Chapter 7: Aldehydes and Ketones II: Enolates

Chapter 7: Aldehydes and Ketones II: Enolates
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
What is the product of the reaction below?

-
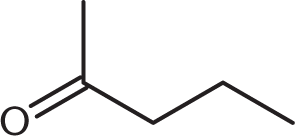
-
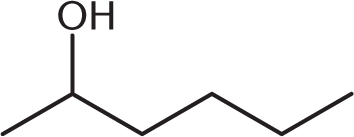
-
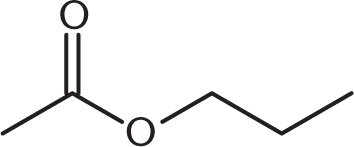
-
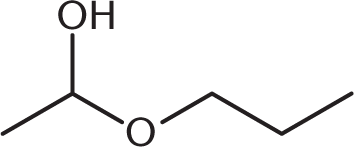
-
The reaction below is an example of:

- esterification.
- tautomerization.
- elimination.
- dehydration.
-
Which of the following reactions would produce the compound below?
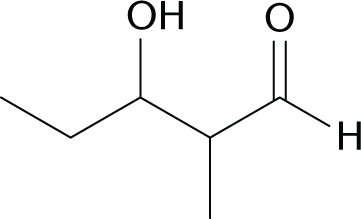
- CH3CHO + CH3CH2CH2CHO →
- CH3COCH3 + CH3CH2CH2CHO →
- CH3CH2COCH3 + CH3CHO →
- CH3CH2CHO + CH3CH2CHO →
-
Why does the equilibrium between keto and enol tautomers lie far to the keto side?
- The keto form is more thermodynamically stable.
- The enol form is lower energy.
- The enol form is more thermodynamically stable.
- I only
- III only
- I and II only
- II and III only
-
The aldol condensation is an example of which reaction type(s)?
- Dehydration
- Cleavage
- Nucleophilic addition
- I only
- I and III only
- II and III only
- I, II, and III
-
Which of the hydrogens in the following molecule is the most acidic?
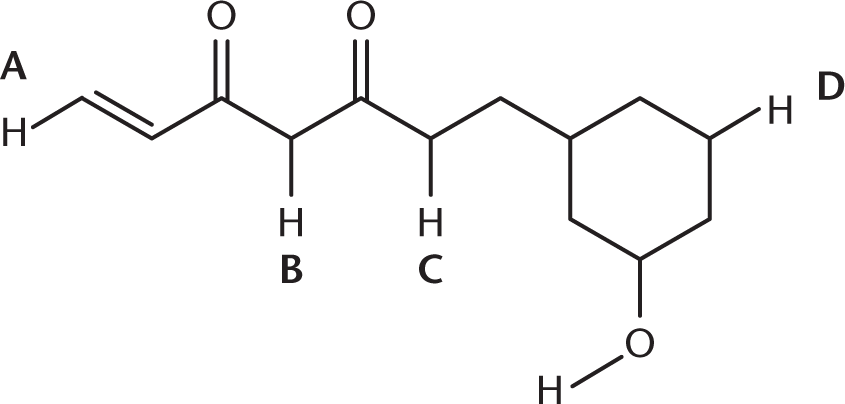
- A
- B
- C
- D
-
When reacted with ammonia (NH3) at 200 °C, which enolate of a carbonyl-containing compound would predominate?
- Kinetic enolate
- Thermodynamic enolate
- Neither enolate; they would be present in roughly equal proportions.
- Neither enolate; these reaction conditions would not form either enolate.
-
Which of the following compounds would be most reactive toward a nucleophile?
- Pentanal
- 3-Pentanone
- Pentane
- 2-Nonanone
-
α-hydrogens of a ketone are acidic due to:
- resonance stabilization.
- the electron-withdrawing properties of the alkyl groups.
- the electronegative carbonyl oxygen.
- I only
- I and III only
- II and III only
- I, II, and III
-
Which of the following is considered a tautomer of the imine functional group?
- Cyanohydrin
- Hydrazone
- Enamine
- Semicarbazone
-
When succinaldehyde is treated with lithium diisopropylamide (LDA), it:
- becomes more nucleophilic.
- becomes less nucleophilic.
- generates a carbanion.
- I only
- II only
- I and III only
- II and III only
-
Which of the following best describes the final product of an aldol condensation?
- 1,3-dicarbonyl
- 1,2-dicarbonyl
- α,β-unsaturated carbonyl
- β,γ-unsaturated carbonyl
-
When benzaldehyde is reacted with acetone, which will act as the nucleophile?
- Benzaldehyde, after addition of strong acid
- Benzaldehyde, after reaction with strong base
- Acetone, after addition of strong acid
- Acetone, after reaction with strong base
-
3-Hydroxybutanal can be formed by the reaction of:
- methanol in diethyl ether.
- ethanal in base, then in acid.
- butanal in strong acid.
- methanal and ethanal in catalytic base.
-
The catalytic production of dihydroxyacetone and glyceraldehyde 3-phosphate (2-hydroxy-3-oxopropyl dihydrogen phosphate) from fructose-1,6-bisphosphate ([(2S,3S,4S,5R)-2,3,4-trihydroxy-5-(phosphonooxymethyl)oxolan-2-yl]methyl dihydrogen phosphate) is what type of reaction?
- Aldol condensation
- Retro-aldol reaction
- Dehydration
- Nucleophilic attack
Answer Key
- D
- B
- D
- A
- B
- B
- B
- A
- B
- C
- C
- C
- D
- B
- B
Chapter 7: Aldehydes and Ketones II: Enolates
CHAPTER 7
ALDEHYDES AND KETONES II: ENOLATES
In This Chapter
7.1 General Principles
Acidity of α-Hydrogens
Steric Hindrance
7.2 Enolate Chemistry
Keto–Enol Tautomerization
Kinetic and Thermodynamic Enolates
Enamines
7.3 Aldol Condensation
The Retro-Aldol Reaction
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 6% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content category:
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
In the previous chapter, we took a look at a few key properties and reactions of aldehydes and ketones. These molecules have highly predictable chemistry centered on their electrophilic, positively charged carbonyl carbon and will be sure to show up on Test Day. In this chapter, we take a look at several more properties of aldehydes and ketones, and focus on the reactivity of the α-hydrogen of carbonyl-containing compounds. The acidity of this α-hydrogen allows many aldehydes and ketones to act as both electrophiles and nucleophiles. Sometimes, they can even serve both functions in the same reaction. But don’t worry—we’ll also review some tips about how to understand when and how aldehydes and ketones will react.
7.1 General Principles
LEARNING OBJECTIVES
After Chapter 7.1, you will be able to:
- Explain the acidic nature of α-hydrogens on aldehydes and ketones
- Compare the acidity of the α-hydrogens of aldehydes to those of ketones
- Describe the relationship between steric hindrance and reactivity
In the previous chapter, we focused on how the electronegativity of the oxygen atom in a carbonyl pulls electrons away from the carbonyl carbon, making it partially positively charged. In this chapter, we take the electron-withdrawing characteristics of oxygen one bond further, focusing on the α-carbon in an aldehyde or ketone.
Acidity of *α*-Hydrogens
An α-carbon is adjacent to the carbonyl carbon, and the hydrogens connected to the α-carbon are termed α-hydrogens. Through induction, oxygen pulls some of the electron density out of these C–H bonds, weakening them. This makes it relatively easy to deprotonate the α-carbon of an aldehyde or ketone, as shown in Figure 7.1. The acidity of α-hydrogens is augmented by resonance stabilization of the conjugate base. Specifically, when the α-hydrogen is removed, the extra electrons that remain can resonate between the α-carbon, the carbonyl carbon, and the carbonyl oxygen. This increases the stability of this enolate intermediate, described in the next section. Through this resonance, the negative charge can be distributed to the more electronegative oxygen atom. The electron-withdrawing oxygen atom thereby helps stabilize the carbanion (a molecule with a negatively charged carbon atom). When in basic solutions, α-hydrogens will easily deprotonate.
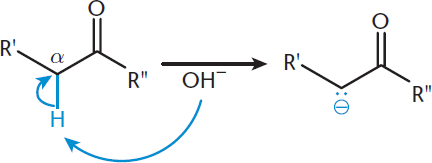
Figure 7.1. Deprotonation of an α-Carbon, Forming a Carbanion
KEY CONCEPT
Electron-withdrawing groups like oxygen stabilize organic anions. Electron-donating groups like alkyl groups destabilize organic anions.
The α-hydrogens of ketones tend to be slightly less acidic than those of aldehydes due to the electron-donating properties of the additional alkyl group in a ketone. This property is the same reason that alkyl groups help to stabilize carbocations—but in this case, they destabilize the carbanion.
Steric Hindrance
In reactions, aldehydes are slightly more reactive to nucleophiles than ketones. This is due in part to steric hindrance in the ketone, which arises from the additional alkyl group that ketones contain. When the nucleophile approaches the ketone or aldehyde in order to react, the additional alkyl groups on the ketone are in the way, more so than the single hydrogen of the aldehyde. This makes for a higher-energy, more crowded intermediate step. Remember, higher-energy intermediates mean that the reaction is less likely to proceed.
KEY CONCEPT
Ketones are slightly less likely to react with nucleophiles than aldehydes because the extra alkyl group destabilizes the carbanion and increases steric hindrance.
MCAT CONCEPT CHECK 7.1:
Before you move on, assess your understanding of the material with these questions.
-
Why are the α-hydrogens of aldehydes and ketones acidic?
________________________
-
Which has a lower pKa: 3-pentanone or pentanal? Why?
________________________
-
How does steric hindrance affect the relative reactivity of aldehydes and ketones?
________________________
7.2 Enolate Chemistry
LEARNING OBJECTIVES
After Chapter 7.2, you will be able to:
- Define tautomerization
- Predict the role of an enolate carbanion in a reaction
- Describe the conditions that favor keto and enol forms
- Identify the thermodynamically favored tautomer of an aldehyde or ketone:
Due to the acidity of the α-hydrogen, aldehydes and ketones exist in solution as a mixture of two isomers: the familiar keto form, and the enol form.
Keto–Enol Tautomerization
The enol form gets its name from the presence of a carbon–carbon double bond (the en– component) and an alcohol (the –ol component). The two isomers, which differ in the placement of a proton and the double bond, are called tautomers. The equilibrium between the tautomers lies far to the keto side, so there will be many more keto isomers in solution. The process of interconverting from the keto to the enol tautomer, shown in Figure 7.2, is called enolization, or, more generally, tautomerization. By extension, any aldehyde or ketone with a chiral α-carbon will rapidly become a racemic mixture as the keto and enol forms interconvert, a phenomenon known as ***α*-racemization**.
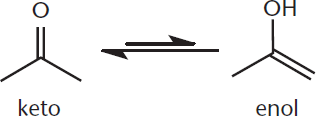
Figure 7.2. Enolization (Tautomerization) On the left is the keto form, which is thermodynamically favored over the enol form on the right.
KEY CONCEPT
Aldehydes and ketones exist in the traditional keto form (C=O) and as the less common enol tautomer (enol = ene + ol). The deprotonated enolate form can act as a nucleophile. Note that tautomers are not resonance structures because they differ in their connectivity of atoms.
Enols are important intermediates in many reactions of aldehydes and ketones. The enolate carbanion results from the deprotonation of the α-carbon by a strong base, as described earlier. Common strong bases include the hydroxide ion, lithium diisopropyl amide (LDA), and potassium hydride (KH). A 1,3-dicarbonyl is particularly acidic because there are two carbonyls to delocalize negative charge and, as such, is often used to form enolate carbanions. Once formed, the nucleophilic carbanion reacts readily with electrophiles. We will see one example of this shortly in the aldol condensation. Another example of this type of reaction is a Michael addition, shown in Figure 7.3, in which the carbanion attacks an α,β-unsaturated carbonyl compound—a molecule with a multiple bond between the α- and β-carbons next to a carbonyl.
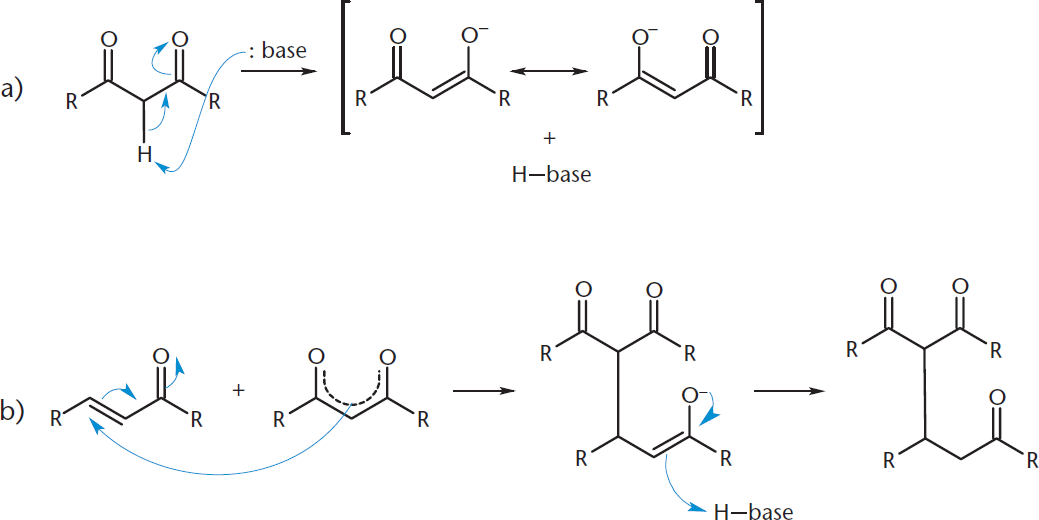
Figure 7.3. Michael Addition (a) The base deprotonates the α-carbon, making it a good nucleophile; (b) The carbanion attacks the double bond, resulting in a Michael addition.
This reaction proceeds as shown due to the resonance stabilization of the intermediates. The better you understand the resonance forms of molecules, the more you will be able to predict the specific location on a molecule where a reaction will occur.
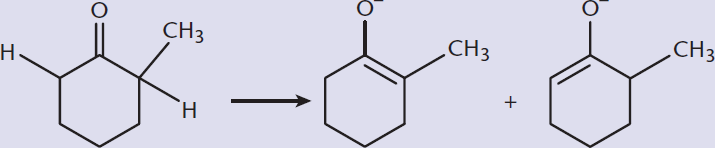
Kinetic and Thermodynamic Enolates
Given a ketone that has two different alkyl groups, each of which may have α-hydrogens, two forms of the enolate can form, with the carbon–carbon double bond between the carbonyl carbon and either the more or less substituted carbon, as shown in Figure 7.4. The equilibrium between these forms is dictated by the kinetic and thermodynamic control of the reaction. The kinetically controlled product is formed more rapidly but is less stable. This form has the double bond to the less substituted α-carbon. As expected, this product is formed by the removal of the α-hydrogen from the less substituted α-carbon because it offers less steric hindrance. The thermodynamically controlled product is formed more slowly, but is more stable and features the double bond being formed with the more substituted α-carbon. Accordingly, this is formed by the removal of the α-hydrogen from the more substituted α-carbon.
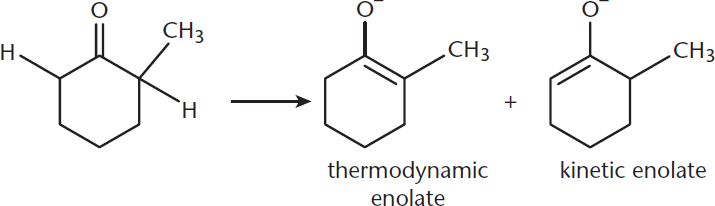
Figure 7.4. Kinetic and Thermodynamic Enolates The kinetic enolate forms more quickly, but is less stable than the thermodynamic enolate.
Each of these two products is favored by different conditions. The kinetic product is favored in reactions that are rapid, irreversible, at lower temperatures, and with a strong, sterically hindered base. If the reaction is reversible, the kinetic product can revert to the original reactant and react again to form the thermodynamic product. The thermodynamic product is favored with higher temperatures; slow, reversible reactions; and weaker, smaller bases.
Enamines
Just as enols are tautomers of carbonyls, enamines are tautomers of imines. An imine is a compound that contains a C=N bond. The nitrogen in the imine may or may not be bonded to an alkyl group or other substituent. Through tautomerization (movement of a hydrogen and a double bond), imines can be converted into enamines, as shown in Figure 7.5.
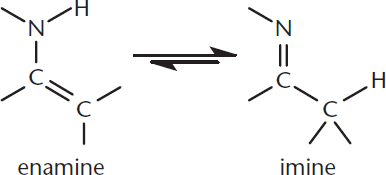
Figure 7.5. Enamination (Tautomerization) On the right is the imine form, which is thermodynamically favored over the enamine form on the left.
MCAT CONCEPT CHECK 7.2:
Before you move on, assess your understanding of the material with these questions.
-
What are tautomers?
________________________
-
Which tautomer of aldehydes and ketones is thermodynamically favored: keto or enol?
________________________
-
Which role does the enolate carbanion play in organic reactions: nucleophile, electrophile, oxidizing agent, or reducing agent?
________________________
-
In the following reaction, which product is the kinetic enolate? The thermodynamic? What conditions favor the formation of each?
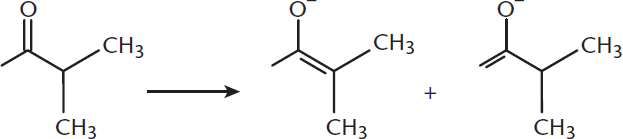
- Kinetic enolate:
______________________________
- Thermodynamic enolate:
______________________________
7.3 Aldol Condensation
LEARNING OBJECTIVES
After Chapter 7.3, you will be able to:
- Identify the species that act as nucleophiles and electrophiles in aldol condensations
- Describe the conditions, reactants, and products involved in a retro-aldol reaction
- List the reaction types associated with aldol condensation
The aldol condensation is another vital reaction for the MCAT. This reaction follows the same general mechanism of nucleophilic addition to a carbonyl as previously described. In this case, however, an aldehyde or ketone acts both as an electrophile (in its keto form) and a nucleophile (in its enolate form), and the end result is the formation of a carbon–carbon bond.
KEY CONCEPT
In aldol condensations, it’s the same nucleophilic addition reaction that we have seen before with carbonyl compounds—just with the carbonyl-containing compound acting as both a nucleophile and an electrophile.
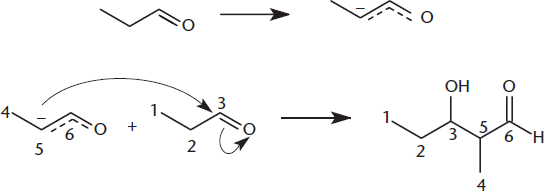
As shown in Figure 7.6, when acetaldehyde (ethanal) is treated with a catalytic amount of base, an enolate ion is produced. The enolate is more nucleophilic than the enol because it is negatively charged.
This nucleophilic enolate ion can react with the electrophilic carbonyl group of another acetaldehyde molecule. The key to this reaction is that both species are in the same flask. The product is 3-hydroxybutanal, which is an example of an aldol (a molecule that contains both aldehyde and alcohol functional groups). Note that the mechanism is still called an aldol reaction even when the reactants are ketones.
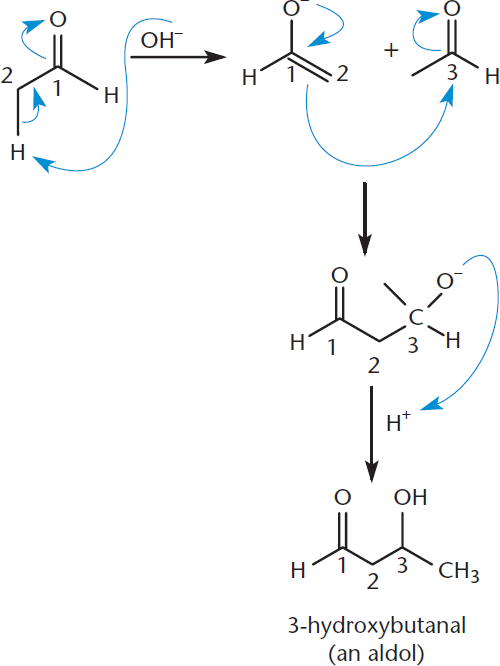
Figure 7.6. Aldol Condensation, Step 1: Forming the Aldol An enolate ion is formed, which then attacks the carbonyl carbon, forming an aldol.
With a strong base and high temperatures, dehydration occurs by an E1 or E2 mechanism: we kick off a water molecule and form a double bond, producing an α, β-unsaturated carbonyl, as shown in Figure 7.7.
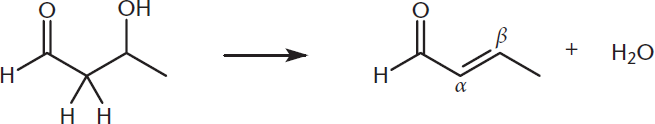
Figure 7.7. Aldol Condensation, Step 2: Dehydration of the Aldol The –OH is removed as water (dehydration), forming a double bond.
Aldol condensations are most useful if we only use one type of aldehyde or ketone. If there are multiple aldehydes or ketones, we cannot easily control which will act as the nucleophile and which will act as the electrophile, and a mixture of products will result. This can be prevented if one of the molecules has no α-hydrogens because the α-carbons are quaternary (like benzaldehyde).
This reaction is referred to as a condensation reaction because two molecules are joined with the loss of a small molecule. This type of reaction is also a dehydration reaction because the small molecule that is lost is water.
The Retro-Aldol Reaction
The reverse of this reaction is called a retro-aldol reaction. To push the reaction in a retro-aldol direction, aqueous base is added and heat is applied. The retro-aldol reaction is useful for breaking bonds between the α-and β-carbons of a carbonyl, as shown in Figure 7.8. This reaction is facilitated if the intermediate can be stabilized in the enolate form, just as in the forward reaction.
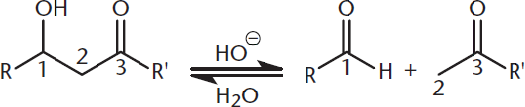
Figure 7.8. Retro-Aldol Reaction The bond between the α- and β-carbons of a carbonyl is broken.
KEY CONCEPT
In a retro-aldol reaction, a bond is broken between the α-and β-carbons of a carbonyl, forming two aldehydes, two ketones, or one aldehyde and one ketone.
MCAT CONCEPT CHECK 7.3:
Before you move on, assess your understanding of the material with these questions.
-
In the following reaction, an aldehyde is treated with a catalytic amount of base and an enolate ion is formed. The enolate then reacts with another aldehyde molecule leading to an aldol condensation. Identify the nucleophile and electrophile in the aldol condensation.
________________________
-
What is a retro-aldol reaction? What conditions favor retro-aldol reactions?
________________________
-
The aldol condensation can be classified under many categories of reactions. List some of these reaction types, and provide a short description of each.
________________________
Conclusion
In this second chapter on aldehydes and ketones, we’ve taken a look at the important resonance structures that the carbonyl of aldehydes and ketones allows. The high electronegativity of the oxygen atom in a carbonyl not only makes the carbonyl carbon electrophilic, but also weakens the C–H bonds on α-carbons. Deprotonation of this α-carbon results in an enolate, a nucleophilic version of carbonyl-containing compounds. Thus, while the carbonyl carbon dictates the electrophilic chemistry of carbonyls, it is the α-carbon, along with its acidic hydrogens, that dictates the nucleophilic chemistry of carbonyls.
Aldehydes and ketones are not the only carbonyl-containing compounds, of course. Carboxylic acids and their derivatives, including esters, anhydrides, and amides, also have chemistry controlled by a carbonyl. But there is one critical difference between aldehydes and ketones, and carboxylic acids and their derivatives: the absence or presence of a leaving group. While aldehydes and ketones lack leaving groups, carboxylic acids and carboxylic acid derivatives have leaving groups with varying degrees of stability. Over the next two chapters, we’ll explore the chemistry of these interesting groups of compounds.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
General Principles
- The carbon adjacent to the carbonyl carbon is termed an α-carbon; the hydrogens attached to the α-carbon are called α-hydrogens.
- α-hydrogens are relatively acidic and can be removed by a strong base.
- The electron-withdrawing oxygen of the carbonyl weakens the C–H bonds on α-carbons.
- The enolate resulting from deprotonation can be stabilized by resonance with the carbonyl.
- Ketones are less reactive toward nucleophiles because of steric hindrance and α-carbanion destabilization.
- The presence of an additional alkyl group crowds the transition step and increases its energy.
- The alkyl group also donates electron density to the carbanion, making it less stable.
Enolate Chemistry
- Aldehydes and ketones exist in the traditional keto form (C=O) and in the less common enol form (ene + ol = double bond + hydroxyl group).
- Tautomers are isomers that can be interconverted by moving a hydrogen and a double bond. The keto and enol forms are tautomers of each other.
- The enol form can be deprotonated to form an enolate. Enolates are good nucleophiles.
- In the Michael addition, an enolate attacks an α,β-unsaturated carbonyl, creating a bond.
- The kinetic enolate is favored by fast, irreversible reactions at lower temperatures with strong, sterically hindered bases. The thermodynamic enolate is favored by slower, reversible reactions at higher temperatures with weaker, smaller bases.
- Enamines are tautomers of imines. Like enols, enamines are the less common tautomer.
Aldol Condensation
- In the aldol condensation, the aldehyde or ketone acts as both nucleophile and electrophile, resulting in the formation of a carbon–carbon bond in a new molecule called an aldol.
- An aldol contains both aldehyde and alcohol functional groups.
- The nucleophile is the enolate formed from the deprotonation of the α-carbon.
- The electrophile is the aldehyde or ketone in the form of the keto tautomer.
- First, a condensation reaction occurs in which the two molecules come together.
- After the aldol is formed, a dehydration reaction (loss of a water molecule) occurs. This results in an α,β-unsaturated carbonyl.
- Retro-aldol reactions are the reverse of aldol condensations.
- Retro-aldol reactions are catalyzed by heat and base.
- In these reactions, the bond between an α- and β-carbon is cleaved.
ANSWERS TO CONCEPT CHECKS
**7.1**
- The α-hydrogens of aldehydes and ketones are acidic, or deprotonate easily, due to both inductive effects and resonance effects. The electronegative oxygen atom pulls electron density from the C–H bond, weakening it. Once deprotonated, the resonance stabilization of the negative charge between the α-carbon, carbonyl carbon, and electron-withdrawing carbonyl oxygen increases the stability of this form.
- The α-hydrogens of aldehydes are slightly more acidic than those of ketones due to the electron-donating characteristics of the second alkyl group in ketones. This extra alkyl group destabilizes the carbanion, which slightly disfavors the loss of the α-hydrogens in ketones as compared to aldehydes. Therefore, pentanal is a stronger acid than 3-pentanone and will have a lower pKa.
- Steric hindrance is one of the two reasons that aldehydes are slightly more reactive than ketones. The additional alkyl group gets in the way and makes for a higher-energy, crowded intermediate.
**7.2**
- Tautomers are isomers that can be interconverted by the movement of a hydrogen and a double bond.
- The keto form is thermodynamically favored.
- Enolate carbanions act as nucleophiles.
- The product with the double bond to the less substituted α-carbon is the kinetically controlled product (the product shown on the right). The thermodynamically controlled product has the double bond formed with the more substituted α-carbon (the product on the left). Because the kinetic enolate forms rapidly and can interconvert with the thermodynamic form if given time, the kinetic form is favored by fast, irreversible reactions, such as with a strong, sterically hindered base, and lower temperatures. The thermodynamic form, on the other hand, is favored by slower, reversible reactions, with weaker or smaller bases, and higher temperatures.
**7.3**
- In the aldol condensation reaction, the enolate carbanion (the deprotonated aldehyde or ketone) acts as the nucleophile and the keto form of the aldehyde or ketone acts as the electrophile.
- A retro-aldol reaction is the reverse of an aldol reaction where instead a bond between the α-and β-carbons of a carbonyl is broken. This can be favored by the addition of base and heat. In this reaction, a bond between the α-and β-carbons of a carbonyl is broken.
- An aldol condensation is a condensation reaction, in which two molecules are joined to form a single molecule with the loss of a small molecule; a dehydration reaction, in which a molecule of water is lost; and a nucleophile–electrophile reaction, in which a nucleophile pushes an electron pair to form a bond with an electrophile.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D

One mole of aldehyde reacts with one mole of alcohol via a nucleophilic addition reaction to form a product called a hemiacetal. In a hemiacetal, an –OH group, an –OR group, a hydrogen atom, and an –R group are attached to the same carbon atom.
2. B

Tautomerization is the interconversion of two isomers in which a hydrogen and a double bond are moved. The keto and enol tautomers of aldehydes and ketones are common examples of tautomers seen on Test Day. Note that the equilibrium lies to the left because the keto form is more stable. Esterification, (A), is the formation of esters from carboxylic acids and alcohols. Elimination, (C), is a reaction in which a part of a reactant is removed and a new multiple bond is introduced. Dehydration, (D), is a reaction in which a molecule of water is eliminated.
3. D
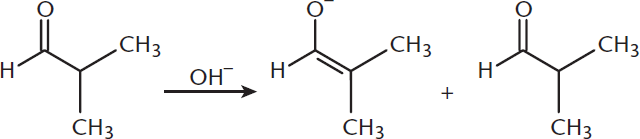
The reactions listed in the answer choices are examples of aldol condensations. In the presence of a base, the α-hydrogen is abstracted from an aldehyde, forming an enolate ion, [CH3CHCHO]–. This enolate ion then attacks the carbonyl group of the other aldehyde molecule, CH3CH2CHO, forming the pictured aldol.
4. A
The keto–enol equilibrium lies far to the keto side because the keto form is significantly more thermodynamically stable than the enol form. This thermodynamic stability stems from the fact that the oxygen is more electronegative than the carbon, and the keto tautomer puts more electron density around the oxygen than the enol tautomer. If the enol tautomer is less thermodynamically stable, it is also higher energy than the keto tautomer.
5. B
The aldol condensation is both a dehydration reaction because a molecule of water is lost, and a nucleophilic addition reaction because the nucleophilic enolate attacks and bonds to the carbonyl carbon.
6. B
This hydrogen is on the carbon between two carbonyls, which means that it is particularly acidic. This is due to both the inductive effects of the two oxygen atoms in the carbonyls and the resonance stabilization of the anion between the carbonyl groups.
7. B
At high temperatures and with a weak base like NH3, the thermodynamic enolate will be favored. The reaction proceeds slowly with the weak base, giving the kinetic enolate time to interconvert to the more stable thermodynamic enolate.
8. A
Aldehydes are generally more reactive than ketones because the additional alkyl group of a ketone is sterically hindering; this alkyl group is also electron-donating, destabilizing the carbanion intermediate. This eliminates (B) and (D). The carbonyl carbon is highly electrophilic; alkanes lack any significant electrophilicity, eliminating (C).
9. B
When α-carbons are deprotonated, the negative charge is resonance stabilized in part by the electronegative carbonyl oxygen, which is electron-withdrawing. Alkyl groups are actually electron-donating, which destabilizes carbanion intermediates; this invalidates statement II.
10. C
All of the answer choices are nitrogen-containing functional groups, but only enamines are tautomers of imines. Imines contain a double bond between a carbon and a nitrogen; enamines contain a double bond between two carbons as well as an amine.
11. C
When succinaldehyde (or any aldehyde or ketone with α-hydrogens) is treated with a strong base like lithium diisopropylamide (LDA), it forms the more nucleophilic enolate carbanion.
12. C
Aldol condensations contain two main steps. In the first step, the α-carbon of an aldehyde or ketone is deprotonated, generating the enolate carbanion. This carbanion can then attack another aldehyde or ketone, generating the aldol. In the second step, the aldol is dehydrated, forming a double bond. This double bond is between the α- and β-carbons, so the molecule is an α,β-unsaturated carbonyl.
13. D
Because benzaldehyde lacks an α-proton, it cannot be reacted with base to form the nucleophilic enolate carbanion. Therefore, acetone will act as our nucleophile, and both (A) and (B) can be eliminated. In order to perform this reaction, which is an aldol condensation, acetone will be reacted with a strong base—not a strong acid—in order to extract the α-hydrogen and form the enolate anion, which will act as a nucleophile.
14. B
This is an example of an aldol condensation, but stopped after aldol formation (before dehydration). After the aldol is formed using strong base, the reaction may be halted by the addition of acid. Butanal in strong acid, described in (C), would be likely to deprotonate without gaining the hydroxyl group. Methanol in diethyl ether would not be reactive because diethyl ether is not a strong enough base to abstract the α-hydrogen, eliminating (A). Reaction of the two aldehydes methanal and ethanal in catalytic base would form 3-hydroxypropanal (which would dehydrate to form propenal), not 3-hydroxybutanal.
15. B
The nomenclature in this question is well above what one needs to be able to draw on the MCAT; however, we can discern that we are forming a ketone and an aldehyde from a single molecule. The hallmark of a reverse aldol reaction is the breakage of a carbon–carbon bond, forming two aldehydes, two ketones, or one of each. In an aldol condensation, (A), we would expect to form a single product by combining two aldehydes, two ketones, or one of each. A dehydration reaction, (C), should release a water molecule, rather than breaking apart a large organic molecule into two smaller molecules. A nucleophilic attack, (D), should feature the formation of a bond between a nucleophile and an electrophile; again, we would not expect to break apart a large organic molecule into two smaller molecules. Note that simply noting how many reactants and products are present in the reaction is sufficient to determine the answer.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- General Chemistry Chapter 5
- Chemical Kinetics
- Organic Chemistry Chapter 4
- Analyzing Organic Reactions
- Organic Chemistry Chapter 6
- Aldehydes and Ketones I
- Organic Chemistry Chapter 8
- Carboxylic Acids
- Organic Chemistry Chapter 9
- Carboxylic Acid Derivatives
- Organic Chemistry Chapter 10
- Nitrogen- and Phosphorus-Containing Compounds