Chapter 9: Solutions

Chapter 9: Solutions
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
An aqueous solution was prepared by mixing 70 g of an unknown nondissociating solute into 100 g of water. The solution has a boiling point of 101.0 °C. What is the molar mass of the solute? (Note: K b = 0.512 K · kg mol )
- 358 . 4 g mol
- 32. 3 g mol
- 123.2 g mol
- 233. 6 g mol
-
Which phases of solvent and solute can form a solution?
- Solid solvent, gaseous solute
- Solid solvent, solid solute
- Gaseous solvent, gaseous solute
- I and II only
- I and III only
- II and III only
- I, II, and III
-
Two organic liquids, pictured in the figure below, are combined to form a solution. Based on their structures, will the solution closely obey Raoult’s law?

- Yes; the liquids differ due to the additional methyl group on toluene and, therefore, will not deviate from Raoult’s law.
- Yes; the liquids are very similar and, therefore, will not deviate from Raoult’s law.
- No; the liquids differ due to the additional methyl group on toluene and, therefore, will deviate from Raoult’s law.
- No; the liquids both contain benzene rings, which will interact with each other and cause deviation from Raoult’s law.
-
Which of the following explanations best describes the mechanism by which solute particles affect the melting point of ice?
- Melting point is elevated because the kinetic energy of the substance increases.
- Melting point is elevated because the kinetic energy of the substance decreases.
- Melting point is depressed because solute particles interfere with lattice formation.
- Melting point is depressed because solute particles enhance lattice formation.
-
The process of formation of a salt solution can be better understood by breaking the process into three steps:
- Breaking the solute into its individual components
- Making room for the solute in the solvent by overcoming intermolecular forces in the solvent
- Allowing solute–solvent interactions to occur to form the solution
Which of the following correctly lists the enthalpy changes for these three steps, respectively?
- Endothermic, exothermic, endothermic
- Exothermic, endothermic, endothermic
- Exothermic, exothermic, endothermic
- Endothermic, endothermic, exothermic
-
The entropy change when a solution forms can be expressed by the term ΔS°soln. When water molecules become ordered around an ion as it dissolves, the ordering would be expected to make a negative contribution to ΔS°soln. An ion that has more charge density will have a greater hydration effect, or ordering of water molecules. In an experiment, an equimolar amount of each of the following aqueous soluble compounds is dissolved in its own test tube containing 100 mL of water. Which will have the most negative contribution to ΔS°soln? (Assume that the charge density of each ion is equal to its actual charge.)
- KCl
- CsI
- BeF2
- NaCl
-
When ammonia, NH3, is used as a solvent, it can form complex ions. For example, dissolving AgCl in NH3 will result in the complex ion [Ag(NH3)]2+. What effect would the formation of complex ions have on the solubility of a compound like AgCl in NH3?
- The solubility of AgCl will increase because complex ion formation will cause more ions to exist in solution, which interact with AgCl to cause it to dissociate.
- The solubility of AgCl will increase because complex ion formation will consume Ag+ ions and cause the equilibrium to shift away from solid AgCl.
- The solubility of AgCl will decrease because Ag+ ions are in complexes, and the Ag+ ions that are not complexed will associate with Cl− to form solid AgCl.
- The solubility of AgCl will decrease because complex ion formation will consume Ag+ ions and cause the equilibrium to shift toward the solid AgCl.
-
One hundred grams of sucrose are dissolved in a cup of hot water at 80 °C. The cup of water contains 300.00 mL of water. What is the percent composition by mass of sugar in the resulting solution? (Note: Sucrose = C12H22O11, density of water at 80 ° C = 0.975 g mL )
- 25.0%
- 25.5%
- 33.3%
- 34.2%
-
Which of the following combinations of liquids would be expected to have a vapor pressure higher than the vapor pressure that would be predicted by Raoult’s law?
- Ethanol and hexane
- Acetone and water
- Isopropanol and methanol
- Nitric acid and water
-
The salt KCl is dissolved in a beaker. To an observer holding the beaker, the solution begins to feel colder as the KCl dissolves. From this observation, one could conclude that:
- ΔS°soln is large enough to overcome the unfavorable ΔH°soln.
- KCl is mostly insoluble in water.
- ΔS°soln must be negative when KCl dissolves.
- boiling point depression will occur in this solution.
-
Which of the following will cause the greatest increase in the boiling point of water when it is dissolved in 1.00 kg H2O?
- 0.4 mol calcium sulfate
- 0.5 mol iron(III) nitrate
- 1.0 mol acetic acid
- 1.0 mol sucrose
-
Reverse osmosis is a process that allows fresh water to be obtained by using pressure to force an impure water source through a semi-permeable membrane that only allows water molecules to pass. What is the minimum pressure that would be required to purify seawater at 25 °C that has a total osmolarity of 1,000 mOsm/L?
- 23.5 atm
- 24.5 atm
- 24,000 atm
- 24,500 atm
-
Lead is a toxic element that can cause many symptoms, including intellectual disabilities in children. If a body of water is polluted with lead ions at 200 ppb (parts per billion), what is the concentration of lead expressed as molarity? (Note: The density of water is 1 g mL , and ppb = grams per 109 grams of solution)
- 9.7 × 10−10 M Pb2+
- 9.7 × 10−7 M Pb2+
- 6.2 × 10−7 M Pb2+
- 6.2 × 10−6 M Pb2+
-
A saturated solution of aqueous cobalt(III) hydroxide (Ksp = 1.6 × 10−44) is added to a saturated solution of aqueous thallium(III) hydroxide (Ksp = 6.3 × 10−46). What is likely to occur?
- Both cobalt(III) hydroxide and thallium(III) hydroxide remain stable in solution.
- Thallium(III) hydroxide precipitates and cobalt(III) hydroxide remains stable in solution.
- Cobalt(III) hydroxide precipitates and thallium(III) hydroxide remains stable in solution.
- Both thallium(III) hydroxide and cobalt(III) hydroxide precipitate.
-
The following equilibrium exists when AgBr (Ksp = 5.35 × 10−13) is in solution:
AgBr (s) ⇌ Ag+ (aq) + Br− (aq)
What is the solubility of AgBr in a solution of 0.0010 M NaBr?
- 5 . 35 × 10 − 13 g L
- 1. 04 × 10 − 12 g L
- 5 . 35 × 10 − 10 g L
- 1. 04 × 10 − 7 g L
Answer Key
- A
- D
- B
- C
- D
- C
- B
- B
- A
- A
- B
- B
- B
- A
- D
Chapter 9: Solutions
CHAPTER 9
SOLUTIONS
In This Chapter
9.1 Nature of Solutions
Solvation
Solubility
Aqueous Solutions
Complex Ion Formation
9.2 Concentration
Units of Concentration
Dilution
9.3 Solution Equilibria
Solubility Product Constants
Common Ion Effect
9.4 Colligative Properties
Raoult’s Law
Boiling Point Elevation
Freezing Point Depression
Osmotic Pressure
Concept Summary
CHAPTER PROFILE
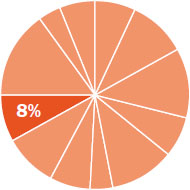
The content in this chapter should be relevant to about 8% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
2A: Assemblies of molecules, cells, and groups of cells within single cellular and multicellular organisms
5A: Unique nature of water and its solutions
Introduction
What do first aid instant cold packs and sweet tea have in common? Not much, you might think—but both, in fact, demonstrate the same principles of solution chemistry. Instant cold packs contain two compartments, one holding water and the other ammonium nitrate. When the barrier between the two compartments is broken, it allows the ammonium nitrate to dissolve into the water. Sweet tea is made by dissolving a large amount of sugar into strongly brewed tea.
The creation of both the ammonium nitrate and sugar solutions is an endothermic process. However, the formation of the ammonium nitrate solution is much more endothermic than the formation of the sugar solution. This is why ammonium nitrate is useful in instant cold packs. When it dissolves in water, the system must absorb an amount of energy equal to 6.14 kcal mol of ammonium nitrate. The heat is absorbed from the surrounding environment, so the pack feels cool to the touch.
Although the dissolution of sugar into water is not as strongly endothermic, we nevertheless have an intuitive understanding that the process is endothermic because we all know that the easiest way to dissolve lots of sugar into water (such as in tea or coffee) is to heat up the water and then add the sugar. Because heating the water increases the solubility of sugar, it must be that the dissolution of sugar into water is an endothermic process—think of Le Châtelier’s principle and changes in temperature from Chapter 6.
In this chapter, our focus will be on the characteristics and behaviors of solutions, the nature of solutions, the formation of aqueous solutions, the measurements of solution concentration, and finally the qualitative and quantitative evaluation of solution equilibria.
9.1 Nature of Solutions
LEARNING OBJECTIVES
After Chapter 9.1, you will be able to:
- Describe the process of solvation
- Define key terms involved in solutions, such as complex, solubility, and saturation
- Explain how to increase solubility of a compound
- Recall the solubility rules and apply them to predict solubility of a compound
Many important chemical reactions, both in the laboratory and in nature, take place in solutions, including almost all reactions in living organisms. Solutions are homogeneous (the same throughout) mixtures of two or more substances that combine to form a single phase, usually the liquid phase. The MCAT will focus almost exclusively on solids dissolved into aqueous solutions, but it’s important to remember that solutions can be formed from different combinations of the three phases of matter. For example, gases can be dissolved in liquids (carbonating soda); liquids can be dissolved in other liquids (ethanol in water); solids can even be dissolved in other solids (metal alloys). Incidentally, gases “dissolved” into other gases can be thought of as solutions, but are more properly defined only as mixtures because gas molecules do not interact all that much chemically, as described by the kinetic molecular theory of gases. As a point of clarification: all solutions are considered mixtures, but not all mixtures are considered solutions.
A solution consists of a solute (such as NaCl, NH3, C6H12O6, or CO2) dissolved (dispersed) in a solvent (such as H2O, benzene, or ethanol). The solvent is the component of the solution that remains in the same phase after mixing. If the two substances are already in the same phase (for example, a solution of two liquids), the solvent is the component present in greater quantity. If the two same-phase components are in equal proportions in the solution, then the component that is more commonly used as a solvent in other contexts is considered the solvent. Solute molecules move about freely in the solvent and interact with it by way of intermolecular forces such as ion–dipole, dipole–dipole, or hydrogen bonding. Dissolved solute molecules are also relatively free to interact with other dissolved molecules of different chemical identities; consequently, chemical reactions occur easily in solution.
Solvation
Solvation is the electrostatic interaction between solute and solvent molecules. This is also known as dissolution, and when water is the solvent, it can be called hydration. Solvation involves breaking intermolecular interactions between solute molecules and between solvent molecules and forming new intermolecular interactions between solute and solvent molecules together, as shown in Figure 9.1 (which was also shown in Chapter 4 of MCAT General Chemistry Review in the context of ions).

Figure 9.1. Solvation of a Polar Covalent Compound Sindicates a solvent particle.**
When the new interactions are stronger than the original ones, solvation is exothermic, and the process is favored at low temperatures. The dissolution of gases into liquids, such as CO2 into water, is an exothermic process because the only significant interactions that must be broken are those between water molecules—CO2, as a gas, demonstrates minimal intermolecular interaction. Le Châtelier’s principle tells us this is the reason that lowering the temperature of a liquid favors solubility of a gas in the liquid.
When the new interactions are weaker than the original ones, solvation is endothermic and the process is favored at high temperatures. Most dissolutions are of this type. Two such examples have already been given: dissolving ammonium nitrate or sugar into water. Because the new interactions between the solute and solvent are weaker than the original interactions between the solute molecules and between the solvent molecules, energy (heat) must be supplied to facilitate the formation of these weaker, less stable interactions. Sometimes the overall strength of the new interactions is approximately equal to the overall strength of the original interactions. In this case, the overall enthalpy change for the dissolution is close to zero. These types of solutions approximate the formation of an ideal solution, for which the enthalpy of dissolution is equal to zero.
The spontaneity of dissolution is dependent not only on the enthalpy change; solutions may form spontaneously for both endothermic and exothermic dissolutions. The second property that contributes to the spontaneity of dissolution is the entropy change that occurs in the process. At constant temperature and pressure, entropy always increases upon dissolution. As with any process, the spontaneity of dissolution depends on the change in Gibbs free energy: spontaneous processes are associated with a decrease in free energy, while nonspontaneous processes are associated with an increase in free energy. Thus, whether or not dissolution will happen spontaneously depends on both the change in enthalpy and the change in entropy for the solute and solvent of the system.
BRIDGE
Proteins dissolve in solution with their most hydrophilic amino acids on the outside and hydrophobic amino acids on the inside because this maximizes the increase in entropy during dissolution. As described in Chapter 1 of MCAT Biochemistry Review, a protein dissolves by forming a solvation layer.
Consider, for example, the formation of another common solution: sodium chloride dissolved in water. When NaCl dissolves in water, its component ions dissociate from each other and become surrounded by water molecules. For this new interaction to occur, ionic bonds between Na+ and Cl− must be broken, and hydrogen bonds between water molecules must also be broken. This step requires energy and is therefore endothermic. Because water is polar, it can interact with each of the component ions through ion–dipole interactions: the partially positive hydrogen end of the water molecules will surround the Cl− ions, and the partially negative oxygen end of the water molecules will surround the Na+ ions, as shown in Figure 9.2. The formation of these ion–dipole bonds is exothermic, but the magnitude is slightly less than the energy required to break the ionic bonds and hydrogen bonds. As a result, the overall dissolution of table salt into water is endothermic ( + 3.87 kJ mol ) and favored at high temperatures.
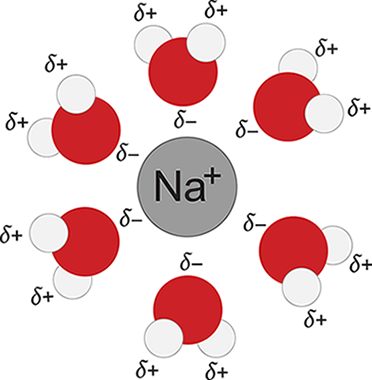
Figure 9.2. Solvation of Na+ Ions in Aqueous Solutions
We’ve considered the enthalpy change for the formation of a sodium chloride solution, and now we need to examine the entropy change. Remember that entropy can be thought of as the degree to which energy is dispersed throughout a system or the amount of energy distributed from the system to the surroundings at a given temperature. Another way to understand entropy is the measure of molecular disorder, or the number of energy microstates available to a system at a given temperature. When solid sodium chloride dissolves into water, the rigidly ordered arrangement of the sodium and chloride ions is broken up as the ion–ion interactions are disrupted and new ion–dipole interactions with the water molecules are formed. The ions, freed from their lattice arrangement, have a greater number of energy microstates available to them (in simpler terms, they are freer to move around in different ways), and consequently, their energy is more distributed and their entropy increases. The water, however, becomes more restricted in its movement because it is now interacting with the ions. The number of energy microstates available to it (that is, the water molecules’ ability to move around in different ways) is reduced, so the entropy of the water decreases. In the end, the increase in the entropy experienced by the dissolved sodium chloride is greater than the decrease in the entropy experienced by the water, so the overall entropy change is positive—energy is, overall, dispersed by the dissolution of sodium chloride in water. Because of the relatively low endothermicity and relatively large positive change in entropy, sodium chloride will spontaneously dissolve in liquid water (ΔG = ΔH – TΔS).
Solubility
We often want to know more than just whether or not dissolution of a solute into a solvent will be spontaneous or nonspontaneous—we also want to know how much solute will dissolve into a given solvent. Thesolubility of a substance is the maximum amount of that substance that can be dissolved in a particular solvent at a given temperature. When this maximum amount of solute has been added, the dissolved solute is in equilibrium with its undissolved state, and we say that the solution is saturated. If more solute is added, it will not dissolve. For example, at 25 °C, a maximum of 90.9 g glucose will dissolve in 100 mL H2O. Thus, the solubility of glucose is 909 g L . If more glucose is added to an already saturated glucose solution, it will not dissolve but rather will remain in solid form,precipitating to the bottom of the container. A solution in which the proportion of solute to solvent is small is said to be dilute, and one in which the proportion is large is said to be concentrated. Note that both dilute and concentrated solutions are still considered unsaturated if the maximum equilibrium concentration (saturation) has not yet been reached.
The solubility of substances in different solvents is ultimately a function of thermodynamics. When the change in Gibbs free energy for the dissolution reaction is negative at a given temperature, the process will be spontaneous, and the solute is said to be soluble. When the change in Gibbs free energy is positive, the process will be nonspontaneous, and the solute is said to be insoluble. Some solute–solvent systems have negative changes in free energy with very large magnitudes, so the equilibrium reaction strongly favors the dissolution of the solute. In general, solutes are considered soluble if they have a molar solubility above 0.1 M in solution. Others have only slightly negative changes in free energy, so the equilibrium position lies closer to the undissociated (reactants) side of the reaction. Those solutes that dissolve minimally in the solvent (molar solubility under 0.1 M) are called sparingly soluble salts.
Aqueous Solutions
The most common type of solution is the aqueous solution, in which the solvent is water. The aqueous state is denoted by the symbol (aq). Aqueous solutions rely on the interactions between water molecules and solutes in solutions. We have mentioned previously that hydration is often the process through which dissolution occurs. It is also important to note that in some solutions, such as acids, the formation of a complex called the hydronium ion (H3O+) can occur. This is facilitated by the transfer of a hydrogen ion (H+) from a molecule in solution to a water molecule (H2O). The reaction of acetic acid (H+ donor) with water is shown in Figure 9.3.
CH3COOH (aq) + H2O (l) ⇌ CH3COO− (aq) + H3O+ (aq)
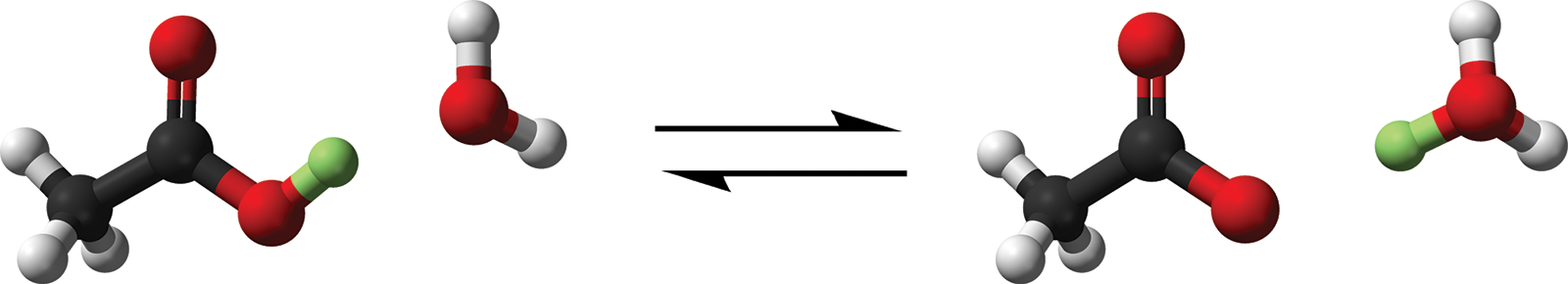
Figure 9.3. Transfer of a Proton in Solution, Forming the Hydronium Ion The transferred proton is highlighted in green.
It is important to realize that H+ is never found alone in solution because a free proton is difficult to isolate; rather, it is found bonded to an electron pair donor (carrier) molecule such as a water molecule. This is an example of a coordinate covalent bond. The hydronium ion and its effects on the solubilities of other compounds using Le Châtelier’s principle will be described further in Chapter 10 of MCAT General Chemistry Review.
Because aqueous solutions are so common and so important to biological systems, the MCAT focuses on them above all others. In aqueous solutions, there are seven general solubility rules:
- All salts containing ammonium (NH4+) and alkali metal (Group 1) cations are water-soluble.
- All salts containing nitrate (NO3−) and acetate (CH3COO−) anions are water-soluble.
- Halides (Cl−, Br−, I−), excluding fluorides, are water-soluble, with the exceptions of those formed with Ag+, Pb2+, and Hg22+.
- All salts of the sulfate ion (SO42−) are water-soluble, with the exceptions of those formed with Ca2+, Sr2+, Ba2+, and Pb2+.
- All metal oxides are insoluble, with the exception of those formed with the alkali metals, ammonium, and CaO, SrO, and BaO, all of which hydrolyze to form solutions of the corresponding metal hydroxides.
- All hydroxides are insoluble, with the exception of those formed with the alkali metals, ammonium, and Ca2+, Sr2+, and Ba2+.
- All carbonates (CO32−), phosphates (PO43−), sulfides (S2–), and sulfites (SO32−) are insoluble, with the exception of those formed with the alkali metals and ammonium.
MCAT EXPERTISE
Because most solutions in the real world involve water as the solvent, it is not a surprise that solutions are common on the MCAT. These solubility rules are not bad to know, but memorizing them all may be a little excessive. It is never a bad thing to know facts, but being able to apply them is more important. Know rules 1 and 2 for sure, and be aware of some of the more common insoluble exceptions, like Pb2+ and Ag+.
The MCAT will not expect memorization of all of the solubility rules, but it is worth knowing two absolutes: all salts of Group 1 metals, and all nitrate salts are soluble. Otherwise, familiaritywith rules listed above will suffice—the MCAT generally supplies solubility information for most compounds. Sodium and nitrate ions are generally used as counterions to what is actually chemically important; for example, if a pH problem gives a sodium formate concentration of 0.10 M, it is really indicating that the concentration of the formate ion is 0.10 M because the sodium ion concentration does not affect pH. The only time one needs to worry about the nitrate ion concentration is in an oxidation–reduction reaction, for the nitrate ion can function—although only weakly—as an oxidizing agent. In all other cases with nitrate ions, only focus on the cation as the chemically reacting species.
Complex Ion Formation
We have mentioned the hydronium ion as a complex that forms in acidic solutions, but it is worthwhile to mention that there are even more varied forms of complex ions that can appear in solution. By definition, a complex ion—or coordination compound—refers to a molecule in which a cation is bonded to at least one electron pair donor (which could include the water molecule). The electron pair donor molecules are called ligands. An example of such a complexation reaction is shown for the tetraaquadioxouranyl cation, which has water (aqua–) and oxygen (oxo–) ligands, in Figure 9.4.
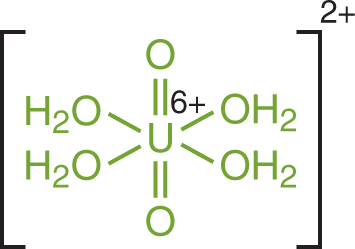
Figure 9.4. Structure of the Tetraaquadioxouranyl Complex Cation Water and oxygen act as ligands with a U6+ cation.
Complexes are held together with coordinate covalent bonds, in which an electron pair donor (a Lewis base) and an electron pair acceptor (a Lewis acid) form very stable Lewis acid–base adducts. Most general chemistry courses do not stress the biological importance of coordination compounds. However, complex ions have profound biological applications in macromolecules such as proteins. For instance, many active sites of proteins utilize complex ion binding and transition metal complexes to carry out their function. One classic example is the iron cation in hemoglobin, which can carry oxygen, carbon dioxide, and carbon monoxide as ligands, as shown in Figure 9.5.
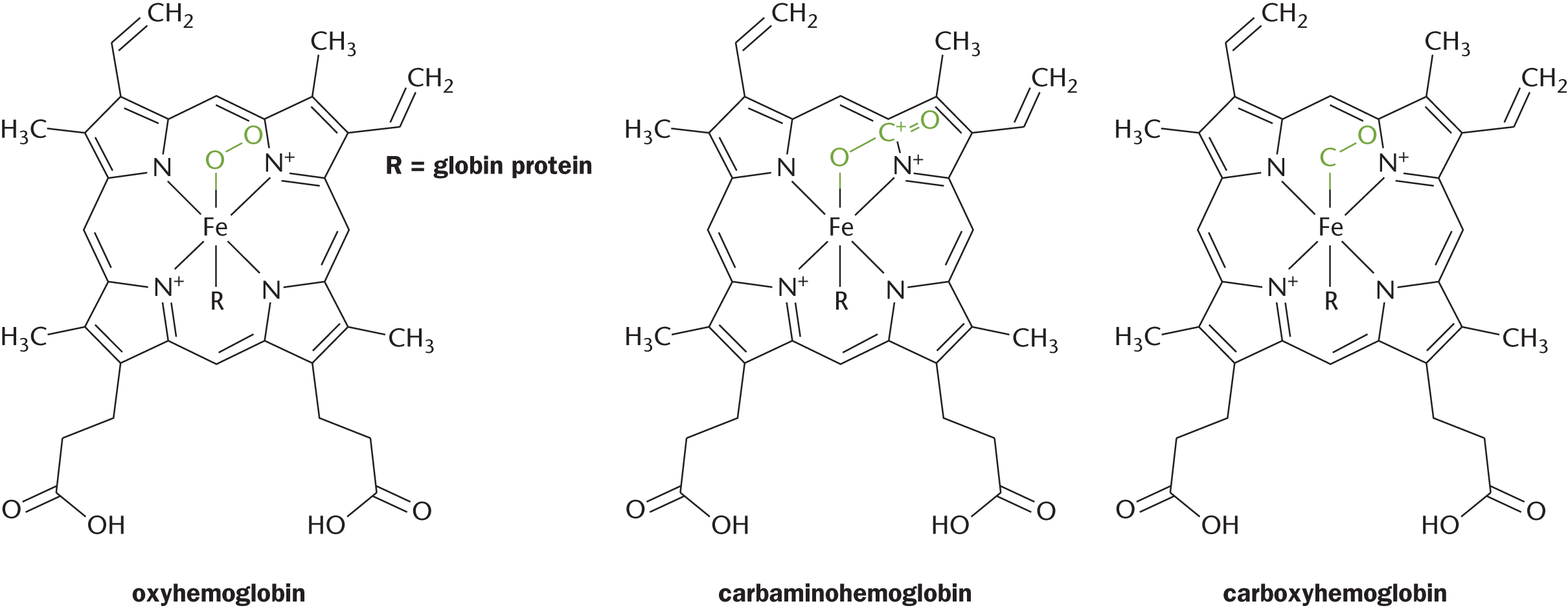
Figure 9.5. Hemoglobin Is a Classic Example of Biochemical Complex Formation The iron in hemoglobin can bind various gases, leading to the formation of oxyhemoglobin (O2), carbaminohemoglobin (CO2), and carboxyhemoglobin (CO).
Many coenzymes (vitamins) and cofactors also contain complexes of transition metals, such as cobalamin (vitamin B12), shown in Figure 9.6. The presence of a transition metal allows coenzymes and cofactors to bind other ligands or assist with electron transfer.
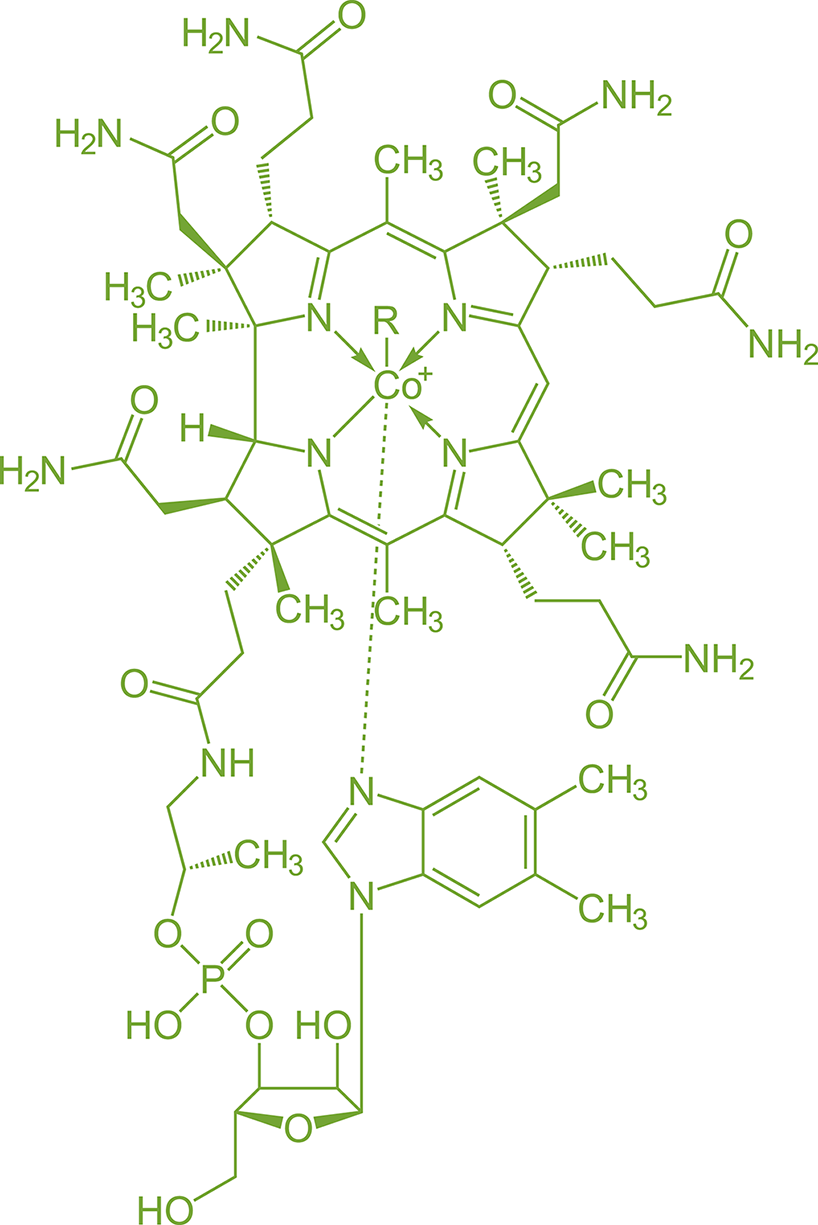
Figure 9.6. Cobalamin (Vitamin B12) Contains a Cobalt Complex
Physical and chemical properties of complex ions are diverse, including a wide range of solubilities and varied chemical reactions. Inorganic complex ions are often fun to characterize because they tend to have vibrant, distinctive colors, as illustrated in Figure 9.7.

Figure 9.7. Nickel(II) Ion Complexes Display Distinctive Colors The characteristic colors of Nickel (II) Ion complexes are, from left to right: 1) hexaamminenickel(II), 2) tris(ethylenediamine)nickel(II), 3) tetrachloronickelate(II), and 4) hexaaquanickel(II)
In some complexes, the central cation can be bonded to the same ligand in multiple places. This is called chelation, and it generally requires large organic ligands that can double back to form a second (or even third) bond with the central cation. Chelation therapy is often used to sequester toxic metals (lead, arsenic, mercury, and so on). Even biologically necessary metals, such as iron, can be toxic in overload states; an example of iron being chelated is shown in Figure 9.8.
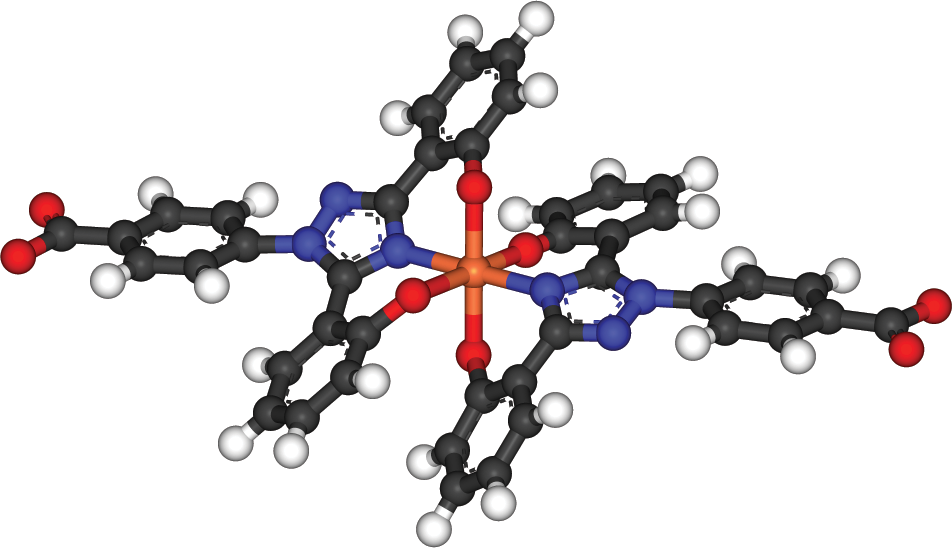
Figure 9.8. Chelation of Iron with Two Molecules of Deferasirox
MCAT CONCEPT CHECK 9.1:
Before you move on, assess your understanding of the material with these questions.
-
Describe the process of solvation.
_____________________________
-
Describe the differences between solubility and saturation:
- ____________________________
- ____________________________
-
What is one way in which solubility of a compound can be increased?
_____________________________
-
Name two ions that form salts that are always soluble:
- ____________________________
- ____________________________
9.2 Concentration
LEARNING OBJECTIVES
After Chapter 9.2, you will be able to:
- Calculate the molality, molarity, or normality of a compound in solution
- Apply MiVi = MfVf to calculate dilution of a solution
- Calculate mole fraction and percent composition by mass
Concentration denotes the amount of solute dissolved in a solvent. There are many different ways of expressing concentration, and different units have been standardized for specific everyday situations. For example, alcohol content in liquors like vodka, gin, or rum is expressed in volume percent (volume of solute divided by volume of solution times 100 percent). Alcoholic proof is twice the volume percent. The sugar content of orange juice and other fruit juices is measured in units of degrees Brix (°Bx), which is a mass percent: mass of glucose divided by mass of solution times 100 percent.
Units of Concentration
On the MCAT, concentrations are commonly expressed as percent composition by mass, mole fraction, molarity, molality, and normality.
Percent Composition by Mass
The percent composition by mass is given by the equation
mass of solute mass of solution × 100 %
Equation 9.1
MCAT EXPERTISE
It is important to have a good idea of how to work with all of these ways of expressing concentration because more than one may show up on Test Day.
Percent composition is used not only for aqueous solutions, but also for metal alloys and other solid-in-solid solutions.
Example: What is the percent composition by mass of a salt water solution if 100 g of the solution contains 20 g of NaCl?
Solution:
mass of solute mass of solution × 100 % = 20 g 100 g × 100 %
=20% NaCl solution
Mole Fraction
The mole fraction (X) of a compound is given by the equation
X A = moles of A total moles of all species
Equation 9.2
The sum of the mole fractions in a system will always equal 1. The mole fraction is used to calculate the vapor pressure depression of a solution, described later in this chapter, as well as the partial pressures of gases in a system, described in Chapter 8 of MCAT General Chemistry Review.
Example: If 184 g glycerol (C3H8O3) is mixed with 180 g water, what will be the mole fractions of the two components? (Note: Molar mass of H 2 O = 18 g mol ; molar mass of C 3 H 8 O 3 = 92 g mol )
Solution: First, determine the number of moles of each compound: 180 g water × [ 1 mol 18 g ] = 10 moles water 184 g glycerol × [ 1 mol 92 g ] = 2 moles glycerol Total number of moles = 10 + 2 = 12 moles
Then, determine the mole fractions:
X water = 10 mol water 12 mol total = 5 6 = 0.83 X glycerol = 2 mol glycerol 12 mol total = 1 6 = 0.17
Molarity
The molarity (M) of a solution is defined as
M = moles of solute liters of solution
Equation 9.3
Solution concentrations are usually expressed in terms of molarity, and this is the most common unit for concentration on the MCAT. Unless otherwise specified, representations of concentration using brackets—such as [Na+]—indicate molarity. Note that the volume term in the denominator of molarity refers to the solution volume, not the volume of solvent used to prepare the solution—although the two values are often close enough to approximate the solution volume using the solvent volume. Molarity is used for rate laws, the law of mass action, osmotic pressure, pH and pOH, and the Nernst equation.
MCAT EXPERTISE
Note that for dilute solutions, the volume of the solution is approximately equal to the volume of solvent used, which simplifies our calculations on Test Day. However, technical questions could ask you to distinguish between these two. For example, when you add two kilograms of sucrose (table sugar) to a liter of water at room temperature (achieving saturation), the volume of solution is certainly larger than 1 L!
Example: If enough water is added to 11 g of CaCl2 to make 100 mL of solution, what is the molarity of the solution?
Solution: First, calculate the number of moles of CaCl2:
11 g CaCl 2 [ 1 mol 111.1 g ] = 0.1 mol CaCl 2
Then determine the molarity:
M = moles of solute liters of solution M = 0.1 mol 100 mL × 1 L 1000 mL = 0 . 1 0 . 1 = 1 M
Molality
The molality (m) of a solution is defined as
m = moles of solute kilograms of solvent
Equation 9.4
For dilute aqueous solutions at 25 °C, the molality is approximately equal to molarity because the density of water at this temperature is 1 kilogram per liter. However, note that this is an approximation and true only for dilute aqueous solutions. As aqueous solutions become more concentrated with solute, their densities become significantly different from that of pure water; most water-soluble solutes have molar masses significantly greater than that of water, so the density of the solution increases as the concentration increases. You won’t use molality very often, so be mindful of the special situations when it is required: boiling point elevation and freezing point depression.
Example: If 10 g NaOH are dissolved in 500 g of water, what is the molality of the solution?
Solution: First, calculate the number of moles of NaOH:
10 g NaOH [ 1 mol 40 g ] = 0.25 mol NaOH
Then determine the molality:
m = moles of solute kilograms of solvent m = 0.25 mol 500 g × 1 kg 1000 g = 0 . 25 0 . 5 = 0.5 m
Normality
We discussed the related concepts of gram equivalent weight, equivalents, and normality in Chapter 4 of MCAT General Chemistry Review. The normality (N) of a solution is equal to the number of equivalents of interest per liter of solution. An equivalent is a measure of the reactive capacity of a molecule. Most simply, an equivalent is equal to a mole of the species of interest—protons, hydroxide ions, electrons, or ions.
To calculate the normality of a solution, we need to know what purpose the solution serves because it is the concentration of the reactive species with which we are concerned. For example, in acid–base reactions, we are most concerned with the concentration of hydrogen ions; in oxidation–reduction reactions, we are most concerned with the concentration of electrons. Normality is unique among concentration units in that it is reaction dependent. For example, in acidic solution, 1 mole of the permanganate ion (MnO4−) will readily accept 5 moles of electrons, so a 1 M solution would be 5 N. However, in alkaline solution, 1 mole of permanganate will accept only 1 mole of electrons, so in alkaline solution, a 1 M permanganate solution would be 1 N.
MCAT EXPERTISE
Simple ideas on Test Day will make things easier. So, when you come across normality, think of it as molarity of the stuff of interest in the reaction.
Dilution
A solution is diluted when solvent is added to a solution of higher concentration to produce a solution of lower concentration. The concentration of a solution after dilution can be determined using the equation
MiVi = MfVf
Equation 9.5
where M is molarity, V is volume, and the subscripts i and f refer to the initial and final values, respectively.
MCAT EXPERTISE
This equation is worthy of memorization. Note that a similar equation is used for the equivalence point in acid–base chemistry, as discussed in Chapter 10 of MCAT General Chemistry Review.
MCAT EXPERTISE
Though not a unique means of measuring concentration or dilution, the term “parts-per” can be used to indicate concentration of a dissolved substance in a solution (most commonly water). Parts-per-million (ppm, 10–6) is the most common usage. If a problem states there is one ppm of substance X in water, that would indicate there is 1 mg/L of water, as there would be 1 millionth of a gram per gram of water, and the density of water is 1 g/mL. On Test Day, prior to converting from ppm, make sure to assess whether conversion to a different unit of measure is actually required, as this conversion can typically be avoided on the MCAT.
Example: A chemist wishes to prepare 300 mL of a 1.1 M NaOH solution from a 5.5 M NaOH stock solution. What volume of stock solution should be diluted with pure water to obtain the desired solution?
Solution: M i V i = M f V f → V i = M f V f M i V i = ( 1. 1 M ) ( 300 mL) ( 5.5 M ) = 300 5 = 60 mL
Note that one can use mL or L in the equation, as long as the units are consistent.
MCAT CONCEPT CHECK 9.2:
Before you move on, assess your understanding of the material with these questions.
-
If you mix 180 g of the following compounds in 250 L of water ( density = 1 kg L ) , what are their concentrations in molality, molarity, and normality (for acid–base chemistry)?
Compound Molality Molarity Normality Glucose ( 180 g mol )
Carbonate ( 60 g mol )
-
You are working in a sewage treatment facility and are assaying chlorine in a water sample. You need to dilute the water sample from 100 ppm stock to 25 ppm and create 100 mL of solution. Calculate the amount of stock solution needed and determine how you would create your final solution:
__________________________________
__________________________________
__________________________________
-
A stock solution for making typical IV saline bags contains 90.0 g of NaCl per 10 liters of water ( density = 1 g mL ) . What is the mole fraction and the percent composition by mass of NaCl in the saline solutions?
- Mole fraction: ______________________
- Percent composition by mass: ____________________________________
9.3 Solution Equilibria
LEARNING OBJECTIVES
After Chapter 9.3, you will be able to:
- Calculate molar solubility for a compound given its Ksp and vice versa
- Calculate ion product for a solution
- Given Ksp, predict when a solute may dissociate or precipitate in order to reach equilibrium
- Predict the impact of a common ion on the dissolution of a compound
The process of solvation, like other reversible chemical and physical processes, tends toward an equilibrium position, defined as the lowest energy state of a system under a given set of temperature and pressure conditions. Systems move spontaneously toward the equilibrium position, and any movement away from equilibrium is nonspontaneous. In the process of creating a solution, the equilibrium is defined as the saturation point, where the solute concentration is at its maximum value for the given temperature and pressure. Immediately after solute has been introduced into a solvent, most of the change taking place is dissociation because no dissolved solute is initially present. However, once solute is dissolved, the reverse process—precipitation of the solute—will begin to occur.
When the solution is dilute (unsaturated), the thermodynamically favored process is dissolution, and initially, the rate of dissolution will be greater than the rate of precipitation. As the solution becomes more concentrated and approaches saturation, the rate of dissolution lessens, while the rate of precipitation increases. Eventually, the saturation point of the solution is reached. The solution now exists in a state of dynamic equilibrium for which the rates of dissolution and precipitation are equal, and the concentration of dissolved solute reaches a steady-state (constant) value. Neither dissolution nor precipitation is more thermodynamically favored at equilibrium because favoring either would necessarily result in the solution no longer being in a state of equilibrium. At this point, the change in free energy is zero, as is the case for all systems at equilibrium.
An ionic solid introduced into a polar solvent dissociates into its component ions, and the dissociation of such a solute in solution may be represented by
AmBn (s) ⇌ m An+ (aq) + n Bm− (aq)
On Test Day, the first step for any solution stoichiometry or solution equilibrium question is to write out the balanced dissociation reaction for the ionic compound in question. This first step is essential for correctly calculating the solubility product constant, ion product, molar solubility, or for determining the outcome of the common ion effect. In other words, it is the essential first step for nearly every solution chemistry problem on the MCAT.
Solubility Product Constants
Most solubility problems on the MCAT deal with solutions of sparingly soluble salts, which are ionic compounds that have very low solubility in aqueous solutions. You may wonder why any ionic compound would not be highly soluble in water. The degree of solubility is determined by the relative changes in enthalpy and entropy associated with the dissolution of the ionic solute at a given temperature and pressure. One common sparingly soluble salt is silver chloride, AgCl, which dissociates in water according to the following equation:
AgCl (s) ⇌ Ag+ (aq) + Cl− (aq)
The law of mass action can be applied to a solution at equilibrium; that is to say, when the solution is saturated and the solute concentration is at a maximum and is dynamically stable. For a saturated solution of an ionic compound with the formula AmBn, the equilibrium constant for its solubility in aqueous solution, called the solubility product constant (Ksp), can be expressed by:
Ksp = [An+]m[Bm–]n
Equation 9.6
where the concentrations of the ionic constituents are equilibrium (saturation) concentrations. For example, we can express the Ksp of silver chloride as:
Ksp = [Ag+][Cl−]
MCAT EXPERTISE
On the MCAT, if you remember that Ksp is just a specialized form of Keq, then you can simplify a lot of problems by using the same concepts that you do for all equilibria, including Le Châtelier’s principle.
You’ll notice that, for the law of mass action of solutions, there is no denominator. Remember that pure solids and liquids do not appear in the equilibrium constant. Because the silver chloride solution was formed by adding pure solid silver chloride to pure water, neither the solid silver chloride nor the water is included. Indeed, dissociation reactions—by definition—have a solid salt as a reactant; thus, Ksp expressions should never have denominators.
Solubility product constants, like all other equilibrium constants (Keq, Ka, Kb, and Kw) are temperature dependent. When the solution consists of a gas dissolved into a liquid, the value of the equilibrium constant, and hence the position of equilibrium (saturation), will also depend on pressure. Generally speaking, the solubility product constant increases with increasing temperature for non-gas solutes and decreases for gas solutes. Higher pressures favor dissolution of gas solutes, and therefore the Ksp will be larger for gases at higher pressures than at lower ones.
REAL WORLD
Because gases become more soluble in solution as pressure increases, divers who have spent time at significant depths under water will have more nitrogen gas dissolved in their blood because nitrogen gas is the main inert gas in the air we breathe. If divers rise to the surface too quickly, the abrupt decompression will lead to an abrupt decrease in gas solubility in the plasma, resulting in the formation of nitrogen gas bubbles in the bloodstream. The gas bubbles can get lodged in the small vasculature of the peripheral tissue, mostly around the large joints of the body, causing pain and tissue damage (hence the name, the bends). The condition is painful and dangerous and can be fatal if not properly treated.
As solute dissolves into the solvent, the system approaches saturation, at which point no more solute can be dissolved and any excess will precipitate to the bottom of the container. We may not know whether the solution has reached saturation, and so to determine where the system is with respect to the equilibrium position, we can calculate a value called the ion product (IP), which is analogous to the reaction quotient, *Q**,* for other chemical reactions. The ion product equation has the same form as the equation for the solubility product constant:
IP = [An+]m[Bm–]n
Equation 9.7
The difference is that the concentrations used in the ion product equation are the concentrations of the ionic constituents at that given moment in time, which may differ from equilibrium concentrations. As with the reaction quotient Q, the utility of the ion product lies in comparing its value to that attained at equilibrium, Ksp. Each salt has its own distinct Ksp at a given temperature. If, at a given set of conditions, a salt’s IP is less than the salt’s Ksp, then the solution is not yet at equilibrium and is considered unsaturated. For unsaturated solutions, dissolution is thermodynamically favored over precipitation. If the IP is greater than the Ksp, then the solution is beyond equilibrium, and the solution is considered supersaturated. It is possible to create a supersaturated solution by dissolving solute into a hot solvent and then slowly cooling the solution. A supersaturated solution is thermodynamically unstable, and any disturbance to the solution, such as the addition of more solid solute or other solid particles, or further cooling of the solution, will cause spontaneous precipitation of the excess dissolved solute. If the calculated IP is equal to the known Ksp, then the solution is at equilibrium—the rates of dissolution and precipitation are equal—and the solution is considered saturated. The molarity of a solute in a saturated solution is called the molar solubility of that substance.
KEY CONCEPT
- IP < Ksp: unsaturated, solute will continue to dissolve
- IP = Ksp: saturated, solution is at equilibrium
- IP > Ksp: supersaturated, precipitation will occur
Example: The molar solubility of Fe(OH)3 in an aqueous solution was determined to be 4 × 10 − 10 mol L . What is the value of the Ksp for Fe(OH)3 at the same temperature and pressure?
Solution: The molar solubility is given as 4 × 10−10 M. The equilibrium concentration of each ion can be determined from the molar solubility and the balanced dissociation reaction of Fe(OH)3. The dissociation reaction is:
Fe(OH)3 (s) ⇌ Fe3+ (aq) + 3 OH− (aq) Ksp = [Fe3+][OH−]3
The molar solubility can be expressed as x, the amount of Fe(OH)3 that dissolves to make a saturated solution at equilibrium. As it dissolves, Fe(OH)3 dissociates to create x of Fe3+ and 3x of OH−. This can be entered into the Ksp equation to give:
Ksp = [x][3x]3
If the molar solubility x is 4 × 10−10 M, then x = 4 × 10−10 M Fe(OH)3 has dissolved, yielding x = 4 × 10−10 M Fe3+ and 3x = 3 × 4 × 10−10 M OH− (because there are three OH− ions released per Fe(OH)3 molecule dissolved). Thus,
Ksp=[Fe3+][OH−]3Ksp=(4×10−10M)(3×4×10−10M)3=(4×10−10)×[33×(4×10−10)3]Ksp=33×(4×10−10)4=27(4×10−10)4=27×256×10−40≈30×250×10−40Ksp≈7500×10−40=7.5×10−37(actual=6.9×10−37)
Example: What are the concentrations of each of the ions in a saturated solution of CuBr, given that the Ksp of CuBr is 6.27 × 10−9 at 25 °C? If 3 g CuBr are dissolved in water to make 1 L of solution at 25 °C, would the solution be saturated, unsaturated, or supersaturated?
Solution: The first step is to write out the dissociation reaction:
CuBr (s) → Cu+ (aq) + Br− (aq) Ksp = [Cu+][Br−]
Let x equal the molar solubility of CuBr, which is the amount of CuBr that dissolves at equilibrium. The concentration of Cu+ and Br– will each equal x.
K sp = x · x = x 2 6 . 27 × 10 − 9 = x 2 6 . 3 × 10 − 9 ≈ x 2 63 × 10 − 10 ≈ x 2 ≈ 64 × 10 − 10 8 × 10 − 5 ≈ x actual value = 7 . 9 × 10 − 5
Therefore, [Cu+] is about 8 × 10−5 M, and [Br−] is also about 8 × 10−5 M. Note that 8 × 10−5 M also represents the molar solubility of copper(I) bromide.
Next, we convert 3 g of CuBr into moles:
3 g × 1 mol CuBr 143 . 5 g ≈ 3 150 ≈ 3 1 . 5 × 10 2 ≈ 2 × 10 − 2 mol
2 × 10−2 mol CuBr in 1 L of solution represents a molarity of 2 × 10−2 M, which is more than 100 times higher than the molar solubility of CuBr. Therefore, this is a supersaturated solution.
KEY CONCEPT
Every sparingly soluble salt of general formula MX will have Ksp = x2, wherex is the molar solubility (assuming no common ion effect).
KEY CONCEPT
Every sparingly soluble salt of general formula MX2 will have Ksp = 4x3, where x is the molar solubility (assuming no common ion effect).
KEY CONCEPT
Every sparingly soluble salt of general formula MX3 will have Ksp = 27x4, where x is the molar solubility (assuming no common ion effect).
Finally, let’s return to our discussion of complex ions and their solubility factors. Much like the examples we have seen previously, the solubility of complex ion solutions is determined by the Ksp. The formation of complex ions increases the solubility of a salt in solution.
For instance, consider free iron(III) (Fe3+) in a solution of water. If a cyanide solution were added, an exceptionally stable iron and cyanide metal complex would form as the water molecules solvating the iron are replaced by excess cyanide ions:
Fe 3 + ( a q ) + 6 CN − ( a q ) ⇌ [ Fe ( CN ) 6 ] 3 – ( a q ) K f = 1.0 × 10 31
Knowing the intricacies of why complexes are more stable in solution than isolated ions is beyond the scope of the MCAT; however, it should make sense that, if a complex ion contains multiple polar bonds between the ligands and the central metal ion, it should be able to engage in a very large amount of dipole–dipole interactions. This stabilizes the dissolution of the complex ion. The end result is that such complexes tend to have very high Ksp values.
When forming a complex ion, one must often use a mixture of solutions. For this reason, a distinction must be made between the Ksp of the solution and that of the complex ion itself. The dissolution of the original solution is termed Ksp, and the subsequent formation of the complex ion in solution is termed Kf (the formation or stability constant of the complex in solution). An example is shown below in which the diamminesilver(I) complex is formed in a silver chloride solution:
AgCl ( s ) ⇌ Ag + ( a q ) + Cl − ( a q ) K sp = 1.8 × 10 − 10 = [ Ag + ] [ Cl − ] Ag + ( a q ) + 2 NH 3 ( a q ) ⇌ [ Ag ( NH 3 ) 2 ] + K f = 1.6 × 10 7 = [ [ Ag(NH 3 ) 2 ] + ] [Ag + ][NH 3 ] 2
Notice that the formation constant (Kf) of the complex ion is significantly larger than the Ksp of the compound providing the metal ion. This is part of the explanation for why the initial dissolution of the metal ion is the rate-limiting step of complex ion formation. However, Le Châtelier’s principle is at play in these reactions as well. Ultimately, complex ions form to become more soluble in solution. And as an amount of silver ion is being used up to form the complex ion itself, the dissociation reaction of AgCl shifts to the right, providing more silver for complex ion formation.
Example: A 0.1 mol sample of CuS is added to 1.00 L of 1.00 M NH3. What is the final concentration of the complex ion, tetraamminecopper(II)?
CuS ( s ) ⇌ Cu 2 + ( a q ) + S 2 − ( a q ) K sp = 8 × 10 − 37 Cu 2 + ( a q ) + 4 NH 3 ( a q ) ⇌ [ Cu ( NH 3 ) 4 ] 2 + ( a q ) K f = 1.1 × 10 13
Solution: First determine the amount of copper ion produced from copper sulfide (CuS) in solution. Comparison of the two equilibrium constants shows that the CuS dissociation has a Ksp of 8 × 10-37, indicating that this reaction is unlikely to proceed as the forward reaction is not favorable. However, if the formation of the complex ion occurs simultaneously, the large Kf of 1.1 × 1013 for this process will drive the dissociation of CuS forward as the Cu2+ ions are consumed in the second reaction due to Le Châtlier's principle. Note that, even without quantitative analysis, the large value of Kf is a sign that the formation of the product of the second reaction will be highly favorable. In fact, if these two reactions are simultaneous, the CuS will ultimately be completely consumed due to this effect.
Thus, the amount of Cu2+ available to react is given by [ Cu 2 + ] = 0.1 mol 1.00 L = 0.1 M . The reactions given show a 1:1 relationship between the Cu2+ available and the [Cu(NH3)4]2+ that is generated. In other words, almost all of the Cu2+ ions will be used up to form the complex. Therefore, the concentration of the [Cu(NH3)4]2+ complex ion is 0.1 M.
Common Ion Effect
The solubility of a substance varies depending on the temperature of the solution, the solvent, and in the case of a gas-phase solute, the pressure. Solubility is also affected by the addition of other substances to the solution. The effect of a complex ion increasing the solubility of a substance is not typical and is—in fact—opposite to the effect seen in many mixtures of solutions.
One of the more challenging solution chemistry problems on the MCAT is calculation of the equilibrium concentration of a salt in a solution that already contains one of the ions in that salt. The solubility of a salt is considerably reduced when it is dissolved in a solution that already contains one of its constituent ions as compared to its solubility in a pure solvent. This reduction in molar solubility is called the common ion effect. As described above, the molar solubility of a compound is its concentration (in moles per liter) at equilibrium at a given temperature. If X moles of AmBn (s) can be dissolved in one liter of solution to reach saturation, then the molar solubility of AmBn (s) is X molar.
Pay attention to the effect of the common ion: its presence results in a reduction in the molar solubility of the salt. Note, however, that the presence of the common ion has no effect on the value of the solubility product constant itself. For example, if a salt such as CaF2 is dissolved into water already containing Ca2+ ions (from some other salt, perhaps CaCl2), the solution will dissolve less CaF2 than would an equal amount of pure water.
The common ion effect is really Le Châtelier’s principle in action. Because the solution already contains one of the constituent ions from the products side of the dissociation equilibrium, the system will shift toward the left side, reforming the solid salt. As a result, molar solubility for the solid is reduced, and less of the solid dissolves in the solution—although the Ksp remains constant.
One can take advantage of the common ion effect to separate out specific compounds in a solution mixture. For example, in a solution of silver salts, one could add sodium or potassium chloride to preferentially precipitate silver(I) chloride. By adding an appropriate counterion in excess, the dissociation reaction shifts to the left, forming the solid salt.
Example: The Ksp of AgI in aqueous solution is 8.5 × 10−17. If a 1 × 10−5 M solution of AgNO3 is saturated with AgI, what will be the final concentration of the iodide ion?
Solution: The concentration of Ag+ in the original AgNO3 solution will be 1 × 10−5 M because AgNO3 will fully dissociate (review the solubility rules from earlier in this chapter). Some small amount of AgI will dissociate into the solution, which is the molar solubility x of AgI under these conditions. The net silver concentration from both AgNO3 and AgI will become 1 × 10−5 M + x. Because no iodide was present in solution until the AgI began dissociating, the concentration of iodide will be x. Thus, the Ksp expression for the dissociation of AgI can be written as:
K sp = [ Ag + ] [ I – ] 8 . 5 × 10 – 17 = [ 1 × 10 – 5 M + x ] [ x ]
Given that the value of Ksp for this reaction is 10-16, only a minuscule amount of AgI will be dissociated. Thus, the value of x is sufficiently small to be negligible when added to 10-5. The math simplifies to:
8 . 5 × 10 – 17 = [ 1 × 10 – 5 M ] [ x ] 8 . 5 × 10 – 12 = x
This question asks for the concentration of iodide, which—based on the equilibrium expression—is represented by x. Thus, [I−] = 8.5× 10−12 M.
MCAT CONCEPT CHECK 9.3:
Before you move on, assess your understanding of the material with these questions.
-
Calculate the Ksp of Ni(OH)2 in water, given that its molar solubility is 5.2 × 10−6 M.
__________________________________
__________________________________
__________________________________
-
The Ksp of Ba(OH)2 is 5.0 × 10−3. Assuming that barium hydroxide is the only salt added to form a solution, calculate the ion product of the following solutions based on the concentration of Ba2+. Then, predict the behavior of the given solutions (dissolution, equilibrium, or precipitation):
[Ba2+] Ion Product Behavior of Solution
0.5 M
0.1 M
0.05 M
-
What is the molar solubility of Zn(OH)2 (Ksp = 4.1 × 10−17) in a 0.1 M solution of NaOH?
__________________________________
__________________________________
9.4 Colligative Properties
LEARNING OBJECTIVES
After Chapter 9.4, you will be able to:
- Recall the names, equations, and applications of the common colligative properties
- Describe the relationship between molality and molarity for a compound
- Calculate the boiling point, freezing point, vapor pressure, or osmotic pressure of a solution
The colligative properties are physical properties of solutions that are dependent on the concentration of dissolved particles but not on the chemical identity of the dissolved particles. These properties—vapor pressure depression, boiling point elevation, freezing point depression, and osmotic pressure—are usually associated with dilute solutions.
Raoult’s Law
Raoult’s law accounts for vapor pressure depression caused by solutes in solution. As solute is added to a solvent, the vapor pressure of the solvent decreases proportionately. For example, consider compound A in Figure 9.9. Compound A in its pure form (mole fraction = 1.0) has a particular vapor pressure, indicated by PA° . At the same temperature, compound B has a lower vapor pressure, indicated by PB° . Note that, as the concentration of B increases, the vapor pressure of A decreases. Indeed, as more solute is dissolved into solvent (as more B is dissolved into A), the vapor pressure of the solvent decreases.
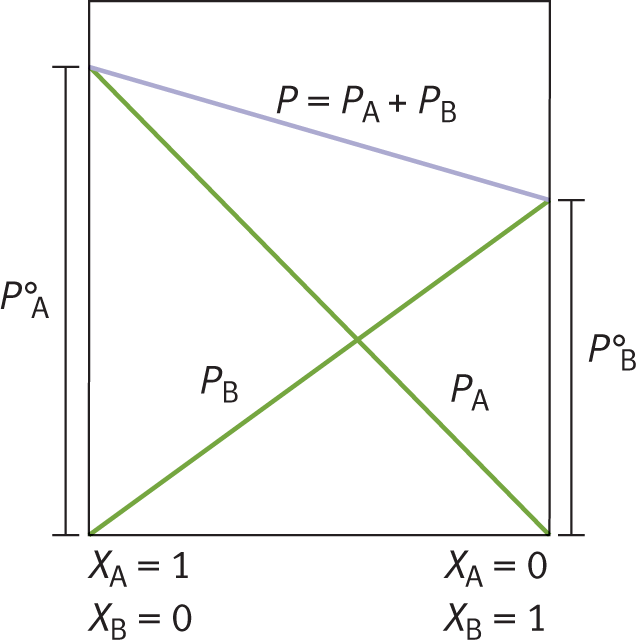
Figure 9.9. Raoult’s Law As more of solute B is dissolved in solvent A, the vapor pressure of solvent A decreases.
On a molecular level, the presence of the solute molecules can block the evaporation of solvent molecules but not their condensation. This reduces the vapor pressure of the solution compared to the pure solvent, as seen in Figure 9.10.
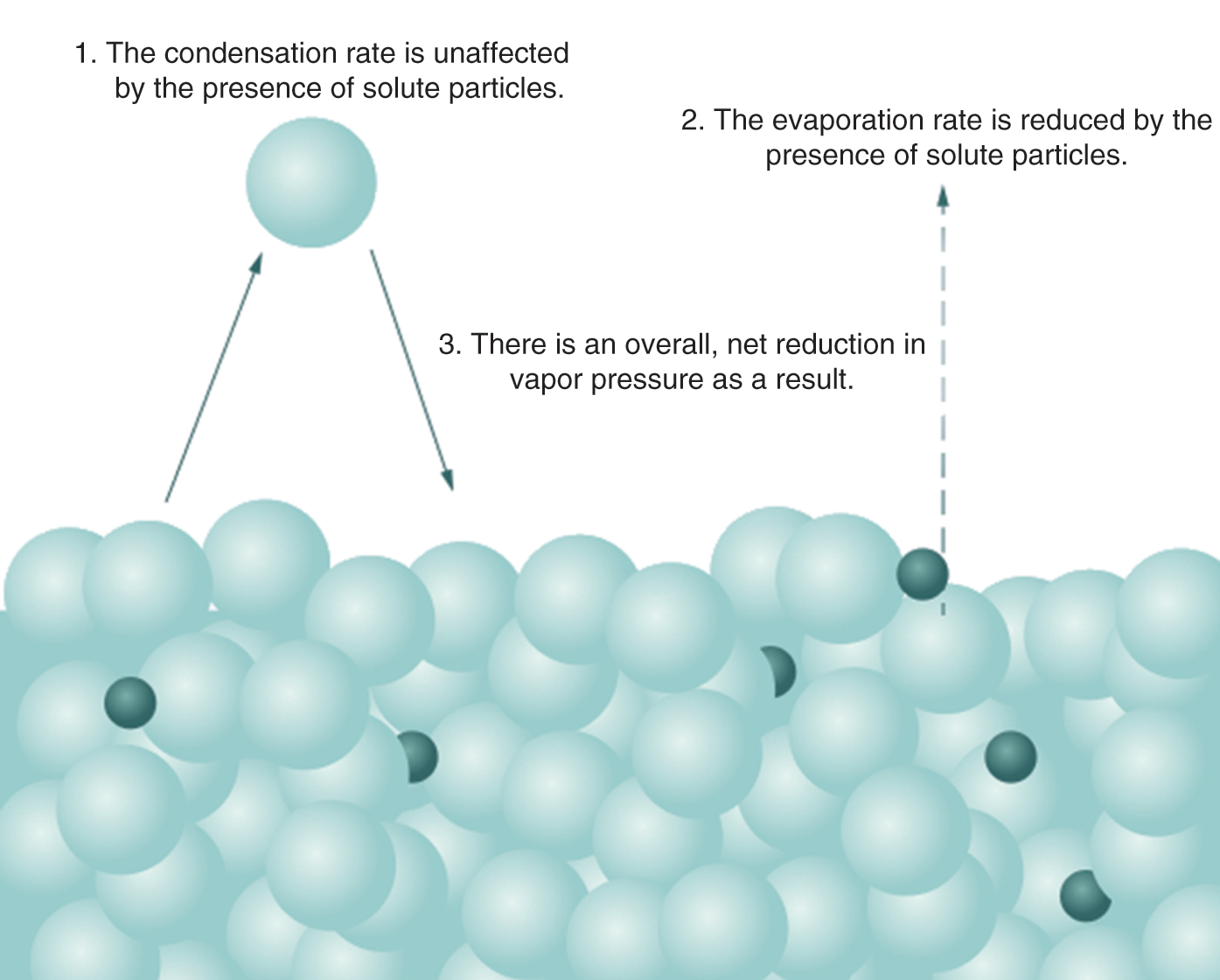
Figure 9.10. Molecular Basis of Raoult’s Law
Raoult’s law is expressed mathematically as:
PA = XAPA°
Equation 9.8
where PA is the vapor pressure of solvent A when solutes are present, XA is the mole fraction of the solvent A in the solution, and PA° is the vapor pressure of solvent A in its pure state.
KEY CONCEPT
Vapor pressure depression goes hand in hand with boiling point elevation. The lowering of a solution’s vapor pressure would mean that a higher temperature is required to match atmospheric pressure, thereby raising the boiling point.
Raoult’s law holds only when the attraction between the molecules of the different components of the mixture is equal to the attraction between the molecules of any one component in its pure state. When this condition does not hold, the relationship between mole fraction and vapor pressure will deviate from Raoult’s law. Solutions that obey Raoult’s law are called ideal solutions.
Example: What is the change in vapor pressure when 180 grams of glyceraldehyde (C3H6O3) are added to 0.18 L of water at 100 °C?
Solution: The density of water at 100 °C is close to 1 g mL , and the vapor pressure of water at the same temperature is 1 atm because this is the boiling point of water.
In order to find the mole fraction of the solvent, first find the molar mass of the solute (glyceraldehyde) and solvent (water). 180 g glyceraldehyde represents 2 moles of glyceraldehyde. 0.18 L of water has a mass around 180 g, which represents 10 moles of water. The mole fraction of water is therefore 10 mol water 12 mol total = 5 6 = 0. 83 .
To find the vapor pressure change, we want to find the difference in the old pressure and the new pressure. The new pressure can be calculated from Raoult’s law:
PA = XAPA° = (0.83)(1 atm) = 0.83 atm.
The change in vapor pressure is therefore 1 atm – 0.83 atm = 0.17 atm.
Example: What is the vapor pressure at room temperature of a mixture containing 58 g butane (C4H10) and 172 g hexane (C6H14)? (Note: The vapor pressures of pure butane and pure hexane are 172 kPa and 17.6 kPa, respectively, at 25 °C.)
Solution: First, determine the number of moles of each substance. 58 g butane represents 1 mole of butane. 172 g hexane represents 2 moles of hexane. Then, determine the mole fractions of each component of the mixture.
X butane = moles of butane total moles = 1 3 X hexane = moles of hexane total moles = 2 3
Then, calculate the vapor pressure of each component:
P butane ∘ = X butane P butane = 1 3 172 kPa ≈ 1 3 1 80 kPa = 60 kPa P hexane ∘ = X hexane P hexane = 2 3 17 . 6 kPa ≈ 2 3 18 kPa = 12 kPa
The total vapor pressure is the sum of the two vapor pressures. Thus, the total vapor pressure is 60 + 12 = 72 kPa (actual = 69.1 kPa).
Boiling Point Elevation
When a nonvolatile solute is dissolved into a solvent to create a solution, the boiling point of the solution will be greater than that of the pure solvent. The boiling point is the temperature at which the vapor pressure of the liquid equals the ambient (incident) pressure. We’ve just seen that adding solute to a solvent results in a decrease in the vapor pressure of the solvent in the solution. If the vapor pressure of a solution is lower than that of the pure solvent, then more energy (and consequently a higher temperature) will be required before its vapor pressure equals the ambient pressure. The extent to which the boiling point of a solution is raised relative to that of the pure solvent is given by the formula
ΔTb = iKbm
Equation 9.9
where ΔTb is the increase in boiling point, i is the van't Hoff factor, Kb is a proportionality constant characteristic of a particular solvent (which will be provided on Test Day), and m is the molality of the solution. The van't Hoff factor corresponds to the number of particles into which a compound dissociates in solution. For example, i = 2 for NaCl because each formula unit of sodium chloride dissociates into two particles—a sodium ion and a chloride ion—when it dissolves. Covalent molecules such as glucose do not readily dissociate in water and thus have i values of 1.
MCAT EXPERTISE
The boiling point elevation formula calculates the amount that the normal boiling point is raised. The value calculated is not the boiling point itself.
Example: 400 g AlCl3 is dissolved in 1.5 L of water at room temperature ( K b = 0.512 K · kg mol ) . How much does the boiling point increase after adding the aluminum chloride?
Solution: Water at room temperature has a density of 1 g mL . Therefore, 1.5 L is the same as 1.5 kg. The van't Hoff factor for aluminum chloride is 4 because it breaks down to form 1 aluminum cation and 3 chloride anions. To determine the molality, we will also need to know how many moles 400 g AlCl3 represents.
400 g AlCl 3 × 1 mol 133.5 g ≈ 3 mol AlCl 3
The molality is therefore 3 mol AlCl 3 1.5 kg = 2 m .
Then, plug into the boiling point elevation equation.
Δ T b = i K b m = ( 4 ) ( 0.512 K · kg mol ) ( 2 m ) ≈ 4 K ( actual = 4.1 K )
Freezing Point Depression
The presence of solute particles in a solution interferes with the formation of the lattice arrangement of solvent molecules associated with the solid state. Thus, a greater amount of energy must be removed from the solution (resulting in a lower temperature) in order for the solution to solidify. For example, pure water freezes at 0 °C, but for every mole of solute dissolved in 1 kg of water, the freezing point is lowered by 1.86 °C. Therefore, the Kf for water is 1.86 K · kg mol . As is the case for Kb, the values for Kf are unique to each solvent and will be provided on Test Day. The formula for calculating the freezing point depression for a solution is
ΔTf = iKfm
Equation 9.10
where ΔTf is the freezing point depression, i is the van't Hoff factor,Kf is the proportionality constant characteristic of a particular solvent, and m is the molality of the solution. Freezing point depression is a colligative property and depends only on the concentration of particles, not on their identities.
Example: 400 g of AlCl3 is dissolved in 1.5 L of water at room temperature ( K f = 1.86 K · kg mol ) . What is the new freezing point of this solution?
Solution: Using the same variables for i and m from the previous example,
Δ T f = i K f m = ( 4 ) ( 1 . 86 K · kg mol ) ( 2 m ) ≈ 15 K ( actual = 14.9 K )
The normal freezing point of water is 273 K. The freezing point is going to be depressed (or decreased) by 15 K. The new freezing point is therefore 273 – 15 = 258 K = –15 °C.
MCAT EXPERTISE
As with boiling point elevation, there is a distinction between the calculated value and a final answer. The freezing point depression calculates the amount that the normal freezing point is lowered. As always, read the question to determine if it is asking for the change in temperature (ΔT) or the new (altered) boiling or freezing point.
This effect is the explanation for why many people in colder climates salt icy roads in the winter. Salt mixes with the snow and ice and initially dissolves into the small amount of liquid water that is in equilibrium with the solid phase (the snow and ice). The solute in solution causes a disturbance to the equilibrium such that the rate of melting is unchanged (because the salt can’t interact with the solid water that is stabilized in a rigid lattice arrangement), but the rate of freezing is decreased (the solute displaces some of the water molecules from the solid–liquid interface and prevents liquid water from entering into the solid phase).
This imbalance causes more ice to melt than water to freeze. Melting is an endothermic process, so heat is initially absorbed from the liquid solution, causing the solution temperature to fall below the ambient temperature. Now, there is a temperature gradient, and heat flows from the warmer air to the cooler aqueous solution; this additional heat facilitates more melting—even though the temperature of the solution is actually colder than it was before the solute was added! The more the ice melts into liquid water, the more the solute is dispersed through the liquid. The resulting salt solution, by virtue of the presence of the solute particles, has a lower freezing point than the pure water and remains in the liquid state even at temperatures that would normally cause pure water to freeze.
Osmotic Pressure
Osmotic pressure is covered primarily in Chapter 8 of MCAT Biochemistry Review, but a brief recap is provided here. Osmotic pressure refers to a “sucking” pressure generated by solutions in which water is drawn into a solution. Formally, the osmotic pressure is the amount of pressure that must be applied to counteract this attraction of water molecules for the solution. The equation for osmotic pressure is:
∏ = iMRT
Equation 9.11
where ∏ is the osmotic pressure, i is the van't Hoff factor, M is the molarity of the solution, R is the ideal gas constant, and T is the temperature.
Water moves in the direction of higher solute concentration. For instance, pure water (no solute concentration) will traverse a semipermeable membrane to a solution containing solute particles (such as NaCl) and increase the level of the water as a result, as shown in Figure 9.11.

Figure 9.11. Change in Water Level Due to Osmotic Pressure
MCAT CONCEPT CHECK 9.4:
Before you move on, assess your understanding of the material with these questions.
-
What is a colligative property?
_____________________________
-
How are molality and molarity related for water? How are they related for other solvents?
- Water:________________________________
- Other solvents:____________________________
-
Determine the vapor pressure of a solution containing 190 g MgCl2 in 540 g water at room temperature: (Note: The vapor pressure of pure water at 25°C is 3.2 kPa.)
_____________________________
-
Determine the new boiling point of a solution containing 190 g MgCl2 in 1500 g water at room temperature: ( K b = 0.512 K · kg mol )
_____________________________
-
Determine the freezing point depression of a solution containing 58.5 g of NaCl in 1800 g of water at room temperature: ( K f = 1.86 K · kg mol )
__________________________________
Conclusion
Our review of solution chemistry has provided an opportunity for us to consider the nature of solutions, solutes, and solvents, and the interactions between solutes and solvents in the formation of solutions. We reviewed solubility and the rules that reflect the solubility of common compounds in water. The different ways of expressing the amount of solute in solution were identified, and examples were given for each unit of concentration, including percent composition, mole fraction, molarity, molality, and normality. Next, we reviewed the thermodynamic principles of solution equilibria and defined unsaturated, saturated, and supersaturated solutions with respect to ion product (IP) and solubility product constant (Ksp). Subsequently, we discussed the common ion effect from the perspective of Le Châtelier’s principle for a solution at equilibrium. And finally, we examined the colligative properties of solutions and the mathematics that govern them. The colligative properties—vapor pressure depression, boiling point elevation, freezing point depression, and osmotic pressure—are physical properties of solutions that depend on the concentration of dissolved particles but not on their chemical identities.
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Nature of Solutions
- Solutions are homogeneous mixtures composed of two or more substances.
- They combine to form a single phase, generally the liquid phase.
- Solvent particles surround solute particles via electrostatic interactions in a process called solvation or dissolution.
- Aqueous solutions are most important for the MCAT; solvation in water can also be called hydration.
- Most dissolutions are endothermic, although the dissolution of gas into liquid is exothermic.
- Solubility is the maximum amount of a solute that can be dissolved in a given solvent at a given temperature; it is often expressed as molar solubility—the molarity of the solute at saturation.
- Complex ions or coordination compounds are composed of metallic ions bonded to various neutral compounds and anions, referred to as ligands.
- Formation of complex ions increases the solubility of otherwise insoluble ions (the opposite of the common ion effect).
- The process of forming a complex ion involves electron pair donors and electron pair acceptors such as those seen in coordinate covalent bonding.
Concentration
- There are many ways of expressing concentration.
- Percent composition by mass (mass of solute per mass of solution times 100%) is used for aqueous solutions and solid-in-solid solutions.
- The mole fraction (moles of solute per total moles) is used for calculating vapor pressure depression and partial pressures of gases in a system.
- Molarity (moles of solute per liters of solution) is the most common unit for concentration and is used for rate laws, the law of mass action, osmotic pressure, pH and pOH, and the Nernst equation.
- Molality (moles of solute per kilograms of solvent) is used for boiling point elevation and freezing point depression.
- Normality (number of equivalents per liters of solution) is the molarity of the species of interest and is used for acid–base and oxidation–reduction reactions.
Solution Equilibria
- Saturated solutions are in equilibrium at that particular temperature.
- Thesolubility product constant (Ksp) is simply the equilibrium constant for a dissociation reaction.
- Comparison of theion product (IP) to Ksp determines the level of saturation and behavior of the solution:
- IP < Ksp: the solution is unsaturated, and if more solute is added, it will dissolve
- IP = Ksp: the solution is saturated (at equilibrium), and there will be no change in concentrations
- IP > Ksp: the solution is supersaturated, and a precipitate will form
- Formation of a complex ion in solution greatly increases solubility.
- The formation or stability constant (Kf) is the equilibrium constant for complex formation. Its value is usually much greater than Ksp.
- The formation of a complex increases the solubility of other salts containing the same ions because it uses up the products of those dissolution reactions, shifting the equilibrium to the right (the opposite of the common ion effect).
- Thecommon ion effect decreases the solubility of a compound in a solution that already contains one of the ions in the compound. The presence of that ion in solution shifts the dissolution reaction to the left, decreasing its dissociation.
Colligative Properties
- Colligative properties are physical properties of solutions that depend on the concentration of dissolved particles but not on their chemical identity.
- Vapor pressure depression follows Raoult’s law.
- The presence of other solutes decreases the evaporation rate of a solvent without affecting its condensation rate, thus decreasing its vapor pressure.
- Vapor pressure depression also explains boiling point elevation—as the vapor pressure decreases, the temperature (energy) required to boil the liquid must be raised.
- Freezing point depression and boiling point elevation are shifts in the phase equilibria dependent on the molality of the solution.
- Osmotic pressure is primarily dependent on the molarity of the solution.
- For solutes that dissociate, the van't Hoff factor (i) is used in freezing point depression, boiling point elevation, and osmotic pressure calculations.
ANSWERS TO CONCEPT CHECKS
**9.1**
- Solvation refers to the breaking of intermolecular forces between solute particles and between solvent particles, with formation of intermolecular forces between solute and solvent particles. In an aqueous solution, water is the solvent.
- Solubility is the amount of solute contained in a solvent. Saturation refers to the maximum solubility of a compound at a given temperature; one cannot dissolve any more of the solute just by adding more at this temperature.
- Solubility of solids can be increased by increasing temperature. Solubility of gases can be increased by decreasing temperature or increasing the partial pressure of the gas above the solvent (Henry’s law).
- Group I metals, ammonium, nitrate, and acetate salts are always soluble.
**9.2**
-
Compound Molality Molarity Normality Glucose ( 180 g mol ) 1 mol 250 kg solvent = 0.004 m 1 mol 250 L solution = 0.004 M 0.004 N (glucose does not dissociate)
Carbonate ( 60 g mol ) 3 mol 250 kg solvent = 0.012 m 3 mol 250 L solution = 0.012 M Approximately 0.024 N(twice the molarity)
Please note that the denominator for molality uses kilograms of solvent, whereas the denominator for molarity uses liters of total solution. These quantities are not necessarily the same and so you must be careful!
An example will help clarify the difference: Suppose 1 mol of solute were added to 1 L of water. 1 L of water has mass 1 kg, so you would simply use 1 kg solvent in the denominator of the molality computation, because molality's denominator is the mass of the solvent itself. However, when adding 1 mol solute to 1 L water, the volume of the final solution will likely be significantly larger than 1 L, because the solute itself takes up space. Therefore, you could not simply plug 1 L into the molarity equation, because molarity's denominator depends on the total volume of the final, mixed solution. Instead, you would need to remeasure the total, final volume, and use that number when computing molarity.
All that said, in the problem in the concept check, the initial volume of solvent is so large (250 L) that the added solute will not appreciably change the volume of final solution. Therefore, in this case, we were able to add 250 L directly into the molarity computation.
- M i V i = M f V f → V i = M f V f M i = ( 25 ppm ) ( 100 mL) ( 100 ppm ) = 25 mL .
Thus, start with 25 mL of the stock solution and add 75 mL pure water to get 100 mL of solution with 25 ppm Cl2.
- X NaCl = moles of NaCl total moles = 90 g [ 1 mol 58.5 g ] 90 g [ 1 mol 58.5 g ] + 10 4 g [ 1 mol 18 g ] ≈ 90 1 60 90 1 60 + 10 4 1 20 ≈ 1.5 1.5 + 500 ≈ 1 . 5 500 ≈ 1 . 5 5 × 10 2 ≈ 1 3 × 10 - 2 ≈ 3 × 10 − 3 (actual = 2.8 × 10 − 3 ) Percent composition = mass of solute mass of solution × 100 % = 90 g 90 g + 10 4 g × 100 % ≈ 90 10 4 × 100 % = 0.9 % ( actual = 0 .89%)
**9.3**
- First, write out the balanced equation: Ni(OH) 2 → Ni 2 + + 2OH -
Next, identify that the molar solubility x represents the amount of Ni(OH)2 that dissociates, creating x of Ni2+ and 2x of OH–. Write out the Ksp equation, and plug in the values of x to solve for Ksp:
K sp = [ Ni 2 + ] [ OH - ] 2 = x ( 2 x ) 2 K sp = ( 5 . 2 × 10 - 6 ) ( 2 × 5 . 2 × 10 - 6 ) 2 K sp = ( 5 × 10 - 6 ) ( 10 × 10 - 6 ) 2 ≈ ( 5 × 10 - 6 ) ( 10 - 5 ) 2 K sp ≈ 5 × 10 - 6 × 10 - 10 = 5 × 10 - 16 ( actual value = 5 . 6 × 10 - 16 )
Ba(OH) 2 → Ba 2 + + 2 OH - K sp = [Ba 2 + ][OH - ] 2
- Start with the balanced reaction and calculation for Ksp, which can also be used to calculate Q. Keep in mind that for every x of Ba(OH)2 that dissolves, x of Ba2+ will be produced, and 2x of OH–.
[Ba2+] Ion Product Behavior of Solution
0.5 M (0.5 M)(1 M)2 = 0.5 0.5 > 5.0 × 10−3 → precipitation
0.1 M (0.1 M)(0.2 M)2 = 4.0 × 10−3 4.0 × 10−3 < 5.0 × 10−3 → dissolution
0.05 M (0.05 M)(0.1 M)2 = 5.0 × 10−4 5.0 × 10−4 < 5.0 × 10−3 → dissolution
Note that the concentration of hydroxide is double that of barium. While there will be a very small contribution of hydroxide from the autoionization of water, this amount is negligible compared to the values given in the question.
- Start by writing the balanced reaction for the least soluble salt in the problem: Zn(OH)2 → Zn2+ + 2 OH– . Next, write out the Ksp equation and enter the variables for the concentrations. The Zn2+ concentration will equal x, the molar solubility under these conditions, but the OH- concentration will come from two contributors: the dissociated Zn(OH)2, and the 0.1 M NaOH solution. This results in the following Ksp expression: Ksp = [Zn2+][OH−]2 = (x)(0.1 + 2x)2. Since the Ksp for Zn(OH)2 is 4.1 x 10–17, x will be negligible compared to the 0.1 M from NaOH. The Ksp expression simplifies to: Ksp = (x)(0.1)2. Thus, 4.1 × 10−17 = 0.01x. x = molar solubility of Zn(OH)2= 4.1 × 10−15.
**9.4**
- Colligative properties are those that depend on the amount of solute present, but not the actual identity of the solute particles. Examples include vapor pressure depression, boiling point elevation, freezing point depression, and osmotic pressure.
- Molarity (M) and molality (m) are nearly equal at room temperature. This is only because 1 L solution is approximately equal to 1 kg solvent for dilute solutions (the denominators of the molarity and molality equations, respectively). For other solvents, molarity and molality differ significantly because their densities are not 1 g mL like water.
- 190 g MgCl 2 × 1 mol 95.3 g = 2 mol MgCl 2 . 540 g H 2 O × 1 mol 18 g = 30 mol H 2 O X water = 30 moles water 32 total moles P water = X water P ∘ water = 30 moles water 32 total moles × 3 . 2 kPa = 3 . 0 kPa
- ∆ T b = i K b m = 3 0 . 512 K · kg mol 2 mol MgCl 2 1 . 5 kg water ≈ 2 K actual = 2.04 K
The new boiling point will be 373 + 2 = 375 K.
- Δ T f = i K f m = ( 2 ) ( 1.86 K · kg mol ) ( 1 mol NaCl 1 . 8 kg water ) ≈ 2 K ( actual = 2 . 06 K )
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. A
The equation ΔTb = iKbm can be used to solve this problem. The change in boiling point is 101.0 − 100 = 1.0 °C. Then, plug that into:
Δ T b = i K b m → m = Δ T b i K b = 1. 0 K ( 1 ) ( 0.512 K · kg mol ) ≈ 2 m
The van’t Hoff factor for this solute is 1 because the molecule does not dissociate into smaller components. Then, convert to grams of solute using the definition of molality:
molality = moles of solute kg of solvent → moles of solute = ( 2 m ) ( 0.1 kg) = 0.2 mol .
The mass used in this equation is 0.1 kg because 100 mL of water has a mass of 0.1 kg. Then, determine the molar mass: molar mass = 70 g 0.2 mol = 350 g mol , which is closest to (A).
2. D
All three choices can make a solution as long as the two components create a mixture that is of uniform appearance (homogeneous). Hydrogen in platinum is an example of a gas in a solid. Brass and steel are examples of homogeneous mixtures of solids. The air we breathe is an example of a homogeneous mixture of gases; while these are more commonly referred to as mixtures, they still fit the criteria of a solution.
3. B
Benzene and toluene are both organic liquids and have very similar properties. They are both nonpolar and are almost exactly the same size. Raoult’s law states that ideal solution behavior is observed when solute–solute, solvent–solvent, and solute–solvent interactions are all very similar. Therefore, benzene and toluene in solution will be predicted to behave as a nearly ideal solution.
4. C
Melting point depresses upon solute addition, making (A) and(B) incorrect. Solute particles interfere with lattice formation, the highly organized state in which solid molecules align themselves. Colder-than-normal conditions are necessary to create the solid structure.
5. D
The first step will most likely be endothermic because energy is required to break molecules apart. The second step is also endothermic because the intermolecular forces in the solvent must be overcome to allow incorporation of solute particles. The third step will most likely be exothermic because polar water molecules will interact with the dissolved ions, creating a stable solution and releasing energy.
6. C
BeF2 will cause the most negative contribution to ΔS°soln through the hydration effect because Be2+ cation has the highest charge density compared to the other ions. All of the other ions have a charge density of +1 or –1, whereas Be2+ has a charge density of magnitude two.
7. B
Formation of complex ions between silver ions and ammonia will cause more molecules of solid AgCl to dissociate. The equilibrium is driven toward dissociation because the Ag+ ions are essentially being removed from solution when they complex with ammonia. This rationale is based upon Le Châtelier’s principle, stating that when a chemical equilibrium experiences a change in concentration, the system will shift to counteract that change.
8. B
The mass percent of a solute equals the mass of the solute divided by the mass of the total solution times 100%. mass % = mass of solute total mass of solution ×100%.
Plug in the values given for sucrose, the volume of water and the density of water to determine the % mass of sucrose:
100 g sucrose ( 300 mL H 2 O ) ( 0 . 975 g/mL ) + 100 g sucrose × 100 % ≈ 100 300 + 100 × 100 % = 100 400 × 100 % = 25 %
Keep in mind that in rounding while calculating, the denominator was estimated to be larger than the actual value, thus giving an answer that is slightly lower than the actual value. Thus, the correct answer is (B), 25.5%. (A) results if rounding error is not taken into account. While these answers are very close, the mass of the water must be slightly less than 300 g, given the density value, so the percent composition of sucrose must be slightly higher than 25%. If the solute’s mass is not added to the solvent’s, the calculated value is 34.2%, which is(D).(C) neglects both the addition step and the rounding error.
9. A
Mixtures that have a higher vapor pressure than predicted by Raoult’s law have stronger solvent–solvent and solute–solute interactions than solvent–solute interactions. Therefore, particles do not want to stay in solution and more readily evaporate, creating a higher vapor pressure than an ideal solution. Two liquids that have different properties, like hexane (hydrophobic) and ethanol (hydrophilic, small) in (A), would not have many interactions with each other and would cause positive deviation (i.e. higher vapor pressure). (B) and (C) are composed of liquids that are similar to one another and would not show significant deviation from Raoult’s law. (D) contains two liquids that would interact very well with each other, which would actually cause a negative deviation from Raoult’s law—when attracted to one other, solutes and solvents prefer to stay in liquid form and have a lower vapor pressure than predicted by Raoult’s law.
10. A
Dissolution is governed by enthalpy and entropy, which are related by the equation ΔG°soln = ΔH°soln − TΔS°soln. The cooling of the solution indicates that heat is used up in this bond-breaking reaction. In other words, dissolution is endothermic, and ΔH is positive. The reaction is occurring spontaneously, so ΔG must be negative. The only way that a positive ΔH can result in a negative ΔG is if entropy, ΔS, is a large, positive value as in (A). Conceptually, that means that the only way the solid can dissolve is if the increase in entropy is great enough to overcome the increase in enthalpy. (B) is incorrect because it is clearly stated in the question stem that KCl dissolves; further, all salts of Group 1 metals are soluble. (C) is incorrect because ΔS°soln must be positive in order for KCl to dissolve. Finally, (D) is incorrect because solute dissolution would cause the boiling point to elevate, not depress. It is also not a piece of evidence that could be found simply by observing the beaker’s temperature change.
11. B
The equation to determine the change in boiling point of a solution is as follows: ΔTb = iKbm. m is the molality of the solution, and Kb is the boiling point elevation constant. In this case, the solvent is always water, so Kb will be the same for each solution. What is needed is the number of dissociated particles from each of the original species. This is referred to as the van’t Hoff factor (i) and is multiplied by molality to give a normality (the concentration of the species of interest—in this case, all particles). The normality values determine which species causes the greatest change in boiling point.
Species Number of Moles Number of Dissolved Particles i × m (Normality)
CaSO4 0.4 2 0.8
Fe(NO3)3 0.5 4 2.0
CH3COOH 1.0 Between 1 and 2 (acetic acid is a weak acid and a low percentage of the molecules will dissociate into 2 particles) Between 1.0 and 2.0
C12H22O11 1.0 1 1.0
The choice is between iron(III) nitrate and acetic acid. The fact that acetic acid is a weak acid indicates that only a few particles will dissociate into H+ and acetate. Therefore, the normality of the acetic acid will be much closer to 1.0 than 2.0.
12. B
Osmotic pressure is given by the formula Π = i M R T . Entering the values from the question stem gives:
Π = ( 1000 × 10 - 3 mOsm/L)(0.0812 L·atm/mol·K)(298 K) ≈ 1 × 0 . 08 × 300 = 24 atm actual value = 24.2 atm
Notice that the concentration of seawater is given for all solutes, which representsi×M. It is also given in mOsm/L, which is converted to moles per liter by multiplying by 10-3. Also, the question asks for the minimum pressure required, which means that the correct answer choice must be slightly above the calculated pressure in order for reverse osmosis to proceed.
13. B
200 ppb of Pb2+ is equivalent to 200 grams of Pb2+ in 109 grams of solution; given the extremely low concentration of lead, the mass of the water can be assumed to be approximately 109 grams, as well. To solve, set up a dimensional analysis question. The units needed at the end are moles per liter (molarity), so convert from grams of lead to moles of lead and grams of water to liters of water:
[ 200 g Pb 2 + 10 9 g H 2 O ] [ 10 3 g H 2 O 1 L ] [ 1 mol Pb 2 + 207.2 g Pb 2 + ] ≈ 200 × 10 3 200 × 10 9 = 1 × 10 − 6 M = 10 × 10 - 7 M ( actual value = 9 . 67 × 10 - 7 M )
Note that the denominator was rounded to a smaller number, meaning the estimated answer is slightly larger than the actual value.
14. A
This question is actually more complicated than it seems since hydroxide is involved. In aqueous solution, the concentration of hydroxide from the autoionization of water is 10–7 M. This concentration is significantly greater (approximately 10,000× greater) than the amount of hydroxide that would be produced by the dissociation of either salt. Thus, the concentration of hydroxide is set by water, not by the dissociation of cobalt(III) hydroxide and thallium(III) hydroxide. When the two solutions are mixed, the concentration of hydroxide does not change. However, in mixing the two solutions, the concentration of the metal ions decreases as a consequence of dilution. As a result, both solutions would be below saturation, justifying (A) as the correct answer.
15. D
The solubility of AgBr can be determined using the Ksp value given in the equation. Some amount of AgBr will dissolve; this is the molar solubility xfor these conditions. When AgBr dissociates, there will be x amount of silver(I) formed and x amount of bromide—which is added to the 0.0010 M Br- already present from NaBr.
AgBr ⇌ Ag + + Br − K sp = [ Ag + ] [ Br − ] 5 . 35 × 10 − 13 = ( x ) ( 0.0010 M + x )
Given the Ksp of 5.4 × 10–13, x will be negligible compared to 0.0010 M. Thus, the math can be simplified to:
5 . 35 × 10 - 13 = ( x ) ( 0 . 0010 ) x = 5 . 35 × 10 - 13 10 - 3 = 5 . 35 × 10 - 10
Therefore, x, the molar solubility, is 5.35 × 10−10, which looks like (C). However, the units of the answer choices are grams per liter, not molarity, and the result must be multiplied by the molar mass ( 187.8 g mol ) :
5 . 35 × 10 − 10 mol L × 187.8 g mol ≈ 5 × 10 − 10 × 200 = 1 , 000 × 10 − 10 = 1 × 10 − 7
which is close to (D). Note that a very accurate approximation was reached by rounding down the first number and rounding up the second, balancing the error.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(9.1) Percent composition by mass: mass of solute mass of solution × 100 %
(9.2) Mole fraction: X A = moles of A total moles of all species
(9.3) Molarity: M = moles of solute liters of solution
(9.4) Molality: m = moles of solute kilograms of solvent
(9.5) Dilution formula: MiVi = MfVf
(9.6) Solubility product constant: Ksp = [An+]m[Bm−]n
(9.7) Ion product: IP = [An+]m[Bm−]n
(9.8) Raoult’s law (vapor pressure depression): PA = XAPA°
(9.9) Boiling point elevation: ΔTb = iKbm
(9.10) Freezing point depression: ΔTf = iKfm
(9.11) Osmotic pressure: ∏ = iMRT
SHARED CONCEPTS
Biology Chapter 10
Homeostasis
General Chemistry Chapter 3
Bonding and Chemical Interactions
General Chemistry Chapter 6
Equilibrium
General Chemistry Chapter 7
Thermochemistry
General Chemistry Chapter 10
Acids and Bases
General Chemistry Chapter 12
Electrochemistry