number ratio of the elements by dividing the number of moles for each compound by the smallest number out of all obtained in the previous step.
C: 3.4 3.4 = 1.00 ; H : 4.6 3.4 ≈ 1.33 ; O: 3.4 3.4 = 1.00
Finally, the empirical formula is obtained by converting the numbers obtained into whole numbers by multiplying them by an integer value.
Empirical formula = C1H1.33O1 × 3 = C3H4O3
To determine the molecular formula, divide the molar mass (264 g/mol, given in the question stem) by the empirical formula weight. The resulting value gives the number of empirical formula units in the molecular formula.
The formula weight of the empirical formula C3H4O3 is:
( 3 × 12.0 g mol C ) + ( 4 × 1.0 g mol H ) + ( 3 × 16.0 g mol O ) = 88 g mol total ratio of molecular formula empirical formula = 264 g mol 88 g mol = 3
Finally, find the molecular formula by multiplying by this ratio:
C3H4O3 × 3 = C9H12O9. The molecular formula is C9H12O9.
Method Two: When the molar mass is given, it is generally easier to find the molecular formula first. This is accomplished by multiplying the molar mass by the given percentages to find the mass of each element present in one mole of compound, then dividing by the respective atomic weights to find the mole ratio of the elements: moles C = ( 0.409 ) ( 264 g ) 12 g mol ≈ ( 0 . 4 ) ( 270 ) 12 ≈ 270 30 = 9 mol moles H = ( 0.0458 ) ( 264 g ) 1 g mol ≈ ( 0 . 05 ) ( 270 ) 1 ≈ 13 mol moles O = ( 0.5452 ) ( 264 g ) 16 g mol ≈ ( 0 . 5 ) ( 270 ) 16 ≈ 270 30 = 9 mol
At first glance, this gives a molecular formula of C9H13O9. However, familiarity with carbohydrates indicates that a molecular formula of C9H12O9 fits the ratio CH2O, and takes rounding error into account. The empirical formula can now be found by reducing the subscript ratio to the simplest integer values (C3H4O3).
MCAT EXPERTISE
When there are two methods for approaching a problem, be well-versed in both. Knowing multiple ways to solve a problem will help you tackle questions efficiently.
KEY CONCEPT
The molecular formula is either the same as the empirical formula or a multiple of it. To calculate the molecular formula, you need to know the mole ratio (this will give you the empirical formula) and the molar mass (molar mass divided by empirical formula weight will give the multiplier for the empirical formula-to-molecular formula conversion).
MCAT CONCEPT CHECK 4.2:
Before you move on, assess your understanding of the material with these questions.
-
What are some similarities and differences between molecular and empirical formulas?
- Similarities: ___________________________
- Differences: ____________________________
-
Find the percent composition (by mass) of sodium, carbon, and oxygen in sodium carbonate (Na2CO3):
- Sodium: ____________________________
- Carbon: ____________________________
- Oxygen: ____________________________
-
Experimental data from the combustion of an unknown compound indicates that it is 28.5% iron, 24.0% sulfur, and 49.7% oxygen by mass. What is its empirical formula?
_______________________________________
_______________________________________
_______________________________________
4.3 Types of Chemical Reactions
LEARNING OBJECTIVES
After Chapter 4.3, you will be able to:
- Describe the series of events in a single displacement, double displacement, neutralization, or combustion reaction
- Classify a reaction and predict its products given the reactants: CH 4 + 2 O 2 →
BRIDGE
Many of the reactions we will discuss here have analogs in MCAT Organic Chemistry Review and MCAT Biochemistry Review. Be sure to understand the relationships between the products and reactants because it will help simplify more advanced reactions.
This section reviews the major classes of chemical reactions. We will begin with a classification of major types of reactions seen on the MCAT and then discuss methods to recognize their products. It is important to understand the conventions of reaction mechanisms. In the following section, we will discuss how to properly balance chemical equations.
Combination Reactions
A combination reaction has two or more reactants forming one product. The formation of water by burning hydrogen gas in air is an example of a combination reaction. This reaction is highlighted in Figure 4.2.
2 H2 (g) + O2 (g) → 2 H2O (g)
Figure 4.2. Formation of Water from Hydrogen and Oxygen
KEY CONCEPT
Combination reactions have more reactants than products: A + B→C
Decomposition Reactions
A decompositionreaction is the opposite of a combination reaction: a single reactant breaks down into two or more products, usually as a result of heating, high-frequency radiation, or electrolysis. An example of decomposition is the breakdown of mercury(II) oxide. (The Δ [delta] sign over a reaction arrow represents the addition of heat.)
2 HgO ( s ) → Δ 2 Hg ( l ) + O 2 ( g )
KEY CONCEPT
Decomposition reactions generally have more products than reactants. A→ B + C
An example of a reaction that utilizes high-frequency light is the decomposition of silver chloride crystals, shown in Figure 4.3, in the presence of sunlight. The ultraviolet component of sunlight has sufficient energy to catalyze certain chemical reactions. For silver chloride, exposure to sunlight results in a decomposition reaction that yields a rust-colored product that consists of separated silver and chlorine.

Figure 4.3. Silver Chloride (AgCl) Crystals Silver chloride will decompose to a rust-colored product upon exposure to sunlight.
Combustion Reactions
A combustion reaction is a special type of reaction that involves a fuel—usually a hydrocarbon—and an oxidant (normally oxygen). In its most common form, these reactants form the two products of carbon dioxide and water. For example, the balanced equation expressing the combustion of methane is shown in Figure 4.4.
Figure 4.4. Combustion of Methane
KEY CONCEPT
Combustion involves oxidation (using O2 or similar) of a fuel (typically a hydrocarbon).
MCAT EXPERTISE
Combustion reactions are usually conducted with hydrocarbon fuels, but they can also use elements such as sulfur or other compounds such as sugars. The products can differ, but carbon dioxide and water are almost always present. Therefore, it is important to recognize the reactants and products of this reaction type because you may see it in various contexts.
Single-Displacement Reactions
A single-displacement reaction occurs when an atom or ion in a compound is replaced by an atom or ion of another element. For example, solid copper metal will displace silver ions in a clear solution of silver nitrate to form a blue copper nitrate solution and solid silver metal.
Cu (s) + AgNO3 (aq) → Ag (s) + CuNO3 (aq)
Single-displacement reactions are often further classified as oxidation–reduction reactions, which will be discussed in greater detail in Chapter 11 of MCAT General Chemistry Review. For example, Ag in AgNO3 has an oxidation state of +1, but when it leaves the compound, it gains one electron (the Ag+ is reduced to Ag). On the other hand, copper loses an electron (oxidation) when it joins the nitrate ion.
Double-Displacement Reactions
In double-displacement reactions, also called metathesis reactions, elements from two different compounds swap places with each other to form two new compounds. This type of reaction occurs when one of the products is removed from the solution as a precipitate or gas or when two of the original species combine to form a weak electrolyte that remains undissociated in solution. For example, when solutions of calcium chloride and silver nitrate are combined, insoluble silver chloride forms in a solution of calcium nitrate.
CaCl2 (aq) + 2 AgNO3 (aq) → Ca(NO3)2 (aq) + 2 AgCl (s)
A series of double-displacement reactions is depicted in Figure 4.5. Shown are illustrations of test tubes in which Zn(NO3)2 is dissolved in solutions to precipitate solid zinc salts. From left to right, the solutions are (NH4)2S, NaOH, and Na2CO3.
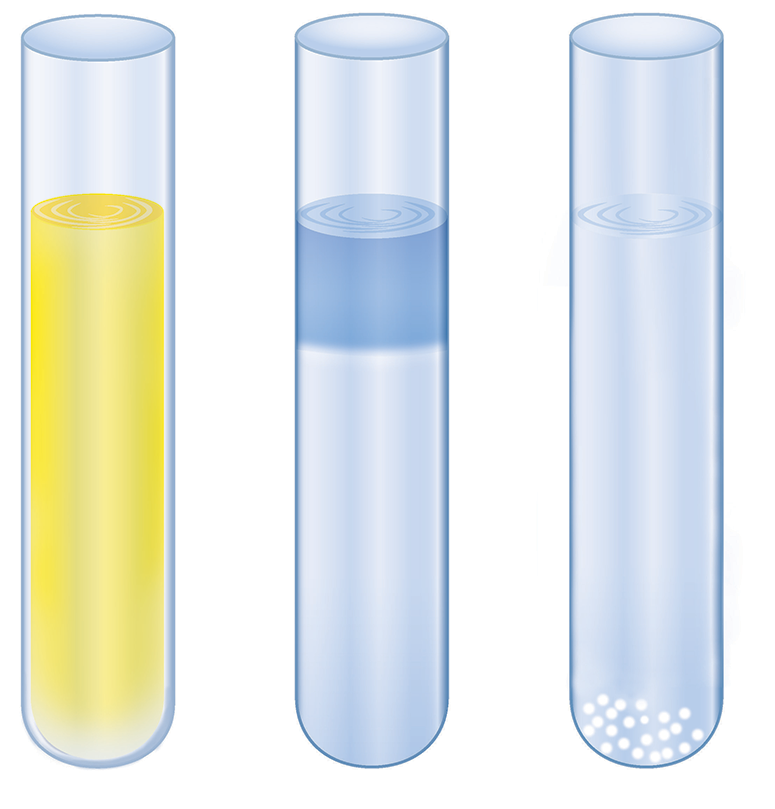
Figure 4.5. Illustration of Double-Displacement Reactions Forming Zinc Salts Left: (NH4)2S solution, producing ZnS (s). Middle: NaOH solution, producing Zn(OH)2 (s). Right: Na2CO3 solution, producing ZnCO3 (s).
Neutralization Reactions
Neutralization reactions are a specific type of double-displacement reaction in which an acid reacts with a base to produce a salt (and, usually, water). For example, hydrochloric acid and sodium hydroxide will react to form sodium chloride and water:
HCl (aq) + NaOH (aq) → NaCl (aq) + H2O (l)
BRIDGE
Acids and bases combine in neutralization reactions to produce salts (and, usually, water). Acid–base chemistry is discussed in Chapter 10 of MCAT General Chemistry Review.
Reactions between acids and bases are not always visible. The addition of an indicator or use of indicator strips, as shown in Figure 4.6, can determine when the reaction has occurred.

Figure 4.6. Indicator Strip Tested in Solutions of Varying pH After an indicator strip is placed in a solution, the indicator strip (right) can be read using the indicator key.
MCAT CONCEPT CHECK 4.3:
Before you move on, assess your understanding of the material with these questions.
-
Describe in words what occurs when Zn(NO3)2 is dissolved in (NH4)2S:
_____________________________
-
Complete and classify the most likely reactions in the table below:
Reactants Conditions Products Reaction Type
2 H2 + O2 →
Al(OH)3 + H3PO4 →
2 H2O → electricity
NaNO3 + CuOH →
Zn + AgCl →
4.4 Balancing Chemical Equations
LEARNING OBJECTIVES
After Chapter 4.4, you will be able to:
- Balance a chemical equation
Because chemical equations express how much and what types of reactants must be used to obtain a given quantity of product, it is of utmost importance that the reaction be balanced so as to reflect the laws of conservation of mass and charge. The mass of the reactants consumed must equal the mass of products generated. More specifically, one must ensure that the number of atoms of each element on the reactant side equals the number of atoms of that element on the product side. Stoichiometric coefficients, which are the numbers placed in front of each compound, are used to indicate the relative number of moles of a given species involved in the reaction. For example, the balanced equation expressing the combustion of nonane is:
C9H20 (g) + 14 O2 (g) → 9 CO2 (g) + 10 H2O (l)
The coefficients indicate that one mole of C9H20 gas must be reacted with fourteen moles of O2 gas to produce nine moles of carbon dioxide and ten moles of water. In general, stoichiometric coefficients are given as whole numbers.
MCAT EXPERTISE
It is unlikely that you will come across a question that explicitly asks you to balance an equation. However, you will need to recognize unbalanced reactions and quickly add the necessary coefficients. To balance a reaction, look at the number of atoms of each element and the charge on both sides (especially for oxidation–reduction reactions).
The steps taken to balance a chemical reaction are necessary to ensure that calculations regarding the reaction are performed correctly. Let’s review the steps involved in balancing a chemical equation, using an example.
Example: Balance the following reaction:
C4H10 (l) + O2 (g) → CO2 (g) + H2O (l)
Method One: First, balance the carbons (4 on reactant side) in the products. Carbons are a good choice to start with because they appear only once on both sides of the reaction:
C4H10 (l) + O2 (g) → 4 CO2 (g) + H2O (l)
Then, balance the hydrogens (10 on reactant side) in the products. Again, hydrogens appear only once on each side, making them a good choice to work on next:
C4H10 (l) + O2 (g) → 4 CO2 (g) + 5 H2O (l)
Next, balance the oxygens (now 13 on product side) in the reactants. Note that oxygens appear in multiple reactants and products, making them the most complex atom to balance and therefore the best to leave until the end: C 4 H 10 ( l ) + 13 2 O 2 ( g ) → 4 CO 2 ( g ) + 5 H 2 O ( l )
Next, produce a whole number ratio. In this case, double each coefficient.
2 C4H10 (l) + 13 O2 (g) → 8 CO2 (g) + 10 H2O (l)
Finally, check that all of the elements and the total charges are balanced correctly. If there is a difference in total charge between the reactants and products, then the charge will also have to be balanced. (Instructions for balancing charge in oxidation–reduction reactions are found in Chapter 11 of MCAT General Chemistry Review.)
Method Two: First, if in doubt, take a guess. Assume there are 4 of the first reactant and balance the carbons appropriately.
4C4H10 (l) + O2 (g) → 16CO2 (g) + H2O (l)
Second, balance the hydrogens (40 on reactant side) in the products.
4 C4H10 (l) + O2 (g) → 16 CO2 (g) + 20H2O (l)
Third, balance the oxygens (now 52 on product side) in the reactants.
4 C4H10 (l) + 26O2 (g) → 16 CO2 (g) + 20 H2O (l)
Fourth, produce the simplest whole number ratio through the greatest common factor. In this case, divide each side by 2.
2 C4H10 (l) + 13 O2 (g) → 8 CO2 (g) + 10 H2O (l)
Finally, check that all of the elements and the total charges are balanced correctly. Notice that both methods produce a multiple of our final answer. These ratios are both correct, but in terms of the stoichiometry one performs on the MCAT, the simpler the numbers are, the easier calculations will become.
KEY CONCEPT
When balancing equations, focus on the least represented elements first and work your way to the most represented element of the reaction (usually oxygen or hydrogen). If you’re stuck, take a guess for the coefficient of the first reactant and balance the remainder appropriately.
MCAT CONCEPT CHECK 4.4:
Before you move on, assess your understanding of the material with this question.
-
Balance the following reactions:
- _ Fe + _ Cl2 → ___ FeCl3
- _ Zn + _ HCl → _ ZnCl2 + _ H2
- _ C5H12 + _ O2 → _ CO2 + _ H2O
- _ Pb(NO3)2 + _ AlCl3 → _ PbCl2 + _ Al(NO3)3
4.5 Applications of Stoichiometry
LEARNING OBJECTIVES
After Chapter 4.5, you will be able to:
- Calculate the grams of product produced given the quantities of reactant
- Identify the limiting reagent within a reaction
- Calculate the mass of excess reagent in a reaction with a limiting reagent
- Calculate the percent yield of a reaction
Perhaps the most useful information to glean from a balanced reaction is the mole ratio of reactants consumed-to-products generated. One can also generate the mole ratio of one reactant to another or one product to another. All of these ratios can be generated using the stoichiometric coefficients. In the formation of water (2 H2 + O2 → 2 H2O), for example, one can determine that, for every one mole of hydrogen gas consumed, one mole of water can be produced; for every one mole of oxygen gas consumed, two moles of water can be produced. Furthermore, mole-to-mole, hydrogen gas is being consumed at a rate twice that of oxygen gas.
KEY CONCEPT
Stoichiometry, an application of dimensional analysis, is often simplified to a series of three fractions. These fractions demonstrate an underlying three-step process:
- Convert from the given units to moles
- Use the mole ratio
- Convert from moles to the desired units
Stoichiometry problems usually involve at least a few unit conversions, so take care when working through these types of problems to ensure that units cancel out appropriately to lead to the desired units of the answer choices. Pay close attention to the following problem, which demonstrates a clear and easy-to-follow method for keeping track of the numbers, calculations, and unit conversions.
Example: How many grams of calcium chloride are needed to prepare 71.7 g of silver chloride according to the following equation?
CaCl2 (aq) + 2 AgNO3 (aq) → Ca(NO3)2 (aq) + 2 AgCl (s)
Solution: Noting first that the equation is balanced, 1 mole of CaCl2 is reacted with 2 moles of AgNO3 to yield 2 moles of AgCl. The molar mass of CaCl2 is 111.1 g, and the molar mass of AgCl is 143.4 g. The given quantity is 71.7 g AgCl.
71 .7 g AgCl × [ 1 mol AgCl 143 .4 g AgCl ] [ 1 mol CaCl 2 2 mol AgCl ] [ 111 .1 g CaCl 2 1 mol CaCl 2 ] ≈ 72 144 × 1 2 × 110 1 = 1 2 × 1 2 × 110 1 = 110 4 = 27. 5 g CaCl 2 (actual value = 27.775 g)
Thus, about 27.8 g CaCl2 are needed to produce 71.7 g AgCl.
MCAT EXPERTISE
Common conversions used in stoichiometry include:
- 1 mole of any ideal gas at STP = 22.4 L
- 1 mole of any substance = 6.022 × 1023 particles (Avogadro’s number)
- 1 mole of any substance = its molar mass in grams (from the periodic table)
Limiting Reagent
Rarely are reactants added in the exact stoichiometric proportions shown in the balanced equation of a reaction. As a result, in most reactions, one reactant will be used up or consumed first. This reactant is known as the limiting reagent(or reactant) because it limits the amount of product that can be formed in the reaction. The reactants that remain after all the limiting reagent is used up are called excess reagents(or reactants).
MCAT EXPERTISE
When the quantities of two reactants are given on the MCAT, expect to have to figure out which is the limiting reagent.
Figure 4.7 shows a reaction vessel that has significant amounts of reactants A and B, which react in equal amounts to produce product C. On the left, before the reaction, there is more reactant A than B. After the reaction is over, there is more product C but there is reactant A left over. Thus, reactant A is considered in excess, and reactant B is considered limiting.
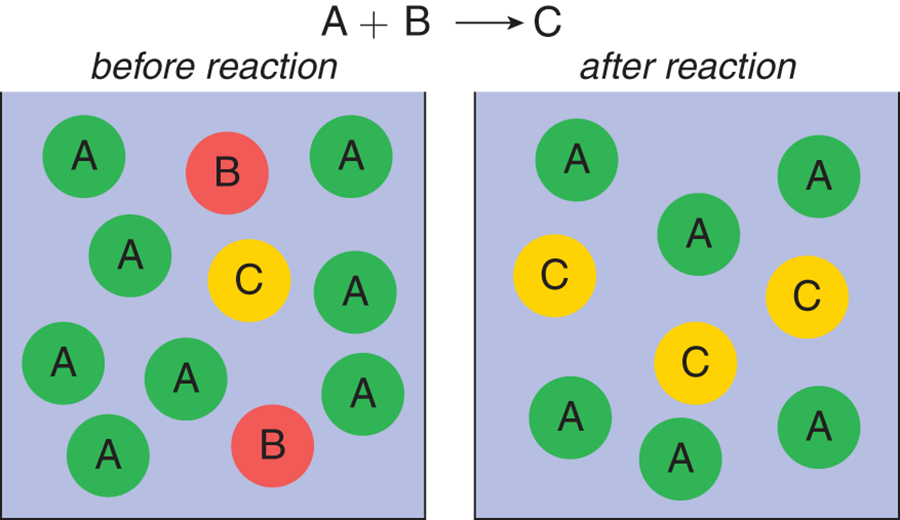
Figure 4.7. Reaction with a Limiting Reagent A is considered an excess reagent; B is the limiting reagent.
For problems involving the determination of the limiting reagent, keep in mind two principles:
- All comparisons of reactants must be done in units of moles. Gram-to-gram comparisons will be useless and may even be misleading.
- It is not the absolute mole quantities of the reactants that determine which reactant is the limiting reagent. Rather, the rate at which the reactants are consumed (the stoichiometric ratios of the reactants), combined with the absolute mole quantities determines which reactant is the limiting reagent.
Example: If 27.9 g of Fe react with 24.1 g of S to produce FeS, what would be the limiting reagent? How many grams of excess reagent would be present in the vessel at the end of the reaction?
The balanced equation is Fe + S → FeS.
Solution: First, determine the number of moles for each reactant.
27.9 g Fe × 1 mol Fe 55 .8 g Fe ≈ 28 × 1 56 = 0.5 mol Fe 24 .1 g S × 1 mol S 32.1 g S ≈ 24 × 1 32 = 0.75 mol S
Because 1 mole of Fe is needed to react with 1 mole of S and there are 0.5 moles Fe for the given 0.75 moles S, the limiting reagent is Fe. Thus, 0.5 moles of Fe will react with 0.5 moles of S, leaving an excess of 0.25 moles of S in the vessel. The mass of the excess reagent will be:
0.25 mol × 32.1 g S 1 mol S = 8 g S
Yield
The yield of a reaction can refer to either the amount of product predicted (theoretical yield) or actually obtained (raw or actual yield) when a reaction is carried out. Theoreticalyield is the maximum amount of product that can be generated as predicted from the balanced equation, assuming that all of the limiting reactant is consumed, no side reactions have occurred, and the entire product has been collected. Theoretical yield is rarely ever attained through the actual chemical reaction. Actual yield is the amount of product one actually obtains during the reaction. The ratio of the actual yield to the theoretical yield, multiplied by 100 percent, gives the percent yield:
Percent yield = Actual yield Theoretical yield × 100 %
Equation 4.6
MCAT EXPERTISE
An experimentally based passage that involves a chemical reaction may include a pseudo-discrete question that involves finding the percent yield.
Example: What is the percent yield for a reaction in which 28 g of Cu is produced by reacting 32.7 g of Zn in excess CuSO4 solution?
Solution: The balanced equation is as follows:
Zn (s) + CuSO4 (aq) → Cu (s) + ZnSO4 (aq)
Calculate the theoretical yield for Cu.
32 .7 g Zn × [ 1 mol Zn 65. 4 g Zn ] [ 1 mol Cu 1 mol Zn ] [ 63.5 g Cu 1 mol Cu ] ≈ 33 × 1 66 × 1 1 × 64 1 = 1 2 × 64 1 = 32 ( actual value = 31.8 g Cu )
This 31.8 g represents the theoretical yield. Finally, determine the percent yield.
28 g 31 .8 g × 100 % ≈ 28 32 × 100 % = 7 8 × 100 % = 87 . 5 %
MCAT CONCEPT CHECK 4.5:
Before you move on, assess your understanding of the material with these questions.
Questions 1–3 refer to the following unbalanced equation:
Na (s) + O2 (g) → Na2O (s)
-
Balance the chemical equation:
_ Na (s) + _ O2 (g) → ___ Na2O (s)
-
If 46 g Na and 32 g O2 are provided, find the maximum number of moles of sodium oxide produced.
_____________________________
-
Identify the limiting reagent, and find the mass of the excess reagent left over once the reaction has run to completion.
______________________________________
-
Be(OH)2 is produced when water reacts with BeO. Starting with 2.5 kg BeO in excess water, and producing 1.1 kg Be(OH)2, what is the percent yield of this reaction?
____________________________________
4.6 Ions
LEARNING OBJECTIVES
After Chapter 4.6, you will be able to:
- Determine whether a molecule will act as an electrolyte in solution
- Recall the common polyatomic ions, including their names and charges
Ionic compounds are of particular interest to chemists because certain important types of chemical reactions—acid–base and oxidation–reduction reactions, for instance—commonly take place in ionic solutions. For stoichiometry problems, the goal with ions is to identify oxidation states. This will allow us to determine electron equivalents, balance equations, and deduce chemical formulas from nomenclature.
Cations and Anions
In Chapter 3 of MCAT General Chemistry Review, we discussed how ionic compounds are made up of positively charged cations, usually metals, and negatively charged anions, usually nonmetals. This rule does not always hold true for elements like hydrogen, which can act like an anion or cation but is still classified as a nonmetal, as shown in Figure 4.8. Ionic compounds are held together by ionicbonds, which rely on the force of electrostatic attraction between oppositely charged particles.
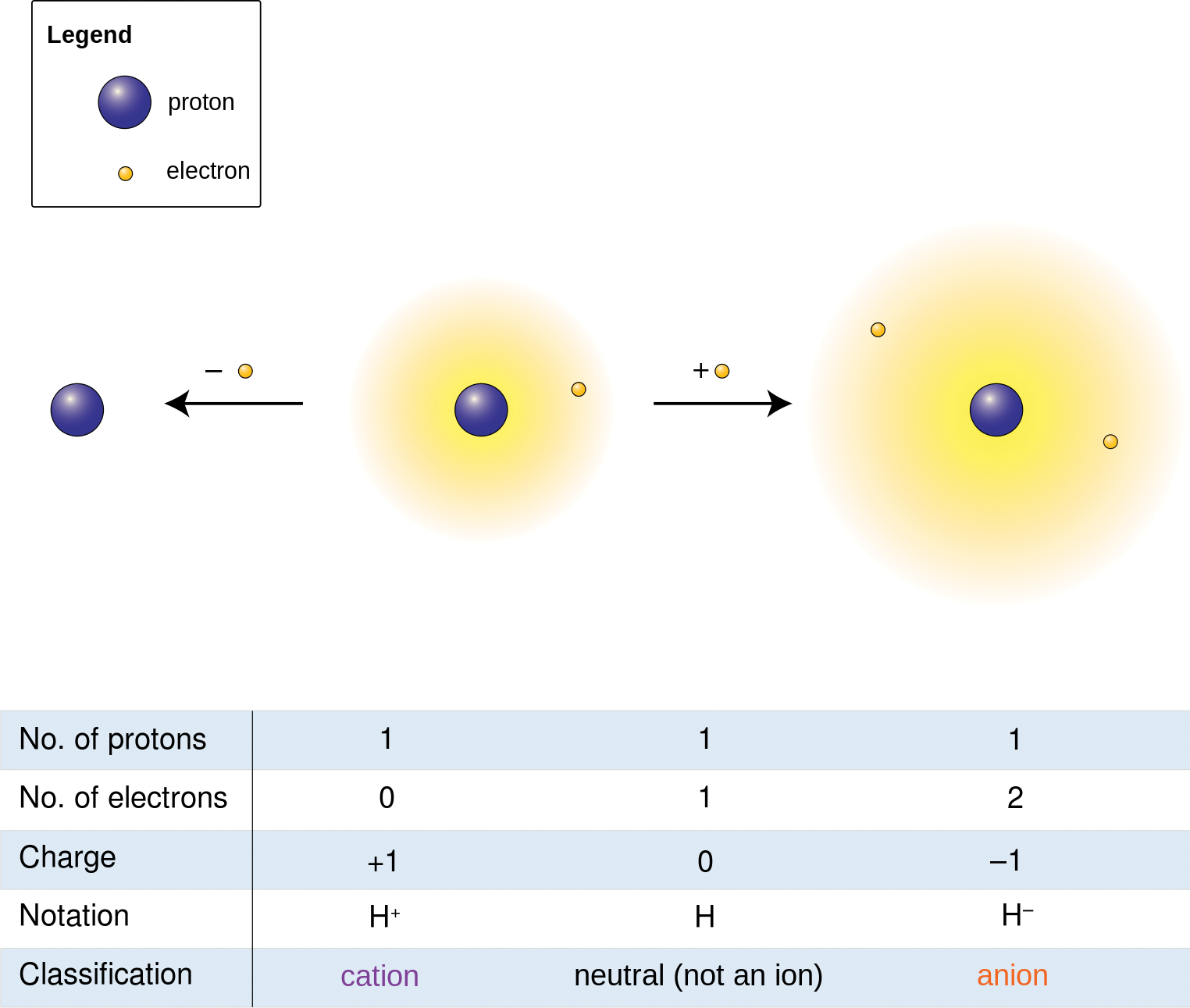
Figure 4.8. Oxidation States of Hydrogen
BRIDGE
The magnitude of the electrostatic force in an ionic bond follows the same conventions described for Coulomb’s law in Chapter 5 of MCAT Physics and Math Review. The distance between nuclei in ionic bonds is inversely proportional to the force. Therefore, ionic compounds with long bond distances are much more weakly held together.
The nomenclature of ionic compounds is based on the names of the component ions:
- For elements (usually metals) that can form more than one positive ion, the charge is indicated by a Roman numeral in parentheses following the name of the element.
Fe2+ Iron(II) Cu+ Copper(I)
Fe3+ Iron(III) Cu2+ Copper(II)
- An older, less commonly used method is to add the endings –ous or –ic to the root of the Latin name of the element to represent the ions with lesser and greater charge, respectively.
Fe2+ Ferrous Cu+ Cuprous
Fe3+ Ferric Cu2+ Cupric
- Monatomic anions are named by dropping the ending of the name of the element and adding –ide.
H− Hydride S2− Sulfide
F− Fluoride N3− Nitride
O2− Oxide P3− Phosphide
- Many polyatomic anions contain oxygen and are therefore called oxyanions. When an element forms two oxyanions, the name of the one with less oxygen ends in –ite, and the one with more oxygen ends in –ate.
NO2− Nitrite SO32− Sulfite
NO3− Nitrate SO42− Sulfate
- In extended series of oxyanions, prefixes are also used. Hypo– and hyper, written as per–, are used to indicate less oxygen and more oxygen, respectively.
ClO− Hypochlorite
ClO2− Chlorite
ClO3− Chlorate
ClO4− Perchlorate
- Polyatomic anions often gain one or more H+ ions to form anions of lower charge. The resulting ions are named by adding the word hydrogen or dihydrogen to the front of the anion’s name. An older method uses the prefix bi– to indicate the addition of a single hydrogen ion.
HCO3− Hydrogen carbonate or bicarbonate
HSO4− Hydrogen sulfate or bisulfate
H2PO4− Dihydrogen phosphate
- Other common polyatomic ions that may be useful to know are in Table 4.1.
Table 4.1. Other Common Polyatomic Ions
CHARGE FORMULA NAME
+1 NH4+ Ammonium
−1 C2H3O2− CN− MnO4− SCN− Acetate Cyanide Permanganate Thiocyanate
−2 CrO42− Cr2O72− Chromate Dichromate
−3 BO33− Borate
MCAT EXPERTISE
It is unlikely that –ous or –ic endings will be required for most problem-solving. Passages tend to provide reaction schemes that allow you to deduce any unfamiliar compound’s formulas. However, it is still important to understand the nomenclature for discrete questions.
MNEMONIC
The “litest” anions have the fewest oxygens; the heaviest anions ate the most oxygens.
Ion Charges
Ionic species, by definition, have charge. Cations have positive charge, and anions have negative charge. Some elements are only found naturally in their charged forms, while others may exist naturally in the charged or uncharged state. Some elements can even have several different charges or oxidationstates, depending on the other atoms in a compound.
Some of the charged atoms or molecules that are on the MCAT include the active metals—the alkali metals (Group IA or Group 1) and the alkaline earth metals (Group IIA or Group 2), which have charges of +1 and +2, respectively, in the natural state.
BRIDGE
Remember that alkali metals are not typically found in nature in their uncharged state because they are highly reactive with moisture. Instead, they are found as cations in salts (like NaCl).
Nonmetals, which are found on the right side of the periodic table, generally form anions. For example, all the halogens (Group VIIA or Group 17) form monatomic anions with a charge of –1 because they already have 7 electrons and aim to fill an octet.
In summary, all elements in a given group tend to form monatomic ions with the same charge (for example, all Group IA elements have a charge of +1 in their ionic state). Note that there are anionic species that contain metallic elements (for example, MnO4− [permanganate] and CrO42− [chromate]); even so, the metals have positive oxidation states. Also note that in the oxyanions of the halogens, such as ClO– and ClO2−, the halogen is assigned a positive oxidation state.
BRIDGE
Oxyanions of transition metals like the MnO4− and CrO42− ions have an inordinately high oxidation number on the metal. As such, they tend to gain electrons in order to reduce this oxidation number and thus make good oxidizing agents. Good oxidizing and reducing agents are discussed in Chapter 4 of MCAT Organic Chemistry Review.
For nonrepresentative elements like many of the transition metals, such as copper, iron, and chromium, there are numerous positively charged states. These states need not be memorized. Qualitatively, the color of a solution can be indicative of the oxidation state of a given element in the solution. The same element in different oxidation states can undergo different electron transitions and therefore absorb different frequencies of light. In Figure 4.9, this phenomenon is shown for various plutonium salts with different oxidation states for plutonium indicated in Roman numerals.

Figure 4.9. Solutions with Various Plutonium Oxidation States
The trends of ionicity, as we’ve described here, are helpful but are complicated by the fact that many elements have intermediate electronegativity and are consequently less likely to form ionic compounds, and by the left-to-right transition from metallic to nonmetallic character on the periodic table.
Electrolytes
In spite of the fact that ionic compounds are composed of ions, solid ionic compounds tend to be poor conductors of electricity because the charged particles are rigidly set in place by the lattice arrangement of the crystalline solid. In aqueous solutions, however, the lattice arrangement is disrupted by the ion–dipole interactions between the ionic components and the water molecules. The cations and anions are now free to move, and as a result, the solution of ions is able to conduct electricity.
Solutes that enable solutions to carry currents are called electrolytes. The electrical conductivity of aqueous solutions is governed by the presence and concentration of ions in the solution. Subsequently, the number of electron equivalents being transferred in such a system, such as in electrochemical cells, varies. Pure water, which has no ions other than the very few hydrogen ions and hydroxide ions that result from water’s low-level autodissociation, is a very poor conductor.
MCAT EXPERTISE
Ionic compounds make good electrolytes because they dissolve most readily. Nonpolar covalent compounds are the weakest because they do not form current-carrying ions.
The tendency of an ionic solute to dissolve, or solvate, into its constituent ions in water may be high or low. A solute is considered a strong electrolyte if it dissociates completely into its constituent ions. Examples of strong electrolytes include certain ionic compounds, such as NaCl and KI, and molecular compounds with highly polar covalent bonds that dissociate into ions when dissolved, such as HCl in water. An example of solvation of such compounds is shown in Figure 4.10.
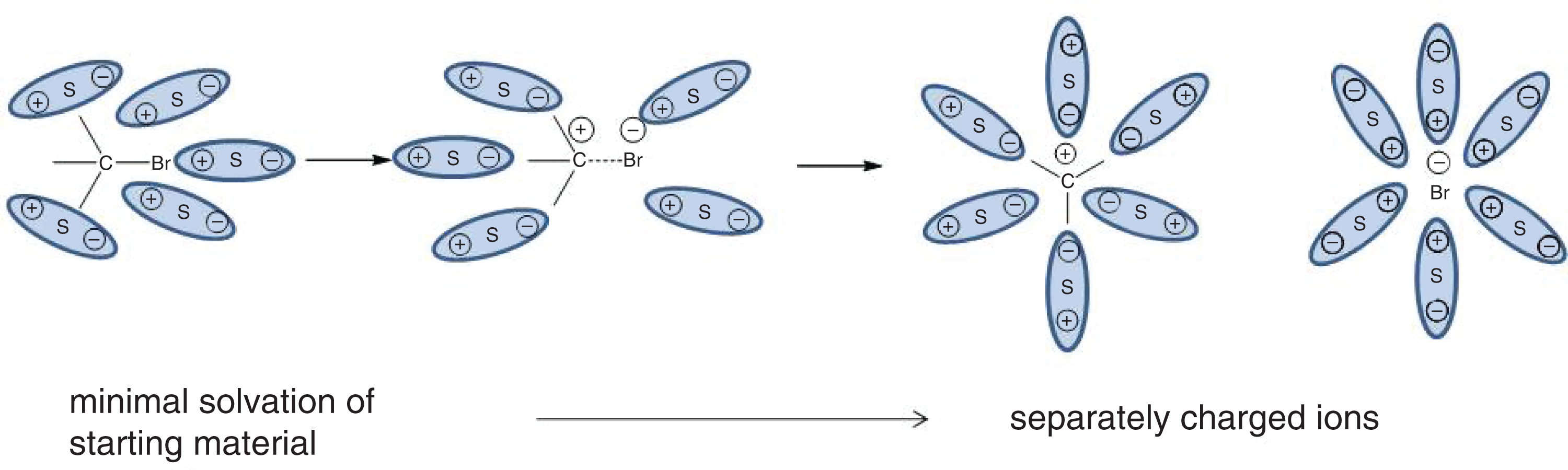
Figure 4.10. Solvation of a Polar Covalent Compound S indicates a solvent particle.
A weak electrolyte, on the other hand, ionizes or hydrolyzes incompletely in aqueous solution, and only some of the solute is dissolved into its ionic constituents. Examples include Hg2I2 (Ksp = 4.5 × 10–29), acetic acid and other weak acids, and ammonia and other weak bases. Many compounds do not ionize at all in water, retaining their molecular structure in solution, which may also limit their solubility. These compounds are called nonelectrolytes and include many nonpolar gases and organic compounds, such as O2 (g), CO2 (g), and glucose.
BRIDGE
Because electrolytes ionize in solution, they will produce a larger effect on colligative properties, described in Chapter 9 of MCAT General Chemistry Review, than one would expect from the given concentration.
MCAT CONCEPT CHECK 4.6:
Before you move on, assess your understanding of the material with these questions.
-
Label the following solutions as electrolytes or nonelectrolytes: (Note: Assume these compounds are all in aqueous solution.)
Electrolyte Nonelectrolyte
- HCl
Electrolyte Nonelectrolyte
- Sucrose
Electrolyte Nonelectrolyte
- MgBr2
Electrolyte Nonelectrolyte
- CH4
-
Identify the following ions as cations or anions, and then provide the formula or chemical symbol:
Ion Cation or Anion Formula Phosphate Hypochlorite Ammonium Phosphide Bicarbonate Nitrite Chromium(II)
Conclusion
We began our consideration of compounds with benzaldehyde. As a compound, it is made from constituent atoms of different elements in a set ratio defined by its empirical or molecular formula. Each molecule of a compound has a defined mass that is measured as its molecular weight. The mass of one mole of any compound is determined from its molar mass in the units of grams per mole. We reviewed the basic classifications of reactions commonly tested on the MCAT: combination, decomposition, combustion, single-displacement, double-displacement, and neutralization reactions. Furthermore, we are now confident in our understanding of the steps necessary to balance any chemical reaction; we are ready to tackle more stoichiometric problems in preparation for Test Day.
Before moving to the next chapters discussing chemical kinetics and thermodynamics, let us offer our congratulations to you. By completing these first four chapters, you have been introduced to the fundamental concepts of chemistry—everything from the structure of the atom and trends of the elements to bonding and the formation of compounds. The understanding you have gained so far will be the foundation for your comprehension of even the most difficult general chemistry concepts tested on the MCAT. Keep moving forward with your review of general chemistry; don’t get stuck in the details. Those details will be learned best through the application of the basic principles to MCAT practice passages and questions.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Molecules and Moles
- Compounds are substances composed of two or more elements in a fixed proportion.
- Molecular weight is the mass (in amu) of the constituent atoms in a compound as indicated by the molecular formula.
- Molar mass is the mass of one mole (Avogadro’s number or 6.022 × 1023 particles) of a compound; usually measured in grams per mole.
- Gram equivalent weight is a measure of the mass of a substance that can donate one equivalent of the species of interest.
- Normality is the ratio of equivalents per liter; it is related to molarity by multiplying the molarity by the number of equivalents present per mole of compound.
- Equivalents are moles of the species of interest; equivalents are most often seen in acid–base chemistry (hydrogen ions or hydroxide ions) and oxidation–reduction reactions (moles of electrons or other ions).
Representation of Compounds
- The law of constant composition states that any pure sample of a compound will contain the same elements in the same mass ratio.
- The empirical formula is the smallest whole-number ratio of the elements in a compound.
- The molecular formula is either the same as or a multiple of the empirical formula; it gives the exact number of atoms of each element in a compound.
- To calculate percent composition by mass, determine the mass of the individual element and divide by the molar mass of the compound.
Types of Chemical Reactions
- Combinationreactions occur when two or more reactants combine to form one product.
- Decomposition reactions occur when one reactant is chemically broken down into two or more products.
- Combustion reactions occur when a fuel and an oxidant (typically oxygen) react, forming the products water and carbon dioxide (if the fuel is a hydrocarbon).
- Displacement reactions occur when one or more atoms or ions of one compound are replaced with one or more atoms or ions of another compound.
- Single-displacement reactions occur when an ion of one compound is replaced with another element.
- Double-displacement reactions occur when elements from two different compounds trade places with each other to form two new compounds.
- Neutralization reactions are those in which an acid reacts with a base to form a salt (and, usually, water).
Balancing Chemical Equations
- Chemical equations must be balanced to perform stoichiometric calculations.
- Balanced equations are determined using the following steps in order:
- Balancing the least common atoms
- Balancing the more common atoms (usually hydrogen and oxygen)
- Balancing charge, if necessary
Applications of Stoichiometry
- Balanced equations can be used to determine the limiting reagent, which is the reactant that will be consumed first in a chemical reaction.
- The other reactants present are termed excess reagents.
- Theoretical yield is the amount of product generated if all of the limiting reactant is consumed with no side reactions.
- Actual yield is typically lower than theoretical yield.
- Percent yield is calculated by dividing actual yield by theoretical yield and converting to a percentage.
Ions
- Like organic chemistry, ions in general chemistry have a system of nomenclature:
- Roman numerals are used for nonrepresentative elements to denote ionic charge.
- –ous endings can also be used to indicate lesser charge, while –ic endings indicate greater charge.
- All monatomic anions end in –ide.
- Oxyanions are given a suffix indicating how oxidized the central atom is. Those that contain a lesser amount of oxygen are given the suffix –ite, and those with a greater amount are given the suffix –ate.
- Oxyanion series with more than two members are given an additional level of nomenclature. The species with the fewest oxygens is given the prefix hypo–, and the species with the most oxygens is given the prefix per–.
- Polyatomic ions containing hydrogen denote the number of hydrogens using hydrogen or bi– to denote one, or dihydrogen to denote two.
- Ionic charges are predictable by group number and type of element (metal or nonmetal) for representative elements, but are generally unpredictable for nonrepresentative elements.
- Metals form positively charged cations based on group number.
- Nonmetals form negatively charged anions based on the number of electrons needed to achieve an octet.
- Electrolytes contain equivalents of ions from molecules that dissociate in solution. The strength of an electrolyte depends on its degree of dissociation or solvation.
ANSWERS TO CONCEPT CHECKS
**4.1**
- NaBr: 23 + 79 . 9 ≈ 23 + 80 = 103 g mol
SrCl2: 87 . 6 + ( 2 × 35 . 5 ) = 87 . 6 + 71 = 158 . 6 g mol
C6H12O6: ( 6 × 12 ) + ( 12 × 1 ) + ( 6 × 16 ) = 180 g mol
- NaBr: 100 g 103 g mol = 0 . 97 moles
Note that the denominator is greater than the numerator by approximately 3 percent, and the actual value is less than 1 by approximately 3 percent. This approximation can be used to quickly estimate answers that are close to one.
SrCl2: 100 g 158 . 6 g mol ≈ 100 160 = 10 16 = 5 8 = 0 .625 moles
Note that the answer is a fraction based on eighths. As these are commonly used on the MCAT, knowing the values for 1/8 through 7/8 can be useful for many problems.
C6H12O6: 100 g 180 g mol = 10 18 = 5 9 = 0 . 555 moles
Note that the final fraction is in ninths. Dividing by nine follows a standard pattern that is useful to know for the MCAT: 1/9 = 0.111, 2/9 = 0.222, 3/9 = 0.333, etc.
- Both values equal one mole of the given substance. The number of entities in a mole is always the same (Avogadro’s number, 6.022 × 1023 mol–1).
-
Normality is calculated as N = M × equivalents mole . For H 3 PO 4 : 0.25 M H 3 PO 4 × 3 equiv H + mol H 3 PO 4 = 0.75 N H 3 PO 4 .
For PO43- the grams must first be converted to moles, then to normality. PO43- has a molecular mass of 95, giving
95 g 100 mL = 950 g 1 L = 10 mol 1 L = 10 M PO 4 3 - 10 M PO 4 3 − × 3 equiv H + mol PO 4 3 − = 30 N PO 4 3 − .
**4.2**
- Both molecular and empirical formulas contain the same elements in the same ratios. They differ in that molecular formulas give the actual number of atoms of each element in the compound; empirical formulas give only the ratio and therefore may or may not give the actual number of atoms.
- The molar mass of sodium carbonate is given by (2×23)+(1×12)+(3×16)=106 g mol . The percent compositions are: Na : 2 × ( 23 g mol ) 106 g mol × 100 % ≈ 46 100 × 100 % = 46 % ( actual value = 43.4 % ) O : 3 × ( 16 g mol ) 106 g mol × 100 % ≈ 48 100 × 100 % = 48 % ( actual value = 45.3 % ) C : 1 × ( 12 g mol ) 106 g mol × 100 % ≈ 12 100 × 100 % = 12 % ( actual value = 11.3 % )
Note that in all three cases, the estimation reduces the value of the denominator, thus making the calculated value larger than the actual value.
- Start by assuming a 100 g sample, which represents 28.5 g Fe, 24.0 g S, and 49.7 g O. Next, divide each number of grams by the atomic weight to determine the number of moles: Fe: 28 . 5 g 55 . 8 g mol ≈ 28 56 = 0.5 moles S: 24 g 32 . 1 g mol ≈ 24 32 = 3 4 = 0.75 moles O: 49 . 7 g 16 g mol ≈ 48 16 = 3 moles
Next, find the multiplier that gives all three compounds integer values of moles. Using sulfur, multiplying 0.75 moles × 4 = 3 moles. Using 4 as a multiplier for all three compounds gives the ratio 2 Fe : 3 S : 12 O. This gives an empirical formula of Fe2S3O12.
**4.3**
- Ammonium cations swap places with (or displace) zinc cations yielding ammonium nitrate and zinc(II) sulfide. Zinc(II) sulfide then precipitates out of solution as a solid salt.
-
Reactants Conditions Products Reaction Type
2 H2 + O2 → 2 H2O Combination
Al(OH)3 + H3PO4 → 3 H2O + AlPO4 Neutralization (a type of double-displacement)
2 H2O → electricity 2 H2 + O2 Decomposition
NaNO3 + CuOH → NaOH + CuNO3 Double-displacement (metathesis)
Zn + AgCl → ZnCl + Ag Single-displacement
**4.4**
-
- 2 Fe + 3 Cl2 → 2 FeCl3
- Zn + 2 HCl → ZnCl2 + H2
- C5H12 + 8 O2 → 5 CO2 + 6 H2O
- 3 Pb(NO3)2 + 2 AlCl3 → 3 PbCl2 + 2 Al(NO3)3
**4.5**
- 4 Na (s) + O2 (g) → 2 Na2O (s)
32 g O 2 × [ 1 mol O 2 32 g O 2 ] = 1 mol O 2 Because 4 sodium atoms are needed for every oxygen molecule, sodium will run out first. To determine the amount of Na2O formed:
- 46 g Na × [ 1 mol Na 23 g Na ] = 2 mol Na;
2 mol Na × [ 2 mol Na 2 O 4 mol Na ] = 1 mol Na 2 O
- The limiting reagent is Na because 4 sodium atoms are needed for every oxygen molecule.
2 mol Na × [ 1 mol O 2 4 mol Na ] = 0.5 mol O 2
will be used, so 1.0 – 0.5 mol O2 = 0.5 mol O2 will remain. In grams, this is:
0.5 mol O 2 × [ 32 g O 2 1 mol O 2 ] = 16 g excess O 2
-
Reaction: BeO + H2O → Be(OH)2
Theoretical yield:
2500 g BeO × [ 1 mol BeO 25 g BeO ] [ 1 mol Be(OH) 2 1 mol BeO ] [ 43 g Be(OH) 2 1 mol Be(OH) 2 ] = 4300 g Be(OH) 2
Percent yield = Actual yield Theoretical yield × 100 % = 1100 g 4300 g × 100 % ≈ 11 44 × 100 % = 25 % yield ( actual value = 25 . 6 % )
**4.6**
- Electrolytes: HCl, MgBr2; Nonelectrolytes: sucrose, CH4
-
Ion Cation or Anion Formula Phosphate Anion PO43−
Hypochlorite Anion ClO−
Ammonium Cation NH4+
Phosphide Anion P3−
Bicarbonate Anion HCO3−
Nitrite Anion NO2−
Chromium(II) Cation Cr2+
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
Ionic compounds are composed of atoms held together by ionic bonds. Ionic bonds associate charged particles with large differences in electronegativity. Rather than forming molecules or being measured by molecular weight, as in (A) and (B), ionic compounds form large arrays of ions in crystalline solids and are measured with formula weights. In ionic bonds, electrons are not really shared but rather are donated from the less electronegative atom to the more electronegative atom, eliminating (C).
2. A
Of the compounds listed, both (B) and (D) are covalent compounds and thus are measured in molecular weights, not formula weights. The formula weight of MgCl2 is much too high (24.3 amu + 2 × 35.5 amu = 95.3 amu per formula unit), eliminating (C). Only KCl fits the criteria (39.1 amu + 35.5 amu = 74.6 amu).
3. A
First, it is helpful to know the molar mass of one mole of H2SO4, which is found by adding the atomic weights of the atoms that constitute the molecule:
( 2 × 1.0 g mol H ) + ( 1 × 32.1 g mol S ) + ( 4 × 16.0 g mol O ) = 98.1 g mol H 2 SO 4 .
Gram equivalent weight is the mass (in grams) that would release one mole of protons. Because sulfuric acid has two hydrogens per molecule, the gram equivalent weight is 98.1 g divided by 2, or 49.1 g.
4. C
The definition of an empirical formula is a formula that represents a molecule with the simplest ratio, in whole numbers, of the elements comprising the compound. In this case, given the empirical formula CH, any molecule with carbon and hydrogen atoms in a 1:1 ratio would be accurately represented by this empirical formula. (C)has three carbon atoms and four hydrogen atoms. Both its molecular and empirical formulas would be C3H4 because this formula represents the smallest whole-number ratio of its constituent elements.
5. A
The percent composition by mass of any given element within a molecule is equal to the mass of that element in the molecule divided by the molar mass of the compound, times 100%. In this case, acetone, C3H6O, has
3 × 12 g mol C (3 × 12 g mol C ) + ( 6 × 1 g mol H ) + ( 16 g mol O ) = 36 g mol 58 g mol = 18 29 ≈ 2 3 ≈ 66 . 7 % .
This is an overestimation, and the actual value will be lower; it is closest to 62% out of the four choices available. (B), ethanol, is 24 g mol C 46 g mol total ≈ 50 % . This is an underestimation, and the actual value will be higher but nowhere near 62%. (C), propane, is C3H8, and calculates to be 36 g mol c 44 g mol total ≈ 9 11 ≈ 80 % carbon . This is an underestimation, and therefore the actual value cannot be 62%. Finally, (D), methanol, is 12 g mol C 32 g mol total = 3 8 = 37.5 % carbon .
Note that all four of these compounds are commonly encountered on the MCAT, and you should be familiar with the structure and composition of each, including their common names.
6. B
This reaction is a classic example of a neutralization reaction, in which an acid and a base react to form a salt and, usually, water. Although this reaction also fits the criteria for a double-displacement reaction, (C), in which two molecules essentially exchange ions with each other, neutralization is a more specific description of the process.
7. A
In this question, you are first given the masses of both reactants used to start the reaction. To figure out what will be left over, we must first determine which species is the limiting reagent. The formula weight of Na2S is
( 2 × 23 g mol Na ) + ( 1 × 32 . 1 g mol S ) = 78 . 1 g mol Na 2 S .
The formula weight of AgNO3 is
( 1 × 107 . 9 g mol Ag ) + ( 1 × 14 g mol N ) + ( 3 × 16 g mol O ) = 169 . 9 g mol AgNO 3 .
From this, we can determine that we are given:
39 . 05 g Na 2 S 78 . 1 g mol = 0 . 5 mol Na 2 S and 85 . 5 g AgNO 3 169 . 9 g mol = 0.5 mol AgNO 3 .
Because we need two moles of AgNO3 for every mole of Na2S, AgNO3 is the limiting reagent, and the correct answer choice will be in grams of Na2S. If 0.5 mol of AgNO3 are used up, and Na2S will be consumed at half the rate of AgNO3 (based on their mole ratio), then 0.25 mol Na2S will be used up. We then have 0.25 mol excess Na2S, which has a mass of
0 . 25 mol × 78 . 1 g mol ≈ 1 4 × 80 1 = 20 g actual value = 19.5 g .
8. A
This is a question best answered by dimensional analysis. Keeping in mind that molar mass is measured in grams of a substance per moles of that substance, only (A) comes out with the units of grams of oxygen. (B) has the units of grams per mole of oxygen, not grams of oxygen. (C) has the units of moles per gram of oxygen. (D) has the units of mol2 per gram of oxygen.
9. D
In the reaction, there is a single displacement, with the silver in silver oxide being replaced by the aluminum to form aluminum oxide. This single-displacement reaction also necessitates a transfer of electrons in an oxidation–reduction reaction; silver, for example, changes from the +2 oxidation state to neutral. Aluminum changes from neutral to the +3 oxidation state.
10. C
Typically, both single-displacement and double-displacement reactions have two reactants that swap either one or two components between the two species. Combination reactions, on the other hand, have more reactants than products because the reactants combine together to form the product.
11. A
In the unbalanced reaction given in the question stem, zinc is already balanced. Next check carbon. There are 4 carbon atoms in the reactants and 1 in the product, so add a coefficient of 4 to carbon dioxide on the product side. Next, check hydrogen. There are 10 hydrogen atoms in the reactants and 2 in the products, so add a coefficient of 5 to water on the product side. Finally, check oxygen. There are 2 oxygen atoms in the reactants and 14 in the products, so add a coefficient of 7 to molecular oxygen on the reactant side. These alterations yield the balanced reaction
Zn(C 2 H 5 ) 2 + 7 O 2 → ZnO + 4 CO 2 + 5 H 2 O
Because of the coefficient of 4 in front of the CO2 molecule on the product side, the answer is (A).
12. B
The equation given is unbalanced, so the first step must be to balance it:
6 CO 2 + 6 H 2 O → h ν C 6 H 12 O 6 + 6 O 2
The theoretical yield is the amount of product synthesized if the limiting reagent is completely used up. This question therefore asks how much glucose is produced if the limiting reagent is 30 grams of water. Using the three-fraction method discussed in this chapter to solve for the mass of glucose produced gives:
30.0 g H 2 O × [ 1 mol H 2 O 18 g H 2 O ] [ 1 mol C 6 H 12 O 6 6 mol H 2 O ] [ 180 g C 6 H 12 O 6 1 mol C 6 H 12 O 6 ] = 50 g C 6 H 12 O 6
Thus, 50 grams of glucose are produced.
13. B
A limiting reagent is by definition a reactant. Because Au and H2S are products, they cannot act as limiting reagents, eliminating (C) and (D). Next, note that the given equation is unbalanced and the first step is to balance it:
Au2S3 (s) + 3 H2 (g) → 2 Au (s) + 3 H2S (g)
The problem states that 2 moles of gold(III) sulfide and 5 moles of hydrogen gas are available. To use up both moles of gold(III) sulfide, 6 moles of hydrogen gas are needed because there is a 1:3 ratio between these reactants. Since only 5 moles of hydrogen gas are present, that will have to be the limiting reagent.
14. B
The best electrolytes dissociate readily (have a high dissociation constant) and are ionic compounds with large amounts of cations and anions. This rules out (A) and (C). (D) has fewer total ions with a smaller total magnitude of charge and therefore is not as strong an electrolyte as (B).
15. A
One approach to answering this question might be to write out an unbalanced equation using the description in the question stem, and then balance that equation. But a faster method is elimination. Three of the given chemical equations are not properly balanced, while one is. Check one element at a time. Observe that in this question, the oxygen atoms in equation (A) are balanced, but oxygen is not balanced in (B), (C), and (D). Thus (A) must be the correct answer.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(4.1) Moles from mass: Moles = Mass of a sample Molar mass
(4.2) Gram equivalent weight: GEW = Molar mass n
(4.3) Equivalents from mass: Equivalents = Mass of compound Gram equivalent weight
(4.4) Molarity from normality: Molarity = Normality n
(4.5) Percent composition: % composition = Mass of element in formula Molar mass × 100 %
(4.6) Percent yield: % yield = Actual yield Theoretical yield × 100 %
SHARED CONCEPTS
General Chemistry Chapter 2
The Periodic Table
General Chemistry Chapter 3
Bonding and Chemical Interactions
General Chemistry Chapter 9
Solutions
General Chemistry Chapter 10
Acids and Bases
General Chemistry Chapter 11
Oxidation–Reduction Reactions
Physics and Math Chapter 5
Electrostatics and Magnetism