Chapter 9: Carboxylic Acid Derivatives

Chapter 9: Carboxylic Acid Derivatives
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of the following would be the best method of producing methyl propanoate?
- Reacting propanoic acid and methanol in the presence of a mineral acid
- Reacting methanoic acid and propanol in the presence of a mineral acid
- Reacting propanoic anhydride with an aqueous base
- Reacting propanoic acid with an aqueous base
-
What would be the product(s) of the reaction below?
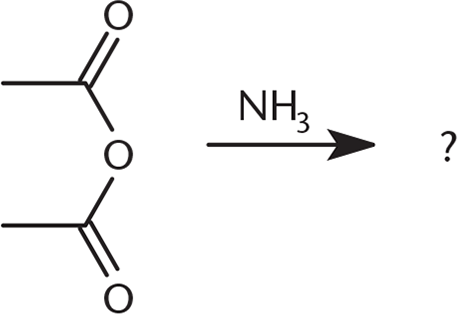
-
-
-
-
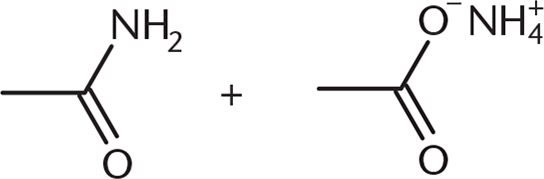
-
Which of the following undergoes a Fischer esterification most rapidly?
-
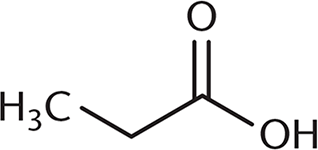
-
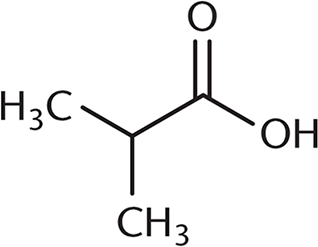
-
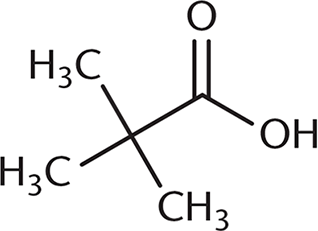
-
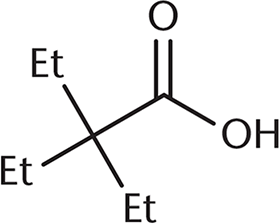
-
Each of the acyl compounds listed below contains a six-membered ring EXCEPT:
- δ-lactam.
- cyclohexane carboxylic acid.
- γ-butyrolactone.
- the anhydride formed from intramolecular ring closure of pentanedioic acid.
-
Which of the following would be most reactive toward nucleophiles?
- Propyl ethanoate
- Propanoic acid
- Propanamide
- Propanoic anhydride
-
How might succinic anhydride, shown below, be formed from succinic acid (butanedioic acid)?
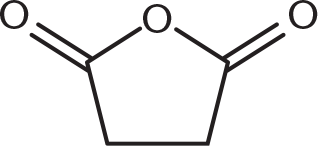
- Catalytic acid
- Catalytic base
- Heat
- Oxidation
-
Which of the following would react most readily with a carboxylic acid to form an amide?
- Methylamine
- Triethylamine
- Diphenylamine
- Ethylmethylamine
-
If propanamide were treated with water, what product(s) would be observed?
- Propanamide
- Propanoic acid
- Equal concentrations of propanamide and propanoic acid
- Propyl propanoate
-
β-lactams are:
- cyclic forms of the least reactive type of carboxylic acid derivative.
- more reactive than their straight-chain counterparts.
- molecules with high levels of ring strain.
- I only
- II only
- II and III only
- I, II, and III
-
The acid-catalyzed conversion of propyl ethanoate to benzyl ethanoate is likely:
- reduction.
- hydrolysis.
- transesterification.
- oxidation.
-
The reaction shown, which is important for the breakdown of polypeptides, would be favored under what conditions?
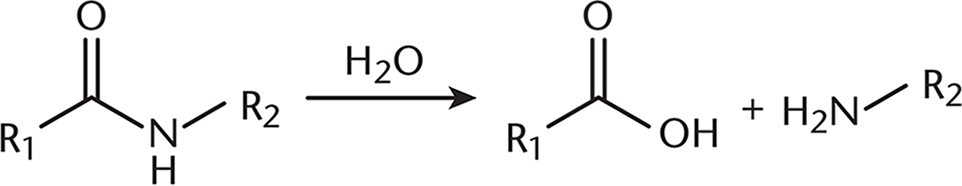
- Mild heat
- Acid environment
- Anhydrous environment
- Nonpolar solvent
-
A positive charge on the molecule shown would have greater stability than a positive charge on a straight-chain alkane version of the same molecule. What property most explains this effect?
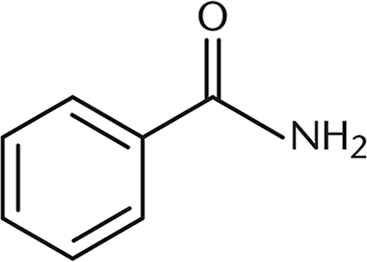
- Steric hindrance
- Nitrogen electronegativity
- Induction
- Conjugation
-
The molecule shown is:
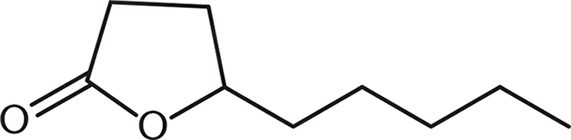
- synthesizable from a γ-hydroxycarboxylic acid.
- a lactone.
- a form of an ester.
- I only
- I and II only
- II and III only
- I, II, and III
-
Which reactant could be combined with butanol to form butyl acetate?
- (CH3CO)2O and catalytic acid
- (CH3CH2CO)2O and catalytic acid
- CH3CH2CONH2 and catalytic acid
- CH3CONH2 and catalytic acid
-
Why should esterification reactions NOT be carried out in water?
- Carboxylic acids, from which esters are made, are generally insoluble in water.
- The polar nature of water overshadows the polar nature of the leaving group.
- The extensive hydrogen bonding of water interferes with the nucleophilic addition mechanism.
- Water molecules would hydrolyze the desired products back into the parent carboxylic acid.
Answer Key
- A
- D
- A
- C
- D
- C
- A
- A
- D
- C
- B
- D
- D
- A
- D
Chapter 9: Carboxylic Acid Derivatives
CHAPTER 9
CARBOXYLIC ACID DERIVATIVES
In This Chapter
9.1 Amides, Esters, and Anhydrides
Descriptions
9.2 Reactivity Principles
Relative Reactivity of Derivatives
Steric Effects
Electronic Effects
Strain in Cyclic Derivatives
9.3 Nucleophilic Acyl Substitution Reactions
Anhydride Cleavage
Transesterification
Hydrolysis of Amides
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 11% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1A: Structure and function of proteins and their constituent amino acids
5D: Structure, function, and reactivity of biologically-relevant molecules
Introduction
We saw in the previous three chapters that carbonyls are susceptible to attack by everything from water to amines to other carbonyl-containing compounds (in the enol or enolate form). These reactions often result in the formation of carboxylic acid derivatives. Our focus in this chapter will be on describing the carboxylic acid derivatives that appear on the MCAT: amides, esters, and anhydrides. Each of these molecules replaces the –OH on the carboxyl group with another leaving group (–NR2, –OR, and –OCOR, respectively). These each react in similar ways to carboxylic acids. Many of these functional groups are also critical for biochemical processes.
9.1 Amides, Esters, and Anhydrides
LEARNING OBJECTIVES
After Chapter 9.1, you will be able to:
- Apply the rules for naming carboxylic acid derivatives, including the nomenclature for the cyclic version of the molecule
- Describe a condensation reaction
Amides, esters, and anhydrides are all carboxylic acid derivatives. Each of these is formed by a condensation reaction with a carboxylic acid—a reaction that combines two molecules into one, while losing a small molecule. In this case, the small molecule is water, which is created from the hydroxyl group of the carboxylic acid and a hydrogen associated with the incoming nucleophile.
KEY CONCEPT
In a condensation reaction, two molecules are combined to form one, with the loss of a small molecule—water, in our case. Carboxylic acid derivatives are formed by this mechanism.
Descriptions
For each of the carboxylic acid derivatives described in this section, focus on the relevant nucleophile that forms the derivative and the nomenclature of the functional group. In the next section, we’ll focus more directly on the relative reactivity of these compounds.
Amides
Amides are compounds with the general formula RCONR2. They are named by replacing the –oic acid suffix with –amide. Alkyl substituents on the nitrogen atom are listed as prefixes, and their location is specified with the letter N–. Figure 9.1 shows a few examples.
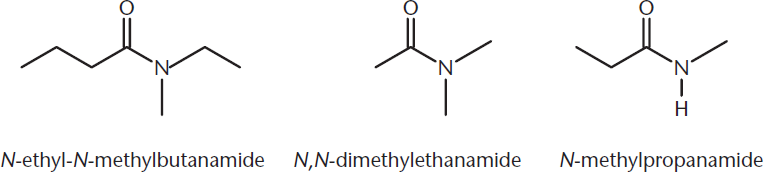
Figure 9.1. Naming Amides
Amides are generally synthesized by the reaction of other carboxylic acid derivatives with either ammonia or an amine. Note that loss of hydrogen from the nucleophile is required for this reaction to take place. Thus, only primary and secondary amines will undergo this reaction.
KEY CONCEPT
Amides are formed by the condensation reaction of other carboxylic acid derivatives and ammonia or an amine.
Cyclic amides are called lactams. These are named according to the carbon atom bonded to the nitrogen: β-lactams contain a bond between the β-carbon and the nitrogen, γ-lactams contain a bond between the γ-carbon and the nitrogen, and so forth. Structures of lactams are shown in Figure 9.2.
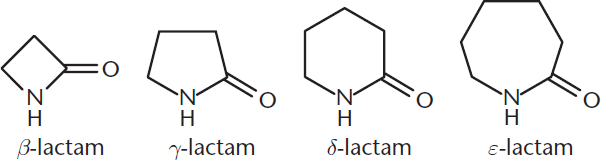
Figure 9.2. Examples of Lactams
Amides may or may not participate in hydrogen bonding depending on the number of alkyl groups they have bonded, and therefore their boiling points may be lower or on the same level as the boiling points of carboxylic acids.
Esters
Esters are the dehydration synthesis products of other carboxylic acid derivatives and alcohols. They are named by placing the esterifying group (the substituent bonded to the oxygen) as a prefix; the suffix –oate replaces –oic acid. Two examples are shown in Figure 9.3. As mentioned in the last chapter, ethyl acetate, derived from the condensation of acetic acid and ethanol, is called ethyl ethanoate according to IUPAC nomenclature.
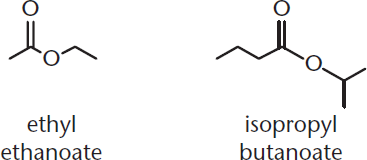
Figure 9.3. Naming Esters
KEY CONCEPT
Esters are formed by the condensation reaction of carboxylic acids or anhydrides with alcohols.
Under acidic conditions, mixtures of carboxylic acids and alcohols will condense into esters. This reaction, called a Fischer esterification, is shown in Figure 9.4. Esters can also be obtained from the reaction of anhydrides with alcohols.
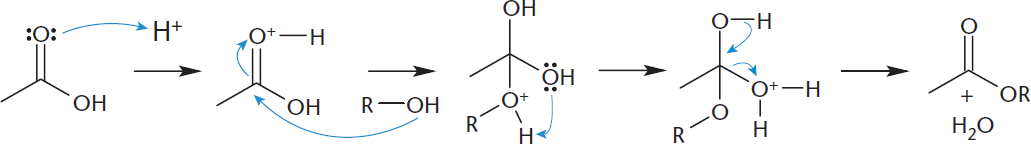
Figure 9.4. Fischer Esterification
Cyclic esters are called lactones and are named in the same manner as lactams, with the name of the precursor acid molecule also included. Examples are shown in Figure 9.5.
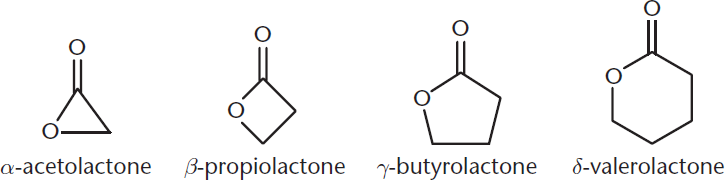
Figure 9.5. Examples of Lactones
Because they lack hydrogen bonding, esters usually have lower boiling points than their related carboxylic acids.
Triacylglycerols, the storage form of fats in the body, are esters of long-chain carboxylic acids (fatty acids) and glycerol (1,2,3-propanetriol). Saponification is the process by which fats are hydrolyzed under basic conditions to produce soap; saponification of a triacylglycerol is shown in Figure 9.6. Subsequent acidification of the soap regenerates the fatty acids.
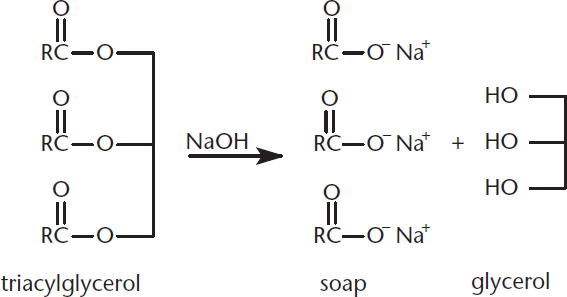
Figure 9.6. Saponification of a Triacylglycerol Treating triacylglycerols with NaOH will produce fatty acid salts (soap) as well as glycerol.
Anhydrides
Anhydrides, also called acid anhydrides, are the condensation dimers of carboxylic acids. These molecules have the general formula RC(O)OC(O)R. Symmetrical anhydrides are named by substituting the word anhydride for the word acid in a carboxylic acid. When anhydrides are asymmetrical, simply name the two chains alphabetically, followed by anhydride, as shown in Figure 9.7. Phthalic and succinic anhydrides are cyclic anhydrides arising from intramolecular condensation or dehydration of diacids; their structures need not be memorized, but recognize them as cyclic anhydrides.
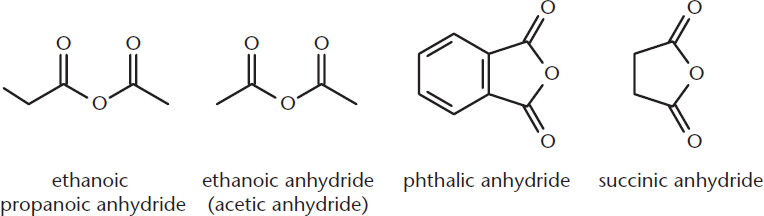
Figure 9.7. Naming Anhydrides
KEY CONCEPT
Anhydrides are usually formed by the condensation reaction of two carboxylic acids.
Acid anhydrides are synthesized, as mentioned previously, by a condensation reaction between two carboxylic acids, with one molecule of water lost in the condensation, as shown in Figure 9.8.
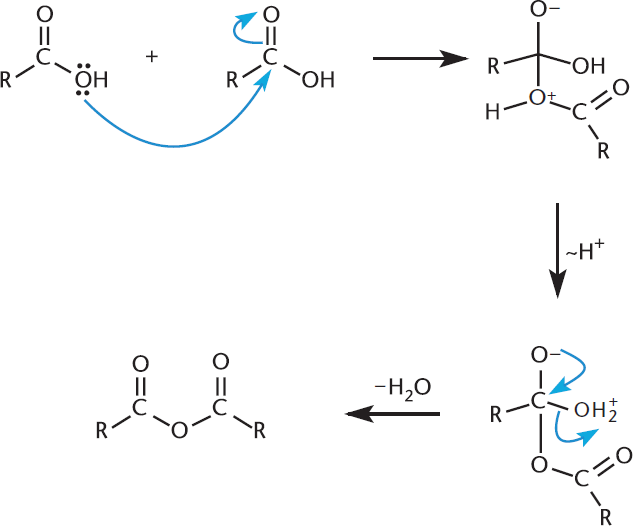
Figure 9.8. Synthesis of an Anhydride via Carboxylic Acid Condensation Two molecules of carboxylic acid come together and lose a molecule of water in the formation of an anhydride.
Certain cyclic anhydrides can be formed simply by heating carboxylic acids, as shown in Figure 9.9. The reaction is driven forward by the increased stability of the newly formed ring; as such, only anhydrides with five- or six-membered rings are easily made. Just as with all anhydride formations, the hydroxyl group of one –COOH acts as the nucleophile, attacking the carbonyl on the other –COOH.
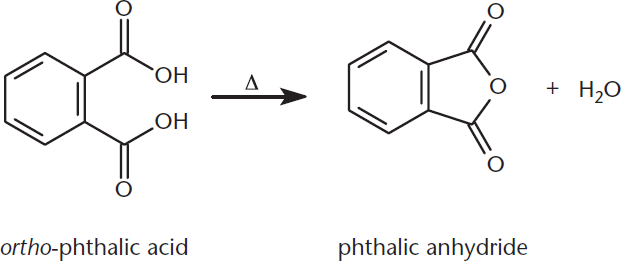
Figure 9.9. Intramolecular Anhydride Formation Heat and the increased stability of the newly formed ring drive this intramolecular ring formation reaction forward.
Anhydrides often have higher boiling points than their related carboxylic acids, based solely on their much greater weight.
MCAT CONCEPT CHECK 9.1:
Before you move on, assess your understanding of the material with these questions.
-
For each of the carboxylic acid derivatives below, list the relevant nucleophile that reacts with a carboxylic acid to generate the derivative, the derivative’s suffix, and the name of the derivative in cyclic form.
Carboxylic Acid Derivative Formed from –COOH by… Suffix Cyclic Naming Amide Ester Anhydride
-
What is the definition of a condensation reaction?
________________________
9.2 Reactivity Principles
LEARNING OBJECTIVES
After Chapter 9.2, you will be able to:
- Order carboxylic acid derivatives, including anhydrides, esters, and amides, based on their reactivity
- Explain the relatively high rate of hydrolysis in β-lactams
- Identify the properties of the carboxylic acid derivatives that cause their reactivities to differ
Regardless of the carboxylic acid derivative at hand, there are some rules that govern the reactivity of these molecules.
Relative Reactivity of Derivatives
In a nucleophilic substitution reaction, the reactivity of the carbonyl is determined by its substituents. Anhydrides are most reactive, followed by esters (which are essentially tied with carboxylic acids), then finally amides. This can be explained by the structure of these molecules. Anhydrides, with their resonance stabilization and three electron-withdrawing oxygen atoms, are the most electrophilic. Esters, by comparison, lack one electron-withdrawing carbonyl oxygen and are slightly less reactive. Finally, amides, with an electron-donating amino group, are the least reactive toward nucleophiles.
KEY CONCEPT
When considering the reactivity of carboxylic acid derivatives toward nucleophilic attack, anhydrides are the most reactive, followed by esters and carboxylic acids, and then amides.
Steric Effects
Steric hindrance is always worth keeping in mind when considering reactivity. Steric hindrance describes when a reaction does not proceed due to the size of the substituents. A good example of this is in SN2 reactions, which will not occur at tertiary carbons. This effect, which might sound detrimental, can be used to our advantage—for example, if we want to push a reaction in an SN1 direction, rather than SN2, we can use a tertiary substrate. Another way that this is used synthetically is in the creation of protecting groups. As we saw in Chapter 6 of MCAT Organic Chemistry Review, aldehydes and ketones will readily react with strong reducing agents like LiAlH4—but this can be prevented by first reacting the aldehyde or ketone with two equivalents of alcohol, producing a nonreactive acetal or ketal. After we complete the rest of the desired reactions, we can then regenerate the carbonyl with aqueous acid. In the context of carboxylic acid derivatives, the size and substitution of the leaving group can affect the ability of a nucleophile to access the carbonyl carbon, thus affecting the reactivity of the derivative to nucleophilic acyl substitution.
KEY CONCEPT
Steric hindrance can be used to control where a reaction occurs in a molecule. Protecting groups may make it too hard for a nucleophile, oxidizing agent, or reducing agent to access or react with a part of the molecule.
Electronic Effects
There are several electronic effects that must be considered in organic chemistry on the MCAT, and all of them come into play when considering carboxylic acid derivatives. Induction refers to the distribution of charge across σ bonds. Electrons are attracted to atoms that are more electronegative, generating a dipole across the σ bond. The less electronegative atom acquires a slightly positive charge, and the more electronegative atom acquires a slightly negative charge. This effect is relatively weak and gets increasingly weaker as one moves further away within the molecule from the more electronegative atom. This effect is responsible for the dipole character of the carbonyl group, as well as the increased dipole character (and, therefore, susceptibility to nucleophilic attack) of carboxylic acids—which contain an additional oxygen atom in their leaving group. This also explains the overall relative reactivity of anhydrides, esters, and amides toward nucleophilic attack. Anhydrides have two electron-withdrawing groups, which leave a significant partial positive charge on the electrophilic carbon. This effect is smaller in amides because nitrogen is less electronegative than oxygen, and the dipole is not as strong.
Resonance and conjugation also affect the reactivity of a molecule. Conjugation refers to the presence of alternating single and multiple bonds. This setup implies that all of the atoms involved in these bonds are either sp2- or sp-hybridized—and therefore have unhybridized p-orbitals. When these p-orbitals align, they can delocalize π electrons through resonance, forming clouds of electron density above and below the plane of the molecule. This type of electron sharing is most commonly demonstrated using benzene, as shown in Figure 9.10.
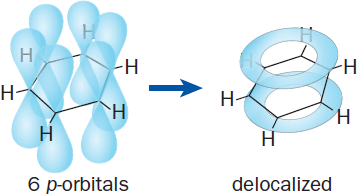
Figure 9.10. Conjugation in Benzene Parallel unhybridized p-orbitals combine to form delocalized electron clouds above and below the plane of the molecule.
KEY CONCEPT
Induction is the distribution of charge across σ bonds. Conjugation and resonance are much more powerful effects and occur in systems with alternating single and multiple bonds.
In carbonyl-containing compounds, conjugation can be established with the carbonyl group itself. α,β-unsaturated carbonyls or (enones) are common examples, as shown in Figure 9.11.
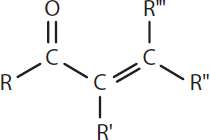
Figure 9.11. Conjugation in a Carbonyl-Containing Compound
This type of electron sharing makes for very stable compounds because these compounds have multiple resonance structures. This characteristic allows for the stabilization of a positive charge once the nucleophile has bonded, making these compounds more susceptible to nucleophilic attack.
Strain in Cyclic Derivatives
Lactams and lactones are cyclic amides and esters, respectively. Certain lactams and lactones are more reactive to hydrolysis because they contain more strain. β-lactams, for example, are four-membered cyclic amides and are highly reactive due to significant ring strain; four-membered rings have both torsional strain from eclipsing interactions and angle strain from compressing the normal sp3 angle of 109.5°. These molecules are part of the core structure of several antibiotic families, as shown in Figure 9.12. The ring strain, and therefore the reactivity, is increased by fusion to a second ring. The four-membered structure of a β-lactam also forces a trigonal pyramidal bond geometry on the nitrogen atom in the ring, which reduces resonance, making hydrolysis more likely.
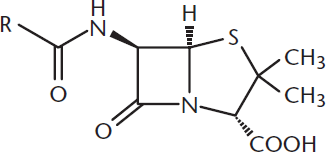
Figure 9.12. Penicillin, a β-Lactam-Containing Antibiotic
REAL WORLD
Many antibiotic families contain β-lactams, including the penicillin family, cephalosporins, carbapenems, and monobactams. Many bacteria have developed β-lactamases, which break β-lactam rings, as a resistance mechanism against these antibiotics. Therefore, β-lactams are sometimes given with β-lactamase inhibitors to increase their efficacy.
MCAT CONCEPT CHECK 9.2:
Before you move on, assess your understanding of the material with these questions.
-
Rank the following molecules by decreasing reactivity to OR–: acetamide, acetic anhydride, and ethyl acetate.
- ____________________________
- ____________________________
- ____________________________
-
What is responsible for the increased rate of hydrolysis in β-lactams?
________________________
-
What properties account for the differences in reactivity seen between anhydrides, esters, and amides with nucleophiles?
________________________
9.3 Nucleophilic Acyl Substitution Reactions
LEARNING OBJECTIVES
After Chapter 9.3, you will be able to:
- Describe the mechanism for transesterification reactions
- Explain how strong acid and strong base conditions would impact the mechanism of hydrolysis of an amide
- Identify the nucleophile and electrophile within nucleophilic acyl substitution reactions such as:

Although there are a seemingly infinite number of reactions in which carboxylic acid derivatives can participate, a much smaller group of reactions will appear on the MCAT. As we will observe, these reactions have much in common with those of carboxylic acids and other carbonyl-containing compounds. Many of the properties we have already discussed determine the ways in which these reactions proceed.
Anhydride Cleavage
As with carboxylic acids, nucleophilic acyl substitution involves nucleophilic attack of the carbonyl carbon with displacement of a leaving group. All carboxylic acid derivatives can participate in nucleophilic substitution reactions at different relative rates. Specifically, anhydrides are most reactive toward nucleophiles, followed by esters, and finally amides. One example of this is the formation of amides from the nucleophilic substitution reaction between ammonia and any carboxylic acid or derivative. The example shown in Figure 9.13 is not only a nucleophilic substitution reaction, but also a cleavage reaction because it splits an anhydride in two. In this reaction, ammonia acts as the nucleophile, one of the carbonyl carbons acts as the electrophile, and a carboxylic acid is the leaving group.
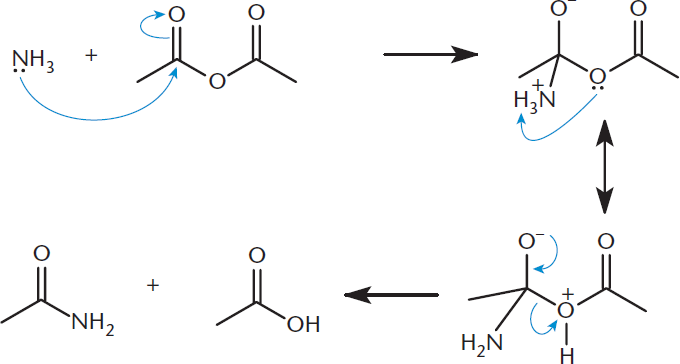
Figure 9.13. Nucleophilic Acyl Substitution: Anhydride to Amide and Carboxylic Acid
Alcohols can also act as nucleophiles toward anhydrides; this nucleophilic substitution reaction will result in the formation of esters and carboxylic acids, as shown in Figure 9.14.
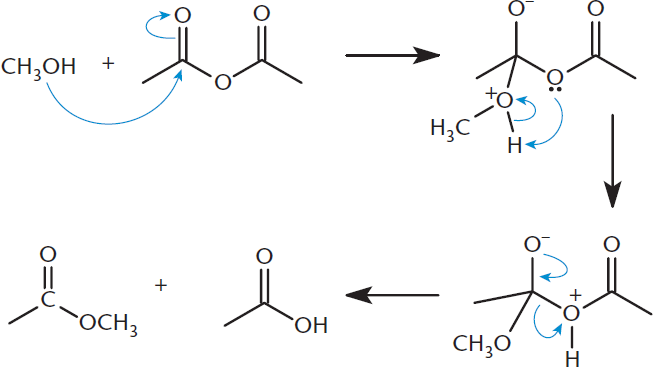
Figure 9.14. Nucleophilic Acyl Substitution: Anhydride to Ester and Carboxylic Acid
Anhydrides can also be reverted to carboxylic acids by exposing them to water, as shown in Figure 9.15. For these reactions to be useful, the anhydride should be symmetric; otherwise, one forms a mixture of products.
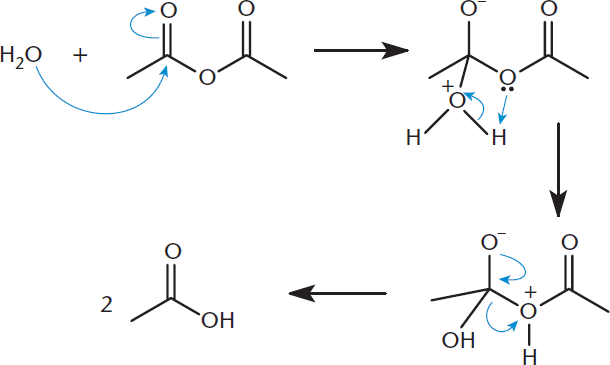
Figure 9.15. Nucleophilic Acyl Substitution: Anhydride to Carboxylic Acids
Transesterification
Alcohols can act as nucleophiles and displace the esterifying group on an ester. This process is called transesterification. In this reaction, one ester is simply transformed into another, as shown in Figure 9.16.
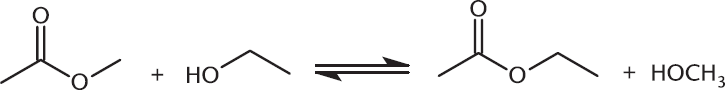
Figure 9.16. Nucleophilic Acyl Substitution: Transesterification Different alcohol chains are swapped into and out of the esterifying group position.
Hydrolysis of Amides
Amides can be hydrolyzed under highly acidic conditions via nucleophilic substitution. The acidic conditions allow the carbonyl oxygen to become protonated, making the molecule more susceptible to nucleophilic attack by a water molecule. The product of this reaction is a carboxylic acid and ammonia. This should be no surprise because this is the reverse of the condensation reaction by which amides are formed. This reaction is shown in Figure 9.17.
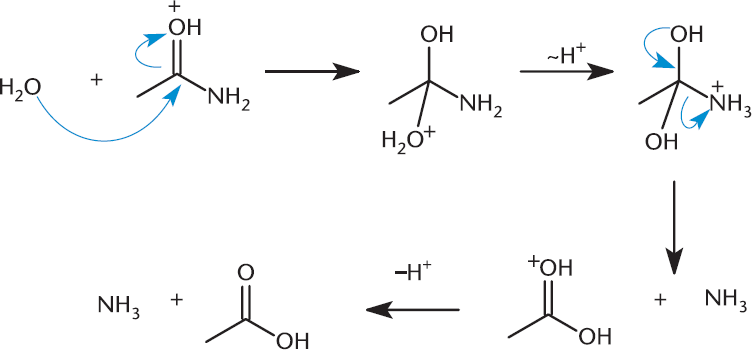
Figure 9.17. Nucleophilic Acyl Substitution: Amide to Carboxylic Acid Strong acid or base is needed to catalyze the hydrolysis of amides, which are normally quite stable.
Hydrolysis can also occur if conditions are basic enough. The reaction is similar to an acid-catalyzed reaction, except that the carbonyl oxygen is not protonated and the nucleophile is a hydroxide ion. The product of this reaction would be the deprotonated carboxylate anion.
MCAT CONCEPT CHECK 9.3:
Before you move on, assess your understanding of the material with these questions.
-
In the formation of an amide from ammonia and an anhydride, what serves as the nucleophile? The electrophile?
- Nucleophile:
________________________
- Electrophile:______________________________
-
What are the products of the transesterification reaction between isopropyl butanoate and ethanol?
________________________
-
How do strongly acidic and strongly basic conditions catalyze the hydrolysis of an amide?
- Strongly acidic conditions:
________________________
- Strongly basic conditions:______________________________
Conclusion
We’re sure you’ve noticed that this chapter covers only a few reactions happening in a wide variety of contexts. The MCAT test makers don’t want you to memorize all the possible reactions; they simply want you to truly understand the trends and the underlying reasons for these reactions. Make sure you know the order of reactivity of the derivatives (from anhydrides, the most, to amides, the least). Also, learn the special reactions of esters and amides. Your study of amides will pay off right away as we explore amino acids and other nitrogen- and phosphorus-containing compounds in the next chapter.
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Amides, Esters, and Anhydrides
- Amides are the condensation products of carboxylic acids and ammonia or amines.
- Amides are given the suffix –amide. The alkyl groups on a substituted amide are written at the beginning of the name with the prefix N–.
- Cyclic amides are called lactams. Lactams are named by the Greek letter of the carbon forming the bond with the nitrogen (β-lactam, γ-lactam, and so on).
- Esters are the condensation products of carboxylic acids with alcohols (Fischer esterification).
- Esters are given the suffix –oate. The esterifying group is written as a substituent, without a number.
- Cyclic esters are called lactones. Lactones are named by the number of carbons in the ring and the Greek letter of the carbon forming the bond with the oxygen (α-acetolactone, β-propiolactone, and so on).
- Triacylglycerols, which are a form of fat storage, include three ester bonds between glycerol and fatty acids. Saponification is the breakdown of fat using a strong base to form soap (salts of long-chain carboxylic acids).
- Anhydrides are the condensation dimers of carboxylic acids.
- Symmetric anhydrides are named for the parent carboxylic acid, followed by anhydride. Asymmetric anhydrides are named by listing the parent carboxylic acids alphabetically, followed by anhydride.
- Some cyclic anhydrides can be synthesized by heating dioic acids. Five- or six-membered rings are generally stable.
Reactivity Principles
- In nucleophilic substitution reactions, anhydrides are more reactive than esters, which are more reactive than amides.
- Steric hindrance describes when a reaction cannot proceed (or significantly slows) because of substituents crowding the reactive site. Protecting groups, such as acetals, can be used to increase steric hindrance or otherwise decrease the reactivity of a particular portion of a molecule.
- Induction refers to uneven distribution of charge across a σ bond because of differences in electronegativity. The more electronegative groups in a carbonyl-containing compound, the greater its reactivity.
- Conjugation refers to the presence of alternating single and multiple bonds, which creates delocalized π electron clouds above and below the plane of the molecule. Electrons experience resonance through the unhybridized p-orbitals, increasing stability. Conjugated carbonyl-containing compounds are more reactive because they can stabilize their transition states.
- Increased strain in a molecule can make it more reactive. β-lactams are prone to hydrolysis because they have significant ring strain. Ring strain is due to torsional strain from eclipsing interactions and angle strain from compressing bond angles below 109.5°.
Nucleophilic Acyl Substitution Reactions
- All carboxylic acid derivatives can undergo nucleophilic substitution reactions. The rates at which they do so are determined by their relative reactivities.
- Anhydrides can be cleaved by the addition of a nucleophile.
- Addition of ammonia or an amine results in an amide and a carboxylic acid.
- Addition of an alcohol results in an ester and a carboxylic acid.
- Addition of water results in two carboxylic acids.
- Transesterification is the exchange of one esterifying group for another on an ester. The attacking nucleophile is an alcohol.
- Amides can be hydrolyzed to carboxylic acids under strongly acidic or basic conditions. The attacking nucleophile is water or the hydroxide anion.
ANSWERS TO CONCEPT CHECKS
**9.1**
-
Carboxylic Acid Derivative Formed from –COOH by… Suffix Cyclic Naming Amide Ammonia or an amine –amide Lactam
Ester An alcohol –oate Lactone
Anhydride Another carboxylic acid anhydride Anhydride
- A condensation reaction is one in which two molecules are joined with the loss of a small molecule. In all of the examples in this section, the small molecule lost was water.
**9.2**
- Anhydrides are the most reactive to nucleophiles, followed by esters, and then amides. Therefore, acetic anhydride will be the most reactive, followed by ethyl acetate, and finally acetamide.
- β-lactams are susceptible to hydrolysis due to the high level of ring strain, which is due to both torsional strain (eclipsing interactions) and angle strain (deviation from 109.5°).
- Electronic effects like induction have some effect on the reactivity of the carbonyl in these three functional groups. Differences in resonance also explain the increased reactivity of anhydrides, in particular. Steric effects could also be significant, depending on the specific leaving group present.
**9.3**
- Ammonia acts as the nucleophile. One of the carbonyl carbons of the anhydride serves as the electrophile.
- Transesterification is the exchange of one esterifying group for another in an ester. This reaction requires an alcohol as a nucleophile. In this case, the ethyl group of ethanol will replace the isopropyl group of isopropyl butanoate, resulting in ethyl butanoate and 2-propanol.
- Strongly acidic conditions catalyze amide hydrolysis by protonating the oxygen in the carbonyl. This increases the electrophilicity of the carbon, making it more susceptible to nucleophilic attack. Strongly basic conditions greatly increase the concentration of OH–, which can act as a nucleophile on amide carbonyls.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. A
Methyl propanoate is an ester; it can be synthesized by reacting a carboxylic acid with an alcohol in the presence of acid. Here, the parent chain is propanoate, and the esterifying group is a methyl group. (B) reverses the nomenclature and would form propyl methanoate. The other reactions listed would not form esters.
2. D
This question asks for the products when ammonia reacts with acetic anhydride. Recall from the chapter that an amide and a carboxylic acid will be formed. However, the carboxylic acid—an acid—is in the same environment as ammonia—a base. The two will react, forming the ammonium carboxylate shown.
3. A
A Fischer esterification involves reacting a carboxylic acid and an alcohol with an acid catalyst. Under these conditions, the carbonyl carbon is open to attack by the nucleophilic alcohol. The rate of this reaction depends on the amount of steric hindrance around the carbonyl carbon because there must be room for the alcohol to approach the carboxylic acid substrate. (B), (C), and (D) all have more crowding around the carbonyl carbon, which will decrease reactivity. The additional alkyl groups in these other choices also donate electron density to the carbonyl carbon, making it slightly less electrophilic.
4. C
This question requires knowledge of the nomenclature of cyclic molecules. A δ-lactam, (A), has a bond between the nitrogen and the fourth carbon away from the carbonyl carbon. This ring will have six elements: the nitrogen, the carbonyl carbon, and the four carbons in between. Cyclohexane carboxylic acid, (B), has cyclohexane, a six-membered cycloalkane. The anhydride formed from pentanedioic acid, (D), will have the five carbons in the parent chain and one oxygen atom closing the ring, meaning there are still six elements. γ-butyrolactone will have five elements because it contains a bond between the ester oxygen and the third carbon away from the carbonyl carbon. The five elements will be the oxygen, the carbonyl carbon, and the three carbons in between.
5. D
With the same R groups, steric influence is the same in each case, so we can therefore rely solely on electronic effects. When this is all that is taken into account, reactivity toward nucleophiles is highest for anhydrides, followed by esters and carboxylic acids, then amides.
6. C
Anhydrides, particularly cyclic anhydrides, will form spontaneously from dicarboxylic acids when heated.
7. A
Methylamine would react readily to form an amide. The less substituted the nucleophile, the easier it will be for the nucleophile to attack the carbonyl carbon and form the amide. In fact, triethylamine, (B), would not be able to form an amide at all because it does not have a hydrogen to lose while attaching to the carbonyl carbon.
8. A
Propanamide is an amide; as such, it is the least reactive of the carboxylic acid derivatives discussed in this chapter. Without a strong acid or base, propanamide will not be able to undergo nucleophilic acyl substitution and no reaction will occur.
9. D
β-lactams are amides in the form of four-membered rings; amides are generally the least reactive type of carboxylic acid derivative. β-lactams experience significant ring strain from both eclipsing interactions (torsional strain) and angle strain and are therefore more susceptible to hydrolysis than the linear form of the same molecule.
10. C
As far as we can tell, we are converting one ester to another in this reaction. The fact that this reaction is acid-catalyzed should confirm the suspicion that this is a transesterification reaction.
11. B
This reaction, which is the hydrolysis of an amide, is favored in strong acid. Acid protonates the carbonyl oxygen, which increases the electrophilicity of the carbonyl carbon. This allows water to serve as the nucleophile, attacking the bond and hydrolyzing the molecule.
12. D
This molecule is more stable with a positive charge than a straight-chain alkane due to the conjugation of the benzene ring. This permits delocalization of the charge through resonance. Although induction, (C), does have an effect on the stabilization of the molecule, this effect is much less significant than the impact of having a conjugated system. The electronegativity of nitrogen, (B), which primarily affects induction, is also not a vital component of the stabilization by this molecule of a positive charge because oxygen is more electronegative. Steric hindrance, (A), would affect the reactivity of a molecule, but not its ability to stabilize charge.
13. D
The molecule shown, γ-nonalactone, is a cyclic ester, also called a lactone. This molecule could arise from intramolecular attack in a γ-hydroxycarboxylic acid.
14. A
In order to prepare butyl acetate from butanol, we need to perform a nucleophilic acyl substitution reaction. If the product is an ester, we need to start with a reactant that is more reactive than the ester itself, or the reaction will not proceed. Anhydrides are more reactive than esters, but amides are less reactive, eliminating (C) and (D). Reaction with propanoic anhydride, as in (B), would result in butyl propanoate.
15. D
The presence of water in an esterification reaction would likely revert some of the desired esters back into carboxylic acids. Small carboxylic acids, like formic or acetic acid, are easily dissolved in water, eliminating (A). The polarity of water plays little role in affecting the leaving group; if anything, water can be used to increase the electrophilicity of the carbonyl carbon by protonating the carbonyl oxygen—eliminating (B). Finally, this is a nucleophilic substitution mechanism, not a nucleophilic addition mechanism, as mentioned in (C). Further, hydrogen bonding would likely augment the reaction.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 11
- Lipid and Amino Acid Metabolism
- Organic Chemistry Chapter 1
- Nomenclature
- Organic Chemistry Chapter 3
- Bonding
- Organic Chemistry Chapter 4
- Analyzing Organic Reactions
- Organic Chemistry Chapter 6
- Aldehydes and Ketones I
- Organic Chemistry Chapter 8
- Carboxylic Acids