Chapter 6: Equilibrium

Chapter 6: Equilibrium
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
A reaction is found to stop just before all reactants are converted to products. Which of the following could be true about this reaction?
- The reaction is irreversible, and the forward rate is greater than the reverse rate.
- The reaction is irreversible, and the reverse rate is too large for products to form.
- The reaction is reversible, and the forward rate is equal to the reverse rate.
- The reaction is reversible, and the reverse rate is greater than the forward rate.
-
What is the equilibrium expression for the reaction Cu2SO4 (s) ⇌ 2 Cu+ (aq) + SO42− (aq)?
- [Cu + ] 2 [ SO 4 2 − ] [ Cu 2 SO 4 ]
- 2 × [Cu + ] [ SO 4 2 − ] [ Cu 2 SO 4 ]
- [Cu + ] [ SO 4 2 − ]
- [Cu + ] 2 [ SO 4 2 − ]
-
Carbonated beverages are produced by dissolving carbon dioxide in water to produce carbonic acid:
CO2 (g) + H2O (l) ⇌ H2CO3 (aq)
When a bottle containing carbonated water is opened, the taste of the beverage gradually changes as the carbonation is lost. Which of the following statements best explains this phenomenon?
- The change in pressure and volume causes the reaction to shift to the left, thereby decreasing the amount of aqueous carbonic acid.
- The change in pressure and volume causes the reaction to shift to the right, thereby decreasing the amount of gaseous carbon dioxide.
- Carbonic acid reacts with environmental oxygen and nitrogen.
- Carbon dioxide reacts with environmental oxygen and nitrogen.
-
Which of the following best describes how to favor the production of the kinetic product over the thermodynamic product?
- High temperature and short reaction time
- Low temperature and short reaction time
- High temperature and long reaction time
- Low temperature and long reaction time
-
If Kc ≫ 1:
- the equilibrium mixture will favor products over reactants.
- the equilibrium mixture will favor reactants over products.
- the equilibrium concentrations of reactants and products are equal.
- the reaction is essentially irreversible.
-
Acetic acid dissociates in solution according to the following equation:
CH3COOH ⇌ CH3COO– + H+
If sodium acetate is added to a solution of acetic acid in excess water, which of the following effects would be observed in the solution?
- Decreased pH
- Increased pH
- Decreased pKeq (pKa)
- Increased pKeq (pKa)
Questions 7 and 8 refer to the reaction below:
FeI (aq) + I2 (g) → FeI3 (aq)
- Which of the following would increase the formation of product?
- Decreasing the volume of the container
- Decreasing the pressure of the container
- Increasing the volume of the container
- Decreasing the volume of the container while maintaining a constant pressure
-
If this reaction were exothermic, what effect would decreasing the temperature have on the equilibrium?
- The forward reaction rate and the reverse reaction rate both increase.
- The forward reaction rate decreases while the reverse reaction rate increases.
- The forward reaction rate increases while the reverse reaction rate decreases.
- The forward reaction rate and the reverse reaction rate both decrease.
- Which of the following actions does NOT affect the equilibrium position of a reaction?
- Adding or removing heat.
- Adding or removing a catalyst.
- Increasing or decreasing concentrations of reactants.
- Increasing or decreasing volumes of reactants.
-
In a sealed 1 L container, 1 mole of nitrogen gas reacts with 3 moles of hydrogen gas to form 0.05 moles of NH3 at equilibrium. Which of the following is closest to the Kc of the reaction?
- 0.0001
- 0.001
- 0.01
- 0.1
-
Increasing temperature can alter the Keq of a reaction. Why might increasing temperature indefinitely be unfavorable for changing reaction conditions?
- The equilibrium constant has a definite limit that cannot be surpassed.
- The products or reactants can decompose at high temperatures.
- Increasing temperature would decrease pressure, which may or may not alter reaction conditions.
- If a reaction is irreversible, its Keq will resist changes in temperature.
-
How does the change in free energy compare between the kinetic and thermodynamic product?
- The free energy change for the kinetic product is greater since it is less stable.
- The free energy change for the kinetic product is greater since it is more stable.
- The free energy change for the thermodynamic product is greater since it is less stable.
- The free energy change for the thermodynamic product is greater since it is more stable.
-
Compound A has a Ka (equilibrium constant of acid dissociation) of approximately 10–4. Which of the following compounds is most likely to react with a solution of compound A?
- HNO3
- NO2
- NH3
- N2O5
-
Consider the following two reactions:
3 A + 2 B ⇌ 3 C + 4 D (Reaction 1) 4 D + 3 C ⇌ 3 A + 2 B (Reaction 2)
If Keq for reaction 1 is equal to 0.1, what is Keq for reaction 2?
- 0.1
- 1
- 10
- 100
-
Which of the following statements best describes the effect of lowering the temperature of the following reaction?
A + B ⇌ C + D Δ H = − 1.12 kJ mol
- [C] and [D] would increase.
- [A] and [B] would increase.
- ΔH would increase.
- ΔH would decrease.
Answer Key
- C
- D
- A
- B
- A
- B
- A
- C
- B
- A
- B
- D
- C
- C
- A
Chapter 6: Equilibrium
CHAPTER 6
EQUILIBRIUM
In This Chapter
6.1 Equilibrium
Dynamic Equilibria and Reversibility
Law of Mass Action
Reaction Quotient
Properties of the Law of Mass Action
Equilibrium Calculations
6.2 Le Châtelier’s Principle
Changes in Concentration
Changes in Pressure (and Volume)
Changes in Temperature
6.3 Kinetic and Thermodynamic Control Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 4% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1D: Principles of bioenergetics and fuel molecule metabolism
5A: Unique nature of water and its solutions
5E: Principles of chemical thermodynamics and kinetics
MCAT EXPERTISE
The AAMC has shown that it will only rarely directly test the details of equilibrium in this chapter. However, you’ll need an excellent conceptual understanding of equilibrium to master many other high-yield topics, such as acid-base chemistry (Chapter 10 of MCAT General Chemistry Review) and enzymes (Chapter 2 of MCAT Biochemistry Review).
Introduction
You’re on the first call of your pediatrics rotation. You get a page: Come to the emergency room, now, the resident says. They just brought in a kid with DKA. DKA, as you know, stands for diabetic ketoacidosis and is a fairly common way for undiagnosed type I diabetes mellitus to present. You remember from your second-year classes about endocrine pathophysiology that ketoacidosis can arise as a result of the body’s metabolism of fatty acids when insulin production shuts down. Fatty acids are metabolized into ketone bodies as an alternative energy source to glucose. Some of the ketones produced are ketoacids, and as the diabetic crisis continues and worsens, the concentration of these ketoacids increases (termed metabolic acidosis), resulting in a plasma pH below 7.35.
As you enter the child’s room, the examination is already under way; the child is young, about ten years old, conscious but agitated, and the most obvious sign—which you notice immediately—is rapid, shallow breathing. You ask why the child is hyperventilating, and the resident takes a piece of paper and writes the following:
CO2 (g) + H2O (l) ⇌ H2CO3 (aq) ⇌ H+ (aq) + HCO3− (aq)
The child’s breathing begins to slow and realization dawns on you. *It’s Le Châtelier’s principle! The respiratory system is trying to compensate for the metabolic acidosis; the increased breathing rate allows the child to blow off more CO2, which causes the equilibrium to shift to the left. Hydrogen ions combine with bicarbonate ions to produce carbonic acid, which decomposes into CO2** gas that’s expelled from the lungs. The result is a decrease in the plasma hydrogen ion concentration, which stabilizes the pH and keeps it from getting too low. Wow, chemistry really is essential for medical school!*
Chemical equilibrium is the dynamic state of a chemical reaction in which the concentrations of reactants and products stabilize over time in a low-energy configuration. Pay particular attention to the concepts of chemical equilibrium because we will return to these topics during our review of solutions, acid–base chemistry, and oxidation–reduction reactions.
6.1 Equilibrium
LEARNING OBJECTIVES
After Chapter 6.1, you will be able to:
- Determine the sign of ΔG and the direction of a reaction given its Keq constant
- Calculate Keq for a reaction
- Write the equilibrium constant expression for a reaction:
3 H2 (g) + N2 (g) ⇌ 2 NH3 (g)
In the previous chapter, we danced around the term equilibrium. We warned you not to confuse the chemical equilibrium expression for the rate expression. We stressed that catalysts make reactions go faster toward their equilibrium position, but that they can’t actually change the equilibrium position or alter the value of Keq. The principles and concepts that are the focus of this chapter will direct our discussion in the upcoming chapters about some of the most important general chemistry topics for the MCAT: solutions, acids and bases, and oxidation–reduction reactions.
Dynamic Equilibria and Reversibility
So far, most of the reactions we’ve covered are irreversible; that is, the reaction proceeds in one direction only, the reaction goes to completion, and the maximum amount of product formed is determined by the amount of limiting reagent initially present. Reversible reactions are those in which the reaction can proceed in one of two ways: forward (toward the products or “to the right”) and reverse (toward the reactants or “to the left”). Reversible reactions usually do not proceed to completion because the products can react together to reform the reactants. When the reaction system is closed and no reactants or products are added or removed, the system will eventually settle into a state in which the rate of the forward reaction equals the rate of the reverse reaction and the concentrations of the products and reactants remain constant. In this dynamic equilibrium, the forward and reverse reactions are still occurring—they haven’t stopped, as they do in a static equilibrium—but they are going at the same rate; thus, there is no net change in the concentrations of the products or reactants, as shown in Figure 6.1.
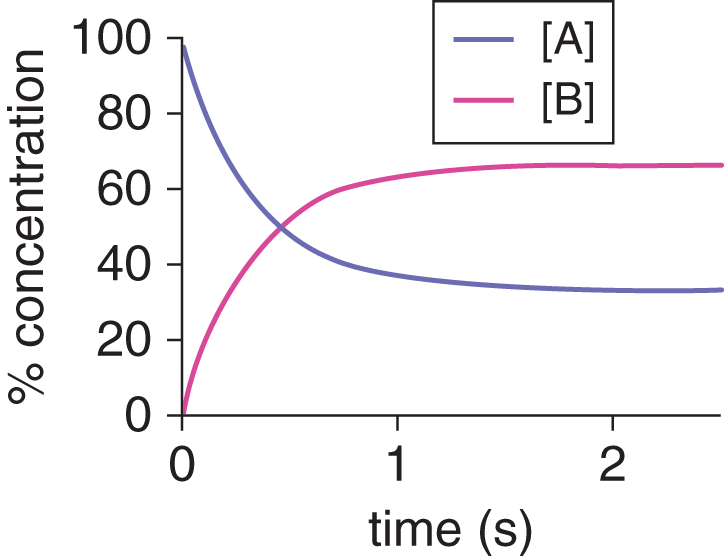
Figure 6.1. Dynamic Equilibrium Occurs when Forward and Reverse Rates Are Equal
BRIDGE
Many biochemical reactions can be classified as reversible, and their activation energies are lowered by enzymes. Irreversible biochemical steps are sometimes termed “committed” because they cannot be reversed in their pathways. These steps also tend to be the rate-limiting steps of metabolic pathways, which are discussed in Chapters 9 through 11 of MCAT Biochemistry Review.
Consider the generic reversible reaction illustrated in Figure 6.1:
A ⇌ B
At equilibrium, the concentrations of A and B are constant (although not necessarily equal), and the reactions A → B and B → A continue to occur at equal rates.
Equilibrium can be thought of as a balance between the forward and reverse reactions. Better still, equilibrium should be understood on the basis of entropy, which is the measure of the distribution of energy throughout a system or between a system and its environment. For a reversible reaction at a given temperature, the reaction will reach equilibrium when the system’s entropy—or energy distribution—is at a maximum and the Gibbs free energy of the system is at a minimum.
BRIDGE
In Chapter 7 of MCAT General Chemistry Review, we will explore the more “classic” MCAT definition of entropy—a measure of the disorder of a system. It is important to realize, though, that the units of entropy ( J K · mol ) imply a distribution of energy in a system.
Law of Mass Action
For a generic reversible reaction aA + bB ⇌ cC + dD, the law of mass action states that, if the system is at equilibrium at a constant temperature, then the following ratio is constant:
K eq = [ C ] c [ D ] d [ A ] a [ B ] b
Equation 6.1
The law of mass action is actually related to the expressions for the rates of the forward and reverse reactions. Consider the following one-step reversible reaction:
2 A ⇌ B + C
Because the reaction occurs in one step, the rates of the forward and reverse reactions are given by:
ratef = kf[A]2 and rater = kr[B][C]
When ratef = rater, the system is in equilibrium. Because the rates are equal, we can set the rate expressions for the forward and reverse reactions equal to each other:
k f [ A ] 2 = k r [ B ] [ C ] → k f k r = [ B ] [ C ] [ A ] 2
KEY CONCEPT
At equilibrium, the rate of the forward reaction equals the rate of the reverse reaction, entropy is at a maximum, and Gibbs free energy is at a minimum. This links the concepts of thermodynamics and kinetics.
Because kf and kr are both constants, we can define a new constant Kc, where Kc is called the equilibrium constant and the subscript c indicates that it is in terms of concentration. When dealing with gases, the equilibrium constant is referred to as Kp, and the subscript p indicates that it is in terms of pressure. For dilute solutions, Kc and Keq are used interchangeably. The new equation can thus be written:
K c = K eq = [ B ] [ C ] [ A ] 2
KEY CONCEPT
For most purposes, you will not need to distinguish between different K values. For dilute solutions, Keq = Kc and both are calculated in units of concentration.
While the forward and the reverse reaction rates are equal at equilibrium, the concentrations of the reactants and products are not usually equal. This means that the forward and reverse reaction rate constants, kf and kr, respectively, are not usually equal to each other. The ratio of kf to kr is Kc:
K c = K eq = k f k r
KEY CONCEPT
Remember the warning in Chapter 5 of MCAT General Chemistry Review about confusing equilibrium expressions and rate laws? In equilibrium expressions, the exponents are equal to the coefficients in the balanced equation. In rate laws, the exponents must be determined experimentally and often do not equal the stoichiometric coefficients.
When a reaction occurs in more than one step, the equilibrium constant for the overall reaction is found by multiplying together the equilibrium constants for each step of the reaction. When this is done, the equilibrium constant for the overall reaction is equal to the concentrations of the products divided by the concentrations of the reactants in the overall reaction, with each concentration term raised to the stoichiometric coefficient for the respective species. The forward and reverse rate constants for the nth step are designated kn and k–n, respectively. For example, if the reaction aA + bB ⇌ cC + dD occurs in three steps, each with a forward and reverse rate, then:
K c = k 1 k 2 k 3 k − 1 k − 2 k − 3 = [ C ] c [ D ] d [ A ] a [ B ] b
Example: What is the expression for the equilibrium constant for the following reaction?
3 H2 (g) + N2 (g) ⇌ 2 NH3 (g)
Solution:
K c = K eq = [ NH 3 ] 2 [ H 2 ] 3 [ N 2 ] . The Kp of this reaction would be: ( P NH 3 ) 2 ( P H 2 ) 3 × P N 2
Reaction Quotient
The law of mass action defines the position of equilibrium; however, equilibrium is a state that is only achieved through time. Depending on the actual rates of the forward and reverse reactions, equilibrium might be achieved in microseconds or millennia. What can serve as a “timer” to indicate how far the reaction has proceeded toward equilibrium? This role is served by the reaction quotient, Q. At any point in time during a reaction, we can measure the concentrations of all of the reactants and products and calculate the reaction quotient according to the following equation:
Q c = [ C ] c [ D ] d [ A ] a [ B ] b
Equation 6.2
This equation looks identical to the equation for Keq. It is the same form, but the information it provides is quite different. While the concentrations used for the law of mass action are equilibrium (constant) concentrations, the concentrations of the reactants and products are not constant when calculating a value for Q of a reaction. Thus, the utility of Q is not the value itself but rather the comparison that can be made between Q at any given moment in the reaction to the known Keq for the reaction at a particular temperature. Le Châtelier’s principle, which will be elaborated upon shortly, will then guide the reaction. For any reaction, if:
- Q < Keq, then the forward reaction has not yet reached equilibrium.
- There is a greater concentration of reactants (and smaller concentration of products) than at equilibrium.
- The forward rate of reaction is increased to restore equilibrium.
- Q = Keq, then the reaction is in dynamic equilibrium.
- The reactants and products are present in equilibrium proportions.
- The forward and reverse rates of reaction are equal.
- Q > Keq, then the forward reaction has exceeded equilibrium.
- There is a greater concentration of products (and smaller concentration of reactants) than at equilibrium.
- The reverse rate of reaction is increased to restore equilibrium.
KEY CONCEPT
- Q < Keq: ΔG < 0, reaction proceeds in forward direction
- Q = Keq: ΔG = 0, reaction is in dynamic equilibrium
- Q > Keq, ΔG > 0: reaction proceeds in reverse direction
Any reaction that has not yet reached the equilibrium state, as indicated by Q < Keq, will continue spontaneously in the forward direction (consuming reactants to form products) until the equilibrium ratio of reactants and products is reached. Any reaction in the equilibrium state will continue to react in the forward and reverse directions, but the reaction rates for the forward and reverse reactions will be equal, and the concentrations of the reactants and products will be constant, such that Q = Keq. A reaction that is beyond the equilibrium state, as indicated by Q > Keq, will proceed in the reverse direction (consuming products to form reactants) until the equilibrium ratio of reactants and products is reached again. Once a reaction is at equilibrium, any further movement in either the forward direction (resulting in an increase in products) or in the reverse direction (resulting in the reformation of reactants) will be nonspontaneous. This trend is illustrated in Figure 6.2.
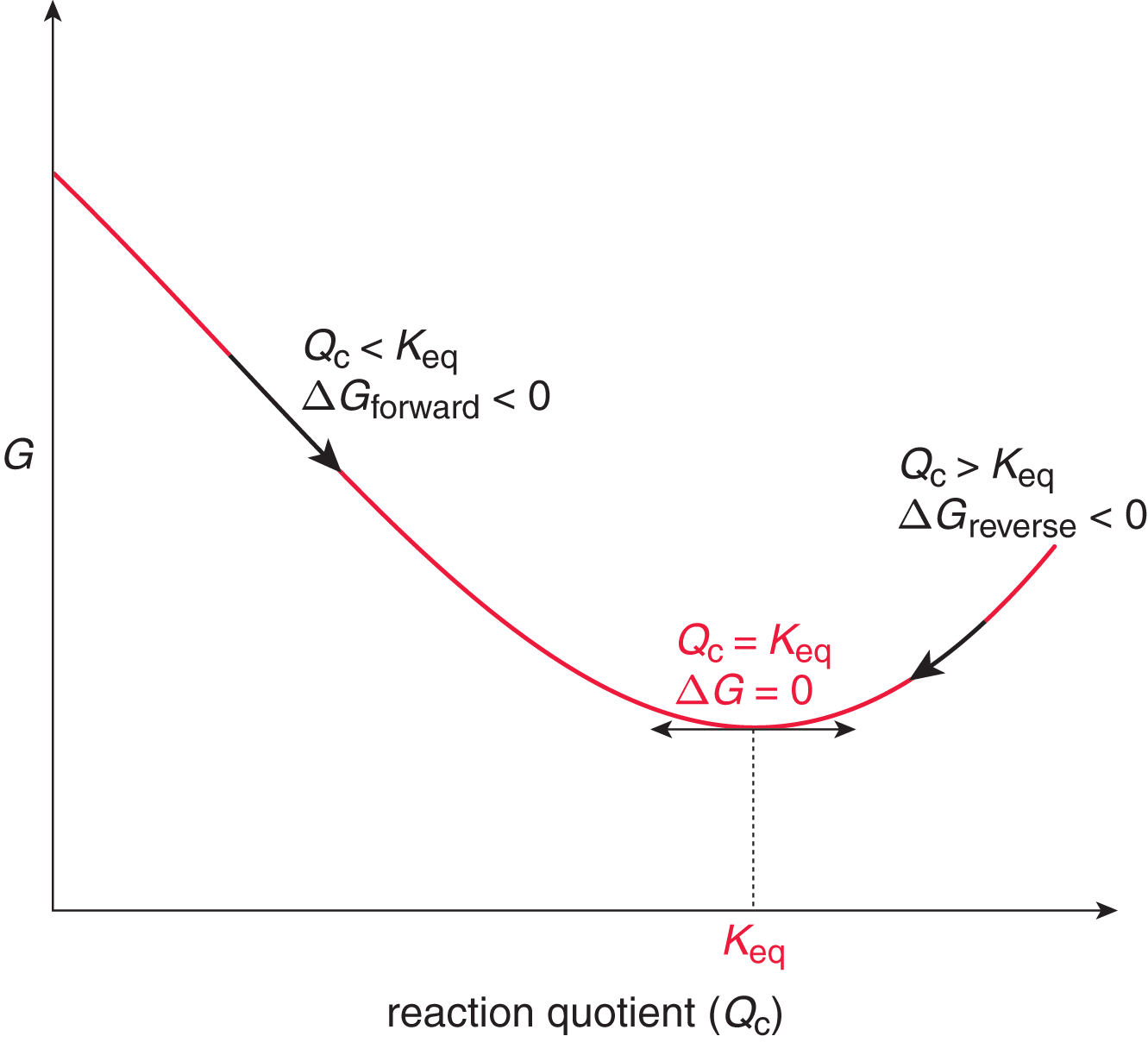
Figure 6.2. Gibbs Free Energy vs. Reaction Quotient
In Chapter 7 of MCAT General Chemistry Review, we’ll further discuss how the spontaneity of these systems is related to enthalpy and entropy.
Properties of the Law of Mass Action
Make sure to remember the following characteristics of the law of mass action and equilibrium constant expressions:
- The concentrations of pure solids and pure liquids do not appear in the equilibrium constant expression. This is because the equilibrium expression is technically based on the activities of compounds, not concentrations; the activities of pure solids and liquids are defined to be 1. For the purposes of the MCAT, there is a negligible difference between concentration and activity.
- Keq is characteristic of a particular reaction at a given temperature; the equilibrium constant is temperature-dependent.
- The larger the value of Keq, the farther to the right the equilibrium position.
- If the equilibrium constant for a reaction written in one direction is Keq, the equilibrium constant for the reverse reaction is 1 K eq .
Equilibrium calculations
Calculations involving the equilibrium constant can take several forms but are also highly repetitive. Although we have discussed Keq in this chapter, equilibrium constants and related calculations appear in solutions (Ksp), acids and bases (Kw, Ka, and Kb), and enzyme kinetics (Kd, Kb, and Ka). Thus, learning the “ins and outs” of equilibrium calculations has a large payoff on the MCAT.
One of the first concepts to grasp is the scale of the equilibrium constant. In an ideal situation, the concentrations of products and reactants would all be the same, and regardless of their actual concentrations would reduce to 1:1 ratios. In this case, Keq would equal 1. In the real world, this situation doesn’t exist, as it is unlikely that any reaction would have exactly equal concentrations of products and reactants at equilibrium. However, a Keq of 1 can be a valuable reference point, given that K eq = products reactants .
If the concentration of products is greater than the concentration of reactants, Keq becomes a “top heavy” fraction and must be greater than 1. On the other hand, if the concentration of reactants is greater than that of products, Keq becomes a “bottom heavy” fraction and must be less than 1. Keep in mind, however, that Keq is often expressed as a single value using exponents, and the sign and scale of these exponents gives even more information about the relative quantities of reactants and products. A reaction that strongly favors products will have a large, positive exponent, and the larger the exponent, the less reactant that will be present at equilibrium. In other words, a large positive exponent indicates a reaction that goes almost to completion.
On the other hand, a large negative exponent indicates a reaction that strongly favors reactants at equilibrium. In this case, only a small amount of reactant is converted to product. When performing equilibrium calculations, a Keq with a large negative exponent allows a very convenient and very necessary shortcut to be used: the amount that has reacted can be considered negligible compared to the amount of reactant that remains.
Consider the reaction A ↔ B + C, with Keq = 10-12 and a starting concentration of [A] = 1 M. The Keq expression can be written as:
K eq = C B A
MCAT EXPERTISE
You may have been previously taught to solve these types of equilibrium problems using a technique referred to as an ICEbox. On Test Day, generating an entire ICEbox table takes valuable time and effort, and this technique can be shortcut using logic alone. Practice solving equilibrium problems without the ICEbox technique, using the methods described in this chapter, for a faster solution on Test Day.
If x amount of A has reacted, x amount of C and x amount of B have been produced at equilibrium, and the equilibrium concentration of A will be [1-x] M. Substituting these values into the Keq expression gives:
K eq = 10 - 12 = x x 1 - x = x 2 1 - x
Unfortunately, performing the calculations required by this equation would give us a polynomial function that would be extremely burdensome to solve. However, the value of Keq has a large negative exponent, allowing us to use the “x is negligible” shortcut. Relative to the 1 M starting concentration, the amount that has reacted is so small, based on Keq = 10-12, that we can assume x is negligible and round the denominator to the starting concentration:
10 - 12 = x 2 1
The problem is much more readily solved, and the value for x is found to be 10-6. This confirms our estimate that x is negligible compared to 1, since x = 0.000001, and [1- (0.00001)] ≈ 1.
If the value for Keq is within one to two orders of magnitude of one, or if the concentration of reactant that goes to product is within two orders of magnitude of the initial concentration of reactant, this estimation will not be valid. Likewise, if the value of Keq is significantly larger than one, this estimation cannot be used. In both cases, the amount that reacts will be significant compared to the starting concentration of reactant. However, these situations are unlikely to be tested on the MCAT.
BRIDGE
Equilibrium constants are calculated for many types of reactions, and go by many different names. For solubility problems (MCAT General Chemistry Review Chapter 9), Keq is known as Ksp. For acids and bases (MCAT General Chemistry Review Chapter 10), Keq is known as Ka, Kb, orKw. For enzyme kinetics (MCAT Biochemistry Review Chapter 2), Keq is known as Kd, Kb, or Ka.
Example: 3 moles of N2O4 is placed in a 0.5 L container and allowed to reach equilibrium according to the following reaction:
N 2 O 4 ( g ) ↔ 2 NO 2 ( g )
What is the equilibrium concentration of NO2, given Keq for the reaction is 6 × 10-6?
Solution: Start by writing the expression for Keq: Keq=[NO2]2[N2O4]
Note that the concentration of NO2 is squared due to its coefficient of 2 in the balanced reaction. Next, determine the starting concentration of N2O4, taking into account that the initial volume is 500 mL = 0.5 L:3M0.5L=[N2O4]1L
[N2O4]=(3)(1)(0.5)=6M
Thus, the starting concentration of N2O4 = 6 M. Next, using x to represent the amount of N2O4 that reacts, 2x to represent the amount of NO2 that is produced, and 10-6 for the value of Keq, plug into the expression for the equilibrium constant:
Keq=6×10−6=[2x]2[6−x]≈[2x]2[6]
Note that the small negative exponent in the value of Keq indicates that x will be negligible in comparison to 6 M, allowing the Keq expression to be simplified, and x to be determined as follows:
6 × 10 − 6 = 4 x 2 6 36 × 10 − 6 = 4 x 2 x 2 = 36 × 10 − 6 4 = 9 × 10 − 6 x = 3 × 10 − 3 M
However, be careful to note that this is the value of x, which represents the amount of N2O4 that reacts. The final answer must represent the amount of NO2 produced, which is twice the amount of N2O4 that reacts, or 2x. Thus the final answer is the concentration of NO2, which is 6 × 10–3 M.
MCAT CONCEPT CHECK 6.1:
Before you move on, assess your understanding of the material with these questions.
-
Given that [product] = 0.075 M and [reactant] = 1.5 M, determine the direction of reaction and the sign of the free energy change for reactions with the following Keq values: (Note: Assume that the reaction has only one product and one reactant, and that the stoichiometric coefficient for each is 1.)
***Keq Direction of Reaction ΔG* 5.0 × 10−2 5.0 × 10−3 5.0 × 10−1**
-
Write the equilibrium constant expression for the following reactions:
- CO (g) + 2 H2 (g) ⇌ CH3OH (g):
________________________________
- H3PO4 (aq) + H2O (l) ⇌ H2PO4− (aq) + H3O+ (aq):________________________________
-
Consider the hypothetical reaction A ↔ B + C .
For each of the following, determine if the amount of reactant A that has converted to product at equilibrium will be negligible compared to the starting concentration of A.
***Keq Initial Concentration of A (M*) Is the amount reacted negligible? 1.0 × 10−121 1.0 × 10−2 0.1
1.0 × 10−3 0.001
1.0 × 10−15 0.001
-
The following reaction has a Keq of 2.1 × 10–7. Given an initial concentration for A equal to 0.1 M and an initial concentration of B equal to 0.2 M, what is the equilibrium concentration of C? Is the approximation that x is negligible valid for this calculation?
A a q + B ( a q ) ↔ C g + D s
6.2 Le Châtelier’s Principle
LEARNING OBJECTIVES
After Chapter 6.2, you will be able to:
- Use Le Châtelier’s principle to determine how changing conditions, including pH, temperature, pressure, and concentration changes, will affect a reaction previously in equilibrium
Le Châtelier’s principle states that if a stress is applied to a system, the system shifts to relieve that applied stress. Regardless of the form the stress takes, the reaction is temporarily moved out of its equilibrium state. This is either because the concentrations or partial pressures of the system are no longer in the equilibrium ratio or because the equilibrium ratio itself has changed as a result of a change in the temperature of the system. The reaction then responds by reacting in whichever direction—either forward or reverse—will result in a reestablishment of the equilibrium state.
MCAT EXPERTISE
Le Châtelier’s principle applies to a wide variety of systems and, therefore, appears as a fundamental concept in all three MCAT science sections.
Changes in Concentration
When reactants or products are added or removed from a reaction in equilibrium, the reaction is moved from its minimum energy state. With the change in concentration of one or more of the chemical species, the system now has a ratio of products to reactants that is not equal to the equilibrium ratio. In other words, changing the concentration of either a reactant or a product results in Qc ≠ Keq. If reactants are added (or products are removed), Qc < Keq, and the reaction will spontaneously react in the forward direction, increasing the value of Qc until Qc = Keq. If reactants are removed (or products are added), Qc > Keq, and the reaction will spontaneously react in the reverse direction, thereby decreasing the value of Qc until once again Qc = Keq. Put simply, the system will always react in the direction away from the added species or toward the removed species.
BRIDGE
The bicarbonate buffer system is a classic example of Le Châtelier’s principle applied to physiology:
CO2 (g) + H2O (l) ⇌ H2CO3 (aq) ⇌ H+ (aq) + HCO3− (aq)
In the tissues, there is a relatively high concentration of CO2, and the reaction shifts to the right. In the lungs, CO2 is lost, and the reaction shifts to the left. Note that blowing off CO2 (hyperventilation) is used as a mechanism for dealing with acidemia (excess H+ in the blood). This buffer system plays a key role in the respiratory, circulatory, and excretory systems, discussed in Chapters 6, 7, and 10 of MCAT Biology Review, respectively.
We often take advantage of Le Châtelier’s principle to improve the yield of chemical reactions. For example, in the industrial production of chemicals, products of reversible reactions are removed as they are formed to prevent the reactions from reaching their equilibrium states. The reaction will continue to react in the forward direction, producing more and more products—assuming reactants are continually replaced as they are consumed. One could also drive a reaction forward by starting with high concentrations of reactants. This will lead to an increase in the absolute quantities of products formed, although the reaction will still eventually reach its equilibrium state unless products are removed as they are formed.
Changes in Pressure (and Volume)
Because liquids and solids are essentially incompressible, only chemical reactions that involve at least one gaseous species will be affected by changes in the system’s pressure and volume. When a system is compressed, its volume decreases and its total pressure increases. This increase in the total pressure is associated with an increase in the partial pressures of each gas in the system, and this results in the system no longer being in the equilibrium state, such that Qp does not equal Keq. The system will move forward or in reverse, always toward whichever side has the lower total number of moles of gas. This is a consequence of the ideal gas law, which tells us that there is a direct relationship between the number of moles of gas and the pressure of the gas. If one increases the pressure of a system, it will respond by decreasing the total number of gas moles, thereby decreasing the pressure. Note that this scenario assumes that the volume of the system was decreased and then held constant while the system returned to its equilibrium state. When one expands the volume of a system, the total pressure and the partial pressures decrease. The system is no longer in its equilibrium state and will react in the direction of the side with the greater number of moles of gas in order to restore the pressure.
Consider the following reaction:
N2 (g) + 3 H2 (g) ⇌ 2 NH3 (g)
The left side of the reaction has a total of four moles of gas molecules, while the right side has only two moles. When the pressure of this system is increased, the system will react in the direction that produces fewer moles of gas. In this case, that direction is to the right, and more ammonia will form. However, if the pressure is decreased, the system will react in the direction that produces more moles of gas; thus, the reverse reaction will be favored, and more nitrogen and hydrogen gas will reform.
Changes in Temperature
Le Châtelier’s principle tells us that changing the temperature of a system will also cause the system to react in a particular way to return to its equilibrium state. However, unlike the effect of changing concentrations or pressures, the result of changing temperature is not a change in the reaction quotient, Qc or Qp, but a change in Keq. The change in temperature does not cause the concentrations or partial pressures of the reactants and products to change immediately, so Q immediately after the temperature change is the same as before the temperature change. Thus, because Keq is now a different value, Q no longer equals Keq. The system has to move in whichever direction allows it to reach its new equilibrium state at the new temperature. That direction is determined by the enthalpy of the reaction. If a reaction is endothermic (ΔH > 0), heat functions as a reactant; if a reaction is exothermic (ΔH < 0), heat functions as a product. Thinking about heat as a reactant or product allows us to apply the same principles we used with concentration changes to temperature changes.
For example, consider the following endothermic reaction, shown in Figure 6.3: N 2 O 4 ( g ) ⇌ Δ 2 NO 2 ( g )
The equilibrium position can be shifted by changing the temperature. When heat is added and the temperature increases, the reaction shifts to the right, and the flask turns reddish-brown due to an increase in [NO2]. When heat is removed and the temperature decreases, the reaction shifts to the left, and the flask turns more transparent due to an increase in N2O4. This demonstrates Le Châtelier’s principle because the equilibrium shifts in the direction that consumes energy.

Figure 6.3. Example of a Reversible Endothermic Reaction, N2O4 (g) ⇌ 2 NO2 (g) Left: As temperature decreases, the equilibrium favors N2O4 production, turning the reaction vessel more transparent. Right: As temperature increases, the equilibrium favors NO2 production, turning the reaction vessel reddish-brown.
KEY CONCEPT
The reaction
A (aq) + 2 B (g) ⇌C (g) + heat Will shift to the right if… Will shift to the left if…
A or B is added C is added
C is removed A or B is removed
the pressure is increased or the volume is reduced the pressure is reduced or the volume is increased
the temperature is reduced the temperature is increased
MCAT CONCEPT CHECK 6.2:
Before you move on, assess your understanding of the material with this question.
-
Describe what would happen in the following situations:
- In the reaction H2SO4 (aq) ⇌ H+ (aq) + HSO4− (aq), the pH has been increased: ________________________________
- In the reaction 2 C (s) + O2 (g) ⇌ 2 CO (g), the pressure of the reaction vessel is decreased: ________________________________
- In the reaction CH4 (g) + 2 O2 (g) ⇌ CO2 (g) + 2 H2O (l) + heat, the reaction vessel is warmed:
________________________________
- In the reaction H3PO4 (aq) + H2O (l) ⇌ H3O+ (aq) + H2PO4− (aq), water is removed (without changing temperature):
__________________________________
6.3 Kinetic and Thermodynamic Control
LEARNING OBJECTIVES
After Chapter 6.3, you will be able to:
- Identify the conditions that will cause a reaction to favor the kinetic or the thermodynamic product
- Distinguish between a kinetic and thermodynamic pathway on a reaction coordinate diagram
Having covered the fundamentals of kinetics and thermodynamics, we come upon a topic that bridges all chemical systems: control of a reaction. In particular, biochemical reactions often require regulation in a precise manner to be useful to an organism. The applications of kinetic and thermodynamic control are common on the MCAT and range from metabolic reactions requiring high-energy phosphate molecules, such as ATP to the effects of temperature and solvents on enzyme activity.
The examples below consider unimolecular systems through the lens of the transition state theory. Figure 6.4 shows starting materials (reactants) at a certain energy level. These reactants can undergo two different sets of reactions. At lower temperatures (with smaller heat transfer), a kinetic product is formed. At higher temperatures (with larger heat transfer), a thermodynamic product is formed.
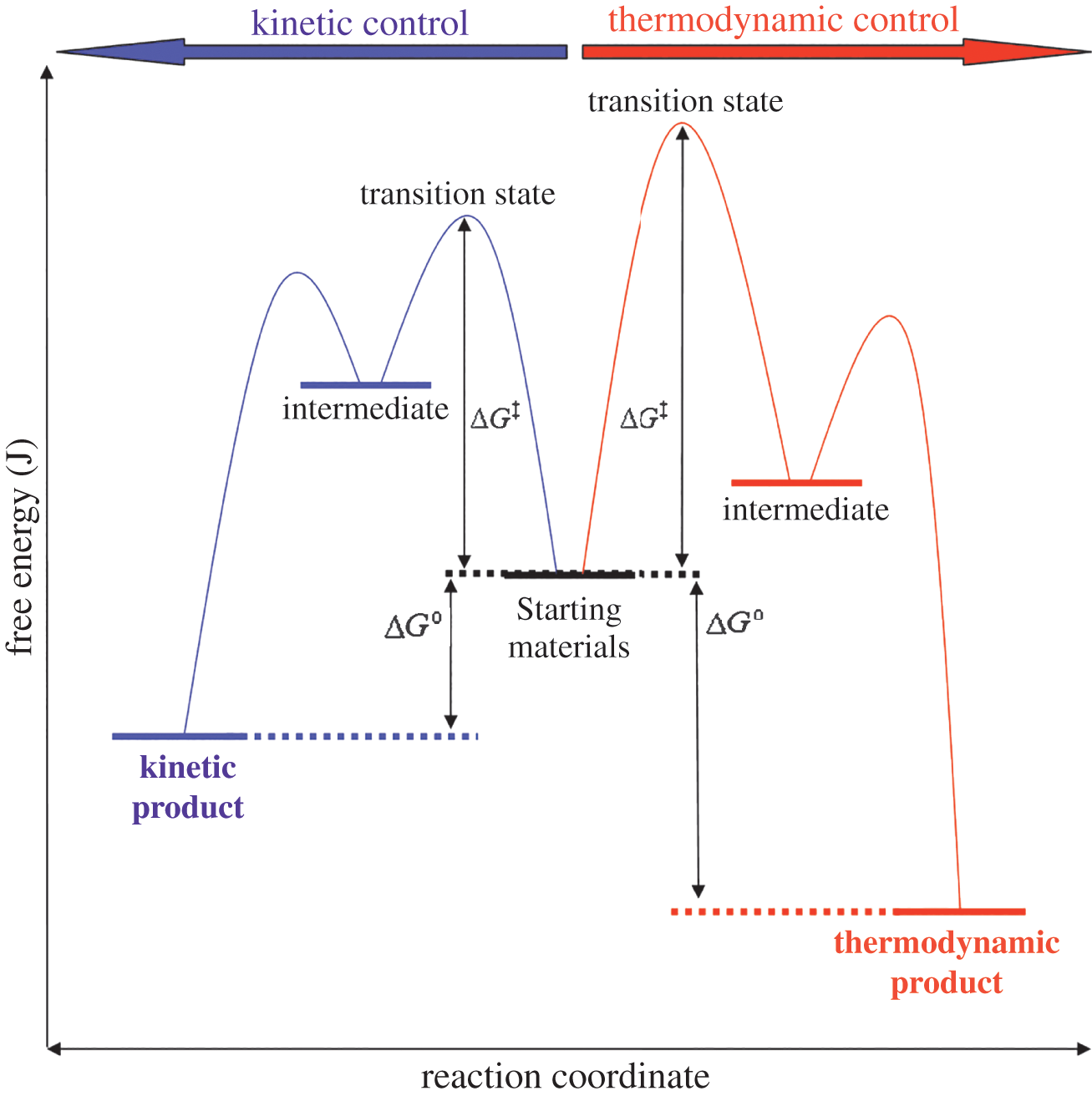
Figure 6.4. Kinetic and Thermodynamic Control of a Reaction The kinetic pathway requires less free energy to reach the transition state, but results in a higher-energy (less stable) product.
Note that the free energy that must be added for the kinetic pathway is lower than that of the thermodynamic pathway. Therefore, the kinetic products often form faster than the thermodynamic products and are sometimes called “fast” products. On the other hand, the free energy of the thermodynamic product is significantly lower than that of the kinetic product. Thermodynamic products are therefore associated with greater stability, and with a more negative ΔG than kinetic products.
The stability of organic molecules is covered in Chapter 2 of MCAT Organic Chemistry Review and is dependent on torsional strain, angle strain, and nonbonded strain. In this example, we consider the conversion of 2-methylcyclohexanone to its thermodynamic product and its kinetic product, as shown in Figure 6.5. Both reactions require a base (B–) in order to catalyze the conversion, yet two different products are produced.
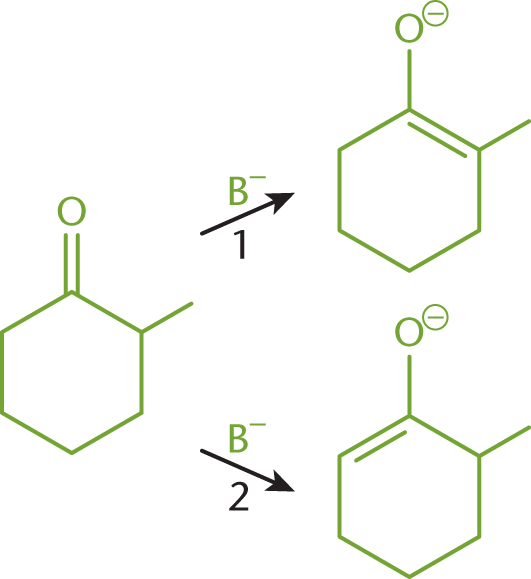
Figure 6.5. Conversion of 2-methylcyclohexanone to (1) Thermodynamic Product and (2) Kinetic Product
For the thermodynamic pathway (1), the double bond is located between C-1 and the methyl group. It requires more energy to form the transition state of this reaction because the base must overcome the steric hindrance created by the methyl group. The base squeezes in to reach the carbon with the methyl group attached to abstract a proton. However, because the double bond is more substituted than the other pathway, the product of this reaction is more stable and less likely to react further.
For the kinetic pathway (2), the double bond is located between C-1 and C-6. This pathway is preferred when there is little heat available because less energy is needed to reach the transition state. The base can more easily reach C-6 to remove a proton, and the resulting enolate can form. This product has a less substituted double bond, which reduces its stability. This lack of stability may leave the ring susceptible to further attack.
MCAT CONCEPT CHECK 6.3:
Before you move on, assess your understanding of the material with these questions.
-
What conditions favor formation of a kinetic product? A thermodynamic product?
- Kinetic product: ________________________________
- Thermodynamic product: ___________________________
-
On a reaction coordinate diagram, how would the kinetic pathway appear as compared to the thermodynamic pathway?
__________________________________
Conclusion
We’ve discussed some very important concepts and principles in the past two chapters related to the studies of reaction rates and chemical equilibria. In this chapter, we began with the law of mass action and the significance of the equilibrium state of a chemical reaction. With our understanding of the significance of Keq and Q, we are able to predict the direction that a reaction will go in response to various stresses—concentration, pressure, or temperature changes—that might be applied to a system.
The concept of homeostasis in biology is a direct result of the energy associated with disturbing equilibria in the body. Reactions are often held slightly out of the equilibrium state to generate energy. Many pathologies you will encounter in your future career in medicine will have a fundamental basis in disturbed chemical equilibria—just wait until you start ordering metabolic panels on your patients!
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Equilibrium
- Reversible reactions eventually reach a state in which energy is minimized and entropy is maximized.
- Chemical equilibria are dynamic—the reactions are still occurring, just at a constant rate.
- The concentrations of reactants and products remain constant because the rate of the forward reaction equals the rate of the reverse reaction.
- The law of mass action gives the expression for the equilibrium constant, Keq. The reaction quotient, Q, has the same form but can be calculated at any concentrations of reactants and products.
- Q is a calculated value that relates the reactant and product concentrations at any given time during a reaction.
- Keq is the ratio of products to reactants at equilibrium, with each species raised to its stoichiometric coefficient. Keq for a reaction is constant at a constant temperature.
- Pure solids and liquids do not appear in the law of mass action; only gases and aqueous species do.
- Comparison of Q to Keq provides information about where the reaction is with respect to its equilibrium state.
- If Q < Keq, ΔG < 0, and the reaction proceeds in the forward direction.
- If Q = Keq, ΔG = 0, and the reaction is in dynamic equilibrium.
- If Q > Keq, ΔG > 0, and the reaction proceeds in the reverse direction.
- Equilibrium calculations are broadly applicable to many areas of chemistry but are often formulaic in their application. The magnitude of Keq determines the balance of a reaction and whether the amount that has reacted can be treated as negligible when compared to other concentrations.
- If Keq > 1, the products are present in greater concentration at equilibrium.
- If Keq ≈ 1, products and reactants are both present at equilibrium at reasonably similar levels.
- If Keq < 1, the reactants are present in greater concentration at equilibrium.
- If Keq <<< 1, the amount of reactants that have been converted to products can be considered negligible in comparison to the initial concentration of reactants.
Le Châtelier’s Principle
- Le Châtelier’s principle states that when a chemical system experiences a stress, it will react so as to restore equilibrium.
- There are three main types of stresses applied to a system: changes in concentration, pressure and volume, and temperature.
- Increasing the concentration of reactants or decreasing the concentration of products will shift the reaction to the right. Increasing the concentration of products or decreasing the concentration of reactants will shift the reaction to the left.
- Increasing pressure on a gaseous system (decreasing its volume) will shift the reaction toward the side with fewer moles of gas. Decreasing pressure on a gaseous system (increasing its volume) will shift the reaction toward the side with more moles of gas.
- Increasing the temperature of an endothermic reaction or decreasing the temperature of an exothermic reaction will shift the reaction to the right. Decreasing the temperature of an endothermic reaction or increasing the temperature of an exothermic reaction will shift the reaction to the left.
Kinetic and Thermodynamic Control
- Reactions may have both kinetic and thermodynamic products that can be regulated by temperature and the presence of a catalyst.
- Kinetic products are higher in free energy than thermodynamic products and can form at lower temperatures. These are sometimes termed “fast” products because they can form more quickly under such conditions.
- Thermodynamic products are lower in free energy than kinetic products and are therefore more stable. Despite proceeding more slowly than the kinetic pathway, the thermodynamic pathway is more spontaneous (more negative ΔG).
ANSWERS TO CONCEPT CHECKS
**6.1**
- First calculate the value of Q from the given concentrations:
Q = products reactants = 0 . 075 1 . 5 = 7 . 5 × 10 - 2 1 . 5 = 5 × 10 - 2 .
Q can now be compared to each value of Keq to predict the direction of the reaction.
***Keq Direction of Reaction ΔG***
5.0 × 10−2 At equilibrium: no net reaction 0
5.0 × 10−3 Qc > Keq: proceeds toward reactants (left) Positive
5.0 × 10−1 Qc < Keq: proceeds toward products (right) Negative
- K p = P CH 3 OH P CO × ( P H 2 ) 2
K c = K a = [ H 2 PO 4 − ] [ H 3 O + ] [ H 3 PO 4 ]
- The concentration of a reactant that converts to product can be considered negligible if it is two or more orders of magnitude less than the initial concentration of the reactant.
***Keq Initial Concentration of A (M*) Is the amount reacted negligible?**
1 x 10-12 1 Yes
1 x 10-2
0.1 No
1 x 10-3
0.001 No
1 x 10-15 0.001 Yes
- The first step in solving is to write the equation for Keq for the reaction:
A(
aq
)+B(
aq
)↔C( g )+D( s ).
K
eq
=
[ C ]
[ A ][ B ]
=2.1×
10
−7
Note that the equation for Keq does not include product D because D is a solid.
Next, using the initial concentrations for A and B and x for the amount that has reacted, plug into the equation for Keq:
K eq = C A B = x 0 . 1 - x 0 . 2 - x
Given that Keq = 2.1 x 10–7, the concentrations of A and B are sufficiently large that x can be considered negligible in comparison to both. This allows the equation for Keq to be simplified and solved:
K eq = 2 . 1 × 10 - 7 ≈ x 0 . 1 0 . 2 x = 2 . 1 × 10 - 7 0 . 1 0 . 2 = 4 . 2 × 10 - 9
The value of x = 4.2 x 10–9 is equal to both the equilibrium concentration of C and the amount of A and B that have reacted. The approximation that x is negligible compared to the initial concentrations of A and B is valid.
**6.2**
-
- Increasing pH of H2SO4 (aq) ⇌ H+ (aq) + HSO4− (aq): [H+] decreases, shifting reaction to the right.
- Decreasing pressure of 2 C (s) + O2 (g) ⇌ 2 CO (g): Reaction shifts right, favoring the side with more moles of gas.
- Warming CH4 (g) + 2O2 (g) ⇌ CO2 (g) + 2 H2O (l) + heat: Reaction shifts left, using the additional heat energy to produce more reactants.
- Removing water from H3PO4 (aq) + H2O (l) ⇌ H3O+ (aq) + H2PO4− (aq): Reaction shifts left. All concentrations would increase proportionately; because there are more products than reactants (and the stoichiometric coefficient is 1 for each reactant and product), the value of Q will increase.
**6.3**
- Kinetic products are favored at low temperatures with low heat transfer. Thermodynamic products are favored at high temperatures with high heat transfer.
- Kinetic pathways require a smaller gain in free energy to reach the transition state. They also have a higher free energy of the products, with a smaller difference in free energy between the transition state and the products.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. C
This scenario likely describes a situation in which a reaction has reached equilibrium very far to the right (with high product concentration and low reactant concentration). This reaction must be reversible because the reaction did not proceed all the way to the right. Any reaction in equilibrium has equal forward and reverse rates of reaction.
2. D
Recall that pure solids and liquids do not appear in the equilibrium expression; thus, this Keq has no denominator because the only reactant is a solid, cuprous sulfate. This could also be called Ksp because a solid is dissociating into ions in solution. The correct Keq should have [Cu+] squared because its stoichiometric coefficient is 2.
3. A
Carbon dioxide gas evolves and leaves the bottle, which decreases the total pressure of the reactants. Le Châtelier’s principle explains that a decrease in pressure shifts the equilibrium to increase the number of moles of gas present. This particular reaction will shift to the left, which in turn will decrease the amount of carbonic acid and increase the amount of carbon dioxide and water. Oxygen and nitrogen are not highly reactive and are unlikely to combine spontaneously with carbon dioxide or carbonic acid, as in (C) and (D).
4. B
Recall that when multiple products are possible and an equilibrium exists between these multiple products, one product will be called the kinetic product and the other the thermodynamic product. The transition state leading to the kinetic product is lower in energy, so that the kinetic product is formed more rapidly in solution. However, the kinetic product is less stable than the thermodynamic product. The end result is that the equilibrium of products initially favors the kinetic product, but eventually favors the more stable thermodynamic product. Taking these factors together, to favor the kinetic product, conduct the reaction for a short duration, so that the thermodynamic product does not have much chance to form. Also, the reaction should be conducted at a lower temperature to help stabilize the otherwise less stable kinetic product. These factors—low temperature and short reaction time—justify choice (B).
5. A
The larger the value of Keq (whether Kc or Kp), the larger the ratio of products to reactants. Therefore, if Kc ≫ 1, there are significantly larger concentrations of products than reactants at equilibrium. Even with a large Keq, the reaction will ultimately reach equilibrium far toward the products side and is therefore reversible, eliminating (D).
6. B
Adding sodium acetate increases the number of acetate ions present. According to Le Châtelier’s principle, this change will push this reaction to the left, resulting in a decrease in the number of free H+ ions. Because pH is determined by the hydrogen ion concentration, a decrease in the number of free protons will increase the pH. An acid’s Ka (which is simply the Keq for acid dissociation) will remain constant under a given temperature and pressure, eliminating (C) and (D).
7. A
Both increasing the pressure of the container and decreasing the volume would favor the side with fewer moles of gas, which is the product side. This makes (B) and (C) incorrect. (D) would not disturb the equilibrium—the significance of decreasing the volume of the container in most equilibria is that there is an increase in pressure; in this case, however, the pressure remains constant despite the change in volume.
8. C
An exothermic reaction produces heat. Decreasing the temperature favors product formation, resulting in an increase in the forward reaction rate with a concomitant decrease in the reverse reaction rate.
9. B
The equilibrium of a reaction can be changed by several factors. Adding or subtracting heat, (A), would shift the equilibrium based on the enthalpy change of the reaction. Increasing reactant concentrations would shift the equilibrium in the direction of the product, and the opposite would occur if reactant concentrations were decreased, eliminating (C). Changing the volume of a reactant would affect any reaction with gaseous reactants or products, eliminating (D). While adding or removing a catalyst would change the reaction rates, it would not change where the equilibrium lies.
10. A
Start with the balanced equation for the reaction of H2 and N2 to produce NH3: N2 + 3 H2 ⇌ 2 NH3.
Next, write out Kc = Keq = [ NH 3 ] 2 [ N 2 ] [ H 2 ] 3 . Because the volume is 1 L, the amount of each gas in moles is equal to the value of the concentration of each gas in moles per liter (M). The relatively small amount of NH3 produced indicates that it will be possible to consider the amount of N2 and H2 that reacts to be negligible compared to their starting concentrations. Putting the amounts of each reactant and product into the Keq expression gives:
K eq = [ NH 3 ] 2 [ N 2 ] [ H 2 ] 3 = [ 0 .05 ] 2 [ 1 - x ] [ 3 - x ] 3 ≈ [ 0 .05 ] 2 [ 1 ] [ 3 ] 3 = 5 × 10 - 2 2 27 K eq = 25 × 10 - 4 27 ≈ 25 × 10 - 4 25 = 1 × 10 - 4 = 0.0001
11. B
At extremely high temperatures, reactants or products may decompose, which will affect the equilibrium and potentially destroy the desired products. (A) implies that reactions have limits, which is true; however, this does not make increasing temperature unfavorable. (C) is false because increasing temperature would also increase pressure, assuming constant volume.(D) is incorrect because it refers to properties of irreversible reactions, which would not be involved in an equilibrium between products and reactants.
12. D
The thermodynamic product is more stable than the kinetic product, making the thermodynamic product lower in energy. Because the energy of the product is lower, the changein free energy is greater for the thermodynamic product compared to the kinetic product. Thus, (D) is correct.
13. C
Ka is equal to the ratio of products to reactants, with each species raised to its stoichiometric coefficient. A compound with a Ka greater than 10–7 contains more H+ cations than HA– anions at equilibrium, which makes it an acid. This means that the compound in question is likely to react with a compound that is basic. Of the four answer choices, NH3 is the only base.
14. C
Reaction 2 is the reverse of reaction 1. This means that Keq for reaction 2 is the inverse of Keq of reaction 1, so the answer is 1 0 . 1 = 1 1 10 = 10 .
15. A
A negative ΔH value indicates an exothermic reaction, meaning that the forward reaction produces heat. Visualize this as follows:
A + B ⇌ C + D + heat
This means that removing heat by decreasing the temperature is similar to removing any other product of the reaction. To compensate for this loss, the reaction will shift to the right, causing an increase in the concentrations of C and D, as well as a decrease in the concentrations of A and B.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(6.1) Equilibrium constant: K eq = [ C ] c [ D ] d [ A ] a [ B ] b
(6.2) Reaction quotient: Q c = [ C ] c [ D ] d [ A ] a [ B ] b
SHARED CONCEPTS
Biochemistry Chapter 2
Enzymes
General Chemistry Chapter 5
Chemical Kinetics
General Chemistry Chapter 7
Thermochemistry
General Chemistry Chapter 9
Solutions
General Chemistry Chapter 10
Acids and Bases
Organic Chemistry Chapter 2
Isomers