Chapter 5: Chemical Kinetics

Chapter 5: Chemical Kinetics
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
In a third-order reaction involving two reactants and two products, doubling the concentration of the first reactant causes the rate to increase by a factor of 2. What will happen to the rate of this reaction if the concentration of the second reactant is cut in half?
- It will increase by a factor of 2.
- It will increase by a factor of 4.
- It will decrease by a factor of 2.
- It will decrease by a factor of 4.
-
In a certain equilibrium process, the activation energy of the forward reaction (ΔGǂf) is greater than the activation energy of the reverse reaction (ΔGǂr). This reaction is:
- endothermic.
- exothermic.
- spontaneous.
- nonspontaneous.
-
A reactant in a second-order reaction at a certain temperature is increased by a factor of 4. By how much is the rate of the reaction altered?
- It is unchanged.
- It is increased by a factor of 4.
- It is increased by a factor of 16.
- It cannot be determined from the information given.
-
The concentrations of all reactants in a zero-order reaction are increased two-fold. What is the new rate of the reaction?
- It is unchanged.
- It is decreased by a factor of 2.
- It is increased by a factor of 2.
- It cannot be determined from the information given.
-
Which of the following experimental methods should NEVER affect the rate of a reaction?
- Placing an exothermic reaction in an ice bath.
- Increasing the pressure of a reactant in a closed container.
- Putting the reactants into an aqueous solution.
- Removing the product of an irreversible reaction.
-
What would increasing the concentration of reactants accomplish in a solution containing a saturated catalyst?
- It would increase the rate constant but not the reaction rate.
- It would decrease the rate constant but increase the reaction rate.
- It would increase the rate constant and increase the reaction rate.
- The reaction rate would be unaffected.
-
A certain chemical reaction has the following rate law:
rate = k[NO2][Br2]
Which of the following statements necessarily describe(s) the kinetics of this reaction?
- The reaction is second-order.
- The amount of NO2 consumed is equal to the amount of Br2 consumed.
- The rate will not be affected by the addition of a compound other than NO2 and Br2.
- I only
- I and II only
- II and III only
- I, II, and III
-
The following data shown in the table were collected for the combustion of the theoretical compound XH4:
XH4 + 2 O2 → XO2 + 2 H2O
**Trial [XH4]initial (M) [O2]initial (M) Rate initial ( M min ) **
1 0.6 0.6 12.4
2 0.6 2.4 49.9
3 1.2 2.4 198.3
What is the rate law for the reaction described here?
- rate = k[XH4][O2]
- rate = k[XH4][O2]2
- rate = k[XH4]2[O2]
- rate = k[XH4]2[O2]2
-
Which of the following best describes the purpose of a catalyst?
- Catalysts are used up in the reaction, increasing reaction efficiency.
- Catalysts increase the rate of the reaction by lowering the activation energy.
- Catalysts alter the thermodynamics of the reaction to facilitate the formation of products or reactants.
- Catalysts stabilize the transition state by bringing it to a higher energy.
-
If the rate law for a reaction is:
rate = k[A]0[B]2[C]1
What is the overall order of the reaction?
- 0
- 2
- 3
- 4
For questions 11–13, consider the following energy diagram shown below:
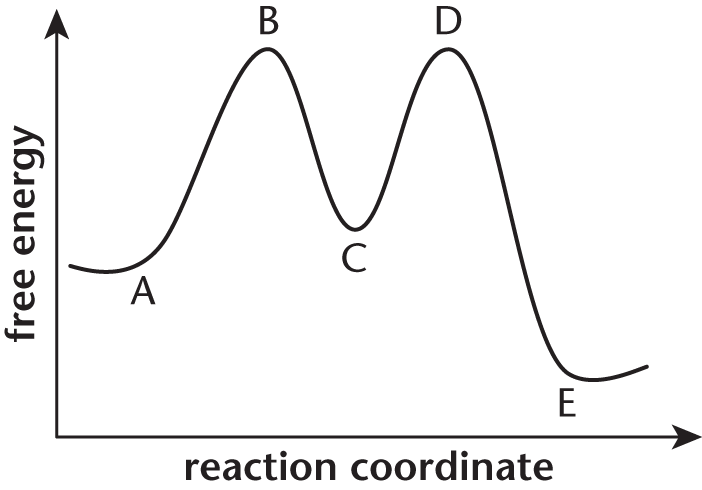
- The overall reaction depicted by this energy diagram is:
- endergonic, because point B is higher than point A.
- endergonic, because point C is higher than point A.
- exergonic, because point D is higher than point E.
- exergonic, because point A is higher than point E.
-
Which process has the highest activation energy?
- The first step of the forward reaction
- The first step of the reverse reaction
- The second step of the forward reaction
- The second step of the reverse reaction
-
Point C in this reaction profile refers to the:
- reactants.
- products.
- transition state.
- intermediates.
- The following system obeys second-order kinetics.
2 NO2 → NO3 + NO (slow)
NO3 + CO → NO2 + CO2 (fast)
What is the rate law for this reaction?
- rate = k[NO2][CO]
- rate = k[NO2]2[CO]
- rate = k[NO2][NO3]
- rate = k[NO2]2
-
The potential energy diagram shown represents four different reactions.
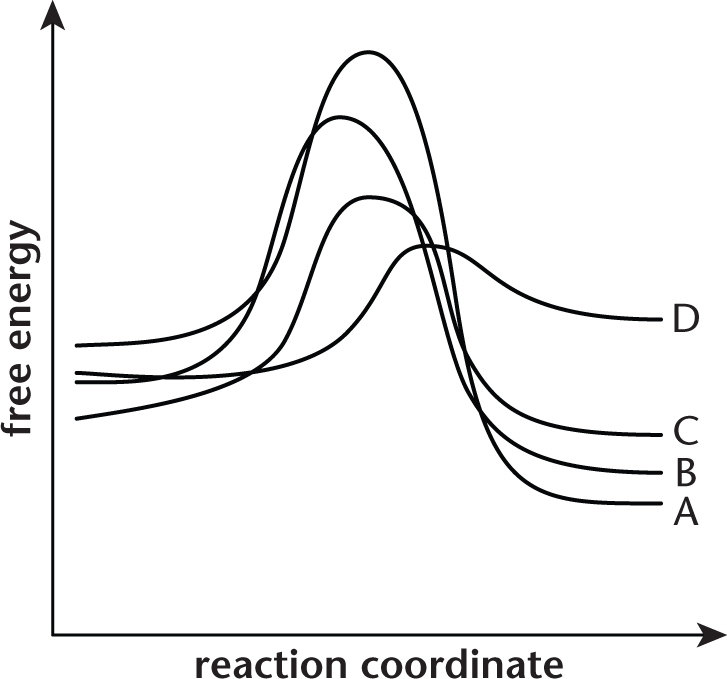
Assuming identical conditions, which of the reactions displayed on the energy diagram proceeds the fastest?
- A
- B
- C
- D
Answer Key
- D
- D
- D
- A
- D
- D
- A
- C
- B
- C
- D
- B
- D
- D
- D
Chapter 5: Chemical Kinetics
CHAPTER 5
CHEMICAL KINETICS
In This Chapter
5.1 Chemical Kinetics
Reaction Mechanisms
Molecular Basis of Chemical Reactions
Factors Affecting Reaction Rate
5.2 Reaction Rates
Definition of Rate
Determination of Rate Law
Reaction Orders
Concept Summary
CHAPTER PROFILE
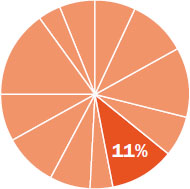
The content in this chapter should be relevant to about 11% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1A: Structure and function of proteins and their constituent amino acids
5E: Principles of chemical thermodynamics and kinetics
Introduction
The following chapters focus on two primary topics: chemical kinetics and chemical equilibrium. As the term suggests, chemical kinetics is the study of reaction rates, the effects of reaction conditions on these rates, and the mechanisms implied by such observations. We start with kinetics because the molecular basis of reactions provides us with a framework of reaction chemistry. Following this, we will explore the equilibria of these reactions, which are related to—but distinct from—the kinetics of the reactions.
You may already have a fairly good understanding of equilibrium and the differences between spontaneous and nonspontaneous reactions. For instance, the utilization of ATP in the body is a spontaneous reaction that can be used to provide thermochemical energy for other reactions. While the equilibrium tells us that ATP will favor dissociation, it tells us nothing about its rate of dissociation. And, in fact, various conditions in the body can alter the rate at which ATP is synthesized and utilized for energy—primarily temperature. Some of the symptoms of hyper- and hypothermia are related to changes in metabolism caused by changes in temperature and reaction kinetics.
More broadly, we will see how multistep reactions, such as those seen in substrate-level and oxidative phosphorylation in biochemistry, have intermediate steps that have crucial kinetic limitations.
5.1 Chemical Kinetics
LEARNING OBJECTIVES
After Chapter 5.1, you will be able to:
- Describe the series of events within a multistep mechanism
- Explain the meaning and importance of a rate-determining step
- Describe activation energy
- Compare and contrast transition state theory and collision theory
Reactions can be spontaneous or nonspontaneous; the change in Gibbs free energy (ΔG) determines whether or not a reaction will occur by itself without outside assistance. However, even if a reaction is spontaneous, this does not necessarily mean that it will run quickly. In fact, nearly every biochemical reaction that enables life to exist, while perhaps spontaneous, proceeds so slowly that, without the aid of enzymes and other catalysts, measurable reaction progress might not actually occur over the course of an average human lifetime. And enzymes, like many other catalyzed reactions, can be saturated and experience a maximal turnover rate, as shown in Figure 5.1. For now, however, let us review reaction mechanisms, rates, rate laws, and the factors that pertain to simple chemical systems.
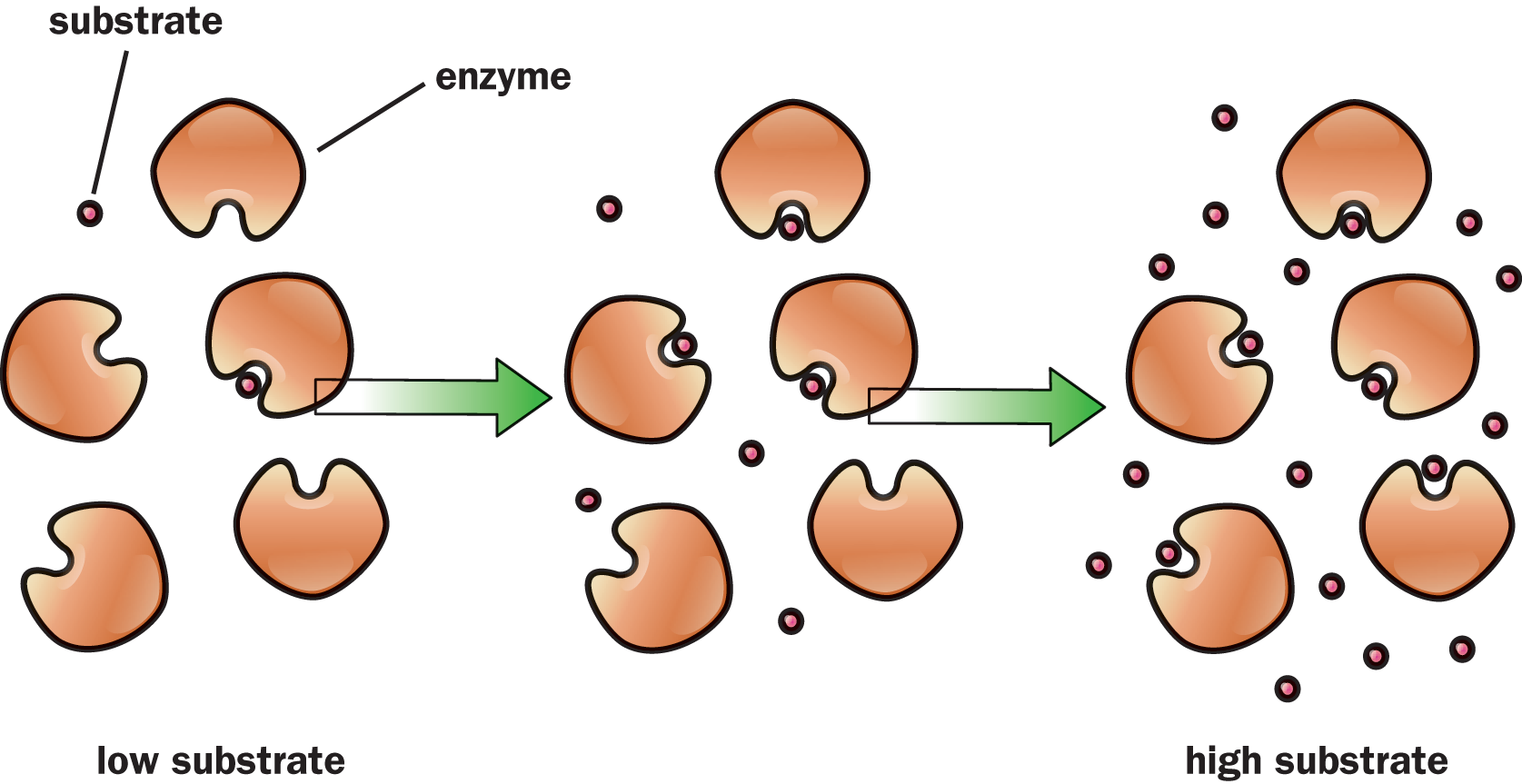
Figure 5.1. Enzymes, as Biological Catalysts, Can Be Saturated High substrate conditions saturate the active sites of the enzyme, leading to maximal turnover.
BRIDGE
Enzymes selectively enhance the rate of certain reactions by a factor of 102 to 1012 over other thermodynamically feasible reaction pathways. Enzyme function is discussed in Chapter 2 of MCAT Biochemistry Review.
Reaction Mechanisms
Very rarely is the balanced reaction equation, used for determining limiting reactants and yields, an accurate representation of the actual steps involved in the chemical process from reactants to products. Many reactions proceed by more than one step, the series of which is known as the mechanism of a reaction, and the sum of which gives the overall reaction. Knowing the accepted mechanism of a reaction may help explain the reaction’s rate, position of equilibrium, and thermodynamic characteristics. Consider this generic reaction:
A2 + 2 B → 2 AB
BRIDGE
Mechanisms are proposed pathways for a reaction that must coincide with rate data information from experimental observation. Reaction mechanisms are a major topic in organic chemistry and metabolism; Chapters 5 through 10 of MCAT Organic Chemistry Review and Chapters 9 through 11 of MCAT Biochemistry Review focus almost exclusively on reaction mechanisms in specific contexts.
On its own, this equation seems to imply a mechanism in which two molecules of B collide with one molecule of A2 to form two molecules of AB. Suppose instead, however, that the reaction actually takes place in two steps:
Step 1: A2 + B → A2B (slow)
Step 2: A2B + B → 2 AB (fast)
Note that the two steps, taken together, give the overall net reaction. The molecule A2B, which does not appear in the overall reaction, is called an intermediate. Reaction intermediates are often difficult to detect because they may be consumed almost immediately after they are formed, but a proposed mechanism that includes intermediates can be supported through kinetic experiments. One of the most important points to remember is that the slowest step in any proposed mechanism is called the rate-determining step because it acts like a kinetic bottleneck, preventing the overall reaction from proceeding any faster than that slowest step.
KEY CONCEPT
The rate of the whole reaction is only as fast as the rate-determining step.
Molecular Basis of Chemical Reactions
It’s one thing to say A2reacts with 2 B to form 2 AB; it’s quite another to be able to describe, as precisely as possible, the actual interactions that occur between A2 and B to produce AB at some rate. Various theories have been proposed to explain the events that are taking place at the atomic level through the process of a reaction.
Collision Theory of Chemical Kinetics
For a reaction to occur, molecules must collide with each other. The collision theory of chemical kinetics states that the rate of a reaction is proportional to the number of collisions per second between the reacting molecules.
The theory suggests, however, that not all collisions result in a chemical reaction. An effective collision (one that leads to the formation of products) occurs only if the molecules collide with each other in the correct orientation and with sufficient energy to break their existing bonds and form new ones. The minimum energy of collision necessary for a reaction to take place is called the activation energy, Ea, or the energy barrier. Only a fraction of colliding particles have enough kinetic energy to exceed the activation energy. This means that only a fraction of all collisions are effective. The rate of a reaction can therefore be expressed as follows:
rate = Z × f
Equation 5.1
whereZ is the total number of collisions occurring per second andf is the fraction of collisions that are effective. A much more quantitatively rigorous analysis of the collision theory can be accomplished through the Arrhenius equation, which is normally given as: k = A e − E a R T
Equation 5.2
where k is the rate constant of a reaction, A is the frequency factor, Ea is the activation energy of the reaction, R is the ideal gas constant, and T is the temperature in kelvin. The frequency factor, also known as the attempt frequency of the reaction, is a measure of how often molecules in a certain reaction collide, with the unit s–1. Activation energy is a subject that will be touched upon briefly in the following subsection and more qualitatively in future chapters.
Overall, what is important here in studying the Arrhenius equation is not the actual calculation (because those involving Euler’s number, e, and natural logs, ln, are not commonly found on the MCAT), but rather the relationships between the variables and the exponent rules that govern the equation.
For example, a simple relationship between A and k is evident in the equation. As the frequency factor of the reaction increases, the rate constant of the reaction also increases in a direct relationship. More complex relationships can also be seen in this equation. For example, if the temperature (T) of a chemical system were to increase to infinity, while all other variables are held constant, the value of the exponent would have a magnitude less than 1. However, before assuming that the rate constant is going to decrease as a result, note the presence of the negative sign. As the magnitude of the exponent gets smaller, it actually moves from a more negative value toward zero. The exponent thus becomes less negative (or more positive), which means that the rate constant actually increases. This should make sense conceptually because the rate of a reaction increases with temperature.
MCAT EXPERTISE
Low activation energy and high temperatures make the negative exponent of the Arrhenius equation smaller in magnitude and thus increase the rate constant k.
The frequency factor can be increased by increasing the number of molecules in a vessel. When there are more molecules, the opportunities for collision are increased, as shown in Figure 5.2.
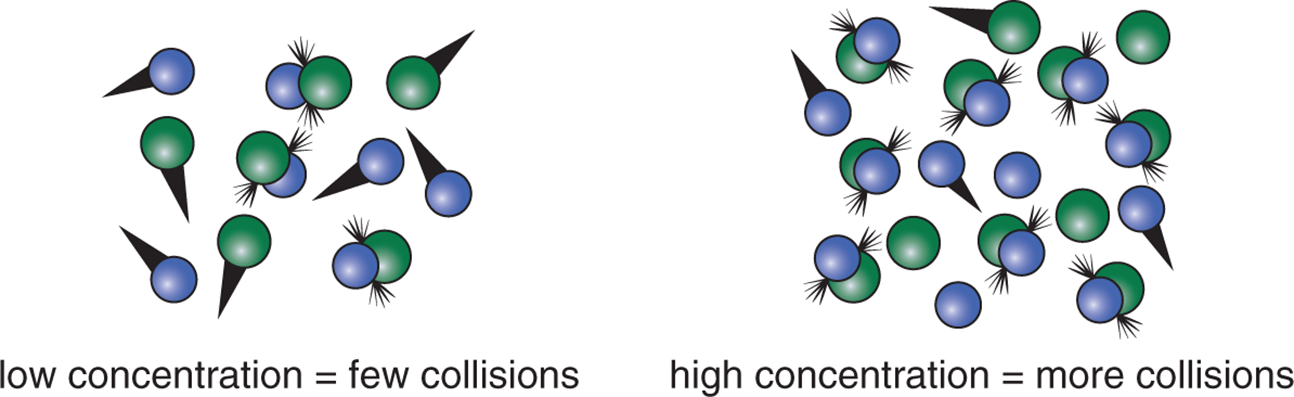
Figure 5.2. Frequency Factor (A) Is Increased by Increasing Concentration
Transition State Theory
When molecules collide with energy equal to or greater than the activation energy, they form a transition state in which the old bonds are weakened and the new bonds begin to form. The transition state then dissociates into products, fully forming the new bonds. For the reaction A2 + 2 B → 2 AB, the progress along the reaction coordinate, which traces the reaction from reactants to products, can be represented as shown in Figure 5.3.
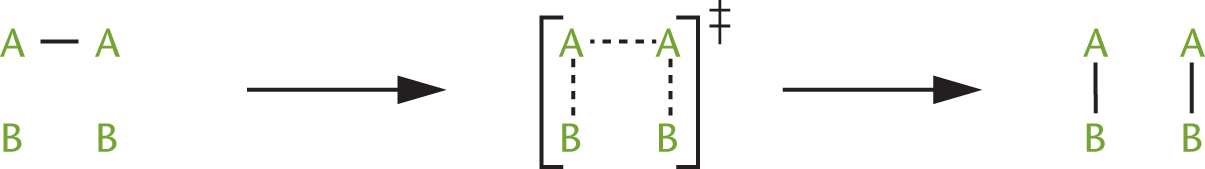
Figure 5.3. The Transition State
The transition state, also called the activated complex, has greater energy than both the reactants and the products and is denoted by the symbol ‡. The energy required to reach this transition state is the activation energy. Once an activated complex is formed, it can either dissociate into the products or revert to reactants without any additional energy input. Transition states are distinguished from reaction intermediates in that transition states are theoretical constructs that exist at the point of maximum energy, rather than distinct identities with finite lifetimes.
KEY CONCEPT
Relative to reactants and products, transition states have the highest energy. They are only theoretical structures and cannot be isolated. Nevertheless, we can still use the proposed structures to better understand the reactions in which they are involved.
A free energy diagram illustrates the relationship between the activation energy, the free energy of the reaction, and the free energy of the system. The most important features to recognize in such diagrams are the relative energies of all of the products and reactants. The free energy change of the reaction (ΔGrxn) is the difference between the free energy of the products and the free energy of the reactants. A negative free energy change indicates an exergonic reaction (energy is given off), and a positive free energy change indicates an endergonic reaction (energy is absorbed). The transition state exists at the peak of the energy diagram. The difference in free energy between the transition state and the reactants is the activation energy of the forward reaction; the difference in free energy between the transition state and the products is the activation energy of the reverse reaction.
KEY CONCEPT
- +ΔG = endergonic = energy absorbed
- –ΔG = exergonic = energy given off
For example, consider the formation of HCl from H2 and Cl2. The overall reaction is:
H2 (g) + Cl2 (g) ⇌ 2 HCl (g)
Figure 5.4 shows that the reaction is exergonic. The free energy of the products is less than the free energy of the reactants; energy is released, and the free energy change of the reaction is negative.
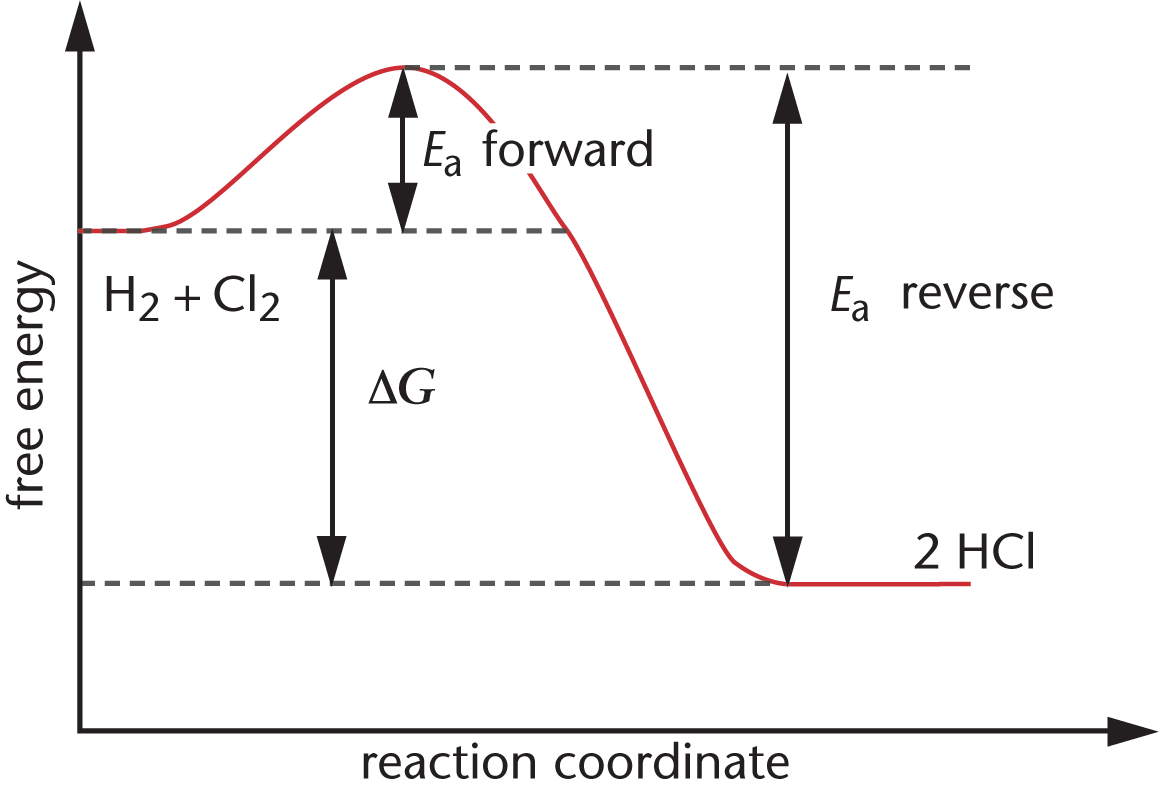
Figure 5.4. Reaction Diagram for the Formation of HCl On the left are the reactants (H2 and Cl2) and on the right are the products (2 HCl); this reaction is exergonic, and forward and reverse activation energies are shown.
MCAT EXPERTISE
Kinetics and thermodynamics should be considered separately. Note that the activation energy can be lowered without affecting the ΔG. In fact, this is how catalysts increase the rate of a reaction.
Factors Affecting Reaction Rate
Before we delve into the specifics of rate calculations, it is helpful to understand the conditions that can alter experimental rates.
Reaction Concentrations
The greater the concentrations of the reactants, the greater the number of effective collisions per unit time. Recall that this leads to an increase in the frequency factor (A) of the Arrhenius equation. Therefore, the reaction rate will increase for all but zero-order reactions, which will be discussed shortly. For reactions occurring in the gaseous state, the partial pressures of the gas reactants serve as a measure of concentration, as discussed in Chapter 8 of MCAT General Chemistry Review.
Temperature
For nearly all reactions, the reaction rate will increase as the temperature increases. Because the temperature of a substance is a measure of the particles’ average kinetic energy, increasing the temperature increases the average kinetic energy of the molecules. Consequently, the proportion of reactants gaining enough energy to surpass Ea (and thus capable of undergoing reaction) increases with higher temperature. All reactions—even the nuclear reactions shown in Figure 5.5—are temperature-dependent and experience an optimal temperature for activity.
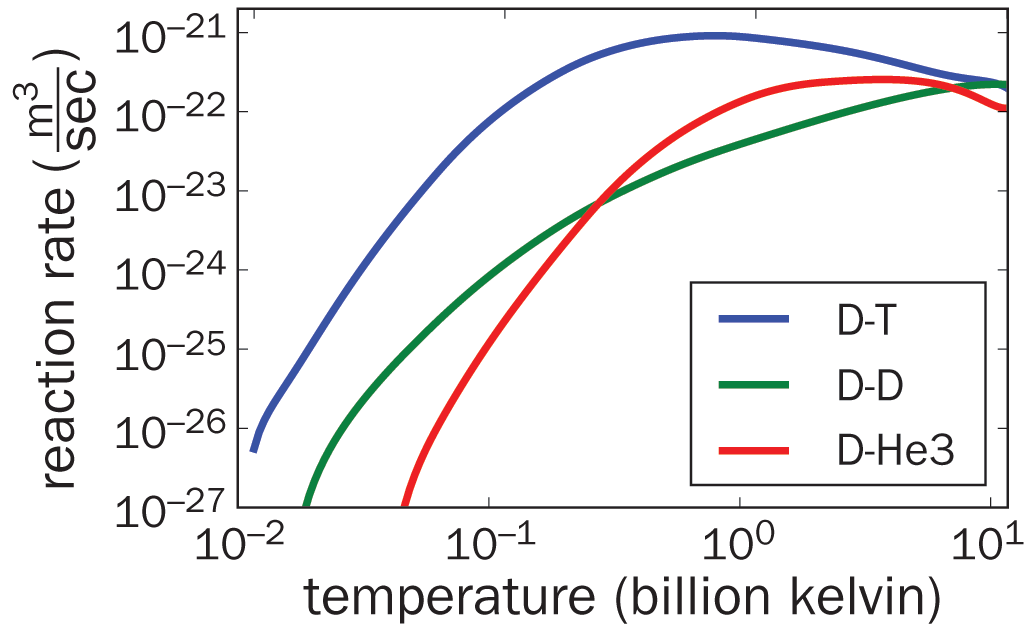
Figure 5.5. All Reactions Are Temperature-Dependent Examples shown are nuclear fusion reactions; at extreme temperatures, the nucleus itself begins to break down.
You’ll often hear that raising the temperature of a system by 10 °C will result in an approximate doubling of the reaction rate. Be careful with this approximation because it is generally true for biological systems but not so for many other systems. Further, even in biological systems, if the temperature gets too high, a catalyst may denature—and then the reaction rate plummets. Figure 5.6 shows a general curve for an enzymatic reaction that is optimal between 35 °C and 40 °C (body temperature). Notice that the curve falls sharply after 40 °C, at which point denaturation has occurred.
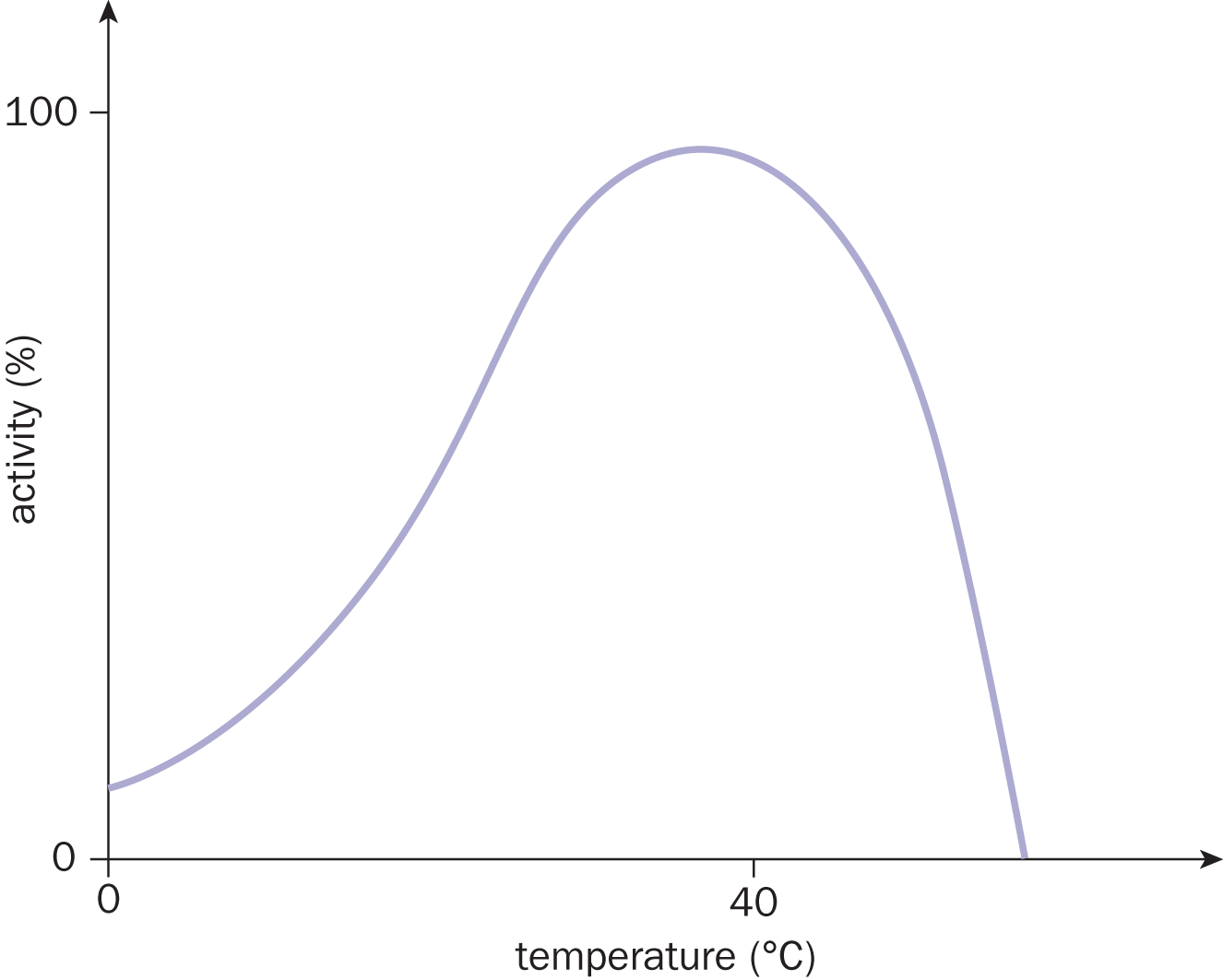
Figure 5.6. An Activity vs. Temperature Curve for a Generic Human Enzyme
Medium
The rate at which a reaction takes place may also be affected by the medium in which it takes place. Some molecules are more likely to react with each other in aqueous environments, while others are more likely to react in nonaqueous solvents, such as in dimethyl sulfoxide (DMSO) or ethanol. Furthermore, the physical state of the medium (liquid, solid, or gas) can also have a significant effect. Generally, polar solvents are preferred because their molecular dipole tends to polarize the bonds of the reactants, thereby lengthening and weakening them, permitting the reaction to occur faster.
Catalysts
Catalysts are substances that increase reaction rate without themselves being consumed in the reaction. Catalysts interact with the reactants, either by adsorption or through the formation of intermediates, and stabilize them so as to reduce the activation energy necessary for the reaction to proceed. While many catalysts, including all enzymes, chemically interact with the reactants, they return to their original chemical state upon formation of the products. They may increase the frequency of collisions between the reactants; change the relative orientation of the reactants, making a higher percentage of the collisions effective; donate electron density to the reactants; or reduce intramolecular bonding within reactant molecules. In homogeneous catalysis, the catalyst is in the same phase (solid, liquid, gas) as the reactants. In heterogeneous catalysis, the catalyst is in a distinct phase. Figure 5.7 compares the energy profiles of catalyzed and uncatalyzed reactions. Note that, depending on context, reaction profiles can use either Gibbs free energy or enthalpy for the y-axis.
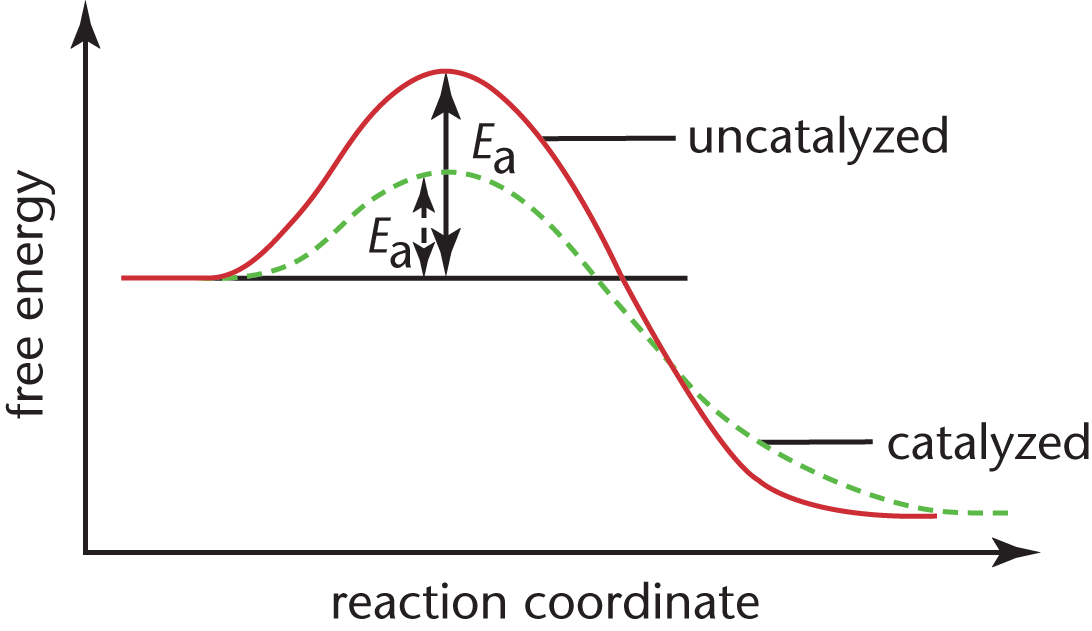
Figure 5.7. Reaction Diagram for a Catalyzed and an Uncatalyzed Reaction
Notice that the only effect of the catalyst is the decrease in the energies of activation, Ea, for both the forward and reverse reactions. The presence of the catalyst has no impact on the free energies of the reactants or the products or the difference between them. This means that catalysts change only the rates of reactions, and in fact, change the forward rate and the reverse rate by the same factor. Consequently, they have no impact whatsoever on the equilibrium position or the measurement of Keq. Remember that, as useful as catalysts are in biological and nonbiological systems, catalysts are not miracle workers: they will not transform a nonspontaneous reaction into a spontaneous one; they only make spontaneous reactions move more quickly toward equilibrium.
BRIDGE
Equilibrium, like biological homeostasis, is a dynamic process that seeks to find balance in all systems. We can use this concept to our advantage on the MCAT in all seven of the basic sciences. Equilibria are dynamic, meaning that they do undergo change but their net change will be zero.
MCAT CONCEPT CHECK 5.1:
Before you move on, assess your understanding of the material with these questions.
-
Describe in words what is occurring in the following two-step mechanism:
Step 1: A 2 B + A 2 B → A 4 B 2 Step 2: A 4 B 2 → 2A 2 + B 2
__________________________________
-
What does it mean for a step in a mechanism to be the rate-determining step?
____________________________________
-
What is activation energy?
_____________________________________
-
How does the transition state theory compare with the collision theory of chemical kinetics?
- Transition state theory: ________________________________
- Collision theory: _____________________________________
5.2 Reaction Rates
LEARNING OBJECTIVES
After Chapter 5.2, you will be able to:
- Predict the impact of changing temperature, concentration, and catalyst presence on rate of reaction for a zero, first, or second-order reaction
- Determine the rate law and rate order for a reaction, given experimental reaction rate data:
**Trial [A]initial (M) [B]initial (M) rate initial ( M s ) **
1 1.00 1.00 2.0
2 1.00 2.00 8.1
3 2.00 2.00 15.9
Reactions, unfortunately, do not come with handy built-in speedometers. To determine the rate at which a reaction proceeds, we must take measurements of the concentrations of reactants and products and note their change over time.
Definition of Rate
If we consider a generic reaction, 2 A + B → C, in which one mole of C can be produced from two moles of A and one mole of B, we can describe the rate of this reaction in terms of either the disappearance of reactants over time or the appearance of products over time. Because the reactants, by definition, are being consumed in the process of formation of the products, we place a negative sign in front of the rate expression for the reactants. For the above reaction, the rate of the reaction with respect to A is − Δ [ A] Δ t , with respect to B is − Δ [ B] Δ t , and with respect to C is + Δ [ C] Δ t . Notice that the stoichiometric coefficients for the reaction are not equal, which means that the rates of change of concentrations are not equal. Because two moles of A are consumed for every mole of B consumed, the rate of consumption of A is twice the rate of consumption of B. Furthermore, for every two moles of A consumed, only one mole of C is produced; thus, we can say that the rate of consumption of A is twice the rate of production of C. Based on the stoichiometry, we can see that the rate of consumption of B is equal to the rate of production of C. To show a standard rate of reaction in which the rates with respect to all reaction species are equal, the rate of concentration change of each species should be divided by the species’ stoichiometric coefficients. Thus, for the general reaction aA + bB → cC + dD:
rate = − Δ [ A] a Δ t = − Δ [ B] b Δ t = Δ [ C] c Δ t = Δ [ D] d Δ t
Equation 5.3
Rate is expressed in the units of moles per liter per second ( mol L · s ) or molarity per second ( M s ) .
Determination of Rate Law
In the Chemical and Physical Foundations of Biological Systems section of the MCAT, it is unlikely that the testmakers will provide a reaction equation that one can merely look at and write the correct rate law. Therefore, on the MCAT, whenever a question asks to determine the rate law for a reaction, the first thing to look for is experimental data.
MCAT EXPERTISE
Remember that the stoichiometric coefficients for the overall reaction are often different from those for the rate law and will, therefore, not be the same as the order of the reaction.
For nearly all forward, irreversible reactions, the rate is proportional to the concentrations of the reactants, with each concentration raised to some experimentally determined exponent. For the general reaction
aA + bB → cC + dD
the rate is proportional to [A]x[B]y. By including a proportionality constant, k, we can say that rate is determined according to the following equation:
rate = k[A]x[B]y
Equation 5.4
where k is the reaction rate coefficient or rate constant and the exponents x and y are the orders of the reaction. This expression is called the rate law. Remember that rate is always measured in units of concentration over time; that is, molarity per second. The exponents x and y (or x,y, and z, if there are three reactants) can be used to state the order of the reaction with respect to each reactant or overall: x is the order with respect to reactant A, and y is the order with respect to reactant B. The overall order of the reaction is the sum ofx and y. These exponents may be integers or fractions and must be determined experimentally. The MCAT will focus almost exclusively on zero-, first-, second-, and third-order reactions. In most cases, the exponents will be integers.
Before we go any further in our consideration of rate laws, we must offer a few warnings about common traps in chemical kinetics. The first—and most common—is the assumption that the orders of a reaction are the same as the stoichiometric coefficients in the balanced overall equation. Pay close attention: On the MCAT, the values of xand yare almost never the same as the stoichiometric coefficients. The orders of a reaction must be determined experimentally. There are only two cases in which the stoichiometric coefficients match the orders of the reaction. The first is when the reaction mechanism is a single step and the balanced overall reaction is reflective of the entire chemical process. The second is when the complete reaction mechanism is given and the rate-determining step is indicated. The stoichiometric coefficients on the reactant side of the rate-determining step are equal to the orders of the reaction. Occasionally, even this can get a little complicated when the rate-determining step involves an intermediate as a reactant, in which case one must derive the intermediate molecule’s concentration by the law of mass action (that is, the equilibrium constant expression) for the step that produced the intermediate.
MCAT EXPERTISE
Note that the exponents in the rate law are not equal to the stoichiometric coefficients, unless the reaction occurs via a single-step mechanism. Note that product concentrations never appear in a rate law. Don’t fall into the exceedingly common trap of confusing the rate law with an equilibrium expression!
The second common trap is mistaking the equilibrium constant expression (law of mass action) for the rate law. The expressions for both look similar; if you’re not alert on Test Day, you may mistake one for the other or use one when you should be using the other. The expression for equilibrium includes the concentrations of all the species in the reaction, both reactants and products. The expression for chemical kinetics—the rate law expression—includes only the reactants. Keq indicates where the reaction’s equilibrium position lies. The rate indicates how quickly the reaction will get there.
The third trap regards the rate constant, k. Technically speaking, k is not a constant because its particular value for any specific chemical reaction will depend on the activation energy for that reaction and the temperature at which the reaction takes place. However, for a specific reaction, at a specific temperature, the rate constant is indeed a constant. For a reversible reaction, the Keq is equal to the ratio of the rate constant for the forward reaction, k, divided by the rate constant for the reverse reaction, k–1.
The fourth and final trap is that the notion and principles of equilibrium apply to the system only at the end of the reaction; that is, after the system has already reached equilibrium. On the other hand, while the reaction rate can theoretically be measured at any time, it is usually measured at or near the beginning of the reaction to minimize the effects of the reverse reaction.
Experimental Determination of Rate Law
This has already been stated a few times, but it bears repeating: The values of k, x, and y in the rate law equation (rate = k[A]x[B]y) must be determined experimentally for a given reaction at a given temperature. Although rate laws can be quite complex and the orders of the reaction difficult to discern, the MCAT limits its coverage of this topic to fairly straightforward reaction mechanisms, experimental data, and rate laws.
On the MCAT, experimental data for determining rate order is usually provided as a chart that includes the initial concentrations of the reactants and the initial rates of product formation as a function of the reactant concentrations. Often, the data for three or four trials are included in this chart.
To use this data, identify a pair of trials in which the concentration of one of the reactants is changed while the concentrations of all other reactants remain constant. Under these conditions, any change in the rate of product formation from one trial to the other (if there is any change) is fully attributable to the change in concentration of that one reactant. Consider a reaction with two reactants, A and B, forming product C. Imagine two trials in which the concentration of A is constant, while the concentration of B doubles. If the rate of the formation of product C has subsequently quadrupled, then the exponent on [B] must be two. Why? Looking at the generic rate law (rate = k[A]x[B]y), the logic should look something like this: Doubling [B] has resulted in a quadrupling of the rate, so to determine the order of the reaction, y,with respect to B, I need to calculate the exponent to which the number 2 must be raised to equal 4. Because 2y = 4, y= 2.
MCAT EXPERTISE
The testmakers love rate problems. Why? Because solving these questions requires a real understanding of proportionality and variable relationships. With practice, you’ll be able to do these quickly in your head with minimal paper-and-pencil calculations. Remember to look for pairs of reaction trials in which the concentration of only one species changes while the others remain constant.
The next step is to repeat this process for the other reactant, using data from a different pair of trials, making sure that the concentration of only the reactant we are trying to analyze is changed from one trial to the other while the concentrations of all other reactants remain the same. Once the orders of the reaction have been determined with respect to each reactant, we can write the complete rate law, replacing the exponents x and y with actual numbers. To determine the value of the rate constant k, plug in actual values from any one of the trials; pick whichever trial has the most arithmetically convenient numbers.
Example: Given the data below, find the rate law for the following reaction at 300 K:
A + B → C + D
Trial [A]initial (M) [B]initial (M) rate initial ( M s )
1 1.00 1.00 2.0
2 1.00 2.00 8.1
3 2.00 2.00 15.9
Solution: First, look for two trials in which the concentrations of all but one of the substances are held constant.
In Trials 1 and 2, the concentration of A is kept constant, while the concentration of B is doubled. The rate increases by a factor of approximately 4. Since k and [A] are constant between the two trials, the rate is proportional to [B] raised to some power (the symbol ∝ means “is proportional to”):
rate = k [ A ] x [ B ] y → k and [A] are constant rate ∝ [ B ] y
Specifically, the relationship between the change in rate and the change in concentration of B can be written as: Δrate=Δ[B]y.For this specific set of data, the proportionality becomes: 4 =
[ 2 ]
y
.
In other words, since the rate was multiplied by 4, and [B] was multiplied by 2, y must be equal to 2. Based on what is known so far, the rate law becomes rate = k [ A ] x [ B ] 2 .
In Trials 2 and 3, the concentration of B is kept constant, while the concentration of A is doubled. The rate increases by a factor of approximately 2. Since k and [B] are constant between the two trials, rate is proportional to [A] raised to some power:
rate = k [ A ] x [ B ] 2 → k and [B] are constant rate ∝ [ A ] x
The relationship between the change in rate and the change in concentration of B can be written as: Δrate=Δ[A]x. For this specific set of data, the proportionality becomes: 2
=
[ 2 ]
x
.
In other words, since the rate was multiplied by 2, and [A] was multiplied by 2, x must be equal to 1.
Therefore, rate = k[A]1[B]2, more typically written as rate = k[A][B]2 as raising a value to the first power is equivalent to the value (e.g., 21 = 2). The order of the reaction is 1 with respect to A and 2 with respect to B; the overall reaction order is thus 1 + 2 = 3.
To calculate k, substitute the values from any one of the trials into the rate law. In this case, trial 1 is chosen because the numbers are straightforward to manipulate:
rate = k [ A ] [ B ] 2 2.0 M s = k [ 1.00 M ] [ 1.00 M ] 2 k = 2.0 M − 2 s − 1
Therefore, the final rate law is rate = 2.0 M–2 s–1 [A][B]2.
Reaction Orders
We classify chemical reactions as zero-order, first-order, second-order, higher-order, or mixed-order on the basis of kinetics. We will continue to consider the generic reaction aA + bB → cC + dD for this discussion.
Zero-Order Reaction
A zero-order reaction is one in which the rate of formation of product C is independent of changes in concentrations of any of the reactants, A and B. These reactions have a constant reaction rate equal to the rate constant (rate coefficient), k. The rate law for a zero-order reaction is:
rate = k[A]0[B]0 = k
where k has units of M s . Remember that the rate constant itself is dependent on temperature; thus, it is possible to change the rate for a zero-order reaction by changing the temperature. The only other way to change the rate of a zero-order reaction is by the addition of a catalyst, which lowers the activation energy, thereby increasing the value of k.
MCAT EXPERTISE
Temperature and the addition of a catalyst are the only factors that can change the rate of a zero-order reaction.
Plotting a zero-order reaction on a concentration vs. time curve results in a linear graph, as shown in Figure 5.8. This line shows that the rate of formation of product is independent of the concentration of reactant. The slope of such a line is the opposite of the rate constant, k.
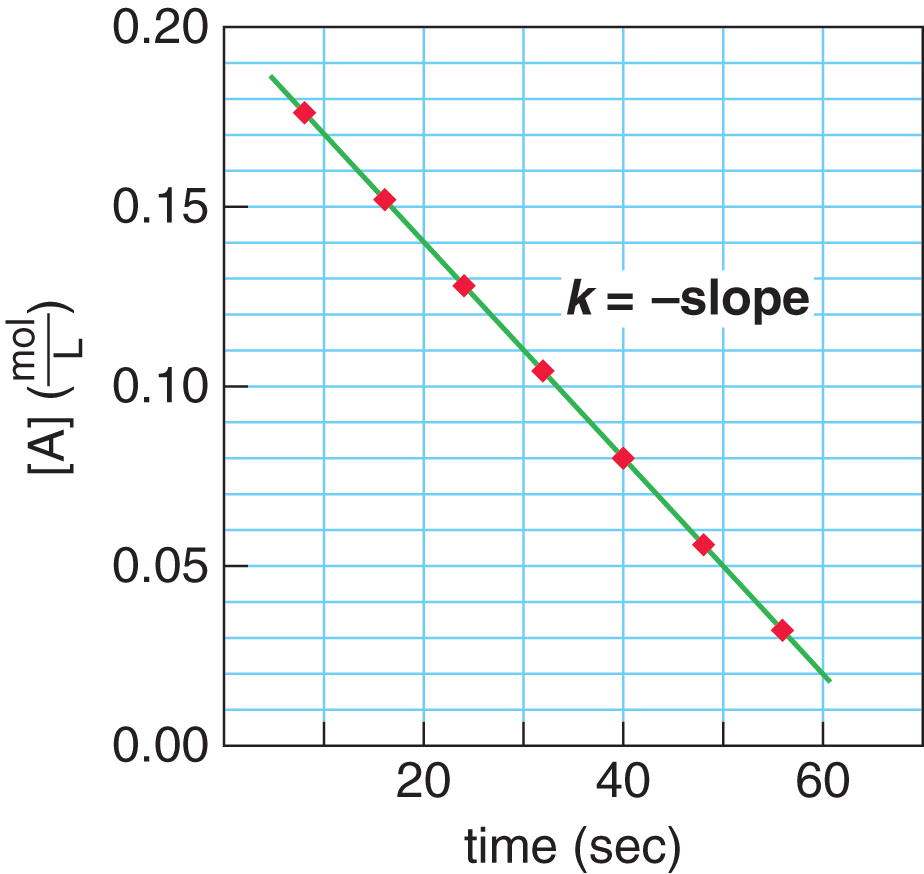
Figure 5.8. Kinetics of a Zero-Order Reaction Note that the rate of reaction,k, is the opposite of the slope.
First-Order Reaction
A first-order reaction has a rate that is directly proportional to only one reactant, such that doubling the concentration of that reactant results in a doubling of the rate of formation of the product. The rate law for a first-order reaction is
rate = k[A]1 or rate = k[B]1
where k has units of s–1. A classic example of a first-order reaction is the process of radioactive decay. From the rate law, in which the rate of decrease of the amount of a radioactive isotope A is proportional to the amount of A:
rate = − Δ [ A ] Δ t = k [ A ]
The concentration of radioactive substance A at any time t can be expressed mathematically as:
[A]t = [A]0e–kt
Equation 5.5
where [A]t is the concentration of A at time t, [A]0 is the initial concentration of A, k is the rate constant, andt is time. It is important to recognize that a first-order rate law with a single reactant suggests that the reaction begins when the molecule undergoes a chemical change all by itself, without a chemical interaction, and usually without a physical interaction with any other molecule.
Plotting a first-order reaction on a concentration vs. time curve results in a nonlinear graph, as shown in Figure 5.9. This curve shows that the rate of formation of product is dependent on the concentration of reactant. Plotting ln [A] vs. time reveals a straight line; the slope of such a line is the opposite of the rate constant, k.
![left: concentration of A vs. time; downsloping line with upward curvature; right: ln[A] vs. time; straight line with slope = -k](../figures/03-general-chemistry/ch05/fig5.9.png)
Figure 5.9. Kinetics of a First-Order Reaction **On the left, note that the rate of reaction is dependent on reactant concentration; on the right, note that the rate constant is the opposite of the slope of a graph of ln [A] vs. time.**
Second-Order Reaction
A second-order reaction has a rate that is proportional to either the concentrations of two reactants or to the square of the concentration of a single reactant. The following rate laws all reflect second-order reactions:
rate = k[A]1[B]1 or rate = k[A]2 or rate = k[B]2
where k has units of M–1 s–1. It is important to recognize that a second-order rate law often suggests a physical collision between two reactant molecules, especially if the rate law is first-order with respect to each of the two reactants.
Plotting a reaction that is second-order with respect to a single reactant on a concentration vs. time curve results in a nonlinear graph, as shown in Figure 5.10. This curve shows that the rate of formation of product is dependent on the concentration of reactant. Plotting 1 [ A ] vs. time reveals a linear curve; the slope of such a curve is equal to the rate constant, k.
![left: concentration of A vs. time; downsloping line with upward curvature; right: 1/[A] vs. time; slope = k](../figures/03-general-chemistry/ch05/fig5.10.png)
Figure 5.10. Kinetics of a Second-Order Reaction **On the left, note that the rate of reaction is dependent on reactant concentration; on the right, note that the rate constant is equal to the slope of a graph of*
1
[ A ]
vs.* time.**
Higher-Order Reactions
Fortunately, there are very few noteworthy reactions in which a single reaction step involves a termolecular process; in other words, there are few processes with third-order rates. This is because it is far more rare for three particles to collide simultaneously with the correct orientation and sufficient energy to undergo a reaction.
Mixed-Order Reactions
Mixed-order reactions sometimes refer to non-integer orders (fractions) and in other cases to reactions with rate orders that vary over the course of the reaction. Fractions are more specifically described as broken-order. In recent times, the term mixed-order has come to refer solely to reactions that change order over time. Knowing those two definitions will be sufficient for Test Day.
An example of a mixed-order rate law is given by:
rate = k 1 [ C ] [ A ] 2 k 2 + k 3 [ A ]
where A represents a single reactant and C, a catalyst. The overall reaction and its mechanism are beyond the relevance and scope of the MCAT, and the derivation of this rate law is even more unnecessary for Test Day; however, understanding what is implied by this equation is important. The result of the large value for [A] at the beginning of the reaction is that k3[A] ≫ k2, and the reaction will appear to be first-order with respect to A. (Note: The symbol "≫" means "much-greater-than", implying, in this context, that the contribution of k2 to the denominator "k2 + [A]k3" is negligible.) At the end of the reaction, when [A] is low, k2 ≫ k3[A], making the reaction appear second-order with respect to A. While the MCAT will not ask you to derive a rate expression for a mixed-order reaction, you are responsible for being able to recognize how the rate order changes as the reactant concentration changes.
MCAT CONCEPT CHECK 5.2:
Before you move on, assess your understanding of the material with these questions.
-
Describe the effects the following conditions would have on the initial rate of reaction, given the reaction order: (Examples: rate increased, rate divided by 2, rate unaffected)
Conditions Zero-Order First-Order Second-Order Temperature lowered All reactants’ concentrations doubled Catalyst added
-
Determine the rate law and rate order for the following reaction:
A + B + C → D
**Trial [A]initial (M) [B]initial (M) [C]initial (M) rate initial ( M s ) **
1 1.00 1.00 1.00 2.0
2 1.00 2.00 1.00 2.1
3 2.00 1.00 1.00 15.9
4 2.00 1.00 2.00 32.2
Conclusion
We began with a consideration of chemical reactions and the mechanisms that illustrate the individual steps necessary to transform reactants into products. We demonstrated the way to derive a reaction’s rate law through the analysis of experimental data, and we looked at the factors that can affect the rates of chemical reactions.
After such an overview, you should begin to appreciate that many chemical principles in the human body rely on the principles of chemical kinetics. Why does the body maintain a certain temperature? Primarily to stabilize the enzymes that catalyze the metabolic reactions necessary for life. Why does the body maintain a pH buffer? Altering the concentration of protons affects not only the ability of an enzyme to maintain its secondary, tertiary, and quaternary structure, but can also directly affect the collisions between reactants. You will begin to appreciate these and many other questions from a clinical perspective throughout your medical career. In the next chapter, we will investigate chemical equilibria, which—although related to kinetics—are distinct (and commonly confused!) topics.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Chemical Kinetics
- The change in Gibbs free energy (ΔG) determines whether or not a reaction is spontaneous.
- Chemical mechanisms propose a series of steps that make up the overall reaction.
- Intermediates are molecules that exist within the course of a reaction but are neither reactants nor products overall.
- The slowest step, also known as the rate-determining step, limits the maximum rate at which the reaction can proceed.
- The collision theory states that a reaction rate is proportional to the number of effective collisions between the reacting molecules.
- For a collision to be effective, molecules must be in the proper orientation and have sufficient kinetic energy to exceed the activation energy.
- The Arrhenius equation is a mathematical way of representing collision theory.
- The transition state theory states that molecules form a transition state or activated complex during a reaction in which the old bonds are partially dissociated and the new bonds are partially formed.
- From the transition state, the reaction can proceed toward products or revert back to reactants.
- The transition state is the highest point on a free energy reaction diagram.
- Reaction rates can be affected by a number of factors.
- Increasing the concentration of reactant will increase reaction rate (except for zero-order reactions) because there are more effective collisions per time.
- Increasing the temperature will increase reaction rate because the particles’ kinetic energy is increased.
- Changing the medium can increase or decrease reaction rate, depending on how the reactants interact with the medium.
- Adding a catalyst increases reaction rate because it lowers the activation energy. Homogeneous catalysts are the same phase as the reactants; heterogeneous catalysts are a different phase.
Reaction Rates
- Reaction rates are measured in terms of the rate of disappearance of a reactant or appearance of a product.
- Rate laws take the form of rate = k[A]x[B]y.
- The rate orders usually do not match the stoichiometric coefficients.
- Rate laws must be determined from experimental data.
- The rate order of a reaction is the sum of all individual rate orders in the rate law.
- Zero-order reactions have a constant rate that does not depend on the concentration of reactant.
- The rate of a zero-order reaction can only be affected by changing the temperature or adding a catalyst.
- A concentration vs. time curve of a zero-order reaction is a straight line; the slope of such a line is equal to –k.
- First-order reactions have a nonconstant rate that depends on the concentration of reactant.
- A concentration vs. time curve of a first-order reaction is nonlinear.
- The slope of a ln [A] vs. time plot is –k for a first-order reaction.
- Second-order reactions have a nonconstant rate that depends on the concentration of reactant.
- A concentration vs. time curve of a second-order reaction is nonlinear.
- The slope of a 1 [ A ] vs. time plot is k for a second-order reaction.
- Broken-order reactions are those with noninteger orders.
- Mixed-order reactions are those that have a rate order that changes over time.
ANSWERS TO CONCEPT CHECKS
**5.1**
- Two molecules of A2B come together in a combination reaction to form an intermediate, A4B2, which subsequently decomposes to produce the final products, two molecules of A2 and one molecule of B2.
- The rate-determining step is the slowest step of a reaction. It determines the overall rate of the reaction because the reaction can only proceed as fast as the rate at which this step occurs.
- The activation energy is the minimum energy needed for a chemical reaction to occur.
- Both theories require a certain activation energy to be overcome in order for a reaction to occur (therefore not all reactions will occur). The transition state theory focuses on forming a high-energy activated complex that can then proceed forward or backward, forming the products or reverting to the reactants, respectively. The collision theory focuses on the energy and orientation of reactants, and considers each potential reaction to be “all-or-nothing” (either there is enough energy to form the products, or there is not).
**5.2**
-
Conditions Zero-Order First-Order Second-Order Temperature lowered rate decreased rate decreased rate decreased
All reactants’ concentrations doubled rate unaffected rate doubled rate multiplied by 4
Catalyst added rate increased rate increased rate increased
- This question asks for the rate law and rate order for the following reaction:
A + B + C → D
**Trial [A]initial (M) [B]initial (M) [C]initial (M) rate initial ( M s ) **
1 1.00 1.00 1.00 2.0
2 1.00 2.00 1.00 2.1
3 2.00 1.00 1.00 15.9
4 2.00 1.00 2.00 32.2
Start by writing the generic rate law for the reaction: rate = k[A]x[B]y[C]z.
In a complex rate law problem, always check for the possibility of a reagent that has no impact on the rate law. Looking at Trials 1 and 2, the concentration of B is doubled with no change in the rate. Thus, reagent B has no impact on the rate law, and its exponent is zero. The rate law can be updated to rate = k[A]x[B]0[C]z.
Next, compare Trials 1 and 3. The concentration of A doubles, the concentrations of B and C remain constant, and the rate increases by a factor of approximately 8. This results in the proportionality Δrate = Δ A x , so 8 = 2 x , giving x = 3. The rate law can now be updated to rate = k[A]3[B]0[C]z.
Finally, compare Trials 3 and 4. The concentration of C doubles, the concentrations of A and B remain constant, and the rate approximately doubles. This results in the proportionality Δrate = Δ C z , so 2 = 2 z , giving z = 1. The rate law can now be updated to rate = k[A]3[B]0[C]1.
Thus, the final rate law is: rate = k[A]3[B]0[C]1 = k[A]3[C]. The rate order is 3 + 0 + 1 = 4.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
Based on the information given in the question, the rate is first-order with respect to the concentration of the first reactant; when the concentration of that reactant doubles, the rate also doubles. Because the reaction is third-order, the sum of the exponents in the rate law must be equal to 3. Therefore, the reaction order with respect to the other reactant must be 3 – 1 = 2. If the concentration of this second reactant is multiplied by 1 2 , the rate will be multiplied by ( 1 2 ) 2 = 1 4 .
2. D
Before you try to answer this question, you should draw a free energy diagram for the system.
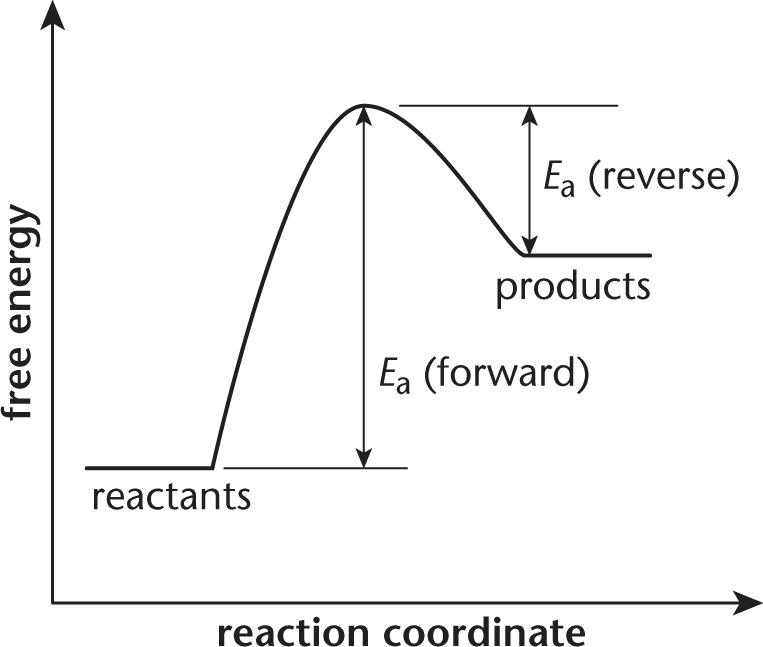
If the activation energy of the forward reaction is greater than the activation energy of the reverse reaction, then the products must have a higher free energy than the reactants. The overall energy of the system is higher at the end than it was in the beginning. The net free energy change is positive, indicating an endergonic (nonspontaneous) reaction. The terms endothermic, (A), and exothermic, (B), are associated with enthalpy. While free energy does depend on enthalpy, it also depends on entropy; there is not enough information in the question stem to reliably determine the sign of the entropy change of the reaction.
3. D
A second-order reaction can be second-order with respect to one reactant, or first-order with respect to two different reactants. In this case, one reactant was increased by a factor of 4. If the reaction is second-order with respect to this reactant, the rate law will be rate = k[A]2[B]0 and the rate will increase by a factor of 16. If it is first-order with respect to this reactant and first-order with respect to another reactant, the rate law will be rate = k[A]1[B]1, and the rate will increase by a factor of 4. We do not know which of these is the correct rate law and, thus, cannot determine the effect on the rate.
4. A
By definition, zero-order reactions are unaffected by the concentrations of any reactants in the reaction. Thus, changing the concentrations of these reactants will not affect the rate.
5. D
The question asks which alteration does NOT affect the rate of the reaction. Temperature directly affects the rate constant (k), making (A) incorrect. Changing the partial pressure of a gas will affect the number of effective collisions per time. This makes (B) incorrect—but note that concentration changes will not affect the rate of zero-order reactions. Solvents affect the rate of reactions depending on how the reactants interact with the solvent, making (C) incorrect. Removing the product of an irreversible reaction, (D), should not affect the rate of the reaction because the rate law does not depend on the concentrations of products.
6. D
While increasing the concentration of reactants can alter the reaction rate in first- or higher-order reactions, saturated solutions containing a catalyst have a maximum turnover rate and cannot increase the rate constant or the reaction rate any higher by adding more reactant molecules.
7. A
If the sum of the exponents (orders) of the concentrations of each species in the rate law is equal to 2, then the reaction is second-order. The exponents in the rate law are unrelated to stoichiometric coefficients, so NO2 and Br2 could have any stoichiometric coefficients in the original reaction and still be a second-order reaction, invalidating statement II. Statement III is incorrect because the rate can be affected by a wide variety of compounds. A catalyst, for example, could increase the rate.
8. C
Start with the generic rate law: rate = k[XH4]x[O2]y. In the first two trials, the concentration of XH4 is held constant while the concentration of O2 is multiplied by 4, and the rate of the reaction also increases by a factor of approximately 4. This gives the proportion Δrate = [O 2 ] y , or 4 = 4 y , meaning y = 1. The rate law can be updated to: rate = k[XH4]x[O2]1.
In the last two trials, the concentration of O2 is held constant while the concentration of XH4 is doubled, and the rate of the reaction is increased by a factor of approximately 4. This gives Δrate = [XH 4 ] x , or 4 = 2 x , and x = 2. The rate law can be updated to: rate = k[XH4]2[O2]1. The final version of the rate law is: rate = k[XH4]2[O2].
9. B
By definition, a catalyst increases the rate of a reaction by lowering the activation energy, making it easier for both the forward and reverse reactions to overcome this energy barrier. Catalysts are neither used up in the reaction, nor do they alter the equilibrium of a reaction, eliminating (A) and (C). Finally, catalysts stabilize the transition state by lowering its energy, not raising it, eliminating (D).
10. C
The overall order of a reaction is the sum of the individual orders in the reaction. Therefore, the rate order is 0 + 2 + 1 = 3.
11. D
A system is exergonic if energy is released by the reaction. For exergonic reactions, the net energy change is negative, and the free energy of the final products is lower than the free energy of the initial reactants. Point E, which represents the energy of the final products, is lower on the energy diagram than point A, which represents the energy of initial reactants. Thus, energy must have been given off, and the reaction is exergonic.
12. B
The activation energy of a reaction is represented by the distance on the y-axis from the energy of the reactants to the peak energy prior to formation of products. The activation energy of the first step of the forward reaction, for example, is equal to the distance along they-axis from point A to point B. The largest energy increase on this graph occurs during the progress from point E to point D, which represents the first step of the reverse reaction.
13. D
Intermediates exist at “valleys” in reaction diagrams. Reactants, (A), are represented by point A. Products, (B), are represented by point E. Transition states, (C), are represented by points B and D.
14. D
To answer this question, recall that the slow step of a reaction is the rate-determining step. The rate is always related to the concentrations of the reactants in the rate-determining step (not the overall reaction), so NO2 is the only compound that should be included in the correct answer. The concentration of NO2 is squared in the rate law because the question stem tells us that the system obeys second-order kinetics.
15. D
The faster a reaction can reach its activation energy, the faster it will proceed to completion. Because this question states that all conditions are equal, the reaction with the lowest activation energy will have the fastest rate. In the diagram, (D) has the lowest activation energy.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(5.1) Collision theory: rate = Z × f
(5.2) Arrhenius equation: k = A e − E a R T
(5.3) Definition of rate: rate = − Δ [ A] a Δ t = − Δ [ B ] b Δ t = Δ [ C ] c Δ t = Δ [ D ] d Δ t for the general reaction a A + b B → c C + d D
(5.4) Rate law: rate= k[A]x[B]y
(5.5) Radioactive decay: [A]t = [A]0e–kt
SHARED CONCEPTS
Biochemistry Chapter 2
Enzymes
General Chemistry Chapter 3
Bonding and Chemical Interactions
General Chemistry Chapter 6
Equilibrium
General Chemistry Chapter 7
Thermochemistry
Organic Chemistry Chapter 4
Analyzing Organic Reactions
Physics and Math Chapter 3
Thermodynamics