Chapter 4: Analyzing Organic Reactions

Chapter 4: Analyzing Organic Reactions
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of the following are Lewis bases?
- Ag+
- H2O
- NH3
- I only
- I and II only
- II and III only
- I, II, and III
-
Rank the following in order of decreasing nucleophilicity in an aprotic solvent: RO–, RCOOH, ROH, HO–
- RCOOH > ROH > RO– > HO–
- HO– > ROH > RO– > RCOOH
- RO– > HO– > ROH > RCOOH
- RCOOH > RO– > HO– > ROH
-
Rank the following in order of decreasing electrophilicity:
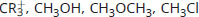
-
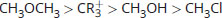
-
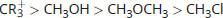
-
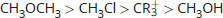
-
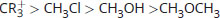
-
Rank the following in order of decreasing leaving group ability: H2O, HO–, Br–, H–
- H2O > Br– > HO– > H–
- H2O > HO– > Br– > H–
- HO– > Br– > H2O > H–
- HO– > H– > H2O > Br–
-
Rank the following in order of decreasing oxidation state: amine, carboxylic acid, aldehyde, alkane
- Aldehyde, amine, alkane, carboxylic acid
- Carboxylic acid, aldehyde, amine, alkane
- Carboxylic acid, amine, aldehyde, alkane
- Alkane, amine, aldehyde, carboxylic acid
-
The last step in the Grignard reaction is acidification, in which the alkoxide ion is protonated to form the final alcohol product. During this reaction, the alkoxide ion acts as a:
- Lewis base only
- Brønsted–Lowry base and Lewis base
- Brønsted–Lowry base and Lewis acid
- Brønsted–Lowry base only
-
If 2-butanol was treated with dichromate, what reaction would occur?
- Reduction, resulting in the formation of butene
- Oxidation, resulting in the formation of butanoic acid
- Oxidation, resulting in the formation of butanone
- No reaction would occur.
-
If 1-hexanol was treated with pyridinium chlorochromate, what would the end product be?
- 2-Hexanol
- 2-Hexanone
- Hexanal
- Hexanoic acid
-
SN1 reactions show first-order kinetics because:
- the rate-limiting step is the first step to occur in the reaction.
- the rate-limiting step involves only one molecule.
- there is only one rate-limiting step.
- the reaction involves only one molecule.
-
In a protic solvent, which of the following halogens would be the best nucleophile?
- Br–
- Cl–
- F–
- I–
-
Which of the compounds below can undergo oxidation by PCC?
-
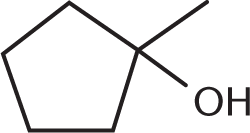
-
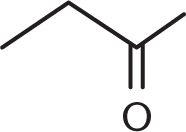
-
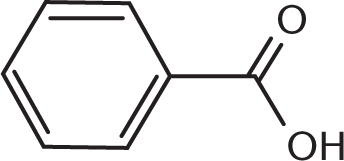
-
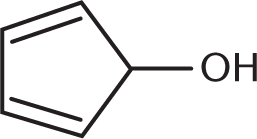
-
Which of the following substrates would most rapidly undergo an SN2 reaction when paired with an appropriate nucleophile?
-
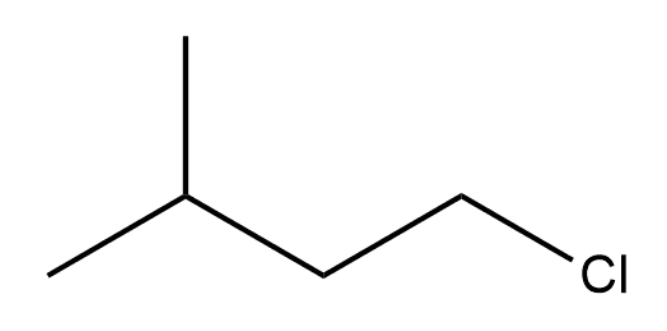
-
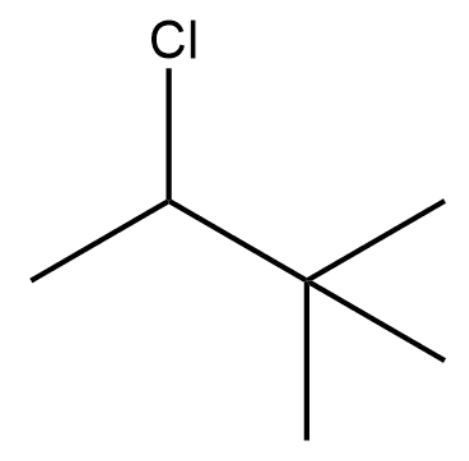
-
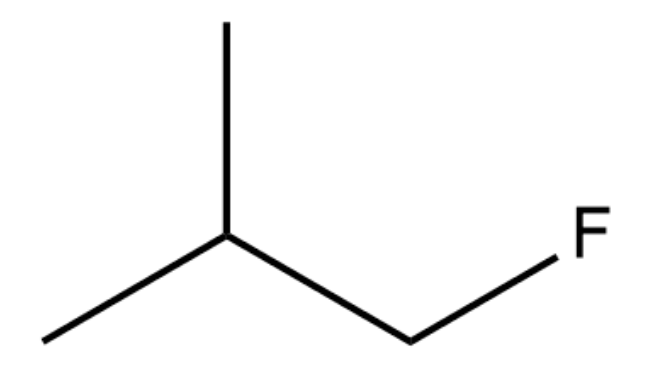
-
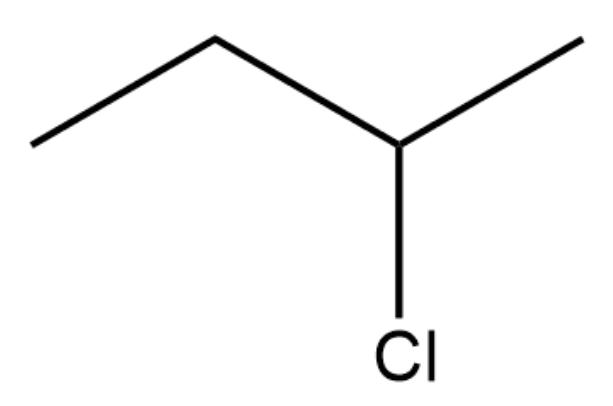
-
Which of the following solvents would be LEAST useful for a nucleophile–electrophile reaction?
- H2O
- CH3CH2OH
- CH3SOCH3
- CH3CH2CH2CH2CH2CH3
-
Aldehydes are generally more reactive than equivalent ketones to nucleophiles. This is likely due to differences in:
- steric hindrance.
- leaving group ability.
- resonance stabilization.
- electron-withdrawing character.
-
Which conversion between carboxylic acid derivatives is NOT possible by a nucleophilic reaction?
- Carboxylic acid to ester
- Ester to carboxylic acid
- Anhydride to amide
- Ester to anhydride
Answer Key
- C
- C
- D
- A
- B
- B
- C
- C
- B
- D
- D
- A
- D
- A
- D
Chapter 4: Analyzing Organic Reactions
CHAPTER 4
ANALYZING ORGANIC REACTIONS
In This Chapter
4.1 Acids and Bases
Definitions
Acid and Base Strength
Common Functional Groups
4.2 Nucleophiles, Electrophiles, and Leaving Groups
Nucleophiles
Electrophiles
Leaving Groups
Nucleophilic Substitution Reactions
4.3 Oxidation–Reduction Reactions
Oxidizing Agents and Reactions
Reducing Agents and Reactions
4.4 Chemoselectivity
Reactive Locations
Steric Protection
4.5 Steps to Problem Solving
Example Reactions
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 5% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1A: Structure and function of proteins and their constituent amino acids
4E: Atoms, nuclear decay, electronic structure, and atomic chemical behavior
5A: Unique nature of water and its solutions
5D: Structure, function, and reactivity of biologically-relevant molecules
MCAT EXPERTISE
This chapter is vital for understanding all the reactions you’ll see in the next six chapters of this book (Chapters 5-10). On top of that, this material is directly tested on the MCAT as often as any other topic in organic chemistry. Make sure you know all of it!
Introduction
Sitting down to solve MCAT organic chemistry problems can be overwhelming at first, particularly if the reactants or reagents are new to you. The reactions on the page may seem like stage magic—fun to watch, but controlled by forces outside of our knowledge. The good news, however, is that organic chemistry isn’t magic—and it is governed by sets of rules that make understanding what will happen much simpler. In this chapter, we will go over several of the aspects that determine how a complex reaction proceeds and take a brief look at functional group reactivity. Then, armed with this knowledge, we lay out the simple, sequential steps that we can use to determine which reactions will take place. Chapters 5 through 10 of MCAT Organic Chemistry Review focus on applications of these principles with different functional groups, so look for patterns between these reactions as you continue preparing for Test Day. With these tools in hand, you can show the MCAT that you know how the trick is done—it isn’t magic.
4.1 Acids and Bases
LEARNING OBJECTIVES
After Chapter 4.1, you will be able to:
- Recall the importance of amphoteric species and common amphoteric molecules
- Describe the meaning of pKa and pKb values in relation to acid and base strength
- Recall common functional groups that act as acids or bases
In an acid–base reaction, an acid and a base react, resulting in the formation of the conjugate base of the acid and the conjugate acid of the base. This reaction proceeds so long as the reactants are more reactive, or stronger, than the products that they form. We will discuss acid and base definitions and strength in the following section. For the MCAT, we will concern ourselves with the broader Lewis and Brønsted–Lowry definitions of acids and bases. The Lewis definition concerns itself with the transfer of electrons in the formation of coordinate covalent bonds; the Brønsted–Lowry definition focuses on proton transfer.
KEY CONCEPT
An acid–base reaction will only proceed if the products that will be formed (the conjugate base of the acid and the conjugate acid of the base) are weaker than the original reactants.
Definitions
A Lewis acid is defined as an electron acceptor in the formation of a covalent bond. Lewis acids also tend to be electrophiles, which we will touch on in the next section. Lewis acids have vacant p-orbitals into which they can accept an electron pair, or are positively polarized atoms.
BRIDGE
Acids and bases are critically important material in organic chemistry, biochemistry, and general chemistry. The most extensive coverage of acids and bases is in Chapter 10 of MCAT General Chemistry Review.
A Lewis base is defined as an electron donor in the formation of a covalent bond. Lewis bases also tend to be nucleophiles, which we will touch on in the next section. Lewis bases have a lone pair of electrons that can be donated, and are often anions, carrying a negative charge.
When Lewis acids and bases interact, they form coordinate covalent bonds—covalent bonds in which both electrons in the bond came from the same starting atom (the Lewis base), as shown in Figure 4.1.
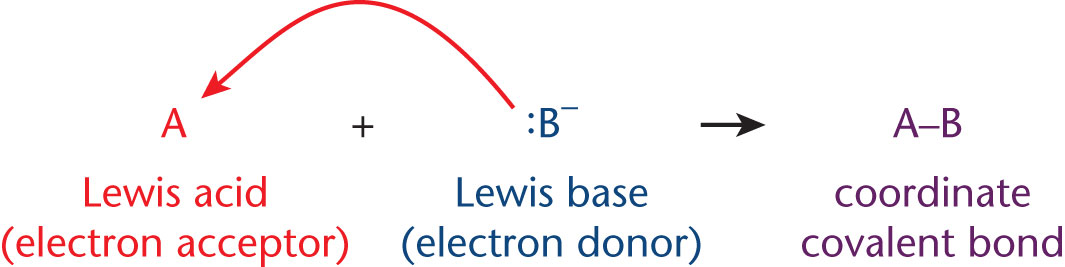
Figure 4.1. Lewis Acid–Base Reactions
In the Brønsted–Lowry definition, an acid is a species that can donate a proton (H+); a base is a species that can accept a proton. Some molecules, like water, have the ability to act as either Brønsted–Lowry acids or bases, making them amphoteric. Water can act as an acid by donating its proton to a base, and thus becoming its conjugate base, OH–. However, water can also act as a base by accepting a proton from an acid to become its conjugate acid, H3O+. The degree to which a molecule acts as an acid or a base is dependent upon the properties of the solution—water can only act as a base in an acidic solution, and only as an acid in a basic solution. Other examples of amphoteric molecules include Al(OH)3, HCO3−, and HSO4−.
Acid and Base Strength
The acid dissociation constant, or Ka, measures the strength of an acid in solution. In the dissociation of an acid HA (HA ⇌ H+ + A–), the equilibrium constant is given by:
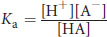
Equation 4.1
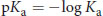
Equation 4.2
Thus, more acidic molecules will have a smaller (or even negative) pKa; more basic molecules will have a larger pKa. Acids with a pKa below –2 are considered strong acids, which almost always dissociate completely in aqueous solution. Weak organic acids often have pKa values between –2 and 20. pKa values for common functional groups are shown in Table 4.1.
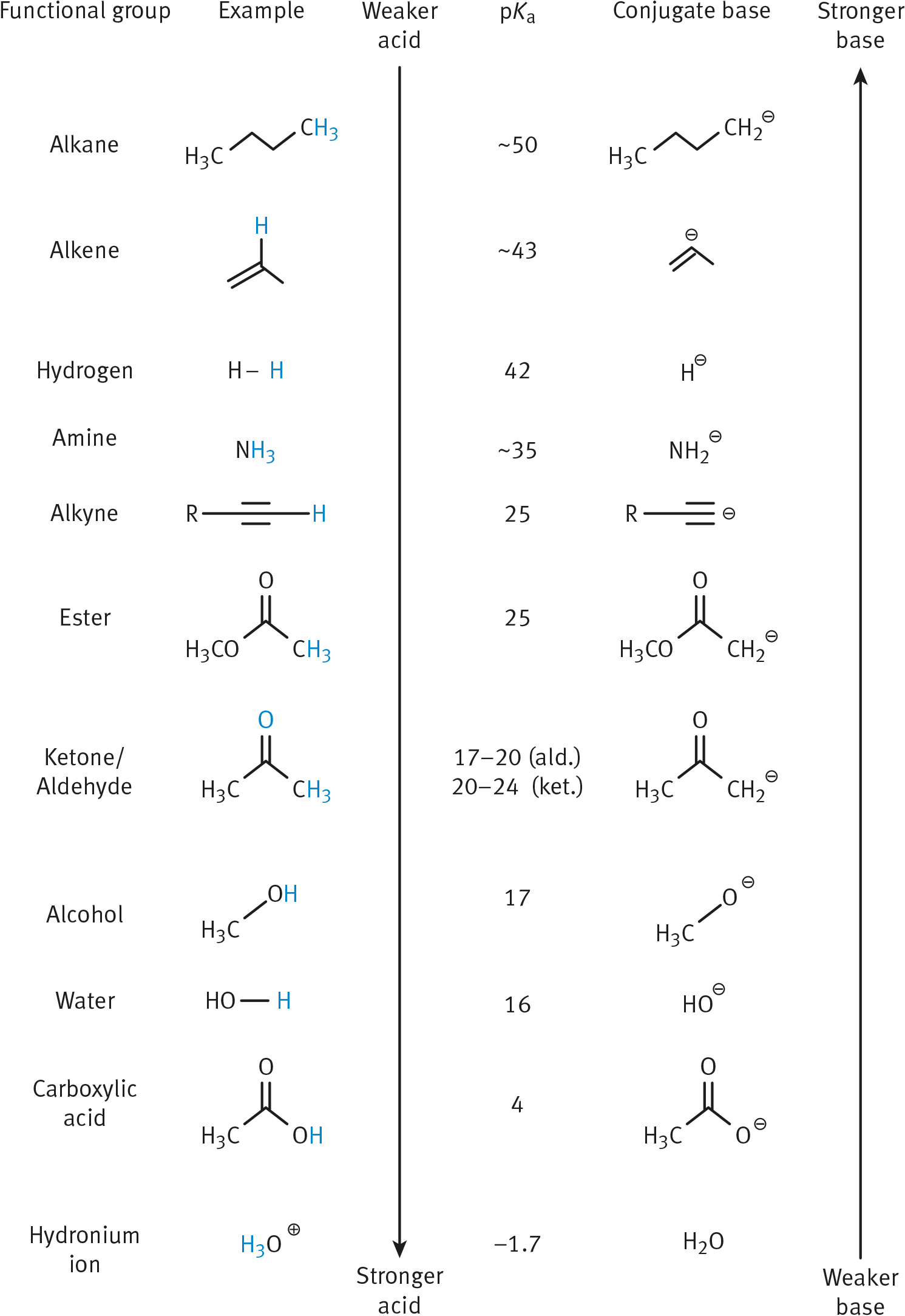
Table 4.1. pKa Values for Common Functional Groups
Generally, bond strength decreases down the periodic table, and acidity therefore increases. Also, the more electronegative an atom, the higher the acidity. When these two trends oppose each other, low bond strength takes precedence.
For the common functional groups on the MCAT, the α-hydrogens of carbonyl compounds deserve special note. α-hydrogens are connected to the α-carbon, which is a carbon adjacent to the carbonyl. Because the enol form of carbonyl-containing carbanions is stabilized by resonance, these are acidic hydrogens that are easily lost. We will go into greater depth about enolate chemistry in Chapters 7 through 9 of MCAT Organic Chemistry Review.
Common Functional Groups
We can also apply these acid and base rules directly to the functional groups that appear on the MCAT. Functional groups that act as acids include alcohols, aldehydes and ketones (at the α-carbon), carboxylic acids, and most carboxylic acid derivatives. These compounds are therefore easier to target with basic (or nucleophilic) reactants because they readily accept a lone pair.
Amines and amides are the main functional groups that act as bases—keep an eye out for these compounds in the formation of peptide bonds. The nitrogen atom of an amine can form coordinate covalent bonds by donating a lone pair to a Lewis acid.
MCAT CONCEPT CHECK 4.1:
Before you move on, assess your understanding of the material with these questions.
-
When will an acid–base reaction proceed, based on the strength of the reactants and products?
______________________________________
-
What does it mean for a molecule to be amphoteric? What biologically relevant molecules are also characteristically amphoteric?
______________________________________
-
How is pKa defined, and what does a low pKa indicate?
______________________________________
-
What are some functional groups that classically act as acids? As bases?
- Acids:
______________________________________
- Bases:
______________________________________
4.2 Nucleophiles, Electrophiles, and Leaving Groups
LEARNING OBJECTIVES
After Chapter 4.2, you will be able to:
- Distinguish nucleophiles and electrophiles from Lewis acids and bases
- Compare nucleophilicity using the four main trends
- Describe the relationship between electrophile and leaving group in a substitution reaction
- Identify the traits that increase electrophilicity
- Recall the traits of a good leaving group
Almost all reactions in organic chemistry can be divided into one of two groups: oxidation–reduction reactions or nucleophile–electrophile reactions. Nucleophiles, electrophiles, and leaving groups are particularly important to the reactions of alcohols and carbonyl-containing compounds, which we will look at in depth in later chapters. Let’s take a look at how each of these terms is defined.
Nucleophiles
Nucleophiles are defined as “nucleus-loving” species with either lone pairs or π bonds that can form new bonds to electrophiles. You may have noted that nucleophilicity and basicity appear to have similar definitions—and this is true! Good nucleophiles tend to be good bases. There is, however, a distinction between the two. Nucleophile strength is based on relative rates of reaction with a common electrophile—and is therefore a kinetic property. Base strength is related to the equilibrium position of a reaction—and is therefore a thermodynamic property. Some common examples of nucleophiles are shown in Figure 4.2.
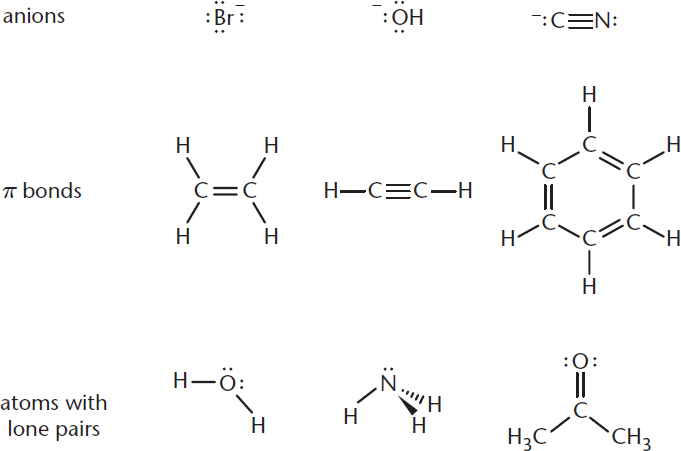
Figure 4.2. Examples of Nucleophiles
KEY CONCEPT
Nucleophiles tend to have lone pairs or π bonds that can be used to form covalent bonds to electrophiles. On Test Day, look for carbon, hydrogen, oxygen, or nitrogen (CHON) with a minus sign or lone pair to identify most nucleophiles.
As long as the nucleophilic atom is the same, the more basic the nucleophile, the more reactive it is. This also holds when comparing atoms in the same row of the periodic table, but not when proceeding down a column in the periodic table. Nucleophilicity is determined by four major factors:
- Charge: Nucleophilicity increases with increasing electron density (more negative charge)
- Electronegativity: Nucleophilicity decreases as electronegativity increases because these atoms are less likely to share electron density
- Steric hindrance: Bulkier molecules are less nucleophilic
- Solvent: Protic solvents can hinder nucleophilicity by protonating the nucleophile or through hydrogen bonding
Solvent Effects
The solvent consideration is worth spending a bit more time on. In polar protic solvents, nucleophilicity increases down the periodic table. In polar aprotic solvents, nucleophilicity increases up the periodic table. Examples of both types of solvents are shown in Figure 4.3.
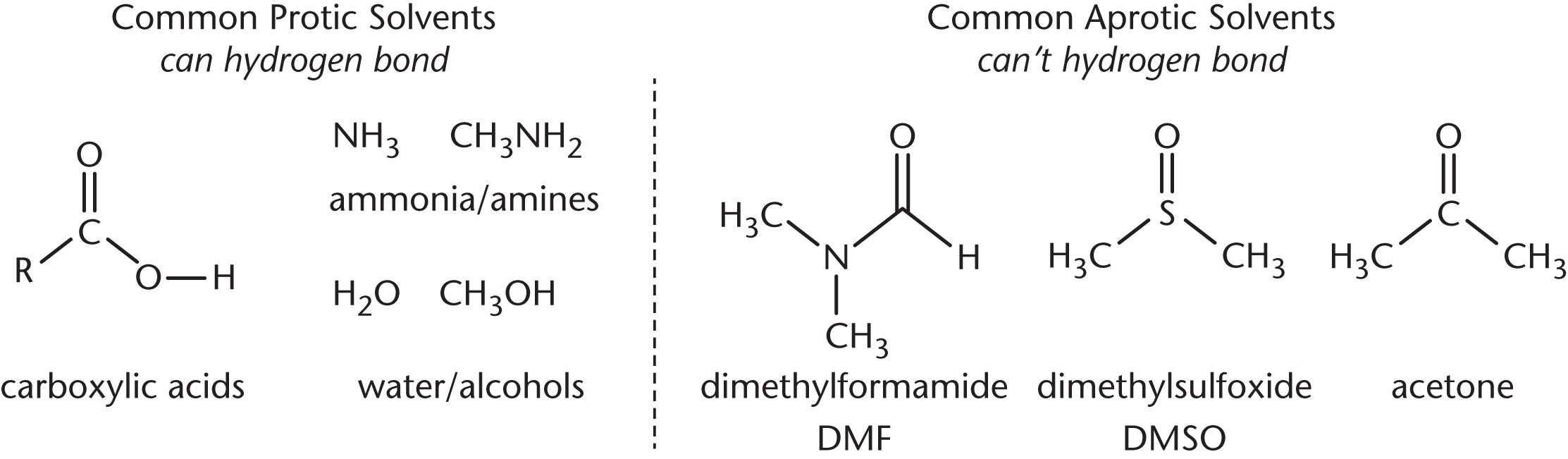
Figure 4.3. Examples of Polar Protic and Polar Aprotic Solvents
The halogens are good examples of the effects of the solvent on nucleophilicity. In protic solvents, nucleophilicity decreases in the order:
I– > Br– > Cl– > F–
This is because the protons in solution will be attracted to the nucleophile. F– is the conjugate base of HF, a weak acid. As such, it will form bonds with the protons in solution and be less able to access the electrophile to react. I–, on the other hand, is the conjugate base of HI, a strong acid. As such, it is less affected by the protons in solution and can react with the electrophile.
MCAT EXPERTISE
If a solvent is not given on Test Day, assume that the reaction occurs in a polar solvent. Polar solvents—whether protic or aprotic—can dissolve nucleophiles and assist in any reaction in which electrons are moved. Organic chemistry is all about moving electrons, so it’s less common for these reactions to be performed in nonpolar solvents.
In aprotic solvents, on the other hand, nucleophilicity decreases in the order:
F– > Cl– > Br– > I–
This is because there are no protons to get in the way of the attacking nucleophile. In aprotic solvents, nucleophilicity relates directly to basicity.
We won’t use nonpolar solvents with this type of reaction because we need our nucleophile to dissolve. Because charged molecules are polar by nature, a polar solvent is required to dissolve the nucleophile as well because like dissolves like . Examples of strong nucleophiles include HO−, RO−, CN−, and N3−. NH3 and RCO2− are fair nucleophiles, and H2O, ROH, and RCOOH are weak or very weak nucleophiles. As far as functional groups go, amine groups tend to make good nucleophiles.
KEY CONCEPT
We can’t use nonpolar solvents in these nucleophile–electrophile reactions because our reactants are polar—they wouldn’t dissolve!
Electrophiles
Electrophiles are defined as “electron-loving” species with a positive charge or positively polarized atom that accepts an electron pair when forming new bonds with a nucleophile. Again, this definition brings to mind Lewis acids. The distinction, as with nucleophiles and bases above, is that electrophilicity is a kinetic property, whereas acidity is a thermodynamic property. Practically, however, electrophiles will almost always act as Lewis acids in reactions. A greater degree of positive charge increases electrophilicity, so a carbocation is more electrophilic than a carbonyl carbon. Some comparisons between electrophiles are drawn in Figure 4.4. Additionally, the nature of the leaving group influences electrophilicity in species without empty orbitals; better leaving groups make it more likely that a reaction will happen. If empty orbitals are present, an incoming nucleophile can make a bond with the electrophile without displacing the leaving group.
Electrophilicity and acidity are effectively identical properties when it comes to reactivity. Just as alcohols, aldehydes and ketones, carboxylic acids, and their derivatives act as acids, they also act as electrophiles and can make good targets for nucleophilic attack.

Figure 4.4. Comparisons of Electrophilicity
The carboxylic acid derivatives are often ranked by electrophilicity. Anhydrides are the most reactive, followed by carboxylic acids and esters, and then amides. In practical terms, this means that derivatives of higher reactivity can form derivatives of lower reactivity but not vice versa, similar to the acid–base reactions described previously.
Leaving Groups
Leaving groups are the molecular fragments that retain the electrons after heterolysis. Heterolytic reactions are essentially the opposite of coordinate covalent bond formation: a bond is broken and both electrons are given to one of the two products. The best leaving groups will be able to stabilize the extra electrons. Weak bases are more stable with an extra set of electrons and therefore make good leaving groups. By this logic, the conjugate bases of strong acids (like I–, Br–, and Cl–) tend to make good leaving groups. Leaving group ability can be augmented by resonance and by inductive effects from electron-withdrawing groups: these help delocalize and stabilize negative charge.
Alkanes and hydrogen ions will almost never serve as leaving groups because they form very reactive, strongly basic anions. We can think of leaving groups and nucleophiles as serving opposite functions. In substitution reactions, the weaker base (the leaving group) is replaced by the stronger base (the nucleophile).
KEY CONCEPT
Just like acid–base reactions, nucleophilic attack will only occur if the reactants are more reactive than the products. Thus, the nucleophile must be more reactive than the leaving group.
Nucleophilic Substitution Reactions
Nucleophilic substitution reactions are perfect examples for demonstrating nucleophile–electrophile reactions. In both SN1 and SN2 reactions, a nucleophile forms a bond with a substrate carbon and a leaving group leaves.
SN1 Reactions
Unimolecular nucleophilic substitution (SN1) reactions contain two steps. The first step is the rate-limiting step in which the leaving group leaves, generating a positively charged carbocation. The nucleophile then attacks the carbocation, resulting in the substitution product. This mechanism is shown in Figure 4.5 below.
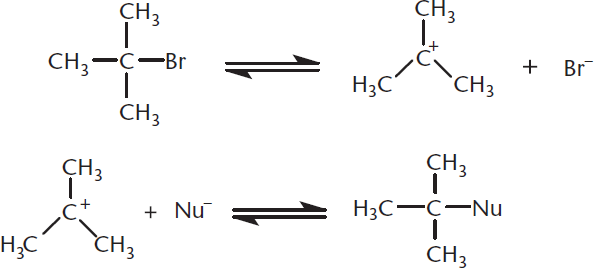
Figure 4.5. Mechanism of SN1 Reaction Step 1: Formation of the carbocation (rate-limiting); Step 2: Nucleophilic attack.
The more substituted the carbocation, the more stable it is because the alkyl groups act as electron donors, stabilizing the positive charge. Because the formation of the carbocation is the rate-limiting step, the rate of the reaction depends only on the concentration of the substrate: rate=k[R–L], where R–L is an alkyl group containing a leaving group. This is a first-order reaction; anything that accelerates the formation of the carbocation will increase the rate of an SN1 reaction.
Because SN1 reactions pass through a planar intermediate before the nucleophile attacks, the product will usually be a racemic mixture. The incoming nucleophile can attack the carbocation from either side, resulting in varied stereochemistry.
SN2 Reactions
Bimolecular nucleophilic substitution (SN2) reactions contain only one step, in which the nucleophile attacks the compound at the same time as the leaving group leaves. Because this reaction has only one step, we call it a concerted reaction. The reaction is called bimolecular because this single rate-limiting step involves two molecules.
In SN2 reactions, the nucleophile actively displaces the leaving group in a backside attack. For this to occur, the nucleophile must be strong, and the substrate cannot be sterically hindered. Therefore, the less substituted the carbon, the more reactive it is in SN2 reactions. Note that this is the opposite of the trend for SN1 reactions. The one-step mechanism is shown in Figure 4.6.
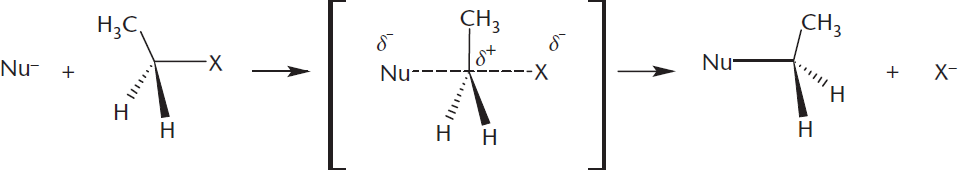
Figure 4.6. Mechanism of SN2 Reaction
The single step of an SN2 reaction involves two reacting species: the substrate (often an alkyl halide, tosylate, or mesylate) and the nucleophile. Therefore, the concentrations of both have a role in determining the rate: rate = k[Nu:][R–L]
SN2 reactions are accompanied by an inversion of relative configuration. Much like an umbrella being turned inside out on a blustery day, the position of substituents around the substrate carbon will be inverted. If the nucleophile and leaving group have the same priority in their respective molecules, this inversion will also correspond to a change in absolute configuration from (R) to (S) or vice-versa. This is an example of a stereospecific reaction, one in which the configuration of the reactant determines the configuration of the product due to the reaction mechanism.
MCAT CONCEPT CHECK 4.2:
Before you move on, assess your understanding of the material with these questions.
-
How do the definitions of nucleophile and electrophile differ from those of Lewis base and acid?
________________________
-
Rank the following molecules in order of increasing nucleophilicity: methoxide, t-butoxide, isopropanolate, ethoxide.
_______< _____< _______< ____________.
-
How must the nucleophile and leaving group be related in order for a substitution reaction to proceed?
________________________
-
What trends increase electrophilicity?
________________________
-
What are some features of good leaving groups?
________________________
4.3 Oxidation–Reduction Reactions
LEARNING OBJECTIVES
After Chapter 4.3, you will be able to:
- Recall common oxidizing agents and the characteristics of a good oxidizing agent
- Recall common reducing agents and the characteristics of a good reducing agent
- Order a given list of molecules from most oxidized to least oxidized:
Another important class of reactions are oxidation–reduction (redox) reactions, in which the oxidation states of the reactants change. Oxidation state is an indicator of the hypothetical charge that an atom would have if all bonds were completely ionic. Oxidation state can be calculated from the molecular formula for a molecule. For example, the carbon in methane (CH4) has an oxidation state of –4 because the hydrogens each have an oxidation state of +1. This is the most reduced form of carbon. In carbon dioxide (CO2), each of the oxygen atoms has an oxidation state of –2, and the carbon has an oxidation state of +4. This is the most oxidized form of carbon. For an ion, the oxidation state is simply the charge—so Na+ and S2– would have oxidation states of +1 and –2, respectively. Carboxylic acids are more oxidized than aldehydes, ketones, and imines, which in turn are more oxidized than alcohols, alkyl halides, and amines.
We won’t need to know too much about how to assign oxidation states in organic chemistry, but should know the definitions of oxidation and reduction. Oxidation refers to an increase in oxidation state, which means a loss of electrons. In organic chemistry, it is often easier to view oxidation as increasing the number of bonds to oxygen or other heteroatoms (atoms besides carbon and hydrogen). Reduction refers to a decrease in oxidation state, or a gain in electrons. In organic chemistry, it is often easier to view reduction as increasing the number of bonds to hydrogen.
Oxidizing Agents and Reactions
As we mentioned above, oxidation refers to an increase in oxidation state. Oxidation of a carbon atom occurs when a bond between a carbon atom and an atom that is less electronegative than carbon is replaced by a bond to an atom that is more electronegative than carbon. In practice, this usually means decreasing the number of bonds to hydrogen and increasing the number of bonds to other carbons, nitrogen, oxygen, or halides.
KEY CONCEPT
One can organize the different functional groups by “levels” of oxidation:
- Level 0 (no bonds to heteroatoms): alkanes
- Level 1: alcohols, alkyl halides, amines
- Level 2: aldehydes, ketones, imines
- Level 3: carboxylic acids, anhydrides, esters, amides
- Level 4 (four bonds to heteroatoms): carbon dioxide
The oxidizing agent is the element or compound in an oxidation–reduction reaction that accepts an electron from another species. Because the oxidizing agent is gaining electrons, it is said to be reduced. Good oxidizing agents have a high affinity for electrons (such as O2, O3, and Cl2) or unusually high oxidation states (like Mn7+ in permanganate, MnO4−, and Cr6+ in chromate, CrO42−).
Primary alcohols can be oxidized by one level to become aldehydes, or can be further oxidized to form carboxylic acids. This reaction commonly proceeds all the way to the carboxylic acid when using strong oxidizing agents such as chromium trioxide (CrO3) or sodium or potassium dichromate (Na2Cr2O7 or K2Cr2O7), but can be made to stop at the aldehyde level using specific reagents such as pyridinium chlorochromate (PCC). Secondary alcohols can be oxidized to ketones.
A number of oxidation reactions and the relevant oxidizing agents are shown in Figure 4.7. Note that the goal at this point should not be memorization of these reactions, but recognition of two themes: oxidation reactions tend to feature an increase in the number of bonds to oxygen, and oxidizing agents often contain metals bonded to a large number of oxygen atoms.
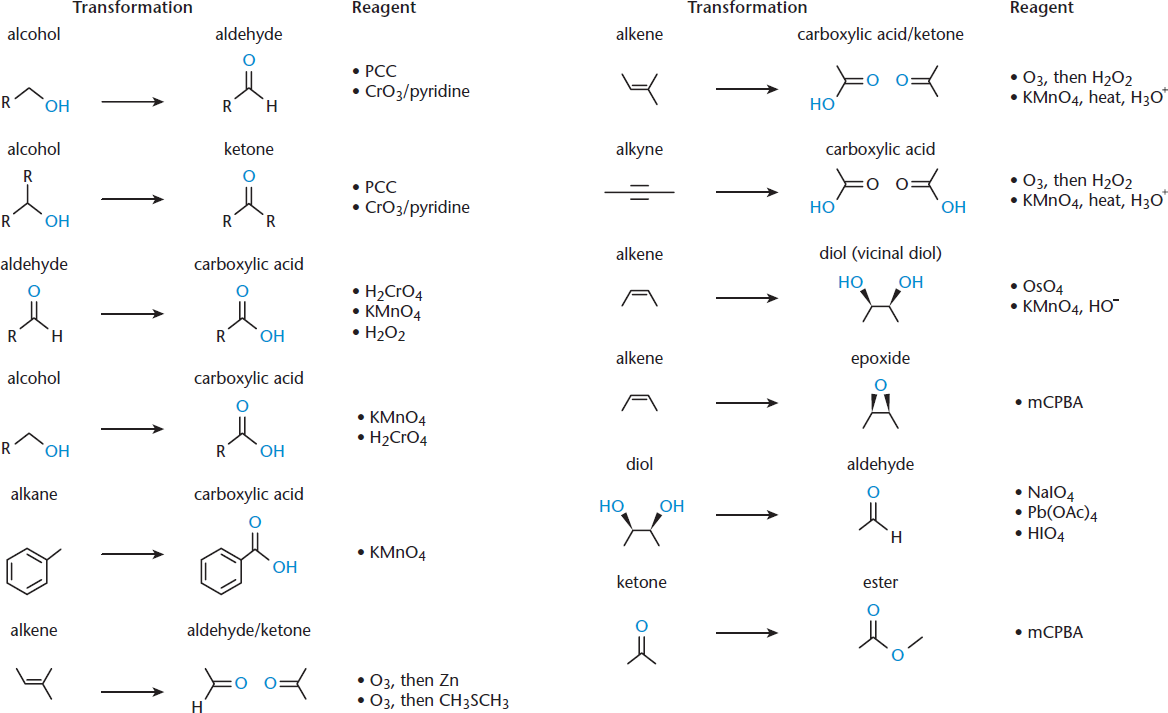
Figure 4.7. Oxidation Reactions and Common Oxidizing Agents
Reducing Agents and Reactions
Conversely, reduction refers to a decrease in oxidation state. Reduction of a carbon occurs when a bond between a carbon atom and an atom that is more electronegative than carbon is replaced by a bond to an atom that is less electronegative than carbon. In practice, this usually means increasing the number of bonds to hydrogen and decreasing the number of bonds to other carbons, nitrogen, oxygen, or halides.
Good reducing agents include sodium, magnesium, aluminum, and zinc, which have low electronegativities and ionization energies. Metal hydrides, such as NaH, CaH2, LiAlH4, and NaBH4, are also good reducing agents because they contain the H– ion.
Aldehydes and ketones will be reduced to primary and secondary alcohols, respectively. This reaction is exergonic, but exceedingly slow without a catalyst. Amides can be reduced to amines using LiAlH4. This same reducing agent will reduce carboxylic acids to primary alcohols and esters to a pair of alcohols. Examples of reduction reactions are shown in Figure 4.8. Again, the focus is not on memorization, but on recognizing that reduction reactions tend to feature an increase in the number of bonds to hydrogen, and reducing agents often contain metals bonded to a large number of hydrides.
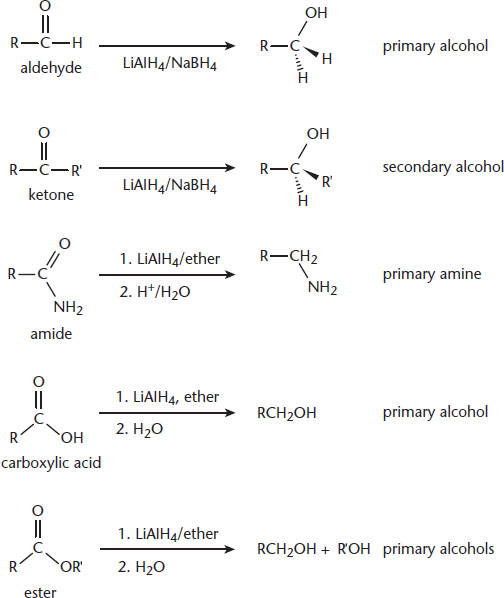
Figure 4.8. Reduction Reactions and Common Reducing Agents
BRIDGE
Note that many of the common oxidizing and reducing agents include transition metals. This is because transition metals can often take on many different oxidation states. Their low ionization energies and presence of d-orbitals allow them to give up and accept electrons easily. Transition metals and periodic trends are discussed in Chapter 2 of MCAT General Chemistry Review.
MCAT CONCEPT CHECK 4.3:
Before you move on, assess your understanding of the material with these questions.
-
What are some characteristics of good oxidizing agents? List some examples of common oxidizing agents.
- Characteristics:
________________________
- Examples:
________________________
-
What are some characteristics of good reducing agents? List some examples of common reducing agents.
- Characteristics:
________________________
- Examples:
________________________
-
List the following carbon-containing compounds from most oxidized carbon to most reduced: methane, carbon dioxide, ketone, alcohol, carboxylic acid
- Most oxidized: ___________________
- ______________________________________
- ______________________________________
- ______________________________________
- Most reduced: ___________________
4.4 Chemoselectivity
LEARNING OBJECTIVES
After Chapter 4.4, you will be able to:
- Describe the types of compounds that are most likely to undergo SN1 and SN2 reactions
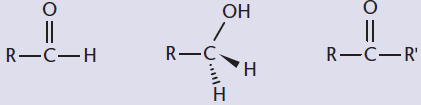
- Identify the two reactive centers of a carbonyl-containing compound:
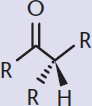
A key skill in recognizing which reactions will occur is recognizing the reactive regions within a molecule. The preferential reaction of one functional group in the presence of other functional groups is termed chemoselectivity.
Reactive Locations
Which site is the reactive site of a molecule depends on the type of chemistry that’s occurring. A redox reagent, as described earlier, will tend to act on the highest-priority functional group. Thus, in a molecule with an alcohol and a carboxylic acid, a reducing agent is more likely to act on the carboxylic acid than on the alcohol. For a reaction involving nucleophiles and electrophiles, reactions also tend to occur at the highest-priority functional group because it contains the most oxidized carbon. A nucleophile is looking for a good electrophile; the more oxidized the carbon, the more electronegative groups around it, and the larger partial positive charge it will experience. Thus, carboxylic acids and their derivatives are the first to be targeted by a nucleophile, followed by an aldehyde or ketone, followed by an alcohol or amine. Aldehydes are generally more reactive toward nucleophiles than ketones because they have less steric hindrance.
KEY CONCEPT
The more oxidized the functional group, the more reactive it is in both nucleophile–electrophile and oxidation–reduction reactions.
One common reactive site on the MCAT is the carbon of a carbonyl, which can be found in carboxylic acids and their derivatives, aldehydes, and ketones. Within a carbonyl-containing compound, the carbon of the carbonyl acquires a positive polarity due to the electronegativity of the oxygen. Thus, the carbonyl carbon becomes electrophilic and can be a target for nucleophiles. Further, the α-hydrogens are much more acidic than in a regular C–H bond due to the resonance stabilization of the enol form. These can be deprotonated easily with a strong base, forming an enolate, as shown in Figure 4.9. The enolate then becomes a strong nucleophile, and alkylation can result if good electrophiles are available.
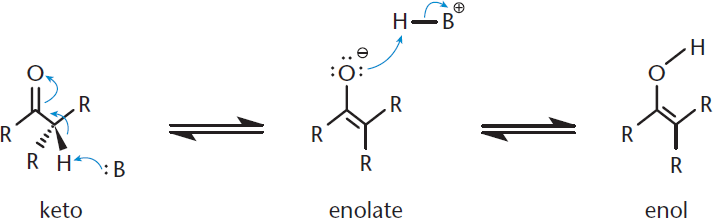
Figure 4.9. Enol and Enolate Forms of a Ketone
A second reactive site for consideration is the substrate carbon in substitution reactions. SN1 reactions, which have to overcome the barrier of carbocation stability, prefer tertiary to secondary carbons as reactive sites, and secondary to primary. For SN2 reactions, which have a bigger barrier in steric hindrance, methyl and primary carbons are preferred over secondary, and tertiary carbons won’t react. This is all because of the mechanism of these two reactions.
Steric Protection
Steric hindrance describes the prevention of reactions at a particular location within a molecule due to the size of substituent groups. For example, SN2 reactions won’t occur with tertiary substrates. This characteristic of steric protection can be a useful tool in the synthesis of desired molecules and the prevention of the formation of alternative products. Effectively, bulky groups make it impossible for the nucleophile to reach the most reactive electrophile, making the nucleophile more likely to attack another region.
Another way that sterics come into play is in the protection of leaving groups. One can temporarily mask a reactive leaving group with a sterically bulky group during synthesis. For example, reduction of a molecule containing both carboxylic acids and aldehydes or ketones can result in reduction of all of the functional groups. To prevent this, the aldehyde or ketone is first converted to a nonreactive acetal or ketal, which serves as the protecting group, and the reaction can proceed. This reaction is shown in Figure 4.10. Another protective reaction is the reversible reduction of alcohols to tert-butyl ethers.
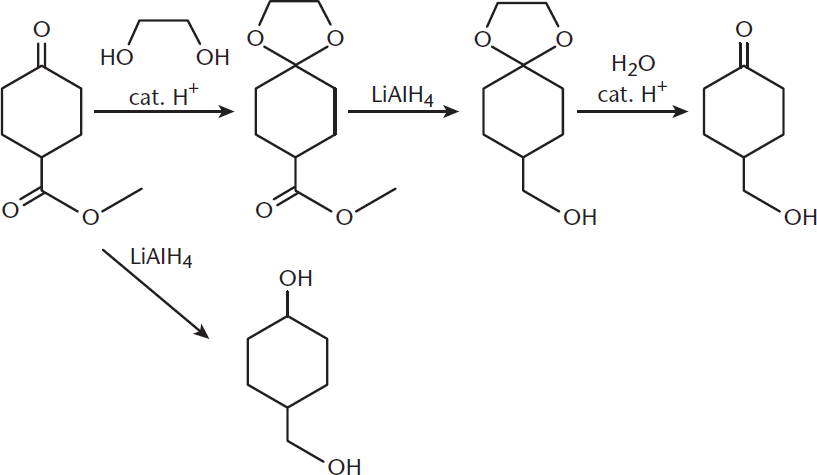
Figure 4.10. Protection of a Ketone by Conversion to an Acetal
BRIDGE
When an aldehyde is mixed with a diol (or two equivalents of alcohol), it forms an acetal. When a ketone is mixed with a diol (or two equivalents of alcohol), it forms a ketal. Acetal and ketal chemistry is discussed in Chapter 6 of MCAT Organic Chemistry Review.
Don’t worry if this seems overwhelming—this is just a preview of what we will see in later chapters, along with a set of rules that will make it easier to understand how chemical reactions will proceed! Feel free to come back to this chapter later to remind yourself of the rules that apply across the board after reading further chapters.
MCAT CONCEPT CHECK 4.4:
Before you move on, assess your understanding of the material with these questions.
-
What are the two reactive centers of carbonyl-containing compounds?
- ____________________________
- ____________________________
-
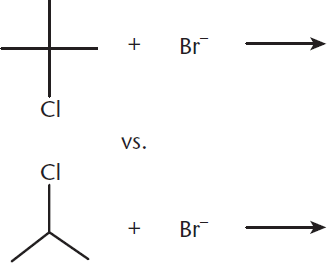
Which pair of reactants will undergo SN1 more quickly? Why?
________________________
-
Which of the following electrophiles will be more favorable to an SN2 reaction? Why?
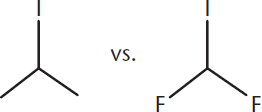
________________________
4.5 Steps to Problem Solving
LEARNING OBJECTIVES
After Chapter 4.5, you will be able to:
- List the six steps for solving organic chemistry reactions
- Predict how a reaction will proceed if you are not given reaction conditions
With all of these rules in hand, we can now apply our knowledge in a systematic way to simplify organic chemistry reactions that appear on the MCAT. These steps are described below.
MCAT EXPERTISE
While studying organic chemistry for the MCAT, don’t permit yourself to simply nod along with the reaction mechanisms—get involved! During each step of a mechanism, ask yourself how the trends and problem-solving steps in this chapter play out.
Step 1: Know Your Nomenclature
Before you can even start to understand what reactions will occur and what products will form, it is vital to know which compounds IUPAC and common names refer to! If you’re still having trouble with nomenclature, be sure to review Chapter 1 of MCAT Organic Chemistry Review.
Step 2: Identify the Functional Groups
Look at the organic molecules in the reaction. What functional groups are in the molecules? Do these functional groups act as acids or bases? How oxidized is the carbon? Are there functional groups that act as good nucleophiles, electrophiles, or leaving groups? This step will help define a category of reactions that can occur with the given functional groups.
Step 3: Identify the Other Reagents
In this step, determine the properties of the other reagents in the reaction. Are they acidic or basic? Are they suggestive of a particular reaction? Are they good nucleophiles or a specific solvent? Are they good oxidizing or reducing agents?
Step 4: Identify the Most Reactive Functional Group(s)
Once you’ve identified the functional groups in the compound and the other reagents present, this step should be relatively quick. Remember that more oxidized carbons tend to be more reactive to both nucleophile–electrophile reactions and oxidation–reduction reactions. Note the presence of protecting groups that exist to prevent a particular functional group from reacting.
Step 5: Identify the First Step of the Reaction
If the reaction involves an acid or a base, the first step will usually be protonation or deprotonation. If the reaction involves a nucleophile, the first step is generally for the nucleophile to attack the electrophile, forming a bond with it. If the reaction involves an oxidizing or reducing agent, the most oxidized functional group will be oxidized or reduced, accordingly.
Once you know what will react, think through how the reaction will go. Did the protonation or deprotonation of a functional group increase its reactivity? When the nucleophile attacks, how does the carbon respond to avoid having five bonds? Does a leaving group leave, or does a double bond get reduced to a single bond (like the opening of a carbonyl)?
Step 6: Consider Stereospecificity/ Stereoselectivity
Though not all reactions are stereospecific or stereoselective, these possibilities should be considered when predicting products. For stereospecificity, consider whether the configuration of the reactant necessarily leads to a specific configuration in the product, as seen in SN2 reactions. Stereoselectivity, on the other hand, occurs in reactions where one configuration of product is more readily formed due to product characteristics. Stereoselectivity is seen in many reactions, as different products often possess different traits which affect their relative stability. If there is more than one product, the major product will generally be determined by differences in strain or stability between the two molecules. More strained molecules (with significant angle, torsional, or nonbonded strain) are less likely to form than molecules without significant sources of strain. Products with conjugation (alternating single and multiple bonds) are significantly more stable than those without.
Example Reactions
Now, we’ll apply these rules to three novel reactions. Focus on the decision-making element of this process so that you will be able to apply the same logic to reactions that appear on Test Day.
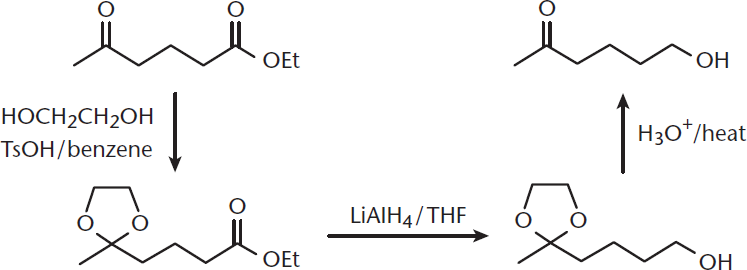
Reaction 1
We’ll start with a series of reactions involving ethyl 5-oxohexanoate. First, it is reacted with 1,2-ethanediol and p-toluenesulfonic acid in benzene; second, with lithium aluminum hydride in tetrahydrofuran, followed by a heated acidic workup. What are the intermediates and final product?
Let’s go through the steps:
- First, let’s draw out the reactants and reaction conditions.

- This molecule has an alkane backbone, a ketone, and an ester. Both of the carbonyl carbons are electrophilic targets for nucleophiles. The carbonyl oxygens can also be reduced. Acidic α-hydrogens are also present.
- For the first part of the reaction, we have a diol, which is commonly used as a protecting group for aldehydes or ketones. Diols are nucleophiles because of lone pairs on the oxygens in the hydroxyl groups. For the second reaction, we have a reducing agent in an organic solvent. Finally, we have an acidic workup—which is often used to remove protecting groups. We’re starting to get hints as to what is happening here.
- In the first reaction, both the ketone and ester carbonyls are highly reactive. One or both of these functional groups will react in the first step.
- The diol is a good nucleophile because it contains lone pairs on the oxygen atoms in the hydroxyl groups. Further, the presence of a diol hints at protecting the ketone carbonyl because diols are commonly used for this function. This gives our first intermediate—the ketone carbonyl will be replaced by a protected diether. The second reaction, then, will only be able to proceed on the ester. LiAlH4 is a strong reducing agent, so the next reaction will be reduction of the carbonyl all the way to an alcohol. In the final reaction, the protecting group will be removed by acidic workup, leaving us with our original ketone group.
- The product and intermediate have no stereoselectivity, so this won’t be a consideration.
Let’s see what we came up with. The first intermediate will have a protective diether at the ketone carbonyl. The second will show the reduction of the ester to an alcohol, with the protecting group still present. The third will be our final product.
Reaction 2
If ethanol is reacted in acidic solution with potassium dichromate, what will the end product be?
Let’s go through the steps again.
- First, let’s draw out our molecules.
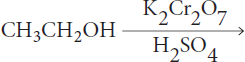
- This molecule has an alkane backbone and a primary alcohol. Alcohols make good nucleophiles and can also be oxidized. The hydroxyl group can also act as a leaving group, especially if it gets protonated.
- Next, the reagents. Dichromate is a good oxidizing agent.
- The alcohol carbon is most likely to react because it is the most oxidized.
- The primary product of a primary alcohol with a strong oxidant like dichromate will be a carboxylic acid. One other possible product could have been an aldehyde, so this could trip us up! Remember, however, that primary alcohols can only be oxidized to aldehydes by reagents specifically designated for this purpose, like pyridinium chlorochromate (PCC). If we start with ethanol, we’ll obtain ethanoic acid (acetic acid) after reaction with dichromate.
- Stereospecificity again isn’t a consideration and won’t change the outcome in this reaction.
Therefore, the primary product of this reaction will be ethanoic acid.
Reaction 3
Determine the product of a reaction between 2-amino-3-hydroxypropanoic acid and 2,6-diaminohexanoic acid in aqueous solution.
Let’s go through the steps one last time.
- First, let’s draw out both molecules.
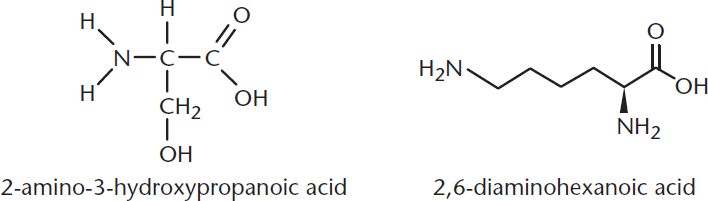
- Both of these molecules have a carboxylic acid (which has an acidic hydrogen and an electrophilic carbonyl carbon) and an amino group (which is nucleophilic). The first molecule also has a hydroxyl group; the second has an additional amino group and a long alkane chain.
- There are no additional reagents listed. Therefore, it will be the properties of the two reactants alone that determine how the reaction will proceed.
- Either of the two molecules could act as the nucleophile in this reaction, and either could be the electrophile. The most reactive species are likely the nucleophilic amino groups attacking the electrophilic carbonyl carbon.
- The first step of this reaction will be nucleophilic attack by the amino group on the electrophilic carbonyl carbon. Carbon cannot have five bonds, so the carbonyl group will have to open up. The hydroxyl group on the carboxylic acid is a poor leaving group, but proton rearrangements in the molecule turn the hydroxyl group into water, improving its leaving group ability. Then, the carbonyl will reform, kicking off the water molecule as a leaving group.
- We might ask why the hydroxyl group on 2-amino-3-hydroxypropanoic acid doesn’t react. Remember that more oxidized groups tend to be more reactive, and the carboxylic acid is significantly more oxidized than the hydroxyl group. Another question to consider is which of the amino groups of 2,6-diaminohexanoic acid will react. This is a question that is perhaps best answered retrospectively; in this case, the amino group closer to the carbonyl will react because the resulting product will be stabilized by resonance.
Does this reaction look familiar? It should! 2-amino-3-hydroxypropanoic acid and 2,6-diaminohexanoic acid are serine and lysine, respectively—in this reaction, we are forming a peptide bond. If we treat them as generic amino acids, this is the reaction:
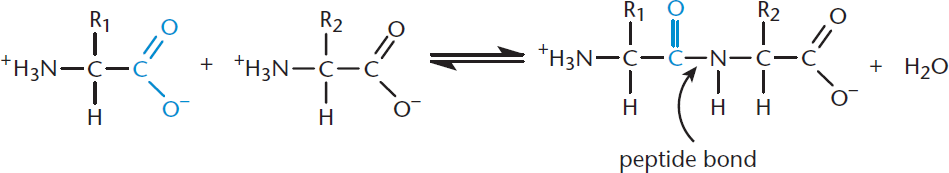
We’ve worked through a few problems here to get a handle on how to use this method. Once you have read further chapters and learned specific mechanisms that we did not touch on here, be sure to come back and see how these rules apply to novel reactions.
MCAT CONCEPT CHECK 4.5:
Before you move on, assess your understanding of the material with these questions.
-
What are the six steps for solving organic chemistry reactions?
-
___________________________
-
___________________________
-
___________________________
-
___________________________
-
___________________________
-
___________________________
-
If there are no reaction conditions listed, what determines how the reaction will proceed?
________________________
Conclusion
In this chapter, we’ve outlined a framework for thinking through organic chemistry questions on the MCAT. We have discussed the various types of reactions that we may see, the properties that make them more or less likely to occur, and a few selective rules that can help us work through reactions, even if they are unfamiliar. Finally, we put this framework to the test on example problems in order to cement its application. With this framework in mind, there’s nothing you can’t tackle—revisit these rules and methods as you continue working through the different functional groups and their reactions in the next six chapters!
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Acids and Bases
- Lewis acids are electron acceptors; they have vacant orbitals or positively polarized atoms.
- Lewis bases are electron donors; they have a lone pair of electrons and are often anions.
- Brønsted–Lowry acids are proton donors; Brønsted–Lowry bases are proton acceptors.
- Amphoteric molecules can act as either acids or bases, depending on reaction conditions. Water is a common example of an amphoteric molecule.
- The acid dissociation constant, Ka, is a measure of acidity. It is the equilibrium constant corresponding to the dissociation of an acid, HA, into a proton (H+) and its conjugate base (A–).
- pKa is the negative logarithm of Ka. A lower (or even negative) pKa indicates a stronger acid.
- pKa decreases down the periodic table and as electronegativity increases.
- Alcohols, aldehydes, ketones, carboxylic acids, and carboxylic acid derivatives are common acidic functional groups. α-hydrogens (hydrogens connected to an α-carbon, a carbon adjacent to a carbonyl) are acidic.
- Amines and amides are common basic functional groups.
Nucleophiles, Electrophiles, and Leaving Groups
- Nucleophiles are “nucleus-loving” and contain lone pairs or π bonds. They have increased electron density and often carry a negative charge.
- Nucleophilicity is similar to basicity; however, nucleophilicity is a kinetic property, while basicity is thermodynamic.
- Charge, electronegativity, steric hindrance, and the solvent can all affect nucleophilicity.
- Amino groups are common organic nucleophiles.
- Electrophiles are “electron-loving” and contain a positive charge or are positively polarized.
- More positive compounds are more electrophilic.
- Alcohols, aldehydes, ketones, carboxylic acids, and their derivatives can act as electrophiles.
- Leaving groups are the molecular fragments that retain the electrons after heterolysis.
- The best leaving groups can stabilize additional charge through resonance or induction.
- Weak bases (the conjugate bases of strong acids) make good leaving groups.
- Alkanes and hydrogen ions are almost never leaving groups because they form reactive anions.
- Unimolecular nucleophilic substitution (SN1) reactions proceed in two steps.
- In the first step, the leaving group leaves, forming a carbocation, an ion with a positively charged carbon atom.
- In the second step, the nucleophile attacks the planar carbocation from either side, leading to a racemic mixture of products.
- SN1 reactions prefer more substituted carbons because the alkyl groups can donate electron density and stabilize the positive charge of the carbocation.
- The rate of an SN1 reaction is dependent only on the concentration of the substrate: rate = k[R–L].
- Bimolecular nucleophilic substitution (SN2) reactions proceed in one concerted step.
- The nucleophile attacks at the same time as the leaving group leaves.
- The nucleophile must perform a backside attack, which leads to an inversion of stereochemistry.
- The absolute configuration is changed—(R) to (S) and vice versa—if the incoming nucleophile and the leaving group have the same priority in the molecule.
- SN2 reactions prefer less-substituted carbons because the alkyl groups create steric hindrance and inhibit the nucleophile from accessing the electrophilic substrate carbon.
- The rate of an SN2 reaction is dependent on the concentrations of both the substrate and the nucleophile: rate = k[Nu:][R–L].
Oxidation–Reduction Reactions
- The oxidation state of an atom is the charge it would have if all its bonds were completely ionic.
- CH4 is the lowest oxidation state of carbon (most reduced); CO2 is the highest (most oxidized).
- Carboxylic acids and carboxylic acid derivatives are the most oxidized functional groups, followed by aldehydes, ketones, and imines, followed by alcohols, alkyl halides, and amines.
- Oxidation is an increase in oxidation state and is assisted by oxidizing agents.
- Oxidizing agents accept electrons and are reduced in the process. They have a high affinity for electrons or an unusually high oxidation state. They often contain a metal and a large number of oxygens.
- Primary alcohols can be oxidized to aldehydes by pyridinium chlorochromate (PCC) or to carboxylic acids by stronger oxidizing agents, like chromium trioxide (CrO3) or sodium or potassium dichromate (Na2Cr2O7 or K2Cr2O7).
- Secondary alcohols can be oxidized to ketones by most oxidizing agents.
- Aldehydes can be oxidized to carboxylic acids by most oxidizing agents.
- Reduction is a decrease in oxidation state and is assisted by reducing agents.
- Reducing agents donate electrons and are oxidized in the process. They have low electronegativity and ionization energy. They often contain a metal and a large number of hydrides.
- Aldehydes, ketones, and carboxylic acids can be reduced to alcohols by lithium aluminum hydride (LiAlH4).
- Amides can be reduced to amines by LiAlH4.
- Esters can be reduced to a pair of alcohols by LiAlH4.
Chemoselectivity
- Both nucleophile–electrophile and oxidation–reduction reactions tend to act at the highest-priority (most oxidized) functional group.
- One can make use of steric hindrance properties to selectively target functional groups that might not primarily react, or to protect functional groups.
- Diols are often used as protecting groups for aldehyde or ketone carbonyls.
- Alcohols may be protected by conversion to tert-butyl ethers.
Steps to Problem Solving
- Know your nomenclature
- Identify the functional groups
- Identify the other reagents
- Identify the most reactive functional group(s)
- Identify the first step of the reaction
- Consider stereoselectivity
ANSWERS TO CONCEPT CHECKS
**4.1**
- An acid–base reaction will proceed when the acid and base react to form conjugate products that are weaker than the reactants.
- Amphoteric species can act as either an acid or a base. Water, bicarbonate, and dihydrogen phosphate are common amphoteric species in biological systems.
- pKa = –log Ka, where Ka is the equilibrium constant for the dissociation of an acid. pKa indicates acid strength: a stronger acid has a lower (or even negative) pKa.
- Alcohols, aldehydes and ketones, carboxylic acids, and most carboxylic acid derivatives act as acids. Amines and amides act as bases.
**4.2**
- Nucleophilicity and electrophilicity are based on relative rates of reactions and are therefore kinetic properties. Acidity and basicity are measured by the position of equilibrium in a protonation or deprotonation reaction and are therefore thermodynamic properties.
- t-Butoxide < isopropanolate < ethoxide < methoxide. The four main determinants of nucleophilicity are charge (more negative = better nucleophile), electronegativity (more electronegative = worse nucleophile), steric hindrance (larger = worse nucleophile), and the solvent (protic solvents can protonate or hydrogen bond with the nucleophile, decreasing its reactivity). Each of these nucleophiles has the same attacking atom (oxygen), but differ in their bulkiness. The molecules with the least steric hindrance will be the more effective nucleophiles.
- A substitution reaction will proceed when the nucleophile is a stronger base (more reactive) than the leaving group.
- Greater positive charge increases electrophilicity, and better leaving groups increase electrophilicity by making the reaction more likely to proceed.
- Good leaving groups can stabilize the extra electrons that result from heterolysis. Weak bases (the conjugate bases of strong acids) are good leaving groups. Resonance stabilization and inductive effects from electron-withdrawing groups also improve leaving group ability.
**4.3**
- Good oxidizing agents have a high affinity for electrons or have very high oxidation states. Examples include O2, O3, Cl2, permanganate (MnO4−), chromate (CrO4−), dichromate (CrO42−), and pyridinium chlorochromate. These compounds often contain a metal and a large number of oxygen atoms.
- Good reducing agents have low electronegativities and ionization energies or contain a hydride ion (H–). Examples include sodium, magnesium, aluminum, zinc, sodium hydride (NaH), calcium dihydride (CaH2), lithium aluminum hydride (LiAlH4), and sodium borohydride (NaBH4). These compounds often contain a metal and a large number of hydrides.
- Carbon dioxide, carboxylic acid, ketone, alcohol, methane
**4.4**
- The two reactive centers are the carbonyl carbon, which is electrophilic, and the α-hydrogens, which are acidic.
- SN1 reactions are most likely to occur on tertiary carbons where a carbocation can be most easily stabilized. The first reaction has a tertiary carbon containing a good leaving group while the second reaction has a secondary carbon containing a good leaving group. The first reaction will proceed more quickly.
- SN2 reactions are easily inhibited by steric hindrance. The fluorides are smaller than the methyls and so the difluoride molecule will be more suitable for an SN2.
**4.5**
-
- Know your nomenclature
- Identify the functional groups
- Identify the other reagents
- Identify the most reactive functional group(s)
- Identify the first step of the reaction
- Consider stereoselectivity
- If there are no reagents other than the reactants, then the properties of the functional groups on the reactants themselves (acid–base; nucleophile–electrophile) will determine the outcome.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. C
NH3 and H2O are Lewis bases because nitrogen and oxygen can donate lone pairs. Ag+ is a Lewis acid because it can accept a lone pair into an unoccupied orbital.
2. C
Remember, good nucleophiles tend to have lone pairs or π bonds and are negatively charged or polarized. Alkoxide (OR–) and hydroxide (OH–) anions are strong nucleophiles. Alcohols (ROH) and carboxylic acids (RCOOH) are weak nucleophiles. The alkyl group of an alkoxide anion donates additional electron density, making it more reactive than the hydroxide ion. The carboxylic acid contains more electron-withdrawing oxygen atoms than the alcohol, making it less nucleophilic.
3. D
Good electrophiles are positively charged or polarized. CR3+ is a tertiary carbocation; it has a positive charge, which makes it very electrophilic. CH3Cl and CH3OH are both polarized; however, the leaving groups differ between these two. Cl– is a weaker base than OH– (HCl is a stronger acid than H2O). As such, Cl– will be more stable in solution than OH–, which increases the electrophilic reactivity of CH3Cl above CH3OH. CH3OCH3 has a much less stable leaving group, CH3O–, and is therefore significantly less electrophilic.
4. A
Good leaving groups are weak bases, which are the conjugates of strong acids. Leaving groups must also be stable once they leave the molecule. H2O is, by far, the most stable leaving group and will be extremely unreactive once it leaves the molecule through heterolysis. Br– is the conjugate base of HBr; HO– is the conjugate base of water. HBr is a much stronger acid than water, so Br– is a better leaving group than HO–. Finally, hydride (H–) is a very poor leaving group because it is extremely unstable in solution.
5. B
Carboxylic acids are the second most oxidized form of carbon (only carbon dioxide is more oxidized). In carboxylic acids, the carbon atom has three bonds to oxygen. In aldehydes, the carbon atom has two bonds to oxygen. In amines, the carbon atom has one bond to nitrogen. In an alkane, the carbon only has bonds to other carbons and hydrogens.
6. B
During this reaction, the alkoxide anion accepts a proton, meaning this molecule acts as a Brønsted-Lowry base. Also, in accepting the proton, the alkoxide anion donates a lone pair of electrons to form a coordinate covalent bond with the incoming hydrogen ion. In this way, the alkoxide anion is also acting as a Lewis base. Together, these observations support (B).
7. C
Because 2-butanol is a secondary alcohol, oxidation by a strong oxidizing agent like dichromate will result in a ketone, butanone.
8. C
Pyridinium chlorochromate is a weak oxidizing agent, and will oxidize an alcohol to an aldehyde. Stronger oxidizing agents are required to convert a primary alcohol to a carboxylic acid.
9. B
An SN1 reaction is a first-order nucleophilic substitution reaction. It is called first-order because the rate-limiting step involves only one molecule. (A) is true, but does not explain why SN1 reactions have first-order kinetics; the rate-limiting step of an SN2 reaction is also the first (and only) step of that reaction, but SN2 reactions have second-order kinetics, not first-order. (C) is a true statement as well, but again does not explain why the reaction is first-order. Finally, (D) is incorrect because it is the rate-limiting step, not the reaction overall, that involves only one molecule.
10. D
In a protic solvent, the protons in solution can attach to the nucleophile, decreasing its nucleophilicity. The larger the nucleophile, and the stronger its conjugate acid, the stronger the nucleophile will be. Of the options given, I– will therefore be the strongest nucleophile because it is least likely to associate with the protons in solution.
11. D
Primary and secondary alcohols can be oxidized by PCC because the central carbon atom can form additional bonds with oxygen while losing bonds to hydrogen, justifying (D) as the right answer. Choices (A), a tertiary alcohol; (B), a ketone; and (C), a carboxylic acid, cannot be oxidized by PCC because the central carbon in these molecules is already making four bonds to other carbon or oxygen atoms, and therefore cannot readily form more bonds to oxygen.
12. A
Steric hindrance slows SN2 pathways, so the molecule that is fastest to react will be the one that is least sterically hindered. The reactivity trend based on steric hindrance is methyl > primary > secondary (note that tertiary substrates are too hindered to react via SN2 at all). The molecule in (A) is a primary substrate, that is, the least sterically hindered, and will therefore react rapidly by an SN2 pathway. Additionally, the molecule in (A) has a good leaving group. Together, these trends make (A) the best substrate for rapid SN2. Note that while (C) also depicts a primary substrate, this molecule does not have a good leaving group, so (C) can be eliminated. Choices (B) and (D) can be eliminated because these molecules are secondary substrates.
12. D
To carry out a nucleophile–electrophile reaction, the nucleophile must be able to dissolve in the solvent. Nucleophiles are nearly always polar, and often carry a charge. Polar solvents are therefore preferred for these reactions. Hexane is a nonpolar solvent and will not be useful for a nucleophile–electrophile reaction.
14. A
Aldehydes have one alkyl group connected to the carbonyl carbon, whereas ketones have two. This creates more steric hindrance in ketones, which lowers their reactivity to nucleophiles. Ketones are also less reactive because their carbonyl carbon has less positive charge character; the additional alkyl group can donate electron density—the opposite of (D)—which decreases the electrophilicity of the compound.
15. D
Remember, there is a hierarchy to the reactivity of carboxylic acid derivatives that dictates how reactive they are toward nucleophilic attack. In order from highest to lowest, this is anhydrides > carboxylic acids and esters > amides. In practical terms, this means that derivatives of higher reactivity can form derivatives of lower reactivity but not vice versa. Nucleophilic attack of an ester cannot result in the corresponding anhydride because anhydrides are more reactive than esters.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
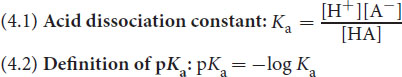
SHARED CONCEPTS
- General Chemistry Chapter 5
- Chemical Kinetics
- General Chemistry Chapter 10
- Acids and Bases
- General Chemistry Chapter 11
- Oxidation–Reduction Reactions
- Organic Chemistry Chapter 1
- Nomenclature
- Organic Chemistry Chapter 5
- Alcohols
- Organic Chemistry Chapter 7
- Aldehydes and Ketones II