Chapter 3: Thermodynamics

Chapter 3: Thermodynamics
Science Mastery Assessment
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what’s important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don’t worry though— skipping something now does not mean you’ll never study it. Later on in your prep, as you complete full-length tests, you’ll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- If an object with an initial temperature of 300 K increases its temperature by 1 °C every minute, by how many degrees Fahrenheit will its temperature have increased in 10 minutes?
- 6°F
- 10°F
- 18°F
- 30 °C
- Which of the following choices correctly identifies the following three heat transfer processes?
- Heat transferred from the Sun to the Earth
- A metal spoon heating up when placed in a pot of hot soup
- A rising plume of smoke from a fire
- I. Radiation; II. Conduction; III. Convection
- I. Conduction; II. Radiation; III. Convection
- I. Radiation; II. Convection; III. Conduction
- I. Convection; II. Conduction; III. Radiation
- A 20 m steel rod at 10 °C is dangling from the edge of a building and is 2.5 cm from the ground. If the rod is heated to 110 °C, will the rod touch the ground? (Note: α = 1.1 × 10−5 K−1)
- Yes, because it expands by 3.2 cm.
- Yes, because it expands by 2.6 cm.
- No, because it expands by 2.2 cm.
- No, because it expands by 1.8 cm.
- What is the final temperature of a 3 kg wrought iron fireplace tool that is left in front of an electric heater, absorbing heat energy at a rate of 100 W for 10 minutes? Assume the tool is initially at 20 °C and that the specific heat of wrought iron is 500 Jkg⋅K.
- 40 °C
- 50 °C
- 60 °C
- 70 °C
- How much heat is required to completely melt a pair of gold earrings weighing 500 g, given that their initial temperature is 25 °C? (The melting point of gold is 1064 °C, its heat of fusion is 6.37×104 Jkg, and its specific heat is 126 Jkg⋅K.)
- 15 kJ
- 32 kJ
- 66 kJ
- 97 kJ
- Given the cycle shown, what is the total work done by the gas during the cycle?
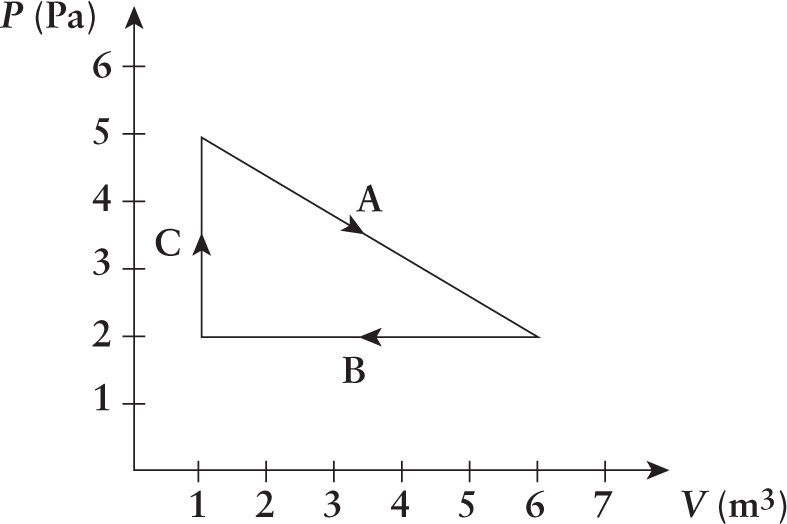
- –10 J
- 0 J
- 7.5 J
- 17.5 J
- In an adiabatic compression process, the internal energy of the gas:
- increases because the work done on the gas is negative.
- increases because the work done on the gas is positive.
- decreases because the work done on the gas is negative.
- decreases because the work done on the gas is positive.
- The entropy of a system can:
- never decrease.
- decrease when the entropy of the surroundings increases by at least as much.
- decrease when the system is isolated and the process is irreversible.
- decrease during an adiabatic reversible process.
- A student making a coffee cup calorimeter fails to use a second coffee cup and inadequately seals the lid. What was the student’s initial goal, and what was the result of this mistake?
- The student was trying to create an isolated system but created an open system instead.
- The student was trying to create an isolated system but created a closed system instead.
- The student was trying to create a closed system but created an open system instead.
- The student was trying to create a closed system but created an isolated system instead.
- A certain substance has a specific heat of 1 Jmol ⋅ K and a melting point of 350 K. If one mole of the substance is currently at a temperature of 349 K, how much energy must be added in order to melt it?
- More than 1 J
- Exactly 1 J
- Less than 1 J but more than 0 J
- Less than 0 J
- Which of the following is NOT a state function?
- Internal energy
- Heat
- Temperature
- Entropy
- The figure shown depicts a thick metal container with two compartments. Compartment A is full of a hot gas, while compartment B is full of a cold gas. What is the primary mode of heat transfer in this system?
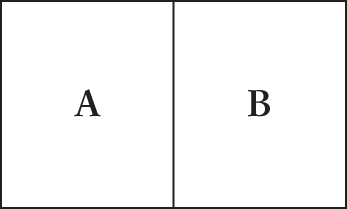
- Radiation
- Convection
- Conduction
- Enthalpy
- Substances A and B have the same freezing and boiling points. If solid samples of both substances are heated in the exact same way, substance A boils before substance B. Which of the following would NOT explain this phenomenon?
- Substance B has a higher specific heat.
- Substance B has a higher heat of vaporization.
- Substance B has a higher heat of fusion.
- Substance B has a higher internal energy.
- In experiment A, a student mixes ink with water and notices that the two liquids mix evenly. In experiment B, the student mixes oil with water; in this case, the liquids separate into two different layers. The entropy change is:
- positive in experiment A and negative in experiment B.
- positive in experiment A and zero in experiment B.
- negative in experiment A and positive in experiment B.
- zero in experiment A and negative in experiment B.
- Which of the following processes is LEAST likely to be accompanied by a change in temperature?
- The kinetic energy of a gas is increased through a chemical reaction.
- Energy is transferred to a solid via electromagnetic waves.
- A boiling liquid is heated on a hot plate.
- A warm gas is mixed with a cold gas.
Answer Key
- C
- A
- C
- C
- D
- C
- B
- B
- A
- A
- B
- C
- D
- B
- C
Chapter 3: Thermodynamics
CHAPTER 3
THERMODYNAMICS
In This Chapter
3.1 Zeroth Law of Thermodynamics
Temperature
Thermal Expansion
3.2 Systems
System Types
State Functions
3.3 First Law of Thermodynamics
Heat
Thermodynamic Processes
3.4 Second Law of Thermodynamics and Entropy
Energy Dispersion
Entropy
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 9% of all questions about physics on the MCAT.
This chapter covers material from the following AAMC content categories:
4B: Importance of fluids for the circulation of blood, gas movement, and gas exchange
5E: Principles of chemical thermodynamics and kinetics
Introduction
Thermodynamics is the study of the flow of energy in the universe, as that flow relates to work, heat, entropy, and the different forms of energy. Classical thermodynamics concerns itself only with observations that can be made at the macroscopic level, such as measurements of temperature, pressure, volume, and work. Although the MCAT will test entropy from a thermodynamic rather than probabilistic understanding, we will briefly discuss the statistical model of entropy because it clarifies much of the confusion that arises from a characterization of entropy as a measure of “disorder.”
This chapter reviews the laws of thermodynamics with a specific focus on the zeroth, first, and second laws. We will examine how the zeroth law leads to the formulation of temperature scales. Thermal expansion will be discussed as an example of the relationship between thermal energy and physical properties like length, volume, and conductivity. We will then take a moment to examine thermodynamic terminology and functions that are also intimately related to the concepts discussed in Chapter 7 of MCAT General Chemistry Review. In the context of the first law—the conservation of energy—we will discuss the relationship between internal energy, heat, and work, and characterize specific heat and heat of transformation. We will also review the various processes by which a system goes from one equilibrium state to another and connect work, discussed in Chapter 2 of MCAT Physics and Math Review, with heat. Finally, we will investigate the second law of thermodynamics through the concepts of entropy and its measurement. The third law of thermodynamics is not directly tested on the MCAT, so we will only briefly mention it.
3.1 Zeroth Law of Thermodynamics
LEARNING OBJECTIVES
After Chapter 3.1, you will be able to:
- Explain the zeroth law of thermodynamics
- Predict the relative expansion of an object undergoing a temperature change
- Describe the basis and the significance of the Kelvin scale
The zeroth law of thermodynamics is based on a simple observation: when one object is in thermal equilibrium with another object, say a cup of warm tea and a metal stirring stick, and the second object is in thermal equilibrium with a third object, such as your hand, then the first and third object are also in thermal equilibrium. As such, when brought into thermal contact, no net heat will flow between these objects. Note that thermal contact does not necessarily imply physical contact, as objects can be in thermal contact across space.
KEY CONCEPT
The zeroth law of thermodynamics states the transitive property in thermal systems: If a = b and b = c, then a = c.
Temperature
The formulation of the zeroth law—that no net heat flows between objects in thermal equilibrium, and the corollary that heat flows between two objects not in thermal equilibrium—actually arose from studies of temperature. At any given time, all substances have a particular temperature. In everyday language, we use the term temperature to describe qualitatively how hot or cold something is, but in thermodynamics, it has a more precise meaning. At the molecular level, temperature is proportional to the average kinetic energy of the particles that make up the substance. At the macroscopic level, it is the difference in temperature between two objects that determines the direction of heat flow. When possible, heat moves spontaneously from materials that have higher temperatures to materials that have lower temperatures. Heat itself refers to the transfer of thermal energy from a hotter object with higher temperature (energy) to a colder object with lower temperature (energy). If no net heat flows between two objects in thermal contact, then we can say that their temperatures are equal and they are in thermal equilibrium.
KEY CONCEPT
Temperature is a physical property of matter related to the average kinetic energy of the particles. Differences in temperature determine the direction of heat transfer.
Since the 18th century, scales have been developed to quantify the temperature of matter with thermometers. Some of these systems are still in common use, including the Fahrenheit (°F), Celsius (°C), and Kelvin (K) scales. Fahrenheit and Celsius are the oldest scales still in common use and are relatively convenient because they are based on the phase changes for water, as shown in Table 3.1. In the Celsius scale, 0° and 100° define the freezing and boiling temperatures of water. In the Fahrenheit scale, these phase change temperatures are defined as 32° and 212°.
Table 3.1 Relevant Points in the Three Major Temperature Scales
°F °C K Absolute Zero −460 −273 0
Freezing Point of Water 32 0 273
Boiling Point of Water 212 100 373
The Kelvin scale is most commonly used for scientific measurements and is one of the seven SI base units. It defines as the zero reference point absolute zero, the theoretical temperature at which there is no thermal energy, and sets the freezing point of water as 273.15 K. The third law of thermodynamics states that the entropy of a perfectly organized crystal at absolute zero is zero. Note that there are no negative temperatures on the Kelvin scale because it starts from absolute zero. Although the Kelvin and Celsius scales have different zero reference points, the size of their units is the same. That is to say, a change of one degree Celsius equals a change of one unit kelvin. Because there are 180 degrees between water’s phase changes on the Fahrenheit scale, rather than 100 degrees as on both the Celsius and the Kelvin scales, the size of the Fahrenheit unit is smaller. The following formulas can be used to convert from one scale to another:
F=95C+32K=C+273
Equation 3.1
where F, C, and K are the temperatures in Fahrenheit, Celsius, and Kelvin, respectively.
MCAT EXPERTISE
The only time Fahrenheit is used routinely on the MCAT is for body temperature, which is 98.6°F or 37 °C. In the rare occasion that it is used for a quantitative analysis question, conversions will be given.
Example: If a meteorologist says that the temperature will reach a high of 86°F today, what will be the high temperature in °C and in K?
Solution: To convert from Fahrenheit to Celsius, use:
F=95C+32→C=59(F−32)C=59(86−32)=59(54)=30 °C
Now convert from Celsius to Kelvin:
K = C + 273 = 30 + 273 = 303 K
Thermal Expansion
It has long been noted that some physical properties of matter change when the matter gets hotter or colder. Length, volume, solubility, and even the conductivity of matter change as a function of temperature. The relationship between temperature and a physical property of some matter was used to develop the temperature scales with which we are familiar today. For example, Daniel Fahrenheit developed the temperature scale that bears his name by placing a thermometer filled with mercury into a bath of ice, water, and ammonium chloride. The cold temperature caused the mercury to contract, and when the level in the glass tube stabilized at a lower level, he marked this as the zero reference on the scale. He then placed the same mercury thermometer in a mixture of ice and water (that is, at the freezing point for water). The slightly warmer temperature of this mixture caused the mercury to rise in the glass column, and when it stabilized at this higher level, Fahrenheit assigned it a value of 32°. When he stuck the thermometer under his (or someone else’s) tongue, he marked the even higher mercury level as 100° (not 98.6°). The details of how and why Fahrenheit came to choose these numbers (and the history of their adjustment since Fahrenheit first developed the scale) are beyond the scope of this discussion; rather, what is important to note is that a change in some physical property of one kind of matter—in this case, the height of a column of mercury—can be correlated to certain temperature markers, such as the phase changes for water. Once the scale has been set in reference to the decided-upon temperature markers, then the thermometer can be used to take the temperature of any other matter, in accordance with the zeroth law.
REAL WORLD
It is because of thermal expansion that bridges and sidewalks have gaps between their segments; they allow for thermal expansion without damaging integrity.
MNEMONIC
When the temperature of an object changes, its length changes a lot (αLΔT).
Because the property of thermal expansion was integral to the development of thermometers, let’s look a little more closely at this phenomenon. A change in the temperature of most solids results in a change in their length. Rising temperatures cause an increase in length, and falling temperatures cause a decrease in length. The amount of length change is proportional to the original length of the solid and the increase in temperature according to the equation
ΔL = αLΔT
Equation 3.2
where ΔL is the change in length, α is the coefficient of linear expansion, L is the original length, and ΔT is the change in temperature. The coefficient of linear expansion is a constant that characterizes how a specific material’s length changes as the temperature changes. This usually has units of K–1, although it may sometimes be quoted as °C–1. This difference is inconsequential because the unit size for the Kelvin and Celsius scales is the same.
Example: A metal rod of length 2 m has a coefficient of linear expansion of 10−6 K−1. It is cooled from 1080 °C to 80 °C. What is the final length of the rod?
Solution: By using the information given in the problem, we can substitute directly into the thermal expansion formula:
ΔL = αLΔT = (10−6 K)(2 m)(80 K − 1080 K) = −2 × 10−3 m
The negative sign represents a decrease in length. The original length was 2 m; therefore, the final length is 2 − (2 × 10−3) = 1.998 m.
Liquids also experience thermal expansion, but the only meaningful parameter of expansion is volume expansion. The formula for volumetric thermal expansion is applicable to both liquids and solids:
ΔV = βVΔT
Equation 3.3
where ΔV is the change in volume, β is the coefficient of volumetric expansion, V is the original volume, and ΔT is the change in temperature. The coefficient of volumetric expansion is a constant that characterizes how a specific material’s volume changes as the temperature changes. Its value is equal to three times the coefficient of linear expansion for the same material (β = 3α).
Example: Suppose that a thermometer with 1 mL of mercury is taken from a freezer with a temperature of −25 °C and placed near an oven at 275 °C. If the coefficient of volume expansion of mercury is 1.8 × 10−4 K−1, by how much will the liquid expand?
Solution: Use the information given to plug into the volumetric expansion formula:
ΔV=βVΔT=(1.8×10−4K−1)(1mL)(275 °C−(−25 °C))=(1.8×10−4)(300)=540×10−4mL=0.054mL
MCAT CONCEPT CHECK 3.1
Before you move on, assess your understanding of the material with these questions.
- What is the zeroth law of thermodynamics?
_________________________________
_________________________________
_________________________________
- What is the maximum distance that two objects can be from one another and still adhere to the zeroth law of thermodynamics?
_________________________________
- How do the initial length of an object and the amount it expands for a given temperature change relate to one another?
_________________________________
_________________________________
- True or False: The Kelvin scale is the most accurate measurement method for temperature because it is based on absolute zero.
3.2 Systems
LEARNING OBJECTIVES
After Chapter 3.2, you will be able to:
- Distinguish between closed, isolated, and open thermodynamic systems
- Compare and contrast state and process functions
- List common state functions
Physicists and chemists tend to classify the world on the basis of observable phenomena and interactions between objects. Before moving on, we need to become familiar with some of the jargon that these fields have in common—specifically, thermodynamic systems and state functions. Note that the same jargon is discussed in Chapter 7 of MCAT General Chemistry Review.
System Types
A system is the portion of the universe that we are interested in observing or manipulating. The rest of the universe is considered the surroundings.
Isolated Systems
Isolated systems are not capable of exchanging energy or matter with their surroundings. As a result, the total change in internal energy must be zero. Isolated systems are very rare, although they can be approximated. A bomb calorimeter attempts to insulate a reaction from the surroundings to prevent energy transfer, and the entire universe can be considered an isolated system because there are no surroundings.
Closed Systems
Closed systems are capable of exchanging energy, but not matter, with the surroundings. The classic experiments involving gases in vessels with movable pistons are examples of closed systems. For thermodynamic purposes, most of what will be encountered on Test Day will be a closed system or will approximate a closed system.
Open Systems
Open systems can exchange both matter and energy with the environment. In an open system, not only does the matter carry energy, but more energy may be transferred in the form of heat or work. A boiling pot of water, human beings, and uncontained combustion reactions are all examples of open systems.
State Functions
State functions are thermodynamic properties that are a function of only the current equilibrium state of a system. In other words, state functions are defined by the fact that they are independent of the path taken to get to a particular equilibrium state. The state functions include pressure (P), density (ρ), temperature (T), volume (V), enthalpy (H), internal energy (U), Gibbs free energy (G), and entropy (S). On the other hand, process functions, such as work and heat, describe the path taken to get to from one state to another.
MCAT CONCEPT CHECK 3.2
Before you move on, assess your understanding of the material with these questions.
- Which of the following thermodynamic systems transfer matter? Transfer energy?
System Type Transfers Matter Transfers Energy Closed Isolated Open
- What is the difference between a state function and a process function?
- State function:
_________________________________
- Process function:
_________________________________
- List at least five common state functions:
- _________________________________
- _________________________________
- _________________________________
- _________________________________
- _________________________________
3.3 First Law of Thermodynamics
LEARNING OBJECTIVES
After Chapter 3.3, you will be able to:
- Recall the mathematical relationships between internal energy, work, and heat
- Describe conduction, convection, and radiation
- Draw a graph of the temperature of a solid as it is heated to a gas
- Calculate work for a P–V diagram:
We have already encountered the first law of thermodynamics in our discussion of the conservation of mechanical energy in Chapter 2 of MCAT Physics and Math Review. Remember that in the absence of nonconservative forces, the sum of kinetic and potential energies is constant in a system. Now, in our present discussion of thermodynamics, we will look more closely at the relationship between internal energy, heat, and work. Essentially, the first law of thermodynamics states that the change in the total internal energy of a system is equal to the amount of energy transferred in the form of heat to the system, minus the amount of energy transferred from the system in the form of work. The internal energy of a system can be increased by adding heat, doing work on the system, or some combination of both processes. The change in internal energy is calculated from the equation
ΔU = Q − W
Equation 3.4
where ΔU is the change in the system’s internal energy, Q is the energy transferred into the system as heat, and W is the work done by the system. To use this equation properly, one must carefully apply the following sign convention shown in Table 3.2.
Table 3.2 Sign Convention for the First Law of Thermodynamics
VARIABLE POSITIVE VALUE NEGATIVE VALUE
**Change in Internal Energy (ΔU)** Increasing temperature Decreasing temperature
**Heat (Q)** Heat flows into system Heat flows out of system
**Work (W)** Work is done by the system (expansion) Work is done on the system (compression)
KEY CONCEPT
The first law of thermodynamics tells us that an increase in the total internal energy of a system is caused by transferring heat into the system or performing work on the system. The total internal energy of a system will decrease when heat is lost from the system or work is performed by the system.
The first law is really just a particular iteration of the more universal physical law of energy conservation: energy can be neither created nor destroyed; it can only be changed from one form to another. Because the first law accounts for all work and all heat processes impacting the system, the presence of nonconservative forces poses no problem because the energy transfer associated with friction, air resistance, or viscous drag will be accounted for in the first law equation. For example, when a car “burns rubber,” all the smoke and noise coming from the back tires is a clear indication that mechanical energy is not being conserved. However, if we include the energy transfers associated with the frictional forces in our consideration of the change in internal energy of the system, then we can confidently say that no energy has been lost at all: there may be a “loss” of energy from the car as a result of the friction, but that precise amount of energy can be “found” elsewhere—as thermal energy in the atoms and molecules of the surrounding road and air.
Heat
In Chapter 2 of MCAT Physics and Math Review, we defined work as the process by which energy is transferred as the result of force being applied through some distance. We noted that work and heat are the only two processes by which energy can be transferred from one object to another. As discussed earlier in this chapter, the zeroth law of thermodynamics says that objects in thermal contact are in thermal equilibrium when their temperatures are the same. The corollary of this is the second law of thermodynamics: objects in thermal contact and not in thermal equilibrium will exchange heat energy such that the object with a higher temperature will give off heat energy to the object with a lower temperature until both objects have the same temperature at thermal equilibrium. Heat, then, is defined as the process by which a quantity of energy is transferred between two objects as a result of a difference in temperature. As we will discuss further in our examination of the second law, heat can never spontaneously transfer energy from a cooler object to a warmer one without work being done on the system.
KEY CONCEPT
Heat is the process of energy transfer between two objects at different temperatures and will continue until the two objects come into thermal equilibrium at the same temperature.
The SI unit for heat is the joule (J), which should not be surprising because it is based on energy. Heat can also be measured in the units of calorie (cal), nutritional Calorie (Cal), or the British thermal unit (BTU). The nutritional Calorie (“big C”) is not the same thing as the calorie (“little c”); one Calorie is equal to 1000 calories or 1 kcal.
KEY CONCEPT
One calorie (little c) is the amount of heat required to raise 1 g of water one degree Celsius. One Calorie (big C) is the amount of heat required to raise 1 kg of water 1 degree Celsius, equal to 1000 calories.
The conversion factors between the units of heat are as follows:
1 Cal ≡ 103 cal = 4184 J = 3.97 BTU
Heat Transfer
For energy to be transferred between objects, they must be in thermal contact with each other. This does not necessarily mean that the objects are touching. Like force, energy can travel tremendous distances and does not require a medium to pass through. There are three means by which heat can transfer energy: conduction, convection, and radiation.
Conduction is the direct transfer of energy from molecule to molecule through molecular collisions. As this definition would suggest, there must be direct physical contact between the objects. At the atomic level, the particles of the hotter matter transfer some of their kinetic energy to the particles of the cooler matter through collisions between the particles of the two materials. Metals are described as the best heat conductors because metallic bonds contain a density of atoms embedded in a sea of electrons, which facilitate rapid energy transfer. Gases tend to be the poorest heat conductors because there is so much space between individual molecules that energy-transferring collisions occur relatively infrequently. An example of heat transfer through conduction is the heat that is rapidly, and painfully, conducted to your fingers when you touch a hot stove.
Convection is the transfer of heat by the physical motion of a fluid over a material. Because convection involves flow, only liquids and gases can transfer heat by this means. In convection, if the fluid has a higher temperature, it will transfer energy to the material. Most restaurants and some home kitchens have convection ovens, which use fans to circulate hot air inside the oven. Because the heat is being transferred to the food by both convection and radiation rather than only by radiation, convection ovens cook more rapidly than radiation-only ovens. Convection may also be used to wick heat energy away from a hot object. In laboratory experiments, for example, a running cold-water bath may be used to rapidly cool a reaction.
Radiation is the transfer of energy by electromagnetic waves. Unlike conduction and convection, radiation can transfer energy through a vacuum. Radiation is the method by which the Sun is able to warm the Earth. Many home kitchens have radiant ovens, which use either electrical coils or gas flames to heat the insulated metal box that forms the body of the oven. The hot metal box then radiates the energy through the open space of the oven, where it is absorbed by whatever food is placed inside.
Specific Heat
When heat energy is added to or removed from a system, the temperature of that system will change in proportion to the amount of heat transfer, unless the system is undergoing a phase change during which the temperature is constant. This relationship between heat and temperature for a substance is called the specific heat. The specific heat (c) of a substance is defined as the amount of heat energy required to raise one gram of a substance by one degree Celsius or one unit kelvin. For example, the specific heat of liquid water is one calorie per gram per unit kelvin (1 calg⋅K). Equivalently, this can be expressed as 4.184 Jg⋅K. The specific heat for a substance changes according to its phase. The MCAT will generally provide specific heat values as necessary, although you are expected to know the specific heat of water in calories. The equation that relates the heat gained or lost by an object and the change in temperature of that object is
q = mcΔT
Equation 3.5
MCAT EXPERTISE
The specific heat of water (in calories) is a constant you are expected to know for Test Day. Its value is 1 calg⋅K.
MNEMONIC
The equation for heat transfer, given a specific heat, is the same as the test you’re studying for! q = mcΔT looks a lot like “q equals MCAT.”
where m is the mass, c is the specific heat of the substance, and ΔT is the change in temperature (in Celsius or kelvins). Because the unit size for the Celsius and Kelvin scales is the same, the change in temperature will be the same for temperatures measured in Celsius or kelvins.
Heat of Transformation
When a substance is undergoing a phase change, such as from solid to liquid or liquid to gas, the heat that is added or removed from the system does not result in a change in temperature. In other words, phase changes occur at a constant temperature, and the temperature will not begin to change until all of the substance has been converted from one phase into the other. For example, water melts at 0 °C. No matter how much heat is added to a mass of ice at 0 °C, the temperature of the equilibrated system will not rise until all the ice has been melted into liquid water.
We’ve determined that adding heat raises the temperature of a system because the particles in that system now have a greater average kinetic energy, and it’s true that molecules have greater degrees of freedom of movement in the liquid state than in the solid state (and even more so in the gas state). However, phase changes are related to changes in potential energy, not kinetic energy. The molecules of water in ice, for example, aren’t truly frozen in place and unable to move. The molecules rotate, vibrate, and wiggle around. The bonds within each molecule are also free to bend and stretch. Of course, the molecules are held in relatively stable positions by the hydrogen bonds that form between them, but they still have a fairly significant amount of kinetic energy. The potential energy, however, is quite low because of the stability provided by the relative closeness of one molecule to another and by the hydrogen bonds.
Now, think about what happens when one adds heat to ice that is at 0 °C. The heat energy causes the water molecules to begin to move away from each other by breaking free of the hydrogen bonds between them. Because the water molecules are being held less rigidly in place, they now have greater degrees of freedom of movement and their average potential energy increases. In statistical mechanics, one would say that this increased freedom of movement permits a greater number of microstates for the water molecules. For example, instead of only being able to move up and down or sway side-to-side, a water molecule may now have more freedom of movement and be able to rock forward and back. In gaining additional directions and forms of motion, however, the amount of up-and-down or side-to-side motion must decrease, thus keeping the average kinetic energy of liquid water at 0 °C the same as solid water at 0 °C. In summary, while liquid water may have a greater number of microstates due to increased freedom of movement, its average kinetic energy is the same as solid water at the same temperature.
When heat energy is added to or removed from a system that is experiencing a phase change, the amount of heat that is added or removed cannot be calculated with the equation q = mcΔT because there is no temperature change during a phase change. Instead, the following equation is used:
q = mL
Equation 3.6
where q is the amount of heat gained or lost from the material, m is the mass of the substance, and L is the heat of transformation or latent heat of the substance.
BRIDGE
It is important to know the common terms used for phase changes:
- Solid to liquid: fusion or melting
- Liquid to solid: freezing or solidification
- Liquid to gas: boiling, evaporation, or vaporization
- Gas to liquid: condensation
- Solid to gas: sublimation
- Gas to solid: deposition
These phase changes are discussed in Chapter 7 of MCAT General Chemistry Review.
The phase change from liquid to solid (freezing or solidification) or solid to liquid (melting or fusion) occurs at the melting point. The corresponding heat of transformation is called the heat of fusion. The phase change from liquid to gas (boiling, evaporation, or vaporization) or gas to liquid (condensation) occurs at the boiling point. The corresponding heat of transformation is called the heat of vaporization. The relevant heats of fusion and vaporization will be provided on Test Day.
REAL WORLD
It is because of the heat of transformation that sweating is such an efficient cooling mechanism. When sweat evaporates, the heat of vaporization is absorbed from the surface of the body. This is also why a hot day seems so much more intense when it is very humid out. The sweat is less likely to evaporate due to the dampness of the environment, so less heat can be lost from the surface of the skin through sweating.
Example: Silver has a melting point of 962 °C and a heat of fusion of approximately 1.05×105 Jkg. The specific heat of silver is 233 Jkg⋅K. Approximately how much heat is required to completely melt a 1 kg silver chain with an initial temperature of 20 °C?
Solution: Before melting the chain, we must first heat the chain to the melting point. To figure out how much heat is required, we use this formula:
q=mcΔT=(1 kg)(233 Jkg ⋅ K)(942 K)≈200×975=1.95×105 J(actual=2.19×105J)
This tells us we have to add 219 kJ of heat to the chain just to get its temperature to the melting point. The chain is still in the solid phase. To melt it, we must continue to add heat in accordance with this formula:
q=mL=(1 kg)(1.05×105 Jkg)=1.05×105 J
The total heat needed to melt the solid silver chain is 219 kJ + 105 kJ = 324 kJ.
Thermodynamic Processes
In the last chapter, we gave significant consideration to work as a change of energy in a system, both as a function of force and displacement and as a function of volume and pressure. We will briefly review the latter and its relationship to heat transfer within a system. Keep in mind that work accomplished by a change in displacement is not likely to be motivated by heat transfer, and any heat transfer that does occur is most likely a result of friction dissipating mechanical energy from the system.
During any thermodynamic process, a system goes from some initial equilibrium state with an initial pressure, temperature, and volume to some other equilibrium state, which may be at a different final pressure, temperature, or volume. These thermodynamic processes can be represented in graphical form with volume on the x-axis and pressure (or temperature) on the y-axis.
Table 3.3 Special Types of Thermodynamic Processes
PROCESS FIRST LAW OF THERMODYNAMICS REDUCES TO:
Isothermal (ΔU = 0) Q = W
Adiabatic (Q = 0) ΔU = −W
Isobaric (constant pressure) (Multiple possible forms)
Isovolumetric (isochoric) (W = 0) ΔU = Q
The MCAT focuses on three particular thermodynamic processes as special cases of the first law (excluding isobaric processes), as shown in Table 3.3. In each of these cases, some physical property is held constant during the process. These processes are isothermal (constant temperature, and therefore no change in internal energy), adiabatic (no heat exchange), and isovolumetric (no change in volume, and therefore no work accomplished; also called isochoric). Isobaric processes are those that occur at a constant pressure, and are of less focus on the MCAT. Figure 3.1 shows the different types of thermodynamic behaviors for a gas.
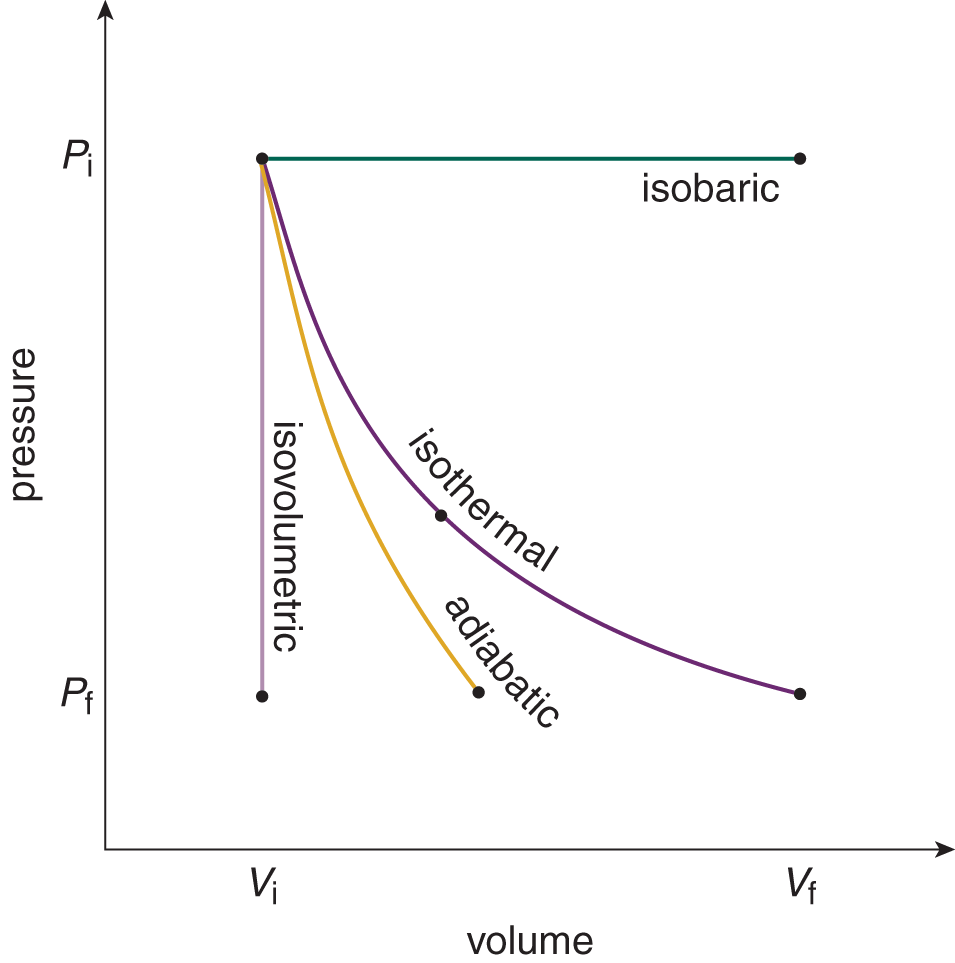
Figure 3.1. Thermodynamic Behaviors of Gases
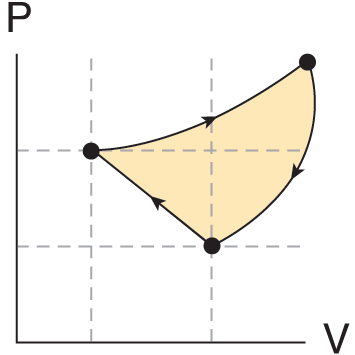
Figure 3.2 shows a closed-loop thermodynamic process. Because the work on a P–V graph is simply the area under the curve, the work done in this closed-loop process is the area inside the loop.
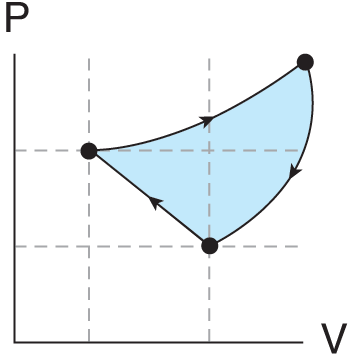
Figure 3.2. A Closed-Loop Process The work is the area inside the loop.
Example: A gas in a cylinder is kept at a constant pressure of 3.6 × 105 Pa while 300 kJ of heat are added to it, causing the gas to expand from 1.0 m3 to 1.5 m3. Find the work done by the gas and the change in internal energy of the gas.
Solution: The pressure is held constant through the entire process so the work can be found using the equation:
W=PΔV=(3.6×105 Pa)(1.5 m3−1.0 m3)=1.8×105 J
The change in internal energy can be found from the first law of thermodynamics:
ΔU=Q−W=(3×105)−(1.8×105 J)=1.2×105 J=120 kJ
MCAT CONCEPT CHECK 3.3
Before you move on, assess your understanding of the material with these questions.
- Describe the relationship between internal energy, work, and heat in words.
_________________________________
- Define the following forms of heat transfer:
- Conduction:
_________________________________
- Convection:
_________________________________
- Radiation:
_________________________________
- Draw a representative graph of the temperature of a solid object as it is heated and goes through two phase changes to become a gas.
- How is work calculated in P–V diagrams?
_________________________________
3.4 Second Law of Thermodynamics and Entropy
LEARNING OBJECTIVES
After Chapter 3.4, you will be able to:
- Describe entropy, on both a macroscopic level and in statistical terms
- Explain the relationship between entropy of a system and entropy of the surroundings for any thermodynamic process
The second law of thermodynamics states that objects in thermal contact and not in thermal equilibrium will exchange heat energy such that the object with a higher temperature will give off heat energy to the object with a lower temperature until both objects have the same temperature at thermal equilibrium. As such, energy is constantly being dispersed.
Energy Dispersion
Consider each of the following scenarios: hot tea cooling down, a frozen drink melting, iron rusting, buildings crumbling, balloons deflating, and living things dying and decaying. These scenarios share a common denominator. In each of them, energy of some form is going from being localized or concentrated to being spread out or dispersed. The thermal energy in the hot tea is spreading out to the cooler air that surrounds it. The thermal energy in the warmer air is spreading out to the cooler frozen drink. The chemical energy in the bonds of elemental iron and oxygen is released and dispersed as a result of the formation of the more stable, lower-energy bonds of iron oxide (rust). The potential energy of the building is released and dispersed in the form of light, sound, and heat as the building crumbles and falls. The energy of the pressurized air is released to the surrounding atmosphere as the balloon deflates. The chemical energy of all the molecules and atoms in living flesh is released into the environment during the process of death and decay.
Entropy
The second law of thermodynamics states that energy spontaneously disperses from being localized to becoming spread out if it is not hindered from doing so. Pay attention to this: the usual way of thinking about entropy as “disorder” must not be taken too literally, a trap that many students fall into. Be very careful in thinking about entropy as disorder. The old analogy between a messy (disordered) room and entropy is deficient and may not only hinder understanding but actually increase confusion.
Entropy is the measure of the spontaneous dispersal of energy at a specific temperature: how much energy is spread out, or how widely spread out energy becomes in a process. In the discussion of microstates earlier, we considered that when ice melts, the freedom of movement of the water molecules increases. If the water remains at the melting point, it will have the same average kinetic energy as molecules of ice; the difference between the two is the number of available microstates. That is, while both water and ice at 0 °C have the same kinetic energy, the energy is dispersed over a larger number of microstates in liquid water. Liquid water therefore has higher entropy and, by extension, it is indeed less organized than ice. The equation for calculating the change in entropy is:
ΔS=QrevT
Equation 3.7
where ΔS is the change in entropy, Qrev is the heat that is gained or lost in a reversible process, and T is the temperature in kelvin. The units of entropy are usually Jmol⋅K. When energy is distributed into a system at a given temperature, its entropy increases. When energy is distributed out of a system at a given temperature, its entropy decreases.
Example: If, in a reversible process, 5.46 × 104 J of heat is used to change a 200 g block of ice to water at a temperature of 273 K, what is the change in the entropy of the system? (Note: The heat of fusion of ice = 333 Jg.)
Solution: We know that during a phase change, the temperature is constant; in this case, 273 K. From the information given:
ΔS=QrevT=5.46×104 J273 K=200 JK
The amount of heat added did not exceed the amount needed to completely melt the block of ice, as calculated by:
q=mL=(200 g)(333 Jg)=6.66×104 J
Therefore, no heat was applied to change the temperature of the resulting liquid and T remained constant.
Notice that the second law states that energy will spontaneously disperse; it does not say that energy can never be localized or concentrated. However, the concentration of energy will not happen spontaneously in a closed system. Work usually must be done to concentrate energy. For example, refrigerators work against the direction of spontaneous heat flow (that is, they counteract the flow of heat from the “warm” exterior of the refrigerator to the “cool” interior), thereby “concentrating” energy outside of the system in the surroundings. As a result, refrigerators consume a lot of energy to accomplish this movement of energy against the temperature gradient.
The second law has been described as time’s arrow because there is a unidirectional limitation on the movement of energy by which we recognize before and after or new and old. For example, you would instantly recognize whether a video recording of an explosion was running forward or backward. Another way of understanding this is to say that energy in a closed system will spontaneously spread out and entropy will increase if it is not hindered from doing so. Remember that a system can be variably defined to include the entire universe; in fact, the second law ultimately claims that the entropy of the universe is increasing.
ΔSuniverse = ΔSsystem + ΔSsurroundings > 0
Equation 3.8
KEY CONCEPT
The universe is a closed, expanding system, so you know that the entropy of the universe is always increasing. The more space that appears with the expansion of the universe, the more space there is for the entire universe’s energy to be distributed and the total entropy of the universe to increase irreversibly.
When describing processes, physicists often use terms such as natural, unnatural, reversible, or irreversible. These terms may sometimes be confusing but they are descriptive of observable phenomena. For example, we expect that when a hot object is brought into thermal contact with a cold object, the hot object will transfer heat energy to the cold object until both are in thermal equilibrium (that is, at the same temperature). This is a natural process and also one that we would describe as irreversible: we are not surprised that the two objects eventually reach a common temperature, but we would be shocked if all of a sudden the hot object became hotter and the cold object became colder. This would be an unnatural process.
To define a reversible reaction, let’s consider a system of ice and liquid water in equilibrium at 0 °C. If we place this mixture of ice and liquid water into a thermostat (device for regulating temperature) that is also at 0 °C and allow infinitesimal amounts of heat to be absorbed by the ice from the thermostat so that the ice melts to liquid water at 0 °C and the thermostat remains at 0 °C, then the increase in the entropy of the system (the water) will be exactly equal to the entropy decrease of the surroundings (the thermostat). The net change in the entropy of the system and its surroundings is zero. Under these conditions, the process is reversible. The key to a reversible reaction is making sure that the process goes so slowly—requiring an infinite amount of time—that the system is always in equilibrium and no energy is lost or dissipated. To be frank, no real processes are reversible; we can only approximate a reversible process. Note how physicists define reversible processes: These are processes that can spontaneously reverse course. For example, while water can be put through cycles of freezing and melting innumerable times, ice melting on the warm countertop would not be expected to suddenly freeze if it remains in the warm environment. The liquid water will need to be placed in an environment that is cold enough to cause the water to freeze, and once frozen in the cold environment, the ice would not be expected to begin melting spontaneously. The freezing and melting of water in real life are therefore irreversible processes in physics while still being chemically reversible.
MCAT CONCEPT CHECK 3.4
Before you move on, assess your understanding of the material with these questions.
- Describe entropy on a macroscopic level and in statistical terms.
- Macroscopic:
_________________________________
_________________________________
- Statistical:
_________________________________
_________________________________
- What is the relationship between the entropy of a system and its surroundings for any thermodynamic process?
_________________________________
_________________________________
Conclusion
This chapter reviewed the zeroth law of thermodynamics, which reflects the observation that objects at the same temperature are in thermal equilibrium and the net heat exchanged between them is zero. We may consider the zeroth law to be ex post facto (after the fact) because it provides the thermodynamic explanation for the function of thermometers and temperature scales, which had been developed many years prior to the law’s formulation. We then took some time to define basic thermodynamic terms for systems and state functions. Examination of the first law of thermodynamics revealed that the energy of a closed system (up to and including the universe) is constant, such that the total internal energy of a system (the sum of all its potential and motional energies) equals the heat gained by the system minus the work done by the system. Finally, we carefully investigated the second law of thermodynamics and the concept of entropy. We understand entropy as a measure not only of “disorder” but of the degree to which energy is spread out through a system, up to and including the universe. We now understand that the constant energy of the universe is progressively and irreversibly spreading out and will continue to spread out until there is an even distribution of energy throughout the universe. Many of these concepts will make a reappearance throughout our discussions of general chemistry, and will certainly be seen on the MCAT. In the next chapter, we’ll investigate fluids, the final mechanical concept for Test Day.
GO ONLINE
You’ve reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Zeroth Law of Thermod ynamics
- The zeroth law of thermodynamics states that objects are in thermal equilibrium when they are at the same temperature.
- Objects in thermal equilibrium experience no net exchange of heat energy.
- Temperature is a qualitative measure of how hot or cold an object is; quantitatively, it is related to the average kinetic energy of the particles that make up a substance.
- Thermal expansion describes how a substance changes in length or volume as a function of the change in temperature.
Systems
- A thermodynamic system is the portion of the universe that we are interested in observing, whereas the surroundings include everything that is not part of the system.
- Isolated systems do not exchange matter or energy with the surroundings.
- Closed systems exchange energy but not matter with their surroundings.
- Open systems exchange both energy and matter with their surroundings.
- State functions are pathway independent and are not themselves defined by a process. Pressure, density, temperature, volume, enthalpy, internal energy, Gibbs free energy, and entropy are all state functions.
- Process functions describe the pathway from one equilibrium state to another. Work and heat are process functions.
First Law of Thermodynamics
- The first law of thermodynamics is a statement of conservation of energy: the total energy in the universe can never decrease or increase.
- For a closed system, the total internal energy is equal to the heat flow into the system minus the work done by the system.
- Heat is the process of energy transfer between two objects at different temperatures that occurs until the two objects come into thermal equilibrium (reach the same temperature).
- Specific heat is the amount of energy necessary to raise one gram of a substance by one degree Celsius or one kelvin.
- The specific heat of water is 1calg⋅K.
- During a phase change, heat energy causes changes in the particles’ potential energy and energy distribution (entropy), but not kinetic energy. Therefore, there is no change in temperature. This is the heat of transformation.
- There are four special types of thermodynamic systems in which a given variable is held constant:
- For isothermal processes, the temperature is constant, and the change in internal energy is therefore 0.
- For adiabatic processes, no heat is exchanged.
- For isobaric processes, the pressure is held constant.
- For isovolumetric (isochoric) processes, the volume is held constant and the work done by or on the system is 0.
Second Law of Thermodynamics and Entropy
- The second law of thermodynamics states that in a closed system (up to and including the entire universe), energy will spontaneously and irreversibly go from being localized to being spread out (dispersed).
- Entropy is a measure of how much energy has spread out or how spread out energy has become.
- On a statistical level, as the number of available microstates increases, the potential energy of a molecule is distributed over that larger number of microstates, increasing entropy.
- Every natural process is ultimately irreversible; under highly controlled conditions, certain equilibrium processes such as phase changes can be treated as essentially reversible.
ANSWERS TO CONCEPT CHECKS
**3.1**
- The zeroth law of thermodynamics states that when two objects are both in thermal equilibrium with a third object, they are in thermal equilibrium with each other. By extension, no heat flows between two objects in thermal equilibrium.
- While there may be a distance at which thermal equilibrium is impractical, there is no theoretical maximum distance. As long as two objects are in thermal contact and at the same temperature, they are in thermal equilibrium.
- Expansion is a result of an increase in dimension at all points along an object. If an object is initially longer, it will experience a greater expansion. This is also represented in the formula for thermal expansion because there is a direct relationship between length change and the initial length of an object.
- False. As we will discuss in Chapter 11 of MCAT Physics and Math Review, accuracy is related to an instrument, rather than the scale. In addition, Kelvin uses the same scale as Celsius, so there are no practical differences in terms of accuracy.
**3.2**
-
System Type Transfers Matter Transfers Energy Closed No Yes
Isolated No No
Open Yes Yes
- State functions are variables independent from the path taken to achieve a particular equilibrium and are properties of a given system at equilibrium; they may be dependent on one another. Process functions define the path (or how the system got to its state) through variables such as Q (heat) or W (work).
- State functions include pressure (P), density (ρ), temperature (T), volume (V), enthalpy (H), internal energy (U), Gibbs free energy (G), and entropy (S).
**3.3**
- The change in the internal energy of a system is equal to heat put into a system minus the work done by the system. This is the first law of thermodynamics.
- Conduction is heat exchange by direct molecular interactions. Convection is heat exchange by fluid movement. Radiation is heat exchange by electromagnetic waves, and does not depend on matter.
-
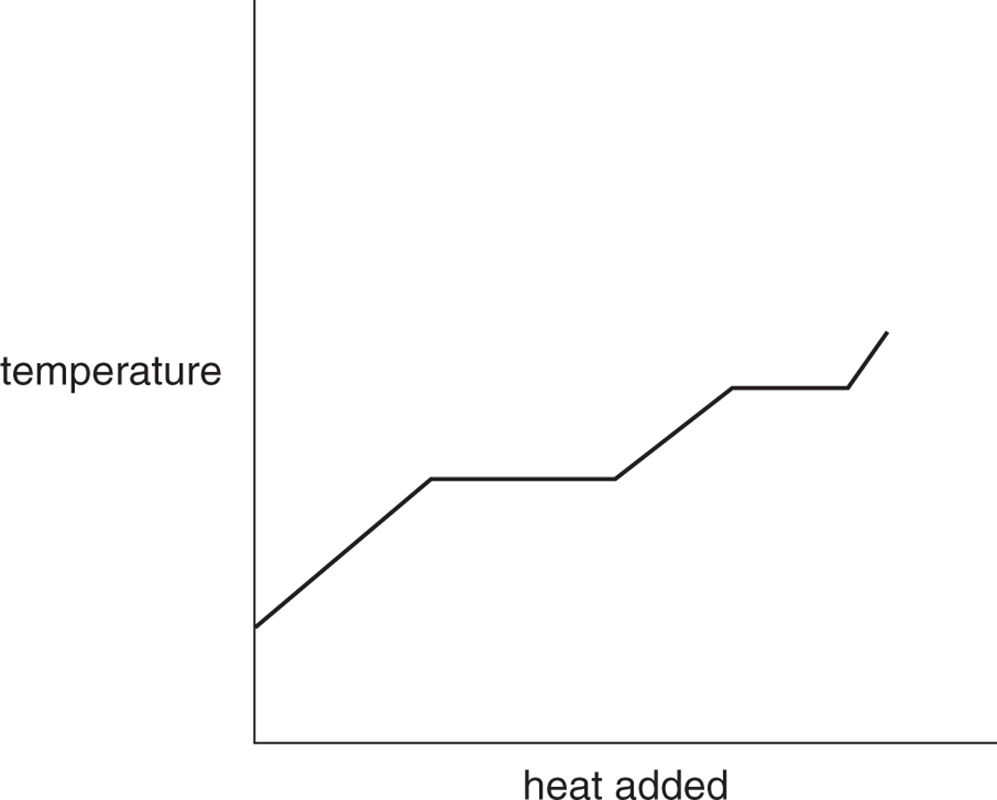
- In a P–V graph, work is the area under the curve (or within a closed loop).
**3.4**
- On a macroscopic level, entropy can be thought of as the tendency toward disorder. Statistically, entropy is the measure of the spontaneous dispersal of energy at a specific temperature, increasing the number of available microstates for a given molecule.
- The entropy of a system and its surroundings will never decrease; entropy will always either remain constant or increase.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. C
The kelvin unit and Celsius degree are the same size; that is, a change of 10 K is equal to a change of 10 °C. One degree Celsius is equal to 1.8 degrees Fahrenheit; therefore, 10 °C = 18°F.
2. A
Because there is essentially only empty space between the Sun and the Earth, the only means of heat transfer is by radiation—electromagnetic waves that propagate across space. When a metal spoon is placed in a pot of hot soup, the molecules in the soup collide with those on the surface of the spoon, thereby transferring heat by conduction. Finally, fire warms the air above it, and the warmed air is less dense than the surrounding air, so it rises. A rising column of warm air means that heat is being transported in the air mass, which is simply the process of convection. The smoke particles ride along with the upward moving air current and create a plume of smoke.
3. C
The magnitude of one degree Celsius equals the magnitude of one unit kelvin. Therefore, a change of 100 °C is equal to a change of 100 K. We can then find the change in length due to thermal expansion:
ΔL=αLΔT=(1.1×10−5 K−1)(20 m)(100 K)=0.022 m=2.2 cm
Because the rod is originally 2.5 cm above the ground and its length increases by 2.2 cm, we can conclude that it will not touch the ground after the thermal expansion process is completed.
4. C
To answer this question, first remember that watts are equal to joules per second; in other words, power is energy transfer over time. In 10 minutes, the tool absorbs the following amount of energy:
E=P×t=(100 W)(10 min)(60 s1 min)=6×104 J
Now we can find the final temperature from this equation:
q=mcΔT6×104 J=(3 kg)(500 Jkg⋅K)ΔT40°C=ΔT
The final temperature is thus 20 + 40 = 60 °C.
5. D
First determine how much heat is required to raise the temperature of the gold earrings to the melting point of gold. Then, calculate the heat required to actually melt the earrings (the latent heat). The total heat required to melt the earrings completely will be the sum of the two heats. The heat required to raise the temperature of the earrings from 25 °C to 1064 °C is
q=mcΔT=(0.5 kg)(126 Jkg ⋅ K)(1039 K)≈60×1000=60 kJ
Thus, it takes about 60 kJ of heat to bring the earrings to their melting point. The next step is to calculate how much heat is needed to melt the earrings. For this, use the heat of fusion (the latent heat) of gold:
q=mL=(0.5 kg)(6.37×104 Jkg)=32 kJ
So overall, it requires approximately 60 + 32 = 92 kJ of heat to melt the gold earrings. Notice that we can heavily approximate the numbers used in our calculations because the answer choices are so spread out. The closest answer is (D).
6. C
The total work done by the cycle is the sum of the work of paths A, B, and C, or the area within the cycle. Because the area bounded by A, B, and C is a triangle with a base of 5 m3 and a height of 3 Pa, we can calculate the area as 12(5 m3)(3 Pa)=7.5 J. Clockwise loops tend to do positive work on the environment, while counterclockwise loops do negative work.
7. B
To answer the question, make sure you understand all the terms. An adiabatic process means that there is no exchange of heat; in other words, Q = 0. When a gas is compressed, positive work is being done on the gas (rather than by the gas), so the value for work done by the gas will be negative (W < 0). Based on this, we can determine how the internal energy of the gas changes by using the first law of thermodynamics (ΔU = Q − W). If Q = 0 and W is negative, then ΔU is positive.
8. B
The entropy of a system can decrease as long as the entropy of its surroundings increases by at least as much. On the other hand, the entropy of an isolated system increases for all real (irreversible) processes. This adheres to the second law of thermodynamics, which says that energy will be dispersed and entropy of the universe will remain constant or increase during all processes.
9. A
Calorimeters are our best approximations of isolated systems, where neither energy nor matter is exchanged with the environment. By failing to use an insulating layer and failing to fully contain the system, heat can be exchanged with the environment and matter may be dispersed, creating an open system.
10. A
To find the amount of heat needed to bring the substance to its melting point, you can use the specific heat. To heat one mole of the substance one unit kelvin, it would take 1 J of heat. After the substance reaches its melting point, additional heat is needed to actually induce the phase change. Therefore, the total amount of heat required is greater than 1 J.
11. B
State functions are any that are independent of the path taken to achieve a given state and which are not themselves defined as a process, such as pressure, density, temperature, volume, enthalpy, internal energy, Gibbs free energy, and entropy. Heat and work are process functions that are pathway dependent.
12. C
In this situation, heat will transfer from the warm gas to the metal and then to the cold gas. Convection requires flow of a fluid to cause heat transfer, invalidating (B) as an answer. In this case, the gas is not flowing, but rather is in contact with the metal. (A) is an invalid answer because heat transfer through radiation is also implausible not only because gases are unlikely to emit heat in the form of waves but also because the radiation would be unlikely to penetrate the thick metal container. Enthalpy, (D), is not a form of heat transfer. Conduction, (C), is the most likely option; it happens when two substances make direct contact with one another. Here, gas A makes contact with the metal container, which makes contact with gas B.
13. D
Saying that substance B has a higher internal energy cannot explain the phenomenon because the internal energy is irrelevant; the heat involved in the process is related only to the specific heat, the heat of fusion, and the heat of vaporization. All of the other choices could explain the phenomenon. The heat required to melt the solid is determined by the heat of fusion, (C). The heat required to bring the liquid to its boiling point is determined by the specific heat, (A). The heat required to boil the liquid is determined by the heat of vaporization, (B).
14. B
When the ink randomly intersperses throughout the water, the final state is more disordered than the initial state, so the entropy change of the system is positive. When the oil separates from the water, the final state is just as ordered as the initial state (because the oil and the water are still completely separate), so the entropy change is zero. You can also answer this question by noticing the reversibility of the two experiments. Experiment A has a positive entropy change because it is irreversible, while experiment B has no entropy change because the reaction is reversible. According to the second law of thermodynamics, the overall entropy change of a system and its surroundings can never be negative in a thermodynamic process that moves from one equilibrium state to another.
15. C
If a substance is undergoing a phase change, any added heat will be used toward overcoming the heat of transformation of the phase change. During the phase change, the temperature will remain constant. Temperature is a measure of the kinetic energy of the molecules in a sample, so a change in kinetic energy, (A), is essentially the same thing as a change in temperature. The heat transfer by radiation described in (B) will definitely change the temperature of the solid as long as it is not in the process of melting. (D) Describes heat transfer by convection, in which the warm gas will transfer heat to the cold gas until they both reach an intermediate temperature.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(3.1) Temperature conversions: F=95C+32K=C+273
(3.2) Thermal expansion equation: ΔL = αLΔT
(3.3) Volume expansion equation: ΔV = βVΔT
(3.4) First law of thermodynamics: ΔU = Q – W
(3.5) Heat gained or lost (with temperature change): q = mcΔT
(3.6) Heat gained or lost (phase change): q = mL
(3.7) Entropy and heat: ΔS=QrevT
(3.8) Second law of thermodynamics: ΔSuniverse = ΔSsystem + ΔSsurroundings > 0
SHARED CONCEPTS
Biochemistry Chapter 12
Bioenergetics and Regulation of Metabolism
General Chemistry Chapter 6
Equilibrium
General Chemistry Chapter 7
Thermochemistry
General Chemistry Chapter 8
The Gas Phase
General Chemistry Chapter 12
Electrochemistry
Physics and Math Chapter 2
Work and Energy