Chapter 5: Alcohols

Chapter 5: Alcohols
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Alcohols have higher boiling points than their analogous hydrocarbons because:
- the oxygen atoms in alcohols have shorter bond lengths.
- hydrogen bonding is present in alcohols.
- alcohols are more acidic than their analogous hydrocarbons.
- alcohols can be oxidized to ketones.
-
Tertiary alcohols are oxidized with difficulty because:
- there is no hydrogen attached to the carbon with the hydroxyl group.
- there is no hydrogen attached to the α-carbon of the carbonyl.
- tertiary alcohols contain hydroxyl groups with no polarization.
- they are relatively inert.
-
The IUPAC name of this molecule is:
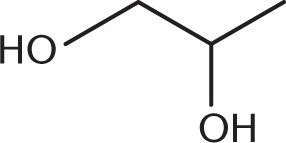
- ethane-1,2-diol.
- propane-1,2-diol.
- dimethanol.
- dipropanol.
-
The IUPAC name of this molecule is:
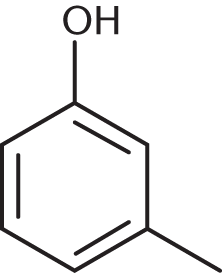
- 2-methylcyclohexanol.
- m-methylphenol.
- p-methylphenol.
- 3-methylcyclohexanol.
-
Which of the following correctly lists methanol, isobutyl alcohol, and propanol by decreasing boiling point?
- Methanol > isobutyl alcohol > propanol
- Isobutyl alcohol > methanol > propanol
- Isobutyl alcohol > propanol > methanol
- Methanol > propanol > isobutyl alcohol
-
Which of the following correctly lists hexanol, phenol, and cyclohexanol by increasing acidity of the hydroxyl hydrogen?
- Phenol < hexanol < cyclohexanol
- Cyclohexanol < hexanol < phenol
- Cyclohexanol < phenol < hexanol
- Phenol < cyclohexanol < hexanol
-
Which of the following will convert CH3CH2CH2OH to CH3CH2CHO?
- CrO3
- PCC
- K2Cr2O7
- I only
- II only
- I and III only
- I, II, and III
-
Which of the following will convert cyclohexanol to cyclohexanone?
- Chromium trioxide
- Pyridinium chlorochromate
- Sodium dichromate
- I only
- II only
- I and III only
- I, II, and III
-
Successfully converting 3-phenylpropanol to 3-phenylpropanoic acid by the Jones oxidation requires the oxidizing agent, the solvent, and:
- dilute sulfuric acid.
- dilute sodium hydroxide.
- anhydrous conditions.
- high temperature.
-
Treating 2-methyl-1-propanol with methylsulfonyl chloride in base, followed by reaction with pyridinium chlorochromate, and a final step in strong acid, will give an end product of:
- 2-methyl-1-propanol.
- 2-methylpropanal.
- 2-methylpropanoic acid.
- 2-methyl-1-propane.
-
Reaction of 1-phenylethanone with ethylene glycol, also known as ethane-1,2-diol, in aqueous H2SO4 will result in the formation of:
- a ketal.
- a carboxylic acid.
- an aldehyde.
- a hemiacetal.
-
Treatment of this molecule with CrO3 under appropriate reaction conditions will yield which of the following molecules?
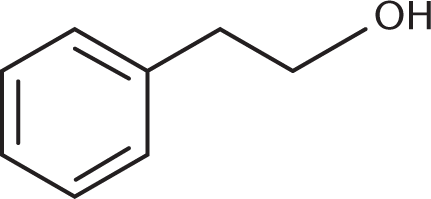
- 2-Phenylethanol
- Phenylacetaldehyde
- Phenylacetone
- 2-Phenylethanoic acid
-
In order to convert phenols into hydroxyquinones, how many steps of oxidation or reduction are required?
- 1 oxidation step
- 2 oxidation steps
- 1 reduction step
- 2 reduction steps
-
The conversion of ubiquinone to ubiquinol requires what type of reaction?
- Condensation
- Oxidation
- Reduction
- Hydrolysis
-
Which of the following will convert a cyclic acetal to a carbonyl and a dialcohol?
- Aqueous acid
- LiAlH4
- CrO3
- Acetone
Answer Key
- B
- A
- B
- B
- C
- B
- B
- D
- A
- A
- A
- D
- B
- C
- A
Chapter 5: Alcohols
CHAPTER 5
ALCOHOLS
In This Chapter
5.1 Description and Properties
Nomenclature
Physical Properties
5.2 Reactions of Alcohols
Oxidation Reactions
Mesylates and Tosylates
Protecting Groups
5.3 Reactions of Phenols
Quinones and Hydroxyquinones
Ubiquinone
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 13% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content category:
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
Alcohols are probably the most popular chemicals you’ll encounter in organic chemistry. Ethanol has been popular with humans for more than 10,000 years. It’s not just humans, either: many animals are known to seek out rotten fruits that have fermented enough to contain moderate levels of ethanol. Note that when we talk about consuming “alcohol,” we are referring exclusively to ethanol (grain alcohol). In fact, consuming other alcohols can have drastically negative effects. Methanol (wood alcohol), for example, is oxidized by the body to formic acid, which is extremely toxic to the optic nerve and retina and can cause blindness when ingested. Isopropyl alcohol, commonly used as an antiseptic, can cause severe central nervous system depression. For organic chemistry purposes, of course, we do not restrict ourselves to only one type of alcohol—many are used synthetically and in analytic techniques.
5.1 Description and Properties
LEARNING OBJECTIVES
After Chapter 5.1, you will be able to:
- Predict the relative pKa values of two given alcohols
- Order alcohols based on boiling point
Alcohols have the general formula ROH, with the functional group –OH referred to as a hydroxyl group.
MCAT EXPERTISE
Alcohols are an important group of compounds. They will be seen on the MCAT as protic solvents, reactants, products, and prime examples of hydrogen bonding.
Nomenclature
Alcohols are named in the IUPAC system by replacing the **–eending of the root alkane with the ending–*ol***. If the alcohol is the highest-priority functional group, the carbon atom attached to it receives the lowest possible number. Some examples are shown in Figure 5.1.
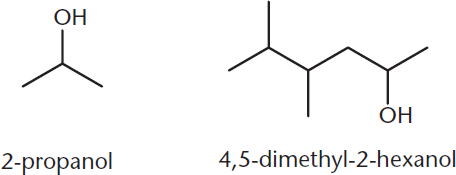
Figure 5.1. IUPAC Names of Alcohols
Alternatively, the common naming practice is to name the alkyl group as a derivative, followed by alcohol, as shown in Figure 5.2.
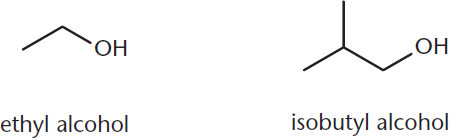
Figure 5.2. Common Names of Alcohols
When the alcohol is not the highest-priority group, it is named as a substituent, with the prefix hydroxy–.
Finally, we will also see that hydroxyl groups can be attached to aromatic rings, as shown in Figure 5.3. These compounds are called phenols. The hydroxyl hydrogens of phenols are particularly acidic due to resonance within the phenol ring. When benzene rings contain two substituents, their relative positions must be indicated. Two groups on adjacent carbons are called ortho–, or simply o–. Two groups separated by a carbon are called meta–, or m–. Two groups on opposite sides of the ring are called para–, or p–.
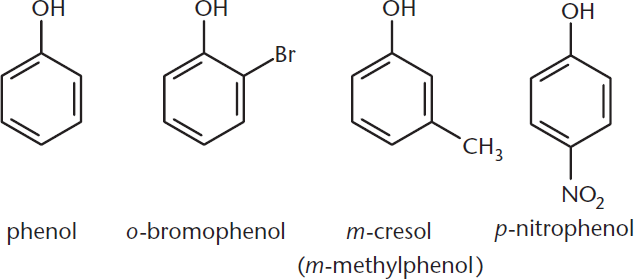
Figure 5.3. Phenols: Aromatic Alcohols
KEY CONCEPT
Aromatic alcohols are called phenols. The possible resonance between the ring and the lone pairs of the oxygen atom in the hydroxyl group make the hydrogen of the alcohol more acidic than other alcohols.
Physical Properties
One of the prominent properties of alcohols is that they are capable of intermolecular hydrogen bonding, which results in significantly higher melting and boiling points than those of analogous hydrocarbons, as shown in Figure 5.4.
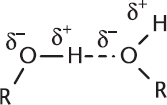
Figure 5.4. Intermolecular Hydrogen Bonding in Alcohols
MCAT EXPERTISE
Hydrogen bonding causes increased melting points, boiling points, and solubility in water.
Molecules with more than one hydroxyl group show greater degrees of hydrogen bonding. This is evident from the boiling points shown in Figure 5.5.
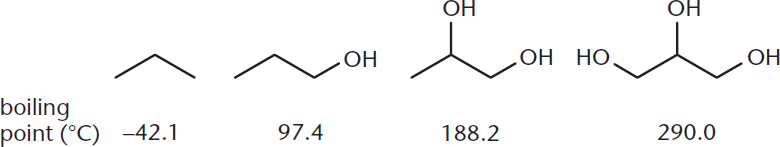
Figure 5.5. Boiling Points for Various Alcohols Boiling point increases significantly with additional hydroxyl groups, which permit more hydrogen bonding.
Hydrogen bonding occurs when hydrogen atoms are attached to highly electronegative atoms like nitrogen, oxygen, or fluorine. Hydrogen bonding is the result of the extreme polarity of these bonds. In the case of a hydroxyl group, the electronegative oxygen atom pulls electron density away from the less electronegative hydrogen atom. This generates a slightly positive charge on the hydrogen and slightly negative charge on the oxygen. Then, the partially positive hydrogen of one molecule electrostatically attracts the partially negative oxygen of another molecule, generating a noncovalent bonding force known as a hydrogen bond.
The hydroxyl hydrogen is weakly acidic, and alcohols can dissociate into protons and alkoxide ions in the same way that water dissociates into protons and hydroxide ions. Table 5.1 gives pKa values of several hydroxyl-containing compounds.
Table 5.1 pKa Values of Hydroxyl-Containing Compounds
DISSOCIATION pKa
H2O
HO− + H+ 14.0
CH3OH
CH3O− + H+ 15.5
C2H5OH
C2H5O− + H+ 15.9
i-PrOH
i-PrO− + H+ 16.5
t-BuOH
t-BuO− + H+ 17.0
CF3CH2OH
CF3CH2O− + H+ 12.5
PhOH
PhO− + H+ ∼10.0
BRIDGE
Remember from Chapter 10 of MCAT General Chemistry Review that pKa = –logKa. Strong acids have high Ka values and low pKa values. Thus, phenol, which has the smallest pKa, is the most acidic of the alcohols listed in Table 5.1.
Looking at Table 5.1, we can see that the hydroxyl hydrogens of phenols are more acidic than those of other alcohols. This is due to the aromatic nature of the ring, which allows for the resonance stabilization of the negative charge on oxygen, stabilizing the anion. Like other alcohols, phenols form intermolecular hydrogen bonds and have relatively high melting and boiling points. Phenols are slightly soluble in water, owing to hydrogen bonding, as are some phenol derivatives. Because phenols are much more acidic than nonaromatic alcohols, they can form salts with inorganic bases such as NaOH.
The presence of other substituents on the ring has significant effects on the acidity, boiling points, and melting points of phenols. As with other compounds, electron-withdrawing substituents increase acidity, and electron-donating groups decrease acidity.
KEY CONCEPT
Charges like to be spread out as much as possible. Acidity decreases as more alkyl groups are attached because they are electron-donating, which destabilizes the alkoxide anion. Resonance or electron-withdrawing groups stabilize the alkoxide anion, making the alcohol more acidic.
Another trend seen in Table 5.1 is that the presence of more alkyl groups in nonaromatic alcohols produces less acidic molecules. Because alkyl groups donate electron density, they destabilize a negative charge. Additionally, alkyl groups help stabilize positive charges, explaining why more substituted carbocations have higher stability than less substituted carbocations.
MCAT CONCEPT CHECK 5.1:
Before you move on, assess your understanding of the material with these questions.
-
Which has a lower pKa: ethanol or p-ethylphenol? Why?
_________________________________
_________________________________
_________________________________
-
Rank the following by decreasing boiling point: 1-pentanol, 1-hexanol, 1,6-hexanediol
- ____________________________
- ____________________________
- ____________________________
5.2 Reactions of Alcohols
LEARNING OBJECTIVES
After Chapter 5.2, you will be able to:
- Predict the reaction of primary and secondary alcohols with strong oxidizing agents
- Recall the reagent(s) used to oxidize primary alcohols to aldehydes
- Explain the purpose of a mesylate or tosylate group
- Describe the process for protecting an aldehyde or ketone
The main reactions that we will see on the MCAT for alcohols include oxidation, preparation of mesylates and tosylates, and protection of carbonyls by alcohols.
Oxidation Reactions
Oxidation of alcohols can produce several products. Primary alcohols can be oxidized to aldehydes, but only by pyridinium chlorochromate (PCC), a mild anhydrous oxidant, as shown in Figure 5.6. This reactant stops after the primary alcohol has been converted to an aldehyde because PCC lacks the water necessary to hydrate the otherwise easily hydrated aldehyde. With other oxidizing agents, aldehydes are rapidly hydrated to form geminal diols (1,1-diols), which can be easily oxidized to carboxylic acids.
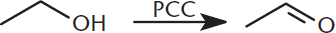
Figure 5.6. Oxidation of a Primary Alcohol to an Aldehyde by Pyridinium Chlorochromate (PCC)
Secondary alcohols can be oxidized to ketones by PCC or any stronger oxidizing agent. Tertiary alcohols cannot be oxidized because they are already as oxidized as they can be without breaking a carbon–carbon bond.
The oxidation of primary alcohols with a strong oxidizing agent like chromium(VI) will produce a carboxylic acid. In the process, chromium(VI) is reduced to chromium(III). Common examples of chromium-containing oxidizing agents include sodium and potassium dichromate salts (Na2Cr2O7 and K2Cr2O7). As with other strong oxidizing agents, these will fully oxidize primary alcohols to carboxylic acids, and secondary alcohols to ketones. An example is shown in Figure 5.7.
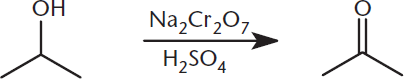
Figure 5.7. Oxidation of a Secondary Alcohol to a Ketone by a Dichromate Salt
KEY CONCEPT
Alcohols are readily oxidized to carboxylic acids by any oxidizing agent other than PCC (which will only oxidize primary alcohols to aldehydes).
Finally, an even stronger chromium-containing oxidizing agent is chromium trioxide, CrO3. When dissolved with dilute sulfuric acid in acetone, this is called the Jones oxidation, as shown in Figure 5.8. As expected, this reaction oxidizes primary alcohols to carboxylic acids and secondary alcohols to ketones.
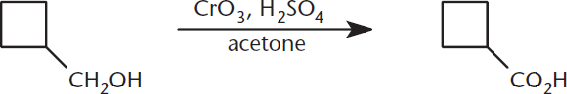
Figure 5.8. Jones Oxidation A primary alcohol is oxidized to a carboxylic acid by CrO3.
Mesylates and Tosylates
The hydroxyl groups of alcohols are fairly poor leaving groups for nucleophilic substitution reactions. However, they can be protonated, or reacted to form much better leaving groups called mesylates and tosylates. A mesylate is a compound containing the functional group –SO3CH3, derived from methanesulfonic acid; its anionic form is shown in Figure 5.9.
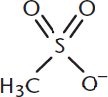
Figure 5.9. Structure of the Mesylate Anion
Mesylates are prepared using methylsulfonyl chloride and an alcohol in the presence of a base.
Tosylates contain the functional group –SO3C6H4CH3, derived from toluenesulfonic acid. These compounds are produced by the reaction of alcohols with p-toluenesulfonyl chloride, forming esters of toluenesulfonic acid. A tosylate is shown in Figure 5.10.
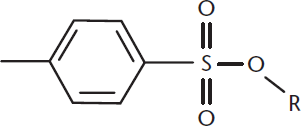
Figure 5.10. Structure of a Tosylate
In addition to making hydroxyl groups of alcohols into better leaving groups for nucleophilic substitution reactions, mesyl and tosyl groups can also serve as protecting groups when we do not want alcohols to react. These groups are protective in that they will not react with many of the other reagents that would attack alcohols, especially oxidizing agents. Thus, reacting an alcohol to form a mesylate or tosylate is sometimes performed before multistep reactions in which the desired products do not derive from the alcohol.
Protecting Groups
Alcohols can also be used as protecting groups for other functional groups themselves. For example, aldehydes and ketones can be reacted with two equivalents of an alcohol or a diol (dialcohol), forming acetals (primary carbons with two –OR groups and a hydrogen atom) or ketals (secondary carbons with two –OR groups). Carbonyls are very reactive with strong reducing agents like lithium aluminum hydride (LiAlH4). Acetals and ketals, on the other hand, do not react with LiAlH4. The acetal or ketal functionality thereby protects the aldehyde or ketone from reaction. After reducing other functionalities in the molecule, the acetal or ketal can be reverted back to a carbonyl with aqueous acid, a step called deprotection. These reactions are shown in Figure 5.11.
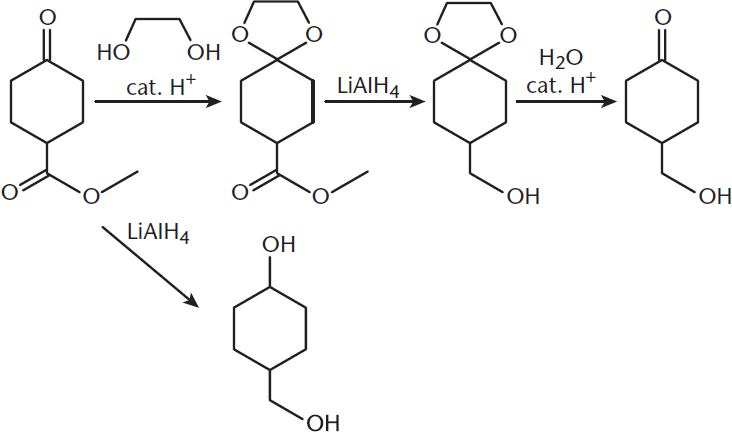
Figure 5.11. Protection of a Ketone by Ketal Formation Using a Dialcohol
MCAT CONCEPT CHECK 5.2:
Before you move on, assess your understanding of the material with these questions.
-
What will happen to primary and secondary alcohols, respectively, in the presence of strong oxidizing agents?
- Primary alcohols:
________________________
- Secondary alcohols:
________________________
-
What is the product when 1-butanol is treated with PCC? With chromium trioxide?
- PCC: ______________________________
- Chromium trioxide: ______________________________
-
What is the purpose of a mesylate or tosylate?
________________________
-
How can aldehydes or ketones be protected using alcohols?
________________________
5.3 Reactions of Phenols
LEARNING OBJECTIVES
After Chapter 5.3, you will be able to:
- Recall the process for production of quinones and hydroxyquinones
- Identify the properties of ubiquinone that allow it to function as an electron carrier:
Reactions of phenols proceed in similar fashion to reactions of alcohols. However, as discussed previously, the hydrogen in the hydroxyl group of phenols is particularly acidic because the oxygen-containing anion is resonance-stabilized by the ring.
Quinones and Hydroxyquinones
Treatment of phenols with oxidizing agents produces compounds called quinones (2,5-cyclohexadiene-1,4-diones), as shown in Figure 5.12.
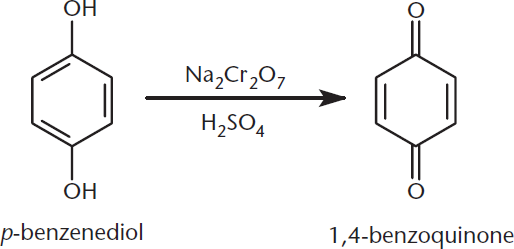
Figure 5.12. Oxidation of p-Benzenediol (a Hydroquinone) to a Quinone
Quinones are named by indicating the position of the carbonyls numerically and adding quinone to the name of the parent phenol. Due to the conjugated ring system, these molecules are resonance-stabilized electrophiles. Remember, however, that these are not necessarily aromatic because they lack the classic aromatic conjugated ring structure. Some quinones do have an aromatic ring, but this is not always the case. Quinones serve as electron acceptors biochemically, specifically in the electron transport chain in both photosynthesis and aerobic respiration. Vitamin K1 is the common name of the quinone 2-methyl-3-[(2E)-3,7,11,15-tetramethylhexadec-2-en-1-yl]naphthoquinone, shown in Figure 5.13. This molecule is also called phylloquinone and is important for photosynthesis and the carboxylation of some of the clotting factors in blood. Vitamin K2, similarly, corresponds to a class of molecules called menaquinones.
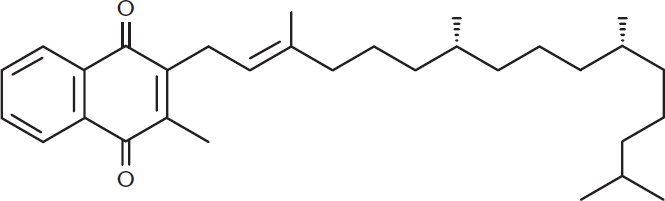
Figure 5.13. Phylloquinone (Vitamin K1)
BRIDGE
Phylloquinone and menaquinone are the common names of Vitamin K1 and Vitamin K2, respectively. These molecules are fat-soluble vitamins that play a role in carboxylation of clotting factors II, VII, IX, and X, and proteins C and S in blood. The functions of fat-soluble vitamins are explored in Chapter 5 of MCAT Biochemistry Review.
These molecules can be further oxidized to form a class of molecules called hydroxyquinones. Hydroxyquinones share the same ring and carbonyl backbone as quinones, but differ by the addition of one or more hydroxyl groups. Many hydroxyquinones have biological activity, and some are used in the synthesis of medications. One classic example is shown in Figure 5.14.
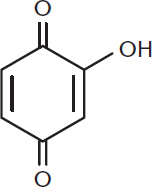
Figure 5.14. 2-Hydroxy-1,4-benzoquinone, a Hydroxyquinone
MCAT EXPERTISE
Note the subtle difference in terminology between Figure 5.12 and Figure 5.14. Ahydroquinone is a benzene ring with two hydroxyl groups. Ahydroxyquinone contains two carbonyls and a variable number of hydroxyl groups.
Because of resonance, hydroxyquinones behave like quinones with electron-donating groups, making these slightly less electrophilic (although still quite reactive). When naming these compounds, the position of the hydroxyl groups is indicated by a number, and the total number of hydroxyl groups (if there is more than one) is indicated by a prefix (such as di–, or tri–) with the substituent name hydroxy–. Several examples are shown in Figure 5.15.
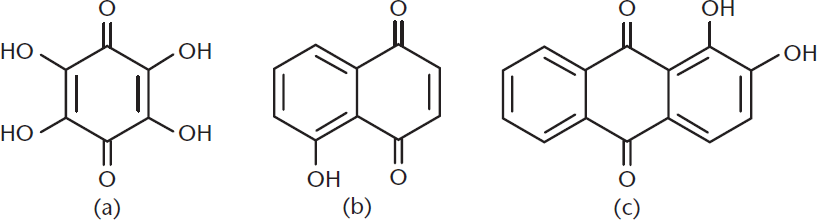
Figure 5.15. Three Examples of Hydroxyquinones (a) Tetrahydroxybenzoquinone; (b) 5-hydroxynaphthoquinone; (c) 1,2-dihydroxyanthraquinone.
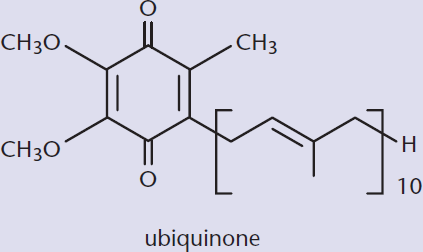
Ubiquinone
Ubiquinone is one example of a biologically active quinone. Ubiquinone is also called coenzyme Q and is a vital electron carrier associated with Complexes I, II, and III of the electron transport chain. Ubiquinone is the most oxidized form that this molecule takes physiologically: it can also be reduced to ubiquinol upon the acceptance of electrons, as shown in Figure 5.16. This oxidation–reduction capacity allows the molecule to perform its physiological function of electron transport.
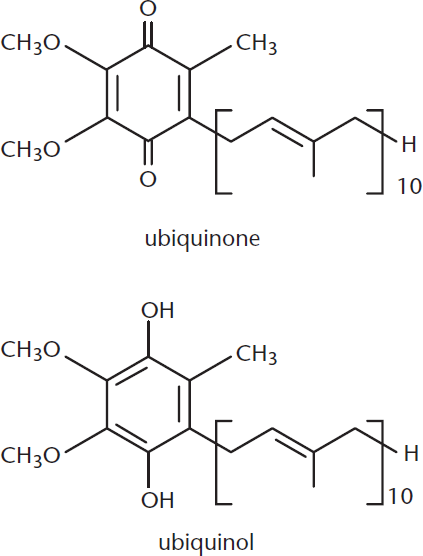
Figure 5.16. Ubiquinone (Coenzyme Q) and Ubiquinol Ubiquinone is the oxidized form; when it picks up electrons, it is converted to its reduced form (ubiquinol).
BRIDGE
Coenzyme Q plays a role in Complexes I, II, and III of the electron transport chain. In Complex III, it is the main player in the Q cycle, which contributes to the formation of the proton-motive force across the inner mitochondrial membrane. The respiratory complexes are discussed in Chapter 10 of MCAT Biochemistry Review.
The long alkyl chain of this molecule allows it to be lipid soluble, which allows it to act as an electron carrier within the phospholipid bilayer.
Other biological molecules that undergo oxidation–reduction reactions as part of their normal function include NADH, FADH2, and NADPH. These molecules accept and donate electrons readily, similar to ubiquinone, and are discussed more thoroughly in Chapters 9 and 10 of MCAT Biochemistry Review.
MCAT CONCEPT CHECK 5.3:
Before you move on, assess your understanding of the material with these questions.
-
How are quinones generally produced?
________________________
-
How are hydroxyquinones produced?
________________________
-
What chemical properties of ubiquinone allow it to carry out its biological functions?
________________________
Conclusion
Alcohols are a particular favorite of the MCAT test makers. We got our first look at the unique properties that stem from hydrogen bonding, an important ability of alcohols. Alcohols can be oxidized to aldehydes, ketones, or carboxylic acids depending on the substitution of the alcohol and the strength of the oxidizing agent. From this point forward, oxidation and reduction will be important reactions with all functional groups. Alcohols can also participate in nucleophilic substitution reactions, which may be facilitated by converting the alcohol into a mesylate or tosylate. Finally, phenols (and their oxidized counterparts, quinones and hydroxyquinones) are involved in a number of biochemical pathways. In particular, their utility is seen in processes that require rapid oxidation and reduction, such as photosynthesis and the electron transport chain.
Over the next four chapters, we’ll explore other oxygen-containing compounds. Recognize that these chapters are put in a specific order: as you move further along in MCAT Organic Chemistry Review, the functional groups will become more oxidized and more reactive. First, we’ll look at aldehydes and ketones (and their deprotonated forms, enols and enolates). Then, we’ll explore carboxylic acids and their derivatives: amides, esters, and anhydrides.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Description and Properties
- Alcohols have the general form ROH and are named with the suffix – ol. If they are not the highest priority, they are given the prefix hydroxy–.
- Phenols are benzene rings with hydroxyl groups. They are named for the relative positions of the hydroxyl groups: ortho– (adjacent carbons), meta– (separated by one carbon), or para– (on opposite sides of the ring).
- Alcohols can hydrogen bond, raising their boiling and melting points relative to corresponding alkanes. Hydrogen bonding also increases the solubility of alcohols.
- Phenols are more acidic than other alcohols because the aromatic ring can delocalize the charge of the conjugate base.
- Electron-donating groups like alkyl groups decrease acidity because they destabilize negative charges. Electron-withdrawing groups, such as electronegative atoms and aromatic rings, increase acidity because they stabilize negative charges.
Reactions of Alcohols
- Primary alcohols can be oxidized to aldehydes only by pyridinium chlorochromate (PCC); they will be oxidized all the way to carboxylic acids by any stronger oxidizing agents.
- Secondary alcohols can be oxidized to ketones by any common oxidizing agent.
- Alcohols can be converted to mesylates or tosylates to make them better leaving groups for nucleophilic substitution reactions.
- Mesylates contain the functional group –SO3CH3, which is derived from methanesulfonic acid.
- Tosylates contain the functional group –SO3C6H4CH3, which is derived from toluenesulfonic acid.
- Aldehydes or ketones can be protected by converting them into acetals or ketals.
- Two equivalents of alcohol or a dialcohol are reacted with the carbonyl to form an acetal (a primary carbon with two –OR groups and a hydrogen atom) or ketal (a secondary carbon with two –OR groups).
- Other functional groups in the compound can be reacted (especially by reduction) without effects on the newly formed acetal or ketal.
- The acetal or ketal can then be converted back to a carbonyl by catalytic acid, which is called deprotection.
Reactions of Phenols
- Quinones are synthesized through oxidation of phenols.
- Quinones are resonance-stabilized electrophiles.
- Vitamin K1 (phylloquinone) and Vitamin K2 (themenaquinones) are examples of biochemically relevant quinones.
- Hydroxyquinones are produced by oxidation of quinones, adding a variable number of hydroxyl groups.
- Ubiquinone (coenzyme Q) is another biologically active quinone that acts as an electron acceptor in Complexes I, II, and III of the electron transport chain. It is reduced to ubiquinol.
ANSWERS TO CONCEPT CHECKS
**5.1**
- Phenols like p-ethylphenol have increased acidity due to resonance and the electron-withdrawing character of the phenol aromatic ring. Because p-ethylphenol is a stronger acid than ethanol, it will have a lower pKa.
- 1,6-Hexanediol will have the highest boiling point; a molecule with two hydroxyl moieties can have more hydrogen bonding. The 1-hexanol boiling point will be next, with 1-pentanol having the lowest boiling point. 1-Hexanol has a higher boiling point than 1-pentanol because the longer hydrocarbon chain has increased van der Waals forces.
**5.2**
- In the presence of strong oxidizing agents, primary alcohols are completely oxidized to carboxylic acids. Secondary alcohols can only be oxidized to ketones.
- Reacting 1-butanol with PCC results in the aldehyde, 1-butanal. Chromium trioxide is a stronger oxidizing agent that will produce the carboxylic acid, butanoic acid.
- Mesylates and tosylates are used to convert an alcohol into a better leaving group. This is particularly useful for nucleophilic substitution reactions because it increases the stability of the product. They can also be used as protecting groups because many reagents (especially oxidizing agents) that would react with an alcohol cannot react with these compounds.
- Aldehydes or ketones can be reacted with two equivalents of alcohol or a diol to form an acetal or ketal. Acetals and ketals are less reactive than aldehydes and ketones (especially to reducing agents), and can thus protect the functional group from reacting. The acetal or ketal can then be reverted back to the carbonyl by catalytic acid.
**5.3**
- Quinones are produced by oxidation of phenols.
- Hydroxyquinones are produced by the oxidation of quinones, adding a variable number of additional hydroxyl groups.
- Ubiquinone has conjugated rings that stabilize the molecule when accepting electrons. Additionally, the long alkyl chain in the molecule allows for lipid solubility, which allows the molecule to function in the phospholipid bilayer.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. B
Alcohols have higher boiling points than their analogous hydrocarbons as a result of their polarized O–H bonds, in which oxygen is partially negative and hydrogen is partially positive. This enables the oxygen atoms of other alcohol molecules to be attracted to the hydrogen, forming a hydrogen bond. Heat is required to overcome these hydrogen bonds, thereby increasing the boiling point. The analogous hydrocarbons do not form hydrogen bonds and, therefore, vaporize at lower temperatures. (A) is irrelevant; oxygen’s bond length is not a factor in determining a substance’s boiling point. (C) and(D) are true statements, but are also irrelevant to boiling point determination.
2. A
Tertiary alcohols can be oxidized only under extreme conditions because their substrate carbons do not have spare hydrogens to give up. Alcohol oxidation involves the removal of such a hydrogen so that carbon can instead make another bond to oxygen. If no hydrogen is present, a carbon–carbon bond must be cleaved, which requires a great deal of energy and will, therefore, occur only under extreme conditions. (B) is incorrect because alcohols are not carbonyl-containing compounds and would more properly describe a carbonyl-containing compound that is unable to form an enolate. (C) is incorrect because the hydroxyl group of the tertiary carbon is still polarized. (D) is a false statement; tertiary alcohols are still involved in other reactions, such as SN1 reactions.
3. B
Remember, diols are named after the parent alkane, with the position of the alcohols indicated, and ending in the suffix –diol. Here the carbon chain is three carbons, with a hydroxyl group on carbons 1 and 2. Thus, the name is propane-1,2-diol.
4. B
This molecule is a phenol, not a hexanol, because the cyclic group has aromatic double bonds rather than single bonds. The methyl group is separated from the hydroxyl carbon by one carbon in between, making this molecule m-methylphenol.
5. C
All else being equal, boiling points increase with increasing size of the alkyl chain because of increased van der Waals attractions. Isobutyl alcohol has the largest alkyl chain and will thus have the highest boiling point; methanol has the smallest chain and will thus have the lowest boiling point.
6. B
Phenols have significantly more acidic hydroxyl hydrogens than other alcohols because of resonance stabilization of the conjugate base, so this will be the most acidic hydroxyl hydrogen. The acidity of hexanol and cyclohexanol are close, but the hydroxyl hydrogen of hexanol is slightly more acidic because the ring structure of cyclohexanol is slightly electron-donating, which makes its hydroxyl hydrogen slightly less acidic.
7. B
CH3CH2CH2OH is 1-propanol, a primary alcohol. The desired end product, CH3CH2CHO, is propanal, an aldehyde. Of the available options, the only reactant capable of oxidizing primary alcohols to aldehydes is pyridinium chlorochromate (PCC). Chromic trioxide and dichromate salts will both oxidize primary alcohols to carboxylic acids.
8. D
Cyclohexanol is a secondary alcohol, so any of the oxidizing agents listed will convert it to a ketone.
9. A
Acidic conditions, provided by dilute sulfuric acid, are required to complete the Jones oxidation. This reaction is carried out in aqueous conditions, eliminating (C). While heat may speed up the reaction, high temperatures are not required for this reaction, eliminating (D).
10. A
Methylsulfonyl chloride serves as a protecting group for alcohols, which are converted into mesylates. Reacting with this reagent before continuing with what would normally be an oxidation reaction keeps the alcohol from reacting; when the protecting group is then removed using strong acid, the resultant product is the same as the initial reactant. Neither of the oxidation products in (B) or (C), nor the reduction product in (D), will be formed.
11. A
This reaction will create a ketal. This is the first step of the protection of aldehydes or ketones using dialcohols.
12. D
This is the Jones oxidation, which will convert a primary alcohol into a carboxylic acid. (A) is the original reacting molecule, and because the reaction will proceed, this is not correct. Because the reaction uses strong oxidizing agents, it won’t stop at the aldehyde, (B). This reaction also cannot make the ketone in (C) because it starts with a primary alcohol.
13. B
In order to convert phenols into hydroxyquinones, they must first be converted to quinones through an oxidation step; a second oxidation step is required to further oxidize quinones to hydroxyquinones.
14. C
The reaction that converts ubiquinone into ubiquinol is a reduction reaction in which two ketones are reduced to two hydroxyl groups.
15. A
An acetal can be converted to a carbonyl and a dialcohol by treatment with aqueous acid. This is the final step when using alcohols as protecting groups, called deprotection.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 5
- Lipid Structure and Function
- General Chemistry Chapter 8
- The Gas Phase
- Organic Chemistry Chapter 1
- Nomenclature
- Organic Chemistry Chapter 4
- Analyzing Organic Reactions
- Organic Chemistry Chapter 6
- Aldehydes and Ketones I
- Organic Chemistry Chapter 8
- Carboxylic Acids