Chapter 8: The Gas Phase

Chapter 8: The Gas Phase
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of the following sets of conditions would be LEAST likely to result in ideal gas behavior?
- High pressure and low temperature
- Low temperature and large volume
- High pressure and large volume
- Low pressure and high temperature
-
What is the density of neon gas in g L at STP?
- 452.3
- 226.0
- 1.802
- 0.9018
-
A leak of helium gas through a small hole occurs at a rate of 3.22 × 10 − 5 mol s . How will the leakage rates of neon and oxygen gases compare to helium at the same temperature and pressure?
- Neon will leak faster than helium; oxygen will leak faster than helium.
- Neon will leak faster than helium; oxygen will leak slower than helium.
- Neon will leak slower than helium; oxygen will leak faster than helium.
- Neon will leak slower than helium; oxygen will leak slower than helium.
-
A 0.10 g piece of magnesium is placed in a beaker of hydrochloric acid. Hydrogen gas is generated according to the following equation:
Mg (s) + 2 HCl (aq) → MgCl2 (aq) + H2 (g)
The gas is collected over water at 25 °C, and the gauge pressure during the experiment reads 784 mmHg.
The gas displaces a volume of 100 mL. The vapor pressure of water at 25 °C is approximately 24.0 mmHg. Based on this data, how many moles of hydrogen are produced in this reaction? (Note: R = 0.0821 L · atm mol · K = 8.314 J K · mol . )
- 4.04 × 10−5 moles hydrogen
- 4.09 × 10−3 moles hydrogen
- 3.07 × 10−2 moles hydrogen
- 3.11 moles hydrogen
-
Ideal gases:
- have no volume.
- have particles with no attractive forces between them.
- have no mass.
- I only
- II only
- I and II only
- I, II, and III
-
Which of the following best explains the difference in ideal behavior between nitrogen and fluoromethane gas?
- Nitrogen gas is more ideal because it is smaller and forms stronger intermolecular interactions.
- Nitrogen gas is more ideal because it is smaller and forms weaker intermolecular interactions.
- Fluoromethane is more ideal because it is larger and forms stronger intermolecular interactions.
- Fluoromethane is more ideal because it is smaller and forms weaker intermolecular interactions.
-
The kinetic molecular theory states that:
- the average kinetic energy of a molecule of gas is directly proportional to the temperature of the gas in kelvin.
- collisions between gas molecules are inelastic.
- gas particles occupy discrete areas of space.
- all gas molecules have the same kinetic energy at the same temperature.
-
The plots of two gases at STP are shown below. One of the gases is 1.0 L of helium, and the other is 1.0 L of bromine. Which plot corresponds to each gas and why?
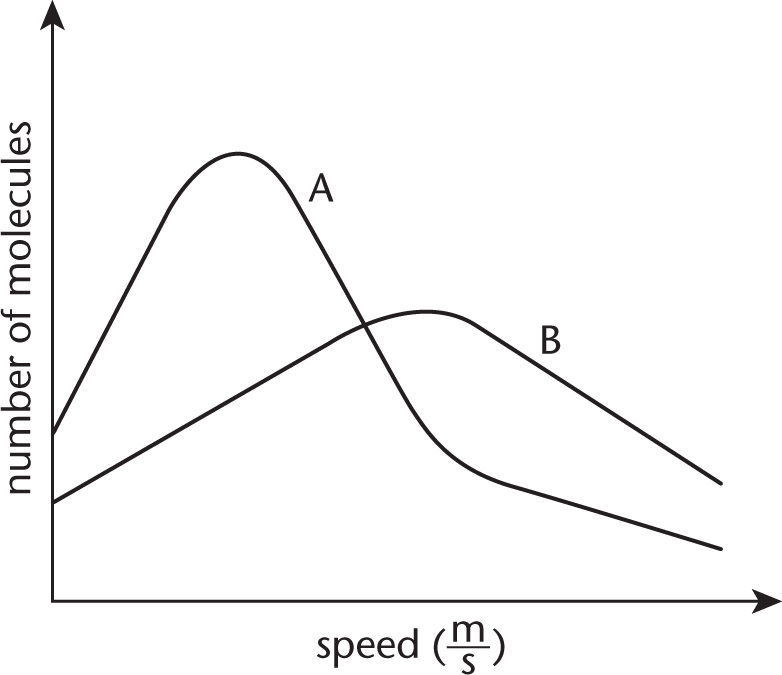
- Curve A is helium and curve B is bromine because helium has a smaller molar mass than bromine.
- Curve A is helium and curve B is bromine because the average kinetic energy of bromine is greater than the average kinetic energy of helium.
- Curve A is bromine and curve B is helium because helium has a smaller molar mass than bromine.
- Curve A is bromine and curve B is helium because the average kinetic energy of bromine is greater than the average kinetic energy of helium.
-
At sea level and 25 °C, the solubility of oxygen gas in water is 1.25 × 10−3 M. In Denver, a city in the United States that lies high above sea level, the atmospheric pressure is 0.800 atm. What is the solubility of oxygen in water in Denver?
- 1.00 × 10−3 M
- 1.05 × 10−3 M
- 1.50 × 10−3 M
- 2.56 × 10−3 M
-
Given that the gases at the center of the sun have an average molar mass of 2.00 g mol , compressed to a density of 1.20 g cm 3 under 1.30 × 109 atm of pressure, what is the temperature at the center of the sun?
- 2.6 × 104 K
- 2.6 × 106 K
- 2.6 × 107 K
- 2.6 × 1010 K
-
The gaseous state of matter is characterized by which of the following properties?
- Gases are compressible.
- Gases assume the volume of their containers.
- Gas particles exist as diatomic molecules.
- I only
- I and II only
- II and III only
- I, II, and III
-
A gas at a temperature of 27 °C has a volume of 60.0 mL. What temperature change is needed to increase this gas to a volume of 90.0 mL?
- A reduction of 150 °C
- An increase of 150 °C
- A reduction of 13.5 °C
- An increase of 13.5 °C
-
How do a significant decrease in temperature and volume affect ideal gas behavior, respectively?
- Both lead to more ideal behavior.
- A lower temperature leads to deviations from ideal behavior, while lower volume causes more ideal behavior.
- A lower temperature leads to more ideal behavior, while lower volume causes more ideal behavior.
- Both lead to deviations in ideal behavior.
-
In which of the following situations is it impossible to predict how the pressure will change for a gas sample?
- The gas is cooled at a constant volume.
- The gas is heated at a constant volume.
- The gas is heated, and the volume is simultaneously increased.
- The gas is cooled, and the volume is simultaneously increased.
-
Experimenters notice that the molar concentration of dissolved oxygen in an enclosed water tank has decreased to one-half its original value. In an attempt to counter this decrease, they quadruple the partial pressure of oxygen in the container. What is the final concentration of the gas?
- Half of the original concentration
- The same as the original concentration
- Double the original concentration
- Quadruple the original concentration
Answer Key
- A
- D
- D
- B
- B
- B
- A
- C
- A
- C
- B
- B
- D
- C
- C
Chapter 8: The Gas Phase
CHAPTER 8
THE GAS PHASE
In This Chapter
8.1 The Gas Phase
Variables
8.2 Ideal Gases
Ideal Gas Law
Special Cases
Dalton’s Law of Partial Pressures
Henry’s Law
8.3 Kinetic Molecular Theory
Assumptions
Applications
8.4 Real Gases
Deviations Due to Pressure
Deviations Due to Temperature
van der Waals Equation of State
Concept Summary
CHAPTER PROFILE
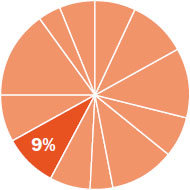
The content in this chapter should be relevant to about 9% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
3B: Structure and integrative functions of the main organ systems
4B: Importance of fluids for the circulation of blood, gas movement, and gas exchange
Introduction
Let’s start this chapter with a thought experiment. Imagine a helium balloon tied to the gearshift lever between the seats of a car and allowed to float freely. What do you think will happen to the balloon as the car accelerates forward? You might think, based on how you feel when you are in an accelerating vehicle, that the balloon will be pushed backwards due to its inertia. However, the balloon’s movement isn’t what we might predict: the balloon shifts forward as the car accelerates!
The molar mass of helium is 4 g mol , while that of air, which is mostly nitrogen and oxygen, is about 29 g mol . This means that air is about seven times denser than helium. Because the air in which the balloon is floating is more dense than the balloon itself, the air has greater inertia. Therefore, as the car accelerates forward, everything that has significant mass, including the air in the car, resists the forward motion (has inertia) and shifts toward the back of the car (even though, of course, everything in the car is accelerating forward, just not as quickly as the car itself). As the air shifts toward the back, a pressure gradient builds up such that there is greater air pressure in the back of the car than in the front, and this pressure difference results in a pushing force against the balloon that is directed from the back toward the front. Responding to this force, the balloon shifts forward in the direction of the car’s acceleration. Who would have thought that general chemistry and physics could be so much fun?
In this chapter, we will discuss some MCAT favorites—the gas phase and the ideal gas laws. We will begin our discussion with ideal gases and the laws that govern their behavior. We will then examine the kinetic molecular theory that describes ideal gases and conclude with an evaluation of the ways in which the behavior of real gases deviates from that predicted by the ideal gas law.
8.1 The Gas Phase
LEARNING OBJECTIVES
After Chapter 8.1, you will be able to:
- Identify the unique characteristics of the gas phase
- Predict how pressure will change in different positions and locations, including underwater
- Recall the conditions at STP and standard conditions
Matter can exist in three different physical forms, called phases or states: gas, liquid, and solid. We have discussed liquids in the context of intermolecular forces and solids in the context of organized crystals in Chapter 3 of MCAT General Chemistry Review. The gaseous phase may be the simplest to understand because all gases display similar behavior and follow similar laws regardless of their particular chemical identities. Like liquids, gases are classified as fluids because they can flow and take on the shapes of their containers. However, the atoms or molecules in a gaseous sample move rapidly and are far apart from each other. In addition, only very weak intermolecular forces exist between gas particles; this results in certain characteristic physical properties, such as the ability to expand to fill any volume. Gases are also easily—although not infinitely—compressible, which distinguishes them from liquids.
Variables
We can define the state of a gaseous sample by four variables: pressure (P), volume (V), temperature (T), and number of moles (n).
Gas pressures are usually expressed in units of atmospheres (atm) or in millimeters of mercury (mmHg), which are equivalent to torr. The SI unit for pressure, however, is the pascal (Pa). The mathematical relationships among all of these units are as follows:
1 atm = 760 mmHg ≡ 760 torr = 101.325 kPa
Medical devices that measure blood pressure are termed sphygmomanometers, and the most clinically relevant unit of measurement for them is mmHg. In fact many medical devices utilize the same conceptual design of a barometer, shown in Figure 8.1, to continuously monitor blood pressure.
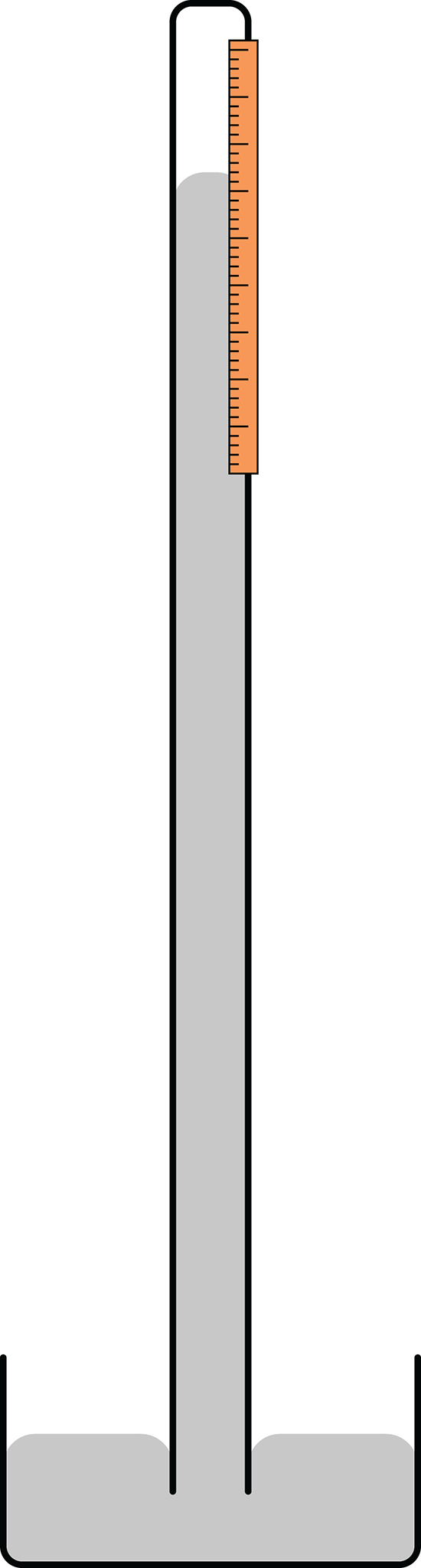
Figure 8.1. Schematic of a Simple Mercury Barometer
REAL WORLD
Blood pressure is measured by a sphygmomanometer, which uses units of mmHg. A normal adult blood pressure is considered less than 120 mmHg systolic and 80 mmHg diastolic (< 120/80). Hypertension (high blood pressure) is defined as having at least two blood pressure readings > 140 mmHg systolic or > 90 mmHg diastolic.
In order to explain why the mercury rises in a barometer, we must summarize the forces at play here. Atmospheric pressure creates a downward force on the pool of mercury at the base of the barometer while the mercury in the column exerts an opposing force (its weight) based on its density. The weight of the mercury creates a vacuum in the top of the tube. When the external air exerts a higher force than the weight of the mercury in the column, the column rises. When the external air exerts a lower force than the weight of the mercury, the column falls. Thus, a reading can be obtained by measuring the height of the mercury column (in mm), which will be directly proportional to the atmospheric pressure being applied.
BRIDGE
Fluid dynamics is an important concept discussed in Chapter 4 of MCAT Physics and Math Review that applies to multiple aspects of the gas laws covered here, including the functionality of a mercury barometer.
It is important to mention here that atmospheric pressure is not the only external pressure that can exert this force. For instance, a clinical blood pressure cuff creates a force that is opposed by the person’s systolic and diastolic arterial blood pressure.
The volume of a gas is generally expressed in liters (L) or milliliters (mL). Temperature is usually given in kelvin (K), although degrees Celsius (°C) may be used instead. Many processes involving gases take place under standard temperature and pressure (STP), which refers to conditions of 273 K (0 °C) and 1 atm.
A note of caution: STP conditions are not identical to standard state conditions. The two standards involve different temperatures and are used for different purposes. STP (273 K and 1 atm) is generally used for gas law calculations; standard state conditions (298 K, 1 atm, 1 M concentrations) are used when measuring standard enthalpy, entropy, free energy changes, and electrochemical cell voltage.
MCAT EXPERTISE
On the MCAT, remember that STP is different from standard state. Temperature at STP is 0 °C (273 K). Temperature at standard state is 25 °C (298 K).
MCAT CONCEPT CHECK 8.1:
Before you move on, assess your understanding of the material with these questions.
-
Name some characteristics that make the gas phase unique:
____________________________
-
A mercury barometer is primarily affected by atmospheric pressure. What would happen to the level of the mercury in the column if:
- the barometer was moved to the top of a mountain?________________________
- the barometer was placed ten meters under water?____________________________
-
What are the conditions for STP?
_______________________________________
-
What are the standard conditions?
__________________________________
8.2 Ideal Gases
LEARNING OBJECTIVES
After Chapter 8.2, you will be able to:
- Apply the ideal gas equation to calculations of pressure, temperature, volume, or number of moles
- Calculate the density of a substance given its molecular formula, current pressure, and current temperature
- Apply Avogadro’s principle, Boyle’s law, Charles’s law, Gay–Lussac’s law, and the combined ideal gas law to given scenarios
- Solve problems using Dalton’s law of partial pressures and Henry’s law
When we examine the behavior of gases under varying conditions of temperature and pressure, we assume that the gases are ideal. An ideal gas represents a hypothetical gas with molecules that have no intermolecular forces and occupy no volume. Although real gases deviate from this ideal behavior at high pressures (low volumes) and low temperatures, many compressed real gases demonstrate behavior that is close to ideal.
KEY CONCEPT
An ideal gas follows the gas laws we will discuss at all pressures and temperatures. A real gas deviates from these laws at high pressures (low volumes) and low temperatures because of intermolecular forces or volume effects.
Ideal Gas Law
The ideal gas law was first stated in 1834 by Benoît Paul Émile Clapeyron, more than 170 years after Sir Robert Boyle performed his experimental studies on the relationship between pressure and volume in the gas state. In fact, by the time the ideal gas law found its expression, Boyle’s law, Charles’s law, and Dalton’s law had already been well-established. Historical considerations aside, it will benefit us to examine the ideal gas law first so that we can then understand the other laws, which had been identified earlier, to be only special cases of the ideal gas law.
The ideal gas law shows the relationship among four variables that define a sample of gas:
PV = nRT
Equation 8.1
where P is the pressure, V is the volume, n is the number of moles, and T is the temperature. R represents the ideal gas constant, which has a value of 8.21 × 10 − 2 L · atm mol · K . Be aware that the gas constant can be expressed in other units. On the MCAT, you may also encounter R given as 8.314 J K · mol , which is derived when SI units of pascal (for pressure) and cubic meters (for volume) are substituted into the ideal gas law. Although the relevant values for R will be provided on Test Day if needed, it is important to recognize the appropriate value for R based on the units of the variables given in a passage or question stem.
The ideal gas law is used to determine the missing term when given all of the others. It can also be used to calculate the change in a term while holding two of the others constant. It is most commonly used to solve for volume or pressure at any given temperature and number of moles; Figure 8.2 shows graphs of P‒V relationships at increasing temperatures.
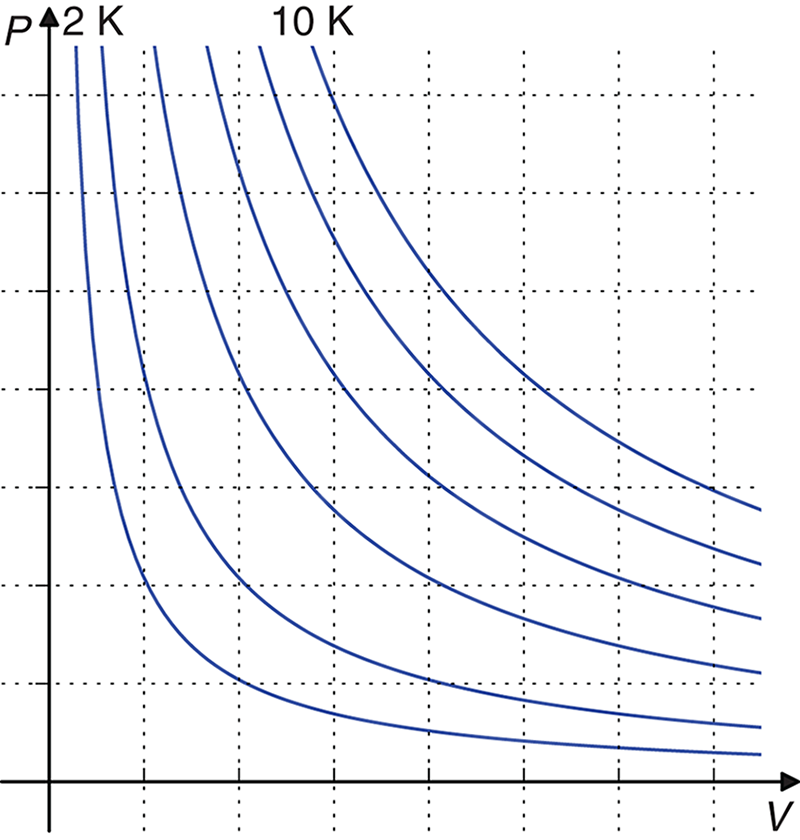
Figure 8.2. Ideal Gas Isothermal Curves Whenn, R, andTare held constant, one can easily analyze the relationship between pressure and volume.
Example: What volume would 12 g of helium occupy at 27 °C and a pressure of 380 mmHg?
Solution: The ideal gas law can be used, but first, all of the variables must be converted to units that will correspond to the expression of the gas constant as 8.21 × 10 − 2 L · atm mol · K . P = 380 mmHg [ 1 atm 760 mmHg ] = 0.5 atm T = 27 ° C + 273 = 300 K n = 12 g He [ 1 mol 4.0 g ] = 3 mol He P V = n R T ( 0.5 atm ) ( V ) = ( 3 mol He ) ( 0.0821 L · atm mol · K ) ( 300 K) 0.5 V ≈ 3 × 8 × 3 V = 144 L ( actual = 148 L )
BRIDGE
Round numbers to speed up your arithmetic on Test Day. For instance, constants such as 0.0821 can be rounded to 0.08. Choices will be sufficiently different so that your estimated answer will be nearly identical to the true answer choice. Arithmetic and math strategies are discussed in Chapter 10 of MCAT Physics and Math Review.
The ideal gas law is useful not only for standard calculations of pressure, volume, or temperature of a gas under a given set of conditions, but also for determinations of gas density and molar mass.
Density
We define density (ρ) as the ratio of the mass per unit volume of a substance. The densities of gases are usually expressed in units of grams per liter. The ideal gas law contains variables for volume and number of moles, so we can rearrange the law to calculate the density of any gas:
P V = n R T where n = m (mass) M (molar mass) Therefore, P V = m M R T and ρ = m V = P M R T
Equation 8.2
A different approach could start with the fact that a mole of an ideal gas at STP occupies 22.4 L. We can then calculate the effect of changes in pressure and temperature when they differ from STP conditions, predicting the volume of the gas. Finally, we’ll calculate the density by dividing the mass by the predicted volume. The following equation, the combined gas law, is an amalgam of some of the special cases we will discuss in the following section. It can be used to relate changes in temperature, volume, and pressure of a gas:
P 1 V 1 T 1 = P 2 V 2 T 2
Equation 8.3
where the subscripts 1 and 2 refer to the two states of the gas (at STP and at the conditions of actual temperature and pressure, for example). This equation assumes the number of moles stays constant.
To calculate a change in volume, the equation is rearranged as follows:
V 2 = V 1 [ P 1 P 2 ] [ T 2 T 1 ]
V2 is then used to find the density of the gas under nonstandard conditions:
ρ = m V 2
On Test Day, it may be helpful to visualize how the changes in pressure and temperature affect the volume of the gas, and this can serve as a check to avoid accidentally switching the values of pressure and temperature in the numerator and denominator. For example, one could predict that doubling the temperature of a gas would result in doubling its volume, and doubling the pressure of a gas would result in halving the volume, so doubling both the temperature and pressure at the same time results in a final volume that is equal to the original volume.
Example: What is the density of CO2 gas at 2 atm and 273 °C?
Solution: At STP, a mole of gas occupies 22.4 L. Because the increase in pressure to 2 atm decreases volume proportionally, 22.4 L must be multiplied by 1 atm 2 atm = 0 . 5 . Because the increase in temperature increases volume proportionally, the temperature factor will be 546 K 273 K = 2 .
V 2 = [ 22.4 L mol ] [ 1 atm 2 atm ] [ 546 K 273 K ] = 22.4 L mol
ρ = 44 g mol 22.4 L mol ≈ 2 g L actual = 1 . 96 g L .
Molar Mass
Sometimes the identity of a gas is unknown, and the molar mass, discussed in Chapter 4 of MCAT General Chemistry Review, can be determined in order to identify it. Using the equation for density derived from the ideal gas law, we can calculate the molar mass of a gas experimentally in the following way:
The pressure and temperature of a gas contained in a bulb of a given volume are measured, and the mass of the bulb with the sample is measured. Then, the bulb is evacuated—the gas is removed—and the mass of the empty bulb is determined. The mass of the bulb with the sample minus the mass of the evacuated bulb gives the mass of the sample. Finally, the density of the sample is determined by dividing the mass of the sample by the volume of the bulb. This gives the density at the given temperature and pressure.
Using V 2 = V 1 [ P 1 P 2 ] [ T 2 T 1 ] , we then calculate the volume of the gas at STP, substituting 273 K for T2 and 1 atm for P2. The ratio of the sample mass divided by V2 gives the density of the gas at STP. The molar mass can then be calculated as the product of the gas’s density at STP and the STP volume of one mole of gas, 22.4 L mol :
M = ( ρ STP ) ( 22.4 L mol )
Example: What is the molar mass of a 22.4 L sample of gas that has a mass of 225 g at a temperature of 273 °C and a pressure of 10 atm?
Solution: Determine how the current conditions compare to STP, and use this to set up a proportional relationship. Be careful to note the differences between degrees C and K, and current versus STP conditions.
P 1 V 1 T 1 = P 2 V 2 T 2 giving V 2 = V 1 [ P 1 P 2 ] [ T 2 T 1 ] V STP = V 1 [ P 1 P STP ] [ T STP T 1 ] = 22 . 4 L [ 10 atm 1 atm ] [ 273 K 546 K ] = 22 . 4 × 10 × 1 2 = 112 L 225 g 112 L ≈ 2 g L at STP M = 2 g L [ 22.4 L mol ] ≈ 44 . 8 g mol
Special Cases
Now that we have considered the ideal gas law as the mathematical relationship between four variables that define the state of a gas (pressure, volume, temperature, and moles of gas), we can examine the other laws that preceded its discovery. Even though the following laws were developed before the ideal gas law, it is conceptually helpful to think of them as special cases of the more general ideal gas law.
Avogadro’s Principle
One important discovery that preceded Clapeyron’s formulation of the ideal gas law was Avogadro’s principle, which states that all gases at a constant temperature and pressure occupy volumes that are directly proportional to the number of moles of gas present. Equal amounts of all gases at the same temperature and pressure will occupy equal volumes. As discussed above, one mole of any gas, irrespective of its chemical identity, will occupy 22.4 liters at STP.
n V = k or n 1 V 1 = n 2 V 2
Equation 8.4
where k is a constant, n1 and n2 are the number of moles of gas 1 and gas 2, respectively, and V1 and V2 are the volumes of the gases, respectively. This can be summarized in the following statement: as the number of moles of gas increases, the volume increases in direct proportion.
Example: A 2.0 L sample at 100 °C and 20 atm contains 5 moles of a gas. If an additional 25 moles of gas at the same pressure and temperature are added, what is the final volume of the gas?
Solution: If pressure and temperature are held constant, the ideal gas law reduces to Avogadro’s principle:
n 1 V 1 = n 2 V 2 5 mol 2.0 L = 5 mol + 25 mol V 2 V 2 = 30 mol × 2 .0 L 5 mol = 12.0 L
Boyle’s Law
Robert Boyle conducted a series of experimental studies in 1660 that led to his formulation of a law that now bears his name: Boyle’s law. His work showed that, for a given gaseous sample held at constant temperature (isothermal conditions), the volume of the gas is inversely proportional to its pressure:
PV = k or P1V1 = P2V2
Equation 8.5
where k is a constant, and the subscripts 1 and 2 represent two different sets of pressure and volume conditions. Careful examination of Boyle’s law shows that it is, indeed, simply the special case of the ideal gas law in which n and T are constant.
KEY CONCEPT
Boyle’s law is a derivation of the ideal gas law and states that pressure and volume are inversely related: when one increases, the other decreases.
A plot of volume vs. pressure for a gas—the inverse of the curves in Figure 8.2—is shown in Figure 8.3.
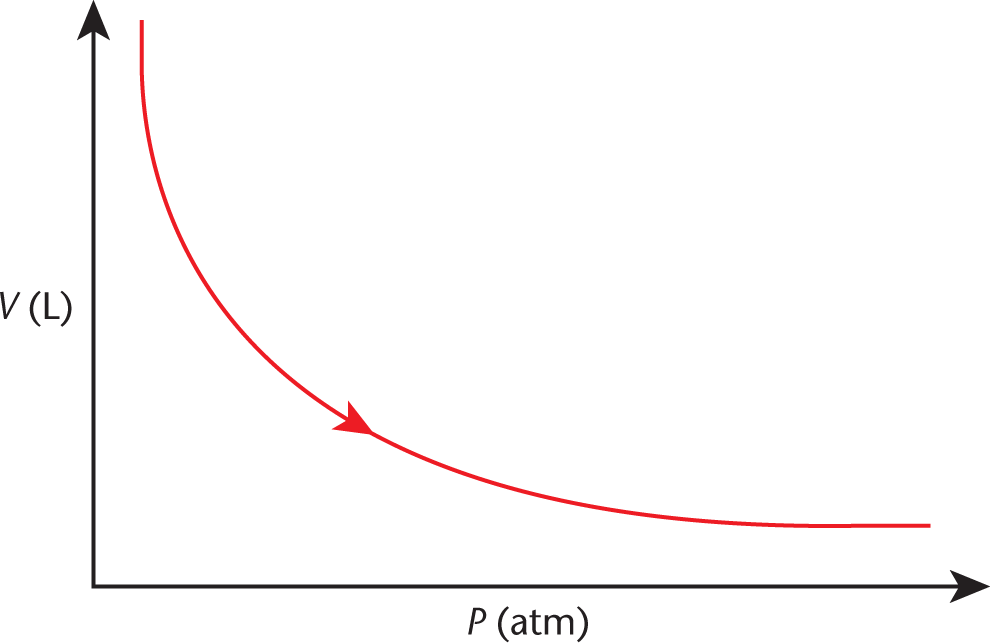
Figure 8.3. Boyle’s Law (Isothermal Compression) As pressure increases, volume decreases.
MCAT EXPERTISE
Sometimes it is easier to remember the shape of the graph to help you recall the variables’ relationship on Test Day. Here we can see that, as pressure increases, the volume decreases, and vice-versa. These ratios and relationships will often allow you to answer questions on the MCAT without having to do much math.
Example: What would be the volume of a 1 L sample of helium if its pressure is changed from 12 atm to 4 atm under isothermal conditions?
Solution: If the number of moles of gas and temperature are held constant, the ideal gas law reduces to Boyle’s law:
P 1 V 1 = P 2 V 2 ( 12 atm)(1 L) = ( 4 atm)( V 2 ) V 2 = 12 atm × 1 L 4 atm = 3 L
Charles’s Law
In the early 19th century, Joseph Louis Gay-Lussac published findings based, in part, on earlier unpublished work by Jacques Charles; hence, the law of Charles and Gay-Lussac is more commonly known simply as Charles’s law. The law states that, at constant pressure, the volume of a gas is proportional to its absolute temperature, expressed in kelvin. Expressed mathematically, Charles’s law is
V T = k or V 1 T 1 = V 2 T 2
Equation 8.6
where, again, k is a proportionality constant and the subscripts 1 and 2 represent two different sets of temperature and volume conditions. Careful examination of Charles’s law shows that it is another special case of the ideal gas law in which n and P are constant.
KEY CONCEPT
Charles’s law is also a derivation of the ideal gas law and states that volume and temperature are directly proportional: when one increases, the other increases in direct proportion.
A plot of temperature vs. volume is shown in Figure 8.4. Note that if one extrapolates the V vs. T plot for a gas back to where T = 0 (absolute zero), we find that V = 0!
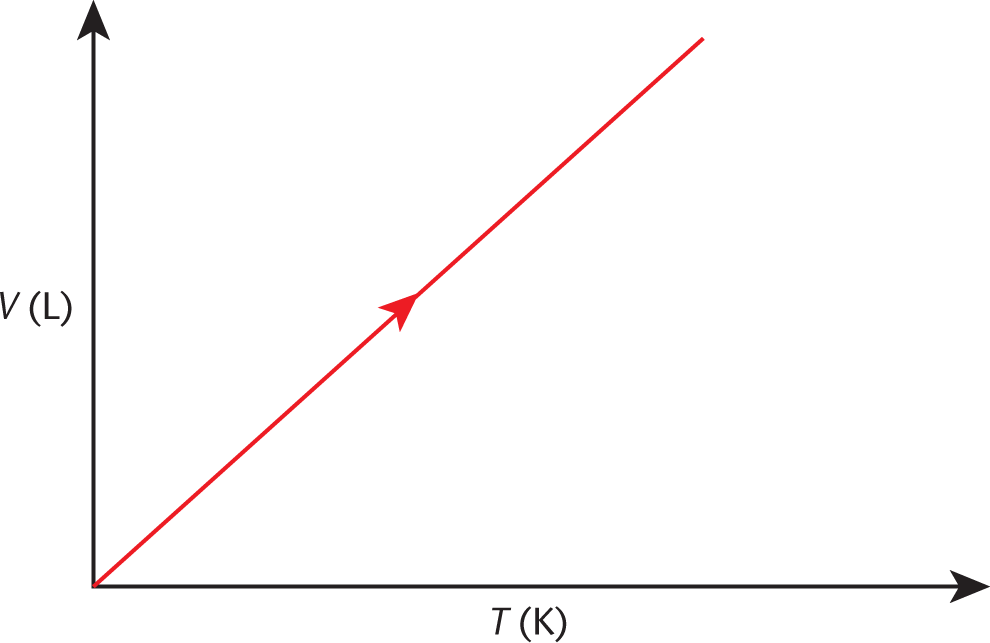
Figure 8.4. Charles’s Law (Isobaric Expansion) As temperature increases, volume increases.
REAL WORLD
While the temperature of 0K cannot be physically attained, curves such as Charles’s law were originally used to figure out its value.
Example: If the temperature of 2 L of gas at constant pressure is changed from 290 K to 580 K, what would be its final volume?
Solution: If the number of moles of gas and pressure are held constant, the ideal gas law reduces to Charles’s law:
V 1 T 1 = V 2 T 2 2 L 290 K = V 2 580 K V 2 = 2 L × 580 K 290 K = 4 L
Gay-Lussac’s Law
Gay-Lussac’s law is complementary to Charles’s Law. It utilizes the same derivation from the ideal gas law, but it relates pressure to temperature instead. Expressed mathematically, Gay-Lussac’s law is
P T = k or P 1 T 1 = P 2 T 2
Equation 8.7
where, again, k is a proportionality constant, and the subscripts 1 and 2 represent two different sets of temperature and pressure conditions. Careful examination of Gay-Lussac’s law shows that it is another special case of the ideal gas law in which n and V are constant.
Figure 8.5 graphs this concept, which is nearly identical to Charles’s law. Again, an increase in temperature will increase the pressure in direct proportion.
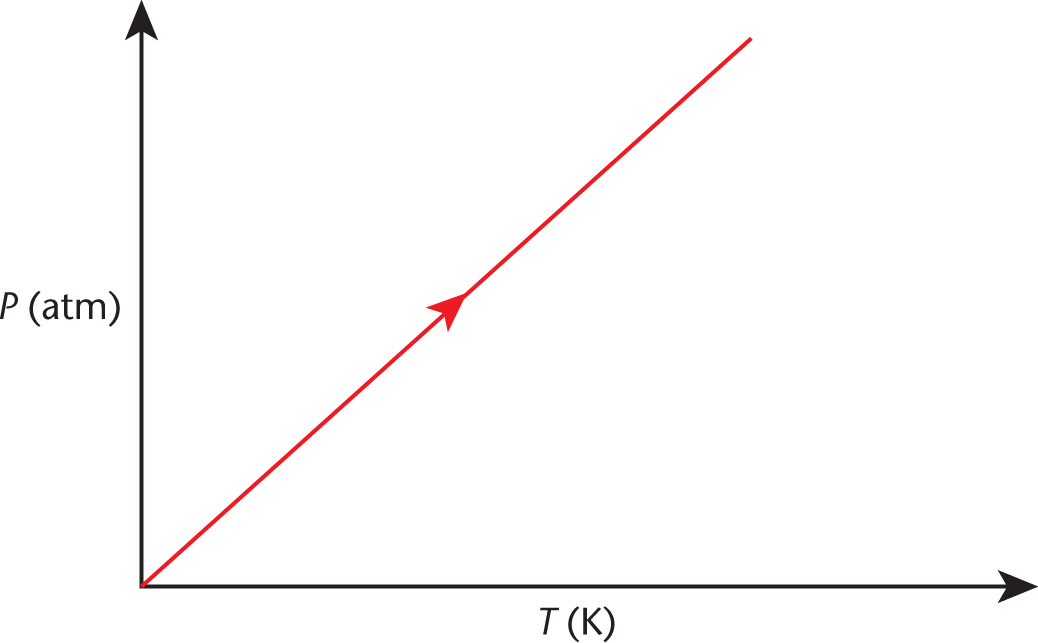
Figure 8.5. Gay-Lussac’s Law (Isovolumetric Heating) As temperature increases, pressure increases.
Example: If the pressure of a sample of gas with a temperature of 300 K changes from 2 atm to 5 atm during heating, what would be the final temperature if volume is held constant?
Solution: If the number of moles of gas and volume are held constant, the ideal gas law reduces to Gay-Lussac’s law:
P 1 T 1 = P 2 T 2 2 atm 300 K = 5 atm T 2 T 2 = 5 atm × 300 K 2 atm = 750 K
Combined Gas Law
As discussed earlier, the combined gas law (Equation 8.3) was a combination of many of the preceding laws. This law relates pressure and volume (Boyle’s law) in the numerator, and relates the variations in temperature to both volume (Charles’s law) and pressure (Gay-Lussac’s law) simultaneously. When using this equation, take care to place all of the variables in the right place.
MCAT EXPERTISE
Understanding how the combined gas law functions helps avoid the need to memorize every other special case of the ideal gas law. Read the question stem or passage with an eye toward the quantities that remain constant to know when assumptions can be made.
Dalton’s Law of Partial Pressures
When two or more gases that do not chemically interact are found in one vessel, each gas will behave independently of the others. That is, each gas will behave as if it were the only gas in the container. Therefore, the pressure exerted by each gas in the mixture will be equal to the pressure that the gas would exert if it were the only one in the container. The pressure exerted by each individual gas is called the partial pressure of that gas. In 1801, John Dalton derived an expression, now known as Dalton’s law of partial pressures, which states that the total pressure of a gaseous mixture is equal to the sum of the partial pressures of the individual components. The equation for Dalton’s law is
PT = PA + PB + PC + …
Equation 8.8
where PT is the total pressure in the container, and PA, PB, and PC are the partial pressures of gases A, B, and C, respectively.
KEY CONCEPT
When more than one gas is in a container, each contributes to the whole as if it were the only gas present. Add up all of the pressures of the individual gases and you get the whole pressure of the system.
The partial pressure of a gas is related to its mole fraction and can be determined using the following equation:
P A = X A P T where X A = moles of gas A total moles of gas
Equation 8.9 Example: A vessel contains 0.75 mol of nitrogen, 0.20 mol of hydrogen, and 0.05 mol of fluorine at a total pressure of 2.5 atm. What is the partial pressure of each gas?
Solution: First calculate the mole fraction of each gas.
X N 2 = 0.75 mol 1.00 mol = 0.75 X H 2 = 0.20 mol 1.00 mol = 0.20 X F 2 = 0.05 mol 1.00 mol = 0.05
Then calculate the partial pressure.
P A = X A P T P N 2 = ( 0.75 ) ( 2.5 atm) = 1.875 atm P H 2 = ( 0.20 ) ( 2.5 atm) = 0.5 atm P F 2 = ( 0.05 ) ( 2.5 atm) = 0.125 atm
Henry’s Law
The difference in gas solubility between fluids was explained by William Henry in 1803. What Henry noticed was that, at various applied pressures, the concentration of a gas in a liquid increased or decreased. This was a characteristic of a gas’s vapor pressure. Vapor pressure is the pressure exerted by evaporated particles above the surface of a liquid. Evaporation, as discussed in Chapter 7 of MCAT General Chemistry Review, is a dynamic process that requires the molecules at the surface of a liquid to gain enough energy to escape into the gas phase.
Vapor pressure from the evaporated molecules forces some of the gas back into the liquid phase, and equilibrium is reached between evaporation and condensation. Mathematically, this is expressed as:
[ A ] = k H × P A or [ A ] 1 P 1 = [ A ] 2 P 2 = k H
Equation 8.10
where [A] is the concentration of A in solution, kH is Henry’s constant, and PA is the partial pressure of A. The value of Henry’s constant depends on the identity of the gas.
KEY CONCEPT
The solubility of a gas will increase with increasing partial pressure of the gas.
According to this relationship, solubility (concentration) and pressure are directly related. In biology, this is a critically important relationship for gas and nutrient exchange. As discussed in Chapter 6 of MCAT Biology Review, lung tissue—at the microscopic level—is organized into grapelike clusters of sacs called alveoli. These sacs are perfused by capillaries that allow for the exchange of carbon dioxide and oxygen, as shown in Figure 8.6. If the atmospheric pressure changes, as it does from sea level to high altitude, then the partial pressure of oxygen in the atmosphere also changes (as explained by Dalton’s law), and the amount of gas exchanged is altered accordingly; if the partial pressure of a particular gas is elevated, such as when giving hyperbaric oxygen, the amount of that gas dissolved in the blood is also elevated.
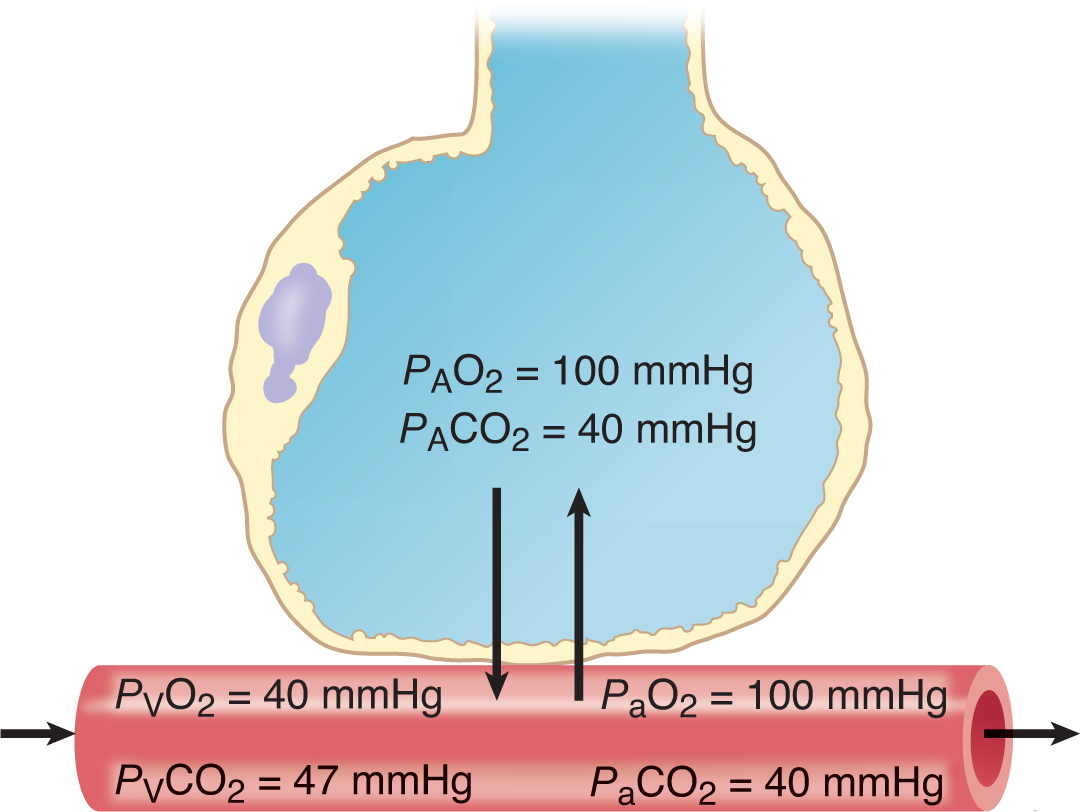
Figure 8.6. Alveolar Capillary Gas Exchange In medicine,Arepresents alveolar concentrations,Vrepresents venous concentrations, andarepresents arterial concentrations.
Example: If 4 × 10−4 moles of gas are dissolved in 2 L of solution under an ambient pressure of 2 atm, what will be the molar concentration of the gas under 10 atm?
Solution: Start by determining the initial concentration of the gas in solution.
[ A ] 1 = 4 × 10 − 4 mol 2 L = 2 × 10 − 4 M
Next, utilize the direct relationship between solubility and pressure according to Henry’s law.
[ A ] 1 P 1 = [ A ] 2 P 2 2 × 10 − 4 M 2 atm = [ A ] 2 10 atm [ A ] 2 = ( 2 × 10 − 4 M ) ( 10 atm ) 2 atm = 10 − 3 M
MCAT CONCEPT CHECK 8.2:
Before you move on, assess your understanding of the material with these questions.
-
A container with 4 moles of a gas at a pressure of 8 atm has a volume of 12 liters. What is its temperature? (Note: R = 8.21 × 10 − 2 L · atm mol · K )
_______________________________________
-
What is the density of argon gas at 4 atm and 127 °C?
_______________________________________
-
A 20 L sample at 300 °C and 5 atm of pressure contains 2 moles of a gas. If an additional 3 moles of gas at the same pressure and temperature are added, what is the final total volume of the gas?
_____________________________
-
What would be the volume of a 2 L sample of neon if its pressure is changed from 1 atm to 40 atm under isothermal conditions?
_____________________________
-
If the temperature of 6 L of gas at constant pressure is changed from 27 °C to 127 °C, what would be its final volume?
_____________________________
-
If the pressure of a sample of gas with a temperature of 227 °C is changed from 5 atm to 2 atm during cooling, what would be the final temperature?
_____________________________
-
A vessel contains 8 mol O2, 3 mol CH4, and 1 mol CO2 at a total pressure of 240 atm. What is the partial pressure of each gas?
_____________________________
-
How can the concentration of carbon dioxide in sodas or other carbonated beverages be so much higher than that of atmospheric carbon dioxide?
__________________________________
8.3 Kinetic Molecular Theory
LEARNING OBJECTIVES
After Chapter 8.3, you will be able to:
- Recall the assumptions made by kinetic molecular theory
- Calculate the average speed of a gas, given its temperature
- Compare the relative speeds of two different gases at the same temperature
- Apply Graham’s law to situations involving gas effusion:
The kinetic molecular theory was developed in the second half of the 19th century, well after the laws describing gas behavior had been developed. The kinetic molecular theory was used to explain the behavior of gases, which the other laws merely described. The gas laws demonstrate that all gases show similar physical characteristics and behavior irrespective of their particular chemical identity. The behavior of real gases deviates from the ideal behavior predicted under the assumptions of this theory, but these deviations can be corrected for in calculations. The combined efforts of James Maxwell, Ludwig Boltzmann, and others led to a simple explanation of gaseous molecular behavior based on the motion of individual molecules. Like the gas laws, the kinetic molecular theory was developed in reference to ideal gases, although it can be applied with reasonable accuracy to real gases as well.
Assumptions
To simplify the model proposed by the kinetic molecular theory, certain assumptions are made:
- Gases are made up of particles with volumes that are negligible compared to the container volume.
- Gas atoms or molecules exhibit no intermolecular attractions or repulsions.
- Gas particles are in continuous, random motion, undergoing collisions with other particles and the container walls.
- Collisions between any two gas particles (or between particles and the container walls) are elastic, meaning that there is conservation of both momentum and kinetic energy.
- The average kinetic energy of gas particles is proportional to the absolute temperature of the gas (in kelvin), and it is the same for all gases at a given temperature, irrespective of chemical identity or atomic mass.
Applications
It is fairly straightforward to imagine gas particles as little rubber balls bouncing off each other and off the walls of the container. Of course, rubber balls, like real gas particles, have measurable mass and volume, and not even the bounciest rubber balls will collide in a completely elastic manner. Still, this provides an apt visualization of the behaviors described by the kinetic molecular theory.
Average Molecular Speeds
According to the kinetic molecular theory of gases, the average kinetic energy of a gas particle is proportional to the absolute temperature of the gas:
K E = 1 2 m v 2 = 3 2 k B T
Equation 8.11
where kB is the Boltzmann constant ( 1.38 × 10 − 23 J K ) , which serves as a bridge between the macroscopic and microscopic behaviors of gases (that is, as a bridge between the behavior of the gas as a whole and the individual gas molecules). This equation shows that the speed of a gas particle is related to its absolute temperature. However, because of the large number of rapidly and randomly moving gas particles, which may travel only nanometers before colliding with another particle or the container wall, the speed of an individual gas molecule is nearly impossible to define. Therefore, the speeds of gases are defined in terms of their average molecular speed. One way to define an average speed is to determine the average kinetic energy per particle and then calculate the speed to which this corresponds. The resultant quantity, known as the root-mean-square speed (urms), is given by the following equation:
u rms = 3 R T M
Equation 8.12
where R is the ideal gas constant, T is the temperature, and M is the molar mass.
MCAT EXPERTISE
Understanding concepts will be much more fruitful on Test Day than memorizing all of the facts. The higher the temperature, the faster the molecules move. The larger the molecules, the slower they move.
A Maxwell–Boltzmann distribution curve shows the distribution of gas particle speeds at a given temperature. Figure 8.7 shows a distribution curve of molecular speeds at two temperatures, T1 and T2, where T2 is greater than T1. Notice that the bell-shaped curve flattens and shifts to the right as the temperature increases, indicating that at higher temperatures, more molecules are moving at higher speeds.
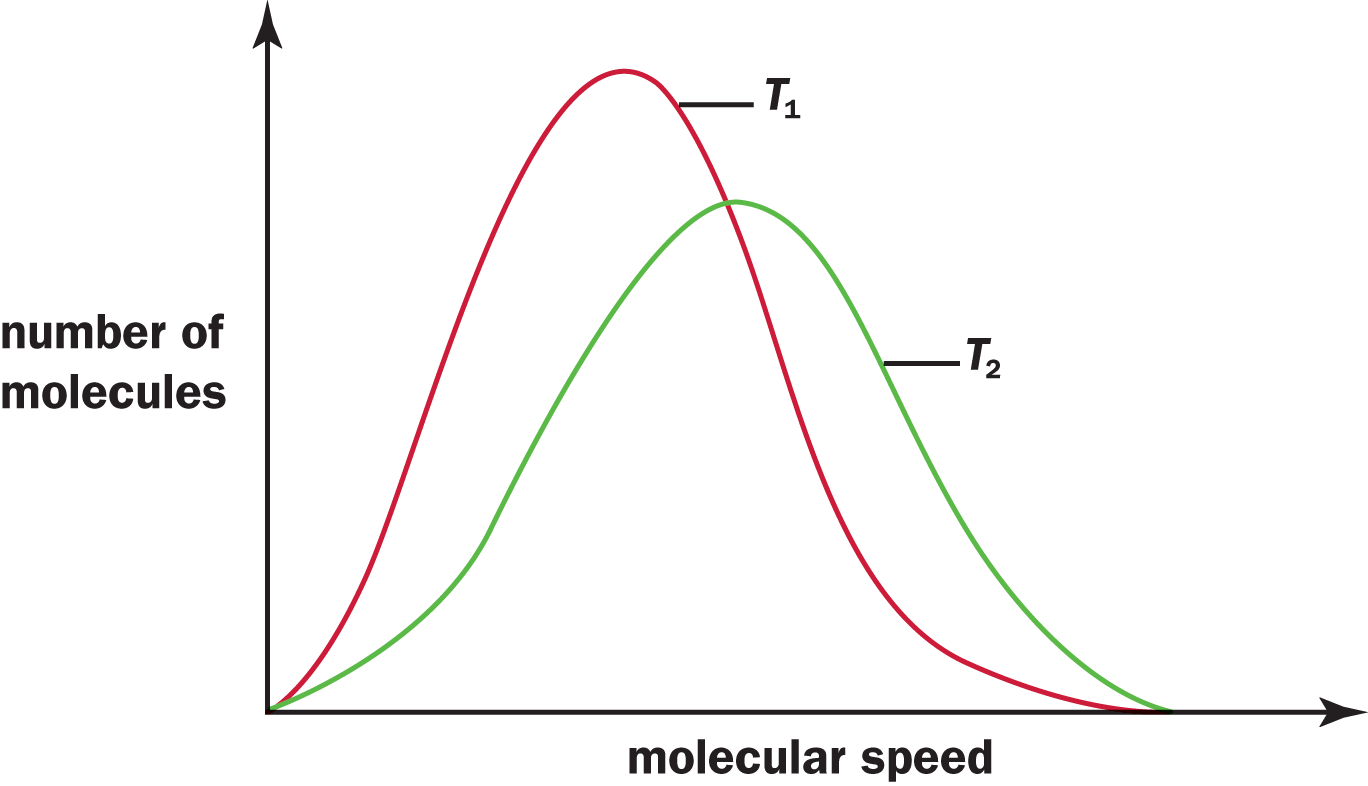
Figure 8.7. Maxwell–Boltzmann Distribution Curves of Molecular Speeds at Two Different Temperatures
Example: What is the average speed of xenon difluoride molecules at 20 °C?
Solution: The ideal gas constant R = 8.314 J K · mol should be used, and the molar mass of xenon difluoride is 169.3 g mol . M must be expressed in kg mol because joules are also derived from kilograms.
u rms = 3 RT M u rms = ( 3 ) ( 8 . 314 J K · mol ) ( 293 K ) 0 . 17 kg mol u rms ≈ ( 3 ) ( 8 ) ( 300 ) 0.18 = ( 8 ) ( 300 ) 0 . 06 = ( 8 ) ( 5 , 000 ) = 40 , 000 u rms ≈ 200 m s ( actual = 208 m s )
Graham’s Law of Diffusion and Effusion
The movement of molecules from high concentration to low concentration through a medium (such as air or water) is called diffusion, as shown in Figure 8.8.
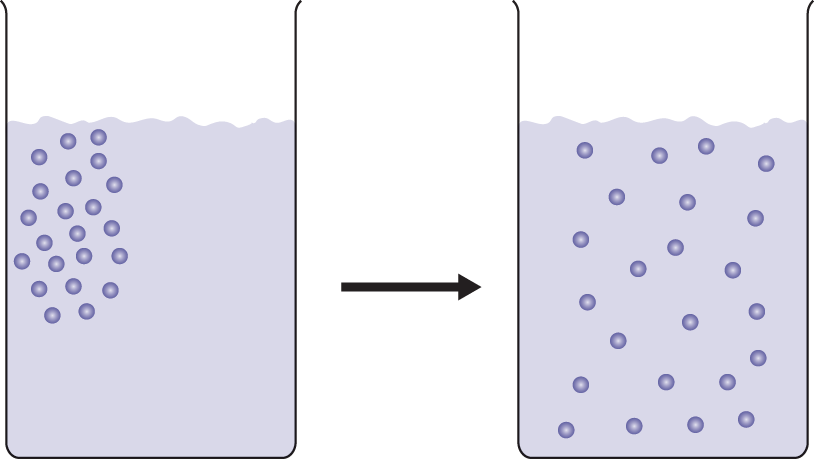
Figure 8.8. Diffusion of Solutes in a Solvent
The kinetic molecular theory of gases predicts that heavier gases diffuse more slowly than lighter ones because of their differing average speeds, as shown in Figure 8.9. Because all gas particles have the same average kinetic energy at the same temperature, it must be true that particles with greater mass travel at a slower average speed.
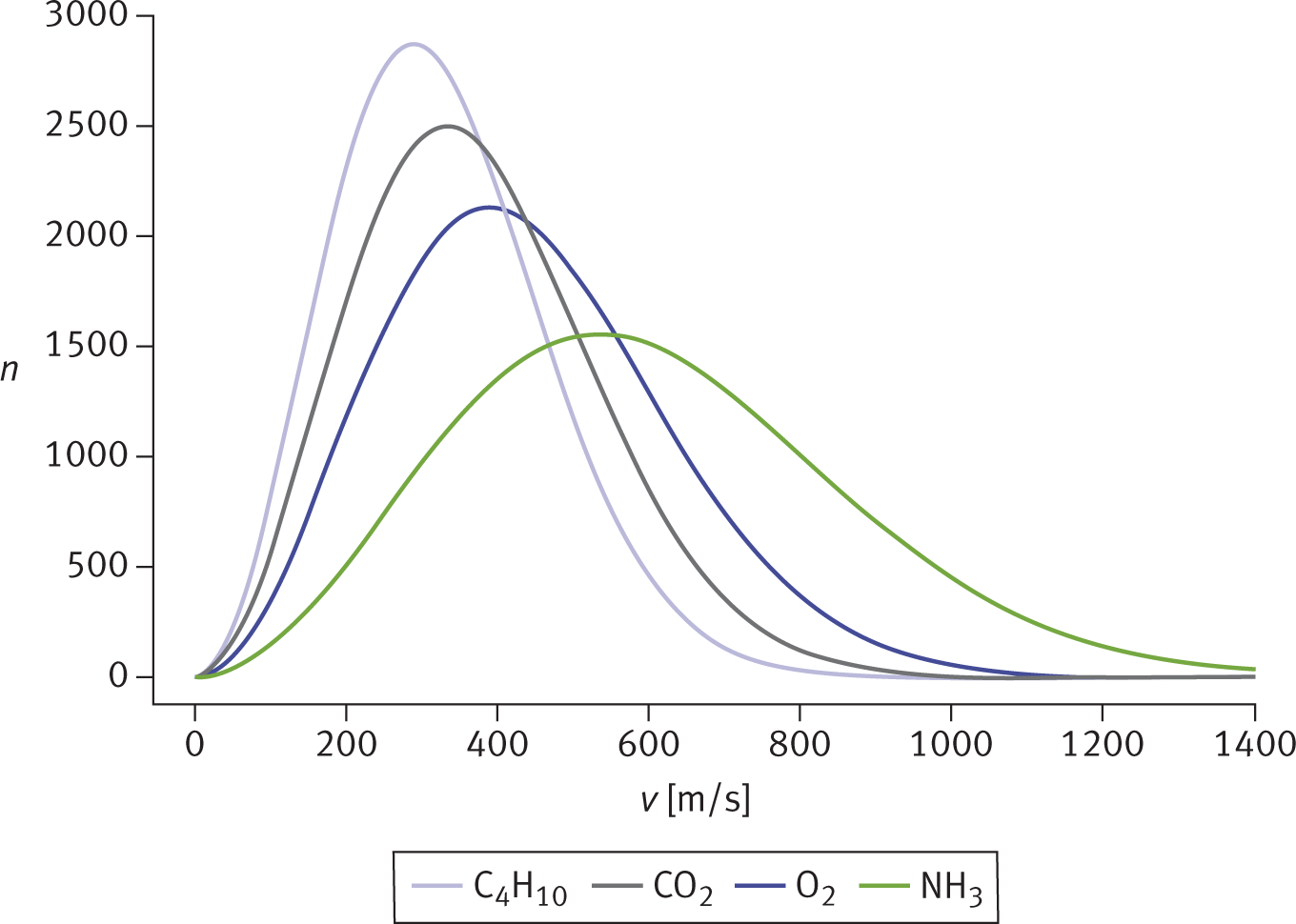
Figure 8.9. Maxwell–Boltzmann Distribution Curves of Molecular Speeds for Gases with Different Molar Masses The more massive the gas particles, the slower their average speed.
In 1832, Thomas Graham showed mathematically that, under isothermal and isobaric conditions, the rates at which two gases diffuse are inversely proportional to the square roots of their molar masses. This is called Graham’s law, which is written mathematically as:
r 1 r 2 = M 2 M 1
Equation 8.13
where r1 and r2 are the diffusion rates of gas 1 and gas 2, respectively, and M1 and M2 are the molar masses of gas 1 and gas 2, respectively. From this equation, we can see that a gas that has a molar mass four times that of another gas will travel half as fast as the lighter gas.
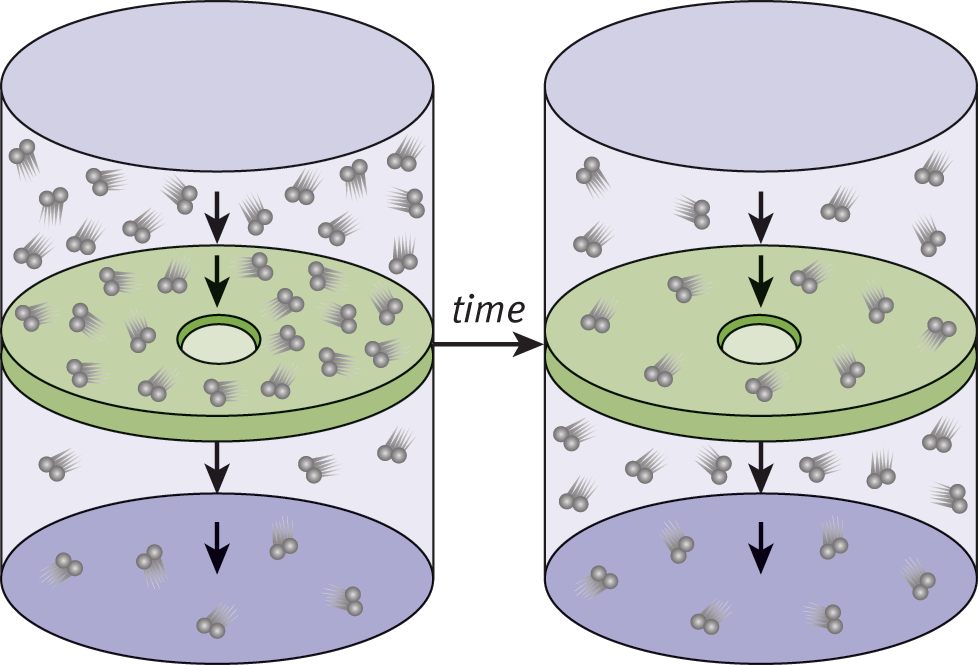
Effusion is the flow of gas particles under pressure from one compartment to another through a small opening, as shown in Figure 8.10. Graham used the kinetic molecular theory of gases to show that, for two gases at the same temperature, the rates of effusion are proportional to the average speeds. He then expressed the rates of effusion in terms of molar mass and found that the relationship is the same as that for diffusion.

Figure 8.10. Effusion of Gas Particles Effusion is the flow of gas particles under pressure from one compartment to another through a small opening.
REAL WORLD
Clinically, a pleural effusion is a condition in which fluid enters the intrapleural space through small openings in the capillaries or lymphatic vessels. This causes a pressure buildup around the lungs that hinders breathing.
KEY CONCEPT
- Diffusion—When gases mix with one another.
- Effusion—When a gas moves through a small hole under pressure.
- Both will be slower for larger molecules.
- Both conditions use the same equation.
Example: Oxygen molecules travel at an average speed of approximately 500 m s at a given temperature. Calculate the average speed of hydrogen molecules at the same temperature.
Solution: Oxygen’s molar mass is 32 g mol . Hydrogen’s molar mass is 2 g mol . Plugging into Graham’s law, we get:
r 1 r 2 = M 2 M 1 → r 2 = r 1 M 1 M 2 r 2 = 500 m s 32 g mol 2 g mol = 500 m s × 4 = 2000 m s
MCAT CONCEPT CHECK 8.3:
Before you move on, assess your understanding of the material with these questions.
-
What are the assumptions made by the kinetic molecular theory?
____________________________
-
What is the average speed of helium atoms at –173 °C?
_____________________________
-
If neon gas travels at 400 m s at a given temperature, calculate the average speed of krypton at the same temperature.
_____________________________
-
Hydrogen sulfide (H2S) has a very strong rotten egg odor. Methyl salicylate (C8H8O) has a wintergreen odor, and benzaldehyde (C7H6O) has a pleasant almond odor. If the vapors for these three substances were released at the same time from across a room, in which order would one smell the odors? Explain your answer.
__________________________________
8.4 Real Gases
LEARNING OBJECTIVES
After Chapter 8.4, you will be able to:
- Distinguish between real gases and ideal gases
- Predict how differences in attractive forces or volumes will affect real gas behavior
Throughout our discussions of the laws and theory that describe and explain the behaviors of gases, we have stressed that the fundamental assumption is a gas that behaves ideally. However, our world is not one of ideal gases but rather real ones. Real gases have particles that occupy nonnegligible volumes and that interact with each other in measurable ways. In general, the ideal gas law is a good approximation of the behavior of real gases, but all real gases deviate from ideal gas behavior to some extent, particularly when the gas atoms or molecules are forced into close proximity under high pressure (at low volume) or at low temperature. These effects are implied by Figure 8.11, which shows isothermal lines on a pressure–volume graph for a real gas. Compare these lines to the ideal isotherms in Figure 8.2. Under these nonideal conditions, the intermolecular forces and the particles’ volumes become significant.
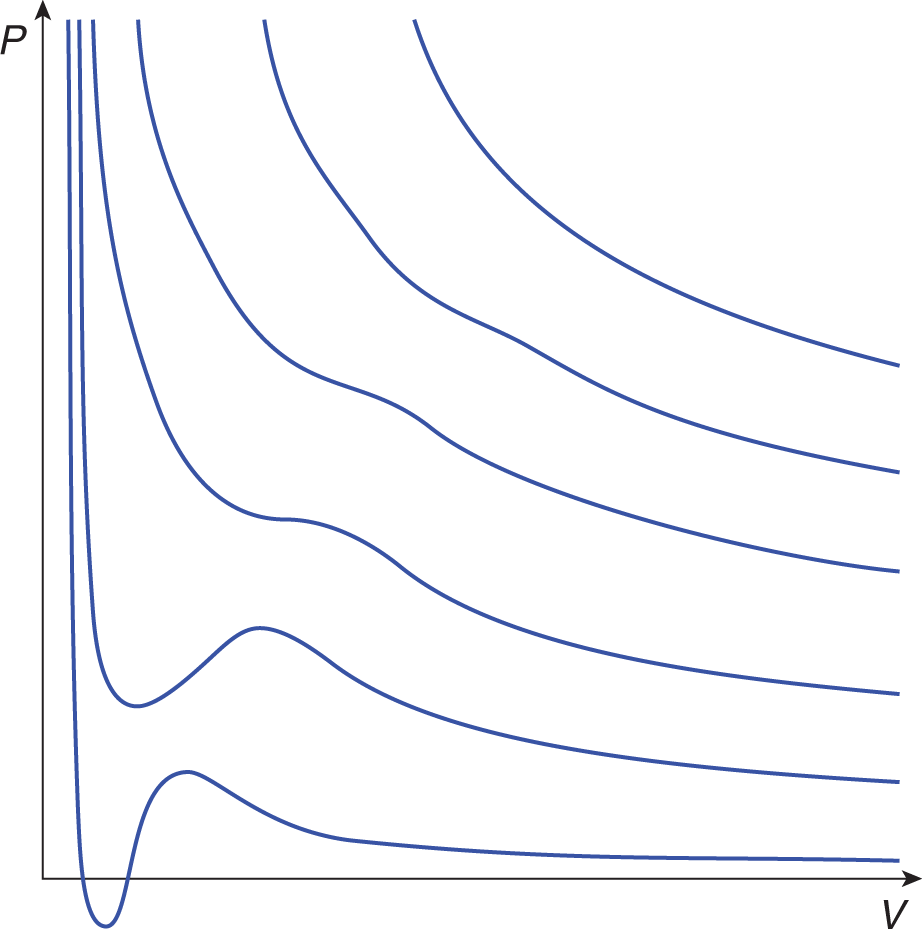
Figure 8.11. Real Gas Isothermal Curves Compare these lines to the ideal gas isotherms in Figure 8.2.
MCAT EXPERTISE
At high temperature and low pressure (high volume), deviations from ideality are usually small; good approximations can still be made from the ideal gas law.
Deviations Due to Pressure
As the pressure of a gas increases, the particles are pushed closer and closer together. As the condensation pressure for a given temperature is approached, intermolecular attraction forces become more and more significant, until the gas condenses into a liquid.
MCAT EXPERTISE
On the MCAT, an understanding of nonideal conditions will help with determining how gases’ behaviors may deviate.
At moderately high pressure (a few hundred atmospheres), a gas’s volume is less than would be predicted by the ideal gas law due to intermolecular attraction. At extremely high pressures, however, the size of the particles becomes relatively large compared to the distance between them, and this causes the gas to take up a larger volume than would be predicted by the ideal gas law. That is, while the ideal gas law assumes that a gas can be compressed to take up zero volume, this is not actually physically possible—the gas particles themselves will take up space.
Deviations Due to Temperature
As the temperature of a gas is decreased, the average speed of the gas molecules decreases and the attractive intermolecular forces become increasingly significant. As the condensation temperature is approached for a given pressure, intermolecular attractions eventually cause the gas to condense to a liquid state.
Like deviations due to pressure, as the temperature of a gas is reduced toward its condensation point (which is the same as its boiling point), intermolecular attraction causes the gas to have a smaller volume than that which would be predicted by the ideal gas law. The closer a gas is to its boiling point, the less ideally it acts. At extremely low temperatures, gases will again occupy more space than predicted by the ideal gas law because the particles cannot be compressed to zero volume.
van der Waals Equation of State
There are several gas equations that attempt to correct for the deviations from ideality that occur when a gas does not closely follow the ideal gas law. The van der Waals equation of state is one such equation:
( P + n 2 a V 2 ) ( V − n b ) = n R T
Equation 8.14
where a and b are physical constants experimentally determined for each gas. The a term corrects for the attractive forces between molecules and, as such, will be smaller for gases that are small and less polarizable (such as helium), larger for gases that are larger and more polarizable (such as Xe or N2), and largest for polar molecules such as HCl and NH3. The b term corrects for the volume of the molecules themselves. Larger molecules thus have larger values of b. Numerical values for a are generally much larger than those for b.
KEY CONCEPT
Note that if a and b are both zero, the van der Waals equation of state reduces to the ideal gas law.
MNEMONIC
- a is the van der Waals term for the attractive forces.
- b is the van der Waals term for big particles.
Example: By what percentage does the real pressure of 1 mole of ammonia in a 1 liter flask at 227 °C deviate from its ideal pressure? (Note: R = 0. 0821 L · atm mol · K ; for NH3, a = 4.2, b = 0.037)
Solution: According to the ideal gas law,
P = n R T V = ( 1 mol ) ( 0.0821 L · atm mol · K ) ( 500 K ) 1 L = ( 0 . 0821 ) ( 1000 ) 2 = 82 . 1 2 = 41 . 5 atm
According to the van der Waals equation of state,
P = n R T V − n b − n 2 a V 2 = [ ( 1 mol ) ( 0.0821 L · atm mol · K ) ( 500 K ) 1 L − ( 1 mol)(0 .037) ] − [ ( 1 mol) 2 ( 4.2 ) ( 1 L) 2 ] P = 41 . 5 0.963 − 4.2 ≈ [ 41 . 5 + 4 % ( 41 . 5 ) ] ≈ 43 - 4 ≈ 39 atm ( actual = 38.8 atm )
The pressure is thus approximately 41.5 - 38.8 = 2.7 atm less than would be predicted from the ideal gas law, representing an error of 2 . 7 atm 41 . 5 atm × 100 % ≈ 3 40 × 100 % = 7 . 5 percent .
MCAT EXPERTISE
Be familiar with the concepts embodied by this equation but do not bother memorizing it; if the testmakers want you to apply the equation, it will be provided in a passage or question stem.
MCAT CONCEPT CHECK 8.4:
Before you move on, assess your understanding of the material with these questions.
-
In what ways do real gases differ from ideal gases?
_____________________________
-
Which gas will exert a higher pressure under the same, nonideal conditions: methane or chloromethane?
_____________________________
-
If methane and isobutane are placed in the same size container under the same conditions, which will exert the higher pressure (consider both as having negligible attractive forces)?
__________________________________
Conclusion
In this chapter, we reviewed the basic characteristics and behaviors of gases. The ideal gas law shows the mathematical relationship among four variables associated with gases: pressure, volume, temperature, and number of moles. We examined special cases of the ideal gas law in which temperature (Boyle’s law), pressure (Charles’s law), or volume (Gay-Lussac’s law) is held constant. Henry’s law helped explain the principles behind dissolution of gases in liquids and gas exchange in biological systems. We also examined Dalton’s law, which relates the partial pressure of a gas to its mole fraction and the sum of the partial pressures of all the gases in a system to the total pressure of the system. The kinetic molecular theory of gases provided the explanation for the behaviors of ideal gases as described by the ideal gas law. Finally, we examined the ways in which real gases deviate from the predicted behaviors of ideal gases. The van der Waals equation of state is a useful equation for correcting deviations caused by molecular interactions and volumes.
From helium-filled balloons to the bubbles of carbon dioxide in a glass of soda, from the pressurized gases used for scuba diving to the air we breathe on land, gases are all around us. And yet, all the different gases that bubble, flow, and settle in and through our daily living experiences behave in remarkably similar ways. Human life is dependent on the exchange of two gases: oxygen and carbon dioxide—to that end, expect that the MCAT will frequently test gases because of their importance in our everyday lives.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
The Gas Phase
- Gases are the least dense phase of matter.
- Gases are fluids and therefore conform to the shapes of their containers.
- Gases are easily compressible.
- Gas systems are described by the variables temperature (T), pressure (P), volume (V), and number of moles (n).
- Important pressure equivalencies include 1 atm = 760 mmHg ≡ 760 torr = 101.325 kPa.
- A simple mercury barometer measures incident (usually atmospheric) pressure. As pressure increases, more mercury is forced into the column, increasing its height. As pressure decreases, mercury flows out of the column under its own weight, decreasing its height.
Ideal Gases
- Standard temperature and pressure (STP) is 273 K (0 °C) and 1 atm.
- Equations for ideal gases assume negligible mass and volume of gas molecules.
- Regardless of the identity of the gas, equimolar amounts of two gases will occupy the same volume at the same temperature and pressure. At STP, one mole of an ideal gas occupies 22.4 L.
- The ideal gas law describes the relationship between the four variables of the gas state for an ideal gas.
- Avogadro’s principle is a special case of the ideal gas law for which the pressure and temperature are held constant; it shows a direct relationship between the number of moles of gas and volume.
- Boyle’s law is a special case of the ideal gas law for which temperature and number of moles are held constant; it shows an inverse relationship between pressure and volume.
- Charles’s law is a special case of the ideal gas law for which pressure and number of moles are held constant; it shows a direct relationship between temperature and volume.
- Gay-Lussac’s law is a special case of the ideal gas law for which volume and number of moles are held constant; it shows a direct relationship between temperature and pressure.
- The combined gas law is a combination of Boyle’s, Charles’s, and Gay-Lussac’s laws; it shows an inverse relationship between pressure and volume along with direct relationships between pressure and volume with temperature.
- Dalton’s law of partial pressures states that individual gas components of a mixture of gases will exert individual pressures in proportion to their mole fractions. The total pressure of a mixture of gases is equal to the sum of the partial pressures of the component gases.
- Henry’s law states that the amount of gas dissolved in solution is directly proportional to the partial pressure of that gas at the surface of a solution.
Kinetic Molecular Theory
- The kinetic molecular theory attempts to explain the behavior of gas particles. It makes a number of assumptions about the gas particles:
- Gas particles have negligible volume.
- Gas particles do not have intermolecular attractions or repulsions.
- Gas particles undergo random collisions with each other and the walls of the container.
- Collisions between gas particles (and with the walls of the container) are elastic.
- The average kinetic energy of the gas particles is directly proportional to temperature.
- Graham’s law describes the behavior of gas diffusion or effusion, stating that gases with lower molar masses will diffuse or effuse faster than gases with higher molar masses at the same temperature.
- Diffusion is the spreading out of particles from high to low concentration.
- Effusion is the movement of gas from one compartment to another through a small opening under pressure.
Real Gases
- Real gases deviate from ideal behavior under high pressure (low volume) and low temperature conditions.
- At moderately high pressures, low volumes, or low temperatures, real gases will occupy less volume than predicted by the ideal gas law because the particles have intermolecular attractions.
- At extremely high pressures, low volumes, or low temperatures, real gases will occupy more volume than predicted by the ideal gas law because the particles occupy physical space.
- The van der Waals equation of state is used to correct the ideal gas law for intermolecular attractions (a) and molecular volume (b).
ANSWERS TO CONCEPT CHECKS
**8.1**
- Gases are compressible fluids with rapid molecular motion, large intermolecular distances, and weak intermolecular forces.
- At the top of the mountain, atmospheric pressure is lower, causing the column to fall. Under water, hydrostatic pressure is exerted on the barometer in addition to atmospheric pressure, causing the column to rise.
- STP: T = 273 K (0 °C), P = 1 atm
- Standard conditions: T = 298 K (25 °C), P = 1 atm, concentrations = 1 M
**8.2**
- P V = n R T → T = P V n R = ( 8 atm ) ( 12 L ) ( 4 mol ) ( 0.0821 L · atm mol · K ) ≈ 8 × 3 0 . 08 = 3 0 . 01 = 300 K ( actual = 292 . 3 K )
- ρ = m V = P M R T = ( 4 atm ) ( 39.9 g mol ) ( 0.0821 L · atm mol · K ) ( 400 K ) ≈ 5 g L ( actual = 4. 85 g L )
- n 1 V 1 = n 2 V 2 → V 2 = n 2 × V 1 n 1 = ( 2 + 3 mol)(20 L) 2 mol = 50 L
- P 1 V 1 = P 2 V 2 → V 2 = P 1 V 1 P 2 = ( 1 atm)(2 L) 40 atm = 0.05 L
- V 1 T 1 = V 2 T 2 → V 2 = V 1 × T 2 T 1 = ( 6 L)(400 K) 300 K = 8 L
- P 1 T 1 = P 2 T 2 → T 2 = P 2 × T 1 P 1 = ( 2 atm)(500 K) 5 atm = 200 K
- There are twelve total moles of gas, so the mole fractions of each gas are:
X O 2 = 8 mol O 2 12 mol = 2 3 = 0.67 ; X CH 4 = 3 mol CH 4 12 mol = 1 4 = 0.25 ; X CO 2 = 1 mol CO 2 12 mol = 0.083
Then multiply each mole fraction by the total pressure to get the partial pressures (this is typically simpler with fractions than with decimals):
P O 2 = 2 3 ( 240 atm) = 160 atm; P CH 4 = 1 4 ( 240 atm) = 60 atm; P CO 2 = 1 12 ( 240 atm) = 20 atm
- High pressures of carbon dioxide gas are forced on top of the liquid in sodas, increasing its concentration in the liquid.
**8.3**
- Assumptions in the kinetic molecular theory include: negligible volume of gas particles, no intermolecular forces, random motion, elastic collisions, and proportionality between absolute temperature and energy.
- u rms = 3 R T M = 3 ( 8.314 J mol · K ) ( 100 K ) 0.004 kg mol ≈ 3 8 100 4 × 10 - 3 = 3 8 25 10 3 u rms = 3 200 10 3 = 600 × 10 3 = 60 × 10 4 ≈ 64 × 10 4 u rms = 8 × 10 2 = 800 m s actual = 790 m s
- r 1 r 2 = M 2 M 1 → r 2 = r 1 M 1 M 2 = 400 m s 20.2 g mol 83.8 g mol ≈ 400 × 1 2 = 200 m s ( actual = 196.4 m s )
- The rotten egg odor (hydrogen sulfide) first, almond (benzaldehyde) next, and wintergreen (methyl salicylate) last. Because all of the gases have the same temperature, they have the same kinetic energy; thus, the lightest molecules travel the fastest.
**8.4**
- Real gas molecules have nonnegligible volume and attractive forces. Real gases deviate from ideal gases at high pressure (low volume) and low temperature.
- According to the van der Waals equation, if a is increased while b remains negligible, the correction term ( n 2 a V 2 ) gets larger, and the pressure drops to compensate. Therefore, methane will behave more ideally than chloromethane because a is smaller for methane. The real pressure of methane will thus be higher (closer to ideal).
- Isobutane is larger and will thus have a larger correction term for the size of the molecule, b. This makes the term V - nb smaller. The pressure or volume must rise to compensate. Because the two gases are in the same size container, isobutane must exert a higher pressure.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. A
Gases deviate from ideal behavior at higher pressures and lower volumes and temperatures, all of which force molecules closer together. The closer they are, the more they can participate in intermolecular forces, which violates the definition of an ideal gas. At low temperatures, the kinetic energy of the particles is reduced, so collisions with other particles or the walls of the container are more likely to result in significant changes in kinetic energy.
2. D
Density equals mass divided by volume. The mass of 1 mole of neon gas equals 20.2 grams. At STP, 1 mole of neon occupies 22.4 L.
density = ρ = mass volume = 20 . 2 g 22 . 4 L ≈ 10 11 < 1 g L ( actual = 0 . 902 g L )
3. D
Graham’s law of effusion states that the relative rates of effusion of two gases at the same temperature and pressure are given by the inverse ratio of the square roots of the masses of the gas particles. In other words, a gas with a higher molar mass will leak more slowly than a gas with a lower molar mass. Both neon and oxygen gases will leak at slower rates than helium because they both have more mass than helium.
4. B
The pressure of the gas is calculated by subtracting the vapor pressure of water from the measured pressure during the experiment: 784 mmHg – 24 mmHg = 760 mmHg, or 1 atm. This is because the reaction is carried out in an aqueous environment; the water present will contribute to the partial pressures of the gas over the liquid. The ideal gas law can be used to calculate the moles of hydrogen gas. The volume of the gas is 0.100 L, the temperature is 298 K, and R = 0.0821 L · atm mol · K . Plugging in gives:
n = P V R T = ( 1 atm)(0 .1 L) ( 0.0821 L · atm mol · K ) ( 298 K) ≈ 1 0.8 × 300 = 1 240 ≈ 1 250 = 0.004
(A) incorrectly substitutes 8.314 into the gas law, rather than 0.0821. Remember that the value of R depends on the other variables in the equation; using 1 atm in the numerator necessitates using 0.0821. (C) incorrectly substitutes the wrong R and keeps the pressure in mmHg. (D) also keeps the pressure in mmHg.
5. B
Ideal gases are said to have no attractive forces between molecules. While each particle within the gas is considered to have negligible volume, ideal gases as a whole certainly do have a measurable volume, thus option I is eliminated. Gases have molar masses, thus option III is eliminated.
6. B
Deviations from ideal behavior are due either to large particle size and/or strong intermolecular forces between molecules. Nitrogen gas consists of two nitrogen atoms triple bonded. This molecule is therefore relatively small and its nonpolar triple bond leads to very weak intermolecular forces. Together, these observations support (B) as the correct answer. By contrast, fluoromethane consists of all single bonds, making this molecule somewhat larger. Furthermore, due to the polarity of the carbon-fluorine bond, this molecule can participate in dipole-dipole interactions. Fluoromethane would therefore exhibit greater deviations from ideal behavior.
7. A
The average kinetic energy is directly proportional to the temperature of a gas in kelvin. The kinetic molecular theory states that collisions between molecules are elastic and thus do not result in a loss of energy, eliminating (B). Gas particles are assumed to take up negligible space in kinetic molecular theory, eliminating (C). While the average kinetic energy of any gas as a whole is the same at a given temperature, the particles themselves have a distribution of speeds (as seen in the Maxwell–Boltzmann distribution curve), eliminating (D).
8. C
At STP, the difference between the distribution of speeds for helium and bromine gas is due to the difference in molar mass. Helium has a smaller molar mass than bromine. Particles with small masses travel faster than those with large masses, so the helium gas corresponds to curve B, which has a higher average speed. Because the gases are at the same temperature (273 K), they have the same average kinetic energy, eliminating (B) and (D).
9. A
The solubility of gases in liquids is directly proportional to the atmospheric pressure, as shown by Henry’s law. O 2 1 P 1 = O 2 2 P 2 1 . 25 × 10 - 3 M 1 . 000 atm = O 2 2 0 . 8000 atm O 2 2 = 0 . 8 × 1 . 25 × 10 - 3 1 = 8 10 × 5 4 × 10 - 3 = 1 × 10 - 3 M
Note that the use of fractions allows this problem to be simplified more readily than it would be with decimals.
10. C
The ideal gas law can be modified to include density (ρ) because the number of moles of gas, n, is equal to the mass divided by the molar mass. Thus, P V = n R T = m M R T → P = m V × R T M = ρ R T M . Isolating for temperature gives:
T = P M ρ R T = ( 1.3 × 10 9 atm) ( 2 .00 g mol ) ( 1.2 g cm 3 ) ( 1000 cm 3 L= ) ( 0.0821 L · atm mol · K ) ≈ ( 1.3 × 2 × 10 9 ) ( 1 ) ( 1000 ) ( 0 . 1 ) ≈ 2 . 6 × 10 9 100 ≈ 2 . 6 × 10 7 K
11. B
Gases are easily compressible because they travel freely with large amounts of space between molecules. Because gas particles are far apart from each other and in rapid motion, they tend to take up the volume of their container. Many gases exist as diatomic molecules, but this is not a property that characterizes all gases, eliminating option III.
12. B
We will use Charles’s law. First, we must convert the temperature to kelvin by adding 273 to get 300 K as the initial temperature. Think of this as a proportionality: If the volume is multiplied by 3 2 , the temperature will also have to be multiplied by 3 2 . Thus the final temperature is 450 K, which represents a 150 K increase (which is equivalent to an increase of 150 °C).
13. D
A decrease in temperature leads to a decrease in the kinetic energy of the particles so intermolecular interactions become more significant. This change leads to deviations from ideal behavior. Similarly, decreases in volume cause the volume of the particles themselves to become significant. This change also leads to deviations from ideal behavior. Thus, (D) is correct.
14. C
Both a change in temperature and a change in volume can affect a gas’s pressure. So if one of those two variables is kept constant, as in (A) and (B), we’ll definitely be able to predict which way the pressure will change. At a constant volume, heating the gas will increase its pressure, and cooling the gas will decrease it. What about when both temperature and volume are changing? If both changes have the same effect on pressure, then we can still predict which way it will change. This is the case in (D). Cooling the gas and increasing its volume both decrease pressure. (C), on the other hand, presents too vague a scenario for us to predict definitively the change in pressure. Heating the gas would amplify the pressure, while increasing the volume would decrease it. Without knowing the magnitude of each influence, it’s impossible to say whether the pressure would increase, decrease, or stay the same.
15. C
Initially the concentration of the gas is decreased to one-half its original value. Recall that concentration (solubility) and partial pressure are directly related—as one increases, the other increases. If the experimenters then quadruple the partial pressure of oxygen in the vessel, the solubility is also increased by a factor of four. One-half times four gives twice the original concentration value. Misreading the answer choices as being related to the concentration before the experimenters increased the partial pressure leads to (D).
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(8.1) Ideal gas law: PV = nRT
(8.2) Density of a gas: ρ = m V = P M R T
(8.3) Combined gas law: P 1 V 1 T 1 = P 2 V 2 T 2
(8.4) Avogadro’s principle: n V = k or n 1 V 1 = n 2 V 2
(8.5) Boyle’s law: PV = k or P1V1 = P2V2
(8.6) Charles’s law: V T = k or V 1 T 1 = V 2 T 2
(8.7) Gay-Lussac’s law: P T = k or P 1 T 1 = P 2 T 2
(8.8) Dalton’s law (total pressure from partial pressures):PT = PA + PB + PC + …
(8.9) Dalton’s law (partial pressure from total pressure): PA = XAPT
(8.10) Henry’s law: [A] = kH × PA or [ A ] 1 P 1 = [ A ] 2 P 2 = k H
(8.11) Average kinetic energy of a gas: K E = 1 2 m v 2 = 3 2 k B T
(8.12) Root-mean-square speed: u rms = 3 R T M
(8.13) Graham’s law: r 1 r 2 = M 2 M 1
(8.14) van der Waals equation of state: ( P + n 2 a V 2 ) ( V − n b ) = n R T
SHARED CONCEPTS
Biology Chapter 6
The Respiratory System
General Chemistry Chapter 3
Bonding and Chemical Interactions
General Chemistry Chapter 6
Equilibrium
Physics and Math Chapter 2
Work and Energy
Physics and Math Chapter 3
Thermodynamics
Physics and Math Chapter 4
Fluids