Chapter 10: Acids and Bases

Chapter 10: Acids and Bases
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you answered correctly, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of the following is not a Brønsted–Lowry base?
-
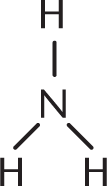
- F−
-
- H — O — N = O
-
Which of the following is closest to the pH of a solution containing 5 mM H2SO4?
- 1
- 2
- 3
- 4
-
Which of the following represents chloric acid?
- HClO3
- ClO3−
- HClO2
- HClO
-
Which of the following bases is the weakest?
- KOH
- NH3
- CH3NH2
- Ca(OH)2
-
The function of a buffer is to:
- maintain a neutral pH.
- resist changes in pH when small amounts of acid or base are added.
- slow down reactions between acids and bases.
- speed up reactions between acids and bases.
-
What is the pH of a solution with an ammonium concentration of 70 mM and an ammonia concentration of 712 mM? (Note: The pKb of ammonia is 3.45.)
- 2.45
- 4.45
- 9.55
- 11.55
Questions 7–9 refer to the titration curve of acid X shown below:
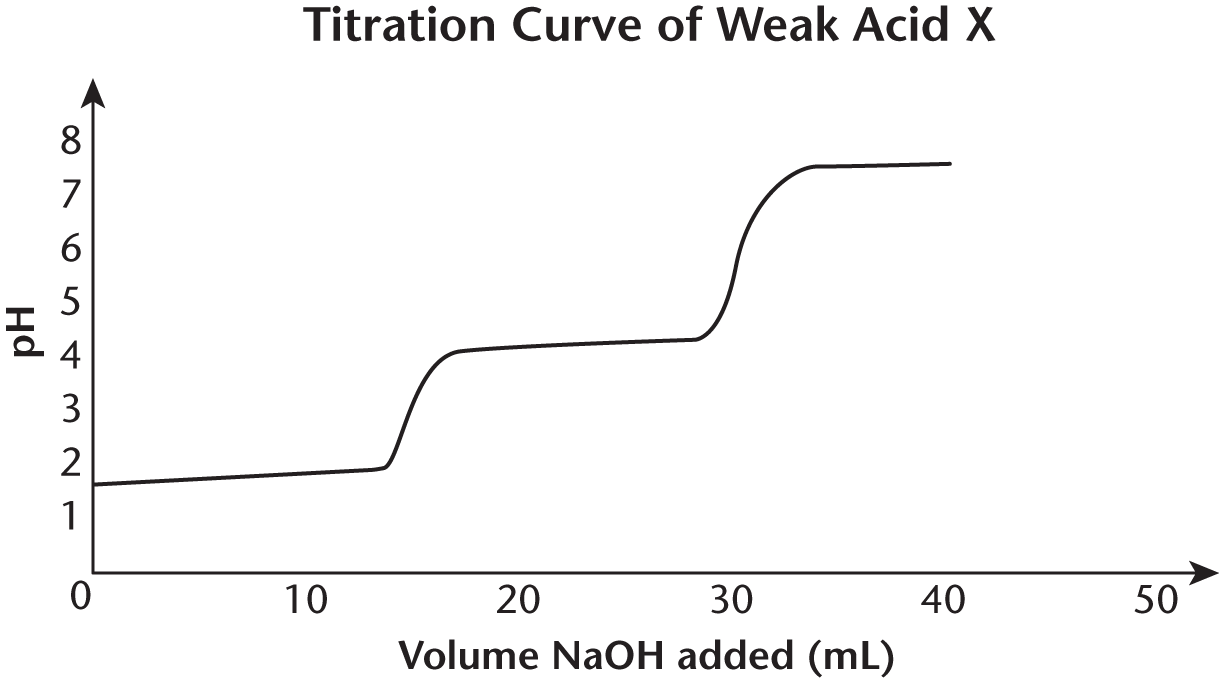
- What is the approximate value of pKa1?
- 1.9
- 2.9
- 3.8
- 4.1
-
At what pH is the second equivalence point?
- pH = 3.0
- pH = 4.1
- pH = 5.9
- pH = 7.2
-
What is the approximate value of pKa2?
- 3.6
- 4.1
- 5.5
- 7.2
-
What is the gram equivalent weight of phosphoric acid?
- 24.5 g
- 32.7 g
- 49.0 g
- 98.0 g
-
What is the [H3O+] of a 2 M aqueous solution of a weak acid HXO2 with Ka = 3.2 × 10−5?
- 6.4 × 10−5 M
- 1.3 × 10−4 M
- 4.0 × 10−3 M
- 8.0 × 10−3 M
-
A solution is prepared with an unknown concentration of a theoretical compound with a Ka of exactly 1.0. What is the pH of this solution?
- Higher than 7
- Exactly 7
- Less than 7
- There is not enough information to answer the question.
-
Which of the following is NOT a characteristic of an amphoteric species?
- Amphoteric species can act as an acid or a base, depending on its environment.
- Amphoteric species can act as an oxidizing or reducing agent, depending on its environment.
- Amphoteric species are sometimes amphiprotic.
- Amphoteric species are always nonpolar.
-
What is the approximate pH of a 1.2 × 10−5 M aqueous solution of NaOH?
- 4.92
- 7.50
- 9.08
- 12.45
-
How many liters of 2 M Ba(OH)2 are needed to titrate a 4 L solution of 6 M H3PO4?
- 1.33 L
- 12 L
- 18 L
- 56 L
Answer Key
- D
- B
- A
- B
- B
- D
- A
- C
- B
- B
- D
- C
- D
- C
- C
Chapter 10: Acids and Bases
CHAPTER 10
ACIDS AND BASES
In This Chapter
10.1 Definitions
Arrhenius
Brønsted–Lowry
Lewis
Amphoteric Species
Acid–Base Nomenclature
10.2 Properties
Autoionization of Water and Hydrogen Ion Equilibria
Strong Acids and Bases
Weak Acids and Bases
Conjugate Acid–Base Pairs
Applications of Ka and Kb
Salt Formation
10.3 Polyvalence and Normality 10.4 Titration and Buffers
General Principles
Strong Acid and Strong Base
Weak Acid and Strong Base
Strong Acid and Weak Base
Weak Acid and Weak Base
Polyvalent Acids and Bases
Buffers
Concept Summary
CHAPTER PROFILE
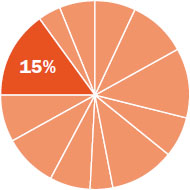
The content in this chapter should be relevant to about 15% of all questions about general chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
4D: How light and sound interact with matter
5A: Unique nature of water and its solutions
Introduction
Some medications can be applied as drops, salves, or creams to mucous membranes. Others are injected. Some employ a transdermal patch, while others are swallowed or inhaled. The route of administration of a drug compound is related to both the location of its target tissue (local or systemic), as well as the chemical and physical properties of the compound. For example, compounds that are water-soluble can be administered intravenously (an aqueous solution dripped directly into the bloodstream), while those that are lipid-soluble can be administered transcutaneously (via a patch or a cream) or orally (in a pill or liquid suspension). The polarity, size, and charge of the drug compound will determine its solubility in polar or nonpolar environments and will be major contributing factors in determining the most effective and efficient route of administration.
Whether a drug compound has an ionic charge is usually a function of the acidic or basic nature of the compound. For example, a basic organic compound that is insoluble in water when neutral can be reacted with an acid to form a salt; because this salt is ionic, it is water-soluble. Correspondingly, an acidic organic compound that is insoluble in water when neutral can be reacted with a base to form a water-soluble salt. On the other hand, the protonated (acidic cationic) form of an organic compound can be reacted with a base to neutralize the compound and release it from its salt, changing (and usually reversing) its solubility in water.
Medical professionals aren’t the only ones concerned about drug solubilities and routes of administration—there’s a science to illicit drugs, too. One of the clearest examples of this is the difference between the two major forms of cocaine, a large alkaloid compound derived from the coca plant. Most commonly, the alkaloid compound is reacted with hydrochloric acid (which protonates its tertiary amine functional group), extracted with water, and dried to a water-soluble powder (cocaine hydrochloride); this powder either is snorted (insufflated) into the nasal cavity, where it is absorbed into the capillary beds, or is injected directly into the venous circulation. The second form of cocaine, the salt form, has a very high boiling point, which is close to the temperature at which cocaine degrades, and cannot be smoked. To produce a vaporizable form of cocaine that can be inhaled from a pipe, the cocaine hydrochloride must be reacted with a base, typically either ammonia (to produce pure freebase cocaine) or sodium bicarbonate (to produce crack cocaine, which is less pure). The base reacts with the protonated tertiary amine, removing the hydrogen ion to reform the neutral alkaloid compound. The freebase cocaine is water-insoluble and usually extracted with ether, or it is left in the aqueous solution, which is heated and evaporated. The freebase and crack forms of cocaine have much lower boiling points; consequently, they can be smoked without risk of degradation.
In this chapter, our focus will be on those two classes of compounds—acids and bases—which are involved in so many important reactions. Acid–base reactions are an important topic for the MCAT; in fact, neutralization reactions are some of the most commonly tested reaction types on Test Day. We will begin with a review of the different definitions of acids and bases and their properties, including their characterization as either strong or weak. Focusing on weak acids and bases, we will discuss the significance of the equilibrium constants Ka and Kb for acids and bases, respectively. Finally, we will review acid–base titrations and buffer systems.
10.1 Definitions
LEARNING OBJECTIVES
After Chapter 10.1, you will be able to:
- Compare and contrast the Arrhenius, Brønsted–Lowry, and Lewis definitions for acids and bases
- Predict the acid formula and name for an anion using Arrhenius acid naming trends
- Identify amphoteric species, and determine whether they are amphiprotic as well
Over the last century, chemists have used different definitions to identify compounds as acids or bases. Three definitions emerged, with each more inclusive than the former one.
Arrhenius
The first and most specific definition of an acid or base is the Arrhenius definition. An Arrhenius acid will dissociate to form an excess of H+ in solution, and an Arrhenius base will dissociate to form an excess of OH− in solution. These behaviors are generally limited to aqueous acids and bases. Arrhenius acids and bases are easily identified; acids contain H at the beginning of their formula (HCl, HNO3, H2SO4, and so on) and bases contain OH at the end of their formula (NaOH, Ca(OH)2, Fe(OH)3, and so on).
MCAT EXPERTISE
Any mention of the Arrhenius definition on Test Day will likely be in comparison to other definitions of acids. The Arrhenius definition is by far the most restrictive; the Brønsted–Lowry and Lewis definitions predominate on the MCAT.
Brønsted–Lowry
A more inclusive definition of acids and bases was proposed independently by Johannes Brønsted and Thomas Lowry in 1923. A Brønsted–Lowry acid is a species that donates hydrogen ions (H+), while a Brønsted–Lowry base is a species that accepts them.
The advantage of this definition over Arrhenius’s is that it is not limited to aqueous solutions. For example, OH−, NH3, and F− are all Brønsted–Lowry bases because each has the ability to accept a hydrogen ion. However, neither NH3 nor F− can be classified as Arrhenius bases because they do not dissociate to produce an excess of OH− ions in aqueous solutions. According to both of these definitions, there is only one way for a species to be an acid: producing hydrogen ions. The only differences between the two definitions for acids are the requirement of an aqueous medium in the Arrhenius definition and the acidity of water. In the Arrhenius definition, water is not considered an acid—it does not produce an excess of H+ in solution. Water is, on the other hand, a Brønsted–Lowry acid because it is able to donate a proton to other species. Most acid–base chemistry reactions on the MCAT will involve the transfer of hydrogen ions in accordance with the Brønsted–Lowry definitions.
MCAT EXPERTISE
Every Arrhenius acid (or base) can also be classified as a Brønsted–Lowry acid (or base). Every Brønsted–Lowry acid (or base) can also be classified as a Lewis acid (or base). This logic does not always work the other way (for example, NH3 is a Brønsted–Lowry base, but not an Arrhenius base).
Brønsted–Lowry acids and bases always occur in pairs because the definitions require the transfer of a proton from the acid to the base. These are conjugate acid–base pairs, as described in the next section. For example, in the autoionization of water, H3O+ is the conjugate acid and OH− is the conjugate base, as shown in Figure 10.1.
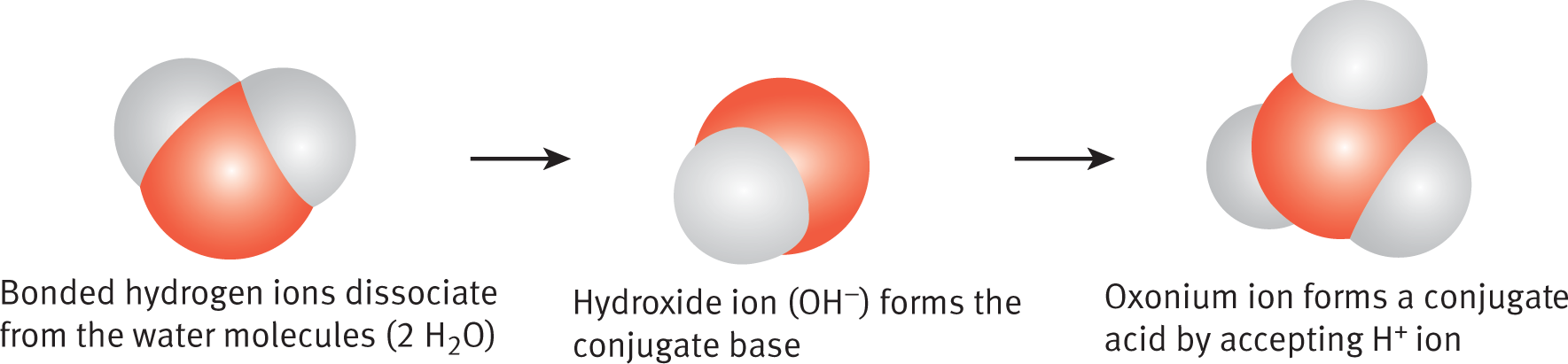
Figure 10.1. Autoionization of Water into Its Conjugate Acid and Conjugate Base The hydroxide ion is the conjugate base; the oxonium (hydronium) ion is the conjugate acid. H2O (l) + H2O (l) ⇌ H3O+ (aq) + OH− (aq)
Lewis
At approximately the same time as Brønsted and Lowry’s publications, Gilbert Lewis also proposed a definition for acids and bases. A Lewis acid is defined as an electron pair acceptor, and a Lewis base is defined as an electron pair donor, as shown in Figure 10.2. The electron pair being donated is a lone pair and is not involved in any other bonds.
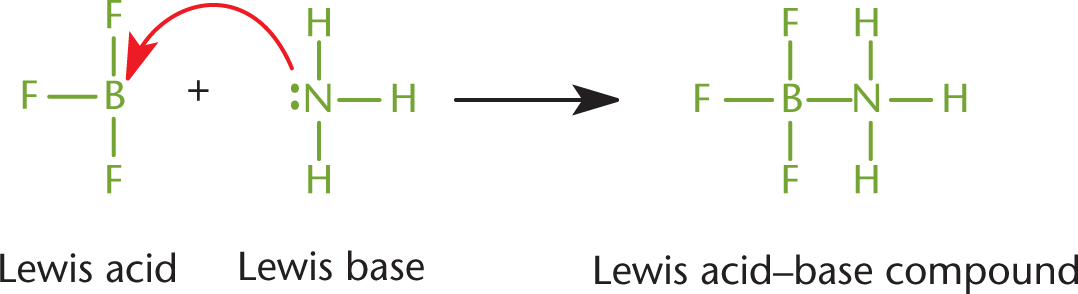
Figure 10.2. Lewis Acid–Base Chemistry Boron trifluoride serves as the Lewis acid, accepting a lone pair. Ammonia serves as a Lewis base, donating a lone pair.
MNEMONIC
The Brønsted-Lowry definition revolves around protons; the Lewis definition around electrons.
On the MCAT, Lewis acid–base chemistry appears with many names. The underlying idea is that one species pushes a lone pair to form a bond with another. This same chemistry can be called coordinate covalent bond formation (discussed in Chapter 3 of MCAT General Chemistry Review), complex ion formation (discussed in Chapter 9 of MCAT General Chemistry Review), or nucleophile–electrophile interactions (discussed in Chapter 4 of MCAT Organic Chemistry Review).
There is an intuitive approach to understanding the differences in the definitions we have discussed so far. The Lewis definition relies on a behavior that is not vastly different from the Brønsted–Lowry interactions—the only difference is the focus. For Brønsted–Lowry acids and bases, we follow the exchange of the hydrogen ion (H+), which is essentially a naked proton. In the Lewis definition, the focus of the reaction is no longer on the proton, but instead the electrons forming the coordinate covalent bond. This difference can be seen using curved arrows, as shown in Figure 10.3.
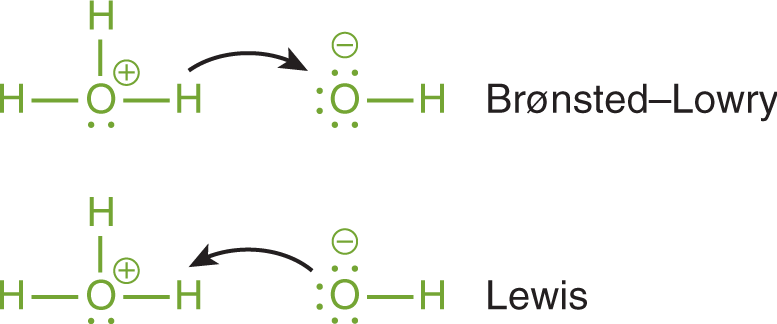
Figure 10.3. Comparison of Brønsted–Lowry and Lewis Definitions of Acids and Bases In the Brønsted–Lowry definition, the focus is on the transfer of the proton. In the Lewis definition, the focus is on the attack of the Lewis acid (electrophile) by the lone pair of the Lewis base (nucleophile).
Note that the Lewis definition is the most inclusive: every Arrhenius acid is also a Brønsted–Lowry acid, and every Brønsted–Lowry acid is also a Lewis acid (and likewise for bases). However, the converse is not necessarily true. The Lewis definition encompasses some species not included within the Brønsted–Lowry definition; for example, BF3 and AlCl3 are species that can each accept an electron pair, which qualifies them as Lewis acids, but they lack a hydrogen ion to donate, disqualifying them as both Arrhenius and Brønsted–Lowry acids.
On the MCAT, you may encounter Lewis acids in the context of organic chemistry reactions because Lewis acids are often used as catalysts.
Amphoteric Species
An amphoteric species is one that reacts like an acid in a basic environment and like a base in an acidic environment. In the Brønsted–Lowry sense, an amphoteric species can either gain or lose a proton, making it amphiprotic as well. On the MCAT, water is the most common example. When water reacts with a base, it behaves as an acid:
H2O + B− ⇌ HB + OH−
When water reacts with an acid, it behaves as a base:
HA + H2O ⇌ H3O+ + A−
KEY CONCEPT
Water, amino acids, and partially deprotonated polyprotic acids such as bicarbonate and bisulfate are common examples of amphoteric and amphiprotic substances. Metal oxides and hydroxides are also considered amphoteric but not necessarily amphiprotic because they do not give off protons.
The partially dissociated conjugate base of a polyvalent acid is usually amphoteric. For example, HSO4− can either gain a proton to form H2SO4 or lose a proton to form SO42−. The hydroxides of certain metals (such as Al, Zn, Pb, and Cr) are also amphoteric. Furthermore, species that can act as both oxidizing and reducing agents are often considered to be amphoteric as well because by accepting or donating electron pairs, they act as Lewis acids or bases, respectively.
Complex amphoteric molecules include amino acids that have a zwitterion intermediate with both cationic and anionic character, as shown in Figure 10.4. Such species are discussed in great detail in Chapter 1 of MCAT Biochemistry Review.

Figure 10.4. Amino Acid Zwitterions Are Complex Amphoteric Species The amino group can release a proton (acid) and the carboxylate group can accept a proton (base).
Acid–Base Nomenclature
The names of most acids are related to the names of their parent anions (the anion that combines with H+ to form the acid). Acids formed from anions with names that end in –ide have the prefix hydro– and the ending –ic.
F− Fluoride HF Hydrofluoric acid
Cl− Chloride HCl Hydrochloric acid
Br− Bromide HBr Hydrobromic acid
Acids formed from oxyanions are called oxyacids. If the anion ends in –ite (less oxygen), then the acid will end with –ous acid. If the anion ends in –ate (more oxygen), then the acid will end with –ic acid. Prefixes in the names of the anions are retained. Some common examples include the following:
ClO− Hypochlorite HClO Hypochlorous acid
ClO2− Chlorite HClO2 Chlorous acid
ClO3− Chlorate HClO3 Chloric acid
ClO4− Perchlorate HClO4 Perchloric acid
NO2− Nitrite HNO2 Nitrous acid
NO3− Nitrate HNO3 Nitric acid
CO32− Carbonate H2CO3 Carbonic acid
SO42− Sulfate H2SO4 Sulfuric acid
PO43− Phosphate H3PO4 Phosphoric acid
BO33− Borate H3BO3 Boric acid
CrO42− Chromate H2CrO4 Chromic acid
CH3COO− Acetate CH3COOH Acetic acid
HCOO− Formate HCOOH Formic Acid
H2NCOO− Carbamate H2NCOOH Carbamic Acid
MCAT EXPERTISE
There are some exceptions to the nomenclature rules. For instance, MnO4− is called permanganate even though there are no MnO3− or MnO2− ions.
MCAT CONCEPT CHECK 10.1:
Before you move on, assess your understanding of the material with these questions.
-
Compare and contrast the three definitions for acids and bases:
Definition Acid Base Arrhenius Brønsted–Lowry Lewis
-
Utilizing Arrhenius acid naming trends, predict the acid formula and name for the following anions:
Anion Acid Formula Acid Name MnO4− Titanate (TiO32−) I− IO4−
-
Identify which reactants are amphoteric species in the following reactions. For those species, determine if the compound is also amphiprotic.
Reaction Amphoteric Reactant Amphiprotic? (Y or N)
HCO3− + HBr ⇌ H2CO3 + Br−
3 HCl + Al(OH)3 ⇌ AlCl3 + 3 H2O
2 HBr + ZnO ⇌ ZnBr2 + H2O
10.2 Properties
LEARNING OBJECTIVES
After Chapter 10.2, you will be able to:
- Predict the behavior of an acid or base in water given its Ka or Kb value, respectively
- Apply the mathematical relationships between pH, pOH, and ion concentration
- Recall the mathematical relationship between Ka, Kb, andKw
- Determine concentration of hydrogen ions given molarity and Ka or Kb of a solution
- Identify acids, bases, conjugate acids, and conjugate bases in a reaction: H2O (l) + H2O (l) ⇌ H3O+ (aq) + OH− (aq)
Acids and bases are usually characterized according to their relative tendencies to either donate or accept hydrogen ions. Furthermore, aqueous acid and base solutions can be characterized according to their concentrations of hydrogen and hydroxide ions.
Autoionization of Water and Hydrogen Ion Equilibria
Because many acid–base reactions take place in water—especially on the MCAT—it is very important to understand the behavior of acidic and basic compounds in water. Only then can one fully appreciate the meaning and significance of such terms as strong acid, weak base, or measurements of pH and pOH.
The Acid–Base Behavior of Water
As described above, water is an amphoteric species: in the presence of a base it reacts as an acid, and in the presence of an acid, it reacts as a base. As an amphoteric compound, water can react with itself in a process called autoionization, seen previously in Figure 10.1. The autoionization of water is represented by the equation:
H2O (l) + H2O (l) ⇌ H3O+ (aq) + OH− (aq)
One water molecule donates a hydrogen ion to another water molecule to produce the hydronium ion (H3O+) and the hydroxide ion (OH−). Many general chemistry courses depict the hydrogen ion simply as H+, rather than as H3O+.
This is acceptable for representing the chemistry, but it is important to remember that the proton is never isolated in the solution; it is always attached to water or some other species that has the ability to accept it. Autoionization of water is a reversible reaction; therefore, the expression above is in equilibrium. For pure water at 298 K, the water dissociation constant, Kw, has been experimentally determined:
Kw = [H3O+][OH−] = 10−14 at 25 °C (298 K)
Equation 10.1
Each mole of water that autoionizes produces one mole each of hydrogen (or hydronium) ions and hydroxide ions, so the concentrations of the hydrogen ions and hydroxide ions are always equal in pure water at equilibrium. Thus, the concentration of each of the ions in pure water at equilibrium at 298 K is 10−7 M.
However, the concentrations of the two ions will not always be equal. In fact, they will only be equal when the solution is neutral. Nevertheless, the product of their respective concentrations will always equal 10−14 when the temperature of the solution is 298 K. For example, if a species donates hydrogen ions to pure water, the hydrogen ion concentration will increase, causing the system to shift toward the reactants in the autoionization process. The result is a decrease in the hydroxide ion concentration and a return to the equilibrium state. This is Le Châtelier’s principle in action: the addition of product to a system at equilibrium causes the system to shift away from the products and toward the reactants. The shift away from the products necessarily decreases the hydroxide ion concentration such that the product of the concentrations of the dissolved ions equals Kw. The addition of a species that accepts hydrogen ions results in a decrease in the hydrogen ion concentration and causes the system to shift toward the products, thereby replacing hydrogen ions. This shift necessarily increases the hydroxide ion concentration and returns the system to equilibrium.
Before we introduce the scales used in measuring concentrations of hydrogen ions and hydroxide ions in different acid–base solutions, it is worthwhile to emphasize an important thermodynamic principle regarding the water dissociation constant (Kw) expression. Kw is an equilibrium constant; unless the temperature of the water is changed, the value for Kw cannot be changed. Thus, the product of the concentrations of the hydrogen ions and the hydroxide ions in an aqueous solution at 298 K must always equal 10−14. However, at different temperatures, the value for Kw changes. At temperatures above 298 K, Kw will increase; this is a direct result of the endothermic nature of the autoionization reaction.
MCAT EXPERTISE
The MCAT loves to test this concept: the value of Kw, like any other equilibrium constant, is dependent only on temperature. Therefore, isolated changes in concentration, pressure, or volume will not affect Kw.
pH and pOH Scales
The concentrations of hydrogen ions and hydroxide ions in aqueous solutions can vary significantly, making the range of measurements on a linear scale unmanageable. The concentration scales for acidic and basic solutions are condensed into something more manageable through expression in logarithmic terms. These logarithmic scales are thepH and the pOH scales for the concentrations of hydrogen and hydroxide ions, respectively.
BRIDGE
Logarithmic scales are used to condense very large absolute differences into small scale differences. Remember that sound level (dB) also uses a logarithmic scale, as discussed in Chapter 7 of MCAT Physics and Math Review.
Using a logarithmic scale is not only mathematically convenient, but also useful for calculations. For instance, the reactivity of an acidic solution is not a function of hydrogen ion concentration but instead of the logarithm of the hydrogen ion concentration. pH and pOH are prototypical examples of p scales. A pscale is defined as the negative logarithm of the number of items.
The pH and pOH of a solution are given by:
pH = –log[H + ] = log 1 [ H + ] pOH = –log[OH − ] = log 1 [ OH − ]
Equation 10.2
For pure water at equilibrium and 298 K, the concentration of hydrogen ions equals the concentration of hydroxide ions (10−7 M). Therefore, pure water at 298 K has a pH of 7 and a pOH of 7 (−log 10−7 = 7). If we take the negative logarithm of the entire water dissociation constant expression ([H3O+][OH−] = 10−14), we find:
pH + pOH = 14
Equation 10.3
Equation 10.3 holds true for all aqueous solutions at 298 K. As pH increases, pOH decreases by the same amount. This relationship can be seen in Figure 10.5 below.
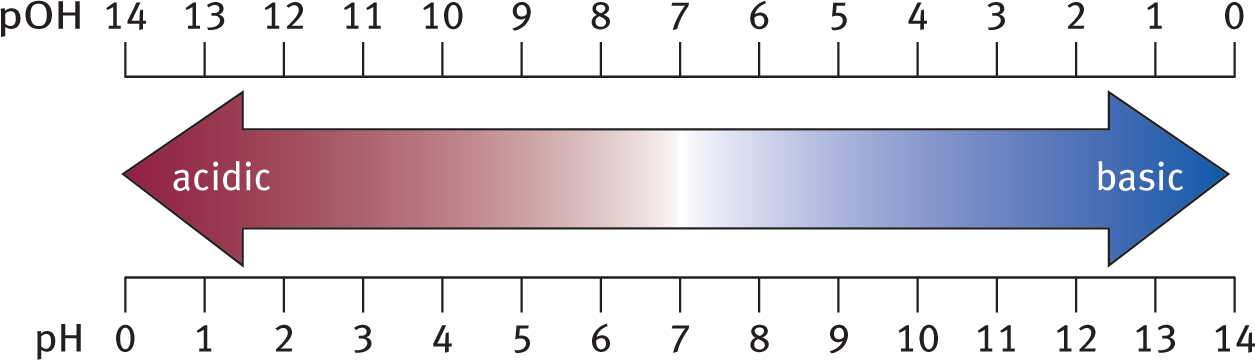
Figure 10.5. pH and pOH Scales pH + pOH = 14 for aqueous solutions at 298 K.
MCAT EXPERTISE
The Kw (like all equilibrium constants) will change if the temperature changes and, in turn, will change the significance of the pH scale. Be careful and read the system conditions given on the MCAT: pH = 7 = neutral is only valid at 25 °C.
For an aqueous solution at 298 K, a pH less than 7 (or pOH greater than 7) indicates a relative excess of hydrogen ions, and the solution is acidic; a pH greater than 7 (or pOH less than 7) indicates a relative excess of hydroxide ions, and the solution is basic. A pH (and pOH) equal to 7 indicates equal concentrations of hydrogen and hydroxide ions, resulting in a neutral solution.
Estimating Scale Values
An essential skill to hone for Test Day is the ability to quickly convert pH, pOH, pKa, and pKb values into nonlogarithmic form and vice-versa.
When the original value is a power of ten, the operation is relatively straightforward: changing the sign on the exponent gives the corresponding p scale value directly. For example, if [H+] = 0.001 or 10−3, then the pH = 3 and pOH = 11. Or, if Kb = 1.0 × 10−12, then pKb = 12.
BRIDGE
In general, math using logarithms frequently appears on the MCAT; make sure to review how these mathematical concepts work in Chapter 10 of MCAT Physics and Math Review. Specifically, the equation pH + pOH = 14 comes from the fact that the log of a product is equal to the sum of logs; that is, log (xy) = log x + log y.
More difficulty arises when the value is not an exact power of ten. Rest assured that the MCAT is not a math test and is not interested in determining your ability to perform complex logarithmic calculations; an exact logarithmic calculation of a number that is not an integer power of ten is unnecessary on the MCAT. The testmakers are interested, however, in testing the ability to apply mathematical concepts appropriately in solving certain problems.
One can obtain a relatively close approximation of a p scale value using the following shortcut: if the nonlogarithmic value is written in proper scientific notation, it will be in the form n × 10−m, where n is a number between 1 and 10. Taking the negative logarithm and simplifying, the p value will be:
– log ( n × 10 − m ) = − log ( n ) − log ( 10 − m ) = m − log ( n )
Because n is a number between 1 and 10, its logarithm will be a decimal between 0 and 1 (log 1 = 0 and log 10 = 1). The closer n is to 1, the closer log n will be to 0; the closer n is to 10, the closer log n will be to 1. As a reasonable approximation, one can say that:
p value ≈ m − 0.n
Equation 10.4
where 0.n represents sliding the decimal point of n one position to the left (dividing n by ten).
Example: If the Ka of an acid is 1.8 × 10−5, then what is its pKa?
Solution: p K a = − log ( 1.8 × 10 − 5 ) = 5 − log 1.8 ≈ 5 − 0.18 = 4.82 ( actual = 4.74 )
MCAT EXPERTISE
Learning how to estimate when using logarithms is an important skill that can save a lot of time on Test Day.
Strong Acids and Bases
Strong acids and bases completely dissociate into their component ions in aqueous solutions. For example, when sodium hydroxide is added to water, the ionic compound dissociates according to the net ionic equation:
NaOH (s) → Na+ (aq) + OH− (aq)
Hence, in a 1 M NaOH solution, complete dissociation yields 1 M Na+ and 1 M OH−. The pH and pOH for this solution can be calculated as follows:
pH = 14 − pOH = 14 − (−log[OH−]) = 14 + log (1 M) = 14 + 0 = 14
MCAT EXPERTISE
Acid–base reactions that consist of a single-headed arrow generally indicate strong acids or bases (complete dissociation with no reversibility).
Virtually no undissociated strong acid or base, such as NaOH, will remain in solution. This is why the dissociation of strong acids and bases is said to go to completion. In the NaOH example above, we assume that the concentration of OH− from the autoionization of water is negligible due to addition of a strong base. The contribution of OH− and H+ ions from the autoionization of water is negligible if the concentration of the acid or base is significantly greater than 10−7 M. On the other hand, if the concentration of acid or base is close to 10−7 M, then the contribution from the autoionization of water is important.
Example: Calculate the pH of a 1 × 10−8 M solution of HCl.
Solution: At first, one may calculate the pH as −log [H+] = −log 10−8 M = 8. However, this answer is not feasible: a pH of 8 cannot describe an acidic solution at 298 K because the presence of the acid will increase the hydrogen ion concentration to above 10−7 M, resulting in an acidic pH below 7.
Recognize that the acid concentration in this question is actually ten times less than the equilibrium concentration of hydrogen ions in pure water generated by the autoionization of water. Consequently, the hydrogen ion concentration from the water itself is significant and cannot be ignored. This can be represented in the equilibrium expression in which x represents the concentration of H3O+ and OH− resulting from the autoionization of water:
Kw = [H3O+][OH−] = [x + 10−8][x] = 10−14
Solving for x (which would require a quadratic equation—math that is beyond the scope of the MCAT) gives x = 9.5 × 10−8 M. The total concentration of hydrogen ions is [H+]total = (9.5 × 10−8) + (1.0 × 10−8) = 1.05 × 10−7 M. Notice that this is extremely close to the concentration of H+ in pure water. The pH of this acidic solution can now be calculated as pH = −log(1.05 × 10−7) ≈ 7 (actual = 6.98). This pH is slightly lower than 7, as expected for a very dilute acidic solution. The point of all of this is: stay alert and keep thinking critically, no matter how familiar the problem setups might seem to you!
Strong acids commonly encountered on the MCAT include HCl (hydrochloric acid), HBr (hydrobromic acid), HI (hydroiodic acid), H2SO4 (sulfuric acid), HNO3 (nitric acid), and HClO4 (perchloric acid). Strong bases commonly encountered include NaOH (sodium hydroxide), KOH (potassium hydroxide), and other soluble hydroxides of Group IA metals. Calculation of the pH and pOH of strong acids and bases assumes complete dissociation of the acid or base in solution.
Example: What is the pH of a solution with [HClO4] = 10 M?
Solution: Because perchloric acid is a strong acid, it will fully dissociate in solution. Therefore, [H+] = 10 M (note that the contribution from the autoionization of water is negligible). pH = −log [H+] = −log 10 M = −1.
This question points out that the pH scale does not “end” at 0 and 14. There can be negative pH values and pH values greater than 14—but this implies a very high concentration of a strong acid or base.
Weak Acids and Bases
Before going any further in our discussion of acids and bases as strong or weak, verify that you are making the distinction between the chemical behavior of an acid or base with respect to its tendency to dissociate (that is, strong bases completely dissociate in aqueous solutions) and the concentrations of acid and base solutions. Although we may casually describe a solution’s concentration as strong or weak, it is preferable to use the terms concentrated and dilute, respectively, because they are unambiguously associated with concentrations, rather than chemical behavior.
Continuing our focus on the chemical behavior of acids and bases, we will now consider those acids and bases that only partially dissociate in aqueous solutions. These are called weak acids and bases. A weak monoprotic acid, HA, will dissociate partially in water to achieve an equilibrium state:
HA (aq) + H2O (l) ⇌ H3O+ (aq) + A− (aq)
Because the system exists in an equilibrium state, we can write the dissociation equation to determine the acid dissociation constant (Ka) as:
K a = [ H 3 O + ] [ A − ] [ HA]
Equation 10.5
The smaller the Ka, the weaker the acid, and consequently, the less it will dissociate. Note that water, as a pure liquid, is not incorporated into the equilibrium expression.
A weak monovalent Arrhenius base, BOH, undergoes dissociation to yield B+ and OH− in solution:
BOH (aq) ⇌ B+ (aq) + OH− (aq)
The base dissociation constant (Kb) can be calculated as:
K b = [ B + ] [ OH − ] [ BOH]
Equation 10.6
The smaller the Kb, the weaker the base, and consequently, the less it will dissociate. As with the acid dissociation expression, water is not included because it is a pure liquid.
Generally speaking, we can characterize a species as a weak acid if its Ka is less than 1.0 and as a weak base if its Kb is less than 1.0. On the MCAT, molecular (nonionic) weak bases are almost exclusively amines.
Conjugate Acid–Base Pairs
Because the Brønsted–Lowry definition of an acid–base reaction is one in which a hydrogen ion (proton) is transferred from an acid to a base, the two always occur in pairs called conjugates. A conjugate acid is the acid formed when a base gains a proton, and a conjugate base is the base formed when an acid loses a proton. For example,
HCO3− (aq) + H2O (l) ⇌ CO32− (aq) + H3O+ (aq)
CO32− is the conjugate base of HCO3−, a weak acid, and H3O+ is the conjugate acid of H2O, a weak base. To find the Ka, we consider the equilibrium concentrations of the dissolved species:
K a = [ CO 3 2 − ] [ H 3 O + ] [ HCO 3 − ]
The reaction between bicarbonate and water is reversible. The reverse reaction would be:
CO32− (aq) + H2O (l) ⇌ HCO3− (aq) + OH− (aq)
We can write the Kb for CO32− as:
K b = [ HCO 3 − ] [ OH − ] [ CO 3 2 − ]
If one adds the previous two reversible reactions, the net reaction is simply the dissociation of water:
HCO 3 − + H 2 O ⇌ CO 3 2 − + H 3 O + CO 3 2 − + H 2 O ⇌ HCO 3 − + OH − 2 H 2 O ⇌ H 3 O + + OH −
Because the net reaction is the autoionization of water, the equilibrium constant for the reaction is Kw = [H3O+][OH−] = 10−14, which is the product of Ka and Kb. Remember: the product of the concentrations of the hydrogen ion and the hydroxide ion must always equal 10−14 for acidic or basic aqueous solutions. Because water is an amphoteric species (both a weak acid and a weak base), all acid–base reactivity in water ultimately reduces to the acid–base behavior of water, and all acidic or basic aqueous solutions are governed by the dissociation constant for water. Thus, if the dissociation constant for one species or its conjugate is known, then the dissociation constant for the other can be determined using the following equations:
K a,acid × K b , conjugate base = K w = 10 − 14 K b,base × K a,conjugate acid = K w = 10 − 14
Equation 10.7
KEY CONCEPT
Be aware of the relationship between conjugate acids and bases because you will need to recognize these entities on the MCAT. Removing a proton from a molecule produces the conjugate base, and adding a proton produces the conjugate acid.
As is evident from these equations, Ka and Kb are inversely related. In other words, if Ka is large, then Kb is small, and vice-versa. By this logic, a strong acid (Ka approaching ∞) will produce a very weak conjugate base (for example, HCl is a strong acid and Cl− is a very weak base), and a strong base will produce a very weak conjugate acid (for example, NaOH is a strong base and H2O is a very weak acid). The conjugate of a strong acid or base is sometimes termed inert because it is almost completely unreactive.
On the other hand, weak acids and bases tend to have conjugates that are also weak. As seen above, CO32− is a weak base; its conjugate acid, HCO3− is a weak acid. As it turns out, for this specific example, the reaction of CO32− with water to produce HCO3− and OH− occurs to a greater extent—is more thermodynamically favorable—than the reaction of HCO3− and water to produce CO32− and H3O+. This fact makes this equilibrium ideal for buffering solutions as part of the bicarbonate buffer system, discussed in Chapter 6 of MCAT Biology Review.
BRIDGE
This thermodynamic preference for the bicarbonate ion intermediate is a major reason why the bicarbonate buffer system in the body is ideal for maintaining a stable pH. The homeostatic mechanisms involved are discussed in Chapter 6 of MCAT Biology Review.
One important theme for acid strength is the effect of induction. Electronegative elements positioned near an acidic proton increase acid strength by pulling electron density out of the bond holding the acidic proton. This weakens proton bonding and facilitates dissociation. Thus, acids that have electronegative elements nearer to acidic hydrogens are stronger than those that do not, as shown in Figure 10.6.
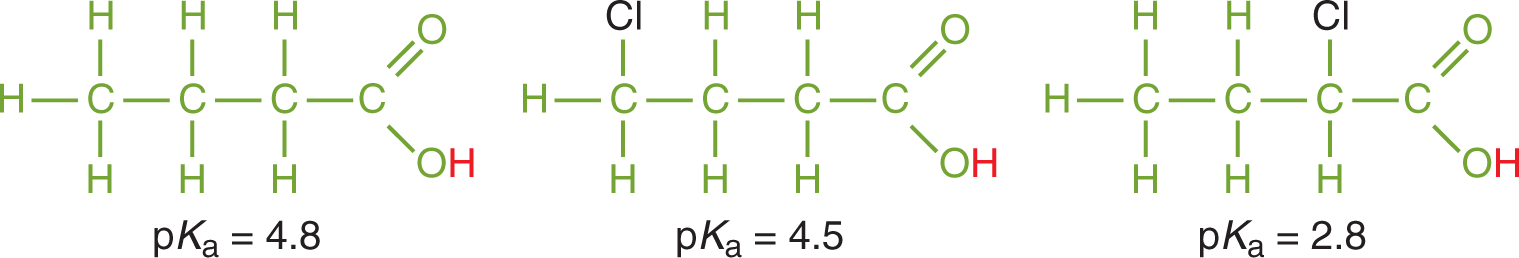
Figure 10.6. Inductive Effects from Electronegative Elements Increase Acidity
Applications of *K*a and *K*b
The most common use of acid and base dissociation constants is to determine the concentration of one of the species in solution at equilibrium. On Test Day, you may be asked to calculate the concentration of the hydrogen ion (or pH), the concentration of the hydroxide ion (or pOH), or the concentration of either the original acid or base.
Example: Calculate the concentration of H3O+ in a 2.0 M aqueous solution of acetic acid, CH3COOH. (Note: Ka = 1.8 × 10−5)
Solution: First, write the equilibrium reaction:
CH3COOH (aq) + H2O (l) ⇌ H3O+ (aq) + CH3COO− (aq)
Next, write the expression for the acid dissociation constant:
K a = [ H 3 O + ] [ CH 3 COO – ] [ CH 3 COOH ] = 1.8 × 10 − 5
Then, recognize that acetic acid is a weak acid, so the concentration of CH3COOH at equilibrium is equal to its initial concentration, 2.0 M, minus the amount dissociated, x. Likewise, [H3O+] = [CH3COO−] =x because each molecule of CH3COOH dissociates into one H+ ion and one CH3COO− ion. Note that the contribution of H3O+ from water is negligible. Thus, the equation can be rewritten as follows:
K a = [ x ] [ x ] [ 2.0 M − x ] = 1.8 × 10 − 5
Remember that the value of x is generally very small. Therefore, we can approximate that 2.0 M − x ≈ 2.0 M. This is further supported because acetic acid is a weak acid and only slightly dissociates in water. This simplifies the calculations:
K a = [ x ] [ x ] 2.0 M = 1.8 × 10 − 5 x 2 = 3.6 × 10 − 5 = 36 × 10 − 6 x = 6 × 10 − 3 M
x represents the concentration of H3O+; therefore, [H3O+] = 6 × 10−3 M. Note: When required to take the square root, adjust the coefficient as needed to make the power of 10 an even number. This way the square root only requires cutting the power of 10 in half.
In this example, note that x is significantly lower than the initial concentration of acetic acid (2.0 M), which validates the approximation; otherwise, it would have been necessary to solve for x using the quadratic formula. Fortunately, the value of x on Test Day is almost always sufficiently small to make this approximation. A rule of thumb is that the approximation is valid as long as x is less than 5 percent of the initial concentration. This typically occurs when Ka is at least 100 times smaller than the concentration of the starting solution. For example, if Ka is 10−4 and the concentration of the starting solution is 0.01 M (10−2 M), then the ratio between the values is 102 or 100. The error in this calculation should be no more than 1 100 = 1 % . On the other hand, if the Ka is 10−3 and the concentration is still 0.01 M, then the ratio between the values becomes 10, which could lead to 1 10 = 10 % error. This degree of error may not be useful when identifying an answer choice on the MCAT.
Students often feel nervous making the assumption that x is negligible because they want to see precise answer choices. However, keep in mind that the MCAT quite deliberately tests students’ ability to make reasonable assumptions under timed conditions to arrive at a feasible answer choice. Part of the skill of taking the MCAT is rounding appropriately to simplify math.
Salt Formation
Acids and bases may react with each other to form a salt and often (but not always) water, in what is termed a neutralization reaction. For example,
HA (aq) + BOH (aq) → BA (s) + H2O (l)
BRIDGE
Remember the reaction types discussed in Chapter 4 of MCAT General Chemistry Review? Go back and review the section on neutralization reactions if this equation doesn’t look familiar to you.
The salt may precipitate out or remain ionized in solution, depending on its solubility and the amount produced. In general, neutralization reactions go to completion. The reverse reaction, in which the salt ions react with water to give back the acid or base, is known as hydrolysis.
Four combinations of strong and weak acids and bases are possible:
- Strong acid + strong base: HCl + NaOH → NaCl + H2O
- Strong acid + weak base: HCl + NH3 → NH4Cl
- Weak acid + strong base: HClO + NaOH → NaClO + H2O
- Weak acid + weak base: HClO + NH3 → NH4ClO
The products of a reaction between equal concentrations of a strong acid and a strong base are equimolar amounts of salt and water. The acid and base neutralize each other, so the resulting solution is neutral (pH = 7), and the ions formed in the reaction will not react with water because they are inert conjugates.
The product of a reaction between a strong acid and a weak base is also a salt, but often no water will be formed because weak bases are often not hydroxides. In this case, the cation of the salt is a weak acid and will react with the water solvent, re-forming some of the weak base through hydrolysis. For example:
- Reaction I: HCl (aq) + NH3 (aq) → NH4+ (aq) + Cl− (aq)
- Reaction II: NH4+ (aq) + H2O (l) → NH3 (aq) + H3O+ (aq)
NH4+ is the conjugate acid of a weak base (NH3) and is stronger than the conjugate base (Cl−) of the strong acid, HCl. NH4+ will then transfer a proton to H2O to form the hydronium ion. The increase in the concentration of the hydronium ion causes the system to shift away from autoionization, thereby reducing the concentration of hydroxide ion. Consequently, the concentration of the hydronium ion will be greater than that of the hydroxide ion at equilibrium, and as a result, the pH of the solution will fall below 7. This should make sense: a strong acid and a weak base produce a slightly acidic solution.
On the other hand, when a weak acid reacts with a strong base, the pH of the solution at equilibrium will be within the basic range because the salt hydrolyzes, with concurrent formation of hydroxide ions. The increase in hydroxide ion concentration will cause the system to shift away from autoionization, thereby reducing the concentration of the hydronium ion. Consequently, the concentration of the hydroxide ion will be greater than that of the hydronium ion at equilibrium, and as a result, the pH of the solution will rise above 7. Consider the reaction of acetic acid, CH3COOH (weak acid) with sodium hydroxide, NaOH (strong base):
- Reaction I: CH3COOH (aq) + NaOH (aq) → Na+ (aq) + CH3COO− (aq) + H2O (l)
- Reaction II: CH3COO− (aq) + H2O (l) → CH3COOH (aq) + OH− (aq)
The pH of a solution containing a weak acid and a weak base depends on the relative strengths of the reactants. For example, the weak acid HClO has a Ka of 3.2 × 10−8, and the weak base NH3 has a Kb = 1.8 × 10−5. Thus, an aqueous solution of HClO and NH3 is basic because the Ka for HClO is less than the Kb for NH3. That is, HClO is weaker as an acid than NH3 is as a base. At equilibrium, therefore, the concentration of hydroxide ions will be greater than the concentration of hydronium ions in the aqueous solution.
In biology and biochemistry, neutralization reactions are often condensation reactions because they form bonds with a small molecule as a byproduct (usually water). The peptide bonds in proteins, for example, are created from the reaction of a carboxyl group (acid) and an amino group (base), while forming a water molecule, as shown in Figure 10.7. The salt in this reaction is the polypeptide itself; breaking it apart requires hydrolysis.
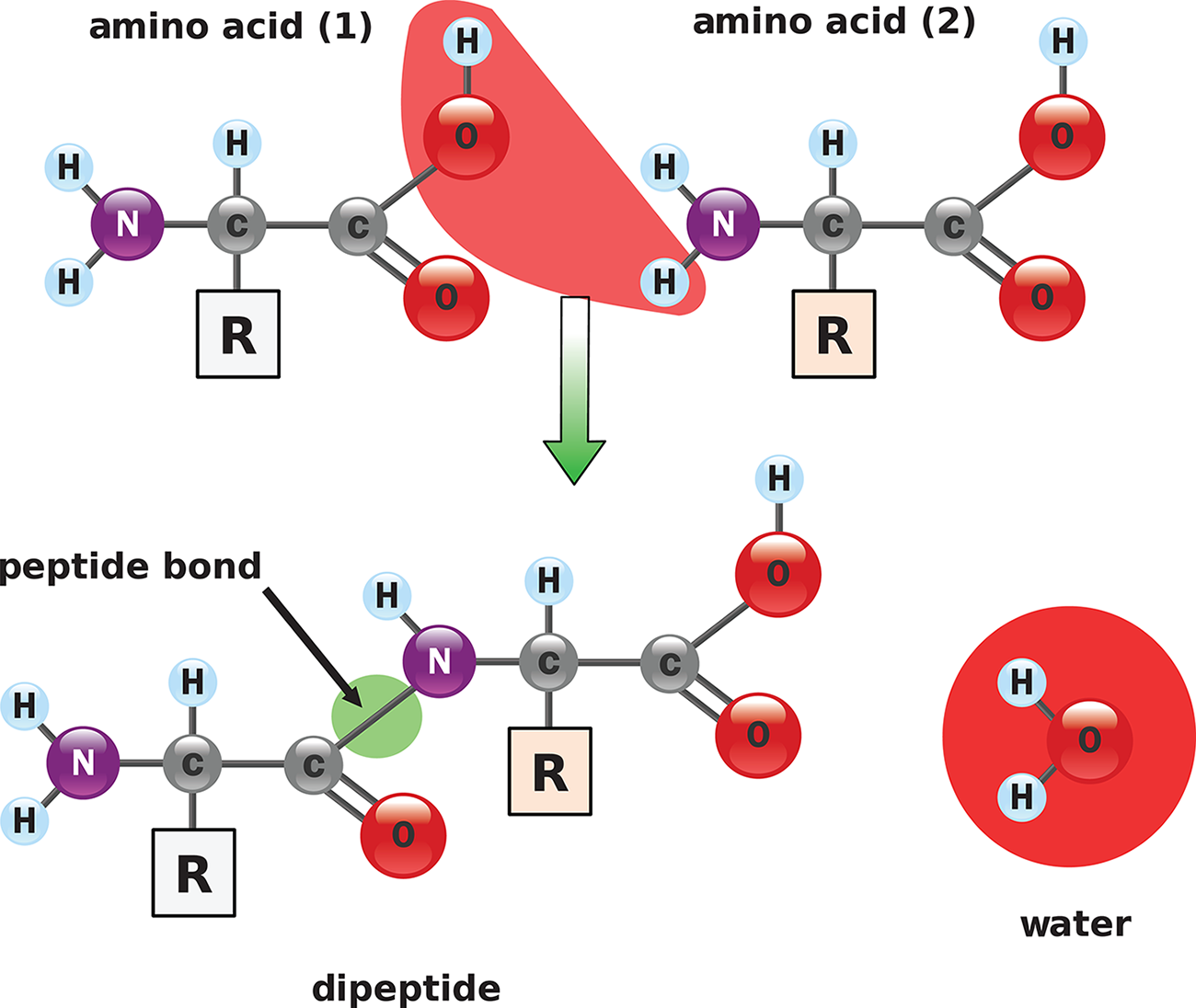
Figure 10.7. Peptide Bond Formation An acidic carboxyl group reacts with a basic amino group.
MCAT CONCEPT CHECK 10.2:
Before you move on, assess your understanding of the material with these questions.
-
What is an amphoteric species?
_____________________________
-
If a compound has a Ka value » water, what does it mean about its behavior in solution? How does this compare with a solution that has only a slightly higher Ka than water?
_____________________________
-
If a compound has a Kb value » water, what does it mean about its behavior in solution? How does this compare with a solution that has only a slightly higher Kb than water?
_____________________________
-
Complete the blank cells in the table by utilizing the mathematical relationships between pH, pOH, and ion concentrations. (Note: Round the numbers given and use logarithmic approximations to determine p values, without a calculator, to simulate Test Day math.)
pH [H3O+] pOH [OH−] Acid or Base?
4
8.89 × 10−4 M
5.19
1.88 × 10−6 M
-
What is the mathematical relationship between Ka, Kb, and Kw?
_____________________________
-
Identify the conjugate acid–base pairings in the reactions below:
Reaction Acid Base Conjugate Acid Conjugate Base
H2CO3 + H2O ⇌ HCO3− + H3O+
H2PO4− + H2O ⇌ H3PO4 + OH−
-
Determine the concentration of hydrogen ions and pH of a solution of 0.2 M acetic acid (Ka = 1.8 × 10−5).
____________________________
10.3 Polyvalence and Normality
LEARNING OBJECTIVES
After Chapter 10.3, you will be able to:
- Describe how equivalents of acid or base are calculated
- Calculate the normality of a solution given its formula and molarity
The relative acidity or basicity of an aqueous solution is determined by the relative concentrations of acid and base equivalents. An acid equivalent is equal to one mole of H+ (or, more properly, H3O+) ions; a base equivalent is equal to one mole of OH− ions. Some acids and bases are polyvalent; that is, each mole of the acid or base liberates more than one acid or base equivalent. Under the Brønsted–Lowry definition, such acids or bases could also be termed polyprotic. For example, the divalent diprotic acid H2SO4 undergoes the following dissociation in water:
H 2 SO 4 ( a q ) + H 2 O ( l ) → H 3 O + ( a q ) + HSO 4 − ( a q ) HSO 4 − ( a q ) + H 2 O ( l ) ⇌ H 3 O + ( a q ) + SO 4 2 − ( a q )
One mole of H2SO4 produces two acid equivalents (2 moles of H3O+). Notice that the first dissociation goes to completion, but the second dissociation reaches an equilibrium state. The acidity or basicity of a solution depends on the concentration of acidic or basic equivalents that can be liberated. The quantity of acidic or basic capacity is directly indicated by the solution’s normality, described in Chapter 9 of MCAT General Chemistry Review. For example, each mole of H3PO4 yields three moles (equivalents) of H3O+. Therefore, a 2 M H3PO4 solution would be 6 N.
Another measurement useful for acid–base chemistry is gram equivalent weight. Chapter 4 of MCAT General Chemistry Review defined and discussed this term extensively. The gram equivalent weight is the mass of a compound that produces one equivalent (one mole of charge). For example, H2SO4 (molar mass: 98 g mol ) is a divalent acid, so each mole of the acid compound yields two acid equivalents. The gram equivalent weight is 98 ÷ 2 = 49 grams. That is, the complete dissociation of 49 grams of H2SO4 will yield one acid equivalent (one mole of H3O+). Common polyvalent acids include H2SO4, H3PO4, and H2CO3. Common polyvalent bases include Al(OH)3, Ca(OH)2, and Mg(OH)2.
BRIDGE
To review normality in more detail, revisit the calculations performed in Chapter 4 of MCAT General Chemistry Review. These are critical calculations for polyvalent acids and bases.
MCAT CONCEPT CHECK 10.3:
Before you move on, assess your understanding of the material with these questions.
-
What species are considered the equivalents for acids and bases, respectively?
- Acids:____________________________
- Bases:____________________________
-
Calculate the normality of the following solutions:
- 2 M Al(OH)3:___________________
- 16 M H2SO4:_____________
10.4 Titration and Buffers
LEARNING OBJECTIVES
After Chapter 10.4, you will be able to:
- Select an appropriate indicator for a given acid–base reaction
- Explain the purpose of a buffer solution
- Identify the pH range of the equivalence point for different combinations of acids and bases, for example, weak acid + weak base
- Calculate the pH or pOH of a known solution
- Identify the buffering region, half-equivalence point, equivalence point, and endpoint of a titration reaction:
Titration is a procedure used to determine the concentration of a known reactant in a solution. There are different types of titrations, including acid–base, oxidation–reduction, and complexometric (metal ion). The MCAT frequently tests the first two types. Complexometric (metal ion) titrations are outside the scope of the MCAT but focus on formation of complex ions, as described in Chapter 9 of MCAT General Chemistry Review.
General Principles
Titrations are performed by adding small volumes of a solution of known concentration (the titrant) to a known volume of a solution of unknown concentration (the titrand) until completion of the reaction is achieved at the equivalence point.
Acid–Base Equivalence Points
In acid–base titrations, the equivalence point is reached when the number of acid equivalents present in the original solution equals the number of base equivalents added, or vice-versa. It is important to emphasize that, while a strong acid/strong base titration will have its equivalence point at a pH of 7, the equivalence point doesnot always occur at pH 7. When titrating polyprotic acids or bases (discussed later in this chapter), there are multiple equivalence points, as each acidic or basic conjugate species is titrated separately. This is shown in the speciation plot in Figure 10.8.
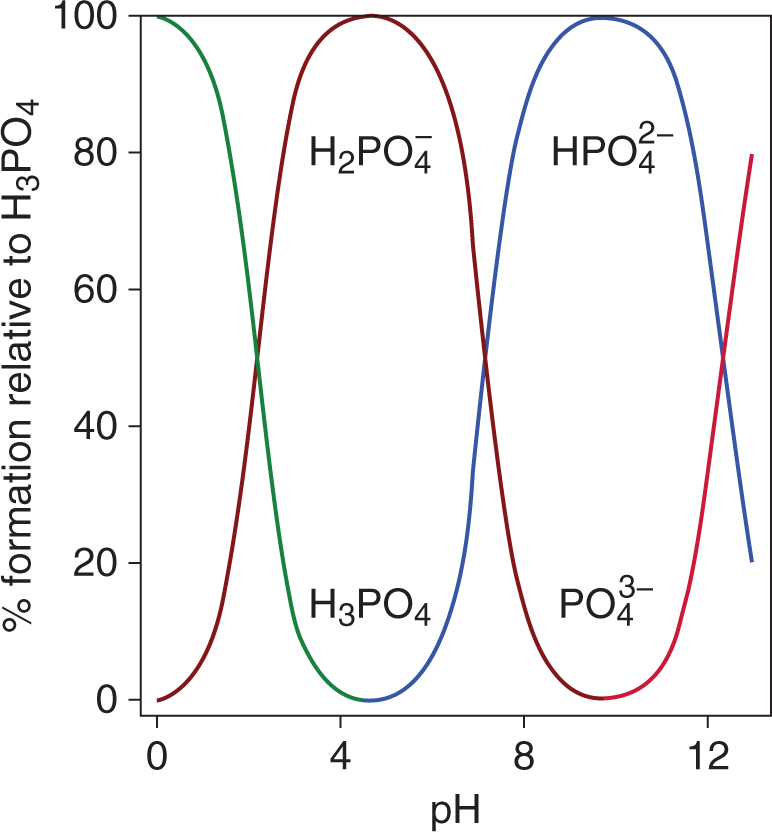
Figure 10.8. Speciation Plot of Phosphoric Acid At any given pH, only two forms of the acid exist in solution; thus, each conjugate is titrated separately.
At the equivalence point, the number of equivalents of acid and base are equal. This fact allows us to calculate the unknown concentration of the titrand through the equation:
NaVa = NbVb
Equation 10.8
where Na and Nb are the acid and base normalities, respectively, and Va and Vb are the volumes of acid and base solutions, respectively. Note that, as long as both volumes use the same units, the units used do not have to be liters.
The equivalence point in an acid–base titration is determined in two common ways: evaluated by using a graphical method, plotting the pH of the unknown solution as a function of added titrant by using a pH meter, or estimated by watching for a color change of an added indicator.
Indicators
Indicators are weak organic acids or bases that have different colors in their protonated and deprotonated states. This small structural change—the binding or release of a proton—leads to a change in the absorption spectrum of the molecule, which we perceive as a color change. Indicators are generally vibrant and can be used in low concentrations without significantly altering the equivalence point. The indicator must always be a weaker acid or base than the acid or base being titrated; otherwise, the indicator would be titrated first! The point at which the indicator changes to its final color is not the equivalence point but rather the endpoint. If the indicator is chosen correctly and the titration is performed well, the volume difference between the endpoint and the equivalence point is negligible and may be corrected for or simply ignored.
KEY CONCEPT
Indicators change color as they shift between their conjugate acid and base forms:
H − Indicator ( color 1 ) ⇌ H + + Indicator ( color 2 ) −
Because this is an equilibrium process, we can apply Le Châtelier’s principle. Adding H+ shifts the equilibrium to the left. Adding OH− removes H+ and therefore shifts the equilibrium to the right.
Acid–base titrations can be performed for different combinations of strong and weak acids and bases. The most useful combinations involve at least one strong species. Weak acid/weak base titrations can be done but are not very accurate and therefore are rarely performed. The pH curve for the titration of a weak acid and weak base lacks the sharp change that normally indicates the equivalence point. Furthermore, indicators are less useful because the pH change is far more gradual.
MCAT EXPERTISE
Any question involving the selection of an ideal indicator will require you to know what the pH of the reaction at the equivalence point will be, whether graphically or mathematically. Once you have determined where the equivalence point is, select the indicator that has the closest pKa value to it.
Strong Acid and Strong Base
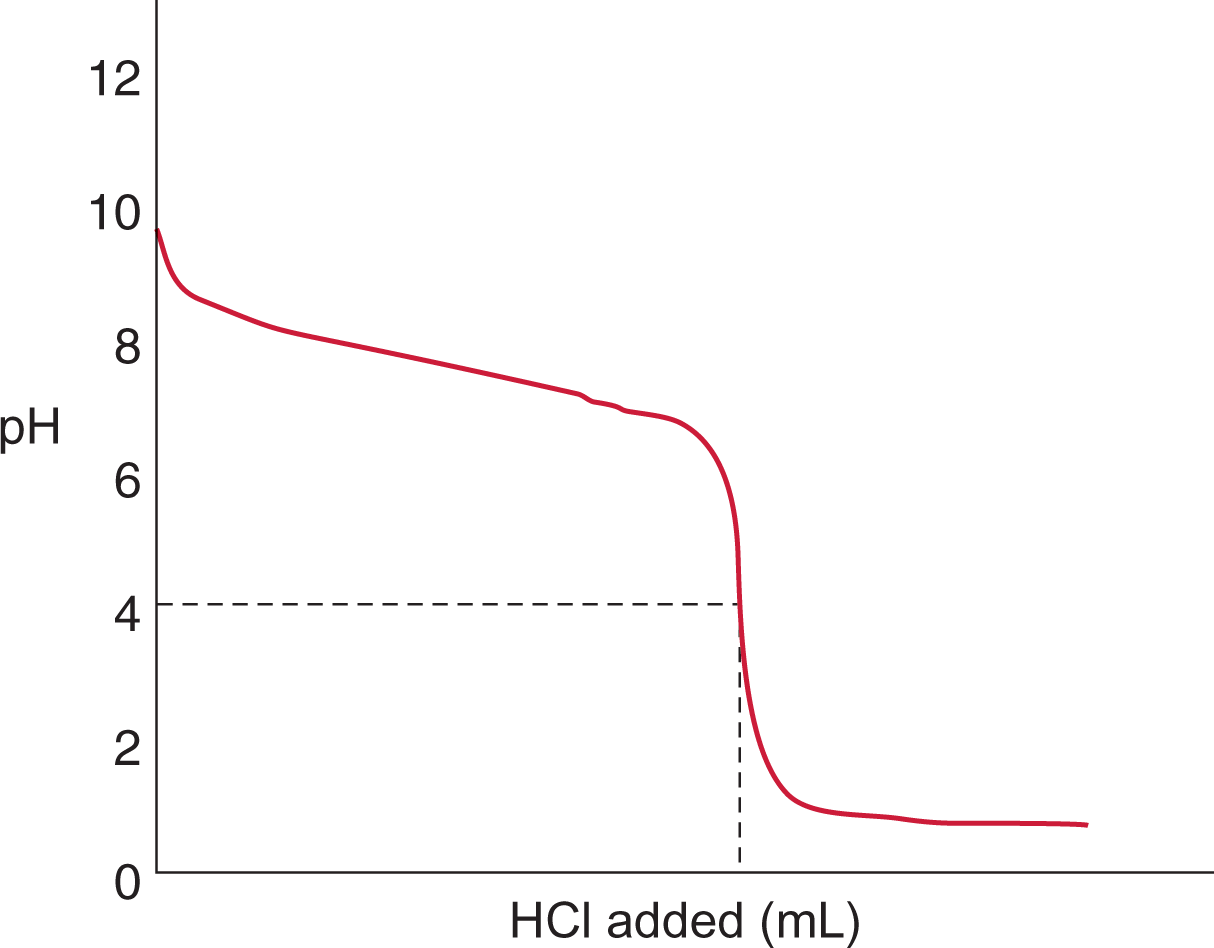
Let’s consider the titration of 10 mL of a 0.1 N solution of HCl with a 0.1 N solution of NaOH. Plotting the pH of the solution vs. the quantity of NaOH added gives the curve shown in Figure 10.9.
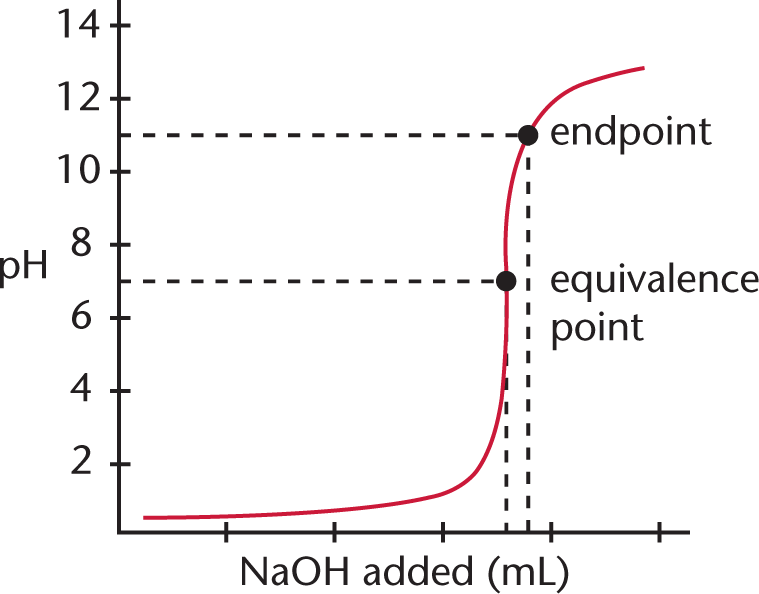
Figure 10.9. Monoprotic Strong Acid and Strong Base Titration Curve A strong base, NaOH, is titrated into a solution of strong acid, HCl, to yield an equivalence point of pH = 7.
KEY CONCEPT
Compare the relative strength of the two solutions in a titration to determine if the pH of the equivalence point is less than, equal to, or greater than 7:
- Strong acid + weak base: equivalence point pH < 7
- Strong acid + strong base: equivalence point pH = 7
- Weak acid + strong base: equivalence point pH > 7
Because HCl is a strong acid and NaOH is a strong base, the equivalence point of the titration will be at pH 7, and the solution will be neutral. Note that the endpoint shown is close to, but not exactly equal to, the equivalence point; selection of a better indicator, one that changes colors at, say, pH 8, would have given a better approximation. Still, the amount of error introduced by the use of an indicator that changes color around pH 11 rather than pH 8 is not especially significant; it represents a mere fraction of a milliliter of excess NaOH solution.
In the early part of the curve when little base has been added, the acidic species predominates, so the addition of small amounts of base will not appreciably change either the [OH−] or the pH. Similarly, in the last part of the titration curve when an excess of base has been added, the addition of small amounts of base will not change the [OH−] significantly, and the pH will remain relatively constant. The addition of base will alter the concentrations of H+ and OH− near the equivalence point, and will elicit the most substantial changes in pH in that region. Remember: the equivalence point for strong acid/strong base titrations is always at pH 7 (for monovalent species).
If one uses a pH meter to chart the change in pH as a function of volume of titrant added, a good approximation can be made of the equivalence point by locating the midpoint of the region of the curve with the steepest slope.
Weak Acid and Strong Base
Titration of a weak acid, such as CH3COOH, with a strong base, such as NaOH, produces the titration curve shown in Figure 10.10.
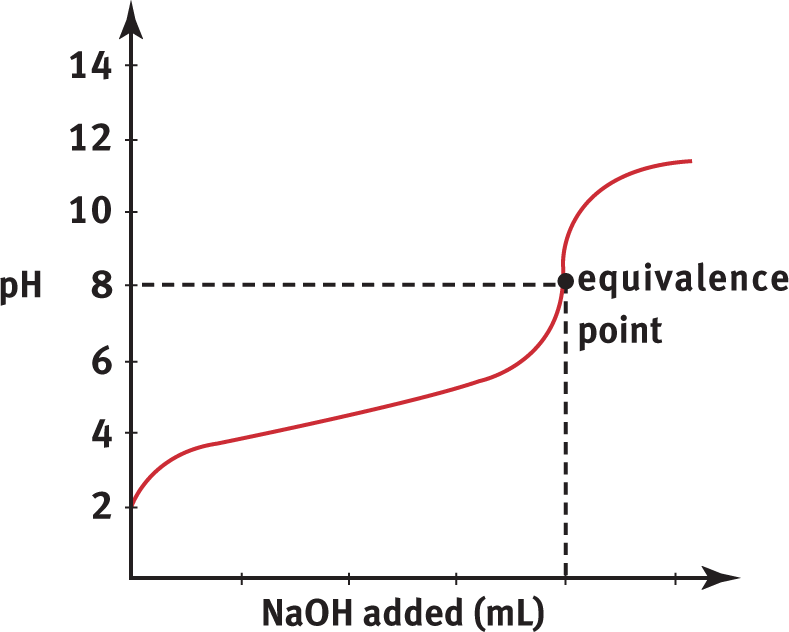
Figure 10.10. Weak Acid and Strong Base Titration Curve A strong base, NaOH, is titrated into a solution of weak acid, CH3COOH, to yield an equivalence point of pH > 7.
Compare Figure 10.10 with the curve in Figure 10.9. The first difference is that the initial pH of the weak acid solution is greater than the initial pH of the strong acid solution. Weak acids do not dissociate to the same degree that strong acids do; therefore, the concentration of H3O+ will generally be lower (and pH will be higher) in an equimolar solution of weak acid. The second difference is the shapes of the curves. The pH curve for the strong acid/strong base titration shows a steeper, more sudden rise in pH at the equivalence point. In the weak acid/strong base titration, the pH changes gradually early on in the titration and has a less sudden rise at the equivalence point. The third difference is the position of the equivalence point. While the equivalence point for a strong acid/strong base titration is pH 7, the equivalence point for a weak acid/strong base titration is above 7. This is because the reaction between the weak acid (HA) and strong base (OH−) produces a weak conjugate base (A−) and even weaker conjugate acid (H2O). This produces a greater concentration of hydroxide ions than hydrogen ions at equilibrium (due to the common ion effect on the autoionization of water). Therefore, the equivalence point for weak acid/strong base titration is always in the basic range of the pH scale.
Strong Acid and Weak Base
The appearance of the titration curve for a weak base titrand and strong acid titrant will look like an inversion of the curve for a weak acid titrand and strong base titrant. The initial pH will be in the basic range (typical range: pH 10–12) and will demonstrate a gradual drop in pH with the addition of strong acid. The equivalence point will be in the acidic pH range because the reaction between the weak base and strong acid will produce a weak conjugate acid and even weaker conjugate base, as shown in Figure 10.11. The stronger conjugate acid will result in an equilibrium state with a concentration of hydrogen ions greater than that of the hydroxide ions. Therefore, the equivalence point for a weak base/strong acid titration is always in the acidic range of the pH scale.
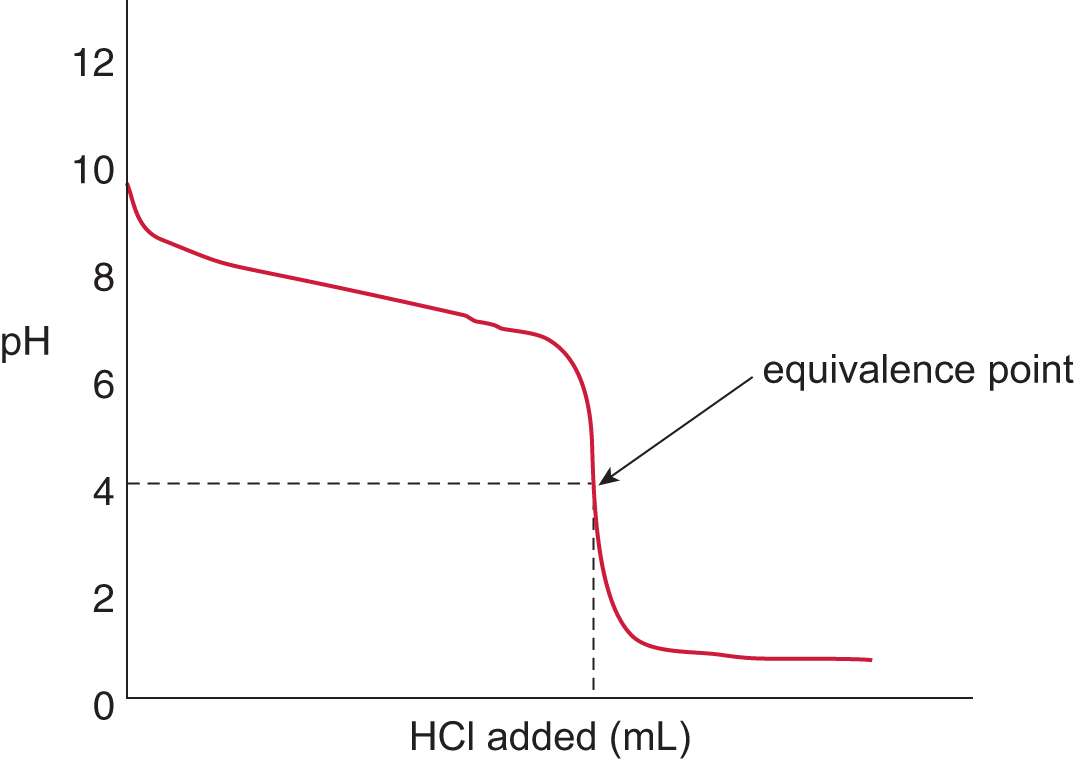
Figure 10.11. Strong Acid and Weak Base Titration Curve A strong acid, HCl, is titrated into a solution of weak base, NH3, to yield an equivalence point of pH < 7.
Weak Acid and Weak Base
The appearance of the titration curve for a weak base titrand and weak acid titrant will look like an intermediate of the previous types discussed. Because both the titrant and the titrand are weak, the initial pH is generally in the 3–11 range and will demonstrate a very shallow drop at the equivalence point. The equivalence point will be near neutral pH because the reaction is partially dissociative for both species.
MCAT EXPERTISE
To identify which type of titration is being shown in a graph, identify the starting position in the graph (pH ≫ 7 = titrand is a strong base, > 7 (slightly) = weak base, < 7 (slightly) = weak acid, and ≪ 7 pH = strong acid), and determine where the equivalence point is. Think of titrations like tug-of-war: the stronger the acid or base, the more it pulls the equivalence point into its pH territory.
Polyvalent Acids and Bases
The titration curve for a polyvalent acid or base looks different from that for a monovalent acid or base. Figure 10.12 shows the titration of Na2CO3 with HCl, in which the divalent (diprotic) acid H2CO3 is the ultimate product.
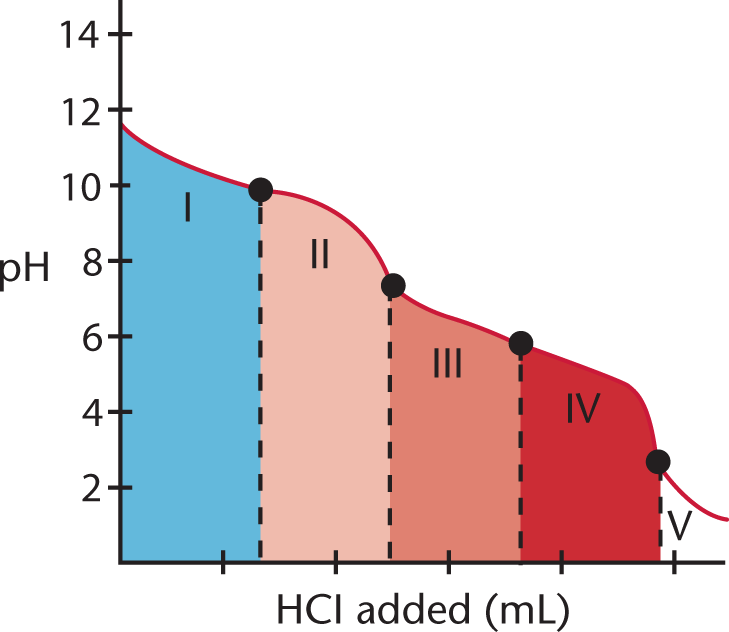
Figure 10.12. Polyvalent Titration The multiple equivalence points indicate that this is a polyvalent titration.
In region I, little acid has been added, and the predominant species is CO32−. In region II, more acid has been added, and the predominant species are CO32− and HCO3−, in relatively equal concentrations. The flat part of the curve is the first buffer region (discussed in the next section), corresponding to the pKa of HCO3− (Ka = 5.6 × 10−11; pKa = 10.25). The center of the buffer region (the point between regions I and II) is sometimes termed the half-equivalence point because it occurs when half of a given species has been protonated (or deprotonated).
Region III begins with the equivalence point, at which all of the CO32− is finally titrated to HCO3−. As the curve illustrates, a rapid change in pH occurs at the equivalence point (the point between regions II and III). In the latter part of region III, the predominant species is HCO3−, although some H2CO3 has formed as well.
At the beginning of region IV, the acid has neutralized approximately half of the HCO3−, and now H2CO3 and HCO3− are in roughly equal concentrations. This flat region is the second buffer region (and second half-equivalence point, between regions III and IV) of the titration curve, corresponding to the pKa of H2CO3 (Ka = 4.3 × 10−7; pKa = 6.37). Region V starts with the second equivalence point, as all of the HCO3− is finally converted to H2CO3. Again, a rapid change in pH is observed near the equivalence point (the point between regions IV and V) as acid is added.
The titrations of the acidic and basic amino acids (which have acidic or basic side chains, respectively) will show curves similar to the one shown in Figure 10.12. But rather than two equivalence points, there will in fact be three: one corresponding to the titration of the carboxyl group and a second corresponding to the titration of the amino group, both of which are attached to the central carbon, as well as a third corresponding to either the acidic or basic side chain.
Buffers
A buffer solution consists of a mixture of a weak acid and its salt (which is composed of its conjugate base and a cation) or a mixture of a weak base and its salt (which is composed of its conjugate acid and an anion). Two examples of buffers that are common in the laboratory—and commonly tested on the MCAT—are a solution of acetic acid (CH3COOH) and its salt, sodium acetate (CH3COO−Na+), and a solution of ammonia (NH3) and its salt, ammonium chloride (NH4+Cl−). The acetic acid/sodium acetate solution is considered an acid buffer, and the ammonium chloride/ammonia solution is a base buffer. Buffer solutions have the useful property of resisting changes in pH when small amounts of acid or base are added. Consider a buffer solution of acetic acid and sodium acetate: (Note: The sodium ion has not been included because it is not involved in the acid–base reaction.)
CH3COOH (aq) + H2O (l) ⇌ H3O+ (aq) + CH3COO− (aq)
When a small amount of strong base, such as NaOH, is added to the buffer, the OH− ions from the NaOH react with the H3O+ ions present in the solution; subsequently, more acetic acid dissociates (the system shifts to the right), restoring the [H3O+]. The weak acid component of the buffer thereby serves to neutralize the strong base that has been added. The resulting increase in the concentration of the acetate ion (the conjugate base) does not create nearly as large an increase in hydroxide ions as the unbuffered NaOH would. Thus, the addition of the strong base does not result in a significant increase in [OH−] and does not appreciably change the pH.
Likewise, when a small amount of HCl is added to the buffer, H+ ions from the HCl react with the acetate ions to form acetic acid. Acetic acid is weaker than the added hydrochloric acid (which has been neutralized by the acetate ions), so the increased concentration of acetic acid does not significantly contribute to the hydrogen ion concentration in the solution. Because the buffer maintains [H+] at approximately constant values, the pH of the solution is relatively unchanged.
The Bicarbonate Buffer System
In the human body, one of the most important buffers is the H2CO3/HCO3− conjugate pair in the plasma component of the blood, called the bicarbonate buffer system. Specifically, carbonic acid (H2CO3) and its conjugate base, bicarbonate (HCO3−), form a weak acid buffer for maintaining the pH of the blood within a fairly narrow physiological range. CO2 (g), one of the waste products of cellular respiration, also has low solubility in aqueous solutions. The majority of the CO2 transported from peripheral tissues to the lungs (where it will be exhaled out) is dissolved in the plasma in a “disguised” form through the bicarbonate buffer system. CO2 (g) and water react in the following manner:
CO2 (g) + H2O (l) ⇌ H2CO3 (aq) ⇌ H+ (aq) + HCO3− (aq)
REAL WORLD
A number of conditions can affect the delicate pH balance of tissues in the body, including chronic obstructive pulmonary disease (COPD), renal tubular acidosis (RTA), diabetic ketoacidosis (DKA), lactic acidosis, metabolic diseases, poisonings and ingestions, and hyperventilation. The buffer system must be well maintained to mitigate these changes.
The bicarbonate buffer system is tied to the respiratory system. In conditions of metabolic acidosis (production of excess plasma H+ not caused by the respiratory system itself), the breathing rate will increase to compensate and blow off a greater amount of carbon dioxide gas; this causes the system to shift to the left, thereby reducing [H+] and buffering against dramatic and dangerous changes to the blood pH. It is interesting to note that the bicarbonate buffer system (pKa = 6.37) maintains a pH around 7.4, which is actually slightly outside the optimal buffering capacity of the system. Buffers have a narrow range of optimal activity (pKa ± 1). This actually makes sense—it is far more common for acidemia (too much acid in the blood) to occur than alkalemia (too much base in the blood). As acidemia becomes more severe, the buffer system actually becomes more effective and more resistant to further lowering of the pH.
The Henderson–Hasselbalch Equation
The Henderson–Hasselbalch equation is used to estimate the pH or pOH of a buffer solution. For a weak acid buffer solution:
pH = p K a + log [ A − ] [ HA]
Equation 10.9
where [A−] is the concentration of the conjugate base and [HA] is the concentration of the weak acid. Note that when [conjugate base] = [weak acid], the pH = pKa because log (1) = 0. This occurs at the half-equivalence points in a titration, and buffering capacity is optimal at this pH.
Likewise, for a weak base buffer solution:
pOH = p K b + log [ B + ] [ BOH]
Equation 10.10
where [B+] is the concentration of conjugate acid and [BOH] is the concentration of the weak base. Similar to acid buffers, pOH = pKb when [conjugate acid] = [weak base]. Buffering capacity is optimal at this pOH.
The Henderson–Hasselbalch equation is, in reality, just a rearrangement of the acid (or base) dissociation constant:
K a = [ H 3 O + ] [ A – ] [ HA] − log K a = − log [ H 3 O + ] [ A – ] [ HA] − log K a = − log [ H 3 O + ] − log [ A – ] [ HA] p K a = pH − log [ A – ] [ HA] pH = p K a + log [ A – ] [ HA]
One subtlety of buffer systems and Henderson–Hasselbalch calculations that usually goes unnoticed or is misunderstood by students is the effect of changing the concentrations of the conjugate pair but not changing the ratio of their concentrations. Clearly, changing the ratio of the conjugate base to the acid will lead to a change in the pH of the buffer solution. But what about changing the concentrations while maintaining a constant ratio? What would happen if the concentrations of both the acid and its conjugate base were doubled? While the pH would not change, the buffering capacity—the ability to which the system can resist changes in pH—has doubled. In other words, addition of a small amount of acid or base to this system will now cause even less deviation in the pH. As mentioned earlier, the buffering capacity is generally maintained within 1 pH unit of the pKa value.
Example: What is the pH of a solution made from 1 L of 0.05 M acetic acid (CH3COOH, Ka = 1.8 × 10−5) mixed with 500 mL of 1 M acetate (CH3COO−)?
Solution: First, determine the concentrations of acetic acid and acetate in the final solution. Because two solutions were mixed, there will be some dilution of both the acetic acid and acetate.
N i,CH 3 COOH V i,CH 3 COOH = N f,CH 3 COOH V f,CH 3 COOH → N f,CH 3 COOH = N i,CH 3 COOH V i,CH 3 COOH V f,CH 3 COOH N f,CH 3 COOH = ( 0.05 N ) ( 1 L) ( 1.5 L) = 0.033 N N i,CH 3 COO − V i,CH 3 COO − = N f,CH 3 COO − V f,CH 3 COO − → N f,CH 3 COO − = N i,CH 3 COO − V i,CH 3 COO − V f,CH 3 COO − N f,CH 3 COO − = ( 1 N ) ( 0.5 L) ( 1.5 L) = 0.33 N
Then, use the Henderson–Hasselbalch equation.
pH = p K a + log [ A – ] [ HA] = − log ( 1.8 × 10 − 5 ) + log 0.33 N 0.033 N pH ≈ 4.82 + 1 = 5.82 (actual = 5 .74)
MCAT CONCEPT CHECK 10.4:
Before you move on, assess your understanding of the material with these questions.
-
Describe each of the following parts of a titration curve:
- Buffering region:________________________________
- Half-equivalence point:________________________________
- Equivalence point:________________________________
- Endpoint:____________________________
-
For a reaction involving a strong base and a weak acid, which of the following indicators would be best to indicate the endpoint of the titration? (Circle the correct answer.)
- Phenolphthalein (pKa = 9.7)
- Bromothymol blue (pKa = 7.1)
- Bromocresol green (pKa = 4.7)
- Methyl yellow (pKa = 3.3)
-
In which part of the pH range (acidic, basic, or neutral) will the equivalence points fall for each of the following titrations?
- Strong acid + weak base:________________________________
- Strong base + weak acid:________________________________
- Strong acid + strong base:________________________________
- Weak acid + weak base:____________________________
-
What is the purpose of a buffer solution?
_____________________________
-
What are the pH and pOH of a solution containing 5 mL of 5 M benzoic acid (Ka = 6.3 × 10−5) and 100 mL of 0.005 M benzoate solution?
_____________________________
Conclusion
In this chapter, we have reviewed the important principles of acid–base chemistry. We clarified the differences among the three definitions of acids and bases, including the nomenclature of some common Arrhenius acids. We investigated important properties of acids and bases, including the important acid–base behavior of water (autoionization) and hydrogen ion equilibria. We explained the mathematics of the pH and pOH logarithmic scales and demonstrated a useful Test Day shortcut for approximating the logarithmic value of hydrogen ion or hydroxide ion concentrations. Strong acids and bases are defined as compounds that completely dissociate in aqueous solutions, and weak acids and bases are compounds that only partially dissociate (to an equilibrium state). We discussed neutralization and salt formation upon reaction of acids and bases, and finally, we applied our fundamental understanding of acid–base reactivity to titrations and buffer systems. Titrations are useful for determining the concentration of a known acid or base solution. Weak acid and weak base buffers are useful for minimizing changes in pH upon addition of a strong acid or base.
You’ve just accomplished a major task in the overall effort to earn points on Test Day. It’s okay if you didn’t understand everything on this first pass. Go back and review the concepts that were challenging for you and then complete the questions at the end of the chapter and MCAT practice passages to test your knowledge. Don’t be alarmed if you find yourself reviewing parts or all of a chapter a second or third time—repetition is the key to success.
You are now two chapters away from completing this review of general chemistry. While we don’t want to offer our congratulations prematurely, we want to acknowledge all the hard work you’ve invested in this process. Keep it up: success on Test Day is within your reach!
GO ONLINE!
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Definitions
- Arrhenius acids dissociate to produce an excess of hydrogen ions in solution. Arrhenius bases dissociate to produce an excess of hydroxide ions in solution.
- Brønsted–Lowry acids are species that can donate hydrogen ions.Brønsted–Lowry bases are species that can accept hydrogen ions.
- Lewis acids are electron-pair acceptors. Lewis bases are electron-pair donors.
- All Arrhenius acids and bases are Brønsted–Lowry acids and bases, and all Brønsted–Lowry acids and bases are Lewis acids and bases; however, the converse of these statements is not necessarily true (that is, not all Lewis acids and bases are Brønsted–Lowry acids and bases, and not all Brønsted–Lowry acids and bases are Arrhenius acids and bases).
- Amphoteric species are those that can behave as an acid or base. Amphiprotic species are amphoteric species that specifically can behave as a Brønsted–Lowry acid or Brønsted–Lowry base.
- Water is a classic example of an amphoteric, amphiprotic species—it can accept a hydrogen ion to become a hydronium ion, or it can donate a hydrogen ion to become a hydroxide ion.
- Conjugate species of polyvalent acids and bases can also behave as amphoteric and amphiprotic species.
Properties
- The water dissociation constant, Kw, is 10−14 at 298 K. Like other equilibrium constants, Kw is only affected by changes in temperature.
- pH and pOH can be calculated given the concentrations of H3O+ and OH− ions, respectively. In aqueous solutions, pH + pOH = 14 at 298 K.
- Strong acids and bases completely dissociate in solution.
- Weak acids and bases do not completely dissociate in solution and have corresponding dissociation constants (Ka and Kb, respectively).
- In the Brønsted–Lowry definition, acids have conjugate bases that are formed when the acid is deprotonated. Bases have conjugate acids that are formed when the base is protonated.
- Strong acids and bases have very weak (inert) conjugates.
- Weak acids and bases have weak conjugates.
- Neutralization reactions form salts and (sometimes) water.
Polyvalence and Normality
- An equivalent is defined as one mole of the species of interest.
- In acid–base chemistry, normality is the concentration of acid or base equivalents in solution.
- Polyvalent acids and bases are those that can donate or accept multiple electrons. The normality of a solution containing a polyvalent species is the molarity of the acid or base times the number of protons it can donate or accept.
Titration and Buffers
- Titrations are used to determine the concentration of a known reactant in a solution.
- The titrant has a known concentration and is added slowly to the titrand to reach the equivalence point.
- The titrand has an unknown concentration but a known volume.
- The half-equivalence point is the midpoint of the buffering region, in which half of the titrant has been protonated (or deprotonated); thus, [HA] = [A−] and a buffer is formed.
- The equivalence point is indicated by the steepest slope in a titration curve; it is reached when the number of acid equivalents in the original solution equals the number of base equivalents added, or vice-versa.
- Strong acid and strong base titrations have equivalence points at pH = 7.
- Weak acid and strong base titrations have equivalence points at pH > 7.
- Weak base and strong acid titrations have equivalence points at pH < 7.
- Weak acid and weak base titrations can have equivalence points above or below 7, depending on the relative strength of the acid and base.
- Indicators are weak acids or bases that display different colors in their protonated and deprotonated forms.
- The indicator chosen for a titration should have a pKa close to the pH of the expected equivalence point.
- The endpoint of a titration is when the indicator reaches its final color.
- Multiple buffering regions and equivalence points are observed in polyvalent acid and base titrations.
- Buffer solutions consist of a mixture of a weak acid and its conjugate salt or a weak base and its conjugate salt; they resist large fluctuations in pH.
- Buffering capacity refers to the ability of a buffer to resist changes in pH; maximal buffering capacity is seen within 1 pH point of the pKa of the acid in the buffer solution.
- The Henderson–Hasselbalch equation quantifies the relationship between pH and pKa for weak acids and between pOH and pKb for weak bases; when a solution is optimally buffered, pH = pKa and pOH = pKb.
ANSWERS TO CONCEPT CHECKS
**10.1**
-
Definition Acid Base Arrhenius Dissociates to form excess H+ in solution Dissociates to form excess OH− in solution
Brønsted–Lowry H+ donor H+ acceptor
Lewis Electron pair acceptor Electron pair donor
-
Anion Acid Formula Acid Name MnO4− HMnO4 Permanganic acid
Titanate (TiO32−) H2TiO3 Titanic acid
I− HI Hydroiodic acid
IO4− HIO4 Periodic acid
-
Reaction Amphoteric Reactant Amphiprotic? (Y or N)
HCO3− + HBr → H2CO3 + Br− HCO3− Yes
3 HCl + Al(OH)3 → AlCl3 + 3 H2O Al(OH)3 No
2 HBr + ZnO → ZnBr2 + H2O ZnO No
**10.2**
- An amphoteric species can act as an acid or a base.
- High Ka indicates a strong acid, which will dissociate completely in solution. Having a Ka slightly greater than water means the acid is a weak acid with minimal dissociation.
- High Kb indicates a strong base, which will dissociate completely in solution. Having a Kb slightly greater than water means the base is a weak base with minimal dissociation.
-
pH [H3O+] pOH [OH−] Acid or Base?
4 10−4 M 10 10−10 M Acid
3.05 8.89 × 10−4 M 10.95 1.12 × 10−11 M Acid
8.81 1.55 × 10−9 M 5.19 6.46 × 10−6 M Base
8.27 5.32 × 10−9 M 5.73 1.88 × 10−6 M Base
(Note: Exact answers are provided; your rounded answers should be relatively close to those listed here.)
- Ka × Kb = Kw
-
Reaction Acid Base Conjugate Acid Conjugate Base
H2CO3 + H2O ⇌ HCO3− + H3O+ H2CO3 H2O H3O+ HCO3−
H2PO4− + H2O ⇌ H3PO4 + OH− H2O H2PO4− H3PO4 OH−
- K a = [ CH 3 COO − ] [ H 3 O + ] [ CH 3 COOH] = [ x ] [ x ] [ 0. 2 M − x ] ≈ x 2 0. 2 . Therefore, x2 = 3.6 × 10−6 → x ≈ 2 × 10−3 M (actual = 1.9 × 10−3 M). Then, pH = −log H3O+ ≈ 3 − 0.2 = 2.8 (actual = 2.72)
**10.3**
- Acids use moles of H+ (H3O+) as an equivalent. Bases use moles of OH− as an equivalent.
- 6 N Al(OH)3; 32 N H2SO4
**10.4**
- The buffering region occurs when [HA] ≈ [A−] and is the flattest portion of the titration curve (resistant to changes in pH). The half-equivalence point is the center of the buffering region, where [HA] = [A−]. The equivalence point is the steepest point of the titration curve, and occurs when the equivalents of acid present equal the equivalents of base added (or vice-versa). The endpoint is the pH at which an indicator turns its final color.
- Phenolphthalein would be the preferred indicator for this titration.
- A strong acid and weak base have an equivalence point in the acidic range. A strong base and weak acid have an equivalence point in the basic range. A strong acid and strong base have an equivalence point at pH = 7 (neutral). A weak acid and weak base can have an equivalence point in the acidic, neutral, or basic range, depending on the relative strengths of the acid and base.
- A buffer solution is designed to resist changes in pH and has optimal buffering capacity within 1 pH point from its pKa.
- Recall from section 10.2 on Properties that the concentrations of the conjugate acid and conjugate base in the final solution must first be calculated due to the dilution from mixing the two solutions together. The calculation below follows that step, but leaves the values unsolved for so that some of their components can be cancelled and simplified:
pH = p K a + log [ A – ] [ HA] = − log 6.3 × 10 − 5 + log [ ( 0.005 N ) ( 100 mL) ( 105 mL) ] [ ( 5 N ) ( 5 mL) ( 105 mL) ] pH ≈ 4.37 + log ( 0.005 N ) ( 100 mL) ( 5 N ) ( 5 mL) = 4.37 + log 0.5 25 = 4.37 + log 2 × 10 − 2 ≈ 4.37 − 1 .8 = 2 .57 (actual = 2 .50) pOH ≈ 14 - 2 . 57 = 11 . 43 (actual = 11.5)
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
A Brønsted–Lowry base is defined as a proton acceptor. Ammonia, fluoride, and water—(A),(B), and(C), respectively—each accept a proton. (D), HNO2, is a far better Brønsted–Lowry acid, donating a proton to a solution.
2. B
First, convert the concentration to 5 × 10−3 M. Next, because sulfuric acid is a strong acid, we can assume that, for the majority of sulfuric acid molecules (although not all), both protons will dissociate. The concentration of hydrogen ions is therefore 2 × 5 × 10−3, or 10−2. The equation for pH is pH = −log [H+]. If [H+] = 10−2 M, then pH = 2.
3. A
Answering this question is simply a matter of knowing nomenclature. Acids ending in –ic are derivatives of anions ending in –ate, while acids ending in –ous are derivatives of anions ending in –ite. ClO3−, (B), is chlorate because it has more oxygen than the other commonly occurring ion, ClO2−, which is named chlorite. Therefore, HClO3 is chloric acid. HClO2, (C), represents chlorous acid. HClO, (D), represents hypochlorous acid.
4. B
Soluble hydroxides of Group IA and IIA metals are strong bases, eliminating (A) and (D). (B) and (C) are both weak bases; however, methylamine contains an alkyl group, which is electron-donating. This increases the electron density on the nitrogen in methylamine, making it a stronger (Lewis) base. Therefore, ammonia is the weakest base.
5. B
The purpose of a buffer is to resist changes in the pH of a reaction. Buffers are not generally used to affect the kinetics of a reaction, so (C) and (D) are incorrect. (A) is true only in specific circumstances where the pH of the buffer solution itself is neutral. Many natural buffer systems maintain pH in the acidic or basic ranges.
6. D
The question is asking for pH, but because of the information given, we must first find the pOH and then subtract it from 14 to get the pH. Use the Henderson–Hasselbalch equation:
pOH = p K b + log [ conjugate acid] [ base] = 3.45 + log 70 m M 712 m M ≈ 3.45 + log 1 10 = 3.45 − 1 = 2.45
If the pOH = 2.45, the pH = 14 − 2.45 = 11.55.
7. A
The first pKa in this curve can be estimated by eye. It is located halfway between the starting point (when no base had yet been added) and the first equivalence point (the first steep portion of the graph, around 15 mL). This point is at approximately 7–8 mL on the x-axis, which corresponds to a pH of approximately 1.9. Notice that this region experiences very little change in pH, which is the defining characteristic of a buffer region.
8. C
The second equivalence point is the midpoint of the second steep increase in slope. This corresponds to approximately pH = 5.9.
9. B
The value of the second pKa is found at the midpoint between the first and second equivalence points. In this curve, that corresponds to pH = 4.1. Just like the first pKa, it is in the center of a flat buffering region.
10. B
Gram equivalent weight is the weight (in grams) that releases 1 acid or base equivalent from a compound. Because H3PO4 contains 3 protons, we find the gram equivalent weight by dividing the mass of one mole of the species by 3. The molar mass of phosphoric acid is 98 g mol , so the gram equivalent weight is 32.7 g.
11. D
This question requires the application of the acid dissociation constant. Weak acids do not dissociate completely; therefore, all three species that appear in the balanced equation will be present in solution. Hydrogen ions and conjugate base anions dissociate in equal amounts, so [H+] = [XO2−]. If the initial concentration of HXO2 was 2 M and some amount x dissociates, we will have x amount of H3O+ and XO2− at equilibrium, with 2 M − x amount of HXO2 at equilibrium.
K a = [ H 3 O + ] [ XO 2 − ] [ HXO 2 ] = ( x ) ( x ) 2 M − x ≈ x 2 2
Note that x was considered negligible when added or subtracted, per usual. Solving for x, we get:
x 2 2 = 3.2 × 10 − 5 → x 2 = 6.4 × 10 − 5 = 64 × 10 − 6 → x = 8 × 10 − 3 M
12. C
A higher Ka implies a stronger acid. Weak acids usually have a Ka that is several orders of magnitude below 1. The pKa of a compound is the pH at which there are equal concentrations of acid and conjugate base; the pKa of this compound would be −log 1 = 0. With such a low pKa, this compound must be an acid. Therefore, the pH of any concentration of this compound must be below 7.
13. D
An amphoteric species is one that can act either as an acid or a base, depending on its environment. Proton transfers are classic oxidation–reduction reactions, so(A) and (B) are true. (C) is true because many amphoteric species, such as water and bicarbonate, can either donate or accept a proton. (D) is false, and thus the correct answer because amphoteric species can be either polar or nonpolar in nature.
14. C
NaOH is a strong base; as such, there will be 1.2 × 10−5 M OH− in solution. Based on this information alone, the pOH must be between 4 and 5, and the pH must be between 9 and 10. Using the shortcut, pOH ≈ 5 − 0.12 = 4.88. pH = 14 − pOH = 9.12 (actual = 9.08).
15. C
Use the equivalence point equation: NaVa = NbVb. Ba(OH)2 can dissociate to give two hydroxide ions, so its normality is 2 M × 2 = 4 N. H3PO4 can dissociate to give three hydronium ions, so its normality is 6 M × 3 = 18 N. Plugging into the equation, we get (18 N)(4 L) = (4 N)(Vb). Therefore, Vb is 18 L.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(10.1) Autoionization constant for water:Kw = [H3O+][OH−] = 10−14 at 25 °C (298 K)
(10.2) Definitions of pH and pOH: pH = –log[H + ] = log 1 [ H + ] pOH = –log[OH − ] = log 1 [ OH − ]
(10.3) Relationship of pH and pOH at 298 K: pH + pOH = 14
(10.4) p scale value approximation: p value ≈ m − 0.n
(10.5) Acid dissociation constant: K a = [ H 3 O + ] [ A − ] [ HA]
(10.6) Base dissociation constant: K b = [ B + ] [ OH − ] [ BOH]
(10.7) **Relationship of *K***a** and *K***bat 298 K: K a , acid × K b,conjugate base = K w = 10 −14 ; K b,base × K a,conjugate acid = K w = 10 −14
(10.8) Equivalence point: NaVa = NbVb
(10.9) Henderson–Hasselbalch equation (acid buffer): pH = pK a + log [ A − ] [ HA]
(10.10) Henderson–Hasselbalch equation (base buffer): pOH = p K b + log [ B + ] [ BOH]
SHARED CONCEPTS
Biology Chapter 6
The Respiratory System
Biology Chapter 10
Homeostasis
General Chemistry Chapter 3
Bonding and Chemical Interactions
General Chemistry Chapter 9
Solutions
Organic Chemistry Chapter 4
Analyzing Organic Reactions
Physics and Math Chapter 10
Mathematics