Chapter 8: Carboxylic Acids

Chapter 8: Carboxylic Acids
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of these compounds would be expected to decarboxylate when heated?
-
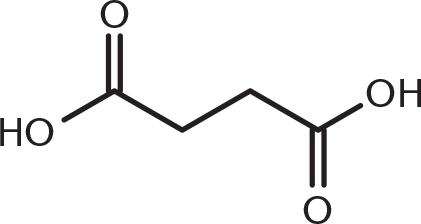
-
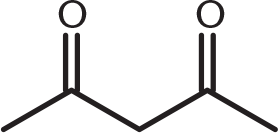
-
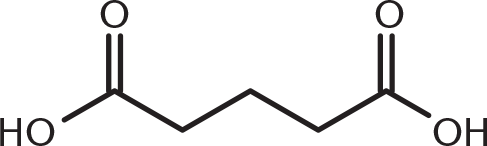
-
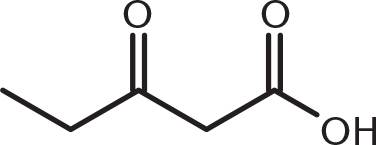
-
Carboxylic acids have higher boiling points than their corresponding alcohols primarily because:
- molecular weight is increased by the additional carboxyl group.
- the pH of the compound is lower.
- acid salts are soluble in water.
- hydrogen bonding is much stronger than in alcohols.
-
Which of the following carboxylic acids will be the most acidic?
- CH3CHClCH2COOH
- CH3CH2CCl2COOH
- CH3CH2CHClCOOH
- CH3CH2CH2COOH
-
Which of the following molecules could be classified as a soap?
- CH3(CH2)19COOH
- CH3COOH
- CH3(CH2)19COO–Na+
- CH3COO–Na+
-
What is the final product of the following reaction?
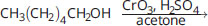
- CH3(CH2)4CHO
- CH3(CH2)4COOH
- CH3(CH2)4CH3
- HOOC(CH2)4COOH
-
Carboxylic acids can be reacted with one reagent (in addition to acid) to form all of the following compounds EXCEPT:
- esters.
- amides.
- alkenes.
- alcohols.
-
The reduction of a carboxylic acid using lithium aluminum hydride will yield what final product?
- An aldehyde
- An ester
- A ketone
- An alcohol
-
Which of the following is true with respect to a micelle in a hydrophilic environment?
- The interior is hydrophilic.
- The structure, as a whole, is hydrophobic.
- It is composed of short-chain fatty acids with polar heads.
- It can dissolve nonpolar molecules deep in its core.
-
In the presence of an acid catalyst, the major product of butanoic acid and 1-pentanol is:
- 1-butoxy-1-pentanol.
- butyl pentanoate.
- 1-pentoxy-1-butanol.
- pentyl butanoate.
-
The α-hydrogen of a carboxylic acid is:
- more acidic than the hydroxyl hydrogen.
- less acidic than the hydroxyl hydrogen.
- relatively acidic, as organic compounds go.
- I only
- II only
- I and III only
- II and III only
-
The reaction of formic acid with sodium borohydride will yield what final product?
- An aldehyde
- A carboxylic acid
- A ketone
- An alcohol
-
The intramolecular reaction of 5-aminopentanoic acid through nucleophilic acyl substitution would result in a(n):
- anhydride.
- lactone.
- lactam.
- carboxylic acid.
-
Butanoic anhydride can be produced by the reaction of butanoic acid with which of the following compounds?
- Butanoic acid
- Ethanoic acid
- Butanol
- Methanal
-
Nucleophilic acyl substitution is favored by:
- basic solution.
- acidic solution.
- leaving groups that are strong bases.
- I only
- II only
- I and II only
- I, II, and III
-
The reaction of ammonia with caprylic acid, found in coconuts, would produce a(n):
- ester.
- anhydride.
- alcohol.
- water molecule.
Answer Key
- D
- D
- B
- C
- B
- C
- D
- D
- D
- D
- B
- C
- A
- C
- D
Chapter 8: Carboxylic Acids
CHAPTER 8
CARBOXYLIC ACIDS
In This Chapter
8.1 Description and Properties
Nomenclature
Physical Properties
8.2 Reactions of Carboxylic Acids
Synthesis of Carboxylic Acids
Nucleophilic Acyl Substitution
Reduction
Decarboxylation
Saponification
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 9% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content category:
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
Carboxylic acids, with both carbonyl and hydroxyl groups, are some of the most reactive organic molecules you’ll encounter on Test Day. As we will see in this chapter, these molecules can react as acids (as their name suggests), nucleophiles, and electrophiles and are integral to many biological processes. Carboxylic acids are found in soaps, oils, preservatives, skin care products, clothing, and—most importantly for the MCAT—amino acids. Carboxylic acids often have strong, unpleasant odors. For example, acetic acid (ethanoic acid) is the main ingredient in vinegar; propionic acid (propanoic acid) gives Swiss cheese its smell; butyric acid (butanoic acid) is found in rancid butter and body odor.
So what makes carboxylic acids so interesting and versatile? First, they’re acids, so they like to give away protons—particularly because when they do so, the remaining negative charge resonates between two oxygen atoms, making the anion very stable. This makes carboxylic acids some of the most acidic compounds encountered in organic chemistry, with pKa values between 3 and 6. Compare this with alcohols, which have an average pKa around 17. Carboxylic acids are also excellent at hydrogen bonding, which results in large intermolecular forces and high boiling points. Finally, carboxylic acids are ubiquitous in nature and are synthesized by all living organisms.
8.1 Description and Properties
LEARNING OBJECTIVES
After Chapter 8.1, you will be able to:
- Explain the underlying cause of the relatively high acidity of carboxylic acids
- Predict the effects of additional substituents on the acidity of a carboxylic acid
- Rank the acidity of comparable carboxylic acids based on their structure
A carboxylic acid contains both a carbonyl group and a hydroxyl group, bonded to the same carbon. With three bonds to oxygen atoms, this is one of the most oxidized functional groups encountered in organic chemistry. Carboxylic acids are always terminal groups.
Nomenclature
In the IUPAC system of nomenclature, carboxylic acids are named by adding the suffix –oic acid to the parent root when the carboxylic acid is the highest-priority functional group. When this is true, the carbonyl carbon becomes carbon number 1. Figure 8.1 shows two examples.
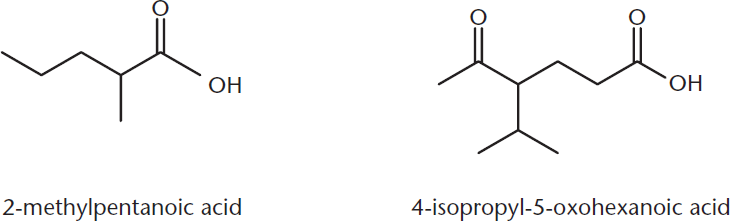
Figure 8.1. IUPAC Names of Carboxylic Acids
Like the other functional groups, many carboxylic acids are also named by their common names. Make note of the common prefixes used in the examples in Figure 8.2.
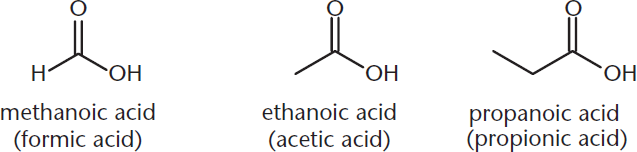
Figure 8.2. IUPAC and Common Names of Carboxylic Acids
MCAT EXPERTISE
The same common-name prefixes are used for both aldehydes and carboxylic acids: form– for one carbon, acet– for two, and propion– for three.
Cyclic carboxylic acids are named by listing the cycloalkane with the suffix carboxylic acid. Salts of carboxylic acids are named beginning with the cation, followed by the name of the acid with the ending –oate replacing –oic acid. Typical examples are shown in Figure 8.3.
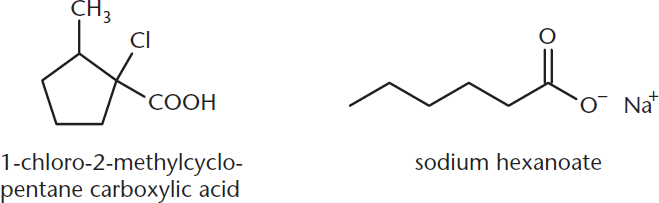
Figure 8.3. Cyclic Carboxylic Acid and Carboxylic Acid Salt
Finally, dicarboxylic acids, which have a carboxylic acid group on each end of the molecule, are common in biological systems. The smallest dicarboxylic acid is oxalic acid, with two carbons. The next five straight-chain dicarboxylic acids are malonic, succinic, glutaric, adipic, and pimelic acids. Their IUPAC names have the suffix –dioic acid: ethanedioic acid, propanedioic acid, butanedioic acid, pentanedioic acid, hexanedioic acid, and heptanedioic acid. Figure 8.4 shows several examples.
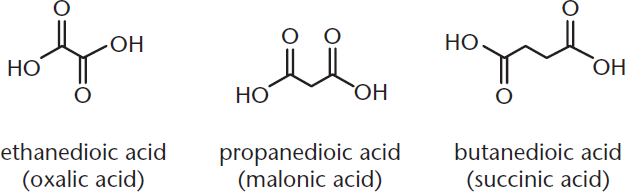
Figure 8.4. IUPAC and Common Names of Dicarboxylic Acids
Physical Properties
Many of the physical properties of carboxylic acids are similar to those of aldehydes and ketones because they both contain carbonyl groups. However, the additional hydroxyl group permits carboxylic acids to hydrogen bond and provides another acidic hydrogen that can participate in reactions.
Hydrogen Bonding
Carboxylic acids are polar because they contain a carbonyl group and can also form hydrogen bonds because they contain a hydrogen bonded to a very electronegative atom (in this case, the hydroxyl oxygen). Carboxylic acids display particularly strong intermolecular attractions because both the hydroxyl oxygen and carbonyl oxygen can participate in hydrogen bonding. As a result, carboxylic acids tend to form dimers: pairs of molecules connected by two hydrogen bonds. Multiple hydrogen bonds elevate the boiling and melting points of carboxylic acids past those of corresponding alcohols. Boiling points also increase with increasing molecular weight.
Acidity
The hydroxyl hydrogen of a carboxylic acid is quite acidic. This results in a negative charge that remains after the hydrogen is removed and resonance stabilization occurs between both of the electronegative oxygen atoms. Delocalization of the negative charge results in a very stable carboxylate anion, which is demonstrated in Figure 8.5.
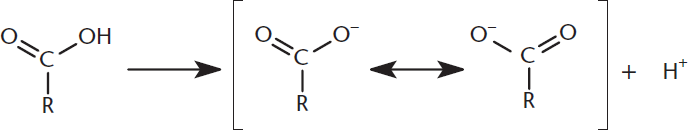
Figure 8.5. Carboxylate Anion Stability The negative charge from deprotonation is stabilized through resonance.
KEY CONCEPT
Carboxylic acids are polar and can form hydrogen bonds. Their acidity is due to resonance stabilization and can be enhanced by the addition of electronegative groups or a greater ability to delocalize charge.
The more stable the conjugate base is, the easier it is for the proton to leave, and thus, the stronger the acid. Carboxylic acids are relatively acidic, with pKa values on the order of 4.8 for ethanoic acid and 4.9 for propanoic acid. However, keep in mind that although these are quite acidic for organic compounds, they do not compare to strong acids like HCl (pKa = –8.0) or even HSO4− (pKa = 1.99). Remember, lower pKa values indicate stronger acids.
Substituents on carbon atoms near a carboxyl group influence anion stability and therefore affect acidity. Groups like –NO2 or halides are electron-withdrawing and increase acidity. In contrast, –NH2 or –OCH3 are electron-donating groups that destabilize the negative charge, decreasing the acidity of the compound. The closer the substituent groups are to the carboxyl group, the greater the effect will be.
In dicarboxylic acids, each –COOH group influences the other –COOH group. Carboxylic acids are electron-withdrawing due to the electronegative oxygen atoms they contain. The net result is that dicarboxylic acids are more acidic than the analogous monocarboxylic acids. However, when one proton is removed from the molecule, the carboxylate anion is formed, resulting in an immediate decrease in the acidity of the remaining carboxylic acid. This makes sense because if the second group were deprotonated, it would create a doubly charged species with two negative charges repelling each other. Due to this instability, the second proton is actually less acidic (harder to remove) than the analogous proton of a monocarboxylic acid.
β-dicarboxylic acids are dicarboxylic acids in which each carboxylic acid is positioned on the β-carbon of the other; in other words, there are two carboxylic acids separated by a single carbon. These compounds are notable for the high acidity of the α-hydrogens located on the carbon between the two carboxyl groups (pKa ≈ 9–14). Loss of this acidic hydrogen atom produces a carbanion, which is stabilized by the electron-withdrawing effect of both carboxyl groups, as shown in Figure 8.6.
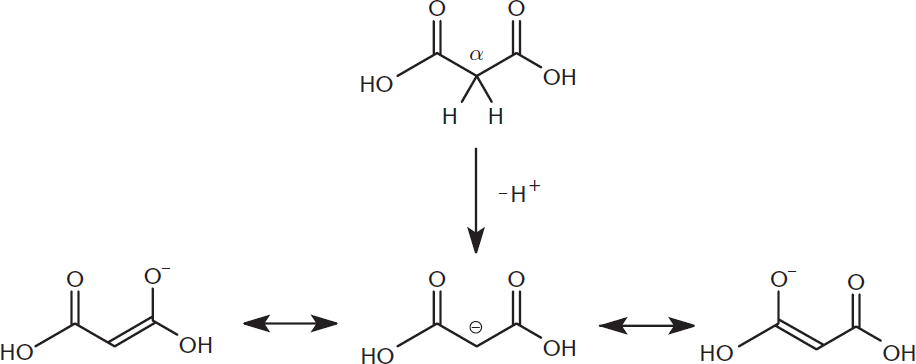
Figure 8.6. Acidity of the α-Hydrogen in β-Dicarboxylic Acids Note that the α-hydrogen is less acidic than the hydroxyl hydrogens; the hydroxyl groups are left protonated in this example for demonstration purposes only.
KEY CONCEPT
The hydroxyl hydrogen is the most acidic proton on a carboxylic acid. However, in 1,3-dicarbonyls, the α-hydrogen is also quite acidic.
Note that this also applies to the α-hydrogens in a β-diketone, β-ketoacids, β-dialdehydes, and other molecules that share the 1,3-dicarbonyl structure shown in Figure 8.7.
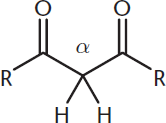
Figure 8.7. General Structure of 1,3-Dicarbonyl Compounds
MCAT CONCEPT CHECK 8.1:
Before you move on, assess your understanding of the material with these questions.
-
What causes the relatively high acidity of carboxylic acids?
________________________
-
Among a monocarboxylic acid, a dicarboxylic acid, and a dicarboxylic acid that has been deprotonated once, which will be the most acidic? Why?
________________________
-
What effects do additional substituents have on the acidity of carboxylic acids?
________________________
8.2 Reactions of Carboxylic Acids
LEARNING OBJECTIVES
After Chapter 8.2, you will be able to:
- Recall the reactant types used in acyl substitution reactions to form the major carboxylic acid derivatives, such as amides and esters
- Describe the mechanism of nucleophilic acyl substitution reactions
- Identify the conditions that would lead to spontaneous decarboxylation of a carboxylic acid
- Predict the products of an acyl substitution reaction:
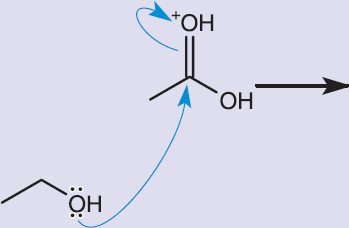
The properties of carboxylic acids make them highly reactive in a number of different categories. Several of the most important reactions are described here.
Synthesis of Carboxylic Acids
As described in earlier chapters, carboxylic acids can be prepared via oxidation of aldehydes and primary alcohols. The oxidant is often a dichromate salt (Na2Cr2O7 or K2Cr2O7), chromium trioxide (CrO3), or potassium permanganate (KMnO4), as shown in Figure 8.8, but several other oxidizing agents can also work. Remember that secondary and tertiary alcohols cannot be oxidized to carboxylic acids because they already have at least two bonds to other carbons.
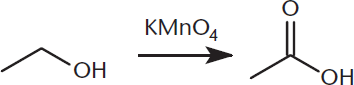
Figure 8.8. Synthesis of a Carboxylic Acid via Oxidation of a Primary Alcohol
There are many other methods of generating carboxylic acids, including organometallic reagents (Grignard reagents) and hydrolysis of nitriles (–C≡N), but these are outside the scope of the MCAT.
Nucleophilic Acyl Substitution
Many of the reactions in which carboxylic acids (and their derivatives) participate proceed via a single mechanism: nucleophilic acyl substitution. This mechanism is similar to nucleophilic addition to an aldehyde or ketone, which was discussed in Chapters 6 and 7 of MCAT Organic Chemistry Review. The key difference, however, focuses on the existence of a leaving group in carboxylic acids and their derivatives. In this case, after opening the carbonyl via nucleophilic attack and forming a tetrahedral intermediate, the carbonyl can reform, thereby kicking off the leaving group. This reaction is shown in Figure 8.9.
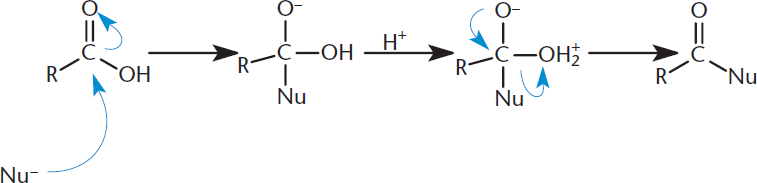
Figure 8.9. Nucleophilic Acyl Substitution Step 1: Nucleophilic addition; Step 2: Elimination of the leaving group and reformation of the carbonyl.
In these reactions, the nucleophilic molecule replaces the leaving group of an acyl derivative. Acyl derivatives encompass all molecules with a carboxylic acid-derived carbonyl, including carboxylic acids, amides, esters, anhydrides, and others. These reactions are favored by a good leaving group. Remember, weak bases, which are often the conjugate bases of strong acids, make good leaving groups. These reactions are also favored in either acidic or basic conditions, which can alter the reactivity of the electrophile and nucleophile.
MCAT EXPERTISE
While you may have learned about other acyl derivatives in your organic chemistry classes, such as acyl halides and nitriles, the official content list for the MCAT restricts its focus to carboxylic acids, amides, esters, and anhydrides.
Amides
Carboxylic acids can be converted into amides if the incoming nucleophile is ammonia (NH3) or an amine, as shown in Figure 8.10. This can be carried out in either an acidic or basic solution to drive the reaction forward.
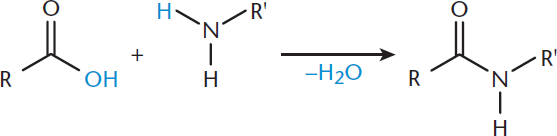
Figure 8.10. Formation of an Amide by Nucleophilic Acyl Substitution
Amides are named by replacing the –oic acid suffix with –amide in the name of the parent carboxylic acid. Any alkyl groups on the nitrogen are placed at the beginning of the name with the prefix N–. Amides exist in a resonance state where delocalization of electrons occurs between the oxygen and nitrogen atoms, as shown in Figure 8.11.
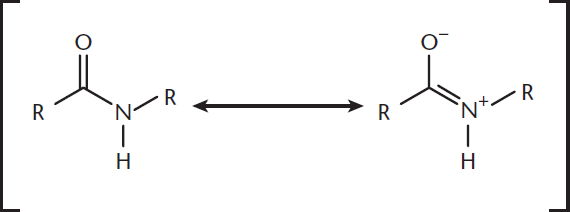
Figure 8.11. Resonance of Amides Resonance between the carbonyl and lone pair on the nitrogen stabilizes this bond and restricts its motion.
Amides that are cyclic are called lactams and are named by replacing –oic acid with –lactam. They may also be named by indicating the specific carbon that is bonded during cyclization of the compound. Several examples are shown in Figure 8.12.
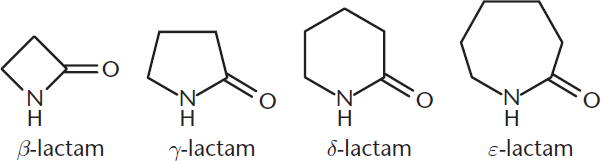
Figure 8.12. Examples of Lactams
Esters
Esters are a hybrid between a carboxylic acid and an ether (ROR′ ), which can be made by reacting carboxylic acids with alcohols under acidic conditions, as shown in Figure 8.13. Esterification is a condensation reaction with water as a side product. In acidic solutions, the carbonyl oxygen can be protonated, which enhances the polarity of the bond, thereby placing additional positive charge on the carbonyl carbon and increasing its susceptibility to nucleophilic attack. This condensation reaction occurs most rapidly with primary alcohols.
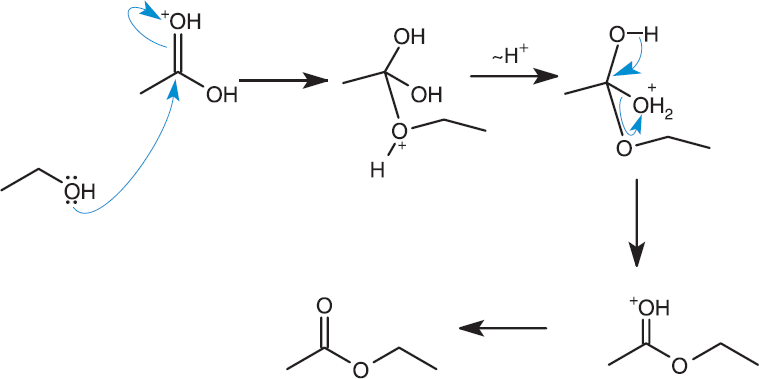
Figure 8.13. Esterification: Reaction of a Carboxylic Acid with an Alcohol
KEY CONCEPT
Protonating the C=O makes the electrophilic carbon even more ripe for nucleophilic attack.
Esters are named in the same manner as salts of carboxylic acids. For example, the ester shown in the reaction in Figure 8.13 has the common name ethyl acetate, or the IUPAC name ethyl ethanoate.
Esters that are cyclic are called lactones and are named by replacing –oic acid with –lactone. Several examples are shown in Figure 8.14.
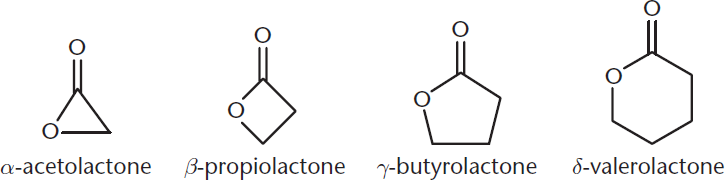
Figure 8.14. Examples of Lactones
Anhydrides
Anhydrides can be formed by the condensation of two carboxylic acids. They are named by replacing the acid at the end of the name of the parent carboxylic acid with anhydride, whether cyclic or linear. One example is the condensation of two molecules of ethanoic acid to form ethanoic anhydride, as shown in Figure 8.15. Just like the above reactions, anhydride formation occurs via nucleophilic acyl substitution.
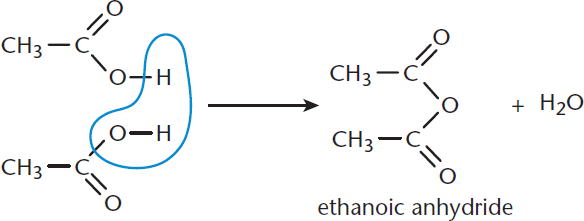
Figure 8.15. Synthesis of an Anhydride via Carboxylic Acid Condensation
Reduction
Carboxylic acids can be reduced to primary alcohols by the use of lithium aluminum hydride (LiAlH4). Aldehyde intermediates may be formed in the course of this reaction, but they, too, will be reduced to the alcohol. The reaction occurs by nucleophilic addition of hydride to the carbonyl group. The reaction mechanism is shown in Figure 8.16.
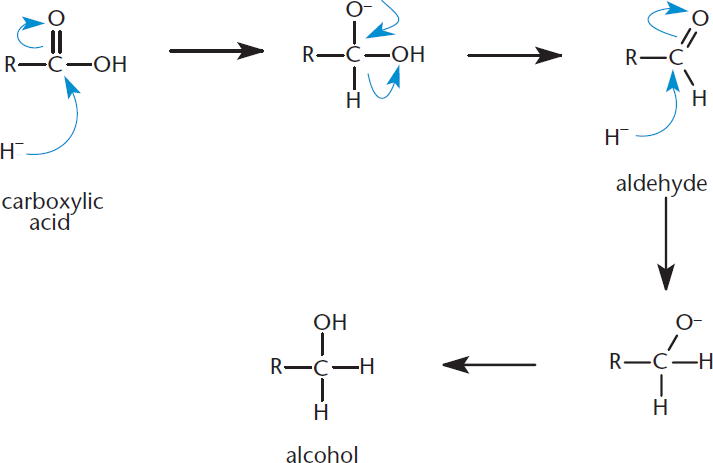
Figure 8.16. Reduction of a Carboxylic Acid to a Primary Alcohol Reaction occurs by nucleophilic addition of hydride and proceeds through an aldehyde intermediate.
Lithium aluminum hydride is a strong reducing agent that can successfully reduce a carboxylic acid; a gentler reducing agent like sodium borohydride (NaBH4) is not strong enough to reduce carboxylic acids.
KEY CONCEPT
Carboxylic acids can be reduced by LiAlH4, but not the less reactive NaBH4.
Decarboxylation
Decarboxylation describes the complete loss of the carboxyl group as carbon dioxide. This is a common way of getting rid of a carbon from the parent chain. 1,3-Dicarboxylic acids and other β-keto acids may spontaneously decarboxylate when heated. Under these conditions, the carboxyl group is lost and replaced with hydrogen. Because both the electrophile and nucleophile are in the same molecule, the reaction proceeds through a six-membered ring in its transition state, as shown in Figure 8.17. The enol that is initially formed from the destruction of the ring tautomerizes to the more stable keto form.
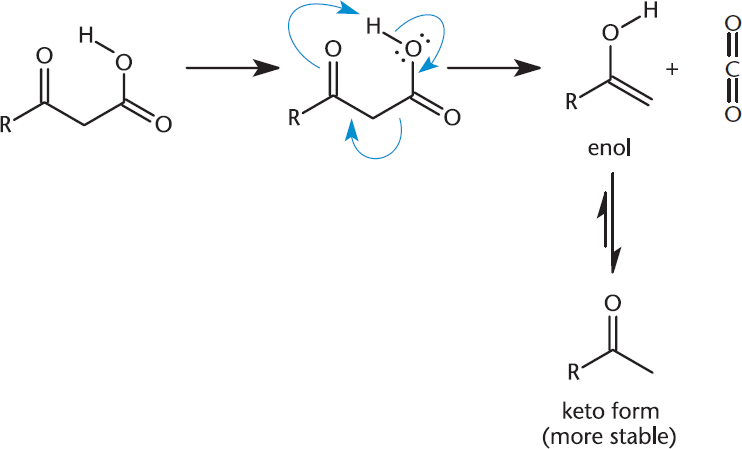
Figure 8.17. Decarboxylation of Carboxylic Acids: Loss of CO2 The intramolecular reaction proceeds via a six-membered ring transition state, and the product tautomerizes from the enol to the more stable keto form.
BRIDGE
Decarboxylation is common in biochemical pathways in the body. *Pyruvate** dehydrogenase complex, described in Chapter 10 ofMCAT Biochemistry Review*, carries out the decarboxylation of pyruvate to help form acetyl-CoA, which can feed into the citric acid cycle.
Saponification
When long-chain carboxylic acids react with sodium or potassium hydroxide, a salt is formed. This process, called saponification, occurs by mixing fatty acids with lye (sodium or potassium hydroxide), resulting in the formation of a salt that we know as soap. Soaps can solvate nonpolar organic compounds in aqueous solutions because they contain both a nonpolar tail and a polar carboxylate head, as shown in Figure 8.18.
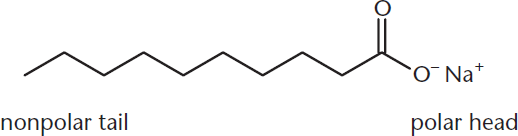
Figure 8.18. Carboxylic Acid Salt (Soap)
When placed in aqueous solution, soap molecules arrange themselves into spherical structures called micelles, as shown in Figure 8.19. The polar heads face outward, where they can be solvated by water, and the nonpolar hydrocarbon chains are oriented toward the inside of the sphere, protected from the solvent. Nonpolar molecules, such as grease, dissolve in the hydrocarbon interior of the spherical micelle; the micelle as a whole then dissolves in water due to the polarity of its exterior surface.

Figure 8.19. Soap Micelle The polar heads interact with the hydrophilic environment; the nonpolar tails are oriented toward the interior of the micelle.
BRIDGE
The formation of the phospholipid bilayer, micelles, and liposomes are all contingent on the bipolar nature of carboxylic acids with long hydrocarbon chains. These structures are discussed in Chapter 5 of MCAT Biochemistry Review.
MCAT CONCEPT CHECK 8.2:
Before you move on, assess your understanding of the material with these questions.
-
For each of the derivatives below, list the nucleophile used to form the derivative in an acyl substitution reaction and the name of the cyclic form of that functional group.
Carboxylic Acid Derivative Formed by Reaction with: Name of Cyclic Form: Amide Ester Anhydride
-
Briefly describe the mechanism of nucleophilic acyl substitution reactions.
________________________
-
What is the result when butanoic acid is reacted with sodium borohydride? With lithium aluminum hydride?
- Sodium borohydride: ______________________________
- Lithium aluminum hydride: ______________________________
-
Under what conditions will a carboxylic acid spontaneously decarboxylate?
________________________
Conclusion
Acids are an important concept on the MCAT: they can be tested in general chemistry, organic chemistry, and biochemistry. The underlying concept in all three subjects is the same: the more stable the conjugate base is, the more likely it is that the proton will leave. This stability is determined by three factors: periodic trends (electronegativity and, thus, induction), size of the anion, and resonance. Understanding these effects is a major key to success on Test Day. The reactions of carboxylic acids, in particular, are dictated by the polarity of the carbonyl group in conjunction with the ability of the hydroxyl group to act as a leaving group. This allows a diversity of reactions through nucleophilic acyl substitution, reduction by lithium aluminum hydride, decarboxylation, and saponification.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Description and Properties
- Carboxylic acids contain a carbonyl and a hydroxyl group connected to the same carbon. They are always terminal groups.
- Carboxylic acids are indicated with the suffix –oic acid. Salts are named with the suffix –oate, and dicarboxylic acids are –dioic acids.
- Physical Properties
- Carboxylic acids are polar and hydrogen bond very well, resulting in high boiling points. They often exist as dimers in solution.
- The acidity of a carboxylic acid is enhanced by the resonance between its oxygen atoms.
- Acidity can be further enhanced by substituents that are electron-withdrawing, and decreased by substituents that are electron-donating.
- β-dicarboxylic acids, like other 1,3-dicarbonyl compounds, have an α-hydrogen that is also highly acidic.
Reactions of Carboxylic Acids
- Carboxylic acids can be made by the oxidation of primary alcohols or aldehydes using an oxidizing agent like potassium permanganate (KMnO4), dichromate salts (Na2Cr2O7 or K2Cr2O7), or chromium trioxide (CrO3).
- Nucleophilic acyl substitution is a common reaction in carboxylic acids.
- A nucleophile attacks the electrophilic carbonyl carbon, opening the carbonyl and forming a tetrahedral intermediate.
- The carbonyl reforms, kicking off the leaving group.
- If the nucleophile is ammonia or an amine, an amide is formed. Amides are given the suffix –amide. Cyclic amides are called lactams.
- If the nucleophile is an alcohol, an ester is formed. Esters are given the suffix –oate. Cyclic esters are called lactones.
- If the nucleophile is another carboxylic acid, an anhydride is formed. Both linear and cyclic anhydrides are given the suffix anhydride.
- Carboxylic acids can be reduced to a primary alcohol with a strong reducing agent like lithium aluminum hydride (LiAlH4).
- Aldehyde intermediates are formed, but are also reduced to primary alcohols.
- Sodium borohydride (NaBH4) is a common reducing agent for other organic reactions, but is not strong enough to reduce a carboxylic acid.
- β-dicarboxylic acids and other β-keto acids can undergo spontaneous decarboxylation when heated, losing a carbon as carbon dioxide. This reaction proceeds via a six-membered cyclic intermediate.
- Mixing long-chain carboxylic acids (fatty acids) with a strong base results in the formation of a salt we call soap. This process is called saponification.
- Soaps contain hydrophilic carboxylate heads and hydrophobic alkyl chain tails.
- Soaps organize in hydrophilic environments to form micelles. A micelle dissolves nonpolar organic molecules in its interior, and can be solvated with water due to its exterior shell of hydrophilic groups.
ANSWERS TO CONCEPT CHECKS
**8.1**
- Carboxylic acids are particularly acidic due to the electron-withdrawing oxygen atoms in the functional group and the high stability of the carboxylate anion, which is resonance stabilized by delocalization with two electronegative oxygen atoms.
- A dicarboxylic acid would be the most acidic, as the second carboxyl group is electron-withdrawing and therefore contributes to even higher stability of the anion after loss of the first hydrogen. However, a monocarboxylic acid is more acidic than a deprotonated dicarboxylic acid because the carboxylate anion is electron-donating and destabilizes the product of the second deprotonation step, resulting in decreased acidity.
- Electron-withdrawing substituents make the anion more stable and therefore increase acidity. Electron-donating substituents, on the other hand, destabilize the anion, causing the carboxylic acid to be less acidic. The closer the substituent is to the carboxylic acid on the molecule, the stronger the effect will be.
**8.2**
-
Carboxylic Acid Derivative Formed by Reaction with: Name of Cyclic Form: Amide Ammonia (NH3) or an amine Lactam
Ester Alcohol Lactone
Anhydride Another carboxylic acid Anhydride
- Nucleophilic acyl substitution is the substitution of an attacking nucleophile for the leaving group of an acyl compound, which includes carboxylic acids, amides, esters, and anhydrides. The nucleophile attacks, opening the carbonyl and forming a tetrahedral intermediate. The carbonyl then reforms, kicking off the leaving group. This reaction is favored by acidic or basic conditions.
- Sodium borohydride is not strong enough to reduce carboxylic acids so there will be no reaction. Lithium aluminum hydride, however, is strong enough to reduce carboxylic acids to primary alcohols—producing 1-butanol.
- 1,3-Dicarboxylic acids will spontaneously decarboxylate when heated, due to the stable cyclic intermediate step.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
This compound is a β-keto acid: a carbonyl functional group at the β-position from a carboxyl group. Decarboxylation occurs with β-keto acids and β-dicarboxylic acids because they can form a cyclic transition state that permits simultaneous hydrogen transfer and loss of carbon dioxide. (B) is a diketone and does not have a single carboxyl group. (A) and (C) are γ- and δ-dicarboxylic acids, respectively, and can decarboxylate but with more difficulty.
2. D
The boiling points of compounds depend on the strength of the attractive forces between molecules. In both alcohols and carboxylic acids, the major form of intermolecular attraction is hydrogen bonding. However, hydrogen bonding is much stronger in carboxylic acids as compared to alcohols because carboxylic acids are more polar, and the carbonyl also contributes to hydrogen bonding in addition to the hydroxyl group. The stronger hydrogen bonds elevate the boiling points of carboxylic acids compared to alcohols. Boiling points also depend on molecular weight, (A), but in this case, the difference in molecular weight is insignificant compared to the effect of hydrogen bonding. (B) and (C) are both true but do not explain the difference in boiling points.
3. B
The acidity of carboxylic acids is significantly increased by the presence of highly electronegative functional groups. Their electron-withdrawing effect increases the stability of the carboxylate anion, favoring proton dissociation. This effect increases as the number of electronegative groups on the chain increases, and it also increases as the distance between the acid functionality and electronegative group decreases. This answer has two halogens bonded to it at a smaller distance from the carboxyl group compared to the other answers.
4. C
Soap is a salt of a carboxylate anion with a long hydrocarbon tail. (A) and (B) are not salts of anionic compounds. (D) is sodium acetate, which is a salt but does not contain the long hydrocarbon tail needed to be considered a soap.
5. B
Jones reagent (chromium trioxide in aqueous sulfuric acid) is an oxidizing agent. As such, it oxidizes primary alcohols directly to carboxylic acids. This reagent is too strong an oxidant to give an aldehyde, so (A) is incorrect; remember that pyridinium chlorochromate (PCC) is a common oxidizing agent used to convert alcohols to aldehydes without progressing to a carboxylic acid. (D), a dicarboxylic acid, cannot form because there is no functional group on the other end of the molecule for the reagent to attack, and it cannot attack an inert alkane. (C) represents reduction, not oxidation.
6. C
Carboxylic acids cannot be converted into alkenes with just one reagent. Esters, (A), are formed in nucleophilic acyl substitution reactions with alcohols. Amides, (B), are formed by nucleophilic acyl substitution reactions with ammonia. Alcohols, (D), may be formed using a variety of reducing agents, most notably lithium aluminum hydride. To form alkenes, carboxylic acids may be reduced to alcohols, which can then be transformed into alkenes by elimination in a second step with addition of another reagent.
7. D
Lithium aluminum hydride (LiAlH4 or LAH) is a strong reducing agent. LAH can completely reduce carboxylic acids to primary alcohols. Aldehydes are intermediate products of this reaction; therefore, (A) is incorrect. The other compounds are not created through the reduction of a carboxylic acid.
8. D
Micelles are self-assembled aggregates of soap in which the interior is composed of long hydrocarbon (fatty) tails, which can dissolve nonpolar molecules. The outer surface is covered with carboxylate groups, which makes the overall structure water-soluble. Soaps, in general, are salts of long-chain hydrocarbons with carboxylate head groups.
9. D
The reaction described is esterification, in which the nucleophilic oxygen atom of 1-pentanol attacks the electrophilic carbonyl carbon of butanoic acid, ultimately displacing water to form pentyl butanoate. The acid catalyst is regenerated from 1-pentanol's released proton. (A) reverses the carbon chains, considering the butyl tail to be the esterifying group. Ethers do not form under these conditions, so (B) and (C) are also incorrect.
10. D
The α-hydrogen of a carboxylic acid is relatively acidic as far as organic compounds go, due to resonance stabilization. However, the hydroxyl hydrogen is significantly more acidic because it is able to share the negative charge resulting from deprotonation between both electronegative oxygen atoms in the functional group.
11. B
The reaction of formic acid, which is a simple carboxylic acid, with sodium borohydride, which is a mild reducing agent, will result in no reaction, and therefore will result in maintenance of the carboxylic acid. Sodium borohydride is too mild to reduce carboxylic acids, and therefore cannot produce the primary alcohols that lithium aluminum hydride, a strong reducing agent, would.
12. C
5-aminopentanoic acid contains a carboxylic acid and an amine. If this molecule undergoes intramolecular nucleophilic acyl substitution, it will form a cyclic amide. These molecules are called lactams. Lactones, (B), are cyclic esters, not amides.
13. A
Butanoic anhydride is an anhydride with two butane R groups. Anhydrides are produced by the reaction of two carboxylic acids with the loss of a water molecule. Therefore, butanoic anhydride would be produced by the reaction of two molecules of butanoic acid.
14. C
Nucleophilic acyl substitutions are favored in basic solution, which makes the nucleophile more nucleophilic; in acidic solution, which makes the electrophile more electrophilic; and by good leaving groups. However, strong bases do not make good leaving groups; weak bases do.
15. D
Based on its name, caprylic acid must be a carboxylic acid. The reaction between a carboxylic acid and ammonia (NH3) would produce an amide—which is not one of the options listed. Instead, we should take a look at the type of reaction occurring. The production of an amide from a carboxylic acid and ammonia occurs through a condensation reaction in which a molecule of water is removed as a leaving group.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 1
- Amino Acids, Peptides, and Proteins
- Biochemistry Chapter 5
- Lipid Structure and Function
- Organic Chemistry Chapter 1
- Nomenclature
- Organic Chemistry Chapter 4
- Analyzing Organic Reactions
- Organic Chemistry Chapter 6
- Aldehydes and Ketones I
- Organic Chemistry Chapter 9
- Carboxylic Acid Derivatives