Chapter 3: Bonding

Chapter 3: Bonding
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Within one principal energy level, which subshell has the least energy?
- s
- p
- d
- f
-
Which of the following compounds possesses at least one σ bond?
- CH4
- C2H2
- C2H4
- All of the above contain at least one σ bond.
-
A carbon atom participates in one double bond. As such, this carbon contains orbitals with:
- hybridization between the s-orbital and one p-orbital.
- hybridization between the s-orbital and two p-orbitals.
- hybridization between the s-orbital and three p-orbitals.
- unhybridized s character.
-
The hybridizations of the carbon and nitrogen atoms in CN− are:
- sp3 and sp3, respectively.
- sp3 and sp, respectively.
- sp and sp3, respectively.
- sp and sp, respectively.
-
Which of the following hybridizations does the Be atom in BeH2 assume?
- sp
- sp2
- sp3
- sp3d
-
Two atomic orbitals may combine to form:
- a bonding molecular orbital.
- an antibonding molecular orbital.
- hybridized orbitals.
- I only
- III only
- I and II only
- I, II, and III
-
Molecular orbitals can contain a maximum of:
- one electron.
- two electrons.
- four electrons.
- 2n2 electrons, where n is the principal quantum number of the combining atomic orbitals.
-
π bonds are formed by which of the following orbitals?
- Two s-orbitals
- Two p-orbitals
- One s- and one p-orbital
- Two sp2-hybridized orbitals
-
How many σ bonds and π bonds are present in the following compound?
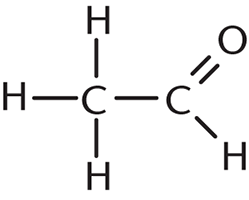
- Six σ bonds and one π bond
- Six σ bonds and two π bonds
- Five σ bonds and one π bond
- Five σ bonds and two π bonds
-
The four C–H bonds of CH4 point toward the vertices of a tetrahedron. This indicates that the hybridization of the carbon atom in methane is:
- sp.
- sp2.
- sp3.
- sp3d.
-
Why is a single bond stronger than a π bond?
- π bonds have greater orbital overlap.
- s-orbitals have more overlap than p-orbitals.
- sp3 hybridization is always unstable.
- I only
- II only
- I and III only
- II and III only
-
The p character of the bonds formed by the carbon atom in HCN is:
- 25%.
- 50%.
- 67%.
- 75%.
-
A resonance structure describes:
- the hybrid of all possible structures that contribute to electron distribution.
- a potential arrangement of electrons in a molecule.
- the single form that the molecule most often takes.
- I only
- II only
- I and II only
- I, II, and III
-
An electron is known to be in the n = 4 shell and the l = 2 subshell. How many possible combinations of quantum numbers could this electron have?
- 1
- 2
- 5
- 10
-
Compared to single bonds, triple bonds are:
- weaker.
- longer.
- made up of fewer σ bonds.
- more rigid.
Answer Key
- A
- D
- B
- D
- A
- D
- B
- B
- A
- C
- B
- B
- B
- D
- D
Chapter 3: Bonding
CHAPTER 3
BONDING
In This Chapter
3.1 Atomic Orbitals and Quantum Numbers 3.2 Molecular Orbitals
σ and π Bonds
3.3 Hybridization
sp3
sp2
sp
Resonance
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 3% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
4E: Atoms, nuclear decay, electronic structure, and atomic chemical behavior
5B: Nature of molecules and intermolecular interactions
Introduction
Now that we have an understanding of nomenclature and how compounds are related, we are ready to start examining the real nature of chemical bonding. Bonding determines how atoms come together to form molecules. It also governs the ways those molecules interact with the other molecules in their environment.
Organic chemistry is the study of carbon and carbon-containing compounds. What makes carbon so special? The simple answer is that carbon has unique bonding properties. Carbon is tetravalent, which means that it can form bonds with up to four other atoms, allowing for the massive versatility required to form the foundation of biomolecules and life itself. This versatility is compounded by the fact that carbon, located near the center of the periodic table, can form bonds with many different elements because of its moderate electronegativity. In addition, because carbon atoms are fairly small, the bonds that they form are strong and stable.
Remember that there are two types of chemical bonds. The first is ionic, in which electrons are transferred from one atom to another and the resulting ions are held together by electrostatic interactions; the second is covalent, in which electrons are shared between atoms. Organic chemistry is deeply rooted in covalent bonding.
3.1 Atomic Orbitals and Quantum Numbers
LEARNING OBJECTIVES
After Chapter 3.1, you will be able to:
- Describe the four quantum numbers, n, l, ml, andms
- Provide ranges of possible values for each quantum number
Bonding occurs in the outermost electron shell of atoms, so an understanding of bonding is contingent on understanding the organization of electrons in an atom. Quantum numbers are discussed in detail in Chapter 1 of MCAT General Chemistry Review, and are briefly summarized here.
The first three quantum numbers, n, l, and *m**l, describe the size, shape, number, and orientation of atomic orbitals an element possesses. The principal quantum number,n, corresponds to the energy level of a given electron in an atom and is essentially a measure of size. The smaller the number, the closer the shell is to the nucleus, and the lower its energy. The possible values ofnrange from 1 to ∞, although the MCAT only tests onn*-values up to 7.
BRIDGE
Recall from Chapter 1 of MCAT General Chemistry Review that quantum numbers describe the location of an electron within an atom and that each electron has a unique combination of quantum numbers according to the Pauli exclusion principle.
Within each electron shell, there can be several subshells. Subshells are described by the azimuthal quantum number, l, which ranges from 0 to n – 1 for a given energy shell. The l-values 0, 1, 2, and 3 correspond to the s, p, d, and f subshells, respectively. Just as with the principal quantum number, energy increases as the azimuthal quantum number increases.
Within each subshell, there may be several orbitals. Orbitals are described by the magnetic quantum number, *m**l, which ranges from –lto +lfor a given subshell. Each type of atomic orbital has a specific shape, which describes the probability of finding an electron in a given region of space. Ans-orbital is spherical and symmetrical, centered around the nucleus. Ap-orbital is composed of two lobes located symmetrically about the nucleus and contains a node—an area where the probability of finding an electron is zero—at the nucleus. Picture thep-orbital as a dumbbell that can be positioned in three different orientations, along thex-,y-, orz-axis. It should make sense that there are threep-orbitals; thepsubshell has thel-value of 1, so there are three possible values form**l: –1, 0, and 1. The shapes of the first fives- andp-orbitals are shown in Figure 3.1. Ad-orbital is composed of four symmetrical lobes and contains two nodes. Four of thed-orbitals are clover-shaped, and the fifth looks like a donut wrapped around the center of ap-orbital. Thankfully, the multiple complex shapes ofd- andf-orbitals are rarely encountered in organic chemistry. Each orbital can hold two electrons, which are distinguished by the spin quantum number,m**s. The only values ofm**s* are
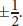
Figure 3.1. The First Five Atomic Orbitals
MCAT CONCEPT CHECK 3.1:
Before you move on, assess your understanding of the material with this question.
-
Summarize the quantum numbers below. The first entry has been completed for clarification.
**Symbol Name Describes… Organizational Level Possible Values *n*** Principal QN Size Shell 1 to ∞
l ml ms
3.2 Molecular Orbitals
LEARNING OBJECTIVES
After Chapter 3.2, you will be able to:
- Describe the stability and energy of bonding and antibonding orbitals
- Explain how the addition of a double or triple bond affects the electron density and molecular orbitals within a molecule
- Order the different orbital types based on strength
When two atomic orbitals combine, they form molecular orbitals. Molecular orbitals are obtained mathematically by adding or subtracting the wave functions of the atomic orbitals. While the mathematics of combining wave functions is outside the scope of the MCAT, some questions may ask for the visualization of molecular orbitals, as shown in Figure 3.2. If the signs of the wave functions are the same, a lower-energy (more stable) bonding orbital is produced. If the signs are different, a higher-energy (less stable) antibonding orbital is produced.
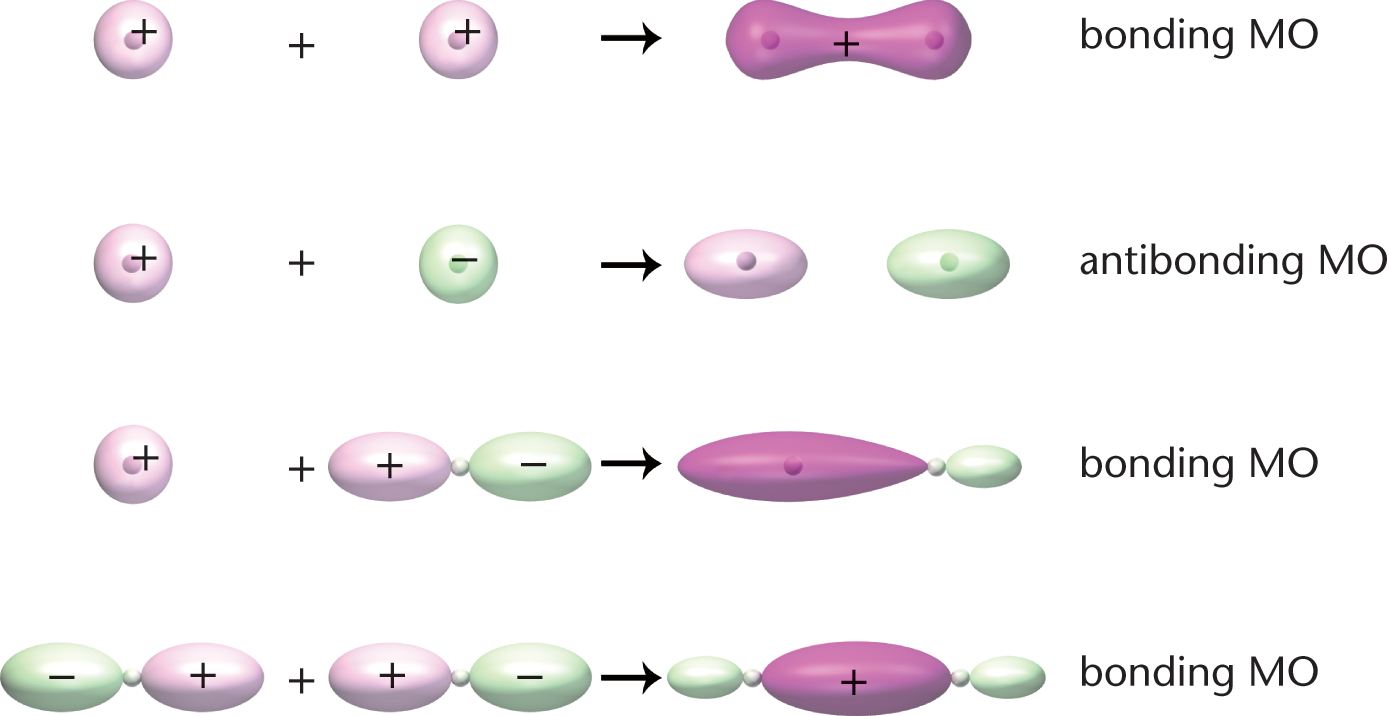
Figure 3.2. Molecular Orbitals Molecular orbitals can be bonding or antibonding, depending on the signs of the atomic orbitals used to form them; head-to-head or tail-to-tail overlap of atomic orbitals results in a σ bond.
*σ* and *π* Bonds
When a molecular orbital is formed by head-to-head or tail-to-tail overlap, as in Figure 3.2, the resulting bond is called a sigma (σ)bond. All single bonds are σ bonds, accommodating two electrons.
KEY CONCEPT
Sigma (σ) bonds are formed by head-to-head or tail-to-tail overlap of atomic orbitals. These bonds are by far the most common in organic compounds and on the MCAT.
When two p-orbitals line up in a parallel (side-by-side) fashion, their electron clouds overlap, and a bonding molecular orbital, called a pi (π)bond, is formed. This is demonstrated in Figure 3.3. One π bond on top of an existing σ bond is a double bond. A σ bond and two π bonds form a triple bond. Unlike single bonds, which allow free rotation of atoms around the bond axis, double and triple bonds hinder rotation and, in effect, lock the atoms into position.
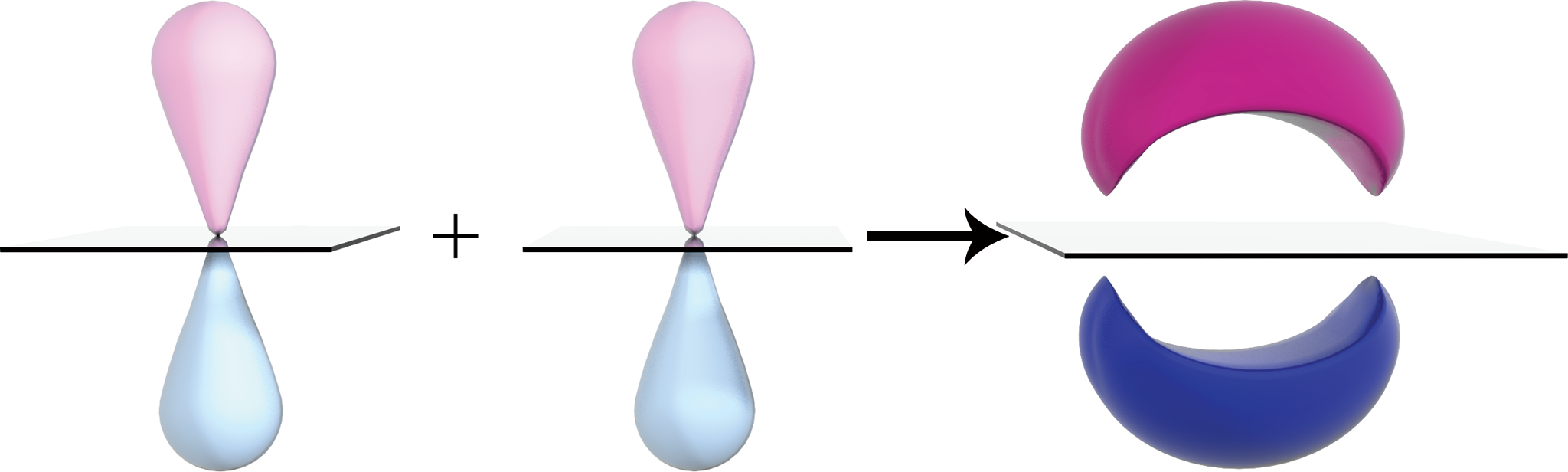
Figure 3.3. Pi (π) Bond Electron density exists above and below the plane of the molecule, restricting rotation about a double bond.
It is important to remember that a π bond cannot exist independently of a σ bond. Only after the formation of a σ bond will the p-orbitals of adjacent carbons be parallel and in position to form the π bond. The more bonds that are formed between atoms, the shorter the overall bond length. Therefore, a double bond is shorter than a single bond, and a triple bond is shorter than a double bond. Shorter bonds hold atoms more closely together and are stronger than longer bonds; shorter bonds require more energy to break.
While double bonds are stronger than single bonds overall, individual π bonds are weaker than σ bonds. Therefore, it is possible to break only one of the bonds in a double bond, leaving a single bond intact. This happens often in organic chemistry, such as when cis–trans isomers are interconverted between conformations. Breaking a single bond requires far more energy.
KEY CONCEPT
A double bond consists of both a σ bond and a π bond; a triple bond consists of a σ bond and two π bonds. π bonds are weaker than σ bonds, but the strength is additive, making double and triple bonds stronger overall than single bonds.
As discussed previously, double and triple bonds do not freely rotate like single bonds. As such, double bonds in compounds make for stiffer molecules. Partial double-bond character in structures with resonance also restricts free rotation, resulting in more rigid structures. Proteins exhibit this kind of limited rotation because there is resonance in the amide linkages between adjacent amino acids.
MCAT CONCEPT CHECK 3.2:
Before you move on, assess your understanding of the material with these questions.
-
Which is more stable: a bonding orbital or an antibonding orbital? Which has higher energy?
- More stable:
________________________
- Higher energy:
________________________
-
What differences would be observed in a molecule containing a double bond compared to the same molecule containing only single bonds?
________________________
-
Rank the following orbitals in decreasing order of strength: σ bond, π bond, double bond, triple bond.
________>______>______>________.
3.3 Hybridization
LEARNING OBJECTIVES
After Chapter 3.3, you will be able to:
- Recall the percentage of s character present in a given hybridization level, such as sp2
- Describe the relationship between electron density and resonance structures
- Identify the hybridization of an atom within a complex molecule:
Carbon has the electron configuration 1s22s22p2 and therefore needs four electrons to complete its octet (2s22p6). A typical molecule formed by carbon is methane, CH4. Experimentation shows that the four σ bonds in methane are equivalent. This may seem inconsistent with what we know about the asymmetrical distribution of carbon’s valence electrons: two electrons in the 2s-orbital, one in the *p**x-orbital, one in thep**y-orbital, and none in thep**z*-orbital. This apparent discrepancy is accounted for by the theory of orbital hybridization.
*sp*3
Hybrid orbitals are formed by mixing different types of orbitals. Just as with molecular orbitals, we can use advanced mathematics to merge three p-orbitals and one s-orbital. The result? As shown in Figure 3.4, this forms four identical sp3 orbitals with new, hybridized shapes.
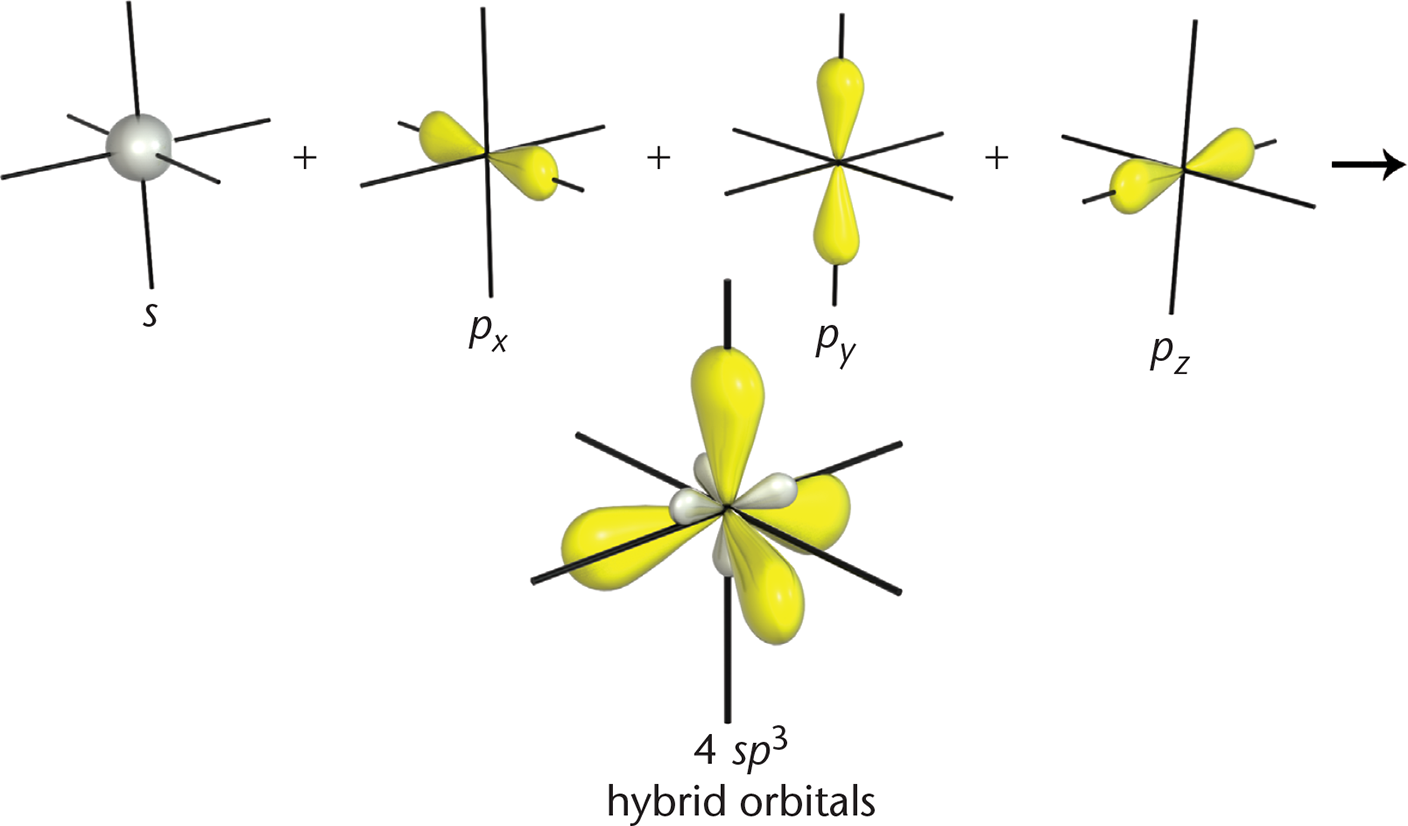
Figure 3.4. sp3-Hybridized Orbitals An atom with these orbitals has tetrahedral geometry, and there are no unhybridized p-orbitals to formπbonds.
All four of these orbitals point toward the vertices of a tetrahedron to minimize repulsion, which explains why carbon prefers tetrahedral geometry. The hybridization is accomplished by promoting one of the 2s electrons into the 2*p**z*-orbital, as shown in Figure 3.5. This produces four valence orbitals, each with one electron, which can be mathematically mixed to model the hybrid orbitals.
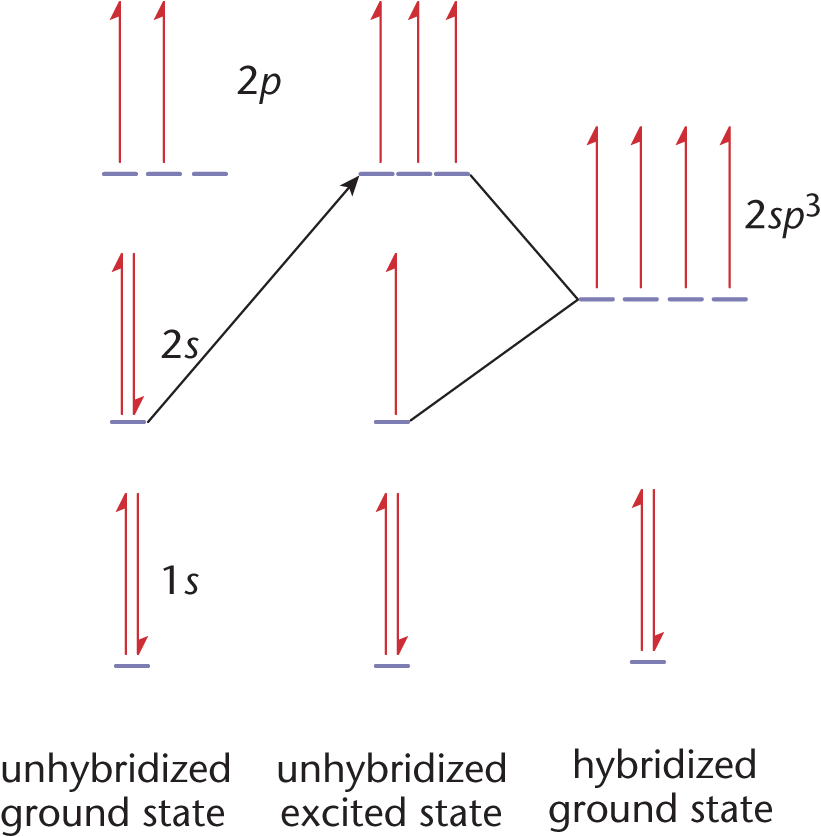
Figure 3.5. Hybridization of Carbon Orbitals
KEY CONCEPT
Hybridization is a way of making all of the bonds to a central atom equivalent to each other. The sp3 orbitals are the reason for the tetrahedral shape that is a hallmark of carbon-containing compounds.
The MCAT sometimes tests how much “s character” a certain hybrid orbital has. To answer such questions, we simply need to determine what type of hybridization exists and use the name to solve the problem. For example, in sp3 orbitals, we have one s- and three p-orbitals, so the bond has 25% s character and 75% p character.
*sp*2
Although carbon is most often bonded with sp3 hybridization, there are two other possibilities. When one s-orbital is mixed with two p-orbitals, three sp2-hybridized orbitals are formed, as shown in Figure 3.6. These orbitals have 33% s character and 67% p character.
This is the hybridization seen in alkenes. The third p-orbital of each carbon is left unhybridized. This is the orbital that participates in the π bond. The three sp2 orbitals are oriented 120° apart, which allows for maximum separation. We know that the unhybridized p-orbital is involved in the π component of the double bond, but what about the hybrid orbitals? In ethene, two of the sp2 hybridized orbitals will participate in C–H bonds, and the other hybrid orbital will line up with the π bond and form the σ component of the C=C double bond.
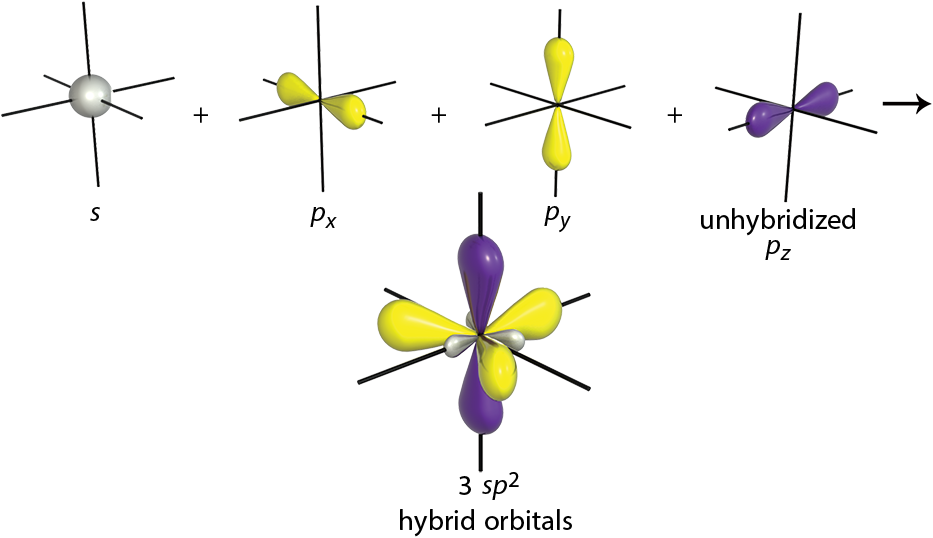
Figure 3.6. sp2-Hybridized Orbitals A molecule with these orbitals has trigonal planar geometry, and the one unhybridized p-orbital can be used to form aπbond.
*sp*
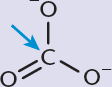
To form a triple bond, we need two of the p-orbitals to form π bonds, and the third p-orbital will combine with the s-orbital to form two sp-orbitals, as shown in Figure 3.7. These orbitals have 50% s character and 50% p character. These orbitals are oriented 180° apart, which explains the linear structure of molecules containing sp-hybridized carbons. The two π bonds can be between the carbon and one other atom (forming a triple bond, like ethyne), or between the carbon and two different atoms (forming two double bonds in a row, like carbon dioxide). In both cases, the molecule is linear about the sp-hybridized carbon.
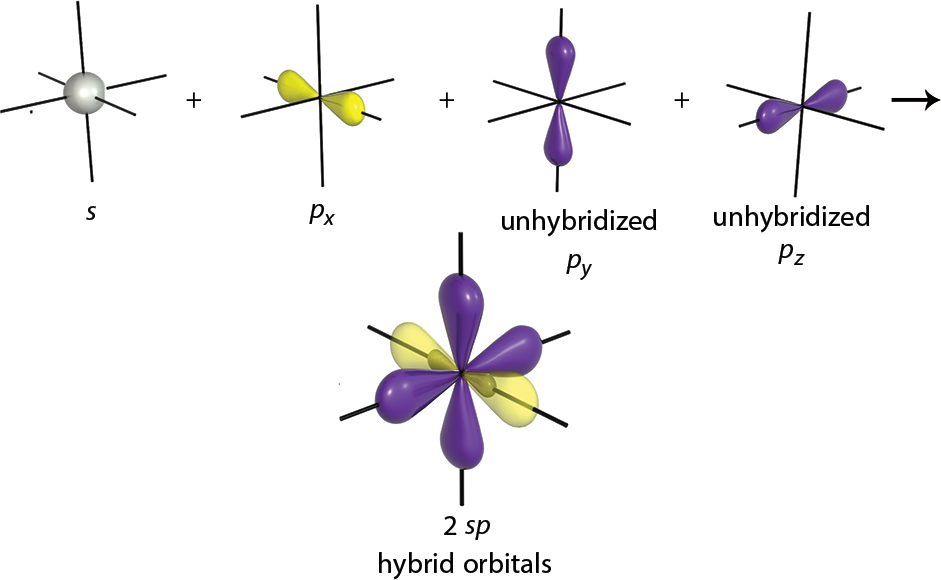
Figure 3.7. sp-Hybridized Orbitals A molecule with these orbitals has linear geometry, and the two unhybridized p-orbitals can be used to formπbonds.
Resonance
Resonance delocalization of electrons occurs in molecules that have conjugated bonds. Conjugation requires alternating single and multiple bonds because this pattern aligns a number of unhybridized p-orbitals down the backbone of the molecule. π electrons can then delocalize through this p-orbital system, adding stability to the molecule. Resonance structures are drawn as the various transient forms the molecule takes, as shown in Figure 3.8.
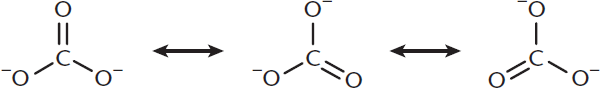
Figure 3.8. Resonance Forms of Carbonate These forms have equal stability and therefore contribute equally to the true electron density of the molecule.
However, these forms aren’t in any sort of equilibrium—the electron density is distributed throughout, making the true form a hybrid of the resonance structures, as shown with ozone in Figure 3.9.
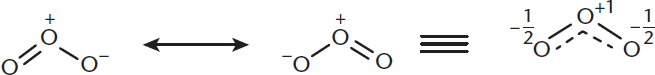
Figure 3.9. Structure of Ozone *The true electron density of ozone is somewhere between the two resonance forms, creating 1.5 bonds between each oxygen, and leaving each oxygen with a
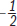
charge.*
If the stability of the various resonance forms differs, then the true electron density will favor the most stable form. Particular resonance structures can be favored because they lack formal charges or form full octets on highly electronegative atoms, like oxygen and nitrogen. Stabilization of positive and negative charges through induction and aromaticity can also favor certain resonance structures.
MCAT CONCEPT CHECK 3.3:
Before you move on, assess your understanding of the material with these questions.
-
What is the s character of sp-, sp2-, and sp3-hybridized orbitals?
-
sp: _________________
-
sp2: _________________
-
sp3: _________________
-
What are resonance structures? How does the true electron density of a compound relate to its resonance structures?
________________________________
Conclusion
The ability of carbon to form single, double, and triple bonds (or to form σ and multiple π bonds) and to form hybrid orbitals gives rise to an entire branch of chemistry—as well as life on Earth. You may have thought this chapter was a bit brief; that’s because the specifics of bonding fall mostly under the domain of general chemistry. However, without a solid grasp of orbitals and bonding, it would be difficult to explain the organic reactions that are tested on the MCAT, which will be the focus of the next seven chapters. Avoid compartmentalizing the information you learn throughout the course of your studies, as bonding plays a role in general chemistry, organic chemistry, and biochemistry, and can therefore be tested in either the Chemical and Physical Foundations of Biological Systems section or theBiological and Biochemical Foundations of Living Systems section. All of the subjects within science blend together into a seemingly complicated, yet beautifully simple, picture of the universe. The sooner you integrate the knowledge you’re accumulating, the more manageable and rewarding your studying will become.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Atomic Orbitals and Quantum Numbers
- Quantum numbers describe the size, shape, orientation, and number of atomic orbitals an element possesses.
- The principal quantum number, n, describes the energy level (shell) in which an electron resides and indicates the distance from the nucleus to the electron. Its possible values range from 1 to ∞.
- The azimuthal quantum number, l, determines the subshell in which an electron resides. Its possible values range from 0 to n – 1. The subshell is often indicated with a letter: l = 0 corresponds to s, 1 is p, 2 is d, and 3 is f.
- The magnetic quantum number, *m**l, determines the orbital in which an electron resides. Its possible values range from –lto +l. Different orbitals have different shapes:s-orbitals are spherical, whilep-orbitals are dumbbell-shaped and located on thex-,y-, orz*-axis.
- The spin quantum number, *m**s*, describes the spin of an electron. Its possible values are
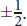
Molecular Orbitals
- Bonding orbitals are created by head-to-head or tail-to-tail overlap of atomic orbitals of the same sign and are energetically favorable.
- Antibonding orbitals are created by head-to-head or tail-to-tail overlap of atomic orbitals that have opposite signs and are energetically unfavorable.
- Single bonds are sigma (σ)bonds, which contain two electrons.
- Double bonds contain one σ bond and one pi (π)bond. π bonds are created by sharing of electrons between two unhybridized p-orbitals that align side-by-side.
- Triple bonds contain one σ bond and two π bonds.
- Multiple bonds are less flexible than single bonds because rotation is not permitted in the presence of a π bond. Multiple bonds are shorter and stronger than single bonds, although individual π bonds are weaker than σ bonds.
Hybridization
- sp3-hybridized orbitals have 25% s character and 75% p character. They form tetrahedral geometry with 109.5° bond angles. Carbons with all single bonds are sp3-hybridized.
- sp2-hybridized orbitals have 33% s character and 67% p character. They form trigonal planar geometry with 120° bond angles. Carbons with one double bond are sp2-hybridized.
- sp-hybridized orbitals have 50% s character and 50% p character. They form linear geometry with 180° bond angles. Carbons with a triple bond, or with two double bonds, are sp-hybridized.
- Resonance describes the delocalization of electrons in molecules that have conjugated bonds.
- Conjugation occurs when single and multiple bonds alternate, creating a system of unhybridized p-orbitals down the backbone of the molecule through which π electrons can delocalize.
- Resonance increases the stability of a molecule.
- The various resonance forms all contribute to the true electron density of the molecule; the more stable the resonance form, the more it contributes. Resonance forms are favored if they lack formal charge, form full octets on electronegative atoms, or stabilize charges through induction and aromaticity.
ANSWERS TO CONCEPT CHECKS
**3.1**
-
**Symbol Name Describes… Organizational Level Possible Values *n*** Principal QN Size Shell 1 to ∞
l Azimuthal QN Shape Subshell 0 to n – 1
ml Magnetic QN Orientation Orbital – l to +l
ms Spin QN Spin —
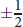
**3.2**
- Bonding orbitals are more stable than antibonding orbitals. Therefore, antibonding orbitals have higher energy than bonding orbitals.
- The differences would be in bond length (shorter in double bond than single), bond energy (higher in double bond than single), and molecular rigidity (higher in double bond than single).
- Triple bond > double bond > σ bond > π bond. Remember that while an individual π bond is weaker than a σ bond, bond strength is additive. Therefore, double bonds are stronger than single, and triple bonds are stronger still.
**3.3**
- sp orbitals have 50% s character and 50% p character, sp2 orbitals have 33% s character and 67% p character, and sp3 orbitals have 25% s character and 75% p character.
- Resonance structures differ in their placement of electrons in hybridized p-orbitals and require bond conjugation to delocalize electrons in a molecule. The true electron density is a weighted average of the resonance structures of a given compound, favoring the most stable structures.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. A
The energies of the subshells within a principal quantum number are as follows: s < p < d < f
2. D
All single bonds are σ bonds; double and triple bonds each contain one σ bond and one or two π bonds, respectively. The compounds CH4, C2H2, and C2H4 all contain at least one single bond and therefore contain at least one σ bond.
3. B
In a carbon with one double bond, sp2 hybridization occurs—that is, one s-orbital hybridizes with two p-orbitals to form three sp2-hybridized orbitals. The third p-orbital of the carbon atom remains unhybridized and takes part in the formation of the π bond of the double bond. Although there is an unhybridized p-orbital, there are no unhybridized s-orbitals, eliminating (D).
4. D
The carbon and nitrogen atoms are connected by a triple bond in CN– (:C≡N:–). A triple-bonded atom is sp hybridized; one s-orbital hybridizes with one p-orbital to form two sp-hybridized orbitals. The two remaining unhybridized p-orbitals take part in the formation of two π bonds.
5. A
Beryllium has only two electrons in its valence shell. When it bonds to two hydrogens, it requires two hybridized orbitals, meaning that its hybridization must be sp. Note that the presence of only single bonds does not mean that the hybridization must be sp3; this is a useful assumption for carbon, but does not apply to beryllium because of its smaller number of valence electrons. The two unhybridized p-orbitals around beryllium are empty in BeH2, which takes on the linear geometry characteristic of sp-hybridized orbitals.
6. D
When atomic orbitals combine, they form molecular orbitals. When two atomic orbitals with the same sign are added head-to-head or tail-to-tail, they form bonding molecular orbitals. When two atomic orbitals with opposite signs are added head-to-head or tail-to-tail, they form antibonding molecular orbitals. Atomic orbitals can also hybridize, forming sp3, sp2, or sp orbitals.
7. B
Like atomic orbitals, molecular orbitals each can contain a maximum of two electrons with opposite spins. The 2n2 rule in (D) refers to the total number of electrons that can exist in a given energy shell, not in a molecular orbital.
8. B
π bonds are formed by the parallel overlap of unhybridized p-orbitals. The electron density is concentrated above and below the bonding axis. A σ bond, on the other hand, can be formed by the head-to-head overlap of two s-orbitals or hybridized orbitals. In a σ bond, the density of the electrons is concentrated between the two nuclei of the bonding atoms.
9. A
Each single bond has one σ bond, and each double bond has one σ and one π bond. In this question, there are five single bonds (five σ bonds) and one double bond (one σ bond and one π bond), which gives a total of six σ bonds and one π bond. Thus, the correct answer is (A).
10. C
The four bonds point to the vertices of a tetrahedron, which means that the angle between two bonds is 109.5°, a characteristic of sp3 orbitals. Hence, the carbon atom of CH4 is sp3-hybridized.
11. B
Bond strength is determined by the degree of orbital overlap; the greater the overlap, the greater the bond strength. A π bond is weaker than a single bond because there is significantly less overlap between the unhybridized p-orbitals of a π bond (due to their parallel orientation) than between the s-orbitals or hybrid orbitals of a σ bond. sp3-hybridized orbitals can be quite stable, as evidenced by the number of carbon atoms with this hybridization forming stable compounds.
12. B
The carbon bond in hydrogen cyanide (H–C≡N:) is triple-bonded, and because triple bonds require two unhybridized p-orbitals, the carbon must be sp-hybridized; sp-hybridized orbitals have 50% s character and 50% p character.
13. B
A resonance structure describes an arrangement of electrons in a molecule. Different resonance structures can be derived by moving electrons in unhybridized p-orbitals throughout a molecule containing conjugated bonds. In molecules that contain multiple resonance structures, some are usually more stable than others; however, each resonance structure is not necessarily the most common form a molecule takes, eliminating statement III. Statement I has reversed the terminology for resonance structures: the electron density in a molecule is the weighted average of all possible resonance structures, not the other way around.
14. D
An electron in the n = 4 shell and the l = 2 subshell can have five different values for *m**l*: –2, –1, 0, 1, or 2. In each of these orbitals, electrons can have positive or negative spin. Thus, there are 5 × 2 = 10 possible combinations of quantum numbers for this electron.
15. D
π bonds do not permit free rotation, unlike σ bonds; this makes triple bonds more rigid than single bonds. Triple bonds are stronger and shorter bonds than single bonds, eliminating (A) and (B). Both single and triple bonds contain one σ bond, eliminating (C).
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 1
- Amino Acids, Peptides, and Proteins
- General Chemistry Chapter 1
- Atomic Structure
- General Chemistry Chapter 3
- Bonding and Chemical Interactions
- General Chemistry Chapter 4
- Compounds and Stoichiometry
- Organic Chemistry Chapter 2
- Isomers
- Organic Chemistry Chapter 11
- Spectroscopy