Chapter 10: Mathematics

Chapter 10: Mathematics
Science Mastery Assessment
Every pre-med knows this feeling: there is so much content I have to know for the MCAT! How do I know what to do first or what’s important?
While the high-yield badges throughout this book will help you identify the most important topics, this Science Mastery Assessment is another tool in your MCAT prep arsenal. This quiz (which can also be taken in your online resources) and the guidance below will help ensure that you are spending the appropriate amount of time on this chapter based on your personal strengths and weaknesses. Don’t worry though— skipping something now does not mean you’ll never study it. Later on in your prep, as you complete full-length tests, you’ll uncover specific pieces of content that you need to review and can come back to these chapters as appropriate.
How to Use This Assessment
If you answer 0–7 questions correctly:
Spend about 1 hour to read this chapter in full and take limited notes throughout. Follow up by reviewing all quiz questions to ensure that you now understand how to solve each one.
If you answer 8–11 questions correctly:
Spend 20–40 minutes reviewing the quiz questions. Beginning with the questions you missed, read and take notes on the corresponding subchapters. For questions you answered correctly, ensure your thinking matches that of the explanation and you understand why each choice was correct or incorrect.
If you answer 12–15 questions correctly:
Spend less than 20 minutes reviewing all questions from the quiz. If you missed any, then include a quick read-through of the corresponding subchapters, or even just the relevant content within a subchapter, as part of your question review. For questions you got correct, ensure your thinking matches that of the explanation and review the Concept Summary at the end of the chapter.
- How would the number 17,060 be written in scientific notation?
- 1706 × 101
- 1.706 × 104
- 1.7060 × 104
- 0.17060 × 105
- How does the number of significant digits differ between 14,320,010 and 3.618000?
- 14,320,010 has more significant digits than 3.618000
- 14,320,010 has fewer significant digits than 3.618000
- 14,320,010 has the same number of significant digits as 3.618000
- A comparison cannot be made because the numbers are not both in scientific notation.
- Using the appropriate number of significant digits, what is the answer to the following math problem? (Note: Assume all numbers are the results of measurements.)
3.060 × 4.10 + 200 =
- 210
- 213
- 212.5
- 212.55
- Which of the following would be the most appropriate setup for estimating the value 3.6 × 4.85 for questions in which answer choices differ by a small margin?
- 3.5 × 5
- 3.5 × 4.5
- 4 × 4
- 4 × 5
- The value of 2000.25 is closest to:
- 4
- 14
- 50
- 800
- If A and B are real numbers, which of the following equations is INCORRECT?
- A3 × B3 = (AB)3
- A5 ÷ A7 = A−2
- (A0.5)4 + A2 = 2A2
- (A3)2 = A9
- How can the value of a natural logarithm be converted to the value of a common logarithm?
- The natural logarithm is divided by a constant.
- A constant is added to or subtracted from the natural logarithm.
- The natural logarithm is raised to an exponent.
- The inverse of the natural logarithm is taken.
- What is the minimum value of 2 cosθ − 1?
- −3
- −2
- −1
- 0
- Which of the following relationships is INCORRECT?
- |sin θ × cos θ| < |sin θ| + |cos θ|
- sin θ ÷ cos θ = tan θ
- tan 90° is undefined
- sin θ = sin (90° − θ)
- What is the approximate pH of a solution with a pKa of 3.6, [HA] = 100 mM, and [A−] = 0.1 M? (Note: pH=pKa+log [A−][HA])
- 1.6
- 3.6
- 5.6
- 7.6
- At what temperature do the Fahrenheit and Celsius scales give equal values?
- 0 K
- 233 K
- 313 K
- 273 K
- In a certain rigid container, pressure and temperature are directly proportional. If the pressure is changed from 540 torr to 180 torr via a temperature change, by what factor has the temperature changed?
- 360
- 3
- 1
- 13
- A 150-pound adult must be given a drug that is dosed at 1.5 mg drugkg body mass. Approximately how many milligrams of the drug should be administered per dose? (Note: 1 lb = 4.45 N)
- 33 mg
- 67 mg
- 100 mg
- 225 mg
- The rate of a reaction is calculated as a change in concentration per time. What are the units of the rate constant, k, in a reaction that is second order overall with respect to one species? (Note: A second-order reaction of this type has a rate law with the form rate = k[A]2, where [A] is the concentration of the species.)
- 1s
- Ms
- Lmol⋅s
- L2mol2⋅s
- One group of people requires a base level of 900 Calories per day plus an additional 12 Calories per kilogram of body mass per day. A second group requires a base level of 500 Calories per day, plus 15 Calories per kilogram of body mass per day. At what mass does the first group and the second group have the same caloric needs?
- 26 kg
- 67 kg
- 133 kg
- 266 kg
Answer Key
- B
- C
- B
- A
- A
- D
- A
- A
- D
- B
- B
- D
- C
- C
- C
Chapter 10: Mathematics
CHAPTER 10
MATHEMATICS
In This Chapter
10.1 Arithmetic and Significant Figures
Scientific Notation
Significant Figures
Estimation
10.2 Exponents and Logarithms
Exponents
Rules of Logarithms
Common vs. Natural Logarithms
10.3 Trigonometry
Definitions and Relationships
Common Values
10.4 Problem-Solving
Use of Relationships
Conversions
Unit Analysis
Algebraic Systems
Concept Summary
MCAT EXPERTISE
You may note that this chapter and the chapters following do not contain the typical chapter profile, as they aren’t directly related to Physics or AAMC content categories. However, this chapter covers fundamental math content required for calculations across all three science test sections. As such, Chapter 10 should be considered a must-review chapter!
Introduction
Going to the grocery store is not so different from solving an MCAT multiple choice question. You begin the process by determining how much of each item you need in the near future. Once you know what you need, you check what you already have in order to determine the quantity you need to buy to reach your goal. When you get to the store, you compare that amount to the containers on the shelves. Often they won’t match exactly. Say, for example, that you need a total of 16 ounces of peas for a recipe. You already have 5 at home, so you only need 11 more. Packages of peas, however, may only come in 10- or 16-ounce packages. At that point, you choose the best one for your needs—the 16-ounce package; better to have a little extra than to run short!
If you’ve ever shopped in an international grocery store, this process can become even more elaborate because the packaging sizes and currency may not match the units with which you are familiar. You may have never taken the time to consider how intensive one’s critical thinking must be to efficiently navigate the grocery store, but recognize that it’s the same process you need to use on Test Day. First, figure out what you want (what is the question looking for), what you have (information in a passage, question stem, or outside knowledge), and what’s needed (calculations and critical thinking), and then make a decision (by matching your answer, eliminating wrong answer choices, or guessing strategically). In this chapter, we’ll be focusing on the calculations and critical thinking of mathematics. The math required for the MCAT is on the level of precalculus. You won’t need any derivatives or integrals on Test Day, but rapid application of arithmetic, exponent and logarithm rules, trigonometry, statistics, and graphical analysis may be necessary to navigate the MCAT efficiently. In this chapter, you won’t see very much new content, but consider this an opportunity to hone your mathematics skills.
10.1 Arithmetic and Significant Figures
LEARNING OBJECTIVES
After Chapter 10.1, you will be able to:
- Convert values to and from scientific notation
- Determine the significant digits within a number, such as: 1,547,200
- Apply rounding techniques in multiplication and division problems
The MCAT often uses numbers that aren’t particularly “nice” looking, especially considering that calculators cannot be used on the test. However, the testmakers also know that calculators aren’t allowed, so even the most complex math still has to be solvable in a reasonable amount of time. We reconcile these two opposing concepts by using a few Test Day tricks: scientific notation, which can help us narrow down the exponent of our answer choice and often gives the answer directly; and judicious estimation, which will differentiate between otherwise similar answers. While significant figures won’t lead us to an answer in the way that the other MCAT skills will, it is a testable topic on the MCAT.
KEY CONCEPT
It is essential to use judicious rounding and math strategies on Test Day to get through the MCAT efficiently. This is particularly true in the Chemical and Physical Foundations of Biological Systems section. Do not try to solve for an exact answer—do only as much as you need to be able to choose the right answer choice!
Scientific Notation
Scientific notation is a method of writing numbers that takes advantage of powers of ten. In scientific notation, a number is written with a significand and an exponent. This is much easier to conceptualize with an example. Consider the number 217. The math using this number can be somewhat cumbersome. By transforming it into scientific notation (2.17 × 102), the number becomes easier to manipulate because the power of 10 has been pulled out. In this case, 2.17 is the significand (also called the coefficient or mantissa), and the 2 in 102 is the exponent.
The significand must be a number with an absolute value in the range [1,10]. This means that it is any real number between –10 and –1 (not including –10) or between 1 and 10 (not including 10). By extension, the significand cannot begin with a 0, nor can it begin with two digits before the decimal point. The exponent, on the other hand, can be any whole number—positive, negative, or 0.
If at any time your calculations are not in scientific notation, consider adjusting them. While there is a small time investment converting to scientific notation, the time saved on subsequent calculations usually makes up for—and often exceeds—this time investment. This is especially true for questions in which the answers differ by powers of ten. The only exception to maintaining scientific notation is in the calculation of square roots, which are discussed later in this chapter.
Significant Figures
Significant figures provide an indication of our certainty of a measurement, and help us to avoid exceeding that certainty when performing calculations. Significant figures are determined by the precision of the instrument being used for measurement. For example, imagine that you are measuring the width of a block of wood with a ruler. The ruler has markings for centimeters and millimeters; you could state with confidence the width of the block in millimeters—say, 55 millimeters.
However, on this ruler, there are no markings smaller than millimeters; you’d be forced to estimate where within the interval between two millimeter markings the block reaches—say 55.2 millimeters. You cannot be 100 percent confident about this decimal, but some information is better than none, and writing it down lets you know that you were confident about the first two digits.
BRIDGE
Significant figures are important because they give an indication of the accuracy of a measurement. Inaccurate measurements can bias research or lead to faulty conclusions. When presented with data, look for accuracy of the measurements in two ways: identifying the number of significant digits in a number, and looking for error margins or statistical significance in graphs. These latter topics are discussed in Chapter 12 of MCAT Physics and Math Review.
In the situation we just described, only the first two digits would be considered significant because we know that they were measured accurately. We can hold on to the third digit during calculations, but by the time we reach a final answer, we need to reduce the answer to an appropriate number of significant figures. To determine the number of significant figures in a number:
- Count all numbers between the first nonzero digit on the left and the last nonzero digit on the right. Any digit between these two markers (including 0) is significant.
- Any zeroes to the left of the first nonzero digit are considered leading zeroes and are not significant.
- If there are zeroes to the right of the last nonzero digit and there is a decimal point in the number, then those zeroes are significant figures. If there is no decimal point, they are not significant. For example, 3,490 has three significant figures, while 3,490.0 has five.
- For measurements, the last digit is usually an estimation and is not considered significant (as in the example above).
Scientific notation can clarify significant figures when it contains a decimal point. When converting between standard numbers and scientific notation, be sure to maintain the number of significant figures. 100.0 is written in scientific notation as 1.000 × 102, while 100 is written as 1 × 102 because the trailing zeroes in the first example are significant while in the second example they are not.
Math with Significant Figures
Significant figure estimations are most important in the laboratory sciences, particularly analytical chemistry. For multiplication and division, maintain as many digits as possible throughout the calculations so that there is very little rounding error, then round to the number of significant digits that is the same as the least number of significant digits in any of the factors, divisors, or dividends. With addition and subtraction, decimal points are maintained rather than maintaining significant figures. The convention for decimal points is the same as for significant figures: the answer may have only as many decimal digits as the initial number with the fewest decimal digits.
MCAT EXPERTISE
Most Test Day math (and, by extension, this Kaplan MCAT Review series) neglects significant figures in the answer choices. These calculations are only necessary when specified by the question stem or passage.
Example: Determine the volume of a cylinder with a radius that is measured as 7.45 m and a height of 8.323 m. (Note: Use 3.14159 as π, and round the answer to the correct number of significant digits.)
Solution:
V=Abase×h=(πr2)×h=(3.14159)(7.45 m)2(8.323 m)=1,451.249
Because all of the factors are multiplied, the answer should have the same number of significant digits as the factor with the fewest number of significant digits. In this case, that is the radius, which has only two significant digits (remember that, in the case of measurements, the last digit is an estimate and is not considered significant). Therefore, the correct answer is 1,500 or 1.5 × 103.
Estimation
On Test Day, much of your math will be determined by the answer choices provided. If the answer choices are very close together, there will be minimal opportunity for rounding; when they are far apart, rough estimations are all that are necessary. While estimation of addition and subtraction are relatively simple rounding choices, we’ll review a few tricks for multiplication and division.
Multiplication
Consider the following multiplication problem: (3.17 × 104) × (4.53 × 105). To three significant digits, the answer to this multiplication problem is 1.44 × 1010, but this precise calculation is beyond the scope of mental math. However, even if the answer choices are close, it is generally acceptable to round to one decimal place, or (3.2 × 104) × (4.5 × 105). When rounding numbers in multiplication, keep in mind whether the rounded number is larger or smaller than the original number. If one number is rounded up, it is best to round the other number down slightly to compensate. Even with this rounding, the answer still comes out as 1.44 × 1010.
If the answer choices are very far apart—differing by, say, powers of ten—we can adjust the numbers so that one contains only one significant digit, further simplifying the math. In this example, the calculations could be adjusted to (3 × 104) × (4.5 × 105), or 1.35 × 1010. This represents an error of 6.25%, which is still close enough to choose the correct answer for most questions on Test Day.
Division
Let’s also consider division as an avenue for estimation. While in our multiplication example we adjusted each number in an opposite direction, with division we are attempting to make proportional adjustments in the same direction. Consider the following example:
Example: Estimate the value of 15.4 ÷ 3.80.
Solution: Estimations in division should be made by shifting both numbers in the same direction. It is often easier to adjust the divisor first to simplify calculations. If we round the divisor up to 4, we should round the dividend up accordingly. In this case, it makes sense to round the dividend up to 16—which is not only a whole number, but also a multiple of 4. Our estimate is 16 ÷ 4 = 4. Note that, despite this very rough adjustment, we are still very close to the true value of 4.05.
KEY CONCEPT
When rounding numbers to be multiplied, round one number up and one number down to compensate. When rounding numbers to be divided, round both numbers in the same direction to compensate.
MCAT CONCEPT CHECK 10.1
- Describe the process for converting a number into scientific notation. What values are possible for the significand?
_________________________________
_________________________________
_________________________________
- Highlight or circle the significant digits in the following numbers:
- 34,600.
- 0.0003201
- 1.10
- 525,600
- When rounding two numbers containing decimals, in which direction(s) should each number go for multiplication? For division?
- Multiplication
_________________________________
- Division
_________________________________
10.2 Exponents and Logarithms
LEARNING OBJECTIVES
After Chapter 10.2, you will be able to:
- Estimate the square root of a given value, like 1942
- Estimate the log value of a given number
- Simplify expressions such as: (a + 2b)3
For many students, exponents and logarithms are topics filed away in the depths of memory. While exponential and logarithmic functions are uncommon for many people in everyday life, a number of science topics and equations regularly tested on the MCAT require use of these concepts, as shown in Table 10.1.
Table 10.1 Common Exponential and Logarithmic Equations on the MCAT
TOPIC EQUATION LOCATION IN KAPLAN MCAT REVIEW SERIES
Sound level β=10 log II0 Chapter 7 of MCAT Physics and Math Review
Exponential decay n = n0e−λt Chapter 9 of MCAT Physics and Math Review
Arrhenius equation for activation energy k=Ae–EaRT Chapter 5 of MCAT General Chemistry Review
Gibbs free energy ΔGrxnο=−RT ln Keq Chapter 7 of MCAT General Chemistry Review
**p scales (pH, pOH, pKa, pKb)** pH = −log [H+] Chapter 10 of MCAT General Chemistry Review
Henderson–Hasselbalch equation pH=pKa+log[A−][HA] Chapter 10 of MCAT General Chemistry Review
Exponents
In addition to exponential equations, exponents appear frequently on the MCAT in the context of scientific notation, discussed earlier. Here, we look at the rules of arithmetic with exponents.
Exponent Identities
Only a basic understanding of exponents is necessary for the MCAT, although it can be helpful to know a few values and basic rules. First, any number to the zeroth power is equal to 1:
X0 = 1
Equation 10.1
When adding or subtracting numbers with exponents, the true value must be calculated before the addition or subtraction can be performed. For example, 32 + 32 ≠ 62; rather, 32 + 32 = 9 + 9 = 18. However, if the base and exponent are the same, we can add the coefficients: 32 + 32 = (1 + 1) × 32 = 2 × 32 = 18.
In cases of multiplication and division, the exponents can be manipulated directly, as long as the base number is the same. When multiplying two numbers with the same base, the exponents are added to determine the new number:
XA × XB = X(A + B)
Equation 10.2
KEY CONCEPT
When adding, subtracting, multiplying, or dividing numbers with exponents, the base must be the same.
In division, we subtract the exponent of the denominator from the exponent in the numerator to find the exponent in the quotient, as long as all bases are the same:
XAXB=X(A−B)
Equation 10.3
For a number that is raised to an exponent and then raised again to another exponent, the two exponents are multiplied:
(XA)B = X(A × B)
Equation 10.4
When a fraction is raised to an exponent, the exponent is distributed to the numerator and denominator:
(XY)A=XAYA
Equation 10.5
Negative exponents represent inverse functions:
X−A=1XA
Equation 10.6
For fractional exponents, the numerator can be treated as the exponent, and the denominator represents the root of the number:
XAB=XAB
Equation 10.7
Estimating Square Roots
On Test Day, you may be expected to calculate approximate square roots. To do so, it is useful to be familiar with the values in Table 10.2.
Table 10.2 Square Values of Integers from 1 to 20
X X2 X X2 X X2 X X2
1 1 6 36 11 121 16 256
2 4 7 49 12 144 17 289
3 9 8 64 13 169 18 324
4 16 9 81 14 196 19 361
5 25 10 100 15 225 20 400
If you are asked to calculate the square root of any number less than 400, you can approximate its value by determining which two perfect squares it falls between. As an alternative method, you can divide the number given to you by known squares to attempt to reduce it:
180=4×9×5=2×3×5=65
One can estimate this value by considering that the square root of five is somewhere between 2 and 3 (22 = 4 and 32 = 9), and is closer to 2 than 3. If we estimate 5 to be about 2.2, then 65≈13.2, which is congruent with our knowledge that the square root of 180 will be between 13 and 14. The true value of 180 is approximately 13.4.
MCAT EXPERTISE
Estimation of square roots and logarithms is generally sufficient to the first decimal place; don’t struggle to become more precise because it won’t be necessary on Test Day.
If you are using a number in scientific notation, adjust the decimal by one place if necessary so that the exponent is easily divisible by two:
4.9×10−7=49×10−8=7×10−4
Finally, it is useful to know the values of 2 and 3:
2≈1.414 (use 1.4)3≈1.732 (use 1.7)
Equation 10.8
Rules of Logarithms
Logarithms follow many of the same rules as exponents because they are inverse functions. The logarithmic rules are described below:
logA1=0logAA=1logA×B=logA+logBlog AB=logA−logBlogAB=BlogAlog1A=−logA
Equations 10.9 to 10.14
It is also useful to know that “p” can be shorthand for −log; thus,pH = −log [H+], pKa = −log Ka, and so on.
Example: Derive the Henderson–Hasselbalch equation from the expression for Ka.
Solution:
Ka=[H+][A−][HA]log Ka=log [H+][A−][HA]log Ka=log [H+]+log [A−]−log [HA]−log [H+]=−log Ka+log [A−]−log [HA]pH=pKa+log [A−][HA]
Common *vs*. Natural Logarithms
Logarithms can use any base, but the most common are base ten, as in our decimal system, and base e (Euler’s number, about 2.718). Base-ten logarithms (log10) are called common logarithms, whereas those based on Euler’s number (loge or ln) are called natural logarithms. Both common and natural logarithms obey the rules discussed above, but it can be easier to estimate common logarithms because of our familiarity with the decimal number system. Therefore, it is useful to be able to convert between natural logarithms and common logarithms:
logx≈lnx2.303
Equation 10.15
KEY CONCEPT
e is Euler’s number, which is 2.718281828459045…. It is also the base for the natural logarithm.
Estimating Logarithms
When estimating the logarithm of a number, use scientific notation. An exact logarithmic calculation of a number that is not an integer power of 10 is unnecessary on the MCAT. The testmakers are interested, however, in testing your ability to apply mathematical concepts appropriately in solving certain problems. Fortunately, there is a simple method of approximation that can be used on Test Day. If a value is written in proper scientific notation, it will be in the form n × 10m, where n is a number between 1 and 10. From this fact, we can use logarithm rules to approximate the value:
log (n×10m)=log (n)+log (10m)=m+log (n)
Because n is a number between 1 and 10, its logarithm will be a decimal between 0 and 1 (log 1 = 0 and log 10 = 1). The closer n is to 1, the closer log n will be to 0; the closer n is to 10, the closer log n will be to 1. As a reasonable approximation, one can say that
log (n × 10m) ≈ m + 0.n
Equation 10.16
where 0.n represents sliding the decimal point of n one position to the left (dividing n by ten). For example, log (9.2 × 108) ≈ 8 + 0.92 = 8.92 (actual = 8.96).
BRIDGE
A similar concept for estimating logarithms is used in calculations of pH, as described in Chapter 10 of MCAT General Chemistry Review. The shortcut is slightly different because we are working with negative logarithms and a negative exponent in the case of pH:
−log (n × 10−m) ≈ m − 0.n.
MCAT CONCEPT CHECK 10.2
Before you move on, assess your understanding of the material with these questions.
- Simplify the following expressions:
- (a + b)2 =
_________________________________
- a2 + 2a25a3=
_________________________________
- loga (a) =
_________________________________
- log (a3) − log (a) =
_________________________________
- Estimate 392:
_________________________________
_________________________________
_________________________________
- Estimate log 7,426,135,420:
_________________________________
10.3 Trigonometry
LEARNING OBJECTIVES
After Chapter 10.3, you will be able to:
- Explain the appropriate way to orient vectors in vector addition
- Calculate the value of sine, cosine, or tangent for a given right triangle
- Recall the sine, cosine, and tangent values of key angles
Very little trigonometry is required for the MCAT, but a basic understanding of definitions and a strong knowledge of two special right triangles is essential for strong performance, especially on physics material.
Definitions and Relationships
For any given right triangle and angle, there are characteristic values of sine, cosine, and tangent that depend on the lengths of the legs of the triangle and of the hypotenuse, as shown in Figure 10.1.
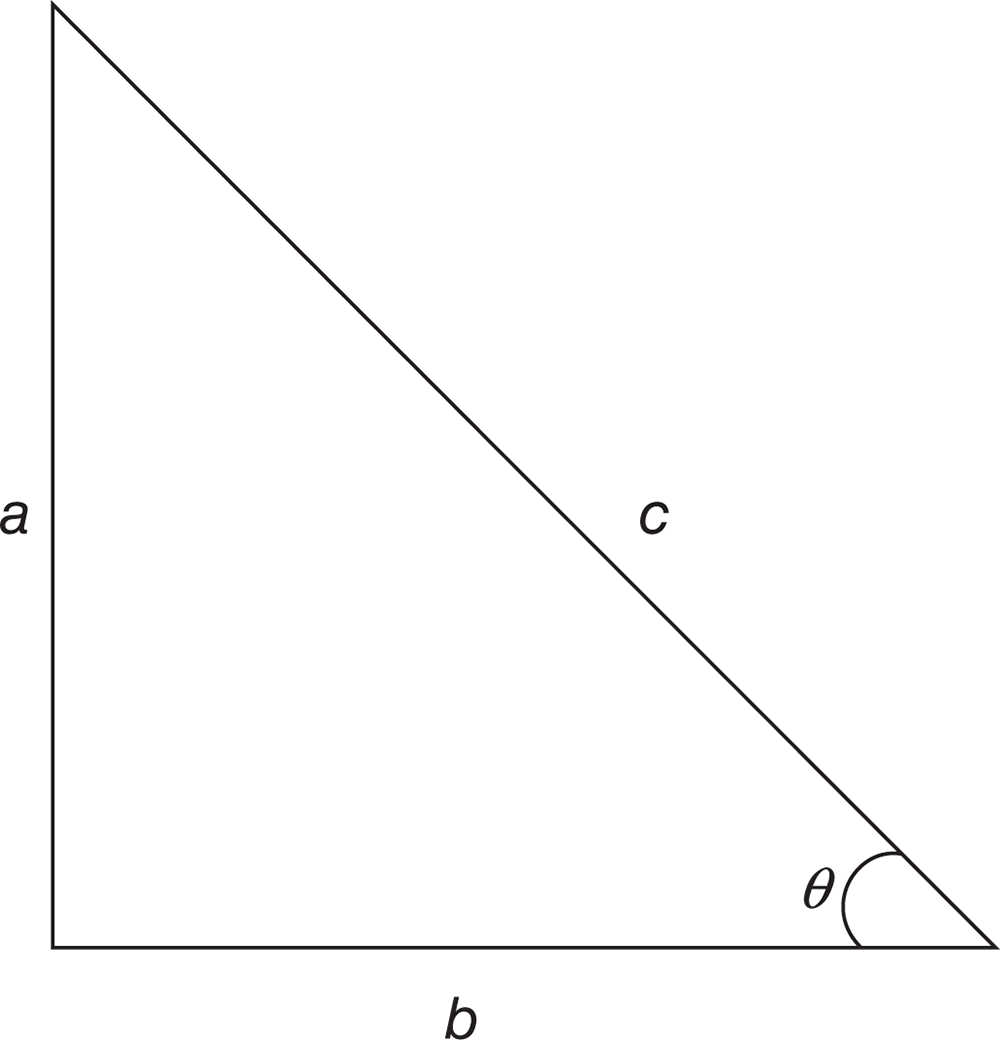
Figure 10.1. Right Triangle and Sides
Sine is calculated as the ratio between the side opposite the angle of interest and the hypotenuse:
sin θ=oppositehypotenuse=ac
Equation 10.17 Cosine is calculated as the ratio between the side adjacent to the angle of interest and the hypotenuse:
cos θ=adjacenthypotenuse=bc
Equation 10.18 Tangent is calculated as the ratio between the side opposite the angle of interest and the side adjacent to the angle of interest:
tan θ=oppositeadjacent=ab
Equation 10.19
MNEMONIC
Trigonometric ratios: SOH CAH TOA:
- Sine = Opposite ÷ Hypotenuse
- Cosine = Adjacent ÷ Hypotenuse
- Tangent = Opposite ÷ Adjacent
The values of both sine and cosine range from −1 to 1. The values of tangent, however, range from −∞ to ∞.
Each trigonometric function also has an inverse function: inverse sine (sin−1 or arcsin), inverse cosine (cos−1 or arccos), and inverse tangent (tan−1 or arctan). These functions use the calculated value of sine, cosine, or tangent, and yield a numerical value for the angle of interest. For the triangle in Figure 10.1, sin−1(ac)=θ. Inverse trigonometric functions are most likely to appear in questions asking for the direction of a resultant in vector addition or subtraction.
KEY CONCEPT
Trigonometric functions are useful for splitting a vector into its components; inverse trigonometric functions are useful for determining the direction of a resultant from its components.
Common Values
On Test Day, you must know the values of sine, cosine, and tangent for all of the angles in the 30−60−90 and 45−45−90 special right triangles, either by memorization or by drawing the triangles. The two triangles are shown in Figure 10.2.
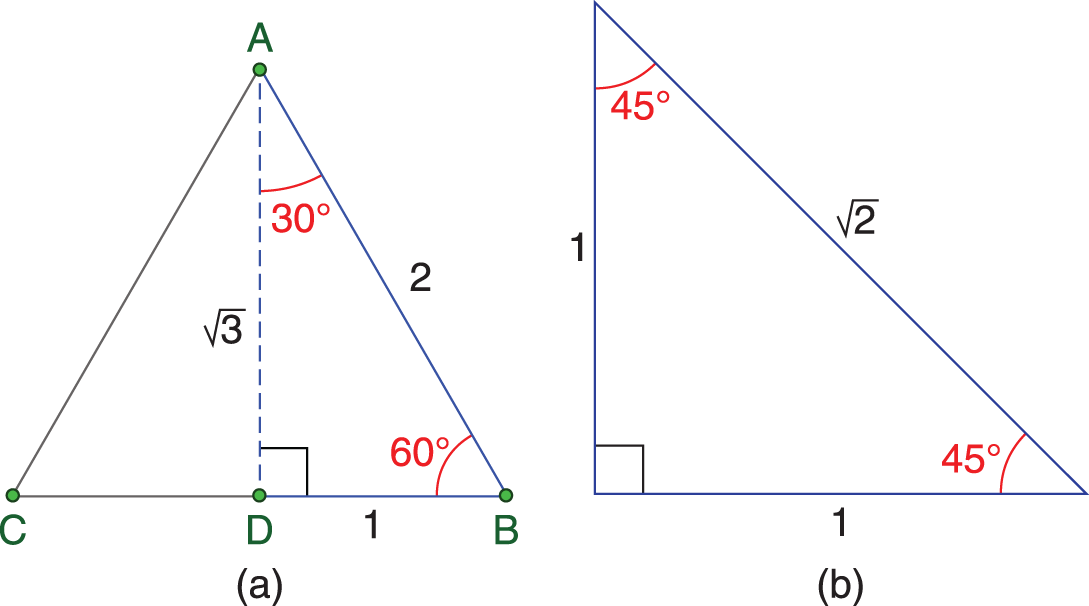
Figure 10.2. Special Right Triangles (a) 30−60−90; (b) 45−45−90.
Important values of the trigonometric ratios at these angles are shown in Table 10.3.
Table 10.3 Common Trigonometric Ratios on the MCAT
θ SIN θ COS θ TAN θ
0° 0 1 0
30° 12 32 33
45° 22 22 1
60° 32 12 3
90° 1 0 undefined
180° 0 −1 0
MCAT CONCEPT CHECK 10.3
Before you move on, assess your understanding of the material with these questions.
- During vector addition, how is the angle of the resultant calculated?
_________________________________
- How are sine, cosine, and tangent calculated when given the dimensions of a right triangle?
- Sine:
_________________________________
- Cosine:
_________________________________
- Tangent:
_________________________________
- True or False: Only angles in right triangles have characteristic values of the trigonometric functions.
- At what angle between 0° and 180° does each trigonometric function have a value of 1?
_________________________________
10.4 Problem-Solving
LEARNING OBJECTIVES
After Chapter 10.4, you will be able to:
- Distinguish between direct and inverse relationships
- Convert between metric prefixes
- Solve a system of equations using substitution, setting equations equal, or elimination, such as: 3x + 4y = 17 and 5x − 2y = 11
Now that we’ve examined some individual mathematical skills, let’s explore some common problem-solving strategies to approach MCAT questions. The use of relationships and proportionality is especially important in passage-based questions, while unit analysis can help determine which formulas are appropriate for a given question. The use of conversion factors is ubiquitous on the MCAT, as answer choices are often given in different units than the information presented. Algebraic systems are less often required, but may underlie passage interpretation and the approach to some questions.
Use of Relationships
Relationships are generally indicated in MCAT passages by formulas or the use of proportionality constants. In other cases they may be implied and require a bit more work on our part to calculate ratios. Calculations of this type are based on multiplication and division, but explaining the relationship in words—rather than math—may make it challenging to decode the connections between the variables. In direct relationships, increasing one variable proportionately increases the other; as one decreases, the other decreases by the same proportion. In inverse relationships, an increase in one variable is associated with a proportional decrease in the other.
BRIDGE
According to Boyle’s law, pressure and volume have an inverse relationship: as one is doubled, the other is cut in half (keeping all else constant). On the other hand, according to Gay-Lussac’s law, pressure and temperature have a direct relationship: as one is doubled, so is the other (keeping all else constant).
Conversions
The MCAT routinely increases the difficulty of a question by requiring the use of conversion factors. Equations may require that variables be in certain formats, or answer choices may differ in units from those given in the question stem. In both cases, it is necessary to convert units. The simplest conversions to perform maintain the same base unit. For example, conversion between grams, kilograms, and milligrams only requires multiplication by an appropriate power of ten. Metric prefixes and their associated powers of ten are found in Table 10.4.
Table 10.4 Metric Prefixes
FACTOR PREFIX PREFIX ABBREVIATION
1012 tera– T
109 giga– G
106 mega– M
103 kilo– k
102 hecto– h
101 deka– da
10−1 deci– d
10−2 centi– c
10−3 milli– m
10−6 micro– μ
10−9 nano– n
10−12 pico– p
In addition to the conversions that are necessary for changes in prefixes, we must often convert between units, particularly between the British system and SI units. Table 10.5 shows several important conversion factors to recognize on Test Day. Conversion factors (except those for time) should not be memorized; the MCAT will provide them as necessary.
Table 10.5 Common Conversion Factors on the MCAT
BASE UNIT EQUIVALENT UNITS
1 mile 5280 feet (ft)
1 ft 12 inches (in)
1 inch (in) 2.54 cm
1 Calorie (Cal) 1000 cal
1 calorie (cal) 4.184 J
1 electron–volt (eV) 1.602 × 10−19 J
1 L 33.8 ounces (oz)
1 pound (lb) 4.45 N
1 atomic mass unit (amu) 1.661 × 10−27 kg
Example: A car’s speedometer registers a speed of 33 miles per hour. What was its speed in meters per second?
Solution: First convert distance measurements, being careful to cancel them out by arranging numerators and denominators.
33 mileshour (5280 feet1 mile)(12 inches1 foot)(2.54 cm1 inch)(1 m100 cm)≈(33)(5280)(30)100≈(1000)(5280)100= 52,800mhour(actual = 53,108 mhour)
Then, repeat the procedure with the time measurements.
53,108mhour (1hour3600s)≈54036=906=15 ms ( actual = 14.8 ms)
One special case of conversions occurs with temperature. Rather than simply multiplying by a conversion factor, there is also a component of addition or subtraction. The following formulas relate the Fahrenheit, Celsius, and Kelvin systems:
F=95 C+32K=C+273
Equation 10.20
where F, C, and K are the temperatures in degrees Fahrenheit, degrees Celsius, and kelvins, respectively.
REAL WORLD
It is important to be able to convert between Fahrenheit and Celsius scales in medicine as different hospitals (and different medical records) may use different units. Body temperature is 98.6°F or 37 °C. A fever is usually defined as a temperature above 100.4°F or 38 °C. Hypothermia is usually defined as a temperature below 95.0°F or 35 °C.
Unit Analysis
Unit analysis, also called dimensional analysis, may help determine the correct answer even if you forget a relevant formula on Test Day. It can also serve as a double check on one’s calculations because the units of the calculated answer must match the units of the answer choices. For example, consider a question in which we are given two quantities: one in NC, and the other in volts. The answer choices for the question are all in meters. Even without remembering the equation V = Ed, we can infer that we must divide the voltage (1 V=1 JC=1 N⋅mC) by the electric field to get a distance in meters:
N⋅mC÷NC=m
Dimensional analysis is not a foolproof strategy; it is always better to know the true relationships between variables than to infer them based on units. Still, this strategy can be effective for narrowing down (or even choosing) answer choices on Test Day.
Example: The ejection fraction is the proportion of the blood volume in the left ventricle expelled with each contraction of the heart. A patient is known to have an ejection fraction of 0.6, a cardiac output of 5 Lmin, and a heart rate of 80 beatsmin. What is the volume of blood in this person’s left ventricle just prior to contraction?
Solution: A formula was not provided in this question, but we can recognize that the desired answer is a volume. We can start with the cardiac output and heart rate terms to determine the volume ejected per beat.
5 Lmin80 beatsmin=116=0.0625 Lbeat
The question also explains that only 60 percent of the blood volume is expelled from the left ventricle per heartbeat. From this, we can determine the volume of blood prior to contraction.
0.0625 Lbeat=0.6 ×ventricular volumebeat0.06250.6 L=ventricular volume≈0.1 L (actual=0.104 L)
Algebraic Systems
The last key mathematical skill for Test Day is the ability to solve systems of linear equations. In order to solve a system of equations, there must be at least as many equations as there are variables. Where there is only one variable (which does not truly constitute a system), only one equation is necessary; for example, 6 − x = 1 reduces to x = 5. In contrast, with an equation like 3x + 4y = 17, there is insufficient data to solve for either variable with only the one equation. If a second equation is introduced, such as 5x − 2y = 11, then we can solve for both variables using one of three methods: substituting one variable in terms of the other, setting equations equal to each other, or manipulating the equations to eliminate one of the variables.
Substitution
In substitution, we solve for one variable in one of the equations, and then insert this term into the other equation. The steps of this method are listed below.
- Solve for one of the variables in one of the equations:
5x−2y=11y=5x−112
- Insert the expression into the other equation:
3x+4(5x − 112)=17
- Isolate the variable and solve the resulting equation:
3x+10x−22=1713x=39x=3
- Solve for the other variable using this value:
3(3)+4y=174y=8y=2
Setting Equations Equal
Setting equations equal to one another is a specialized case of substitution. In this method, we solve for the same variable in both equations and then set the two equations equal to each other. The steps of this method are listed below.
- Solve for the same variable in both equations:
y=17 − 3x4y=5x − 112
- Set the equations equal to each other, isolate the variable, and solve for the variable:
17 − 3x4=5x − 11217−3x=10x−2239=13x3=x
- Solve for the other variable using this value:
3(3)+4y=174y=8y=2
Elimination
In elimination, multiply or divide one (or both) of the equations to get the same coefficient in front of one of the variables in both equations. Then, add or subtract the equations as necessary to eliminate one of the variables. The steps of this method are listed below.
- Multiply or divide one (or both) of the equations by a constant so that the coefficient in front of one of the variables in both equations is the same:
3x+4y=172×[5x−2y=11]
- If the sign of both coefficients is the same, subtract one equation from the other. If the sign is opposite, add the two equations together:
4y+3x=17-4y+10x=2213x=39x=3
- Solve for the other variable using this value:
3(3)+4y=174y=8y=2
Note that each method results in the same answer despite slight differences in the steps taken. As a matter of convention, the answers for systems of equations with the variables x and y are reported as coordinates on the Cartesian plane (x,y); thus, our answer for this system would be (3,2). Systems of equations can have many variables, but it is unlikely that you will encounter a system with more than three variables (x,y,z) on the MCAT.
MCAT CONCEPT CHECK 10.4
Before you move on, assess your understanding of the material with these questions.
- How are conversions between metric prefixes accomplished? How would a distance be converted from millimeters to kilometers?
_________________________________
_________________________________
- What does it mean for two variables to have a direct relationship? An inverse relationship?
- Direct:
_________________________________
- Inverse:
_________________________________
- Each of the three methods for solving systems of equations discussed in this chapter solve for one variable, and then use this value to solve for the other. How does each method solve for the first variable?
- Substitution:
_________________________________
- Setting equations equal:
_________________________________
- Elimination:
_________________________________
Conclusion
In this chapter, we reviewed many of the skills that are necessary for successful performance on the MCAT science sections. We began by examining relevant arithmetic calculations for Test Day, including scientific notation and significant figures. We continued our review by examining logarithms and exponents before discussing the most common trigonometric functions and their values. We finished our math review by working on problem-solving skills that will be valuable in your studying and during the MCAT itself. In the next two chapters, we’ll review Test Day skills in experimental design and data analysis so that you’ll be ready ready to answer all of the Test Day questions.
GO ONLINE
You’ve reviewed the content, now test your knowledge and critical thinking skills by completing a test-like passage set in your online resources!
CONCEPT SUMMARY
Arithmetic and Significant Figures
- Scientific notation is a method of writing numbers in a way that improves the ease of calculations and the comparability of significant digits.
- Scientific notation takes the format [significand] × 10[exponent].
- The significand must be greater than or equal to 1 and less than 10.
- The exponent must be an integer.
- Significant figures include all nonzero digits and any trailing zeroes in a number with a decimal point.
- Measurements are an exception, in that the last digit provided is not significant.
- In addition and subtraction, reduce the answer to have the same number of decimal places as the number with the fewest number of decimal places.
- In multiplication and division, reduce the answer to have the same number of significant digits as the number with the fewest number of significant digits.
- The entire number should be maintained throughout calculations to minimize rounding error.
- Estimation of multiplication and division should be done logically.
- In multiplication, if one number is rounded up, the other should be rounded down in proportion.
- In division, if one number is rounded up, the other should also be rounded up in proportion.
Exponents and Logarithms
- Exponents are a notation for repeated multiplication. They may be manipulated mathematically, especially when the bases are the same.
- Logarithms are the inverse of exponents and are subject to similar mathematical manipulations.
- Natural logarithms, which use base e (Euler’s number) can be converted into common logarithms, which use base 10.
Trigonometry
- Trigonometric relationships can be calculated based on the lengths of the sides of right triangles.
- Sine is the ratio of the length of the side opposite an angle to the length of the hypotenuse.
- Cosine is the ratio of the length of the side adjacent to an angle to the length of the hypotenuse.
- Tangent is the ratio of the side opposite an angle to the side adjacent to it.
- Inverse trigonometric functions use the calculated value from a ratio of side lengths to calculate the angle of interest.
Problem-Solving
- In direct relationships, as one variable increases, the other increases in proportion.
- In inverse relationships, as one variable increases, the other decreases in proportion.
- Conversions between metric prefixes require multiplication or division by corresponding powers of ten.
- Conversions between units of different scales require multiplication or division, and may require addition or subtraction.
- Unit analysis (dimensional analysis) can determine the appropriate computation based on given information.
- Algebraic systems may be solved by substitution, setting equations equal, or elimination. The general ideas are the same in each—solve for one variable, and then substitute the variable into an equation to solve for the other—although the specific methods are different.
ANSWERS TO CONCEPT CHECKS
**10.1**
- First, determine which digits are significant, as these will be preserved in scientific notation. Then, move the decimal point until the significand is greater than or equal to 1 and less than 10. Finally, determine what power of 10 is necessary for multiplication to restore the original number.
- 34,600.; 0.0003201; 1.10; 525,600
- In multiplication, adjust the two decimals in opposite directions. In division, adjust the two decimals in the same direction.
**10.2**
-
- (a + b)2 = a2 + 2ab + b2
- a2 + 2a25a3=3a25a3=35a
- loga (a) = 1
- log(a3)−log(a)=loga3a=loga2=2loga
- 392 is between 361 and 400, so the value is between 19 and 20. We can also simplify this radical:
392=4×49×2≈14×1.4=19.6(actual=19.8)
- log 7,426,135,420 ≈ log (7.4 × 109) ≈ 9 + 0.74 = 9.74 (actual = 9.87). Note that—even with an absurdly large number—we can still get relatively accurate estimations by following basic logarithm rules.
**10.3**
- The value of a trigonometric function calculated from the dimensions of the resultant vector is used in the inverse tangent function to calculate the resultant vector angle. Inverse trigonometric ratios, in general, can be used to calculate angles.
- The sine of an angle is equal to the ratio of the side opposite the angle to the hypotenuse. Cosine is the ratio of the side adjacent to the angle to the hypotenuse. Tangent is the ratio of the side opposite the angle to the side adjacent to the angle.
- False. Even though calculating the values of sine, cosine, and tangent is more complicated in a triangle that does not contain a right angle, all possible angles do still have characteristic trigonometric values.
- Sin is equal to 1 at 90°, cos at 0°, and tan at 45°. Many MCAT questions utilize the trend that sin increases going from 0° to 90°, whereas cos decreases over this range.
**10.4**
- Conversion between metric prefixes is accomplished by multiplication or division by the appropriate power of ten. To convert from millimeters (10−3) to kilometers (103), it is necessary to multiply by 10−6. It’s wise to double-check your work when converting: a kilometer is a larger unit of distance than a millimeter, so the number of kilometers should be smaller than the number of millimeters.
- In direct relationships, as one quantity increases, the other also increases in proportion. In inverse relationships, as one quantity increases, the other decreases in proportion.
- In substitution, solve one equation for one variable in terms of the other; then, substitute this expression into the other equation. In setting equations equal (a modified version of substitution), solve both equations for the same variable and set them equal to each other. In elimination, multiply or divide one (or both) equations so that the coefficient in front of one of the variables is the same in both equations; then, add or subtract the equations to eliminate one of the variables.
SCIENCE MASTERY ASSESSMENT EXPLANATIONS
1. B
This question, while overtly testing the ability to use scientific notation, is also checking on the appropriate use of significant digits. Because there is no decimal point, the last zero is not significant and should not be used in scientific notation. The significand in scientific notation should always be between one and ten.
2. C
Significant digits include all nonzero digits, all zeroes that are between nonzero digits, and trailing zeroes in any number with a decimal point. In 14,320,010 there is no decimal point; thus, the last zero is insignificant and there are seven significant digits. In 3.618000, all of the digits are significant; thus there are also seven significant digits.
3. B
While all digits are preserved during calculations, the final determination of the number of digits is made by both significant figures and decimal places. During multiplication, the answer is maintained to the smallest number of significant digits. During addition, it is maintained to the smallest number of decimal places. By following the order of operations, addition is the last operation; thus we cannot have a decimal in our answer choice. Because multiplication occurred earlier, the result of that multiplication may be shortened according to the two significant figures in 4.10, but not the entire answer.
4. A
When estimating the product of two numbers, it is best to round one up while rounding the other down, as in (A). (B) and (D) each round both numbers in the same direction, which would increase the amount of error in the answer. (C) rounds the numbers in opposite directions, but the degree of rounding is significantly larger than in (A) and too extreme for answer choices that differ by small amounts.
5. A
The fourth root of a number, or a number raised to the one-quarter power, is the square root of the square root of that number:
2000.25=20012×12=200=2004
The square root of 200 should be a bit larger than 14 (142 = 196); therefore, the fourth root of 200 should be a bit less than 4.
6. D
Raising an exponent to another exponent requires multiplying the exponents. Thus, (A3)2 = A6.
7. A
The relationship between the natural logarithm of a number and the common logarithm of a number is logx=lnx2.303. Therefore, the natural logarithm of a number must be divided by the constant 2.303 to obtain the common logarithm of the same number.
8. A
The minimum value of the cosine function is −1 (cos 180° = −1). Therefore, the minimum value of 2 cos θ – 1 is 2 × (−1) −1 = −3.
9. D
sin θ ≠ sin (90° − θ), although sin θ = cos (90° − θ). The other statements must all be true. Because sine and cosine values are always between −1 and 1, the product of sine and cosine will always have a magnitude less than 1. The sum of the absolute value of sine and the absolute value of cosine, on the other hand, will always be greater than 1. Therefore, (A) can be eliminated. Because sine is the ratio of opposite to hypotenuse and cosine is the ratio of adjacent to hypotenuse, the quotient between the two is the ratio of opposite to adjacent, or the tangent of the angle. Therefore, (B) can be eliminated. By the same logic, because sin 90° = 1 and cos 90° = 0, tan 90° is undefined, eliminating (C).
10. B
This question involves both a unit conversion between millimolar values and molar values, and calculation of a logarithm. The relationship between pH and pKa is described by the Henderson–Hasselbalch equation given in the question stem. 100 mM = 0.1 M, so
pH=pKa+log[A−][HA]=3.6+log(1)=3.6
11. B
This question requires not only unit conversions, but algebra as well. Given that F=95 C+32, the temperature T can be calculated as:
T = 95 T + 32 →−45 T = 32 → T =−54(32) =−40
However, the answers are given in kelvin. −40 °C + 273 = 233 K.
12. D
In a direct relationship, a change in one of the variables will be associated with a proportional change in the other. Because the pressure was multiplied by 13, the temperature must also be multiplied by 13. Note that the fractional relationships can only be used with temperatures in kelvin.
13. C
Because grams are a unit of mass and pounds are a unit of force, we must first convert pounds to newtons, and then divide by the acceleration due to gravity to find kilograms. The weight of the person in newtons is 150 lb×4.45Nlb≈150×4.4=600+60=660 (actual = 667.5 N). This corresponds to a mass of 667.5N9.8 ms2=667.5 kg⋅ms29.8 ms2≈67 kg.
Now, we can determine the dose: 67 kg (1.5 mg drugkg body mass)≈100 mg.
14. C
According to the question stem, the rate of a reaction is measured as a change in concentration over time, and thus has the units Ms, where M (molarity) is measured in moles per liter. However, the rate of the reaction is equal to a rate constant times the concentrations of certain reactants squared. In this case, we know the units of everything except the rate constant and must solve for its units:
rate=k[A]2Ms=k[M]2→k=1M⋅s=Lmol⋅s
15. C
This is a system of equations couched in data. From this information, we can construct two equations:
C=900+12mC=500+15m
These equations can be solved by setting them equal:
900+12m=500+15m400=3m133 kg=m
GO ONLINE
Consult your online resources for additional practice.
EQUATIONS TO REMEMBER
(10.1) Zero exponent identity: X0 = 1
(10.2) Multiplying like bases with exponents: *X**A×X**B=X(A+B*)
(10.3) Dividing like bases with exponents: XAXB=X(A − B)
(10.4) Raising an exponent to another exponent: (*X**A)B=X(A×B*)
(10.5) Raising fractions to exponents: (XY)A=XAYA
(10.6) Raising bases to negative exponents: X−A=1XA
(10.7) Raising bases to fractional exponents: XAB=XAB
(10.8) Square root approximations: 2≈1.414(use 1.4)3≈1.732(use 1.7)
(10.9) Logarithm of 1 identity: logA 1 = 0
(10.10) Logarithm of base identity: logA A = 1
(10.11) Logarithm of product: log A × B = log A + logB
(10.12) Logarithm of quotient: logAB=logA−logB
(10.13) Logarithm of exponent-containing expression: log AB = B log A
(10.14) Logarithm of inverse: log1A=−logA
(10.15) Conversion of natural to common logarithm: logx≈lnx2.303
(10.16) Scientific notation logarithm approximation: log (n × 10m) ≈ m + 0.n
(10.17) Definition of sine: sinθ=oppositehypotenuse=ac
(10.18) Definition of cosine: cosθ=adjacenthypotenuse=bc
(10.19) Definition of tangent: tanθ=oppositeadjacent=ab
(10.20) Temperature conversions: F=95 C+32K=C+273
SHARED CONCEPTS
General Chemistry Chapter 5
Chemical Kinetics
General Chemistry Chapter 7
Thermochemistry
General Chemistry Chapter 10
Acids and Bases
Physics and Math Chapter 7
Waves and Sound
Physics and Math Chapter 9
Atomic and Nuclear Phenomena
Physics and Math Chapter 12
Data-Based and Statistical Reasoning