Chapter 10: Nitrogen- and Phosphorus-Containing Compounds

Chapter 10: Nitrogen- and Phosphorus-Containing Compounds
SCIENCE MASTERY ASSESSMENT
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what's important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don't worry though— skipping something now does not mean you'll never study it. Later on in your prep, as you complete full-length tests, you'll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
-
Which of the following amino acids does not have an L-enantiomer?
- Cysteine
- Threonine
- Glutamic acid
- Glycine
-
Which of the following would be formed if methyl bromide were reacted with phthalimide and followed by hydrolysis with an aqueous base?
- C2H5NH2
- CH3NH2
- (C2H5)3N
- (CH3)4N+Br–
-
Which of the following amino acids contain(s) sulfur?
- Cysteine
- Serine
- Methionine
- I only
- I and III only
- II and III only
- I, II, and III
-
Nylon, a polyamide, is produced from hexanediamine and a substance, X. This substance, X, is most probably a(n):
- amine.
- carboxylic acid.
- ketone.
- alcohol.
-
Intermediates in the Strecker synthesis include all of the following nitrogen-containing functional groups EXCEPT a(n):
- nitrile.
- imine.
- amide.
- amine.
-
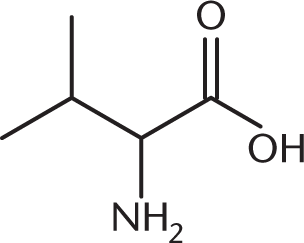
A biochemist is synthesizing valine, shown below, using the Strecker synthesis. Which of the following carbonyl-containing compounds would be an appropriate starting reactant in this synthesis?
- 2-Propanone
- Propanal
- 2-Methylpropanal
- Butanal
-
Why is the C–N bond of an amide planar?
- It has partial double-bond character.
- It is sp3-hybridized.
- It has some sp2 character.
- I only
- II only
- I and II only
- I and III only
-
Which of the primary methods of amino acid synthesis results in an optically active solution?
- The Strecker synthesis only
- The Gabriel synthesis only
- Both the Strecker and Gabriel syntheses
- Neither the Strecker nor the Gabriel syntheses
-
During the Gabriel synthesis, phthalimide serves as the:
- nucleophile.
- base.
- leaving group.
- electrophile.
-
Each of the following reaction types occurs during the Gabriel synthesis EXCEPT:
- decarboxylation.
- nucleophilic substitution.
- dehydration.
- hydrolysis.
-
At physiological pH, which two forms of phosphoric acid have the highest concentrations?
- H3PO4 and H2PO4−
- H2PO4− and HPO42−
- HPO42− and PO43−
- PO43− and H3PO4
-
In aqueous solution, pyrophosphate will likely:
- form insoluble complexes.
- be stable and inert.
- degrade into inorganic phosphate.
- decrease the polarity of the solvent.
-
What would be the charge of aspartic acid at pH 7?
- Neutral
- Negative
- Positive
- There is not enough information to answer the question.
-
When a bond is created between two nucleotide triphosphates in DNA synthesis, the small molecule released from this reaction is:
- pyrophosphate.
- inorganic phosphate.
- ATP.
- organic phosphate.
-
The hydrogens of phosphoric acid have pKa values that:
- allow high buffering capacity over a small pH range.
- allow moderate buffering capacity over a large pH range.
- allow low buffering capacity over a small pH range.
- do not allow buffering.
Answer Key
- D
- B
- B
- B
- C
- C
- D
- D
- A
- C
- B
- C
- B
- A
- B
Chapter 10: Nitrogen- and Phosphorus-Containing Compounds
CHAPTER 10
NITROGEN- AND PHOSPHORUS-CONTAINING COMPOUNDS
In This Chapter
10.1 Amino Acids, Peptides, and Proteins
Description
Properties
**10.2 Synthesis of α-Amino Acids**
Strecker Synthesis
Gabriel Synthesis
10.3 Phosphorus-Containing Compounds
Description
Properties
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 4% of all questions about organic chemistry on the MCAT.
This chapter covers material from the following AAMC content categories:
1A: Structure and function of proteins and their constituent amino acids
5D: Structure, function, and reactivity of biologically-relevant molecules
MCAT EXPERTISE
Note that, even though biological molecules — amino acids, proteins, and DNA — are called out in this chapter, the low percentage rating for this chapter only applies to non-biochemical reactions of these molecules. Mastery of these molecules in biological settings is still very important for Test Day success; that content is found in Chapters 1, 2, 3, 6, and 7 of MCAT Biochemistry Review.
Introduction
Organic chemistry is the study of carbon-containing molecules. But as you’ve seen, carbon is not the only element that plays a role in organic molecules; many of the functional groups we’ve explored up until this point also include hydrogen and oxygen. Together, these three elements make up 93% of the composition of the human body by weight. But other atoms also contribute to biomolecules: nitrogen comprises 3.2% of body weight and phosphorus 1.0%.
In this chapter, we’ll take a look at some biologically important molecules that contain nitrogen and phosphorus. In addition to the amides explored in the previous chapter, amino acids also contain nitrogen. These building blocks of proteins are discussed in depth in Chapter 1 of MCAT Biochemistry Review; in this chapter, we review their structure and focus on laboratory methods for synthesizing amino acids. Finally, we turn our attention to phosphorus-containing molecules, which often are used to transfer energy—or store genetic information.
10.1 Amino Acids, Peptides, and Proteins
LEARNING OBJECTIVES
After Chapter 10.1, you will be able to:
- Identify the functional groups that make amino acids amphoteric
- Recall the mechanisms for forming and cleaving peptide bonds
- Explain why the C–N bond of an amide is planar
- Recall the unique properties of glycine:
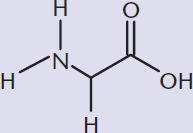
Amino acids are dipolar molecules that come together through a condensation reaction, forming peptides. Larger, folded peptide chains are considered proteins.
Description
Amino acids contain an amino group and a carboxyl group attached to a single carbon atom (the α-carbon). The other two substituents of the α-carbon are a hydrogen atom and a side chain referred to as the R group. This structure is shown in Figure 10.1.
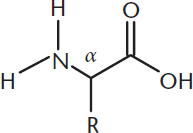
Figure 10.1. Amino Acid Structure
The α-carbon, with its four different groups, is a chiral (stereogenic) center. Glycine, the simplest amino acid, is an exception to this rule because its R group is a hydrogen atom. All naturally occurring amino acids in eukaryotes—except for glycine—are optically active, and all are L-isomers. Therefore, by convention, the Fischer projection for an amino acid is drawn with the amino group on the left, as shown in Figure 10.2. L-amino acids have (S) configurations, except for cysteine, which is (R) because of the change in priority caused by the sulfur in its R group.
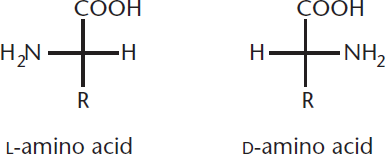
Figure 10.2. L- and D-Amino Acids
Properties
Amino acids, with their acidic carboxyl group and basic amino group, are amphoteric molecules. That is, they can act as both acids and bases. Amino groups can take on a positive charge by being protonated, and carboxyl groups can take on negative charges by being deprotonated. When an amino acid is put into solution, it will take on both of these charges, forming a dipolar ion or **zwitterion, as shown in Figure 10.3. How an amino acid acts depends on the pH of the environment. In basic solutions, the amino acid can become fully deprotonated; in acidic solutions, it can become fully protonated.
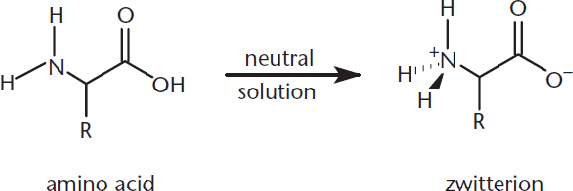
Figure 10.3. Amino Acids Exist as Zwitterions (Dipolar Ions) at Neutral pH
BRIDGE
Amino acids are amphoteric molecules, just like water—they can act as both acids and bases. These acid–base characteristics (and titrations of amino acids) are discussed thoroughly in Chapter 1 of MCAT Biochemistry Review.
Aside from the zwitterionic properties common to every amino acid, each one has properties defined by its R group, or side chain. The 20 eukaryotic proteogenic amino acids are grouped into five categories: nonpolar nonaromatic,aromatic, polar, negatively charged (acidic), and positively charged (basic). Nonpolar nonaromatic amino acids tend to have side chains that are saturated hydrocarbons, like alanine, valine, leucine, and isoleucine; they also include glycine, proline (which is cyclic, with a secondary amine), and methionine (which contains sulfur). Aromatic amino acids include tryptophan, phenylalanine, and tyrosine. Nonpolar amino acids—both nonaromatic and aromatic—are also hydrophobic and tend to be sequestered in the interior of proteins. Polar amino acids tend to have terminal groups containing oxygen, nitrogen, or sulfur. These include serine, threonine, asparagine, glutamine, and cysteine. Negatively charged (acidic) amino acids include aspartic acid and glutamic acid. These amino acids have terminal carboxylate anions in their R groups. Finally, positively charged (basic) amino acids, including arginine, lysine, and histidine, have a protonated amino group in their R groups. Polar, acidic, and basic amino acids are all hydrophilic and tend to form hydrogen bonds with water in aqueous solution.
Amino acids undergo condensation reactions to form peptide bonds. The molecules these bonds form, called polypeptides, are the base unit of proteins. The reverse reaction, hydrolysis of the peptide bond, is catalyzed by a strong acid or base. Both of these reactions are shown in Figure 10.4.
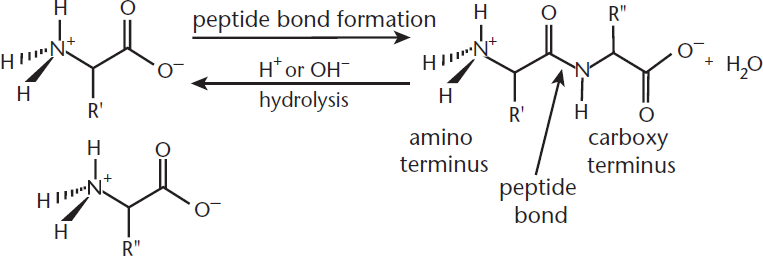
Figure 10.4. Peptide Bond Formation and Cleavage
Like other carbonyl-containing functional groups, amides have two resonance structures, as shown in Figure 10.5. The true structure of the amide bond is therefore a hybrid of these two structures, with partial double-bond character between the nitrogen atom and the carbonyl carbon. This double-bond character limits rotation about the C–N bond, which adds to the rigidity and stability of the backbone of proteins. The single bonds on either side of the peptide bond, on the other hand, permit free rotation.

Figure 10.5. Resonance in the Peptide Bond
KEY CONCEPT
Rotation is limited around the peptide bond because resonance gives the C–N bond partial double-bond character.
MCAT CONCEPT CHECK 10.1:
Before you move on, assess your understanding of the material with these questions.
-
What makes glycine unique among the amino acids?
________________________
-
Amino acids are amphoteric. What does this mean? What functional groups give amino acids this characteristic?
________________________
-
How are peptide bonds formed and cleaved?
________________________
-
Why is the C–N bond of an amide planar?
________________________
10.2 Synthesis of *α*-Amino Acids
LEARNING OBJECTIVES
After Chapter 10.2, you will be able to:
- Recall the required reactants and product types for the Strecker and Gabriel synthesis reactions
- Identify the reaction types found in the Strecker and Gabriel synthesis reactions
Synthesis of amino acids occurs by an astonishing variety of mechanisms in vivo. In the lab, several simple reaction mechanisms are exploited to make amino acids neatly and efficiently.
Strecker Synthesis
In the Strecker synthesis, one starts with an aldehyde, ammonium chloride (NH4Cl), and potassium cyanide (KCN), as shown in Figure 10.6. The carbonyl oxygen is protonated, increasing the electrophilicity of the carbonyl carbon. Then, as seen in Chapter 6 of MCAT Organic Chemistry Review, ammonia can attack the carbonyl carbon, forming an imine. The imine carbon is also susceptible to nucleophilic addition reactions; thus, the CN– anion from KCN attacks, forming a nitrile group (–C≡N). The final molecule at the end of Step 1 is an aminonitrile—a compound containing an amino group (–NH2) and a nitrile group.
MNEMONIC
Nitriles have a triple bond between nitrogen and carbon.
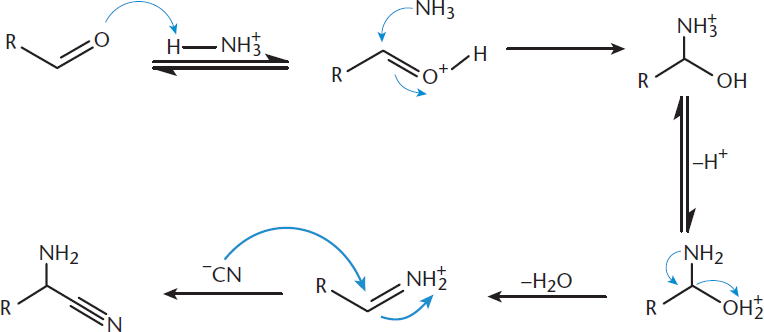
Figure 10.6. Strecker Synthesis: Step 1 An aminonitrile is generated from an aldehyde or ketone.
In Step 2, the nitrile nitrogen is protonated, increasing the electrophilicity of the nitrile carbon. This is similar to protonating the oxygen of a carbonyl. A water molecule attacks, leading to the creation of a molecule with both imine and hydroxyl moieties on the same carbon. This imine is attacked by another equivalent of water. A carbonyl is formed, kicking off ammonia and creating the carboxylic acid functionality. This step, shown in Figure 10.7, is performed in aqueous acid and can be accelerated by the use of heat.
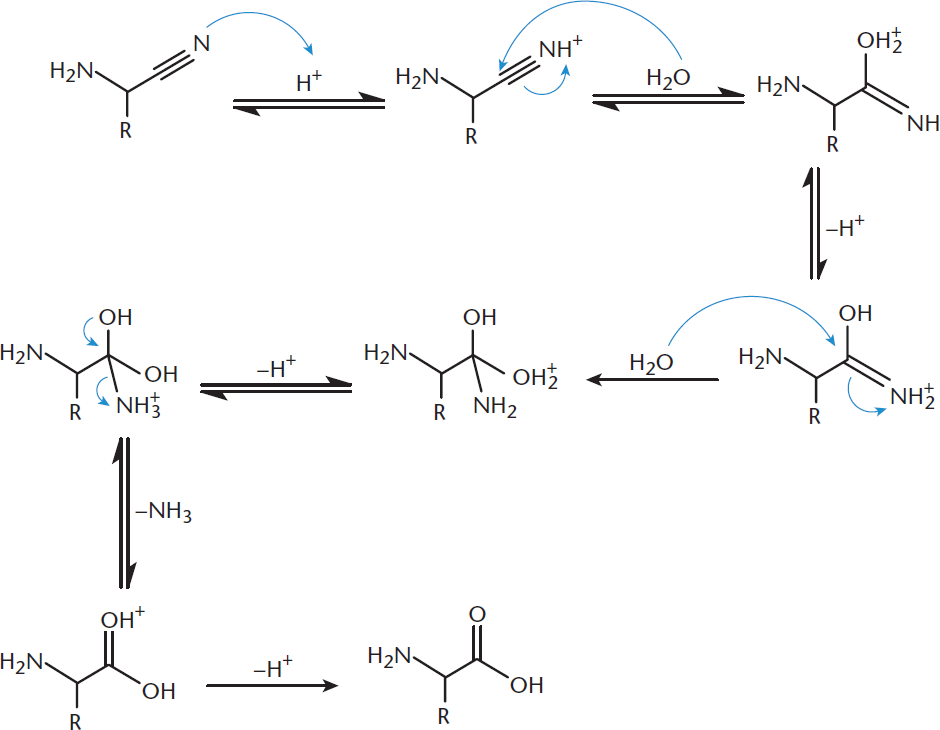
Figure 10.7. Strecker Synthesis: Step 2 An amino acid is generated from the aminonitrile.
The starting material for the Strecker synthesis is a planar carbonyl-containing compound; therefore, the product of this pathway is a racemic mixture. The incoming nucleophiles are equally able to attack from either side of the carbonyl; thus, both L- and D-amino acids can be generated through this process.
Gabriel Synthesis
Another way of synthesizing amino acids is the Gabriel (malonic-ester) synthesis, shown in Figure 10.8.
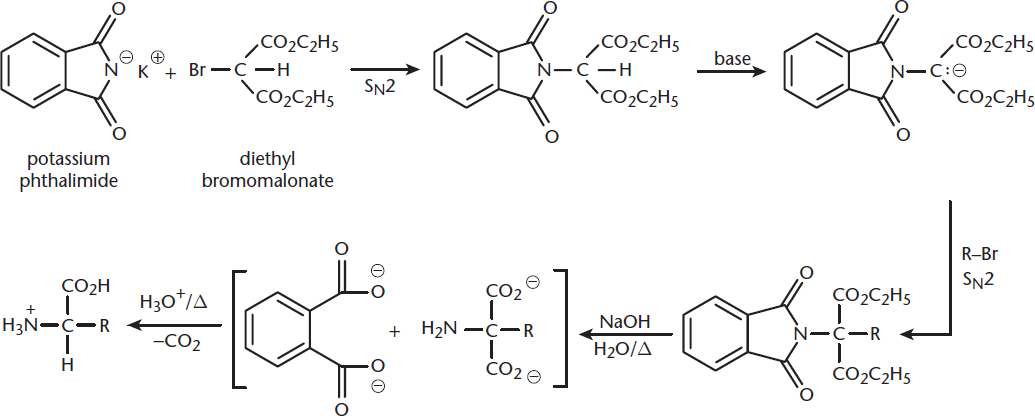
Figure 10.8. Gabriel Synthesis An amino acid is generated from phthalimide and diethyl bromomalonate, using two SN2 reactions, hydrolysis, and decarboxylation.
In this method, potassium phthalimide is reacted with diethyl bromomalonate. Phthalimide is acidic and exists in solution as a nucleophilic anion. Diethyl bromomalonate contains a secondary carbon bonded to bromine, a good leaving group. This setup should sound much like the SN2 reactions discussed in Chapter 4 of MCAT Organic Chemistry Review. With phthalimide as the nucleophile, the (secondary) substrate carbon as the electrophile, and bromine as the leaving group, this reaction generates a phthalimidomalonic ester. Consider the benefits of using such a large nucleophile. The bulkiness of this group creates steric hindrance, which prevents the substrate carbon from undergoing multiple substitutions.
Instead, in the presence of base, this carbon (which is the α-carbon between two carbonyls) can easily be deprotonated. The molecule as a whole can then act as a nucleophile, attacking the substrate carbon of a bromoalkane. This is another example of an SN2 reaction. The nucleophile is the large, deprotonated phthalimidomalonic ester, the electrophile is the substrate carbon, and the leaving group is the bromide anion.
Next, this molecule is hydrolyzed with strong base and heat. Much like converting a cyclic anhydride into a dioic acid, the phthalimide moiety is removed as phthalic acid (with two carboxylic acids). The malonic ester is hydrolyzed to a dicarboxylic acid with an amine on the α-carbon.
Finally, this dicarboxylic acid, which is a 1,3-dicarbonyl, can be decarboxylated through the addition of acid and heat. The loss of a molecule of carbon dioxide results in the formation of the complete amino acid.
Like the Strecker synthesis, the Gabriel synthesis starts with a planar molecule; thus, the product is a racemic mixture of L- and D-amino acids.
KEY CONCEPT
Both the Strecker and Gabriel synthesis methods result in a racemic mixture of amino acids.
MCAT CONCEPT CHECK 10.2:
Before you move on, assess your understanding of the material with these questions.
-
What are the four reactants in the Strecker synthesis of an amino acid?
- ____________________________
- ____________________________
- ____________________________
- ____________________________
-
What are the reaction types used in the Strecker synthesis?
________________________
-
What are the four main reactants in the Gabriel synthesis of an amino acid?
- ____________________________
- ____________________________
- ____________________________
- ____________________________
-
What are the reactions types used in the Gabriel synthesis?
________________________
10.3 Phosphorus-Containing Compounds
LEARNING OBJECTIVES
After Chapter 10.3, you will be able to:
- Recognize the traits that make inorganic phosphate a useful molecule for energy transfer
- Explain why phosphoric acids are good buffers
- Recall what makes a molecule an organic phosphate
Phosphoric acid is an extremely important molecule biochemically. This molecule forms the high-energy bonds that carry energy in adenosine triphosphate (ATP).
Description
In a biochemical context, phosphoric acid is sometimes referred to as a phosphate group or inorganic phosphate, denoted Pi. At physiological pH, inorganic phosphate includes molecules of both hydrogen phosphate (HPO42−) and dihydrogen phosphate (H2PO4−).
In addition to the energy-carrying nucleotide phosphates, phosphorus is also found in the backbone of DNA in phosphodiester bonds linking the sugar moieties of the nucleotides, as shown in Figure 10.9.
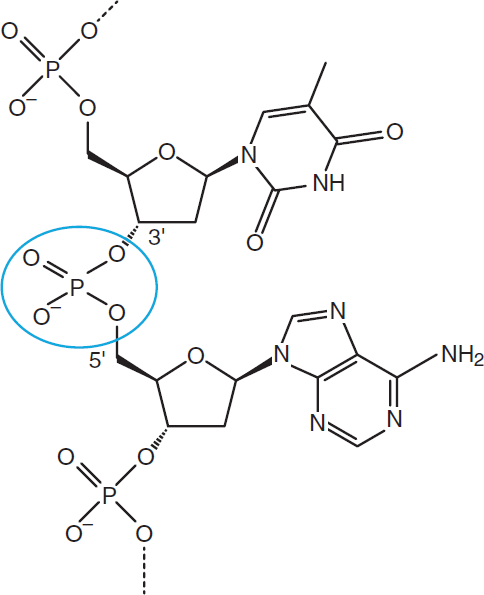
Figure 10.9. Phosphodiester Bond in DNA
When a new nucleotide is joined to a growing strand of DNA by a DNA polymerase, it releases an ester dimer of phosphate, referred to as pyrophosphate (P2O74−), denoted PPi, and shown in Figure 10.10. The hydrolytic release of this molecule provides the energy for the formation of the new phosphodiester bond. Pyrophosphate is unstable in aqueous solution and is hydrolyzed to form two molecules of inorganic phosphate, which can then be recycled to form high-energy bonds in ATP or for other purposes.
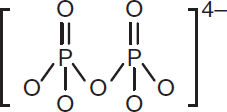
Figure 10.10. Pyrophosphate Anion
BRIDGE
DNA replication is an important process for the MCAT. Nucleotide triphosphates are added to the growing daughter strand with the release of pyrophosphate, PPi. The process of DNA synthesis is described in Chapter 6 of MCAT Biochemistry Review.
Nucleotides, such ATP, GTP, and those in DNA, are referred to as organic phosphates due to the presence of the phosphate group bonded to a carbon-containing molecule.
Properties
Phosphoric acid is unique in that it has three acidic hydrogens, each with its own pKa. Phosphoric acid most properly refers to the form that predominates in strongly acidic conditions, H3PO4. In mildly acidic conditions, it loses a proton to become dihydrogen phosphate, H2PO4−; it will readily lose a second proton to become hydrogen phosphate, HPO42− in weakly basic solutions. The form that exists in strongly basic solutions is phosphate, PO43−. The pKa for the loss of the first hydrogen is 2.15; for the second, 7.20; and for the third, 12.32. At a physiological pH of 7.4, this means that dihydrogen phosphate and hydrogen phosphate predominate in nearly equal proportions. This variety of pKa values also makes phosphates good buffers because they can pick up or give off protons depending on the pH of the solution.
KEY CONCEPT
Phosphoric acid is an excellent buffer because it has three hydrogens with pKa values that span nearly the entire pH scale.
Adjacent phosphate groups on a nucleotide triphosphate experience a large amount of repulsion because they are negatively charged. This, combined with the ability of phosphate to stabilize up to three negative charges by resonance, means that the energy released when a phosphate or pyrophosphate is cleaved is quite high.
MCAT CONCEPT CHECK 10.3:
Before you move on, assess your understanding of the material with these questions.
-
What characteristics make inorganic phosphate so useful for energy transfer biologically?
________________________
-
What is an organic phosphate?
________________________
-
What characteristics of phosphoric acids make them good buffers?
________________________
Conclusion
In this chapter, we spent a lot of time looking at biologically active molecules—but did you notice that these molecules are simply applications of the general principles that we have been learning throughout the chapters of this book? By applying your knowledge of the reactions and properties of different types of molecules, you can understand how biological processes work and how complex organic chemistry mechanisms work, like those of the Strecker and Gabriel syntheses. Many of these processes will fall into categories of reactions that we’ve seen before over and over—nucleophilic substitution, nucleophilic addition, and condensation reactions are just a few examples. The MCAT doesn’t require you to memorize tables of reactants or regurgitate hundreds of named reactions from scratch. Instead, the MCAT asks you to look at the bigger picture and understand the trends—which you’ve now learned!
GO ONLINE
You've reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Amino Acids, Peptides, and Proteins
- The α-carbon of an amino acid is attached to four groups: an amino group, a carboxyl group, a hydrogen atom, and an R group. It is a chiral stereocenter in all amino acids except glycine.
- All amino acids in eukaryotes are L-amino acids. They all have (S) stereochemistry except cysteine, which is (R).
- Amino acids are amphoteric, meaning they can act as acids or bases.
- Amino acids get their acidic characteristics from carboxylic acids and their basic characteristics from amino groups.
- In a neutral solution, amino acids tend to exist as zwitterions (dipolar ions).
- Amino acids can be classified by their R groups.
- Nonpolar nonaromatic amino acids include alanine, valine, leucine, isoleucine, glycine, proline, and methionine.
- Aromatic amino acids include tryptophan, phenylalanine, and tyrosine. Both nonpolar nonaromatic and aromatic amino acids tend to be hydrophobic and reside in the interior of proteins.
- Polar amino acids include serine, threonine, asparagine, glutamine, and cysteine.
- Negatively charged amino acids contain carboxylic acids in their R groups and include aspartic acid and glutamic acid.
- Positively charged amino acids contain amines in their R groups and include arginine, lysine, and histidine.
- Nonpolar nonaromatic and aromatic amino acids tend to be hydrophobic and reside in the interior of proteins.
- Polar, negatively charged (acidic), and positively charged (basic) amino acids tend to be hydrophilic and reside on the surface of proteins, making hydrogen bonds with the aqueous environment.
- Peptide bonds form by condensation reactions and can be cleaved hydrolytically.
- Resonance of the peptide bond restricts motion about the C–N bond, which takes on partial double-bond character.
- Strong acid or base is needed to cleave a peptide bond.
- Polypeptides are made up of multiple amino acids linked by peptide bonds. Proteins are large, folded, functional polypeptides.
Synthesis of *α*-Amino Acids
- Biologically, amino acids are synthesized in many ways. In the lab, certain standardized mechanisms are used.
- TheStrecker synthesis generates an amino acid from an aldehyde.
- An aldehyde is mixed with ammonium chloride (NH4Cl) and potassium cyanide. The ammonia attacks the carbonyl carbon, generating an imine. The imine is then attacked by the cyanide, generating an aminonitrile.
- The aminonitrile is hydrolyzed by two equivalents of water, generating an amino acid.
- The Gabriel synthesis generates an amino acid from potassium phthalimide, diethyl bromomalonate, and an alkyl halide.
- Phthalimide attacks the diethyl bromomalonate, generating a phthalimidomalonic ester.
- The phthalimidomalonic ester attacks an alkyl halide, adding an alkyl group to the ester.
- The product is hydrolyzed, creating phthalic acid (with two carboxyl groups) and converting the esters into carboxylic acids.
- One carboxylic acid of the resulting 1,3-dicarbonyl is removed by decarboxylation.
Phosphorus-Containing Compounds
- Phosphorus is found in inorganic phosphate (Pi), a buffered mixture of hydrogen phosphate (HPO42−) and dihydrogen phosphate (H2PO4−).
- Phosphorus is found in the backbone of DNA, which uses phosphodiester bonds. In forming these bonds, a pyrophosphate (PPi, P2O74−) is released. Pyrophosphate can then be hydrolyzed to two inorganic phosphates.
- Phosphate bonds are high energy because of large negative charges in adjacent phosphate groups and resonance stabilization of phosphates.
- Organic phosphates are carbon-containing compounds that also have phosphate groups. The most notable examples are nucleotide triphosphates (such as ATP or GTP) and DNA.
- Phosphoric acid has three hydrogens, each with a unique pKa. This wide variety in pKa values allows phosphoric acid to act as a buffer over a large range of pH values.
ANSWERS TO CONCEPT CHECKS
**10.1**
- All amino acids, except glycine, have chiral α-carbons. Because the R group of glycine is a hydrogen atom, it is not chiral and therefore is not optically active.
- Amphoteric molecules can act as acids or bases. Carboxylic acids give amino acids their acidic properties because they can be deprotonated. Amino groups give amino acids their basic properties because they can be protonated.
- Peptide bonds are formed by a condensation reaction, in which water is lost, and cleaved hydrolytically by strong acid or base.
- The C–N bond of an amide is planar because it has partial double-bond character due to resonance. Double bonds exist in a planar conformation and restrict movement.
**10.2**
- An aldehyde, ammonium chloride (NH4Cl), and potassium cyanide (KCN) are used to make the aminonitrile; water is used to hydrolyze the aminonitrile to form the amino acid.
- Strecker synthesis is a condensation reaction (formation of an imine from a carbonyl-containing compound and ammonia, with loss of water), followed by nucleophilic addition (addition of the nitrile group), followed by hydrolysis.
- Gabriel synthesis begins with potassium phthalimide and diethyl bromomalonate, followed by an alkyl halide. Water is then used to hydrolyze the resulting compound to form the amino acid. While acids and bases are used at various times as catalysts, they are not main reactants.
- Gabriel synthesis proceeds through two SN2 reactions, hydrolysis, and decarboxylation.
**10.3**
- Inorganic phosphate contains a very negative charge. When bonded to other phosphate groups in a nucleotide triphosphate, this creates repulsion with adjacent phosphate groups, increasing the energy of the bond. Further, inorganic phosphate can be resonance-stabilized.
- Organic phosphates are carbon-containing molecules with phosphate groups; the most common examples are nucleotides, like those in DNA, ATP, or GTP.
- The three hydrogens in phosphoric acid have very different pKa values. This allows phosphoric acid to pick up or give off protons in a wide pH range, making it a good buffer over most of the pH scale.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. D
Glycine’s R group is a hydrogen atom; this amino acid is therefore achiral because the central carbon is not bonded to four different substituents. The other amino acids are all chiral and therefore have both L- and D-enantiomers.
2. B
This reaction is similar to the Gabriel synthesis. Phthalimide acts as a nucleophile, the methyl carbon acts as an electrophile, and bromide acts as the leaving group. Therefore, the reaction between methyl bromide and phthalimide results in the formation of methyl phthalimide. Subsequent hydrolysis then yields methylamine.
3. B
Cysteine is well known for containing a sulfur atom because it is able to form disulfide bridges; however, methionine also contains a sulfur atom in its R group.
4. B
An amide is formed from an amine and a carboxyl group or its acyl derivatives. In this question, an amine is already given; the compound to be identified must be an acyl compound. The only acyl compound among the choices given is a carboxylic acid.
5. C
During the Strecker synthesis, ammonia attacks a carbonyl, forming an imine, (B). This imine is attacked by cyanide, forming an amine, (D), and a nitrile, (A). Amide bonds are formed between amino acids but do not appear during the Strecker synthesis.
6. C
The Strecker synthesis creates an amino acid from an aldehyde. The carbonyl carbon ultimately becomes the α-carbon of the amino acid. Any remaining alkyl chain becomes the R group, as shown below. The starting compound is therefore 2-methylpropanal (isobutyraldehyde).
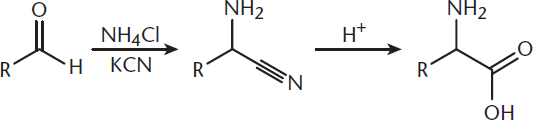
7. D
One resonance structure of a C–N bond in an amide has the double bond between the C and N, not between the C and O. Thus, the C–N bond of an amide has some sp2 character, and sp2-hybridized atoms exhibit planar geometry.
8. D
Both the Strecker and Gabriel syntheses contain planar intermediates, which can be attacked from either side by a nucleophile. This results in a racemic mixture of enantiomers, and the solution will therefore be optically inactive.
9. A
During the Gabriel synthesis, phthalimide attacks a secondary carbon in diethyl bromomalonate. The secondary carbon is the electrophile, (D), and bromide is the leaving group, (C).
10. C
The Gabriel synthesis includes two nucleophilic substitution steps, followed by hydrolysis and decarboxylation. Dehydration—the loss of a water molecule—is not a part of this reaction.
11. B
The pKa2 of phosphoric acid is close to physiological pH; therefore,
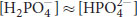
at this pH.
12. C
Pyrophosphate is unstable in aqueous solution and will degrade to form two equivalents of inorganic phosphate. The solvent is water, which should retain its polarity regardless of the presence of solutes, eliminating (D). Pyrophosphate and inorganic phosphate are small, charged molecules which are relatively soluble, eliminating (A).
13. B
The amino acid in question is aspartic acid, which is an acidic amino acid because it contains an extra carboxyl group. At neutral pH, both of the carboxyl groups are ionized, so there are two negative charges on the molecule. Only one of the charges is neutralized by the positive charge on the amino group, so the molecule has an overall negative charge.
14. A
As DNA is synthesized, it forms phosphodiester bonds, releasing pyrophosphate, PPi. Pyrophosphate is an inorganic phosphate-containing molecule, but it is not the single phosphate group commonly referred to as inorganic phosphate, (B). The DNA molecule itself is referred to as an organic phosphate, (D).
15. B
Phosphoric acid has three hydrogens with pKa values spread across the pH range. This allows some degree of buffering over almost the entire standard pH range from 0 to 14.
GO ONLINE
Consult your online resources for additional practice.
SHARED CONCEPTS
- Biochemistry Chapter 1
- Amino Acids, Peptides, and Proteins
- Biochemistry Chapter 6
- DNA and Biotechnology
- Biochemistry Chapter 9
- Carbohydrate Metabolism I
- General Chemistry Chapter 10
- Acids and Bases
- Organic Chemistry Chapter 4
- Analyzing Organic Reactions
- Organic Chemistry Chapter 8
- Carboxylic Acids