Chapter 9: Atomic and Nuclear Phenomena

Chapter 9: Atomic and Nuclear Phenomena
Science Mastery Assessment
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what’s important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don’t worry though— skipping something now does not mean you’ll never study it. Later on in your prep, as you complete full-length tests, you’ll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- If the work function of a metal is 6.622 × 10–20 J and a ray of electromagnetic radiation with a frequency of 1.0 × 1014 Hz is incident on the metal, what will be the speed of the electrons ejected from the metal? (Note: h = 6.626 × 10−34 J·s and *m**e*− = 9.1 × 10−31 kg)
- 2.62×10−6 ms
- 1.07×10−4 ms
- 9.38×103 ms
- 3.81×105 ms
- What is the wavelength of a photon that causes an electron to be emitted from a metal with a kinetic energy of 50 J? (Note: The work function of the metal is 16 J, and h = 6.626 × 10−34 J·s)
- 1.0 × 10−34 m
- 3.0 × 10−27 m
- 3.0 × 10−26 m
- 1.0 × 1035 m
- Which of the following statements is inconsistent with the Bohr model of the atom?
- Energy levels of the electron are stable and discrete.
- An electron emits or absorbs radiation only when making a transition from one energy level to another.
- To jump from a lower energy to a higher energy orbit, an electron must absorb a photon of precisely the right frequency such that the photon’s energy equals the energy difference between the two orbits.
- To jump from a higher energy to a lower energy orbit, an electron absorbs a photon of a frequency such that the photon’s energy is exactly the energy difference between the two orbits.
- When a hydrogen atom electron falls to the ground state from the n = 2 state, 10.2 eV of energy is emitted. What is the wavelength of this radiation? (Note: 1 eV = 1.60 × 10−19 J, and h = 6.626 × 10−34 J·s)
- 5.76 × 10−9 m
- 1.22 × 10−7 m
- 3.45 × 10−7 m
- 2.5 × 1015 m
- The figure below illustrates an electron with initial energy of –10 eV moving from point A to point B. What change accompanies the movement of the electron?
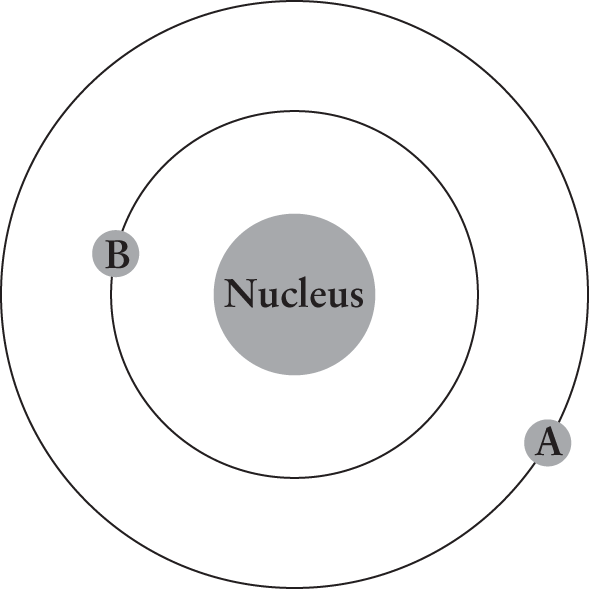
- Absorption of a photon
- Emission of a photon
- Decrease in the atom’s work function
- Increase in the atom’s total energy
- Which of the following four fundamental forces is primarily responsible for holding the nucleons together?
- Binding energy
- Strong nuclear force
- Electrostatic force
- Gravitational force
- All of the following statements about the photoelectric effect are true EXCEPT:
- the intensity of the light beam does not affect the photocurrent.
- the kinetic energies of the emitted electrons do not depend on the light intensity.
- a weak beam of light of frequency greater than the threshold frequency yields more current than an intense beam of light of frequency lower than the threshold frequency.
- for light of a given frequency, the kinetic energy of emitted electrons increases as the value of the work function decreases.
- What is the binding energy of the argon-40 isotope in MeV? (Note: mproton = 1.0073 amu, mneutron = 1.0087 amu, mAr-40 nucleus = 39.9132 amu, c2=932MeVamu)
- 0.4096 MeV
- 40.3228 MeV
- 381.7 MeV
- 643.8 MeV
- Which of the following correctly identifies the following process?
G3167a+e−→Z3067n
- β− decay
- β+ decay
- e− capture
- γ decay
- Consider the following fission reaction.
n011.0087+B51010.0129→L37i7.0160+H24e4.0026
The masses of the species involved are given in atomic mass units below each species, and 1 amu can create 932 MeV of energy. What is the energy liberated due to transformation of mass into energy during this reaction?
- 0.003 MeV
- 1.4 MeV
- 2.8 MeV
- 5.6 MeV
- Element X is radioactive and decays via α decay with a half-life of four days. If 12.5 percent of an original sample of element X remains after n days, what is the value of n ?
- 4
- 8
- 12
- 16
- A graph of an exponential decay process is created. The y-axis is the natural logarithm of the ratio of the number of intact nuclei at a given time to the number of intact nuclei at time t = 0. The x-axis is time. What does the slope of such a graph represent?
- λ
- − λ
- e−λt
- nn0
- A certain carbon nucleus dissociates completely into α-particles. How many particles are formed?
- 1
- 2
- 3
- 4
- The half-life of carbon-14 is approximately 5,730 years, while the half-life of carbon-12 is essentially infinite. If the ratio of carbon-14 to carbon-12 in a certain sample is 25% less than the normal ratio in nature, how old is the sample?
- Less than 5,730 years
- Approximately 5,730 years
- Significantly greater than 5,730 years, but less than 11,460 years
- Approximately 11,460 years
- A nuclide undergoes two alpha decays, two positron decays, and two gamma decays. What is the difference between the atomic number of the parent nuclide and the atomic number of the daughter nuclide?
- 0
- 2
- 4
- 6
Answer Key
- C
- B
- D
- B
- B
- B
- A
- C
- C
- C
- C
- B
- C
- A
- D
Chapter 9: Atomic and Nuclear Phenomena
CHAPTER 9
ATOMIC AND NUCLEAR PHENOMENA
In This Chapter
9.1 The Photoelectric Effect
Threshold Frequency
Kinetic Energy of Ejected Electrons
9.2 Absorption and Emission of Light 9.3 Nuclear Binding Energy and Mass Defect 9.4 Nuclear Reactions
Fusion
Fission
Radioactive Decay
Concept Summary
CHAPTER PROFILE
The content in this chapter should be relevant to about 16% of all questions about physics on the MCAT.
This chapter covers material from the following AAMC content categories:
4D: How light and sound interact with matter
4E: Atoms, nuclear decay, electronic structure, and atomic chemical behavior
Introduction
All of life depends on the photoelectric effect. As a photon of light enters the chloroplast in a plant cell, it reacts with chlorophyll, causing the ejection of an electron from certain magnesium-containing dyes. This electron feeds into synthetic pathways that ultimately result in glucose production. While the MCAT does not include photosynthesis in its content lists, its principles are a primary example of the photoelectric effect. It was Albert Einstein who described this effect, and it was this that won him the Nobel Prize—not the theory of relativity. We now use the photoelectric effect in many industrial applications, such as solar panels.
After discussing the photoelectric effect, we will examine nuclear radiation. Nuclear radiation is curiously full of opposites: it can cause life-threatening diseases such as cancer, but it can also be used in the treatment of cancer. It can be used safely for mass power generation, but it can cause untold devastation in meltdowns or weapons of mass destruction. In addition to nuclear radiation, we will examine the strong nuclear force and the equation of mass defect, perhaps the most quoted equation in all of science. At the end of this chapter, we’ll have covered all of the physics content tested on the MCAT, and will be ready to move on to mathematics and some skills-based practice.
9.1 The Photoelectric Effect
LEARNING OBJECTIVES
After Chapter 9.1, you will be able to:
- Relate the work function to the energy needed to emit an electron from a metal
- Recall the factor(s) that threshold frequency depends on
- Recognize the phenomena that result from the application of the photoelectric effect:
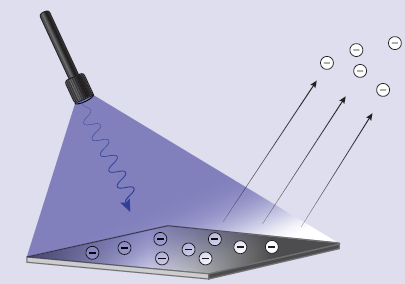
When light of a sufficiently high frequency (typically, blue to ultraviolet light) is incident on a metal in a vacuum, the metal atoms emit electrons. This phenomenon, discovered by Heinrich Hertz in 1887, is called the photoelectric effect. As mentioned earlier, Albert Einstein’s 1905 explanation of the photoelectric effect won him the Nobel Prize.
Electrons liberated from the metal by the photoelectric effect will produce a net charge flow per unit time, or current. Provided that the light beam’s frequency is above the threshold frequency of the metal, light beams of greater intensity produce larger current in this way. The higher the intensity of the light beam, the greater the number of photons per unit time that fall on an electrode, producing a greater number of electrons per unit time liberated from the metal. When the light’s frequency is above the threshold frequency, the magnitude of the resulting current is directly proportional to the intensity (and amplitude) of the light beam.
Threshold Frequency
The minimum frequency of light that causes ejection of electrons is known as the threshold frequency, fT. The threshold frequency depends on the type of metal being exposed to the radiation. The photoelectric effect is, for all intents and purposes, an “all-or-nothing” response: if the frequency of the incident photon is less than the threshold frequency (f < fT), then no electron will be ejected because the photons do not have sufficient energy to dislodge the electron from its atom. But if the frequency of the incident photon is greater than the threshold frequency (f > fT), then an electron will be ejected, and the maximum kinetic energy of the ejected electron will be equal to the difference between hf and hfT (also called the work function). Einstein’s explanation of these results was that the light beam consists of an integral number of light quanta called photons. The energy of each photon is proportional to the frequency of the light:
E = hf
Equation 9.1
where E is the energy of the photon of light, h is Planck’s constant (6.626 × 10−34 J·s), and f is the frequency of the light. Once we know the frequency, we can easily find the wavelength λ according to the equation c = fλ, as described in Chapter 8 of MCAT Physics and Math Review. According to these equations, waves with higher frequency have shorter wavelengths and higher energy (toward the blue and ultraviolet end of the spectrum); waves with lower frequency have longer wavelengths and lower energy (toward the red and infrared end of the spectrum). In nuclear physics, wavelength is commonly measured in nanometers (1 nm = 10−9 m) and ångströms (1 Å = 10−10 m).
KEY CONCEPT
The energy of a photon increases with increasing frequency. The reason that we only discuss electrons being ejected from metals (and not protons or neutrons) is because of the weak hold that metals have on their valence electrons due to their low ionization energies.
Kinetic Energy of Ejected Electrons
If the frequency of a photon of light incident on a metal is at the threshold frequency for the metal, the electron barely escapes from the metal. However, if the frequency of an incident photon is above the threshold frequency of the metal, the photon will have more than enough energy to eject a single electron, and the excess energy will be converted to kinetic energy in the ejected electron. We can calculate the maximum kinetic energy of the ejected electron with the formula:
Kmax = hf − W
Equation 9.2
where W is the work function of the metal in question. The work function is the minimum energy required to eject an electron and is related to the threshold frequency of that metal by:
W = hfT
Equation 9.3
These formulas solve for the maximum kinetic energy of the electron rather than exact kinetic energy because the actual energy can be anywhere between 0 and Kmax, depending on the specific subatomic interactions between the photon and the metal atom. Kmax is only achieved when all possible energy from the photon is transferred to the ejected electron.
MCAT EXPERTISE
The photoelectric effect is not frequently tested on the MCAT, but the underlying principles are simple. This is simply another example of energy transfer in which light energy causes an increase in electrical potential energy in the atom—enough to allow the electron to escape. If any energy is “left over,” it cannot be destroyed. Rather, it is transferred into kinetic energy in the ejected electron.
BRIDGE
Think of the work function like activation energy, in the sense that it must be matched or exceeded to cause the reaction (escape of an electron) to occur. Activation energy is discussed in Chapter 5 of MCAT General Chemistry Review.
Example: If blue light of frequency 6.00 × 1014 Hz is incident on rubidium (W = 2.26 eV), will there be photoejection of electrons? If so, what is the maximum kinetic energy that an ejected electron will carry away? (Note: h = 6.626 × 10−34 J·s = 4.14 × 10−15 eV · s)
Solution: If the photons have a frequency of 6.00 × 1014 Hz, each photon has an energy of:
E = hf = (4.14 × 10−15 eV · s)(6.00 × 1014 Hz) = 2.48 eV
Clearly then, any given photon has more than enough energy to allow an electron in the metal to overcome the 2.26 eV barrier. In fact, the maximum excess kinetic energy carried away by the electron turns out to be:
K = hf − W = 2.48 − 2.26 = 0.22 eV
In general, the photoelectric effect is strong support for the particle theory of light, which states that light is not a continuous wave but acts as discrete bundles of energy called photons, as shown in Figure 9.1.
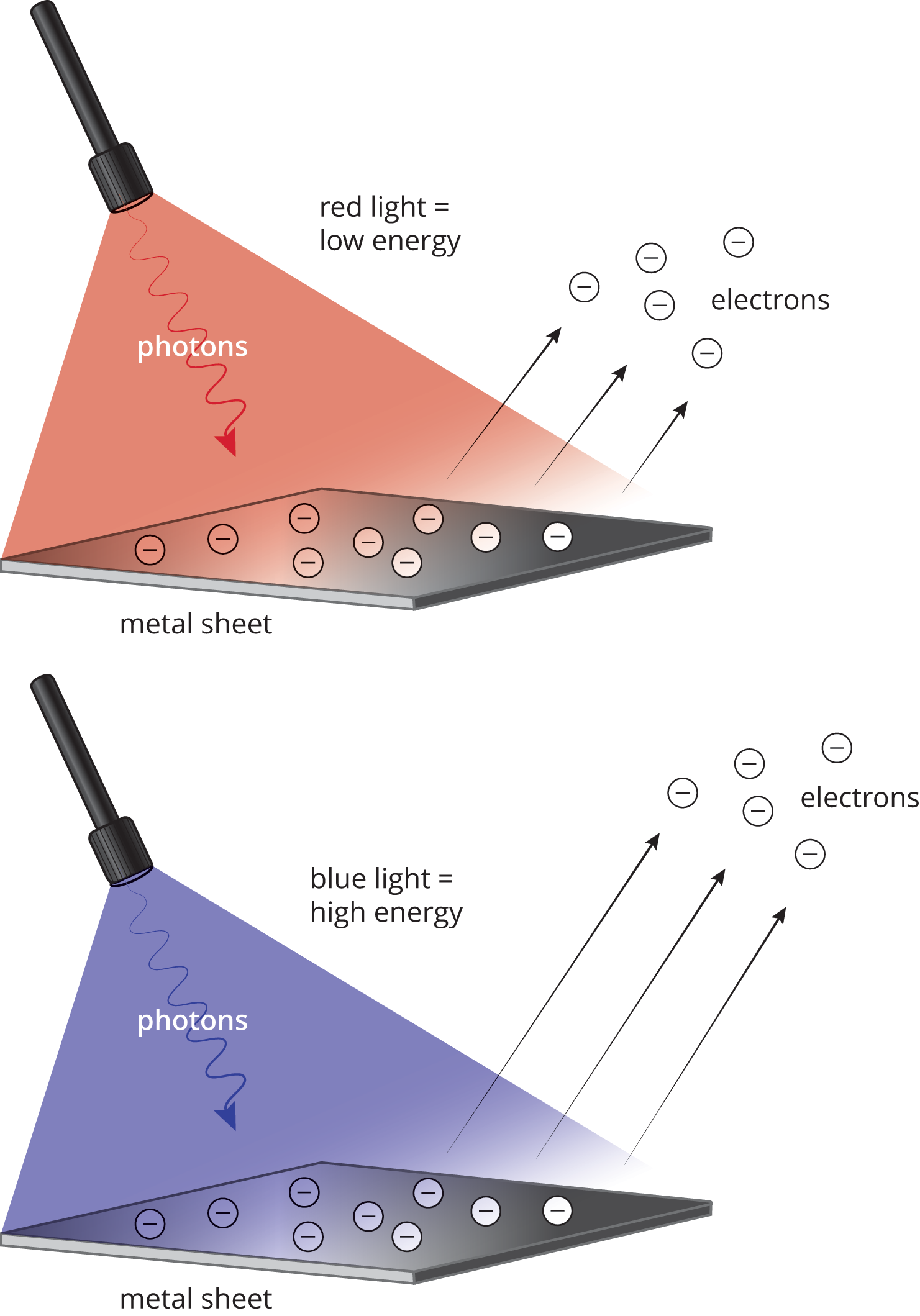
Figure 9.1. The Photoelectric Effect
MCAT CONCEPT CHECK 9.1
Before you move on, assess your understanding of the material with these questions.
- How does the work function relate to the energy necessary to emit an electron from a metal?
_________________________________
- What does the threshold frequency depend upon?
_________________________________
- What electrical phenomenon results from the application of the photoelectric effect?
_________________________________
9.2 Absorption and Emission of Light
LEARNING OBJECTIVES
After Chapter 9.2, you will be able to:
- Describe how the absorption spectrum of a single atom is determined
- Recall when photon emissions are most common during electronic transitions
- Explain the phenomenon of fluorescence
In Chapter 1 of MCAT General Chemistry Review, we explored the Bohr model of the atom. As a reminder, the Bohr model states that electron energy levels are stable and discrete, corresponding to specific orbits. An electron can jump from a lower-energy to a higher-energy orbit by absorbing a photon of light of precisely the right frequency to match the energy difference between the orbits (E = hf). If a photon does not carry enough energy, then the electron cannot jump to a higher energy level. When an electron falls from a higher-energy level to a lower-energy level, a photon of light is emitted with an energy equal to the energy difference between the two orbits. These processes of atomic absorption and emission are shown in Figure 9.2.
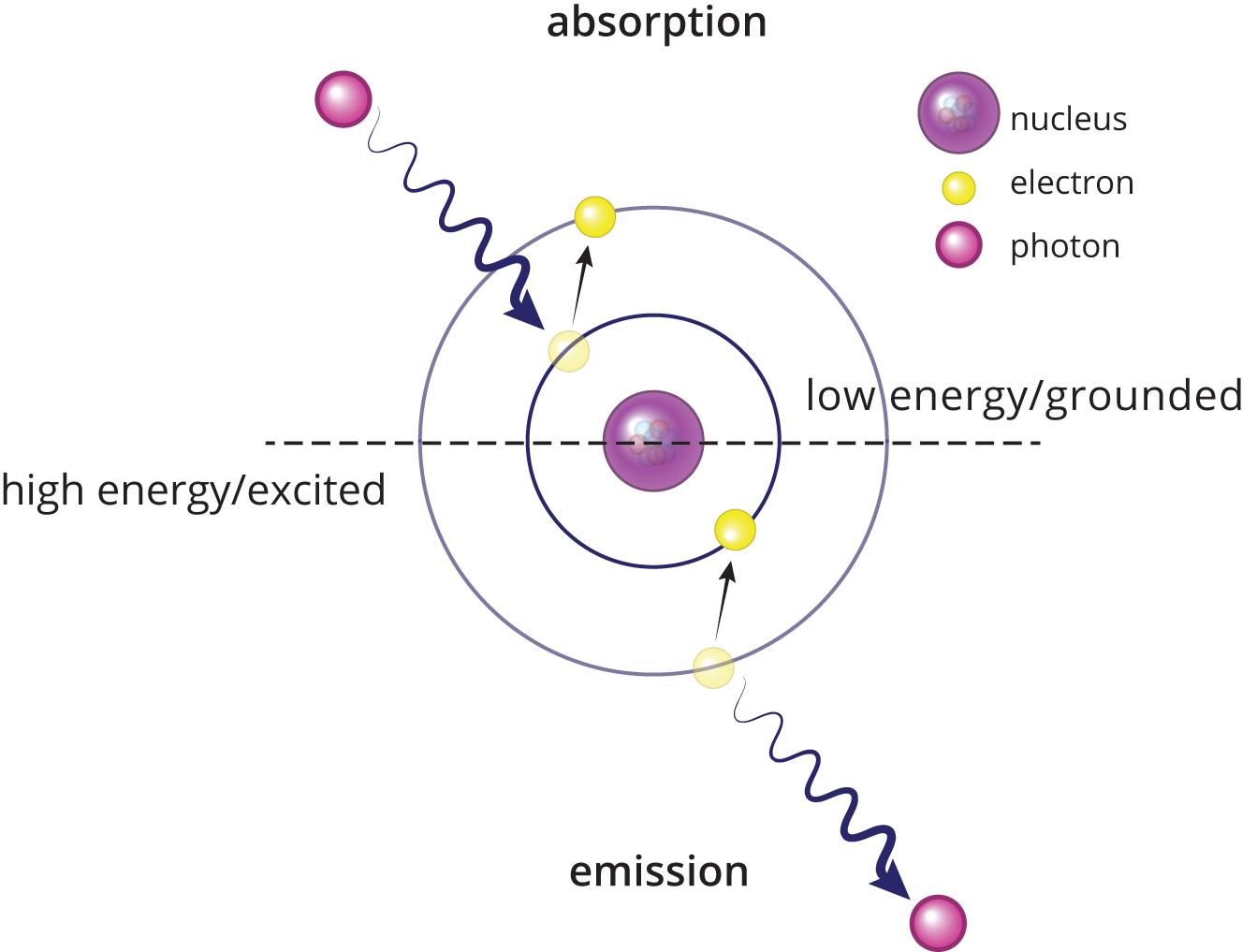
Figure 9.2. Bohr Model: Light Absorption and Emission
While information about a single electron is a great foundation for Test Day, in the real world we’ll often be handling more complex structures. In organic chemistry, we use infrared (IR) spectroscopy to determine chemical structure because different bonds will absorb different wavelengths of light. UV–Vis spectroscopy takes this one step further, looking at the absorption of light in the visible and ultraviolet range. Absorption spectra may be represented as a color bar with peak areas of absorption represented by black lines. It can also be shown as a graph with the absolute absorption as a function of wavelength. This is shown in Figure 9.3, which shows the absorption spectrum for the atmosphere across the entire electromagnetic spectrum.
BRIDGE
Spectroscopy is discussed in greater detail in Chapter 11 of MCAT Organic Chemistry Review.
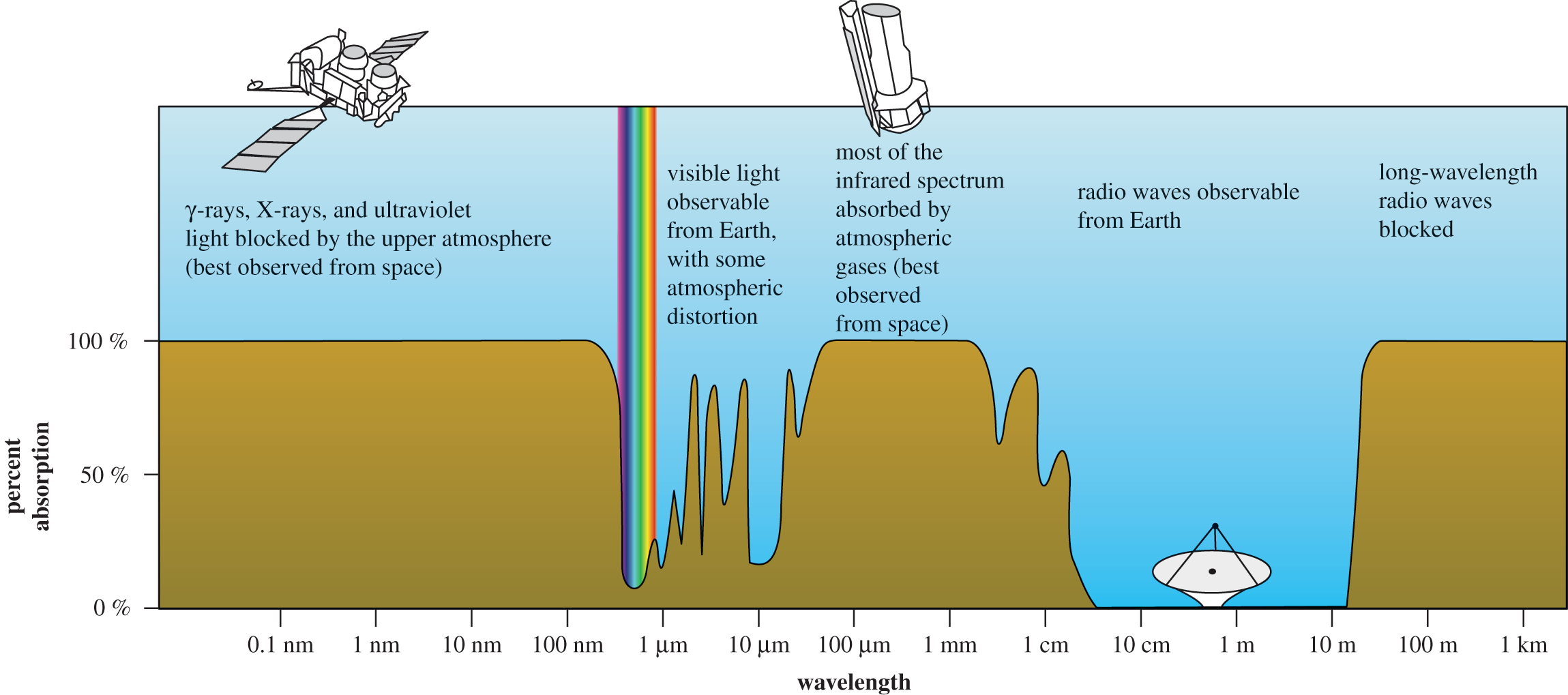
Figure 9.3. Absorption Spectrum of the Atmosphere The sky is blue because blue light is the least absorbed by atmospheric gases.
Changes in molecular structure can cause dramatic shifts in the absorption patterns of a substance. Consider indicators like phenolphthalein. This indicator has a clear appearance in its acidic state, and thus does not absorb any visible light. In its basic state, it is a bright pink, and thus is absorbing all but the longer wavelengths of visible light—remember that we see the colors that are not absorbed. Most indicators contain large organic compounds that have strikingly different absorption patterns based solely on the protonation state of the compound. These compounds often have conjugated double bonds or aromatic ring systems, as this permits the absorption of light from photons in the visible range.
Another phenomenon related to absorption and emission of visible light is fluorescence. If one excites a fluorescent substance (such as a ruby, an emerald, or the phosphors found in fluorescent lights) with ultraviolet radiation, it will begin to glow with visible light. Photons of ultraviolet light have relatively high frequencies (short wavelengths). After being excited to a higher energy state by ultraviolet radiation, the electron in the fluorescent substance returns to its original state in two or more steps. By returning in two or more steps, each step involves less energy, so at each step, a photon is emitted with a lower frequency (longer wavelength) than the absorbed ultraviolet photon. If the wavelength of this emitted photon is within the visible range of the electromagnetic spectrum, it will be seen as light of the particular color corresponding to that wavelength. The wide range of colors in fluorescent lights, from the whitish-green of office lighting to the glaring colors of neon signs, is the result of the distinct multi-step emission spectra of different fluorescent materials.
MCAT CONCEPT CHECK 9.2
Before you move on, assess your understanding of the material with these questions.
- What determines the absorption spectrum of a single atom?
_________________________________
- True or False: Small changes in chemical structure only minimally impact light absorption and emission patterns.
- During which electronic transitions is photon emission most common?
_________________________________
- What causes fluorescence?
_________________________________
9.3 Nuclear Binding Energy and Mass Defect
LEARNING OBJECTIVES
After Chapter 9.3, you will be able to:
- Describe key concepts in nuclear binding energy, including strong nuclear force, mass defect, and binding energy
- Recall the four fundamental forces of nature
- Apply the equation E = mc2
Until this point, we’ve examined the relationships between electromagnetic radiation and matter—particularly electrons. Now, we’ll shift to the energy that is stored in the nucleus, which can be emitted under specific circumstances. While one would assume that the mass of the nucleus is simply the sum of the masses of all of the protons and neutrons within it, the actual mass of every nucleus (other than hydrogen) is slightly smaller than that. This difference is called the mass defect. Scientists had difficulty explaining why this mass defect occurred until Einstein characterized the equivalence of matter and energy, embodied by the equation
E = mc2
Equation 9.4
where E is energy, m is mass, and c is the speed of light. The mass defect is a result of matter that has been converted to energy. Because of the large exponent on the speed of light—which is squared in the equation—a very small amount of mass will yield a huge amount of energy. For example, the conversion of one gram of mass to energy will produce 89.9 terajoules (1 TJ = 1012 joules) or 21.5 billion kilocalories.
When protons and neutrons (nucleons) come together to form the nucleus, they are attracted to each other by the strong nuclear force, which is strong enough to more than compensate for the repulsive electromagnetic force between the protons. Although the strong nuclear force is the strongest of the four fundamental forces, it only acts over extremely short distances, less than a few times the diameter of a proton or neutron. The nucleons have to get very close together in order for the strong nuclear force to hold them together. The bonded system is at a lower energy level than the unbonded constituents, and this difference in energy must be radiated away in the form of heat, light, or other electromagnetic radiation before the mass defect becomes apparent. This energy, called binding energy, allows the nucleons to bind together in the nucleus. Given the strength of the strong nuclear force, the amount of mass that is transformed into the dissipated energy will be a measurable fraction of the initial total mass. The binding energy per nucleon peaks at the element iron, which implies that iron contains the most stable nucleus. In general, intermediate-sized nuclei are more stable than very large or small nuclei.
The weak nuclear force also contributes to the stability of the nucleus, but is about one-millionth as strong as the strong nuclear force. The strong and weak nuclear forces constitute two of the four fundamental forces of nature. The other two are electrostatic forces and gravitation.
Example: Measurements of the atomic mass of a neutron and a proton yield these results:
proton=1.00728 amuneutron=1.00867 amu
H24e contains two protons and two neutrons, which should theoretically give a helium nucleus a mass of 2 × 1.00728 + 2 × 1.00867 = 4.03190 amu. However, the true mass of the helium nucleus is 4.00260 amu. What is the mass defect and binding energy of this nucleus? (Note: c2=932MeVamu)
Solution: The difference 4.03190 − 4.00260 = 0.02930 amu is the mass defect for the helium nucleus. This is the mass that contributed to the binding energy of the nucleus:
E=mc2=(0.02930 amu)(932 MeVamu)≈0.03×900=27MeV (actual=27.3 MeV)
MCAT CONCEPT CHECK 9.3
Before you move on, assess your understanding of the material with these questions.
- Define the following terms:
- Strong nuclear force:
_________________________________
- Mass defect:
_________________________________
- Binding energy:
_________________________________
- What are the four fundamental forces of nature?
- _________________________________
- _________________________________
- _________________________________
- _________________________________
- How does the mass defect relate to the binding energy?
_________________________________
_________________________________
9.4 Nuclear Reactions
LEARNING OBJECTIVES
After Chapter 9.4, you will be able to:
- Compare and contrast nuclear fission and nuclear fusion reactions
- Recall the emissions, ΔZ, and ΔA of different radioactive processes
- Recall the type of decay that could be detected in an atomic absorption spectrum
- Predict the number of half-lives necessary for decay of some portion of a radioactive sample:
Nuclear reactions, such as fusion, fission, and radioactive decay, involve either combining or splitting the nuclei of atoms. Because the binding energy per nucleon is greatest for intermediate-sized atoms (that is, intermediate-sized atoms are most stable), when small atoms combine or large atoms split, a great amount of energy is released.
When written in isotopic notation, elements are preceded by their atomic number as a subscript and mass number as a superscript (ZAX). The atomic number (Z) corresponds to the number of protons in the nucleus; the mass number (A) corresponds to the number of protons plus neutrons. When balancing nuclear equations, it is important to balance the number of nucleons on both sides by balancing the atomic numbers and mass numbers.
Fusion
Fusion occurs when small nuclei combine to form a larger nucleus. As an example, many stars (including the Sun) power themselves by fusing four hydrogen nuclei to make one helium nucleus as shown in Figure 9.4. By this method, the Sun produces 3.85 × 1026 joules per second (385 yottawatts), which accounts for the mass defect that arises from the formation of helium nuclei from hydrogen nuclei. Here on Earth, fusion power plants—which are only in developmental stages—generate energy from deuterium (12H) and lithium nuclei.
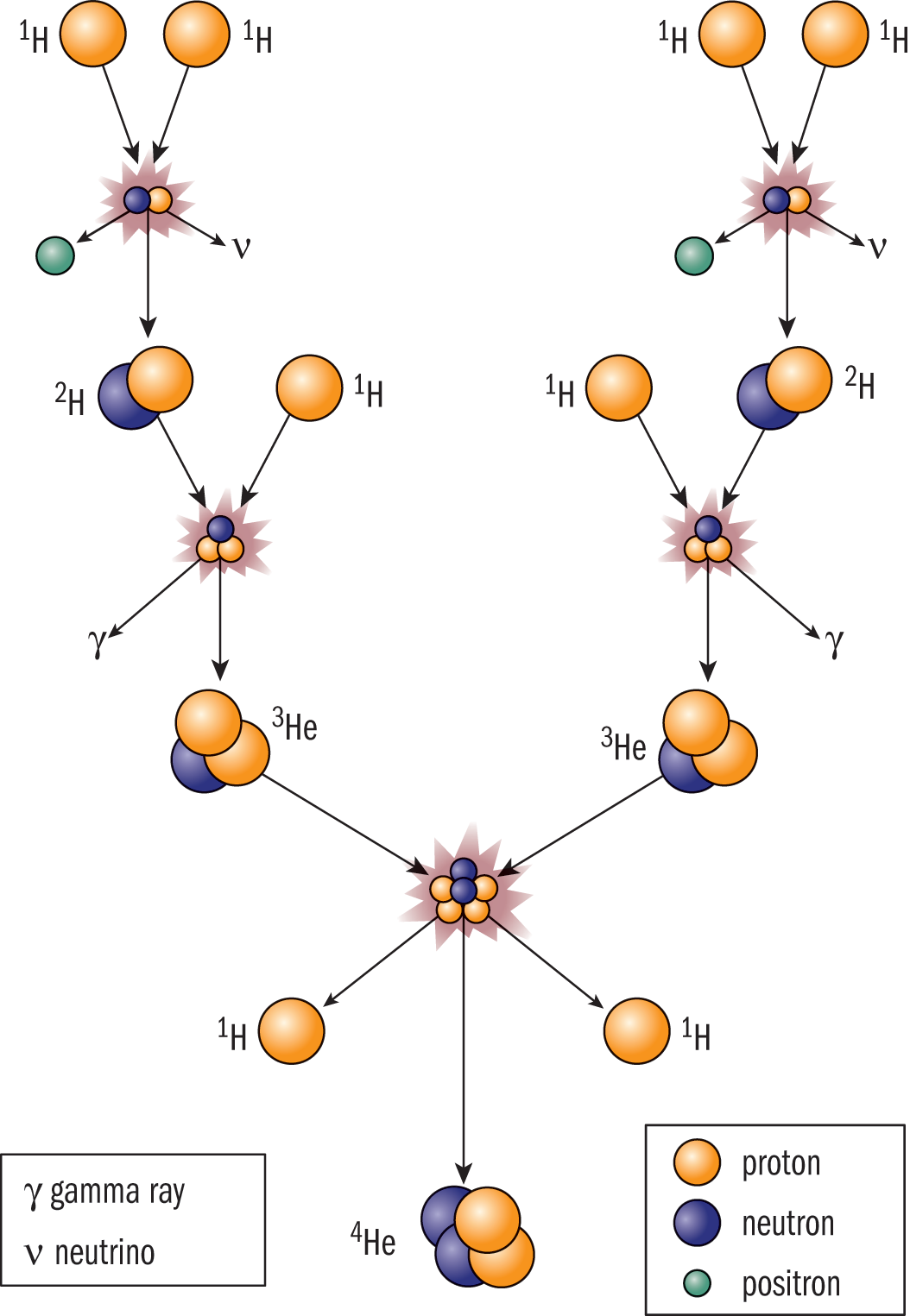
Figure 9.4 Hydrogen Fusion in the Sun, Creating Helium Nuclei
Fission
Fission is a process by which a large nucleus splits into smaller nuclei. Spontaneous fission rarely occurs. However, through the absorption of a low-energy neutron, fission can be induced in certain nuclei. Of special interest are those fission reactions that release more neutrons because these other neutrons will cause a chain reaction in which other nearby atoms can undergo fission. This in turn releases more neutrons, continuing the chain reaction. Such induced fission reactions power most commercial nuclear power plants.
Example: A fission reaction occurs when uranium-235 (U-235) absorbs a low-energy neutron, briefly forming an excited state of U-236, which then splits into xenon-140, strontium-94, and more neutrons. In isotopic notation form, the unbalanced reaction is:
U92235+n01→U92236*→Xe54140+S3894r+n01
When balanced, how many neutrons are produced in the last reaction?
Solution: By treating each arrow as an equals sign, the problem is simply asking us to balance the last equation. The mass numbers (A) on either side of each arrow must be equal. This is an application of nucleon number conservation, which says that the total number of neutrons plus protons remains the same, even if neutrons are converted to protons and vice-versa, as they are in some decays. Because 235 + 1 = 236, the first arrow is indeed balanced. Looking at the atomic numbers, the number of protons are balanced throughout (92 + 0 = 92 = 54 + 38 + 0). To find the number of neutrons, determine how many nucleons remain after accounting for xenon-140 and strontium-94:
236 − (140 + 94) = 236 − 234 = 2 nucleons
Because the protons are balanced, these remaining nucleons are both neutrons. Therefore, two neutrons are produced in this reaction. These neutrons are free to go on and be absorbed by more U-235 and cause more fission reactions. Note that it was not actually necessary to know that the intermediate high-energy state U92236* was formed.
Radioactive Decay
Radioactive decay is a naturally occurring spontaneous decay of certain nuclei accompanied by the emission of specific particles. On the MCAT, you should be prepared to answer three general types of radioactive decay problems:
- The integer arithmetic of particle and isotope species
- Radioactive half-life problems
- The use of exponential decay curves and decay constants
Isotope Decay Arithmetic and Nucleon Conservation
Let the letters X and Y represent nuclear isotopes. When the parent nucleus X undergoes nuclear decay to form daughter nucleus Y, the balanced reaction is:
XZA→YZ′A′+emitted decay particle
Equation 9.5
When balancing nuclear reactions, the sum of the atomic numbers must be the same on both sides of the equation, and the sum of the mass numbers must be the same on both sides as well.
MCAT EXPERTISE
Whenever you approach radioactive decay problems on the MCAT, start by balancing the number of protons (the atomic numbers). Often, wrong answer choices will simply have an error in the number of protons and can be eliminated before even checking the mass numbers.
Alpha Decay
Alpha decay is the emission of an α-particle, which is a He24 nucleus that consists of two protons, two neutrons, and zero electrons. The alpha particle is very massive compared to a beta particle and carries double the charge. Alpha particles interact with matter very easily; hence, they do not penetrate shielding (such as lead sheets) very extensively.
KEY CONCEPT
Alpha particles do not have any electrons, so they carry a charge of +2.
The emission of an α-particle means that the atomic number of the daughter nucleus will be two less than that of the parent nucleus, and the mass number will be four less. This can be expressed in the balanced equation:
ZAX→ Z−2A−4Y+α24
Equation 9.6 Example: Suppose a parent nucleus X alpha decays as follows:
92238X→ Z′A′Y+α
What are the mass number and atomic number of the daughter isotope Y?
Solution: To solve this question, we simply need to balance the atomic numbers and mass numbers:
ZAX→Z−2A−4Y+α24U92238→90234Y+α24
While it is not necessary to identify the elements to answer the question, answers on the MCAT are usually given with the element’s symbol. Y must be thorium (Th) because its atomic number is 90. Therefore, the daughter nucleus is 90234Th.
Beta Decay
Beta decay is the emission of a β-particle, which is an electron and is given the symbol e− or β−. Electrons do not reside in the nucleus, but they are emitted by the nucleus when a neutron decays into a proton, a β-particle, and an antineutrino (ν¯). Because an electron is singly charged and 1836 times lighter than a proton, the beta radiation from radioactive decay is more penetrating than alpha radiation. In some cases of induced decay (positron emission), a positron is released, which has the mass of an electron but carries a positive charge. The positron is given the symbol e+ or β+. A neutrino (ν) is emitted in positron decay, as well. Note that neutrinos and antineutrinos are not tested on the MCAT, and are therefore omitted in subsequent discussion.
KEY CONCEPT
In both types of beta decay, there needs to be conservation of charge. If a negative charge (β−) is produced, a neutron is converted into a proton to maintain charge. Conversely, if a positive charge (β+) is produced, a proton is converted into a neutron to maintain charge. Remember that negative beta decay produces a negative β-particle and positive beta decay produces a positive β-particle.
During β− decay, a neutron is converted into a proton and a β−-particle (Z = −1, A = 0) is emitted. Hence, the atomic number of the daughter nucleus will be one higher than that of the parent nucleus, and the mass number will not change. This can be expressed in the balanced equation:
ZAX → Z+1AY + β−
Equation 9.7
During β+ decay, a proton is converted into a neutron and a β+-particle (Z = +1, A = 0) is emitted. Hence, the atomic number of the daughter nucleus will be one lower than that of the parent nucleus, and the mass number will not change. This can be expressed in the balanced equation:
ZAX → Z−1AY + β+
Equation 9.8 Example: Suppose a promethium-146 nucleus beta-decays as follows:
61146Pm → Z′A′Y + β−
What are the mass number and atomic number of the daughter isotope Y?
Solution: Again, balance the atomic numbers and mass numbers:
61146Pm→YZ + 1A+ −1 0β−61146Pm→62146Y+ −1 0β−
Y must be samarium (Sm) because its atomic number is 62. Therefore, the daughter nucleus is 62146Sm.
Gamma Decay
Gamma decay is the emission of γ-rays, which are high-energy (high-frequency) photons. They carry no charge and simply lower the energy of the parent nucleus without changing the mass number or the atomic number. The high-energy state of the parent nucleus may be represented by an asterisk.
KEY CONCEPT
Gamma decay questions are the easiest on the MCAT. No changes occur in the mass number or atomic number; only a γ-ray is emitted.
This can be expressed in the balanced equation:
ZAX*→ ZAX+γ
Equation 9.9 Example: Suppose an excited parent isotope ZAXgamma decays to Z′A′X, which then undergoes positron emission to form Z″A″Y, which in turn alpha decays to Z‴A‴Z. If Z is americium-241, what is ZAX?
Solution: Because the final daughter nucleus is given, it will be necessary to work backwards through the reactions. The last reaction is the following alpha decay:
Z″A″Y → 95241Am+ 24α
The atomic number of the parent nucleus must be 97, and the mass number is 245. This is berkelium-245. The preceding reaction is the following positron emission:
Z’A’X → 97245Bk + +1 0β+
The atomic number of the parent nucleus must be 98, and the mass number is 245. This is californium-245. Finally, the preceding reaction is the following gamma decay:
ZAX* → 98245Cf+γ
The atomic number of the parent nucleus must be 98, and the mass number is 245. This is a higher-energy form of californium-245: ZAX= 98245Cf
Electron Capture
Certain unstable radionuclides are capable of capturing an inner electron that combines with a proton to form a neutron, while releasing a neutrino. The atomic number is now one less than the original but the mass number remains the same. Electron capture is a rare process that is perhaps best thought of as the reverse of β− decay:
ZAX+e− → Z−1 AY
Equation 9.10
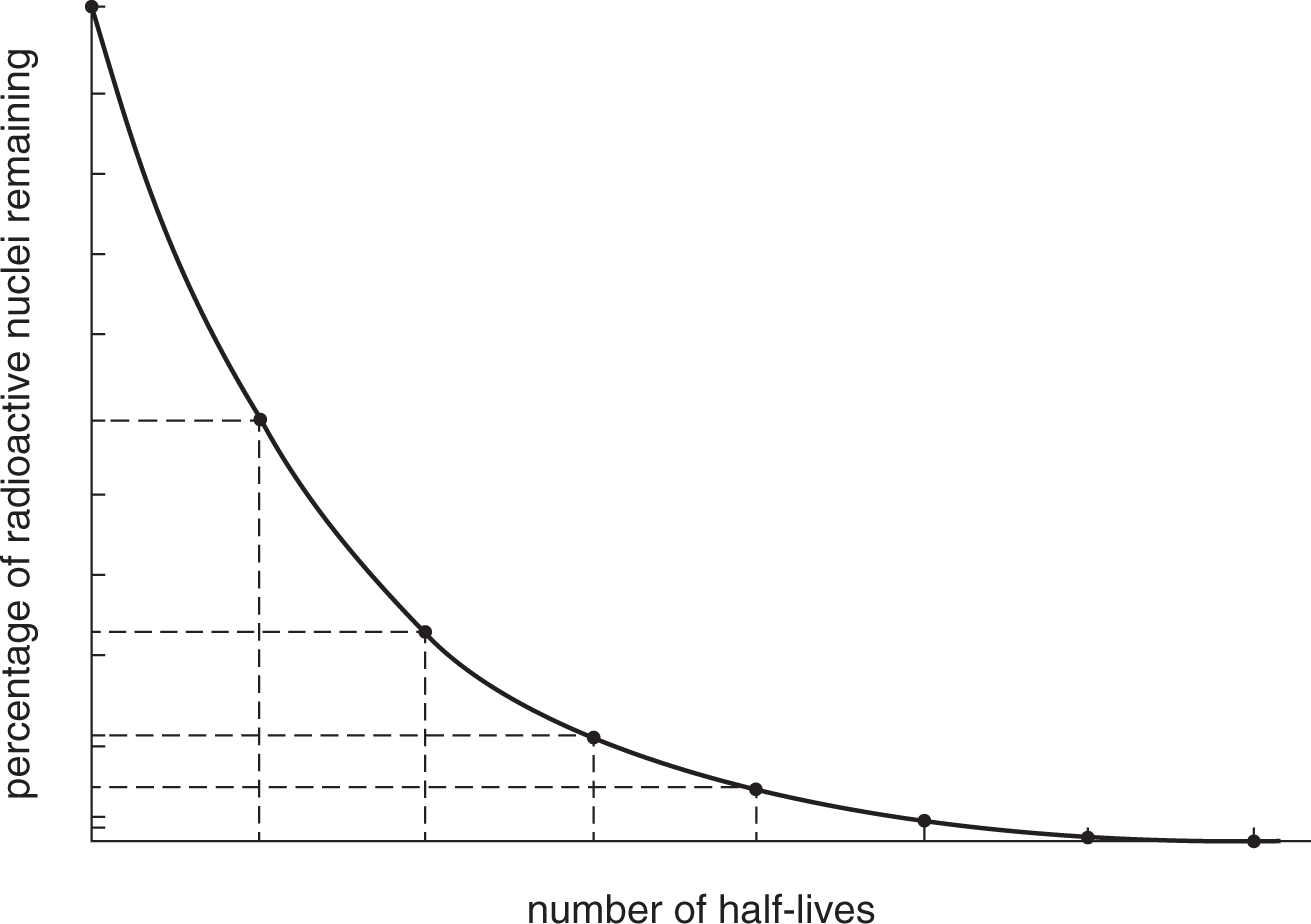
Half-Life
In a sample of radioactive particles, the half-life (T12) of the sample is the time it takes for half of the sample to decay. In each subsequent half-life, one-half of the remaining sample decays so that the remaining amount asymptotically approaches zero.
MCAT EXPERTISE
Half-life problems are common on the MCAT. Make sure you write them out; it’s easy to lose your place when doing them in your head.
Example: If the half-life of a certain isotope is 4 years, what fraction of a sample of that isotope will remain after 12 years?
Solution: If 4 years is one half-life, then 12 years is 3 half-lives. During the first half-life—the first 4 years—half of the sample will decay. During the second half-life (years 4 to 8), half of the remaining half will decay, leaving one-fourth of the original. During the third and final half-life (years 8 to 12), half of the remaining fourth will decay, leaving one-eighth of the original sample. Thus, the fraction remaining after 3 half-lives is (12)3=18.
Exponential Decay
Let n be the number of radioactive nuclei that have not yet decayed in a sample. It turns out that the rate at which the nuclei decay, ΔnΔt, is proportional to the number that remain (n). This suggests the equation
ΔnΔt=−λn
Equation 9.11
where λ is known as the decay constant. The solution of this equation tells us how the number of radioactive nuclei changes with time. This is known as an exponential decay:
n = n0e−*λ**t*
Equation 9.12
where n0 is the number of undecayed nuclei at time t = 0. The decay constant is related to the half-life by
λ=ln2T12=0.693T12
Equation 9.13
A typical exponential decay curve is shown in Figure 9.5.
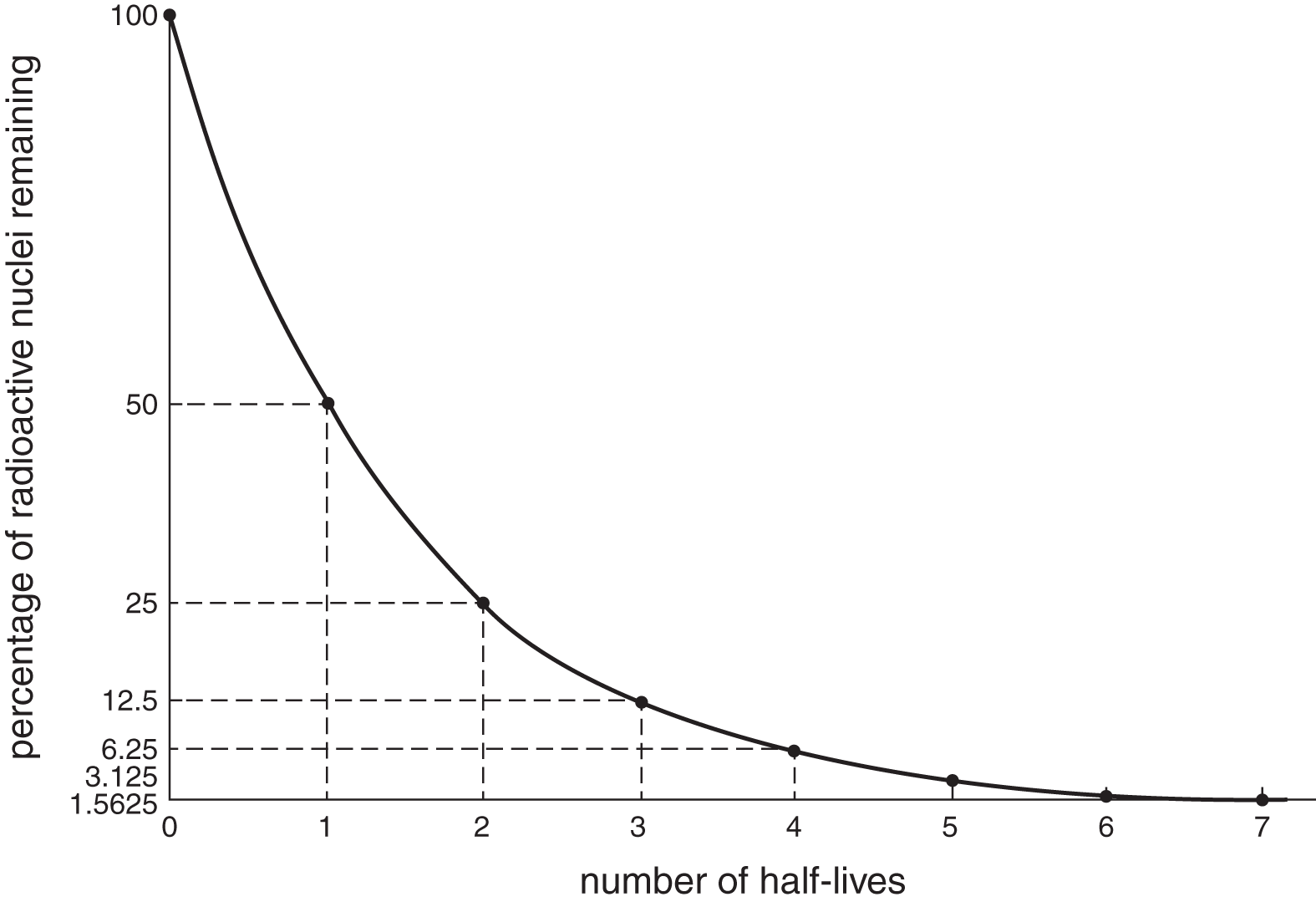
Figure 9.5 Exponential Decay
Example: If at time t = 0, there is a 2 mole sample of radioactive isotopes, how many nuclei remain after 45 minutes, assuming a decay constant of 2 hr−1? (Hint: e-32= 0.22).
Solution: This question is asking for an application of the exponential decay equation:
n = n0e−λt =n0e−(2hr−1)(34hr) = n0e−32
Raising Euler’s number (e) to an exponent—especially a fractional exponent—is beyond the scope of the math on the MCAT, but the value of e−32 is 0.22. Thus, 22% of the original 2 mole sample remains. This constitutes 0.44 mol, which, if multiplied by Avogadro’s number, gives us the number of nuclei remaining:
0.44 mol×6.02×1023 nucleimol=2.64×1023 nuclei
MCAT CONCEPT CHECK 9.4
Before you move on, assess your understanding of the material with these questions.
- True or False: Nuclear fission and nuclear fusion both release energy.
- Compare and contrast nuclear fission and nuclear fusion reactions:
Nuclear Reaction Size of Reactant Particles Change in Nuclear Mass during Reaction (Increase or Decrease) Fission Fusion
- Complete the following chart:
**Nuclear Reaction Emits… ΔZ ΔA Alpha decay Beta-negative decay Beta-positive decay Gamma decay Electron capture**
- How many half-lives are necessary for the complete decay of a radioactive sample?
_________________________________
- Which type of nuclear decay could be detected in an atomic absorption spectrum?
_________________________________
Conclusion
Congratulations! You’ve finished the physics material that will be needed for Test Day. Our last topic was the interaction of energy and matter on the atomic level. We began by examining the photoelectric effect. Further, we took a look at Bohr’s model of the hydrogen ion, and made some generalizations about electronic structure and the permissible states in regards to absorption and emission of light energy. We also noted that on a molecular level, small changes in structure can lead to significant shifts in absorption. We studied the interactions of the nucleus with energy, including the prototypical nuclear reactions of fusion and fission. We finished our discussion of nuclear reactions by examining the most common forms of nuclear decay and some of the mathematics for determining half-life or sample remaining. In the next few chapters, we’ll focus on building Test Day skills, including MCAT math shortcuts that will make many of these concepts more rewarding.
GO ONLINE
You’ve reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
The Photoelectric Effect
- The photoelectric effect is the ejection of an electron from the surface of a metal in response to light.
- The threshold frequency is the minimum light frequency necessary to eject an electron from a given metal.
- The work function is the minimum energy necessary to eject an electron from a given metal. Its value depends on the metal used and can be calculated by multiplying the threshold frequency by Planck’s constant.
- The greater the energy of the incident photon above the work function, the more kinetic energy the ejected electron can possess.
- The ejected electrons create a current; the magnitude of this current is proportional to the intensity of the incident beam of light.
Absorption and Emission of Light
- The Bohr model of the atom states that electron energy levels are stable and discrete, corresponding to specific orbits.
- An electron can jump from a lower-energy to a higher-energy orbit by absorbing a photon of light of the same frequency as the energy difference between the orbits.
- When an electron falls from a higher-energy to a lower-energy orbit, it emits a photon of light of the same frequency as the energy difference between the orbits.
- Absorption spectra may be impacted by small changes in molecular structure.
- Fluorescence occurs when a species absorbs high-frequency light and then returns to its ground state in multiple steps. Each step has less energy than the absorbed light and is within the visible range of the electromagnetic spectrum.
Nuclear Binding Energy and Mass Defect
- Nuclear binding energy is the amount of energy that is released when nucleons (protons and neutrons) bind together.
- The more binding energy per nucleon released, the more stable the nucleus.
- The four fundamental forces of nature are the strong and weak nuclear force, which contribute to the stability of the nucleus, electrostatic forces, and gravitation.
- The mass defect is the difference between the mass of the unbonded nucleons and the mass of the bonded nucleons within the nucleus.
- The unbonded constituents have more energy and, therefore, more mass than the bonded constituents.
- The mass defect is the amount of mass converted to energy during nuclear fusion.
Nuclear Reactions
- Fusion occurs when small nuclei combine into larger nuclei.
- Fission occurs when a large nucleus splits into smaller nuclei.
- Energy is released in both fusion and fission because the nuclei formed in both processes are more stable than the starting nuclei.
- Radioactive decay is the loss of small particles from the nucleus.
- Alpha (α) decay is the emission of an alpha particle (α, 24α, 24Ηe), which is a helium nucleus.
- Beta-negative (***β*−)decay** is the decay of a neutron into a proton, with emission of an electron (e−, β−) and an antineutrino ν¯.
- Beta-positive (***β*+)decay, also calledpositron emission, is the decay of a proton into a neutron, with emission of apositron** (e+, β+) and a neutrino (ν).
- Gamma (γ) decay is the emission of a gamma ray, which converts a high-energy nucleus into a more stable nucleus.
- Electron capture is the absorption of an electron from the inner shell that combines with a proton in the nucleus to form a neutron.
- Half-life is the amount of time required for half of a sample of radioactive nuclei to decay.
- In exponential decay, the rate at which radioactive nuclei decay is proportional to the number of nuclei that remain.
ANSWERS TO CONCEPT CHECKS
**9.1**
- The work function describes the minimum amount of energy necessary to emit an electron. Any additional energy from a photon will be converted to excess kinetic energy during the photoelectric effect.
- The threshold frequency depends on the chemical composition of a material (that is, the identity of the metal).
- The accumulation of moving electrons creates a current during the photoelectric effect.
**9.2**
- The energy differences between ground-state electrons and higher-level electron orbits determine the frequencies of light a particular material absorbs (its absorption spectrum).
- False. Small changes, such as protonation and deprotonation, change in oxidation state or bond order, and others may cause dramatic changes in light absorption in a material.
- When electrons transition from a higher-energy state to a lower-energy state, they will experience photon emission.
- Fluorescence is a special stepwise photon emission in which an excited electron returns to the ground state through one or more intermediate excited states. Each energy transition releases a photon of light. With smaller energy transitions than the initial energy absorbed, these materials can release photons of light in the visible range.
**9.3**
- The strong nuclear force is one of the four primary forces and provides the adhesive force between the nucleons (protons and neutrons) within the nucleus. Mass defect is the apparent loss of mass when nucleons come together, as some of the mass is converted into energy. That energy is called the binding energy.
- The four fundamental forces of nature are the strong and weak nuclear forces, electrostatic forces, and gravitation.
- Mass defect is related to the binding energy such that there is a transformation of nuclear matter to energy with a resultant loss of matter. They are related by the equation E = mc2.
**9.4**
- True. While they may seem like inverses of each other, both nuclear fusion and nuclear fission reactions release energy.
-
Nuclear Reaction Size of Reactant Particles Change in Nuclear Mass during Reaction (Increase or Decrease) Fission Large (actinides, lanthanides) Decrease
Fusion Small (hydrogen, helium) Increase
-
**Nuclear Reaction Emits… ΔZ ΔA Alpha decay** Alpha particle (α, 24α, 24Ηe) −2 −4
Beta-negative decay Electron (e−, β−) and antineutrino (ν¯) +1 0
Beta-positive decay Positron (e+, β+) and neutrino (ν). −1 0
Gamma decay Gamma ray (γ) 0 0
Electron capture Nothing (absorbs an electron from inner shell) −1 0
- Because the amount remaining is cut in half after each half-life, the portion remaining will never quite reach zero. This is mostly a theoretical consideration; “all” of a sample is considered to have decayed after 7 to 8 half-lives.
- Because gamma radiation produces electromagnetic radiation (rather than nuclear fragments), it can be detected on an atomic absorption spectrum.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. C
To determine the speed of the electrons ejected, we must first calculate their kinetic energy:
K=hf−W=(6.626×10−34 J⋅s)(1.0×1014 Hz)−6.622 × 10-20J =(6.626−6.622)×10-20=0.004 × 10-20= 4 × 10-23J
Now with a value for kinetic energy, we can calculate the speed of the ejected electrons:
K=12mv2→v=2 Km=2(4×10−23J)9.1×10−31 kg≈8×10−239×10−31≈108=104 ms
Notice the wide range in the exponents for the answer choices. While the math in this question may seem complex, this allows us to round significantly.
2. B
To determine the wavelength of the light ray, first calculate its frequency from the photoelectric effect equation:
K=hf−W→f=K+Wh=50J+16J6.626×10−34 J⋅s≈6.6×1016.6×10−34=1035 Hz
In this case, estimation of Planck’s constant makes our calculation much simpler without leading us to a nonviable answer. It is worth attempting an estimation first to avoid doing more work than necessary. Now we can determine the wavelength of the incident ray of light by relating the frequency to the speed of light:
c=fλ→λ=cf=3.00×108 ms1035 Hz=3×10−27 m
3. D
The Bohr model is based on a set of postulates originally put forward to discuss the behavior of electrons in hydrogen. In summary, these postulates state that the energy levels of the electron are stable and discrete, and they correspond to specific orbits, eliminating (A). They also state that an electron emits or absorbs radiation only when making a transition from one energy level to another, eliminating (B). Specifically, when an electron jumps from a lower-energy orbit to a higher-energy one, it must absorb a photon of light of precisely the right frequency such that the photon’s energy equals the energy difference between the two orbits, eliminating (C). When falling from a higher-energy orbit to a lower-energy one, an electron emits a photon of light with a frequency that corresponds to the energy difference between the two orbits, This is the opposite of (D), which makes it the right answer.
4. B
To solve this question correctly, one must be careful with the units. First, convert 10.2 eV to joules:
10.2 eV (1.60×10−19 JeV)≈1.6×10−18 J
Next, to determine the wavelength of the radiation, we can combine the formulas E = hf and c = f λ:
E=hf=hcλ→λ=hcE=6.626×10−34J⋅s3.00×108 ms(1.6×10−18 J)≈(4)(3)(10-26)10-18=12×10-8=1.2×10-7 m
5. B
The electron moves from a higher energy level to a lower energy level; this can only occur if the extra energy is dissipated through the emission of a photon. If the electron moved from B to A, it would absorb a photon and increase the atom’s total energy; however, the opposite is occurring, so (A) and (D) can be eliminated. The work function is the amount of energy required to eject an electron from a material; when moving from A to B, the electrical potential energy of the atom decreases, meaning that more energy will be required to free the electron from the atom, eliminating (C).
6. B
The strong nuclear force is the attractive force that holds protons and neutrons together in the nucleus, supporting choice (B). This force is greater than the electrostatic repulsion between protons. Note that binding energy, (A), is not one of the four fundamental forces.
7. A
The greater the intensity, the greater the number of incident photons and, therefore, the greater the number of electrons that will be ejected from the metal surface (provided that the frequency of the light remains above the threshold). This means a larger current. Remember that the frequency of the light (assuming it is above the threshold frequency) will determine the kinetic energy of the ejected electrons; the intensity of the light determines the number of electrons ejected per time (the current).
8. C
To determine the binding energy, we must first determine the mass defect. The mass defect is simply the masses of each of the protons and neutrons in the unbound state added together minus the mass of the formed argon-40 nucleus (which contains 18 protons and 40 − 18 = 22 neutrons):
mass defect=(18×1.0073 amu)+(22×1.0087 amu) −39.9132 amu≈(18×1.007)+(22×1.009)-39.9132=18.126+22.198−39.9132=40.324−39.9132≈0.4 (actual=0.4096 amu)
This math was difficult without a calculator, but by rounding one value down (proton) and one value up (neutron) by similar amounts we ended up very near the actual value. Calculating 18 x 7 = 126 and 22 x 9 = 198 for the decimal values is more manageable than the exact numbers and the spacing of the answer choices allows for our estimation. The binding energy can then be determined from this mass defect:
E=mc2=0.4096 amu×932MeVamu≈0.4×900=360 MeV
The closest answer is (C).
9. C
This process can be described as electron capture. Certain unstable radionuclides are capable of capturing an inner electron that combines with a proton to form a neutron. The atomic number becomes one less than the original, but the mass number remains the same. Electron capture is a relatively rare process and can be thought of as the reverse of β− decay. Notice that the equation is similar to that of β+ decay but not identical because a particle is absorbed, not emitted.
10. C
This problem presents a reaction and asks for the energy liberated due to the transformation of mass into energy. To convert mass into energy, we are told that 1 amu can be converted into 932 MeV of energy. All we need to do now is calculate how much mass, in amu, is converted in the reaction. Because we are given the atomic mass for each of the elements in the reaction, this is simply a matter of balancing the equation:
mass defect=(1.0087 amu+10.0129 amu) –(7.0160 amu+4.0026 amu)=11.0216−11.0186=0.0030 amu
Given both the small magnitude of this value and the small difference of the answer choices, it is best to not round at this point of the calculation. This is the amount of mass that has been converted into energy. To obtain energy from mass, we have to multiply by the conversion factor (1 amu = 932 MeV):
E = 0.003 × 932 ≈ 0.003 × 900 = 2.7 MeV
At this point we were able to round for an easier calculation that keeps us very near the correct answer choice.
11. C
Because the half-life of element X is four days, 50% of an original sample remains after four days, 25% remains after eight days, and 12.5% remains after 12 days. Therefore, n = 12 days. Another approach is to set (12)x=0.125, where x is the number of half-lives that have elapsed. Solving for x gives x = 3. Thus, 3 half-lives have elapsed, and because each half-life is four days, we know that n = 12 days.
12. B
The expression n = n0e−λt is equivalent to nn0=e−λt. Taking the natural logarithm of both sides, we get:
ln(nn0)=−λt
From this expression, it is clear that plotting ln(nn0) vs. t will give a straight line with a slope of −λ.
13. C
A typical carbon nucleus contains 6 protons and 6 neutrons. An α-particle contains 2 protons and 2 neutrons. Therefore, one carbon nucleus can dissociate into 62=3 α-particles.
14. A
Because the half-life of carbon-12 is essentially infinite, a 25% decrease in the ratio of carbon-14 to carbon-12 means the same as a 25% decrease in the amount of carbon-14. If less than half of the carbon-14 has deteriorated, then less than one half-life has elapsed. Therefore, the sample is less than 5,730 years old. Be careful with the wording here—the question states that the ratio is 25% less than the ratio in nature, not 25% of the ratio in nature, which would correspond to (D).
15. D
In alpha decay, an element loses two protons. In positron decay, a proton is converted into a neutron. Gamma decay has no impact on the atomic number of the nuclide. Therefore, two alpha decays and two positron decays will yield a daughter nuclide with six fewer protons than the parent nuclide.
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(9.1) Energy of a photon of light: E = hf
(9.2) Maximum kinetic energy of an electron in the photoelectric effect: Kmax = hf − W
(9.3) Work function: W = hfT
(9.4) Mass defect and energy: E = mc2
(9.5) Nuclear decay (general form): XZA→YZ′A′+ emitted decay particle
(9.6) Alpha decay: XZA→YZ−2A−4+α24
(9.7) Beta-negative decay: XZA→YZ+1A+β−
(9.8) Beta-positive decay (positron emission): XZA→YZ−1A+β+
(9.9) Gamma decay: XZA*→XZA+γ
(9.10) Electron capture: XZA→e−→YZ−1A
(9.11) Rate of nuclear decay: ΔnΔt=−λn
(9.12) Exponential decay: n = n0e−λt
(9.13) Decay constant: λ=ln2T12=0.693T12
SHARED CONCEPTS
General Chemistry Chapter 1
Atomic Structure
General Chemistry Chapter 2
The Periodic Table
Organic Chemistry Chapter 11
Spectroscopy
Physics and Math Chapter 1
Kinematics and Dynamics
Physics and Math Chapter 2
Work and Energy
Physics and Math Chapter 8
Light and Optics