Chapter 6: Aldehydes and Ketones I: Electrophilicity and Oxidation–Reduction

Chapter 6: Aldehydes and Ketones I: Electrophilicity and Oxidation–Reduction
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
All of the following are true with respect to carbonyls EXCEPT:
- the carbonyl carbon is electrophilic.
- the carbonyl oxygen is electron-withdrawing.
- a resonance structure of the functional group places a positive charge on the carbonyl carbon.
- the π electrons are mobile and are pulled toward the carbonyl carbon.
-
Order the following compounds by increasing boiling point: butane, butanol, butanone
- Butanol < butane < butanone
- Butane < butanone < butanol
- Butanone < butane < butanol
- Butane < butanol < butanone
-
What is the product of the reaction below?
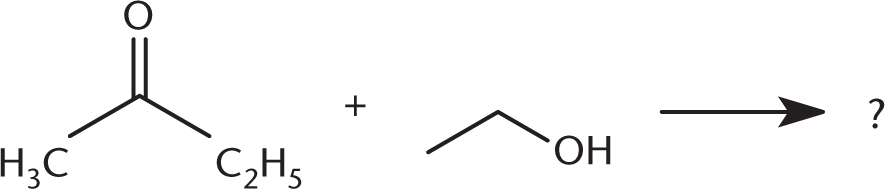
-
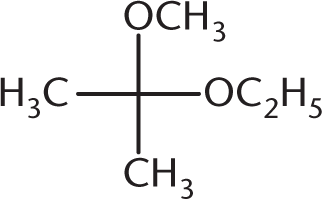
-
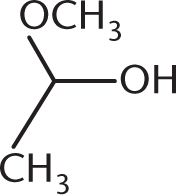
-
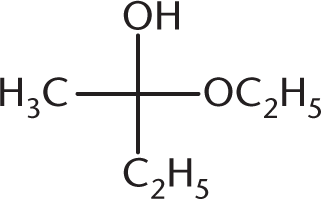
-
-
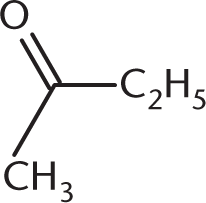
What is the product of the reaction below?
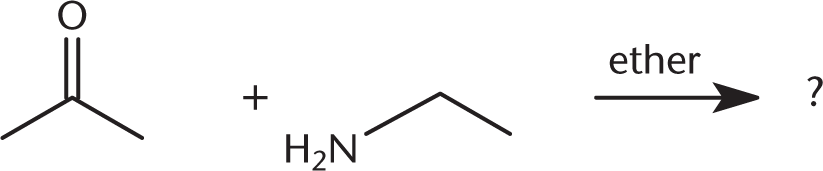
-
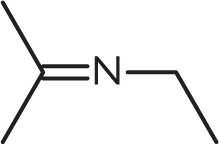
-
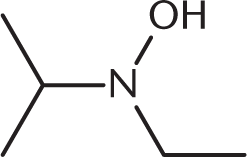
-
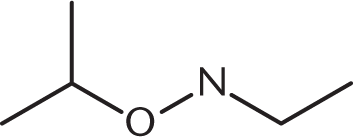
-
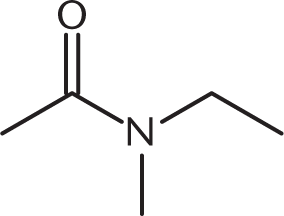
-
What is the product of the reaction below?
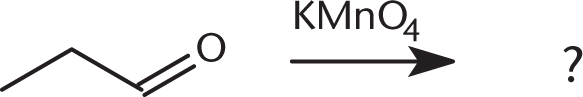
- C3H7OH
- C2H5COOH
- C2H5CHO
- CH3COOH
-
What is the product of the reaction below?
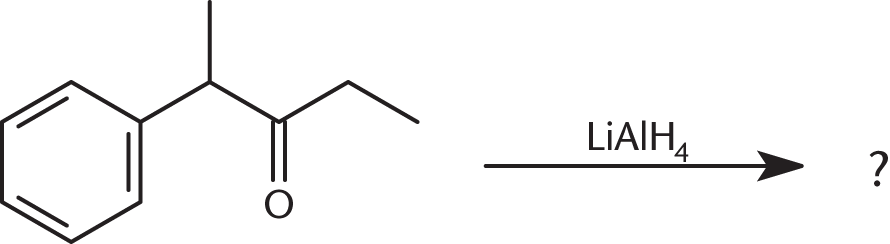
-
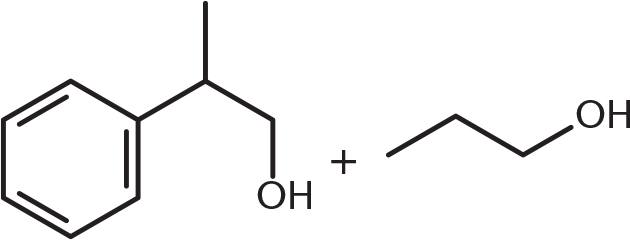
-
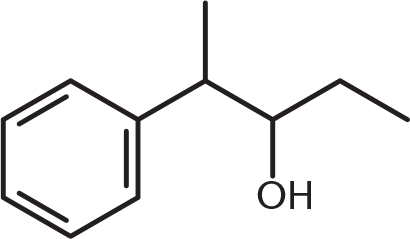
-
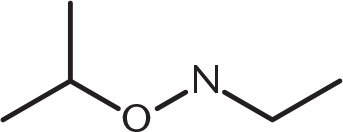
-
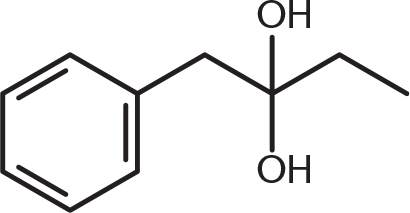
-
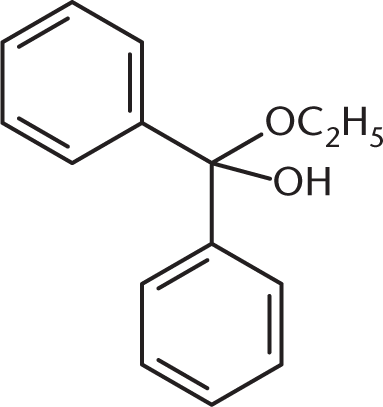
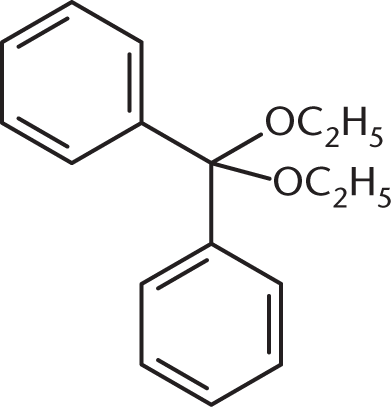
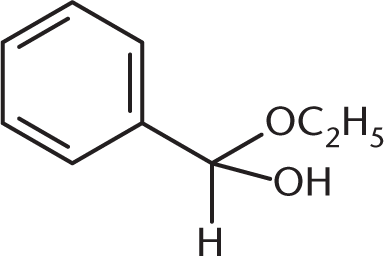
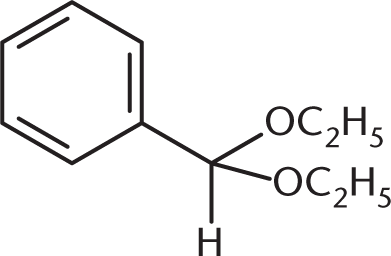
What is the product of the reaction between benzaldehyde and an excess of ethanol (CH3CH2OH) in the presence of anhydrous HCl?
-
-
-
-
-
Hemiacetals and hemiketals usually keep reacting to form acetals and ketals. Why is it difficult to isolate hemiacetals and hemiketals?
- These molecules are unstable.
- The hydroxyl group is rapidly protonated and lost as water under acidic conditions, leaving behind a reactive carbocation.
- The molecules are extremely basic and react rapidly with one another.
- I only
- I and II only
- II and III only
- I, II, and III
-
In a hemiacetal, the central carbon is bonded to:
- –OH, –OR, –H and –R.
- –H, –OR, –OR, and –R.
- –OH, –OR, –R, and –R.
- –OR, –OR, –R, and –R.
-
In a reaction between hydrogen cyanide, butyraldehyde, and ethylmethylketone, which compounds will come together to form the major product?
- Butyraldehyde and hydrogen cyanide
- Ethylmethylketone and butyraldehyde
- Hydrogen cyanide and ethylmethylketone
- No reaction will occur.
-
Which of the following describe(s) pyridinium chlorochromate (PCC)?
- An oxidant that can form aldehydes from primary alcohols
- An oxidant that can completely oxidize primary alcohols
- An oxidant that can completely oxidize secondary alcohols
- I only
- I and II only
- I and III only
- I, II, and III
-
To form a geminal diol, which of the following could attack a carbonyl carbon?
- Hydrogen peroxide
- Water
- Potassium dichromate
- Ethanol
-
In a reaction between ammonia and glutaraldehyde, what is the major product?
- An imine
- A cyanohydrin
- A semicarbazone
- A hydrazone
-
Which of the following can be used to reduce a ketone to a secondary alcohol?
- CrO3
- KMnO4
- LiAlH4
- Ag2O
-
Imines naturally tautomerize to form:
- oximes.
- hydrazones.
- semicarbazones.
- enamines.
Answer Key
- D
- B
- C
- A
- B
- B
- D
- B
- A
- A
- C
- B
- A
- C
- D
Chapter 6: Aldehydes and Ketones I: Electrophilicity and Oxidation–Reduction
CHAPTER 6
ALDEHYDES AND KETONES I: ELECTROPHILICITY AND OXIDATION–REDUCTION
In This Chapter
6.1 Description and Properties
Nomenclature
Physical Properties
Formation
6.2 Nucleophilic Addition Reactions
Hydration
Acetals and Hemiacetals
Imines and Enamines
Cyanohydrins
6.3 Oxidation–Reduction Reactions
Oxidation of Aldehydes
Reduction by Hydride Reagents
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 8% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content category:
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
This chapter focuses on an important functional group for the MCAT: the carbonyl. Aldehydes and ketones, the two functional groups highlighted here, have a lot in common because they both contain a carbonyl—a double bond between a carbon and an oxygen. The only difference between the two is what is attached to the carbonyl carbon.
The carbonyl group is one of the most common functional groups in organic chemistry for two reasons. First, the carbonyl is a component of many different functional groups. In addition to aldehydes and ketones, the carbonyl is found in carboxylic acids, esters, amides, anhydrides, and several other compounds. More importantly, the carbonyl has the unique ability to behave as either a nucleophile (as in condensation reactions) or an electrophile (as in nucleophilic addition reactions). In this chapter, we will investigate the overall properties of aldehydes and ketones, as well as their oxidation–reduction reactions and electrophilic properties. In the following chapter, we will investigate their nucleophilic properties through enolate chemistry.
6.1 Description and Properties
LEARNING OBJECTIVES
After Chapter 6.1, you will be able to:
- Name aldehydes and ketones using the proper suffixes
- Describe the reactivity of the carbonyl carbon
- Recognize common reactions used to form aldehydes and ketones
- Predict the boiling point of a compound based on its oxidation level
A ketone has two alkyl groups bonded to the carbonyl, whereas an aldehyde has one alkyl group and one hydrogen. This means that the carbonyl in a ketone is never a terminal group, whereas it always is in an aldehyde. Like many organic compounds, aldehydes and ketones are often strong-smelling compounds. Volatile carbonyls are found in many spices, including cinnamon (cinnamaldehyde), vanilla (vanillin), cumin (cuminaldehyde), dill (carvenone), and ginger (zingerone).
KEY CONCEPT
An aldehyde is a terminal functional group. A ketone, on the other hand, will always be internal and can never be a terminal functional group.
Nomenclature
Aldehydes are named by replacing the –e at the end of the alkane name with the suffix –al. Common names for the first five aldehydes, shown in Figure 6.1, are formaldehyde, acetaldehyde, propionaldehyde, butyraldehyde, and valeraldehyde. When aldehydes are named as substituents, use the prefix oxo–.
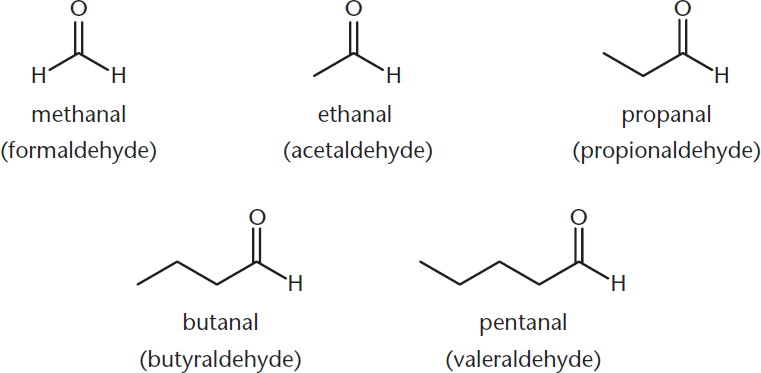
Figure 6.1. Naming Aldehydes
MCAT EXPERTISE
Notice that these common names have a pattern that can help us: form– will also be seen in formic acid (a one-carbon carboxylic acid), and acet– is seen in many two-carbon compounds (acetylene, acetic acid, and acetyl-CoA).
If the aldehyde is attached to a ring, the suffix –carbaldehyde is used instead. This is shown in Figure 6.2.
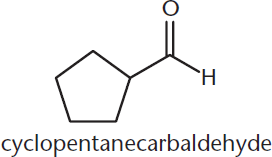
Figure 6.2. Naming Cyclic Aldehydes
Ketones are named by replacing the –e with the suffix –one. When naming ketones by their common names, the two alkyl groups are named alphabetically, followed by –ketone. When ketones are named as substituents, use either the prefix oxo– or keto–. Figure 6.3 shows some examples of ketones.
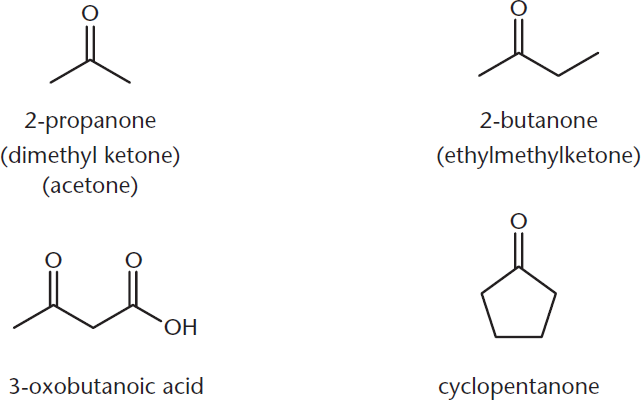
Figure 6.3. Naming Ketones
Physical Properties
The physical properties of aldehydes and ketones are governed by the presence of the carbonyl group. The dipole of the carbonyl is stronger than the dipole of an alcohol because the double-bonded oxygen is more electron-withdrawing than the single bond to oxygen in the hydroxyl group. In solution, the dipole moments associated with these polar carbonyl groups increase intermolecular attractions, causing an elevation in boiling point relative to their parent alkanes. However, even though aldehydes and ketones have dipoles more polar than those of alcohols, the elevation in boiling point is less than that in alcohols because no hydrogen bonding is present. In reactions, aldehydes and ketones both act as electrophiles, making good targets for nucleophiles. This is due to the electron-withdrawing properties of the carbonyl oxygen, which leaves a partial positive charge on the carbon, as shown in Figure 6.4. Generally, aldehydes are more reactive toward nucleophiles than ketones because they have less steric hindrance and fewer electron-donating alkyl groups.
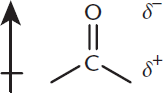
Figure 6.4. Polarity of the Carbonyl Group
KEY CONCEPT
While the dipole moment in the carbonyl group increases the intermolecular forces (and therefore boiling points) of aldehydes and ketones relative to alkanes, this is not as significant as the impact of hydrogen bonding seen in alcohols.
MCAT EXPERTISE
The carbonyl carbon is the most common electrophile you’ll see on Test Day. Remember why this group has a dipole moment: oxygen is more electronegative and pulls electrons away from the carbon, making the carbon electrophilic and a good target for nucleophiles.
Formation
Aldehydes and ketones can be produced by several mechanisms. An aldehyde can be obtained from the partial oxidation of a primary alcohol, although only by pyridinium chlorochromate (PCC; C5H5NH[CrO3Cl]). With any stronger oxidants, aldehydes will continue to be oxidized to carboxylic acids. A ketone can be obtained from the oxidation of a secondary alcohol. This can be performed using reagents ranging from sodium or potassium dichromate salts (Na2Cr2O7 or K2Cr2O7) to chromium trioxide (CrO3) to PCC. When oxidizing a secondary alcohol, there is no concern for oxidizing too far because the reaction will stop at the ketone stage.
MCAT CONCEPT CHECK 6.1:
Before you move on, assess your understanding of the material with these questions.
-
Name the following two compounds:
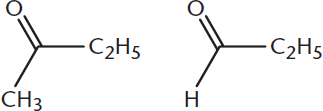
________________________
-
Given an alkane, an aldehyde, and an alcohol with equal-length carbon chains, which will have the highest boiling point? Why?
________________________
-
Is the carbon of a carbonyl electrophilic or nucleophilic? Why?
________________________
-
What is one method for forming an aldehyde? A ketone?
- Aldehyde:
________________________
- Ketone:
______________________________
6.2 Nucleophilic Addition Reactions
LEARNING OBJECTIVES
After Chapter 6.2, you will be able to:
- Predict the products of reacting aldehydes or ketones with alcohols, in the presence or absence of acidic conditions
- Recall the functional group formed when nitrogen-containing derivatives react with aldehydes or ketones
- Predict the product of the reaction of HCN with aldehydes or ketones, including the reactivity of the product:

In each of the following reactions, the general reaction mechanism is the same: nucleophilic addition to a carbonyl. This is one of the most important reaction mechanisms on the MCAT, and many of the reactions of aldehydes, ketones, and more complex molecules share this general reaction mechanism. Rather than memorizing each reaction individually, focus on the overall pattern—then learn how a particular reaction exemplifies it.
MCAT EXPERTISE
Memorizing one reaction may help you to get one question right on the MCAT, but understanding trends and overarching concepts will allow you to answer many more questions correctly. You will see that the carbonyl carbon is a great target for nucleophilic attack in many of the reactions in this chapter.
As we have seen, the C=O bond is polarized, with a partial positive charge on the carbonyl carbon and a partial negative charge on the oxygen. This makes the carbonyl carbon an electrophile, ripe for nucleophilic attack.
When the nucleophile attacks, it forms a covalent bond to the carbon, breaking the π bond in the carbonyl. The electrons from the π bond are pushed onto the oxygen atom. Oxygen happily accepts extra electrons due to its electronegativity. Breaking the π bond forms a tetrahedral intermediate. Any time a carbonyl is opened, one should ask: Can I reform the carbonyl? If no good leaving group is present (as is the case with aldehydes and ketones), the carbonyl will not reform. Generally, O– will accept a proton from the solvent to form a hydroxyl group, resulting in an alcohol. However, if a good leaving group is present (as is the case with carboxylic acids and their derivatives), the carbonyl double bond can reform, pushing off the leaving group. Figure 6.5 shows the reaction mechanism of nucleophilic addition for an aldehyde.
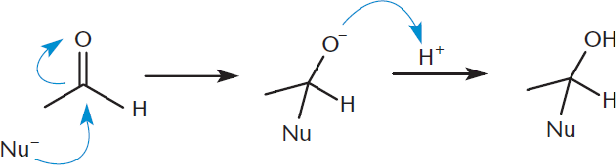
Figure 6.5. Nucleophilic Addition Reaction Mechanism The nucleophile attacks the carbonyl carbon, opening the carbonyl. The carbonyl cannot reform because there is no good leaving group; thus, the O− is protonated to generate an alcohol.
Hydration
In the presence of water, aldehydes and ketones react to form geminal diols (1,1-diols), as shown in Figure 6.6. In this case, the nucleophilic oxygen in water attacks the electrophilic carbonyl carbon. This hydration reaction normally proceeds slowly, but we can increase the rate by adding a small amount of catalytic acid or base. In acidic conditions, protonation of the carbonyl oxygen creates an activated electrophilic carbonyl carbon, which allows for the addition of a relatively weak nucleophile like water. By the end of the reaction, the proton is regenerated, making the acid a catalyst. In basic conditions, hydroxide is a relatively strong nucleophile and can directly attack the electrophilic carbonyl carbon without any activation. The resulting negative charge on the oxygen (after gaining a pair of electrons from the carbonyl bond) picks up a proton from water, regenerating a hydroxide.
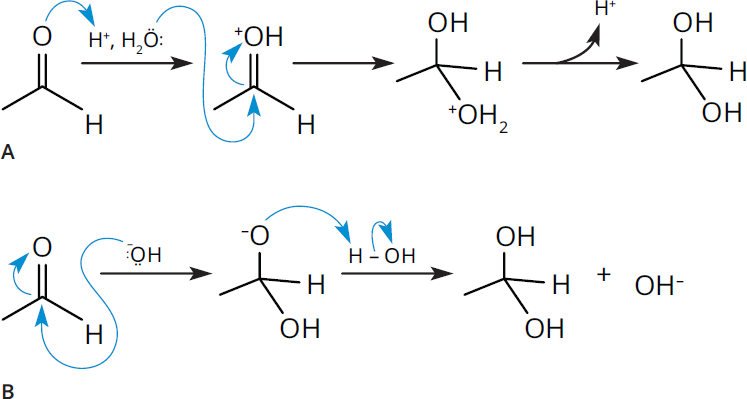
Figure 6.6. Nucleophilic Addition Reaction Mechanism in Acidic and Basic Conditions
Acetals and Hemiacetals
A similar reaction occurs when aldehydes and ketones are treated with alcohols. When one equivalent of alcohol (the nucleophile in this reaction) is added to an aldehyde or ketone, the product is a hemiacetal or hemiketal, respectively, as shown in Figure 6.7. Hemiacetals and hemiketals can be recognized by the retention of the hydroxyl group. This “halfway” step (hence the hemi– prefix) is the endpoint in basic conditions.
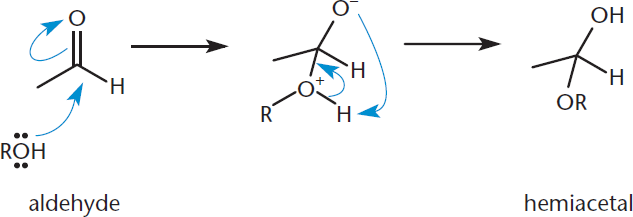
Figure 6.7. Hemiacetal Formation The oxygen in the alcohol functions as the nucleophile, attacking the carbonyl carbon and generating a hemiacetal.
When two equivalents of alcohol are added, the reaction proceeds to completion, resulting in the formation of an acetal or ketal, as shown in Figure 6.8. This reaction proceeds by a nucleophilic substitution reaction (SN1) and is catalyzed by anhydrous acid. The hydroxyl group of a hemiacetal or hemiketal is protonated under acidic conditions and lost as a molecule of water. A carbocation is thus formed, and another equivalent of alcohol attacks this carbocation, resulting in the formation of an acetal or ketal. Acetals and ketals, which are comparatively inert, are frequently used as protecting groups for carbonyl functionalities. Molecules with protecting groups can easily be converted back to carbonyls with aqueous acid and heat.
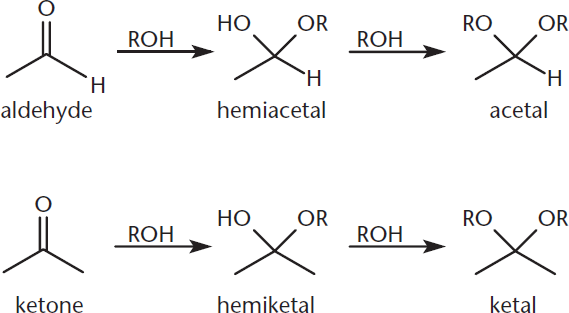
Figure 6.8. Acetal and Ketal Formation Once a hemiacetal or hemiketal is formed, the hydroxyl group is protonated and released as a molecule of water; alcohol then attacks, forming an acetal or ketal.
KEY CONCEPT
In the formation of hemiacetals and hemiketals, alcohol is the nucleophile and the carbonyl carbon is the electrophile. In the formation of acetals and ketals, alcohol is the nucleophile, and the carbocation carbon (formerly the carbonyl carbon) is the electrophile.
Imines and Enamines
Nitrogen and nitrogen-based functional groups act as good nucleophiles due to the lone pair of electrons on nitrogen, and react readily with the electrophilic carbonyls of aldehydes and ketones. In the simplest case, ammonia adds to the carbon atom and water is lost, producing an imine, a compound with a nitrogen atom double-bonded to a carbon atom. This reaction is shown in Figure 6.9. Because a small molecule is lost during the formation of a bond between two molecules, this is an example of a condensation reaction. Because nitrogen replaces the carbonyl oxygen, this is also an example of a nucleophilic substitution. Some common ammonia derivatives that react with aldehydes and ketones are hydroxylamine (H2N–OH), hydrazine (H2N–NH2), and semicarbazide (H2N–NH–C(O)NH2); these form oximes, hydrazones, and semicarbazones, respectively.
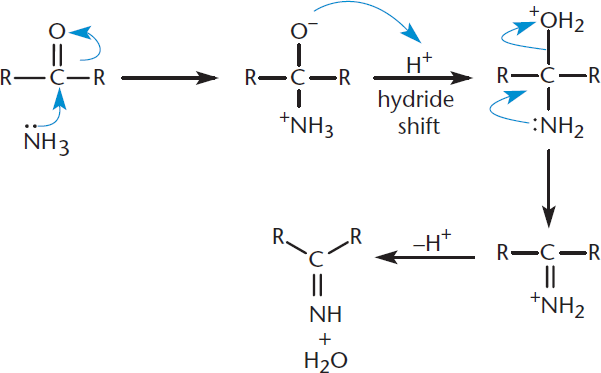
Figure 6.9. Imine Formation Ammonia is added to the carbonyl, resulting in the elimination of water and generation of an imine.
Imines and related compounds can undergo tautomerization to form enamines, which contain both a double bond and a nitrogen-containing group. This is analogous to the keto–enol tautomerization of carbonyl compounds and will be explored in Chapter 7 of MCAT Organic Chemistry Review.
Cyanohydrins
Hydrogen cyanide (HCN) is a classic nucleophile on the MCAT. HCN has both triple bonds and an electronegative nitrogen atom, rendering it relatively acidic with a pKa of 9.2. After the hydrogen dissociates, the nucleophilic cyanide anion can attack the carbonyl carbon atom, as shown in Figure 6.10. Reactions with aldehydes and ketones produce stable compounds called cyanohydrins once the oxygen has been reprotonated. The cyanohydrin gains its stability from the newly formed C–C bond.
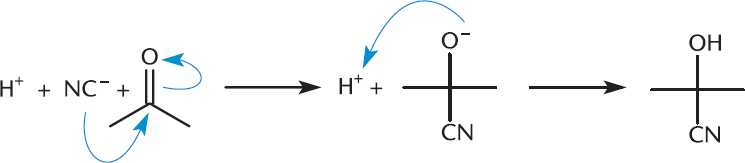
Figure 6.10. Cyanohydrin Formation Cyanide functions as a nucleophile, attacking the carbonyl carbon and generating a cyanohydrin.
KEY CONCEPT
In a reaction with HCN,
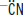
is the nucleophile; the carbonyl carbon is the electrophile.
MCAT CONCEPT CHECK 6.2:
Before you move on, assess your understanding of the material with these questions.
-
When an aldehyde or ketone is reacted with one equivalent of an alcohol, what occurs? What would be different if it were reacted with two equivalents in acidic conditions?
- Aldehyde or ketone + 1 equivalent of alcohol:
________________________
- Aldehyde or ketone + 2 equivalents of alcohol: ______________________________
-
When nitrogen or nitrogen-containing derivatives react with aldehydes and ketones, what type of reaction happens, and what functional group is formed?
________________________
-
When HCN reacts with an aldehyde or ketone, what functional group is produced? Is the product stable?
________________________
6.3 Oxidation–Reduction Reactions
LEARNING OBJECTIVES
After Chapter 6.3, you will be able to:
- Recall common oxidizing and reducing agents used on aldehydes and ketones
- Predict the products of redox reactions involving aldehydes and ketones
Aldehydes occupy the middle of the oxidation–reduction spectrum; they are more oxidized than alcohols but less oxidized than carboxylic acids. Ketones, on the other hand, are as oxidized as secondary carbons can get.
Oxidation of Aldehydes
When aldehydes are further oxidized, they form carboxylic acids. Any oxidizing agent stronger than PCC can perform this reaction. Some examples include potassium permanganate (KMnO4), chromium trioxide (CrO3), silver(I) oxide (Ag2O), and hydrogen peroxide (H2O2). This is shown in Figure 6.11.
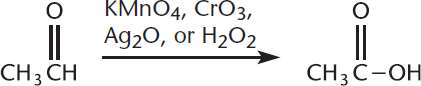
Figure 6.11. Aldehyde Oxidation Most oxidizing agents will turn aldehydes into carboxylic acids; PCC, however, is anhydrous and is not strong enough to oxidize past the point of an aldehyde.
Reduction by Hydride Reagents
Aldehydes and ketones can also undergo reduction to form alcohols. This is often performed with hydride reagents. The most common of these seen on the MCAT are lithium aluminum hydride (LiAlH4) and sodium borohydride (NaBH4), which is often used when milder conditions are needed. This reaction is shown in Figure 6.12.
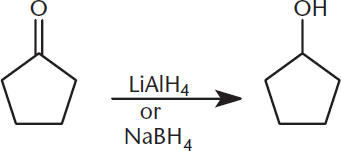
Figure 6.12. Ketone Reduction Ketones are easily reduced to their respective alcohols using hydride reagents.
MCAT CONCEPT CHECK 6.3:
Before you move on, assess your understanding of the material with these questions.
-
What functional group is formed when an aldehyde is oxidized? What are some common oxidizing agents that assist this reaction?
- Functional group:
________________________
- Oxidizing agents:
________________________
-
What functional group is formed when aldehydes and ketones are reduced? What are some common reducing agents that assist this reaction?
- Functional group:
________________________
- Reducing agents:
________________________
-
A chemistry student reacts butanone and butanal each with PCC and KMnO4. What are the expected products of each reaction?
- Butanone:
________________________
- Butanal:
________________________
Conclusion
In this chapter, we have examined the properties of aldehydes and ketones. Specifically, we have taken a look at the reactivity of the carbonyl carbon in nucleophilic addition reactions and examined how aldehydes and ketones can be oxidized and reduced. Carbonyls are common reaction sites in many biosynthetic processes, which helps explain their importance on the MCAT.
In the following chapter, we will continue our exploration of aldehydes and ketones by looking at the chemistry of enolates, which are nucleophilic carbonyl-containing compounds.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Description and Properties
- Aldehydes are terminal functional groups containing a carbonyl bonded to at least one hydrogen. In nomenclature, they use the suffix –al and the prefix oxo–. In rings, they are indicated by the suffix –carbaldehyde.
- Ketones are internal functional groups containing a carbonyl bonded to two alkyl chains. In nomenclature, they use the suffix –one and the prefix oxo– or keto–.
- The reactivity of a carbonyl (C=O) is dictated by the polarity of the double bond. The carbon has a partial positive charge and is therefore electrophilic.
- Carbonyl-containing compounds have higher boiling points than equivalent alkanes because of dipole interactions. Alcohols have higher boiling points than carbonyls because of hydrogen bonding.
- Aldehydes and ketones are commonly produced by oxidation of primary and secondary alcohols, respectively.
- Weaker, anhydrous oxidizing agents like pyridinium chlorochromate (PCC) must be used for synthesizing aldehydes, or the reaction will continue oxidizing to the level of the carboxylic acid.
- Various oxidizing agents can be used for ketones, such as dichromate, chromium trioxide, or PCC because ketones are the most oxidized functional group for secondary carbons.
Nucleophilic Addition Reactions
- When a nucleophile attacks and forms a bond with a carbonyl carbon, electrons in the π bond are pushed to the oxygen atom.
- If there is no good leaving group (aldehydes and ketones), the carbonyl will remain open and is protonated to form an alcohol.
- If there is a good leaving group (carboxylic acids and derivatives), the carbonyl will reform and kick off the leaving group.
- In hydration reactions, water adds to a carbonyl, forming a geminal diol.
- When one equivalent of alcohol reacts with an aldehyde (via nucleophilic addition), a hemiacetal is formed. When the same reaction occurs with a ketone, a hemiketal is formed.
- When another equivalent of alcohol reacts with a hemiacetal (via nucleophilic substitution), an acetal is formed. When the same reaction occurs with a hemiketal, a ketal is formed.
- Nitrogen and nitrogen derivatives react with carbonyls to form imines, oximes, hydrazones, and semicarbazones. Imines can tautomerize to form enamines.
- Hydrogen cyanide reacts with carbonyls to form cyanohydrins.
Oxidation–Reduction Reactions
- Aldehydes can be oxidized to carboxylic acids using an oxidizing agent like KMnO4, CrO3, Ag2O, or H2O2. They can be reduced to primary alcohols via hydride reagents (LiAlH4, NaBH4).
- Ketones cannot be further oxidized, but can be reduced to secondary alcohols using the same hydride reagents.
ANSWERS TO CONCEPT CHECKS
**6.1**
- The molecule on the left is butanone. The molecule on the right is propanal.
- The alkane will have the lowest boiling point, followed by the aldehyde, and then the alcohol. The boiling point of the aldehyde is elevated by its dipole, but the boiling point of an alcohol is further elevated by hydrogen bonding.
- The carbon in a carbonyl is electrophilic; it is partially positively charged because oxygen is highly electron-withdrawing.
- Aldehydes can be formed by the oxidation of primary alcohols, but can only be produced using weaker (and anhydrous) oxidizing agents like PCC—otherwise, they will oxidize fully to carboxylic acids. Ketones can be formed by the oxidation of secondary alcohols. Other methods can be used as well (ozonolysis, Friedel–Crafts acylation), but these are outside the scope of the MCAT.
**6.2**
- With one equivalent of alcohol, aldehydes and ketones will form hemiacetals and hemiketals, respectively. With two equivalents of alcohol, the reaction will run to completion, forming acetals and ketals, respectively.
- The reaction that occurs is a condensation reaction because a small molecule is lost, and also a nucleophilic substitution reaction. This reaction results in the formation of an imine (or, for nitrogen-containing derivatives: oximes, hydrazones, or semicarbazones).
- When HCN reacts with an aldehyde or ketone, a cyanohydrin is produced, which is a stable product.
**6.3**
- Oxidizing an aldehyde yields a carboxylic acid. Common oxidizing agents include KMnO4, CrO3, Ag2O, and H2O2.
- Reducing an aldehyde or ketone yields an alcohol. Under certain conditions not tested on the MCAT, aldehydes and ketones can be reduced all the way to alkanes. Common reducing agents include LiAlH4 and NaBH4.
- Butanone reacts with neither PCC nor KMnO4 because ketones cannot be oxidized with common oxidizing reagent that cannot break the carbon–carbon bond. Butanal is oxidized by KMnO4 to form butanoic acid, but does not react with PCC, which is not a strong enough oxidant.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
The reactivity of the carbonyl can be attributed to the difference in electronegativity between the carbon and oxygen atoms. The more electronegative oxygen atom attracts the bonding electrons and is therefore electron-withdrawing. Thus, the carbonyl carbon is electrophilic. One resonance structure of the carbonyl pushes the π electrons onto the oxygen, resulting in a positively charged carbonyl carbon.
2. B
Assuming the length of the carbon chain remains the same, the alkane consistently has the lowest boiling point. The boiling point of the ketone is elevated by the dipole in the carbonyl. The boiling point of the alcohol is elevated further by hydrogen bonding.
3. C
The reaction between a ketone and one equivalent of alcohol produces a hemiketal. This has an –OR group, an –OH group, and two alkyl groups attached at the same carbon. (A) is a ketal, with two –OR groups and two –R groups. (B) is a hemiacetal, with an –OH group, an –OR group, one R group, and a hydrogen atom (not drawn). (D) is a ketone. Note that a hemiketal is a very unstable compound, and will react rapidly with a second equivalent of alcohol to form a ketal in acidic conditions.
4. A
Aldehydes and ketones react with ammonia and nitrogen-based derivatives to form imines—compounds with a double bond between carbon and nitrogen.
5. B
Aldehydes are easily oxidized to the corresponding carboxylic acids by KMnO4. In (A), the aldehyde has been reduced to an alcohol. In (C), the molecule has not reacted. In (D), the aldehyde has been oxidized, but a –CH2– group has been removed.
6. B
LiAlH4 reduces aldehydes to primary alcohols and ketones to secondary alcohols. In this reaction, a ketone is converted to a secondary alcohol.
7. D
Because an excess of ethanol is present, the product of the reaction between this aldehyde and ethanol will be an acetal. The benzaldehyde will first be converted to a hemiacetal, shown in (C), but will then proceed to completion as an acetal. (A) and (B) are incorrect because they show the presence of two benzene rings in the final product.
8. B
Hemiacetals and hemiketals are usually short-lived because the –OH group will rapidly become protonated in acidic conditions and is lost as water, leaving behind a carbocation that is very susceptible to attack by an alcohol. Once the alcohol has been added, the acetal or ketal becomes more stable because the newly added group is less likely to become protonated and leave as compared to –OH.
9. A
A hemiacetal is a molecule in which one equivalent of alcohol has been added to a carbonyl (–OR) and the carbonyl oxygen has been protonated (–OH). Otherwise, there is the same alkyl group (–R) and hydrogen atom (–H) as the parent aldehyde. (B) describes an acetal, (C) a hemiketal, and (D) a ketal.
10. A
Although both the aldehyde and ketone listed will be reactive with the strongly nucleophilic hydrogen cyanide, aldehydes are slightly more reactive toward nucleophiles than ketones for steric reasons, so the aldehyde and HCN will form the major product (which will be a cyanohydrin).
11. C
PCC is a mild anhydrous oxidant that can oxidize primary alcohols to aldehydes, and secondary alcohols to ketones. It is not strong enough to oxidize alcohols or aldehydes to carboxylic acids.
12. B
In a hydration reaction, water adds to a carbonyl, forming a geminal diol—a compound with two hydroxyl groups on the same carbon. Hydrogen peroxide and potassium dichromate are oxidizing agents that can convert an aldehyde to a carboxylic acid. Ethanol will react with a carbonyl compound to form an acetal or a ketal, if excess ethanol is available.
13. A
Ammonia, or NH3, will react with an aldehyde like glutaraldehyde to form an imine. This is a condensation and a substitution reaction, as the C=O of the carbonyl will be replaced with a C=N bond.
14. C
Hydrides like LiAlH4 and NaBH4 are reducing agents; as such, they will reduce aldehydes and ketones to alcohols. The other reagents listed are oxidizing agents, which will not act on a ketone.
15. D
During tautomerization, the double bond between the carbon and nitrogen in an imine is moved to lie between two carbons. This results in an enamine—a compound with a double bond and an amine.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 4
- Carbohydrate Structure and Function
- General Chemistry Chapter 11
- Oxidation–Reduction Reactions
- Organic Chemistry Chapter 1
- Nomenclature
- Organic Chemistry Chapter 4
- Analyzing Organic Reactions
- Organic Chemistry Chapter 7
- Aldehydes and Ketones II
- Organic Chemistry Chapter 8
- Carboxylic Acids